Abstract
The biggest issues in the preparation of carbon nanotube (CNT)-reinforced composites reside in efficient dispersion of CNT into polymer matrix. In our work, a simple acid treatment method was adopted to obtain carboxylic functionalized multiwalled nanotubes (MWNTs), thus improving the dispersion of CNT and interaction between particles and polymer matrix. X-ray photoelectron spectroscopy and Fourier transform infrared showed that the carboxylic groups were introduced onto the surface of MWNTs. The Raman spectroscopy showed that the amorphous carbon materials and impurities decreased after acid treatment. The electrical and dielectric properties of the Polyamide-11 (PA11)-based composites filled with pristine multiwalled nanotubes (p-MWNTs) and carboxylic functionalized multiwalled nanotubes (c-MWNTs) were investigated. The biggest dielectric constant of PA11/c-MWNTs composites, which was about twice as high as that of PA11/p-MWNTs composites at room temperature and 103 Hz (345–610), was obtained, accompanied by a lower dielectric loss. The formation of abundant microcapacitors and improved interfacial polarization effect by improving the dispersion of MWNTs in the composites via carboxylic functionalization was the main reason for the excellent dielectric properties of PA11/c-MWNTs composites.
Introduction
In recent years, dielectric materials have attracted a great attention on account of their potential applications such as electric energy storage devices, dielectric actuators, and sensors. to name a few. 1 –5 The feature of dielectric materials is to transmit, store, and record the effects of electricity in the form of electric polarization, that is, electric field-induced separation and alignment of electric charges. As the real application of dielectric materials, dielectric constant, and dielectric loss are the two main points focused by the researchers, for example, the large dielectric constant and low dielectric loss are demanded when these materials are employed as high charge-storage capacitors, “smart skins” for drag reduction and other functional materials. Compared with the traditional inorganic dielectric materials such as ceramic, the polymer-based dielectric materials could be potential replacement due to their unique combination of lightness, durability, high flexibility and processability. Recently, a universal strategy to prepare polymer-based dielectric materials is introducing electric conductive fillers, and among which, carbon nanotubes (CNTs) is the widely used conductive filler due to their large aspect ratio, high electrical conductivity, and unique mechanical properties. 6 –10 Wu et al. 8 prepared multiwalled nanotubes/poly (vinylidene fluoride (MWNTs/PVDF) composites via the melt-blending method, and found that when the MWNTs content was 12 vol%, a giant dielectric constant (3800) could be obtained, which was three orders of magnitude higher than that of pure PVDF. Zhang et al. 11 reported that the dielectric constant for the poly (vinylidenefluoride-trifluoride-trifluoroethylene-chlorofluoroethylene) (P(VDF-TrFE-CFE))/CNTs nanocomposites with only 1.0 wt% CNTs was improved by 30% compared with pure P(VDF-TrFE-CFE).
It is known that the CNTs have a large surface area and are easy to aggregate in the composites. A considerable research effort has been put into improving the dispersion of CNTs in composites from the view of surface chemistry of CNTs via a non-covalent method (surfactants adsorption) 12 or covalent method (chemical functionalization). 13 The remarkable enhancement of dielectric properties of composites would be expected when a homogeneous dispersion of CNTs into polymer matrix was obtained to ensure strong interfacial interaction between CNTs and the polymer, thus opening the way to charge transfer process, and improving the electrical and dielectric properties of the composites. Dang et al. 14 prepared MWNTs/PVDF nanocomposites by modifying the MWNTs with 3,4,5-trifluorobromobenzene. The giant dielectric constant (∼8000) of composites derived from the good dispersion of MWNTs and strong interaction between 3,4,5-trifluorobromobenzene-MWNTs and PVDF because of fluoride groups existing on the surface of modified particles. Han et al. 15 studied the surface nature of MWNTs effects on the properties of composites based on cyanate ester (CE) and found that after Fenton oxidation treatment, the surface functional groups of MWNTs could improve the interfacial adhesion between MWNTs and CE matrix, and promote dispersion of MWNTs in CE resin, which was beneficial to form more microcapacitor networks and thus enhanced the dielectric and thermal properties of composites. Zhou et al. 16 reported a comparative study of the dielectric properties of PVDF-based nanocomposites with pristine MWNTs and surface-modified MWNTs with core/shell structure as fillers. Compared with the MWNTs/PVDF composites, the surface-modified MWNTs/PVDF composites exhibited lower dielectric loss and higher dielectric permittivity. The conductive/nonconductive core/shell structure of the MEB filler was the main cause of the improved dielectric properties. As mentioned above, it can be seen that the chemical functionalization of fillers is an effective method to improve the dielectric properties of polymer-based composites.
Polyamide-11 (PA11) is an important commercial polyamide with high dielectric constant and very low dielectric loss even under high temperature and low frequency and has been widely used as various industrial fields such as automotive and offshore applications. 17 The internal amide groups of PA11 make up the intrinsic dipole moment, which contributes to the dielectric behaviors of materials. Our previous work showed that quenched MWNTs/PA11 composites obtained excellent dielectric properties, and the highest dielectric constant could be 350 at 103 Hz when the MWNTs content was 1.21 vol%, 18 which was believed to be resulted from the formation of nano-capacitors and between PA11 and MWCNTs of the interfacial polarization effect. For the purpose of increasing the dielectric constant of PA11-based composites further, improving the dispersion state of MWNTS and interfacial interaction between MWNTs and PA11 has come into our sight. In this work, the carboxylic functionalized MWNTs were obtained by a simple acid treatment and then introduced into PA11 by melt-blending method. The effect of MWNTs carboxylic functionalization on the dielectric properties and electrical conductivity of PA11/carboxylic MWNTs composites were systematically investigated. This work proved a convenient approach to improve the dielectric properties by promoting the dispersion of MWNTs in composites.
