Abstract
Toxic product in fire disasters is the most important reason for fire casualties. With wide application of polymer material, the toxic products in fire effluents are getting more and more diversified and complicated. Polyurethane is one of the most widely used materials. In this article, the fire toxicant release has been evaluated for thermoplastic polyurethane (TPU) and its flame-retardant composites. Ammonium polyphosphate (APP), aluminum hydroxide (ATH), and nano-montmorillonite (MMT) were combined into different flame-retardant combinations at certain ratios. Three kinds of flame-retardant combinations (APP-ATH, APP-MMT, and APP-ATH-MMT) were blended to reduce toxicity of TPU. The properties of thermal stability and decomposition were characterized by thermogravimetric analysis (TGA). TGA/infrared spectrometry, static tube furnace, and steady-state tube furnace were used to evaluate the toxic gases, including CO and HCN. Fractional effective dose (FED) was calculated based on the concentrations of CO, CO2, and HCN. The results showed that more than 50% toxicity effect in FED was accounted for HCN. The comprehensive toxicity of TPU was reduced in the samples with APP-ATH and APP-ATH-MMT. The yields of CO, CO2, and O2 consumption were indicated much lower in the samples with APP-ATH-MMT than the other two combinations.
Introduction
Thermoplastic polyurethane (TPU) is widely used in wires, cables, and protective coatings for its good performance. However, TPU is highly combustible with high release of heat and smoke which will prevent people to escape from fire. Large amounts of toxic gas, such as CO, NO, NO2, CH2O, and hydrogen cyanide (HCN) are produced during the thermal decomposition and combustion of TPU, which can easily cause poisoning and respiratory damages. 1 Many tests have identified that CO is not the only primary toxicant in smoke from burning materials, 2,3 HCN and HCl are also identified as primary toxicants depending on the chemical composition of burning materials. 2 Incapacitation followed by death will be due to the combined effect of CO and HCN. 4 A series of experiments show that 92% of toxicity in polymer fire is accounted for CO, 2% for HCN, and 4% for the combined action between CO and HCN. 5
Flame retardants are the most important additives widely used in polyurethane to reduce its flammability and toxicity and affect the release of smoke and toxicity with different mechanism for different additives. Shi et al. 6 –8 have performed a lot of studies about the fire hazard of plastic material, such as TPU and polystyrene (PS). It approved that the modified combination of flame retardants had high efficiency in reduction of heat release, smoke suppression, and toxicant decrease. Wang et al. 9 used ammonium polyphosphate (APP) as a flame-retardant agent to reduce toxicity and fire hazard of TPU, the synthesized zinc hydroxystannate (ZnHS) demonstrated obviously decreased toxic volatiles, such as CO, HCN, and NOX. Yang and Lu 10 used sodium hydroxide solution to absorb HCN to study the effect of the type of fire retardants. It was found that NH4Cl, (NH4)SO4, and (NH4)3PO4 increased HCN yield, whereas MgCl2, C2H4N4, NH4H2PO4, KH2PO4, and Na2B4O7·10H2O reduced HCN yield. Duquesne et al. 11 reported that the emissions of CO and CO2 were significantly decreased, the volume of smoke production was reduced by 40%, and the production of CO was decreased by 80% with the load of 40 wt% APP. Liu et al. 12 have added 5 wt% ATH in rigid polyurethane-polyisocyanurate (PUR-PIR) with 10 wt% dimethyl methylphosphonate (DMMP). The emissions of CO and CO2 were decreased, respectively. In addition, some natural or synthetic hybrid compounds, such as vermiculite, layered double hydroxide (LDH), and montmorillonite (MMT), have been employed in PU to decrease smoke production.
The greatest yields of toxic product usually occur under the oxygen-depleted conditions in large-scale tests. Small-scale toxicity assessment can replicate large-scale fires when burning can be conducted under oxygen-depleted conditions. Blais and Carpenter 5 have performed a series of experiments to measure the toxic vapor emissions of fires with flexible polyurethane foams. Test was performed both to maximize detection of gases in small-scale test and measure the exposure concentrations in realistic fire conditions in a room sized enclosures. Some analytical techniques are commonly used to assess quantitatively and qualitatively the smoke and toxicity of the gasses evolved during PU decomposition. By using an fourier transform infrared spectroscopy (FTIR) attachment, the production of specific gasses such as HCN, CO, and CO2 can be monitored. 13 Thermogravimetric analysis–mass spectrometry (TG-MS), thermogravimetry analysis–infrared spectroscopy (TG-FTIR), and pyrolysis gas chromatography–mass spectrometry (GC-MS) are also widely used for gas-phase analyses. Sun et al. 14 have spatially and temporally resolved in situ measurements of HCN in a steady-state tube furnace (SSTF) using midinfrared polarization spectroscopy, it was found that high temperatures and good ventilation result in relatively high peak concentrations of HCN inside the tube furnace. Purser and Purser 15 studied the relationships between equivalence ratio and HCN yields in the ISO19700 tube furnace under steady flaming conditions, enabling the relationships between toxic product yields and equivalence ratio to be investigated. Stec and Hull 16 studied six insulation materials under a range of fire conditions, the toxicities of the effluents were compared using the fractional effective dose (FED) model and lethal concentration 50 (LC50). It showed that HCN resulted in doubling of the overall toxicity, as the fire condition changes from well-ventilated to under-ventilated. Hartzell and Emmons 17 proved that the concept of the FED model can be used with either individual fire toxicants or with smoke produced from the burning of materials.
TPU is widely used because of its good performance, and the application of flame retardant additives is very important to reduce the risk of combustion. So it is necessary to evaluate the composites comprehensively, perform the comparative analysis, and determine the main factors of toxicity release. Most of previous works focused on the small-scale experiments, including animal test, SSTF, and TGA/infrared spectrometry to quantitatively and qualitatively assess the smoke and toxicity of the gasses evolved in the decomposition of TPU. But it is found that animal studies can only observe the final toxic results, instead of continuous exposures in the real time. In addition, most experiments conditions are from well-ventilated to under-ventilated, but the greatest toxic emission is appeared in the under-ventilated condition in a series of tests. Therefore, TGA/infrared spectrometry, static tube furnace, and SSTF are used to evaluate the toxic gases continuously in the under-ventilated conditions. This work mainly aims at the comprehensive toxic gases suppression properties and fire toxicity reduction between APP, ATH, and MMT in TPU. The comprehensive assessment of toxicity (especially CO and HCN) are evaluated by TGA/infrared spectrometry, static tube furnace, and SSTF.
Experimental work
Preparation of flame-retardant TPU composites
TPU, APP (powder), aluminum hydroxide (ATH) (powder), and MMT (powder) are obtained from material market in Hefei (Anhui, China). TPU, APP, ATH, and MMT are dried in vacuum drier (Hebei, China) at 80°C for 10 h before test. APP, ATH, and MMT are preliminarily mixed and then are added into TPU to prepare TPU composites using a master batch-melt compounding approach at the temperature of 175°C for 10 min by a blende (model: Rheomixer XSS-300, Dongguan, China). After mixing, the samples are pressed at 180°C under 10 MPa for 5 min into sheets of suitable thickness for further analysis. The formulations are given in Table 1.
The formulation of flame-retardant TPU composites.
