Abstract
This study investigates the influence of clay nanofiller on morphological, thermal, and mechanical behaviors of post-consumer polypropylene (PCPP)-based nanocomposites. Waste polypropylene was mixed with 1%, 3%, 5%, and 7% of Tunisian clay nanoparticles using twin-screw extruder. The structure and morphology of raw, purified, and organo-modified clays were characterized by Fourier-transform infrared spectroscopy and X-ray diffraction (XRD). The influence of clay nanoparticles content on the thermal behavior of waste polypropylene was studied by means of thermogravimetric analysis. The effect of nanoclay on morphology and properties of PCPP/clay nanocomposites was also studied. It was found from transmission electron microscopy and XRD analysis that clay nanoparticles are well dispersed into PCPP matrix and the addition of organo-modified Tunisian clay did not change the crystal structure of the PCPP polymer. Thus, organically modified Tunisian clay can be used for preparing organic–inorganic hybrids by melt processing with waste PP polymers. In dead, by varying the nanofiller loading, mechanical properties (Young’s modulus, tensile strength, and elongation at break) and thermal stability of filled PCPP showed an increase as compared to unfilled polymer. Optimal mechanical performances were obtained with 5% clay loading.
Introduction
Polypropylene is widely used in various industries such as packaging, geotextile, health care, aerospace, and automotive. 1 Because of this enormous use, every year millions of tons of polypropylene are being consumed and tons of plastic scraps are collected in the landfills causing environmental threats affecting ecology. 2 To overcome these problems, recycling process is one of the most successful solutions and transforms the collected materials into raw materials to manufacture new products. Nevertheless, the performance of reused PP proved to be insufficient for many applications, particularly with regard to its thermomechanical properties. 3
However, post-consumer polypropylene (often called recycled PCPP) could acquire desired properties by incorporating nanofillers into polymer matrix. 4 Most commonly used nanoparticles to reinforce recycled PP are montmorillonite (MMT), 5 graphene, 6 carbon nanotubes, 7–8 silica, 9–10 Boehmite alumina, 11 calcium carbonate (CaCO3), 12 and zinc oxide. 13 Among all nanoadditives, clay is the most preferred one for preparing polymer/layered silicate composites. 14
There are several techniques used for the manufacturing of polymer/layered silicate composites: solution intercalation, in situ interactive polymerization, and melt intercalation. 15,16 The last technique is the most relevant approach for preparing the materials used in this work because of its high productivity, low cost, its solvent-free operation type, compatibility with current industrial processes, such as extrusion and injection molding, and ease of application proper for any type of polymer matrices including polar ones. 17 In the processing equipment, the agglomerates of clays are broken down due to the external forces from the polymer melt and the diffusion of macromolecules into the gaps.
Nanoclays like purified Na+ or Ca2+ MMTs are hydrophilic in nature, which lead to incompatibility with the hydrophobic polymer matrix with subsequent poor composite properties. Therefore, to obtain a nanocomposite with requisite properties, the inorganic clay must be modified with some organic surfactant, usually onium salt or an alkyl amine which, compatibilizes the surface chemistry of the clay and polymer matrix at the interface by replacing the inorganic cation and making the gallery space of the clay sufficiently organophilic to permit the entry of polymer matrix. 18
Polypropylene, which is a thermoplastic linear hydrocarbon polymer, does not contain any polar groups on its backbone; it is not quite compatible with clay. In this case, homogeneous dispersion of the silicate layers in PCPP would not be realized, and improved properties are not obtained using modified hydrophobic MMT. For this purpose, the addition of an appropriate compatibilizer is also recommended to overcome this problem. Polar functional oligomers, such as maleic anhydride-grafted polypropylene (PP-g-MA), glycidyl methacrylate, diethyl maleate, acrylic acid, or maleic anhydride (MA), are used to facilitate exfoliation in addition to modifying clay surfaces. Best results have been obtained with PP-g-MA as compatibilizer. 19–20
Toyota group reported synthesis of PP/clay nanocomposites by direct melt mixing of organo-modified clays with PP-g-MA as an additive to enhance properties. 21 In their synthesis, a polyolefin oligomer with polar telecheric OH groups was inserted into the interlayer spacing of the clay. The basal spacing of the clay particles increased, and, as a result, the interaction of the layers should be weakened. At the second stage, PP was inserted into the intercalation between surfactant agent and polymer oligomer, and the PP/clay nanocomposites were obtained. 22,23
In the present work, the organically modified Tunisian clay (organoclay) was prepared by treating Na-clay with a bromide surfactant by solid state method. The solid state technique has some advantages, like cheapness, simple processing, and being harmless to the environment. Then, organo-modified Tunisian clay was used as reinforcement to formulate PCPP/clay nanocomposites, using PP-g-MA as compatibilizer. Throughout the study, the mineralogical and morphological characteristics of PCPP/Tunisian clay nanocomposites were evaluated. Stress–strain behavior was used to evaluate mechanical properties of obtained materials. Thermal analysis of the nanocomposites was performed using differential calorimeter apparatus.
Experimental
Materials
PCPP used in this study is obtained from woven bags used for food packaging. It has a melt flow rate of 11 g/10 min−1 (230°C, 2.16 kg), a density of 0.905, and a melting temperature around 167°C.
These woven bags are cleaned, washed, and dried. Then, polymer was converted into fine powders using an MFI-MVI 3350 machine (Matériaux Ingénierie Limited, France) and a chisel.
Tunisian clay investigated in this study was smectitic clay obtained from sit of Coniacian–Santonian outcroppings of Jebel Meloussi (Sidi Bouzid region) located in the meridional Atlas of Tunisia.
Clay particles were organo-modified using trimethyloctadecylammonium (TOMA) bromide salt (99% purity) of as surfactant. Cationic surfactant, with an average molecular weight 392.5 g mol−1, was purchased from Sigma-Aldrich (St. Louis, Missouri).
In the current study, a PP-g-MA was used as a compatibilizing agent. It was purchased from Sigma-Aldrich, Lyon, France (3.1 wt% of MA content, Mw = 47,100 g mol−1, Mw/Mn = 1.9).