Experimental
Materials and sample preparation
Commercially available MWCNTs (NC7000) grown by chemical vapor deposition was purchased from Nanocyl S.A. (Sambreville, Belgium). According to the manufacturer, the MWNTs have an average diameter of 10 nm, length of 1.5 μm, and specific surface area of 250–300 m2 g−1. 19 The PA11 pellets (BESN-P40TL) with a density of 1.05 g cm−3 was purchased from Arkema Group (France). The concentrated sulfuric acid and nitric acid were purchased from Tianjin Chemical Co. Ltd., China.
The carboxylic functionalized MWNTs were functionalized following the route reported by Qin et al. 20 1 g of the pristine MWNTs was suspended in 100 mL mixture of concentrated sulfuric acid and concentrated nitric acid (3:1, v/v) and sonicated for 8 h at 20°C. Then, the MWNTs were filtrated and washed with deionized water until the pH value of the water is 6–7. After being dried at 80°C in a vacuum oven for 24 h, the carboxylic functionalized MWNTs were obtained. In this article, we denoted pristine MWNTs and carboxylic functionalized MWNTs as p-MWNTs and c-MWNTs, respectively.
All PA11 pellets were dried in vacuum oven at 80°C for 24 h before use. A range of PA11/MWNTs composites containing different content of pristine MWNTs and carboxylic functionalized MWNTs were prepared via melt blending using a torque rheometer (XSS-300, Shanghai, China) at 210°C for 15 min with a screw speed of 60 rpm. Disk-shape samples with 10 mm in diameter and 1.5 mm in thickness were obtained by compression molding under the pressure of 10 MPa at 210°C for 5 min, and then cold pressed to room temperature under the same pressure for alternating current (AC) conductivity and dielectric properties measurement.
Measurement and characterization
The surface chemical structure of p-MWNTs and resulted c-MWCNTs were characterized by X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FTIR). XPS characterization was conducted on AXIS Ultra DLD system (Japan) using Al Kα radiation (93.9 eV photons). The X-ray source was operated at 14 KV and 20 mA. The MWNTs were grounded into fine powder before being mounted on standard sample holders by means of double-sided adhesive tapes. FTIR testing was confirmed using a Nicolet 6700 FTIR spectrometer (Nicolet Instrument Company, Madison, WI, USA) in the transmission mode at the wavelength range of 400–4000 cm−1 with the resolution of 4 cm−1. The MWNTs powder was mixed with KBr powders and pressed into a disk for IR measurement. Raman spectra at the wavelength range of 800–2000 cm−1 were obtained using a LabRAM HR spectrophotometer (Horiba Jobin Yvon LabRAM HR, France), coupled to an optical microscopy that focuses the incident radiation down to nearly 1 μm spot. X-ray diffraction (XRD) spectra of the MWNTs were obtained with DX-1000 X-ray diffractometer using nickel-filtered Cu Kα radiation (40 KV, 40 mA). The measurements were performed within a 2θ range of 5°–45° at scan speed of 1° min−1. The morphology of the bulk fillers and composites were visually characterized via a high-resolution transmission electron microscopy (HR-TEM) (Tecnai G2 F20, FEI Company, Hillsboro, TX, USA) with operating voltage of 200 kV and a scanning electron microscopy (SEM) instrument (INSPECT F, FEI, USA) at an accelerating voltage of 20 kV, respectively. The sample surfaces of composite for SEM examination were prepared by a cryogenic fracture in liquid nitrogen and coated with a thin of layer of gold. Prior to the AC conductivity and dielectric properties measurement, silver paste was painted on both sides of the samples to provide a good contact with filler network within the samples. The measurements were carried out in the frequency range of 103–107 Hz and room temperature, using an Agilent 4294A precision LCR meter (Concept 50, Germany).
Results and discussion
Chemical functionalization of MWNTs and its dispersion in composites
Figure 1(a) and (b) is the full XPS and narrow scan spectra of O 1 s of p-MWNTs and c-MWNTs, respectively, which could indicate the elements and their concentration on the surface of MWNTs. As shown in Figure 1(a), the intensity of the O 1 s (555 eV) peak of the c-MWNTs dramatically increased compared to that of the p-MWNTs. As shown in Figure 1(b), the area of the peak of c-MWNTs was larger than that of p-MWNTs, and it was calculated that the oxygen element concentration was1.47 at% for the p-MWNTs and 8.53 at% for the c-MWNTs. The oxygen element concentration of c-MWNTs increased obviously, giving a clear evidence of the H2SO4/HNO3 treatment on the MWNTs.