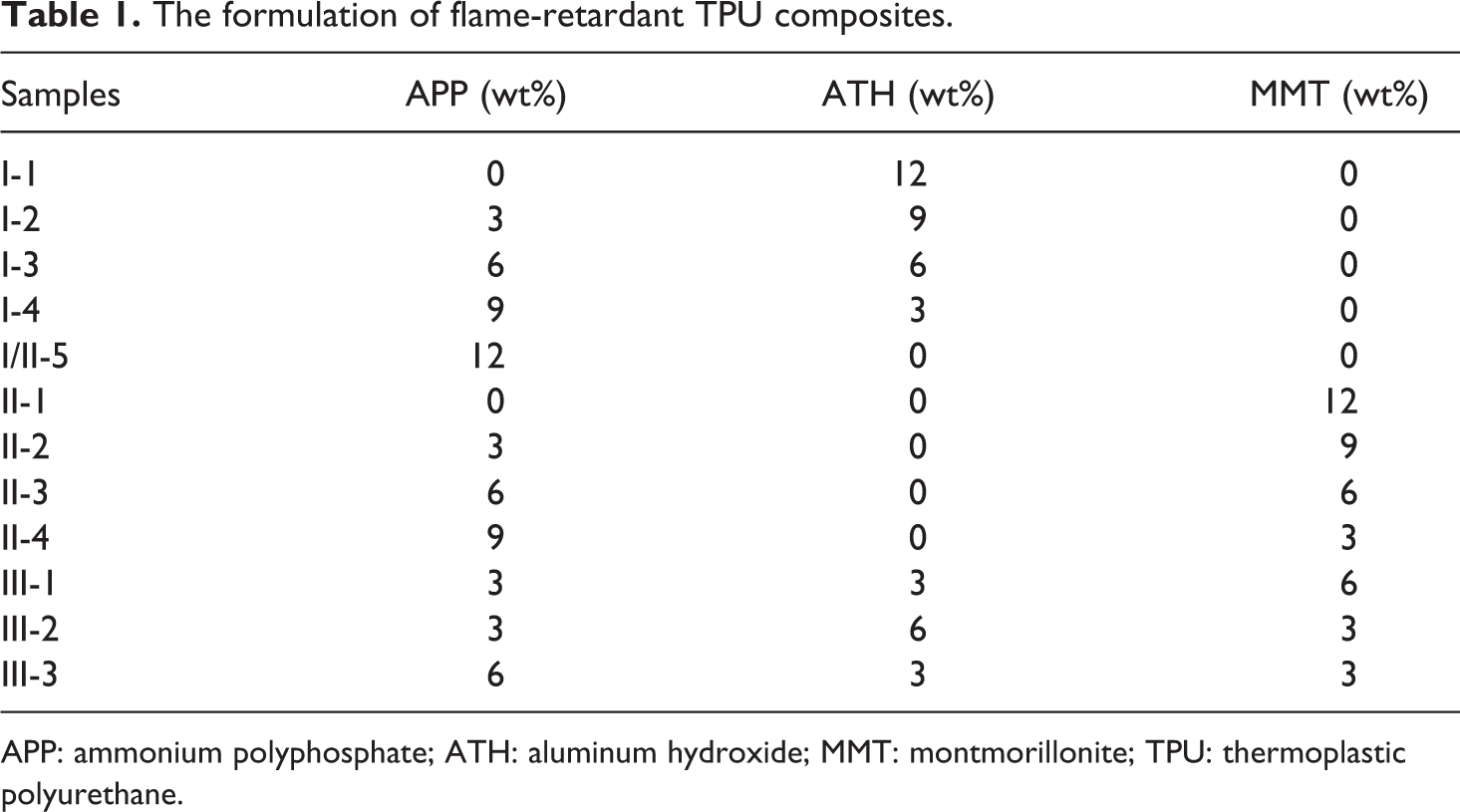
APP: ammonium polyphosphate; ATH: aluminum hydroxide; MMT: montmorillonite; TPU: thermoplastic polyurethane.
Experimental conditions
TG-FTIR has been used to evaluate the gaseous products of the TPU and its composites during thermal degradation in nitrogen. The FTIR spectra of samples are recorded by a Nicolet MAGNA-IR 750 spectrophotometer. Each sample is examined in air flowing at 45 mL min−1 on a Q5000 (model: TA, USA) thermogavimetric analyzer at the heating rate of 20°C min−1. The weight of all samples is in the range of 5–10 mg in an open porcelain pan and is heated from 30°C to 825°C in nitrogen atmosphere.
The toxicity of HCN during the thermal degradation of polymer materials in air is performed by the static tubular furnace method, then is detected by HCN detector tube (model: 12 L, sensitivity 0.1 ppm) to quantitatively analyze the combustion gases. Experiments have been carried out at 825°C for 20 min. In all cases, the mass of the sample used is around 1 g. The samples are positioned in an open porcelain pan placed in a tube. Gases are continuously extracted with a pump (rate: 2L min−1) (Figure 1).
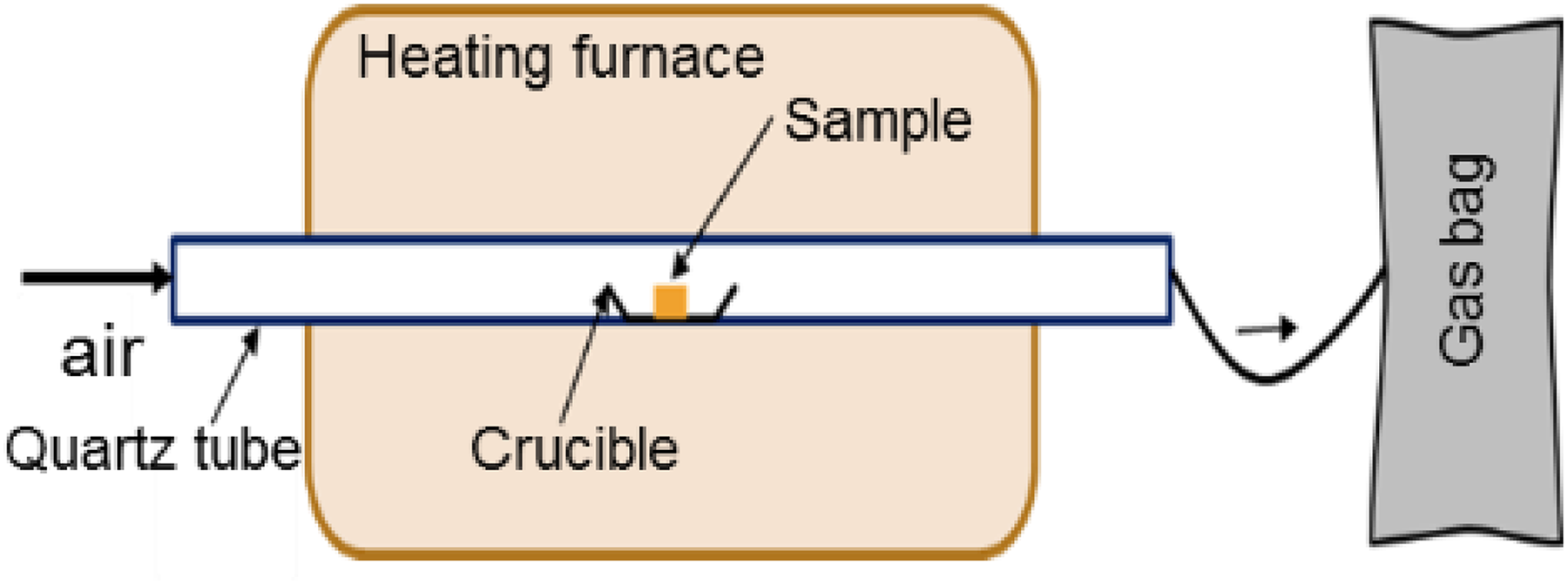
Schematic diagram of the static tube furnace.
The SSTF tests are measured according to standard ISO TS 19700 (Figure 2). About 20 g samples loaded into the quartz boat are fed into the furnace at around 40 mm min−1. To realize flashover condition in ISO 19706, 18 the experimental temperature is set at 825°C, the ventilation condition is set with the primary air flow rate at 3.5 L min−1 and second air flow rate at 46.5 L min−1. Oxygen depletion and yields of carbon dioxide, carbon monoxide, and smoke are determined using standalone detectors.