Processing
Purification of Tunisian clay nanoparticles
There are two steps necessary to prepare purified clay: activated clay and the sodium-saturated clay sample. 24 For preparing acid-activated clays, a suspension was made by dispersing 10 g of raw sample in 100 mL of 2.5 mol L−1 H2SO4 at 60°C for 1 h. The resulting suspension was stirred continuously in a jacketed glass reactor, equipped with water circulation condenser and a thermometer, at 80°C for 2 h 30 min. After cooling at room temperature, the suspensions were centrifuged at 3500 r min−1 for 10 min. The solid content was immediately filtered off and then washed several times with double-distilled water until neutral point obtained with pH indicator (5% BaCl2 test). The clay residue was dried in oven at 110°C for 24 h prior to use. For preparation of sodium-saturated clay sample (Na-clay), 1 L of 2 mol L−1 NaCl solution is added to the 20 g of purified clay and was mixed at 500 r min−1 with magnetic stirrer for 24 h for five times at room temperature. Na-saturated clay was filtered and washed with distilled water several times until chloride-free as confirmed by the AgNO3 test. The product was dried in the oven at 80°C. Dry activated and purified clay was then crushed, ground, and then sieved to obtain a particle size of 40 µm using stainless steel testing sieve.
Organo-modification of Tunisian clay
The intercalation of the surfactant into Tunisian clay was carried out by solid–solid reactions. 25 Syntheses of surfactant-modified Tunisian clay were performed by mixing an amount of purified clay, initially dispersed in 100 mL of deionized water at 70 ± 5°C, with an organic surfactants dispersed in 200 mL of deionized water, added slowly to the clay suspension. The resulting organophilic clay suspension were stirred for 30 min and allowed to stand at room temperature for 24 h.
After maturation, centrifugation, and several successive dialyses, the prepared organo-modified clay materials were filtered, washed with distilled water four times to remove any excess surfactants, until free of bromide anions (tested by AgNO3). Then, the resulting organoclay was dried at 80°C temperature for 48 h under vacuum and ground in an agate mortar at room temperature to pass through a 40 µm mesh sieve.
Elaboration of PCPP/Tunisian clay nanocomposites
Nanocomposites were prepared via melt mixing using a Haake–Rheocard twin-screw extruder under the following conditions: die temperature 180°C, extrusion speed 50 r min−1, and residence time of 5 min. Compositions of elaborated composites are reported in Table 1.
Compositions of prepared nanocomposites.

PCPP: post-consumer polypropylene.
Characterization
Infrared spectroscopy
The Fourier-transform infrared (FTIR) in diffuse reflectance mode was used to investigate the structural changes of different clays. The infrared (IR) spectra were recorded using a Perkin-Elmer model (783) spectrometer with a smart endurance single bounce diamond Attenuated Total Reflectance (ATR) cell. Spectra over the 4000–400 cm−1 ranges were obtained by the coaddition of 64 scans with a resolution of 4 cm−1 and a mirror velocity of 0.63 cm s−1. Potassium bromide (KBr) pellet technique was prepared by mixing 2 mg anionic clay with 200 mg KBr and pressing.
X-ray diffraction analysis
The structure of the raw PCPP and its nanocomposites was examined by X-ray diffraction (XRD) analysis at ambient temperatures using an X’PERT PRO X-ray diffractometer (Spectris Co Ltd, Japan). An acceleration voltage of 40 kV and a current of 30 mA were applied using nickel-filtered Cu-Kα radiation at a wavelength of λ = 1.541 Å. Diffraction angle 2θ was scanned from 1° to 50° at a scan rate of 1° min−1 with a step size of 0.01°.
XRD analyzed samples are thin films with 2 mm thickness that were obtained by compression molding at 180°C.
Transmission electron microscopy
Morphologies of prepared PCPP/clay nanocomposites were examined by Transmission electron microscopy (TEM). TEM observations were performed on a Jeol-2000CX TEM (Arishima, Tokyo, Japan) at an accelerating voltage of 120 kV.
TEM imaging was carried out on ultrathin sections prepared using an Ultra cut cryo-microtome (Leica Microsystems, Wetzlar, Germany) equipped with Cryotrim and 2 mm Cryo diamond knives (Diatome, Biel, Switzerland). Tested ultra thin samples were with nominal thickness of 50 µm.
Thermal analysis
Thermal behavior of our samples was evaluated by thermogravimetric and differential thermal analysis (TGA-DTA) methods. Thermal analysis tests were performed using differential calorimeter apparatus TG-DTA 92 SETARAM with a heating rate of 10°C min−1 and using α-Al2O3 as the inert marker. Thermogravimetric data were recorded from 25°C to 600°C.
Mechanical characterization
Mechanical properties are related with the response and resistance capacity of the materials to load deformation applied in various manners. In this study, tensile tests were conducted in order to evaluate the degree of improvements in stiffness and toughness properties owing to modifications. The effects of organo-modified clay and compatibilizer compositions on performance of the composites were analyzed. The tensile strength tests for PCPP and its related composites were carried out according to the ASTM D-638 on a Universal Tensile Testing Machine Hounsfield H10KS (Tinius Olsen Company, Redhill, United Kingdom). Test samples were prepared by compression molding at 180°C and cooled at room temperature. Then, specimens were conditioned at the temperature of 25°C and the relative humidity of 50% for 24 h. The standard samples dimensions are shown in Figure 1. For each material, five samples were tested and the mean values were considered. The crosshead speed was 50 mm min−1.