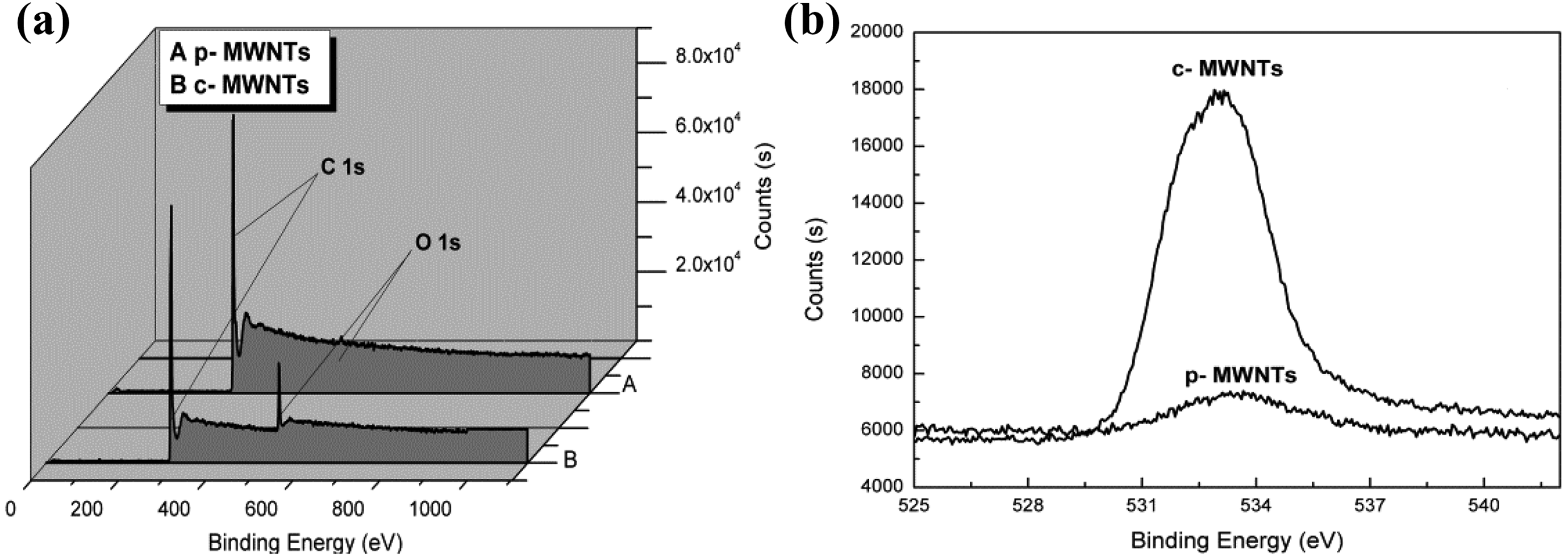
Full XPS (a) and narrow scan spectroscopy of O 1 s (b) for p-MWNTs and c-MWNTs. XPS: X-ray photoelectron spectroscopy; p-MWNT: pristine multiwalled nanotube; C-MWNT: carboxylic multiwalled nanotube.
The FTIR spectrum of c-MWCNTs (Figure 2(a)) showed a relatively strong C=O stretching at 1727 cm−1 corresponding to the carboxylic acid groups as a result of the acid treatment process. The peaks around 1340 and 1450 cm−1 of c-MWNTs represented C–O stretching vibration band and O–H bending vibration band, which further demonstrated that the MWNTs were modified successfully by chemical reaction.
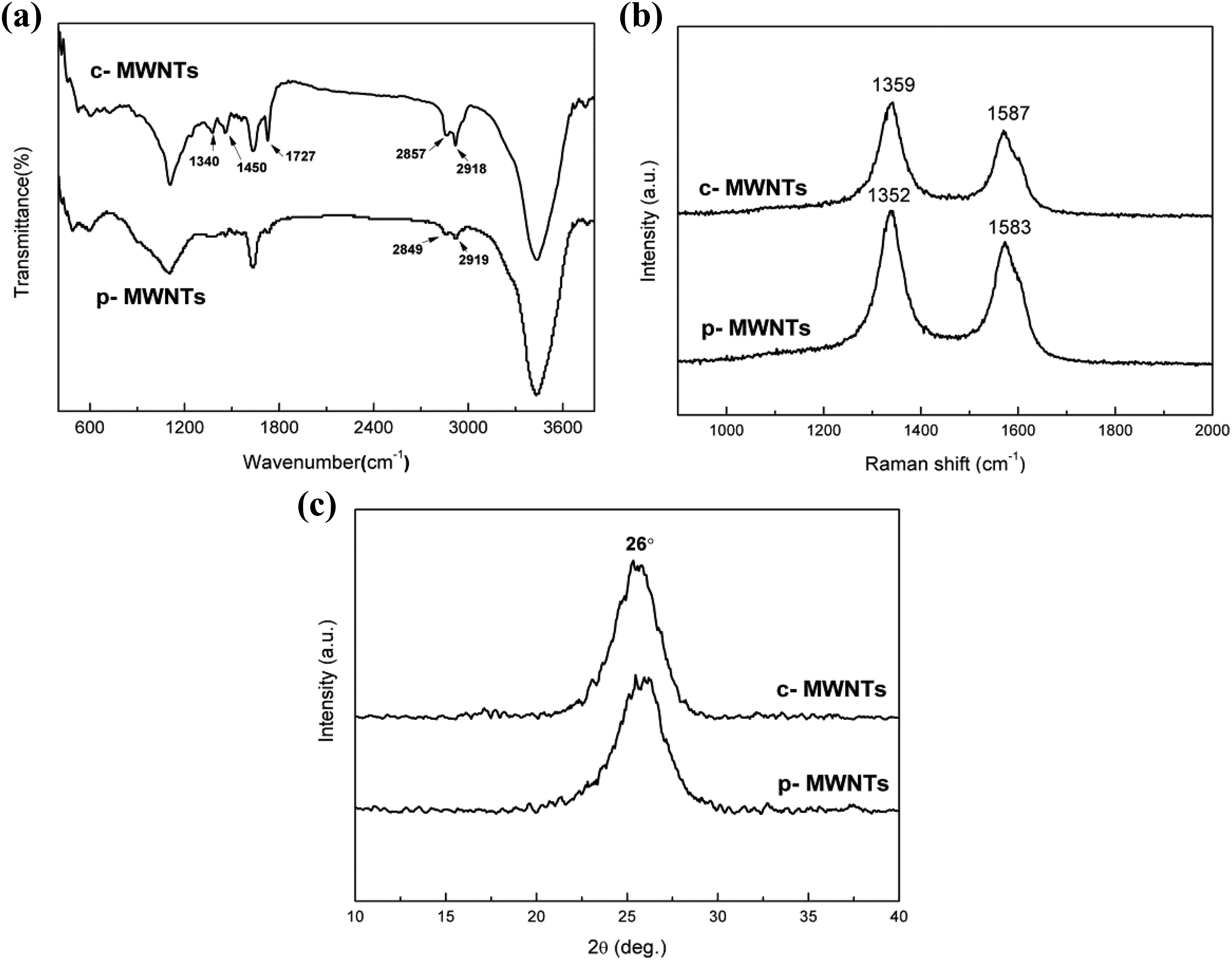
FTIR spectroscopy (a), Raman spectroscopy (b), and XRD pattern (c) of p-MWNTs and c-MWNTs.