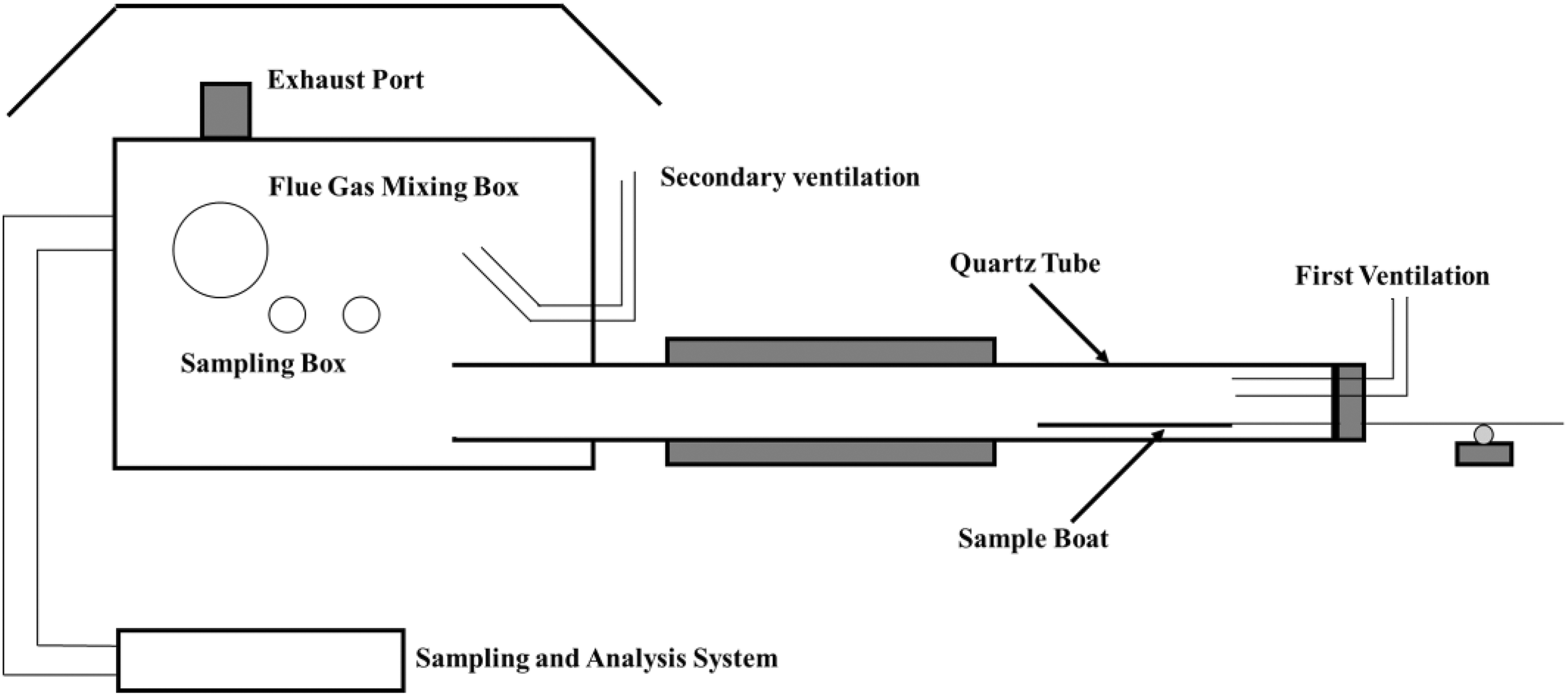
Schematic diagram of the SSTF.
Results and discussion
Thermogravimetric analysis
The TGA curve of pure TPU is presented in Figure 3 for an example to interpret how to obtain the initial decomposition temperature: the temperature at 5% weight loss (T−5wt%), the temperature at the first maximum decomposition rate (TI max), and the temperature at the second maximum decomposition rate (TII max). The summaries of the thermal data of TPU and its composites and three flame retardants (APP, ATH, MMT) are displayed in Table 2. The initial decomposition temperature of pure TPU is 330°C. Two-stage decomposition of pure TPU is observed between 367°C and 430°C, respectively. Approximately 3.76% of the char residues remains at 825°C. Compared with pure TPU, the TPU composites have lower initial decomposition temperature, as a result of ATH and MMT begin to decompose at 277°C and 280°C, respectively. When it is more than 350°C, the char residues of TPU composites are increased to 26.9–30 wt%. So there is a synergism effect between APP and ATH for the char formation.
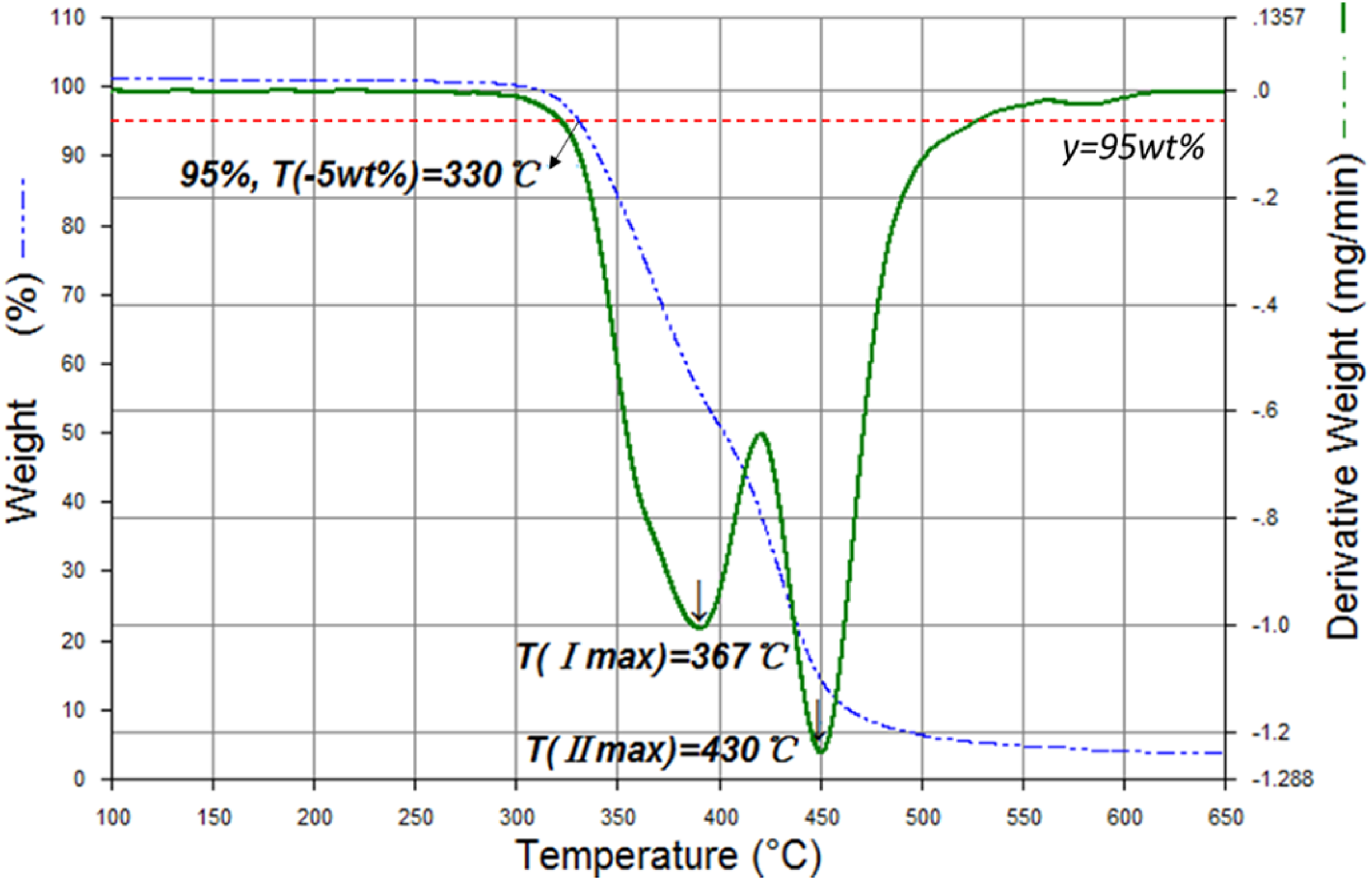
Temperature profiles of TPU under nitrogen atmosphere.
The summaries of the thermal data of TPU and its composites.