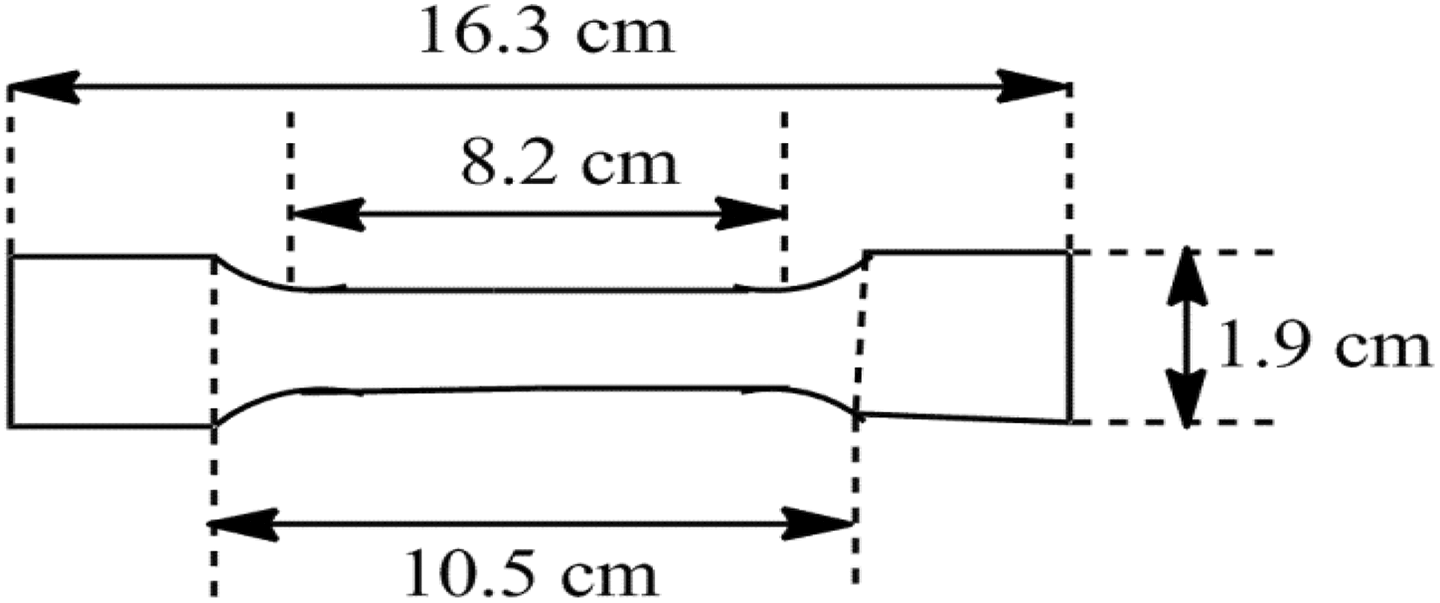
Standard sample dimensions.
Results and discussion
Characterization of raw, purified, and organo-modified clays
Infrared spectroscopy analysis
FTIR spectroscopy was used as sensitive tool to assess the molecular environment of the intercalated surfactant within the organoclay, based on the comparison of the different absorption bands of the functional groups present in the clay before and after its modification.
On FTIR spectrum of raw clay (Figure 2(a)), we can see that the bands at 420 cm−1 are characteristic of quartz. The band appeared at 1433, 711.6, and 871.6 cm−1 confirms the presence of carbonate (dolomite (Ca, Mg (CO3)2) or calcite (CaCO3)). 26 Absorption bands at 3627 and 912.1 cm−1, attributable to Al–Al–OH− stretching and bending modes of vibrations show that the smectitic phase has a dioctaedric character. 27,28 The bands of vibration of deformation of hydroxyl groups on the edges of the mineral clay layers are observed at 871.6 cm−1 for (Fe, Al–OH), at 796 cm−1 for (Mg, Al–OH), and at 693 cm−1 for (Mg, Fe–OH). 29 The broad bands at 1026 and 1100 cm−1 are characteristic of the vibrations of Si–O valence in the plan and apart from the plan. 30 Other bands appeared at 471, 420, and 527 cm−1 are attributed to the banding vibration of Si–O–Mg, Si–O–Si, and Si–O–Al (octahedral Al), respectively. 31 The peaks of absorbance from the purified clay and organo-modified clay appear approximately at similar wave numbers; however, their intensities and widths are different. The absence of band attributed to carbonate residues in the spectrum of the purified clay suggests that this residue is removed during the organic clay modification, possibly during the washing step.
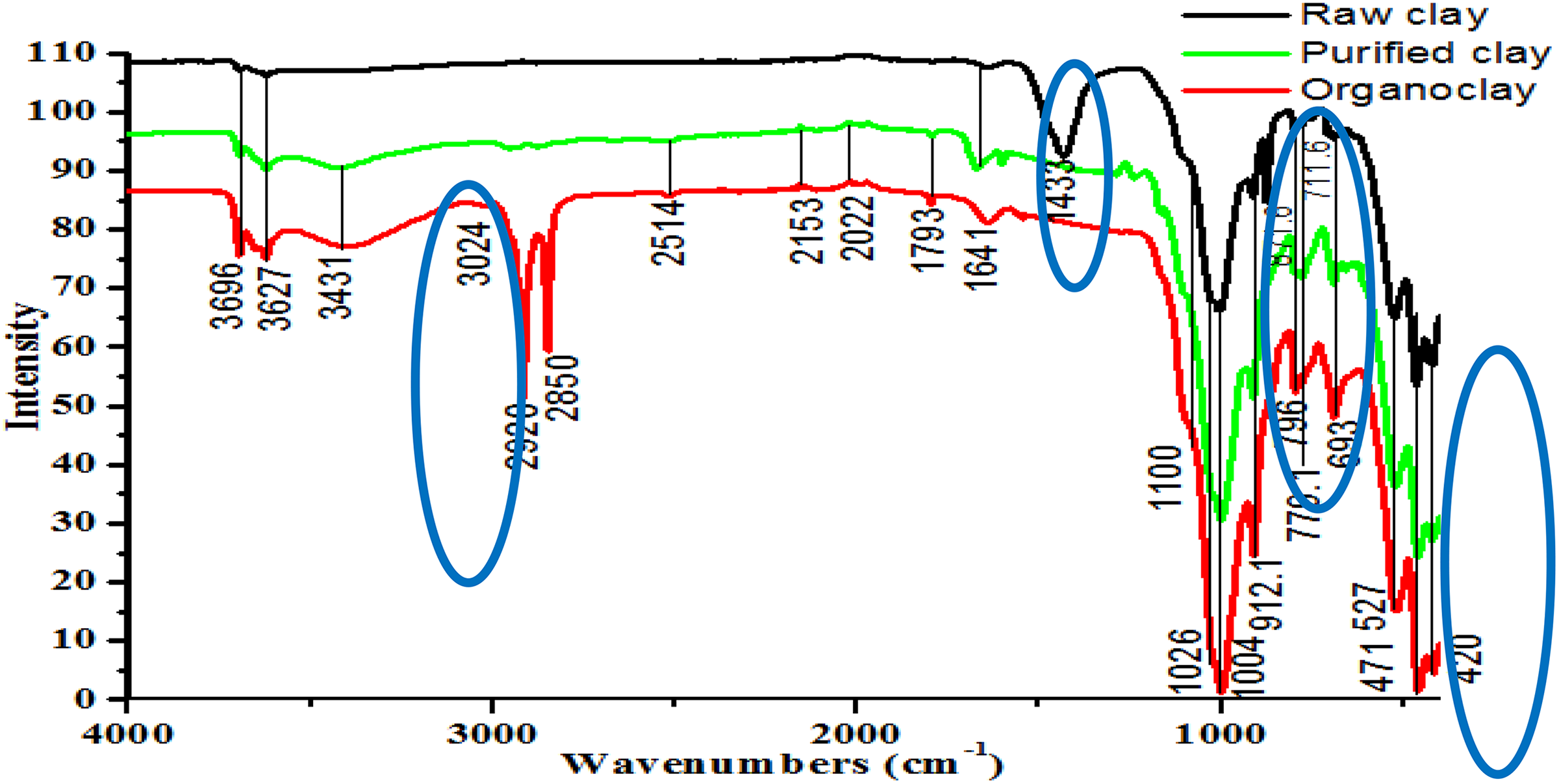
IR spectra of raw, purified, and organoclay. IR: infrared.