The structural features of MWNTs before and after modification were detected by Raman spectroscopy, as shown in Figure 2(b). The peak near 1350 cm−1 assigned to the disorder-induced D band, may be attributed to lattice distortions and/or the presence of structure defects. The peak at 1580 cm−1, called G band, indicates the graphite crystal structure in carbon materials. As shown in the Raman spectrum, the D band around 1350 cm−1 was more intense than the G band around 1580 cm−1 for both MWNTs. Comparing the relative intensity ratio of D band and G band (ID/IG) of the samples, which were 1.23 for c-MWNTs and 1.48 for p-MWNTs, revealed that the amorphous carbon materials and impurities of the MWNTs decreased during the carboxylic functionalization. Moreover, the two-peak position of c-MWNTs shifted to higher wave numbers, which might derive from the electro-withdrawing group (carboxylic group). Furthermore, as shown in Figure 2(c), XRD pattern indicated the persistence of the main reflection of both MWNTs at angle of 2θ ≈ 26°, which was assigned to the (002) reflection of the host lattice. 21 From the results of FTIR, XPS, Raman, and XRD analyses, it was clear that the carboxylic groups (–COOH) were attached onto the surface of the MWNTs and the structure of cylindrical concentric carbon layers had not been destroyed in this experiment.
Figure 3(a) and (b) shows TEM images of the pristine MWNTs and carboxylic functionalized MWNTs, respectively. The amorphous carbon materials in c-MWNTs were less than that of p-MWNTs, and also the length of the MWNTs. Figure 3(c) and (d) shows the SEM images of the composites containing MWCNTs before and after modification, respectively. For the p-MWNTs/PA11 composites, a certain amount of MWNTs aggregates were observed (in the black circle areas) and the MWNTs were absent in some areas of the composites within our field of vision. It illustrated that the p-MWNTs were not homogeneously dispersed in the polymer matrix. Notably, for the c-MWNTs/PA11 composites, the c-MWNTs exhibited an improved dispersion and distribution state in the polymer matrix and no obvious MWNTs aggregates were observed. Therefore, we believe that the presence of carboxylic groups (–COOH) allowed the MWNTs to be more easily dispersed into polymers when preparing polymer/MWNTs composite materials due to the strong interaction between the carboxylic group on the surface of c-MWNTs and the polar amide groups of the PA11. The dispersed MWNTs could afford excellent electrical properties for the resulted composites.

TEM images of p-MWNTs (a) and c-MWNTs (b); SEM images of fractured surface of p-MWNTs/PA11 composites (c) and c-MWNTs/PA11composites (d) with f MWNTs = 0.0121. TEM: transmission electron microscopy. p-MWNT: pristine multiwalled nanotube; C-MWNT: carboxylic multiwalled nanotube; SEM: scanning electron microscopy; PA11: Polyamide-11.
Electrical properties of PA11/MWCNTs composites
Figure 4(a) and (b) shows the frequency dependence of AC conductivity of the p-MWNTs/PA11 composites and c-MWNTs/PA11 composites, respectively. As expected, the electrical conductivity of composites with p-MWNTs or c-MWNTs increased with the increase of MWNTs content. Furthermore, with increasing the MWNTs concentration, a frequency-independent plateau up to a critical frequency (ω c) was observed and the ω c extended to a higher frequency. The onset of frequency-independent behavior of composites was considered as insulator-semiconductor transition, which indicated the formation of conductive paths in the composite. Therefore, 0.61 vol% and 0.91 vol% were observed to be the frequency-independent plateau for p-MWNTs/PA11 composites and c-MWNTs/PA11 composites, respectively. Moreover, the conductivity of p-MWNTs/PA11 was higher than that of c-MWNTs/PA11 composites with the same MWNTs concentration. When the MWNTs concentration was 1.21 vol%, the conductivity of p-MWNTs/PA11 composite was ∼10−3 S m−1, nearly 10 times of the c-MWNTs/PA11 composite (10−4 S m−1). In fact, besides the high dielectric constant, the desired composites serving as dielectrics should also have low conductivity. Furthermore, in both the composites, above the percolation threshold, the conductivity values were found to further increase with MWNT content, indicating the formation of multiple conducting paths. It was observed that when the MWNTs are 1.82 vol% and 2.44 vol%, the AC conductivities were almost invariable over a wide range of frequency, indicating the conquering role of Ohmic conduction and stable conductive network in the composites.