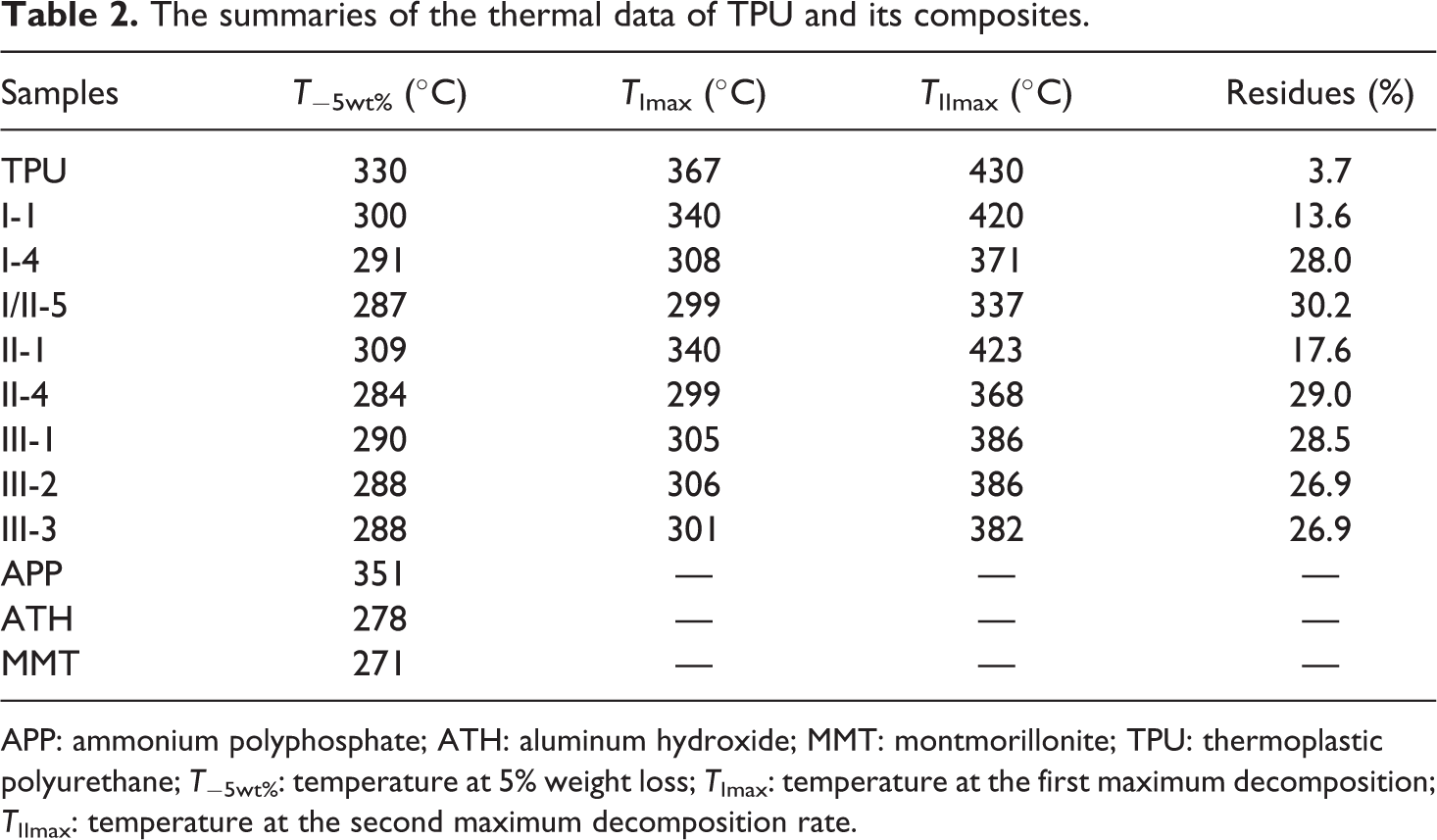
APP: ammonium polyphosphate; ATH: aluminum hydroxide; MMT: montmorillonite; TPU: thermoplastic polyurethane; T−5wt%: temperature at 5% weight loss; TImax: temperature at the first maximum decomposition; TIImax: temperature at the second maximum decomposition rate.
Volatile gases analysis
FTIR spectra of pyrolysis products at the temperature of maximum decomposition rates (368°C and 429°C) are presented in Figure 4(a). The evolved gaseous products for pure TPU at two rates exhibit characteristic bands of water (3575 cm−1), 19 –CH3 and –CH2–(2960 cm−1), 20 CO2 (2358 and 2310 cm−1), 21,22 –C=O (1754 cm−1), 20 –NHC=O (1528 cm−1), 20 esters (1237 cm−1), 22 –24 and HCN (722 cm−1). As shown in Figure 4(b), similar volatilized products are exhibited in sample III-1 at maximum decomposition rates as pure TPU. However, the intensity of absorption peak of some groups is stronger. For example, the intensity of CO2 is obviously decreased at 385°C, an absorption peak at 722 cm−1 (HCN) is decreased at the second maximum decomposition rate. It can be inferred that the release of CO2 and HCN is mainly occurred at the temperature of the first maximum decomposition rate.
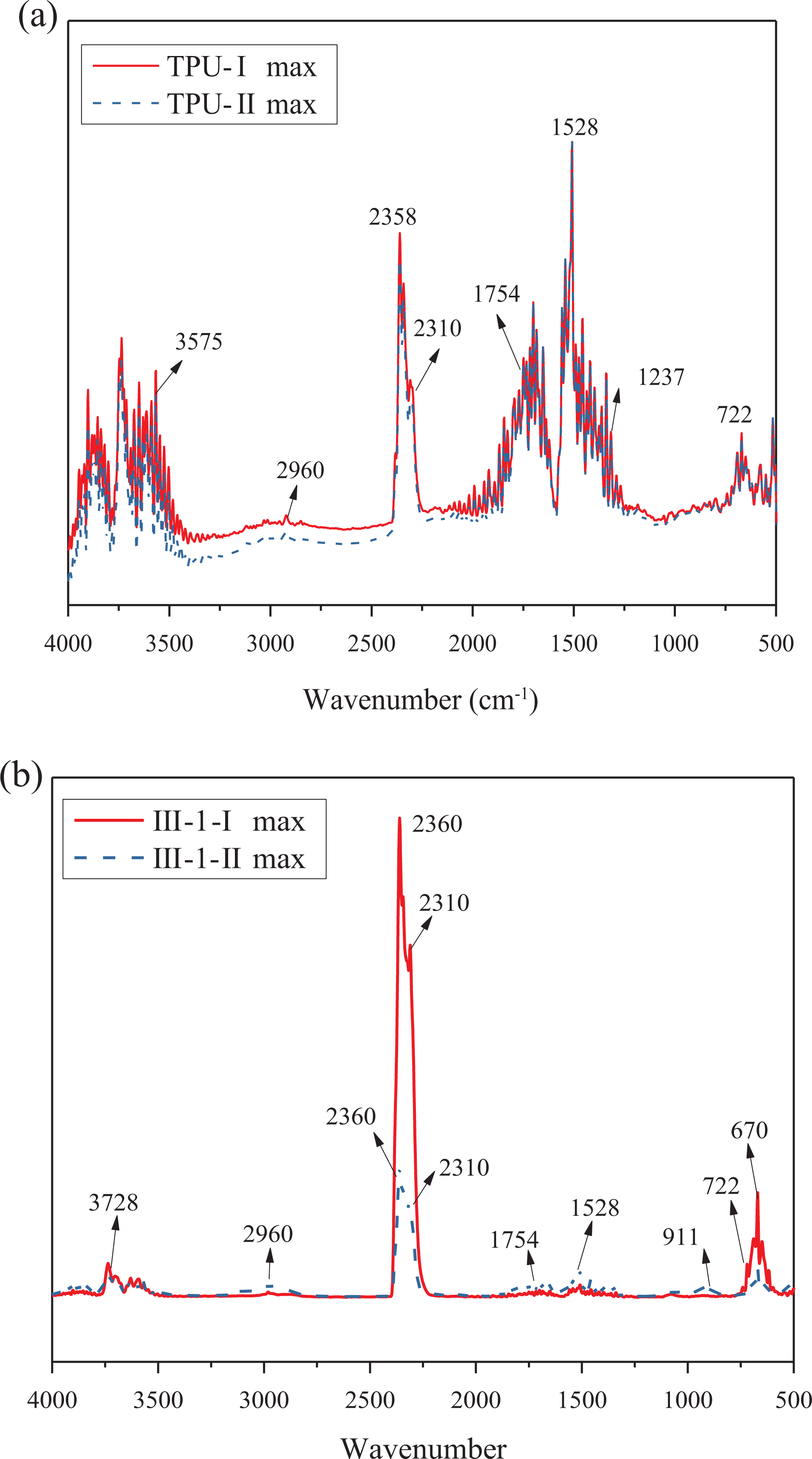
TG-IR spectra of pyrolysis products at maximum decomposition rate of TPU (a) and sample III-1 (b).