As shown for organophilic clay spectrum (Figure 2(c)), the new peaks that appeared at 2920 and 2850 cm−1 are associated with C–H stretching of –CH2 groups and aliphatic C–H stretching, respectively, which are absent in the purified and the raw clay. These absorption bands indicate that the surface modification of exchanged clay is achieved by the surfactant. Therefore, the surfactant molecule is incorporated within the nano-space of the purified clay without altering its chemical structure. 32
In the IR spectra of purified and organo-modified clay, there was a distinct increase in the IR bands at 3696, 3627, and 912.1 cm−1 compared to the raw clay, confirming the dominant presence of dioctahedral smectites with (Al, Al–OH) stretching and bending bands. 33,34 The bands near 3431 and 1641 cm−1 which persist in both the purified and the modified clay are attributed to H–O–H stretching vibrations of free water molecules adsorbed on the clay surface and to the bending mode of the water molecule adsorbed on the mineral clay, respectively. Moreover, the intensity of stretching and bending band at 420 cm−1 that appears in the spectrum of the crude sample intensively decreased in the purified and the organoclay, indicating some elimination of the amount of quartz. 35
Carbonate calcite (Ca, CO3) or dolomite (Ca, Mg(CO3)2) traces were strongly evident at 1433, 711.6, and 871.6 cm−1 in the raw clay disappeared in the spectrum associated with the purified and organo-modified clay samples. 36 This is explained by cation exchange between Ca2+ cations of the clay and H+ from the acid. This result, compared to that obtained for raw clay, shows that the purification treatment allowed almost a total elimination of calcites. 37,38 The position of the band at 3431 cm−1 and of the peaks at 3627, 3696, 1641, 912.1, 1004, 1026, 796, 779.1, 693, 527, and 471 cm−1 did not shift after the organophilization, which is an indication that these procedures did not modify the Tunisian clay structure.
Mineralogical properties
XRD patterns of raw, purified, and modified clays investigated in this study are reported in Figure 3. A natural clay sample is a mixture of different clay mineral types with major proportions of kaolinite. The peaks at 7.17, 2.56, and 1.49 Å are the characteristics peaks of kaolinite clay mineral. Calcite (reflection at 3.03 Å) and Quartz (reflection at 3.33 Å) are the major impurities. 39 The characteristic diffraction pattern d001 of Tunisian clay (Figure 3(a)) was displayed at 7.12 Å without showing any significant change for purified clay (Figure 3(b)) and organoclay (Figure 3(c)), indicating that the layered structure of raw Tunisian clay is preserved after the functionalization process. This can be explained by that Tunisian clay has a strong interaction of hydrogen bonds between the lamellae which hinders the organo-modification process without a previous intercalation with surfactant molecules.
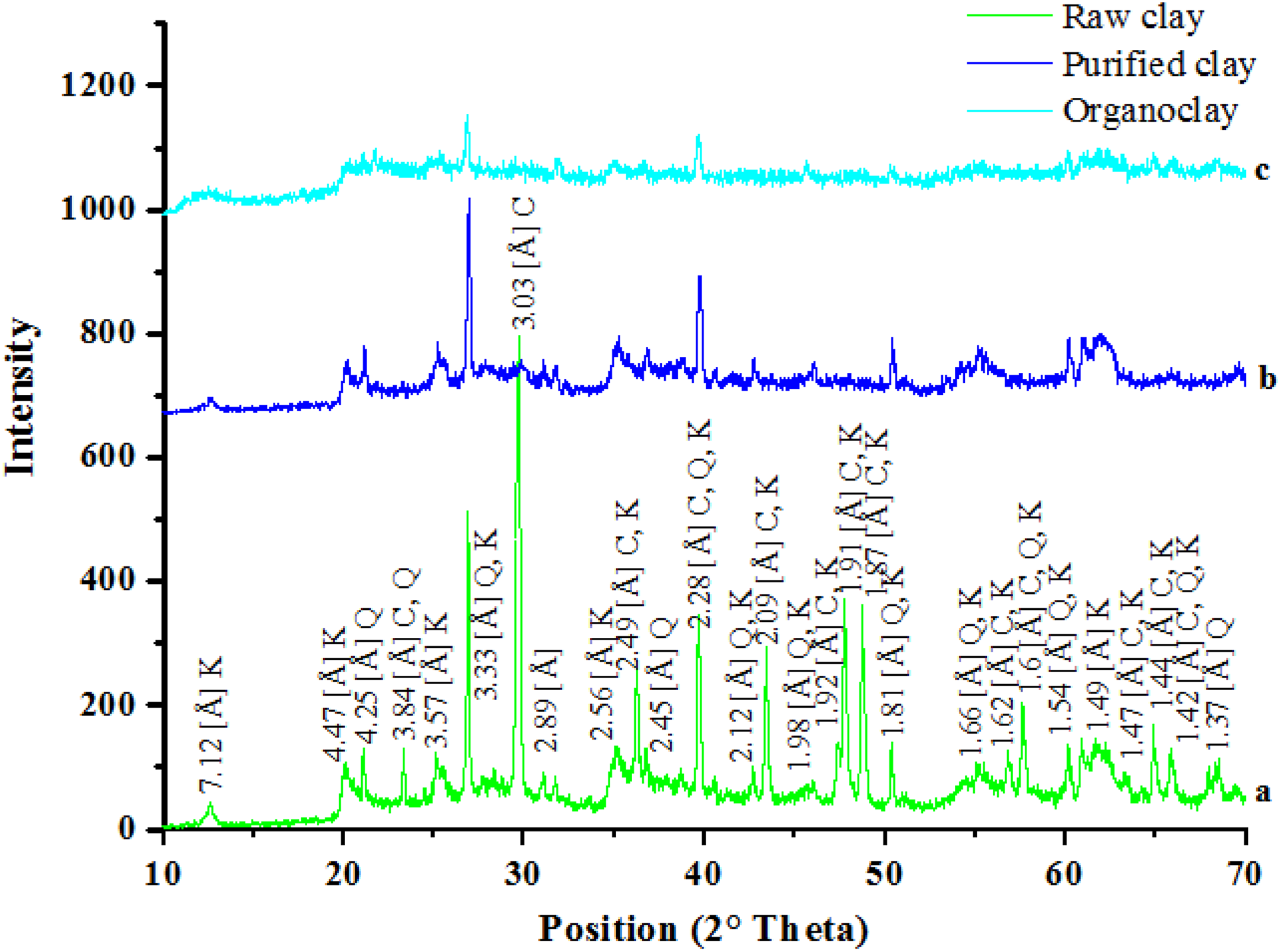
XRD patterns of raw, purified, and organo-modified clays. XRD: X-ray diffraction.