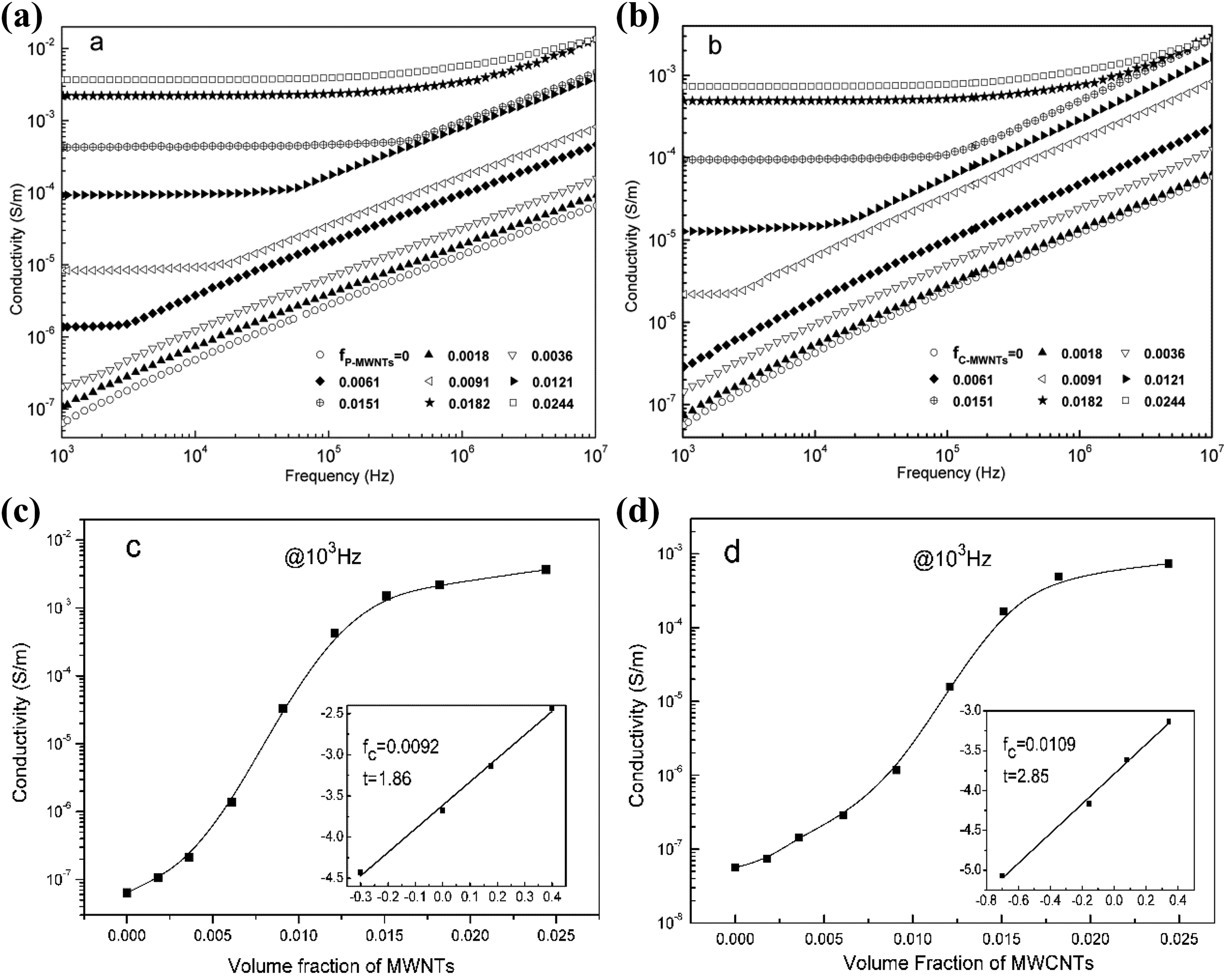
Frequency dependence of the AC conductivity of (a) p-MWNTs/PA11 composites and (b) c-MWNTs/PA11 composites with different MWNT volume concentration; dependence of the AC conductivity of (c) p-MWNTs/PA11 composites and (d) c-MWNTs/PA11 composites on the MWNTs volume concentration; the inset shows a log–log plot of the conductivity as a function of fMWNTs − fc according to equation (1).