Gaseous toxicant analysis
The repetitive unit in polyurethanes is the urethane bond (–NH–COO–) obtained from the reaction between isocyanates (–NCO–), 25 so CO and HCN are considered as two principle toxicants in this part. Well-ventilated fire scenarios are routinely used for assessment of flammability, because the object is to stop the fire developing to the stage out of control. While fire toxicity is concerned, the important fire stages are under-ventilated. There are two reasons for this: (1) The volume of effluent is much greater, and (2) the yields of the major toxic products will be much greater. 26 According to the previous work, 27 tube furnace (static and stead state) is performed at 825°C, and the equivalence ratio is set more than 1.
HCN measurement
The total concentration of HCN for TPU and its composites in static tube furnace is shown in Figure 5. The total concentration of HCN for pure TPU is 38 ppm. In sample group I, the lowest total concentration of HCN is shown in I -2 sample at 15 ppm. As for sample group II, the second lowest total concentration of HCN is shown in II-2 sample around 32 ppm. In addition, the total concentration of HCN of sample group III is appeared as similar as pure TPU. The total concentration of HCN for samples just added one of three flame retardants are higher than the pure TPU. It is shown that the release of HCN is reduced under certain addition (more than 9 wt%) of ATH and MMT, and ATH performs better than MMT in this way.
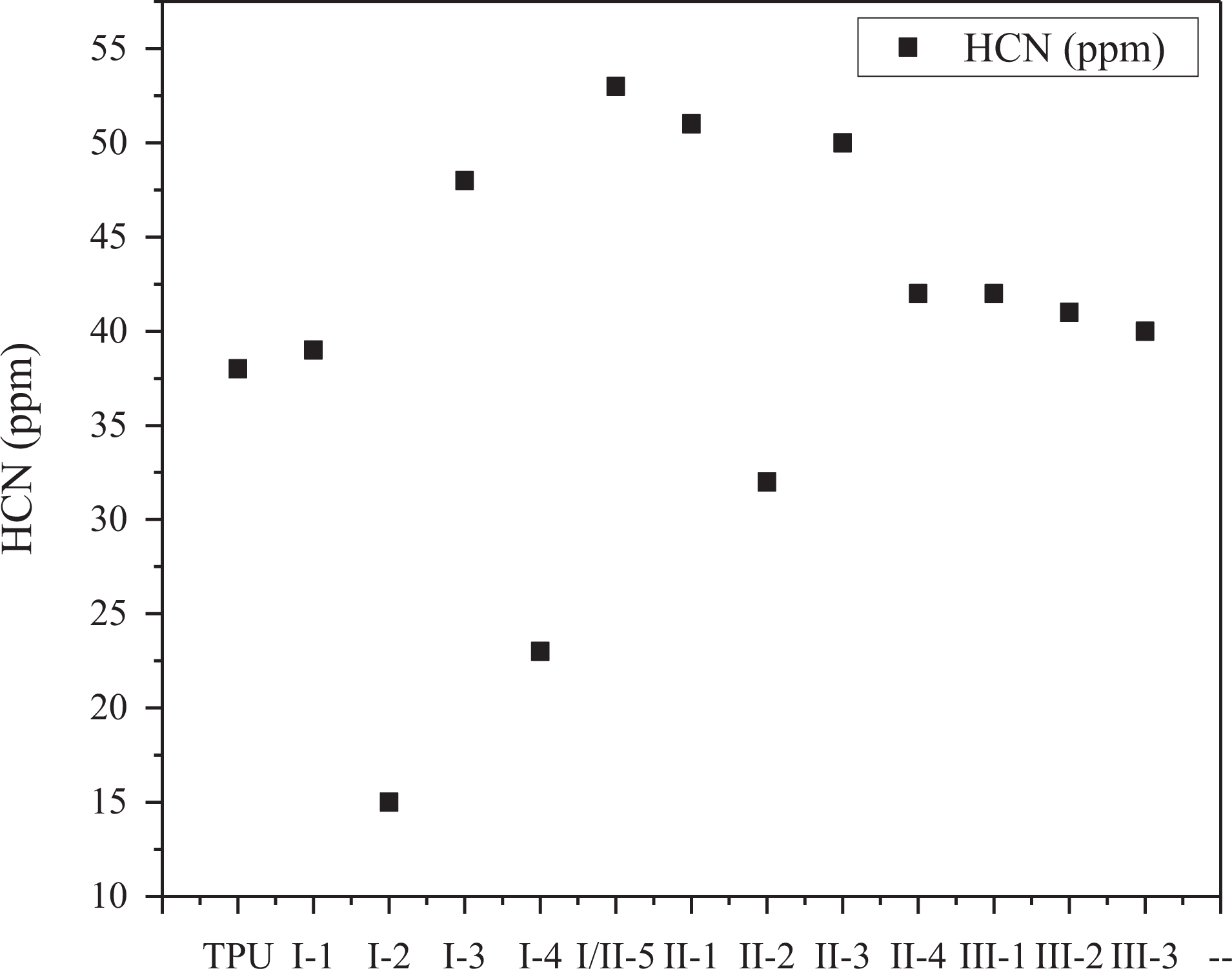
The total concentration of HCN for TPU and its composites.
CO and CO2 measurement
The amount of CO2, CO, and the depletion of O2 on SSTF test are shown in Figure 6. The results show that each sample has a certain time range (from 122 to 1746 s) as a relative steady stage. At that stage, the yield of gaseous product reaches at a steady value, which can be regarded as volume concentration. The steady yields of all gaseous product in the steady stage are represented in Table 3.
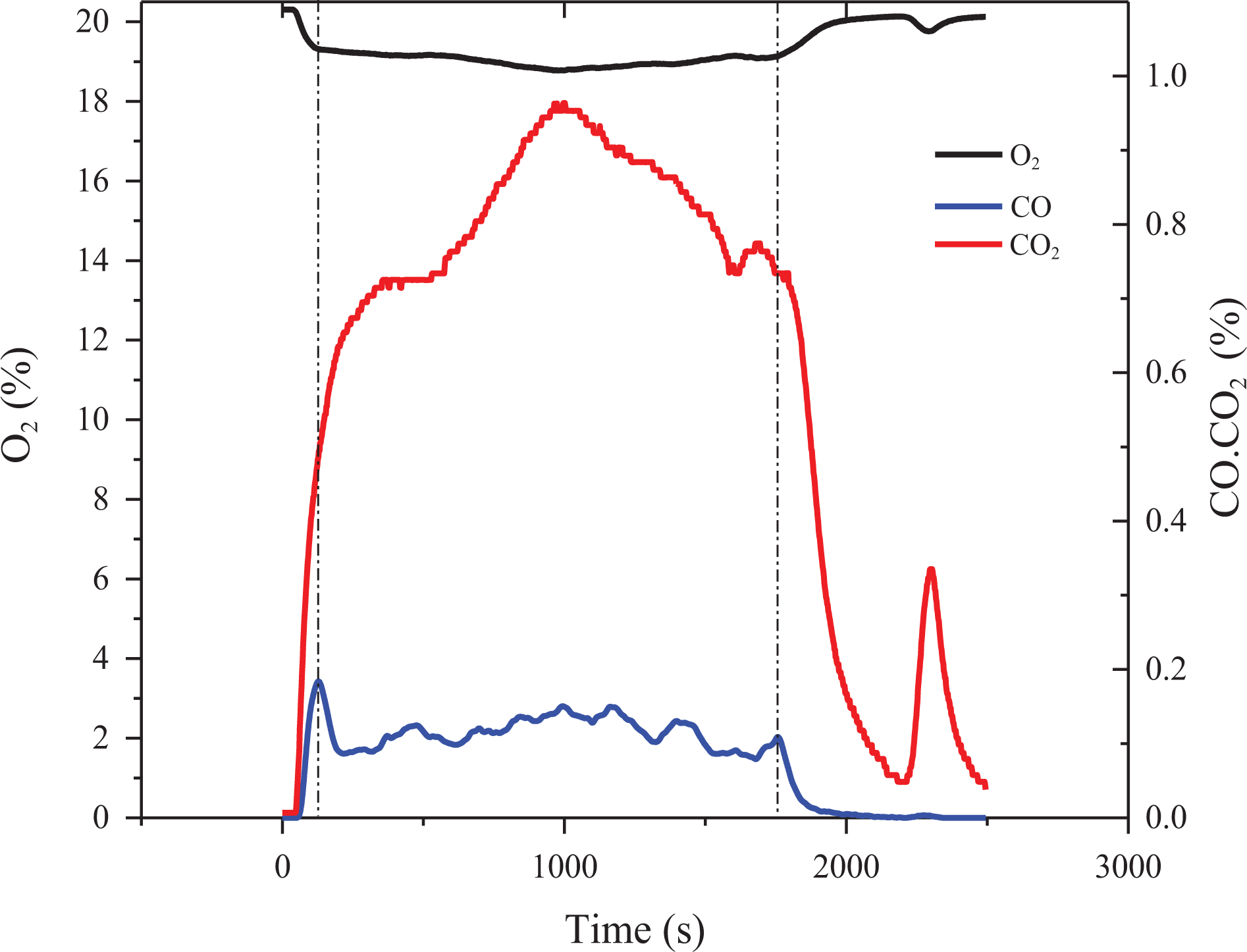
The example of curves of the CO, CO2, and O2 concentration versus time of TPU composites in SSTF test.