We also note that only the peaks relating to the argillaceous fractions are present in the diffractogram of purified clay. We notice the total disappearance of the peaks at 3.03, 3.84, 2.49, 2.09, 1.91, and 1.87 Å relating to calcite: This is explained by the cation exchange that occurs between the cations Ca2+ of clay and H+ of the hydrochloric acid which is proved by the chemical composition by the reduction of fraction of CaO of activated clay sample. 40
Indeed, purified treatment generates a better crystallization of quartz present in the layer of Tunisian clay, this is observed by the smoothness of the peaks relating to this phase. Quartz as the major impurity was not totally eliminated by purification. 41
The peaks from the purified clay and organo-modified clay appear approximately at similar position; however, their intensities are different. Indeed, the intensity of peak at 4.25, 3.33, 2.28, 1.81, 1.66, and 1.54 Å that appear in the spectrum of the crude sample intensively decreased in the purified and the organophilic clay, indicating some elimination of the amount of quartz after organophilization treatment.
From morphological and structural analysis of clay nanoparticles, we can affirm that organo-modified clay is suitable to elaborate PCPP/clay nanocomposites.
Structural analysis of PCPP/clay nanocomposites
Mineralogical properties
Figure 4 shows the XRD patterns of raw PCPP and its related nanocomposites. The XRD pattern shows that the addition of clay did not affect the crystallographic nature of the PCPP matrix. In both raw PCPP and PCPP loaded with Tunisian clay, the crystal plane of PCPP is monoclinic, showing reflections assigned to planes (110), (040), (130), and others. 42 It is observed that five characteristic peaks of PCPP structure appear in the raw PCPP and nanocomposites at 2° angles of about 14.0°, 16.8°, 18.6°, 21.2°, and 21.9°, corresponding to lattice planes of (110), (040), (130), (111), and (041), respectively. 43
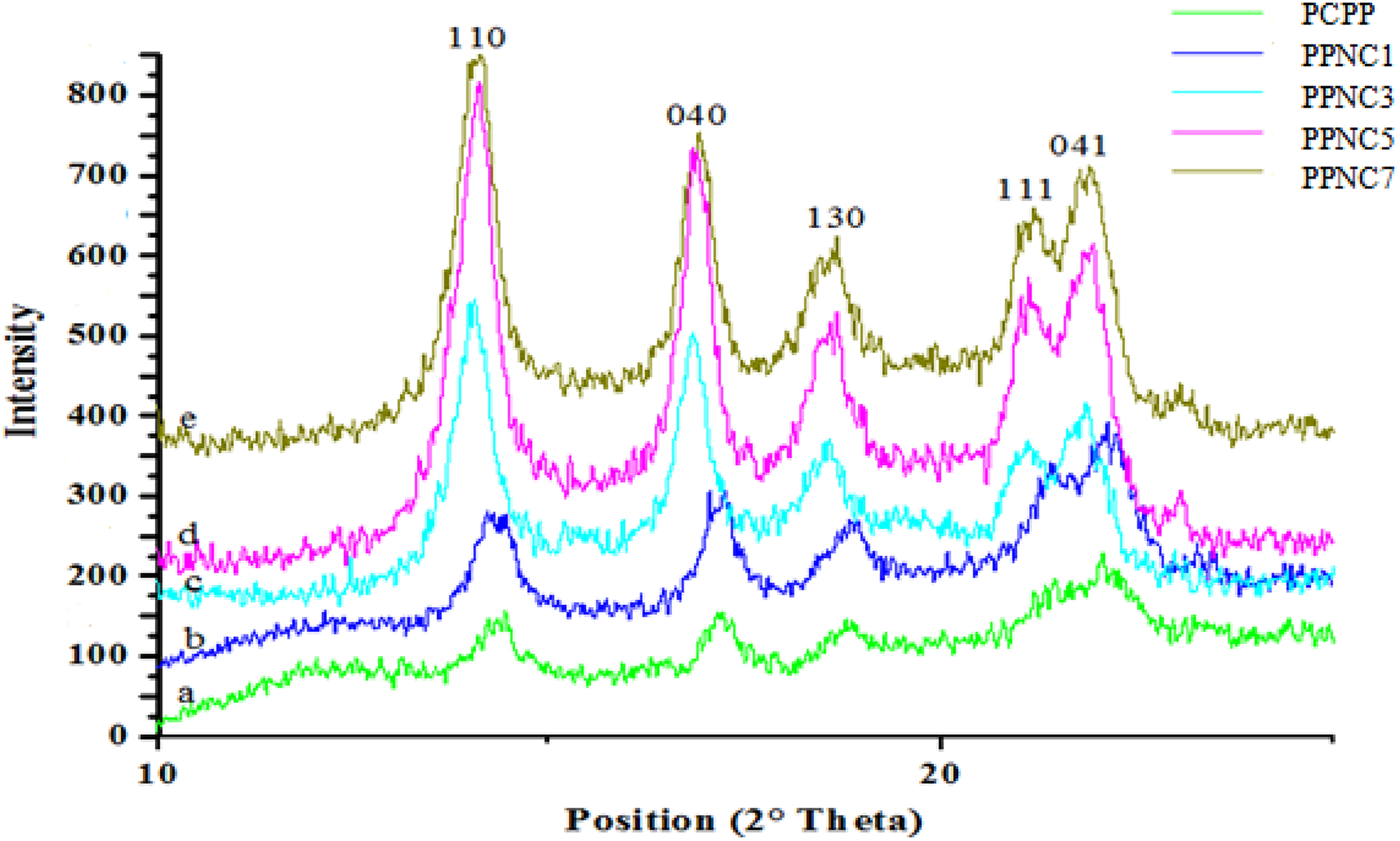
Crystal structures of PCPP polymer and PCPP/clay nanocomposites. PCPP: post-consumer polypropylene.