Figure 4(c) and (d) displays the evolution of the AC conductivity of p-MWNTs/PA11 and c-MWNTs/PA11 composites, respectively, as a function of MWNTs volume concentration, measured at room temperature and 103 Hz. It was observed that the conductivity of both composites increased with increasing the MWNTs volume concentration, and exhibited a typical insulator–conductor transition near the percolation threshold. The electrical conductivity can be further analyzed with the critical MWNTs concentration fc by the scaling law near the conductor–insulator transition as follows: 22,23
where σeff is the effective conductivity of the composites, σi is the conductivity of the insulating PA11 polymer, fMWNTs is the MWNTs volume fraction, fc is the percolation threshold, and t is the conductivity exponent. The percolation thresholds fc of 0.92 vol% and 1.09 vol%, and the value t of 1.86 and 2.85, for p-MWNTs/PA11 and c-MWNTs/PA11 composites, respectively, were then obtained (inset of Figure 4(c) and (d)). The larger percolation threshold of c-MWNTs/PA11 composites suggested the difficult formation of the conductive network in composite with modified MWNT, which should be attributed to the reduced length of MWNTs, reduced intrinsic electrical properties of MWNTs by the acid treatment, and the interaction enhancement between MWCNTs and PA11. Besides, the larger conductivity exponent t of c-MWNTs/PA11 composites also resulted from the acid chemical functionalization, which can be explained by the Swiss-Chess model. 24
Dielectric properties of PA11/MWCNTs composites
Figure 5 shows the dielectric properties of p-MWNTs/PA11 and c-MWNTs/PA11composites as function of MWCNTs concentration at room temperature and 103 Hz. For both kinds of composites, the dielectric constant increased distinctly with increasing MWNTs content at the MWNTs contents near the percolation threshold. For the p-MWNTs/PA11 composites, the dielectric constant increased from ∼7 of pure PA11 to ∼345 of the composites with 1.21 vol%. For the c-MWNTs/PA11composites, the dielectric constant increased from ∼7 of pure PA11 to ∼610 of the composites with 1.51 vol%. However, the dielectric constant decreased sharply with further increasing MWNTs contents because of the formation of conductive network of MWCNTs nanoparticles. Moreover, the dielectric loss increased with increasing MWNTs contents, especially when MWNTs content was higher than percolation threshold, the dielectric loss increased sharply. It was attributed to the MWCNT materials could produce electrical current under electric field which was resulted in part of electrical energy transforming into the thermal energy. For example, the dielectric loss increased from 2.1 of the composites with 1.21 vol% to 12.4 of the composites with 2.44 vol% for p-MWNTs/PA11 composite and from 1.1 of the composites with 1.21 vol% to 8.9 of the composites with 2.44 vol% for c-MWNTs/PA11 composites, respectively. With such high dielectric loss, the composites became semi-conductive and could not be used as dielectrics due to the large leakage. It was worth noting three obvious differences in dielectric properties comparing two kinds of MWNTs/PA11 composites. The first one was that with the same MWNTs contents, the dielectric constants of c-MWNTs/PA11 composites were higher than those of p-MWNTs/PA11 composites. For example, when the MWNTs content was 1.21 vol%, the dielectric constants were 345 and 410 for the p-MWNTs/PA11 composites and c-MWNTs/PA11 composites, respectively. The second one was that the maximum dielectric constant of c-MWNTs/PA11 composites was 610, higher than the maximum dielectric constant of p-MWNTs/PA11 composites (345) which can be attributed to the larger MWCNTs content and the carboxylic groups on the surface of the MWCNTs to attain more microcapacitors in the c-MWNTs/PA11 composites. The third one was that although the dielectric loss also increases with increasing MWNTs contents for c-MWNTs/PA11 composites, it was lower than that of the p-MWNTs/PA11 composites with the same MWNTs content. The results suggested that a homogeneous dispersion of c-MWNTs in the PA11 matrix enhanced the dielectric constant and reduced the dielectric loss of composites.
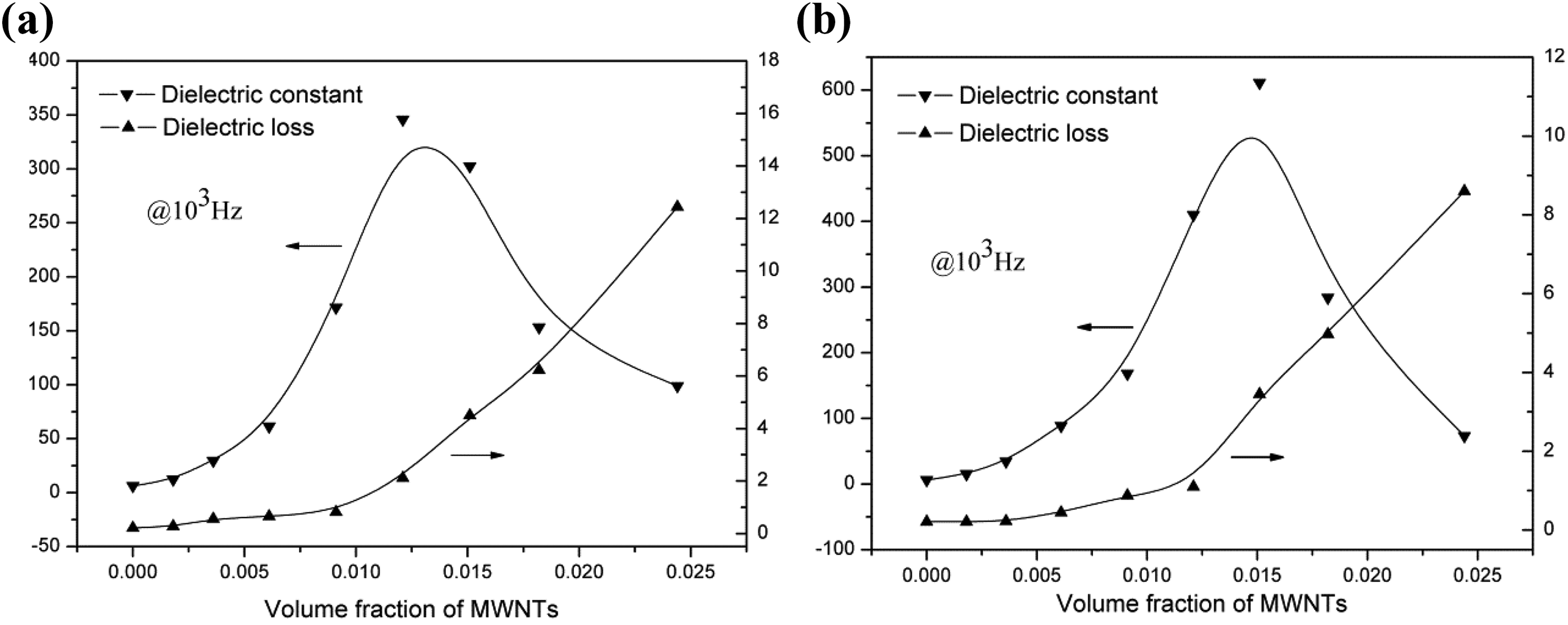
Dielectric constant and dielectric loss of (a) p-MWNTs/PA11 composites and (b) c-MWNTs/PA11 composites at 103 Hz. p-MWNT: pristine multiwalled nanotube; C-MWNT: carboxylic multiwalled nanotube; PA11: Polyamide-11.