CO and CO2 and O2 concentration of TPU composites in SSTF test.
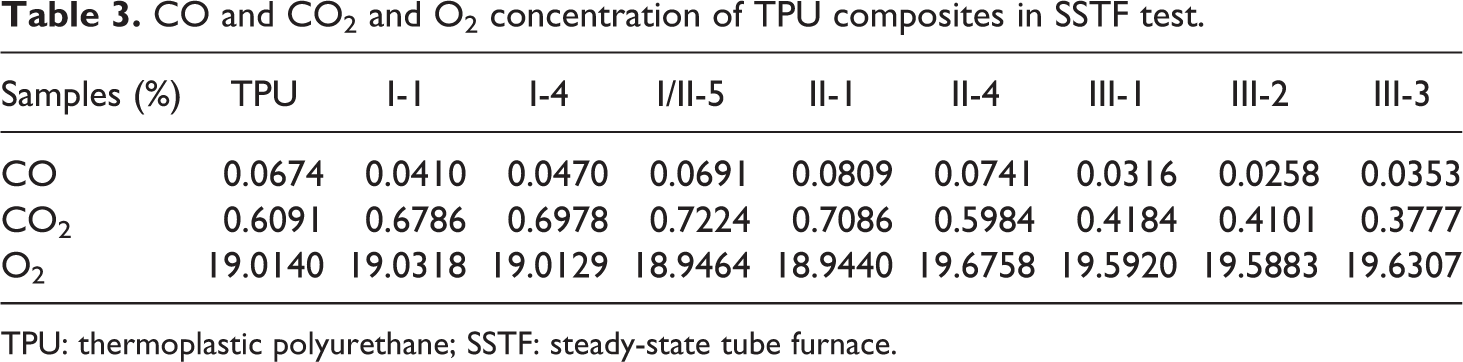
TPU: thermoplastic polyurethane; SSTF: steady-state tube furnace.
Yield of combustion product
The yield of combustion product is the ratio of the combustion product mass concentration and the combustion material mass. It can be evaluated by formula (1), the unit is g g−1
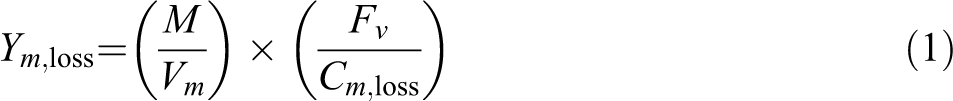
Ym,loss is the yield of certain component, g g−1. M is the molar mass of certain component, g mol−1. Vm=24.055 L mol−1, assumed as ideal gas which stands molar volume of certain component at 20°C and 1.013 bar. Fv is the volume concentration of certain component in smoke mixing box, %. Cm,loss is mass loss concentration, which can be got from the ratio of mass loss rate (g min−1) and total airflow velocity (50 L min−1 in this paper) in mixing box, g L−1.
In the under-ventilated fire condition, the yields of CO, CO2, the consumption of O2, and the ratio of CO2 and CO are shown in Figures 7 to 10. In Figure 10, the ratio of CO2 and CO is around 15. Compared with previous work, the ratio of CO2 and CO of common polymer in the well-ventilated condition is far more than 15, such as PMMA in the same situation reaches more than 200. 27 This phenomenon is mainly attributed to the incomplete combustion of TPU at the under-ventilated condition, low combustion efficiency contributes to a large amount of CO. Lower yield of CO and CO2 and lower consumption of O2 are appeared in sample group III than other groups, this is because of the two decomposition processes, including the decomposition from TPU to hydrocarbon and the decomposition from TPU to CO at 825°C. The two decompositions are competitive fiercely with each other, and the decomposition from TPU to hydrocarbon is strengthened with the interaction of APP, ATH, and MMT. Therefore, the yields of CO and CO2 have been both decreased, resulting in the decrease of O2 consumption, which is used in the transformation from CO to CO2.
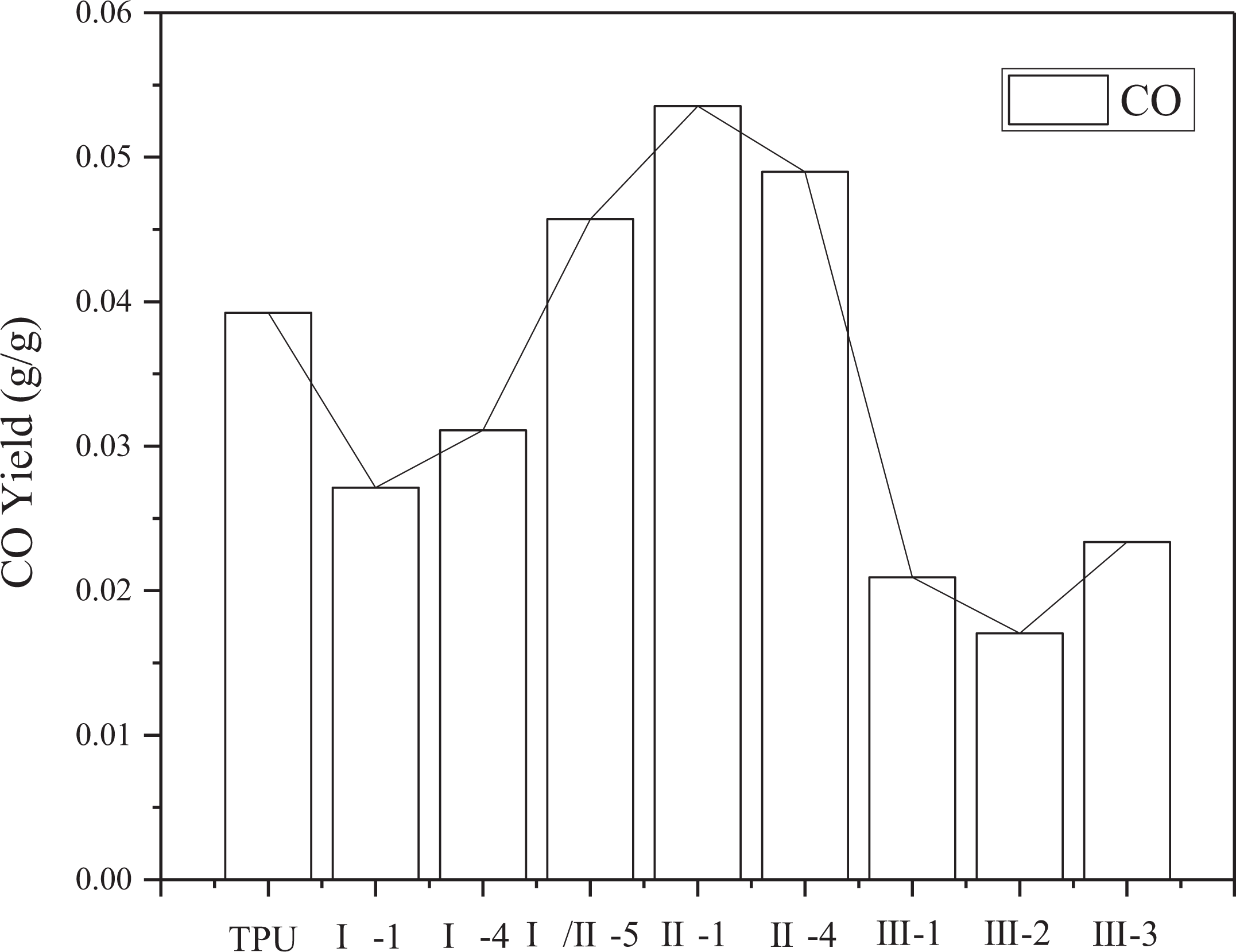
CO2 yield of TPU composites.