In addition, the same numbers of XRD peaks are detected apart from the different relative peak intensities, indicating that the addition of clay particles does not greatly alter the crystalline structures of PCPP matrix. 44 Peaks intensity of examined nanocomposites is considerably enhanced, especially in PP nanocomposite with 5% clay content (PPNC5), leading to the significant improvement of properties as previously indicated. 45
Transmission electron microscopy
XRD analysis is useful in characterization of nanostructures and dispersion properties if diffraction peaks are observed in the small angle region. Such peaks represent the d-spacing and presence of ordered-intercalated and ordered-delaminated nanocomposites. However, in the case of disordered structures of our nanocomposites, XRD peaks are no longer visible due to loss of structural registry or large d-spacings or both. In such cases, TEM analysis could provide definitive information regarding to the structure of the nanocomposites and direct observation of filler arrangements in the composite structure.
Figure 5 shows representative TEM photographs for various clay contents, in which the dark areas represent the individual Tunisian clay layers or agglomerates (tactoids) while the gray part represents the recycled polypropylene matrix.

TEM micrographs of (a) PPNC1, (b) PPNC3, (c) PPNC5, and (d) PPNC7. TEM: transmission electron microscopy.
According to transmission electron micrographs, clay particles are finely dispersed within the PCPP matrix indicating that the morphology changes from an intercalated structure to a more delaminated morphology.
Comparing Figure 5(a) to (d), it was observed that the state of dispersion was enhanced and particles became smaller and were dispersed more uniformly as the Tunisian clay was increased. Indeed, composites having high percentage of clay (over 5 wt%) meanwhile very little and small sized agglomerates which tends to prove that clay particles were well finely dispersed. 44
For PPNC5 (Figure 5(c)), the individual silicate layers are really observed. Indeed, the reduction in particle size is significant. These results could be attributed to enhanced interfacial adhesion due to the increased amount of PP-g-MA and also to the well dispersion of organo-modified clay layers inhibiting possible coagulation of the dispersed phase. 46 The ability of PP-g-MA polar functional groups to interact with the edges of nanoclay polar layers facilitates the intercalation of polymer chains in the clay galleries. Moreover, interaction between the MA group and the organoclay would cause an increase in viscosity value and reduction in the particle size as a result of application of high shear and enhanced dispersion. Results are in agreement with the ones found by Kim et al., 47 whose stated that the degree of dispersion of the MMT increases as the ratio of compatibilizer PP-g-MA to organoclay increases.
Mechanical behavior of nanocomposites
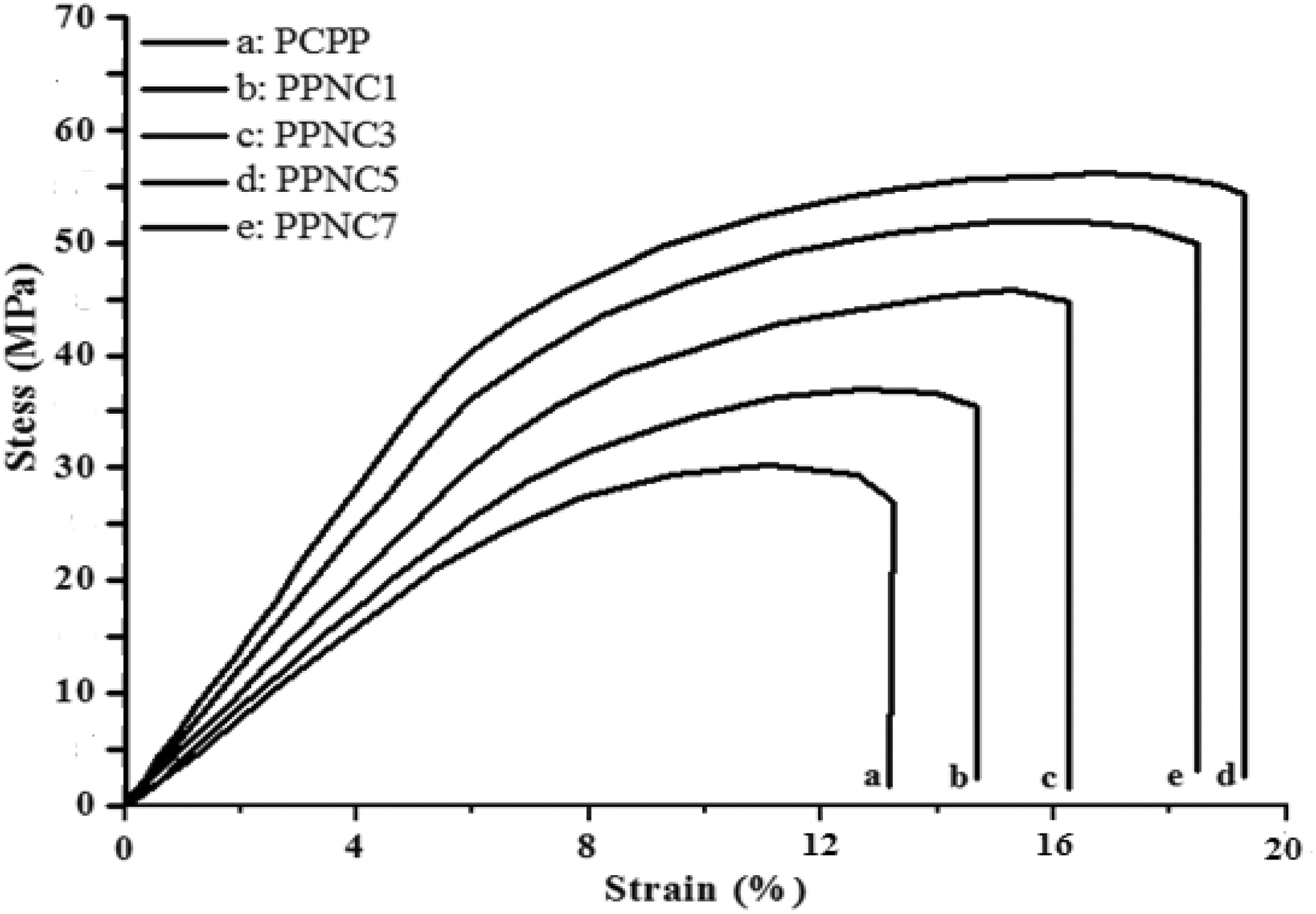
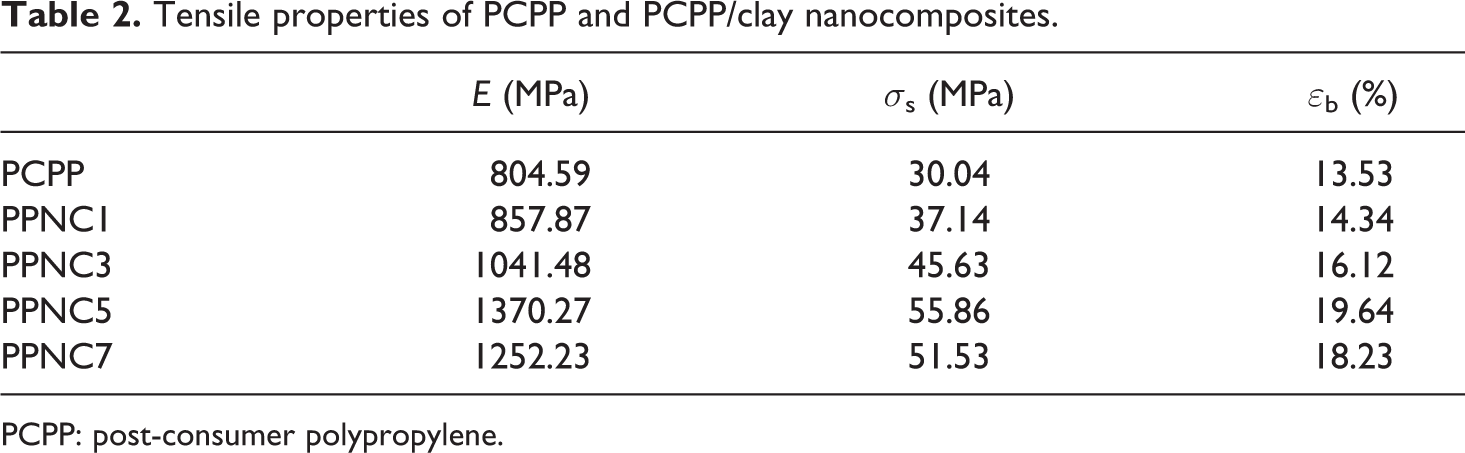
A large aspect ratio, good compatibility at filler–matrix interface, and high quality of dispersion of clay in the polymer matrix are the key points needed to achieve polymer nanocomposites with enhanced properties. The variation in the mechanical behaviors of PCPP/Tunisian clay nanocomposites with clay content is shown in Figure 6. The extracted values of the Young’s modulus (E), tensile strength (σs), and elongation at break (εb) are summarized in Table 2.
Stress–strain curves of PCPP and PCPP/clay nanocomposites. PCPP: post-consumer polypropylene.
Tensile properties of PCPP and PCPP/clay nanocomposites.
PCPP: post-consumer polypropylene.