The frequency dependence of dielectric properties of a dielectric material is also quite important for its real application. Therefore, the frequency dependence of dielectric properties of the p-MWNTs/PA11 composites and c-MWNTs/PA11 composites were investigated at room temperature and shown in Figure 6.
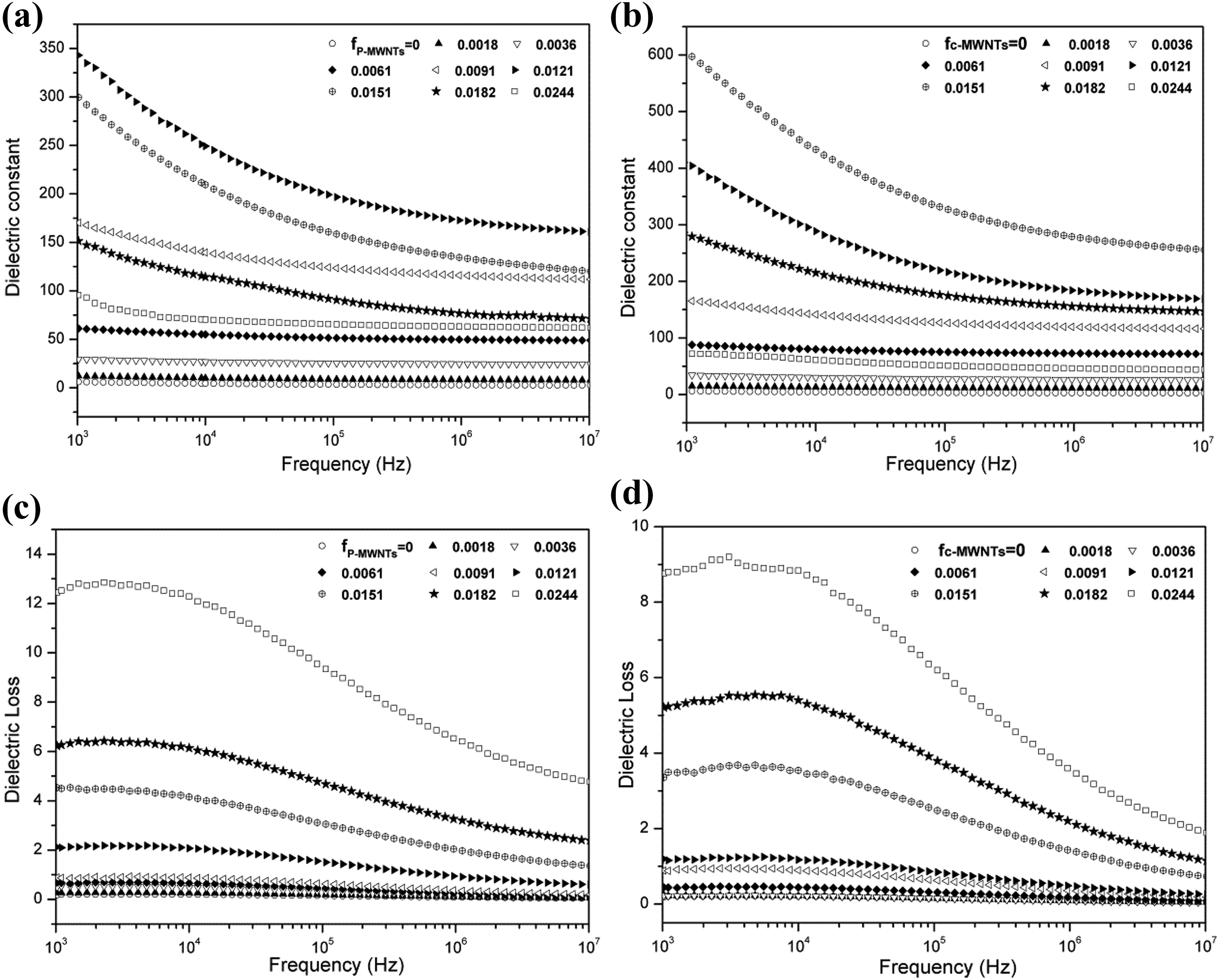
Frequency dependence of the (a, b) dielectric constants and (c, d) dielectric loss of p-MWNTs/PA11 composites and c-MWNTs/PA11 composites. p-MWNT: pristine multiwalled nanotube; c-MWNT: carboxylic multiwalled nanotube; PA11: Polyamide-11.