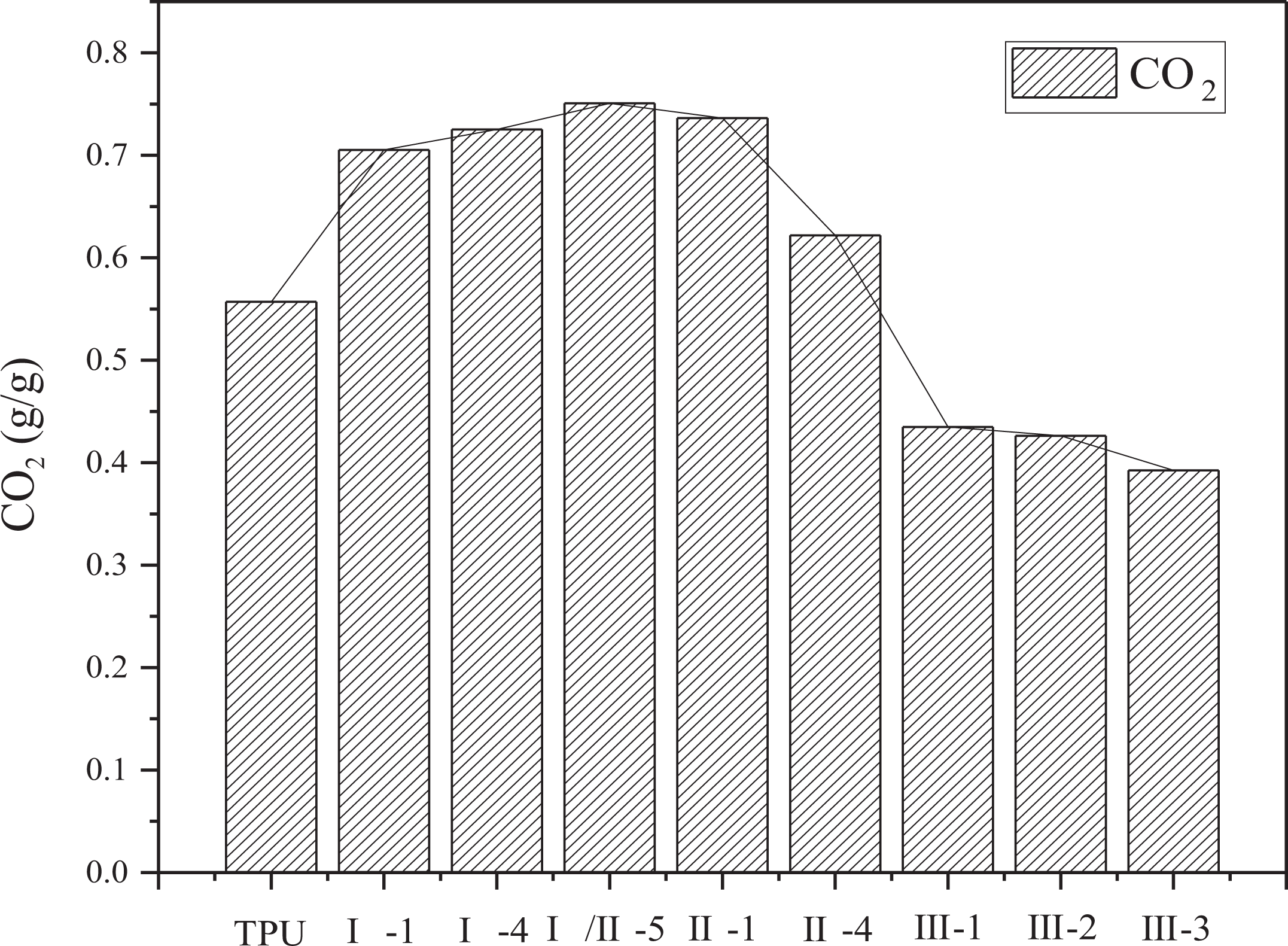
CO yield of TPU composites.
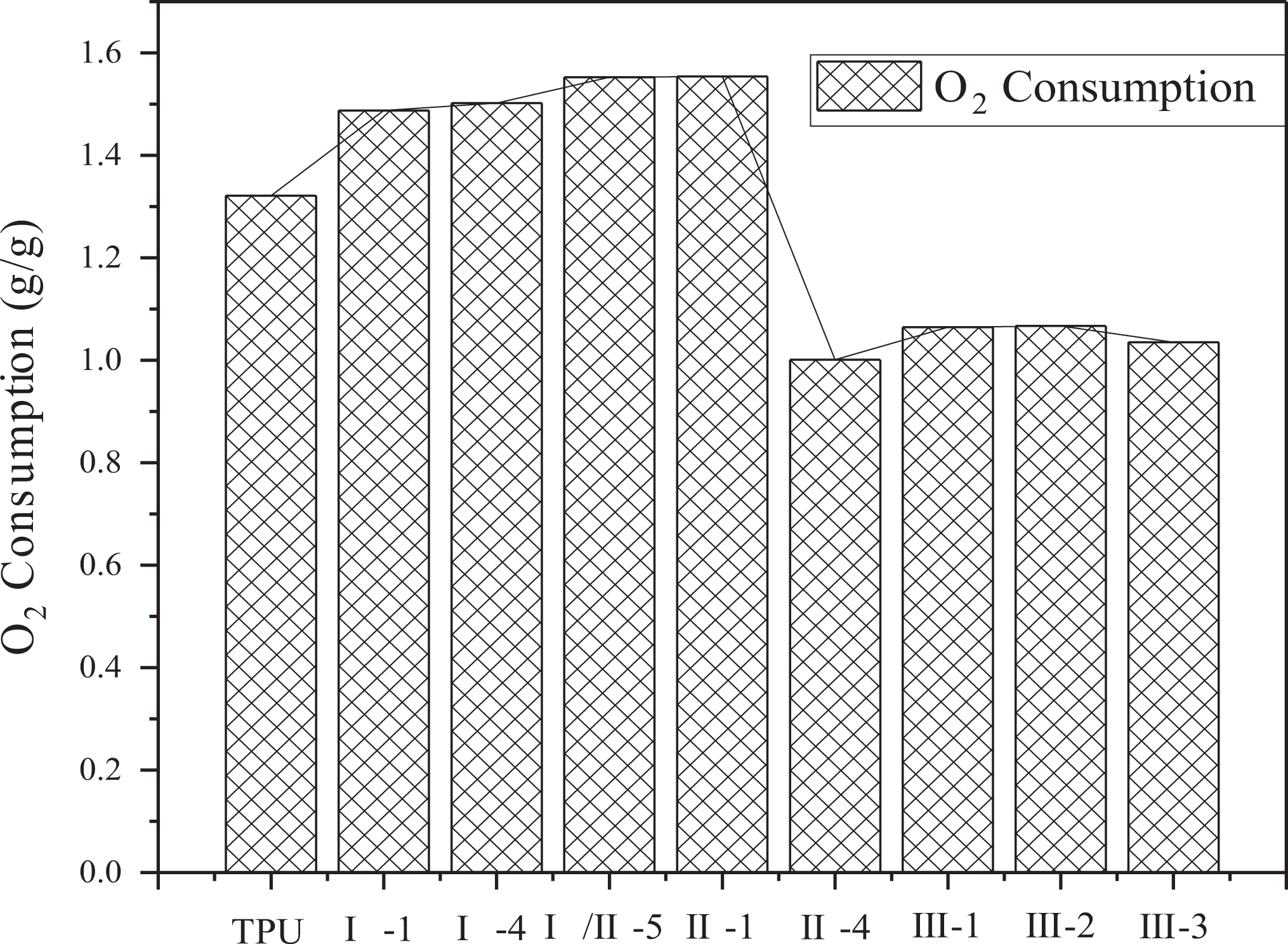
O2 consumption of TPU composites.