It can be seen on stress–strain curves of Figure 6, that the increase in the clay loading improves the tensile strength and modulus of the composites compared to raw PCPP. The tensile strength and modulus increases about 71.53% and 55.63%, respectively, with the incorporation of 7 wt% and reaches a maximum value (85.95% and 70.3%, respectively) with a clay content of 5 wt% compared with that of the raw PCPP. Intercalation of the polymer chain inside the Tunisian clay layers enables to an increase in contact area between polymer matrix and clay filler and confines the segmental movement of the PCPP macromolecules; the increase in modulus value for this composition is expected. 48 The increase of the tensile strength of nanocomposites indicates the interaction of clay within the PCPP matrix which is possibly due to the presence of PP-g-MA that facilitated the expansion of the gallery space of layers by enclosure of polar groups to intercalate between the clay layers through secondary interaction. 49
In fact, the compatibilizer (PP-g-MA) has a positive effect in the composite reinforcement and provides an additional improvement in Young’s modulus of composite. This may be due to increased bonding between the anhydride group and the oxygen in the surface of the lamellae. The increased tensile properties may be attributed to the high aspect ratio of stiff silicate layers in the PCPP that results in a higher extent of interaction with the polymer chains and good interfacial adhesion between the clay particles and the polypropylene matrix, so that the mobility of polymer chains is restricted under loading. It is well known that the tensile strength depends on many factors such as various types of constituent of the system, dispersion degree, interactions between the clay layer and PCPP matrix and also particle to particle interactions. 50
With further increase in clay loading from at 7 wt%, a decrease in the tensile strength and modulus of the nanocomposites was observed but remained higher than raw PCPP mechanical characteristics. This effect could be due to the fact that as the filler content increases agglomeration among clay particles inside the polymer matrix also increases. This agglomeration results in a reduction of the organo-modified clay aspect ratio, thereby decreasing the contact surface between the clay filler and the polymer matrix. However, the agglomeration of clay particles also induces a local stress concentration within the composite. Thus, during tensile deformation, nanocomposites containing higher filler content deform in a brittle manner and show relatively lower tensile strength. These results were similar to those of Chandran et al. 51 Indeed, the tensile strength of PPNC7 is 51.53 MPa, whereas it is 55.86 MPa for PPNC5 and only 30.04 MPa for the neat PCPP. This indicates that clay was dispersed more uniformly through the PCPP matrix at low concentrations (1–5 wt%) to increase the surface attraction between the clay and the polymer matrix and then decreased for clay concentrations over 5 wt%. In addition, these results are in accord with those of Risite. 52
The PPNC elongation at break increases with the increase of the clay loading and it is always lower than that of the raw PCPP. The elongation at break increases with the incorporation of clay nanofillers and reaches a maximum value (19.64%) with a clay content of 5 wt% compared with that of the raw PCPP. This can be explained by the presence of PP-g-MA as compatibilizer. The low molecular weight of PP-g-MA gives plasticization effect at the interface causing increase in elongation at break for nanocomposites. In addition, alignment of the filler during flow and some plasticizing action due to the presence of the long chain organic modifier in the clay particle might have contributed to an increase in elongation at break. 53
On the other hand, the greater interaction between filler and polymer matrix should be responsible also for the lower elongation at break that was showed for all nanocomposites tested. In fact, this greater interaction probably leads to a lower polymeric chain mobility making the material more rigid. 54
Thermal analysis of PCPP/clay nanocomposites
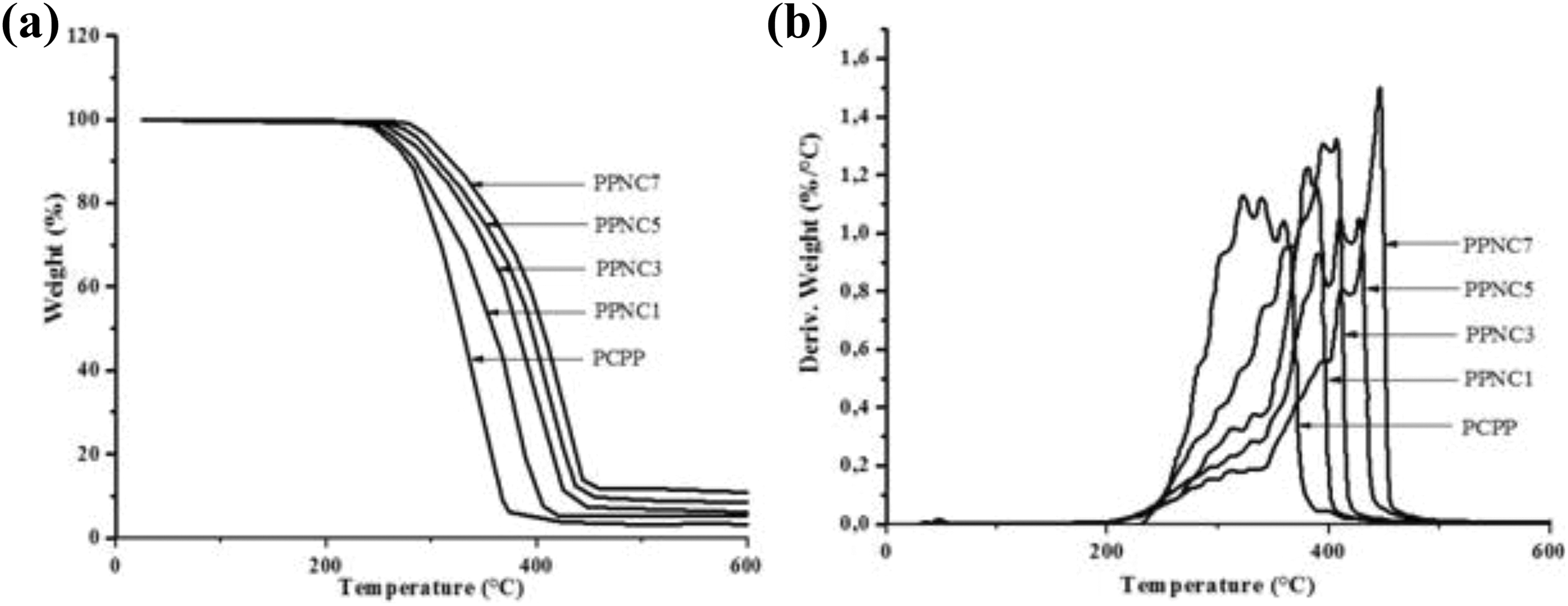
Thermal gravimetric analyses were carried to investigate the behavior of PCPP/clay nanocomposites during heating. Thermal tests of pure PCPP and PCPP/Tunisian clay nanocomposites were carried out using thermogravimetric analysis (TGA) and differential thermal analysis (DTA) within the temperature range 0–600°C and the results are shown in Figure 7.