As shown in Figure 6(a) and (b), for both composites systems, the dielectric constant demonstrated a slight correlation to the frequency when the MWCNT concentration was far lower and larger than the percolation threshold. The dielectric constant decreased dramatically from low frequency to high frequency for the composite with particle content near the percolation threshold. Furthermore, in the whole frequency region, the dielectric constants increased firstly but then turned decreasing with increasing MWNTs contents due to the formation of the conductive network above the percolation threshold, and a maximum dielectric constant appeared at certain MWNTs contents which were 1.21 vol% and 1.51 vol% for the p-MWNTs/PA11 composites and c-MWNTs/PA11 composites, respectively. Figure 6(c) and (d) presents the frequency dependence of dielectric loss of composites with different MWCNTs contents. For both composites systems, the dielectric loss increased with increasing MWNTs contents and showed a slight correlation to the frequency with low MWNTs contents. But when the MWNTs contents were relatively high, the dielectric loss showed a strong frequency dependence. By comparison, the dielectric loss of c-MWNTs/PA11 composites was lower than that of p-MWNTs/PA11 composites in the whole frequency region especially at high MWNTs contents, because the carboxylic groups on the surface of MWNTs prevented the MWNTs from direct connecting with each other and the formation of conductive paths would be blocked effectively.
Based on the discussion above, dielectric constant of MWNTs/PA11 composites was improved and dielectric loss of composite was reduced when particle was dispersed into the matrix homogeneously by surface modification of MWNTs. With regard to polymer composites, the large dielectric constant can be comprehended by two mechanisms. The first one is nano-capacitor principle,14 which proposes that many parallel or serial nano-capacitors connect with one another. The MWNTs distribute in the insulated matrix can be considered as nano-electrodes and the thin layer of polymer between MWNTs acts as the nano-dielectrics. The second one is the interfacial polarization effect (Maxwell-Wager-Sillars effect), 25 which is contributed to accumulation of many nomadic charge carriers at the interfaces between MWNTs and polymer. The interfacial polarization effect dominates the frequency dependence of dielectric constant in the low-frequency range, while the nano-capacitor principle is frequency-independent, as reported in previous studies. 26 For both mechanisms, the dispersion and distribution state of MWNTs in the polymer matrix are key factors influencing the dielectric properties. Figure 3 reveals that the c-MWNTs exhibited an improved dispersion and distribution state in the composites compared with p-MWNTs, and increased the conduction percolation threshold as shown in Figure 4. On the one hand, when the percolation threshold increased, the number of the nano-capacitors increased, and the isolation distance between MWNTs decreased so that the average capacitance of the single nano-capacitor increased. Consequently, the dielectric constant could be enhanced remarkably. On the other hand, more MWNTs was needed to provide large domains for nomadic electrons when uniform dispersion of c-MWNTs was obtained in the PA11 matrix, and the electro-withdrawing carboxylic groups on the c-MWNTs reinforced the MWS effect which enhanced the dielectric constant accordingly.
As we know, for the conductor/polymer composites, the dielectric loss is mainly considered as the contribution of two effects: direct current conductance and interfacial polarization, as reported in previous works. 27,28 The loss of interfacial polarization dominates at high frequency, and the loss of direct current conductance dominates at low frequency. In our work, when the MWNTs contents were below the percolation threshold, because of the low direct current conductance, the dielectric loss was relatively low and almost displayed a frequency-independent behavior. However, as the MWNTs contents increased, the dielectric loss also gradually increased resulting from the progressive formation of conductive paths, which could give rise to current leakage. And when the MWNTs contents were much higher than the percolation, the direct connection of MWNTs network in the composites or the much smaller distance between MWNTs led to a high current leakage, resulted in the high dielectric loss and strong frequency dependence of dielectric loss at the low-frequency region. Because the interfacial polarization effect dominates at the high-frequency region, the contribution of direct current conductance to dielectric loss was suppressed, the dielectric loss decreased with increasing frequency.
Conclusions
We investigated the electrical and dielectric properties of the pristine and carboxylic functionalized MWNTs/PA11 composites. The XPS and FTIR spectroscopy showed that the carboxylic groups were successfully introduced onto the MWNTs during the carboxylic functionalization. The Raman spectroscopy and XRD indicated that amorphous carbon materials and impurities decreased while the structure of cylindrical concentric carbon layers was preserved. The SEM micrographs showed that the carboxylic functionalized MWNTs exhibited a better dispersion in the composites. The percolation threshold of carboxylic functionalized MWNTs/PA11 composites was slightly higher than that of pristine MWNTs/PA11. After carboxylic functionalization, not only the highest dielectric constant was enhanced significantly at room temperature, but also the dielectric loss decreased at room temperature (12.4–8.9 at 103 Hz). The enhanced dielectric properties of carboxylic functionalized MWNTs/PA11 composites can be understood by the formation of more nano-capacitors and improved interfacial polarization effect in the composites resulted from better dispersion of carboxylic functionalized MWNTs in the composites.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant nos. 51103087, 51421061).