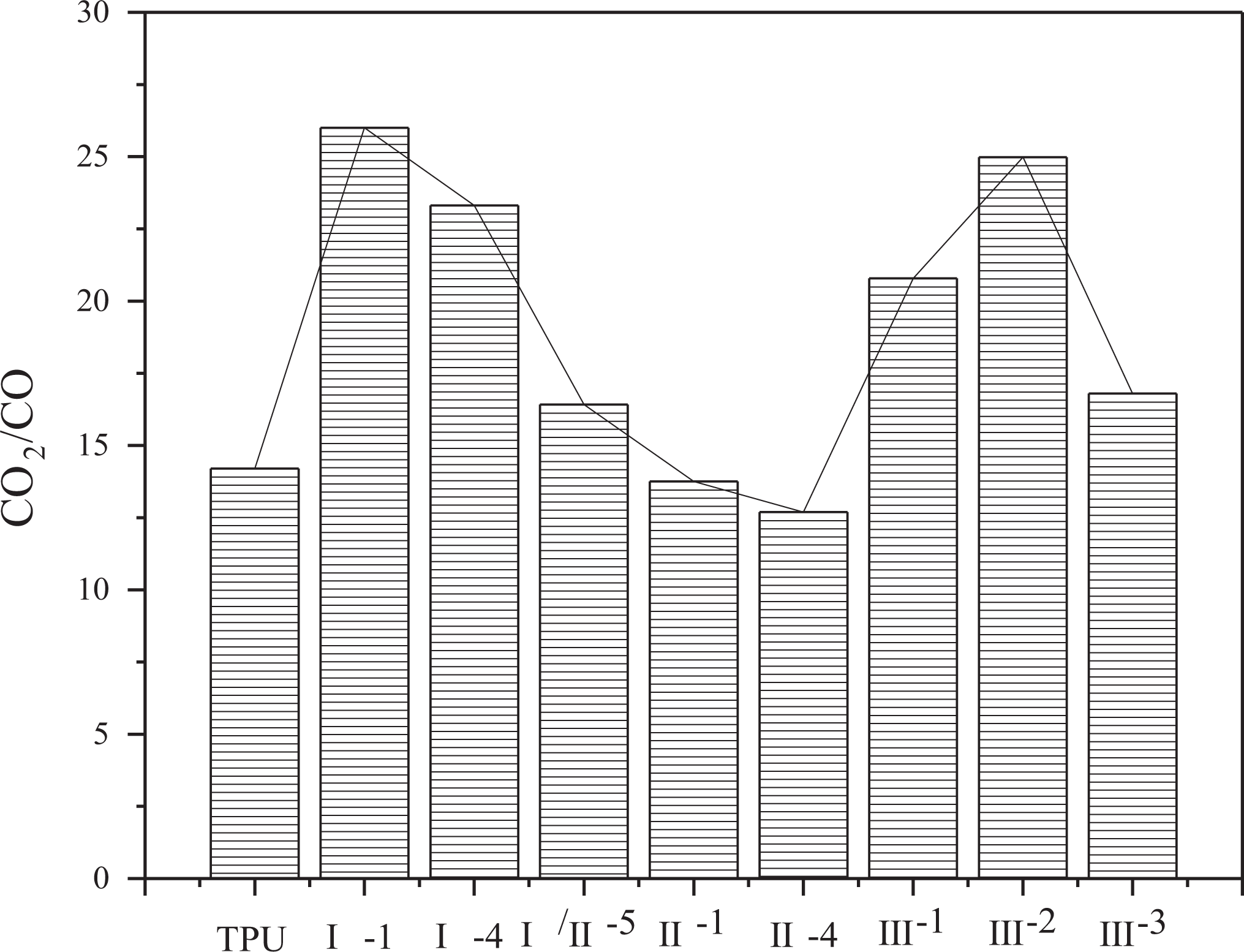
The ratio of CO2 and CO of TPU composites.
Fractional effective dose
Purser’s FED model expresses the ratio of the concentration of each toxicant to its lethal concentration and then multiplies the sum of this ratio by the hyperventilation factor. The higher the FED values, the greater the toxicity of the effluent is.
To analysis the toxicity of TPU composites, FED is calculated by formula (2). The concentrations of CO, CO2, and O2 are from SSTF, the concentration of HCN is from static tube furnace. The FED of TPU composites is shown in Figure 11. As shown in Figure 11, the comprehensive toxicity of pure TPU is reduced in sample groups I and III, the lowest toxicity is appeared at sample I-4. In sample group II, the reduce of toxicant is not obvious. As for the contribution of each gaseous toxicant, group I toxicant is reduced mainly by the decreased release of CO and HCN, the addition of MMT can help lessen the release of CO. Furthermore, more than 50% toxicity in FED for all samples is accounted by HCN, which infers that HCN plays the most important role than other toxicants.
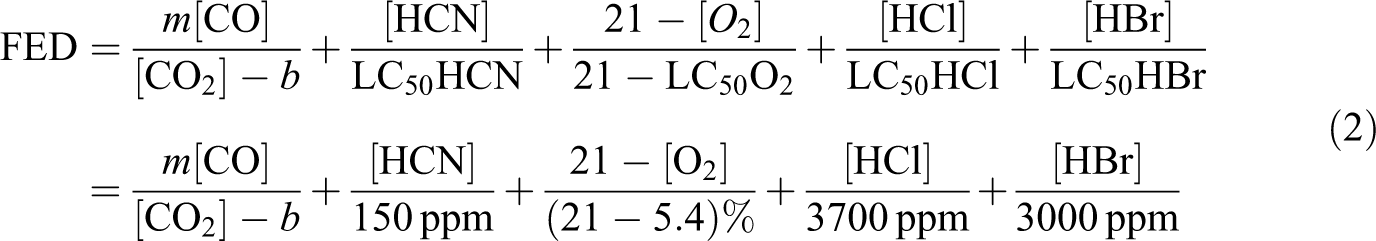

FED of TPU composites.
In this article, the toxic of HCN and CO is concerned primarily, so the HCl and HBr can be omitted. Then the formula (2) can be abbreviated as follows
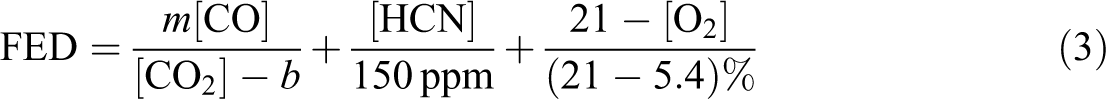
[ ] means each toxicity gaseous products volume concentration. When CO2 concentration is below 5%, m = 18, b = 122,000.
Conclusions
APP, ATH, and MMT are combined into three types of flame retardant at different prescriptions. APP-ATH, APP-MMT, and APP-ATH-MMT are melting blended into TPU to prepare TPU composites which have lower fire risk. TG-IR, static tube furnace, and SSTF are used to analyze the toxicity of TPU and its composites. The FED is calculated based on the concentrations of CO, CO2, and HCN. It can be concluded that.
The thermal stability of TPU at low temperature is weakened after the addition of flame retardants (APP, ATH, and MMT), but it is strengthened at high temperature. The char residue is increased.
HCN release of TPU composites is decreased with the amount (more than 9 wt%) of addition of ATH and MMT, whereas the effect of ATH is more obvious than MMT. The FED results of TPU and its composites reveal that the toxicity of HCN accounts for more than 50% of the total toxicity, so HCN is the major source of toxicant. The toxicity is cut down as a primary result of the decreased release of CO and HCN by APP-ATH, and the release of CO is reduced with MMT. The comprehensive toxicity of pure TPU is reduced in APP-ATH and APP-ATH-MMT TPU composites. Lower yields of CO, CO2, and O2 consumption can be found in APP-ATH-MMT composites than the other two sample groups.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (grant no. 51576185), the National Key Technology R&D Program (No. 2016YFC0802101).