(a) TGA and (b) DTA curves of raw PCPP and related nanocomposites. TGA: thermogravimetric analysis; DTA: differential thermal analysis.
As observed in the thermograms (Figure 7(a)), the weights of the samples decreased with increasing temperature, indicating the thermal destruction of the samples. It is clear that all weight loss temperatures for samples containing clay particles are higher than that of raw PCPP. Indeed, the thermal degradation temperature of PPNC7 is 280°C, whereas it is 250°C for PPNC1 and only 245°C for the neat PCPP. This improvement in thermal stability can be justified by the effect of these particles on the nucleation stage. 55 Similar behavior, regarding the improvement of the thermal stability of PP nanocomposites, has been reported in the literature and was attributed to the shielding effect of nanoparticles inhibiting the elimination of the formed volatile by-products. 56
It can be evident that composite systems containing layered fillers like clays are known to create tortuous paths that have the effect of increasing the barrier properties and therefore hinder the mobility of volatile decomposition products. Therefore, the formation of such paths that attributed to better dispersion of filler in matrix has the direct effect of slowing the matrix degradation by the oxidation processes. This observation is compatible with the study of Costache et al, 57 which had reported that the addition of clay particles increases the thermal degradation temperature of poly methyl methacrylate/MMT composites. These authors confirmed that this observation was due to the high slenderness ratio of clay filler and creation of barrier mechanism.
Figure 7(b) showed that the addition of clay particles reduces the intensity of the mass loss rate during the thermal degradation. Moreover, when the polymer containing clay burns, coal layer is formed on the external surface of the plastic by clay. This allows in insulation of the sublayer and prevents the composite from burning resulting in the fewer released heat. In general, due to the high degradation temperature, the clay particles improve the thermal stability of the composites and reduce the amount of mass loss rate during combustion. 58
Conclusion
Organically modified Tunisian clay was prepared by treating purified Tunisian clay with TOMA bromide through solid state method. Then, composite systems were prepared by melt blending of PCPP with various amounts of organo-modified Tunisian clays (1, 3, 5, 7 mass%). The mineralogical, morphological, and thermomechanical properties of the PCPP/clay nanocomposites have been carefully investigated.
XRD shows no crystallographic change of the PCPP matrix in the composite. TEM analysis revealed that good dispersion of fillers was achieved inside the post-consumer polymer matrix. It became apparent that PP-g-MA intercalated between silicate layers of Tunisian clay and therefore improved the dispersibility of the clay particle in the PPNC.
Mechanical analysis of PCPP/Tunisian clay nanocomposites also showed substantial improvements in tensile strength, Young modulus, and elongation at break by incorporation of 5 wt% clay loading followed by a decrease. The increased tensile modulus is attributed to the increased stiffness and brittleness of composites by the addition of clay. On the other hand, the high tensile strength is related to an intercalation phenomenon of clay layers by PP-g-MA. In addition, the increased elongation at break can be attributed to the low molecular weight of PP-g-MA, which gives plasticization effect at the interface.
A significant enhancement in thermal stability was observed by the incorporation of Tunisian clay. This improvement of thermal stability can be explained by the occurrence of a diffusion effect which limits the emission of the gaseous degradation products.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
