Abstract
In this work, the effects of inorganic phosphinate flame retardant of aluminum hypophosphite (AP) and organic phosphinate flame retardant of ethyl substituted phosphinates (AP-ethyl) on the thermal degradation, flame performance, and mechanical properties of polyamide 6 (PA6) were investigated. Scanning electron micrograph showed AP with the shape of bulk and the mean size of 8 μm while AP-ethyl with irregular shape and the mean size of 30 μm. Thermal analysis indicated that the thermal degradation behavior of flame-retardant PA6 was different from pure PA6. Moreover, the cone calorimeter test results revealed that peak heat release rate (PHRR) of PA6/AP (85/15) and PA6/AP-ethyl (85/15) decreased by 51% and 64%, respectively, compared with pure PA6. Furthermore, pure PA6 showed ductile stress–strain curve with the tensile strength of 54.8 MPa. However, PA6/AP and PA6/AP-ethyl displayed brittle stress–strain curve and their tensile strength decreased to 52.3 and 47.1 MPa, respectively. In addition, pure PA6 showed a glossy and tough fracture surface morphology. The rough fracture surface morphologies for PA6/AP and PA6/AP-ethyl were observed, and the interface of PA6/AP was more obscure than that of PA6/AP-ethyl. Consequently, the small particle size of AP had a more uniform dispersion in PA6 matrix.
Introduction
The improvement of fire resistance of polyamide is always an important research field due to their wide application in electrons, automotives, and so on. In recent years, phosphorus-containing flame retardant of metal phosphinates was proven to be effectively in polyamide active in both condensed and gaseous phases flame inhibition mechanism. 1 –3 Among the reported metal phosphinates, the inorganic phosphinate flame retardant of aluminum hypophosphite (AP) and organic phosphinate flame retardant of alkyl substituted phosphinates were mostly applied in polyamide 6 (PA6). Zhao et al. 4,5 investigated flame-retardant PA6 with AP and aluminum isobutylphosphinate (AP-isobutyl). Both the flame-retardant PA6 composites can achieve a V-0 rating in UL-94 test at 20 wt% concentration. And their results indicated that AP mainly exhibited condensed phase mechanism, whereas AP-isobutyl showed more gaseous phase flame inhibition mechanism. After a formulation (AP–AP-isobutyl) of 1:1 was incorporated in PA6 with the content of 15 wt%, good flame-retardant synergism was found.
However, their results were based on the flame retardant of AP-isobutyl; the wide application flame retardant of aluminum diethylphosphinate (AP-ethyl) had not been considered. Previous investigation showed that the activity of phosphorus depends on the molecular structure and their dispersion degree in polymer. 6 –8 Hence, in order to have a systematic description and understanding, in this work, AP versus AP-ethyl applied in PA6 was studied. The structures of AP and AP-ethyl were shown in Figure 1. This article not only comprehensively illuminated the effect of these materials but also gave insight into effects of the disperse degree of flame retardant with different particle sizes on the properties of applied polymer.
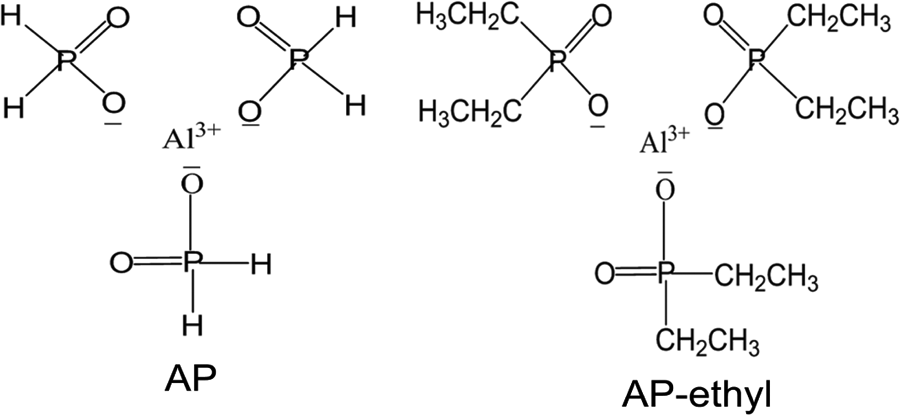
Chemical structures of AP and AP-ethyl. AP: aluminum hypophosphite.
Materials and characterization
Materials
Pure PA6 was obtained from UBE Industrials Ltd, Japan. AP was purchased from Hubei Tianhu Chemistry Co. Ltd, China. Aluminum diethylphosphinate (AP-ethyl) was purchased from Clarinet Co. Ltd, German. All the materials were dried in an oven at 100°C for 4 h before used. PA6 and AP or AP-ethyl were mixed by extrusion and then molded into plates for mechanical and flame tests. The temperature was in the range of 220–240°C.
Characterization
Scanning electron micrograph (SEM, KYKY-2008B; KYKY Technology Co., China) was performed. Vertical burning test was conducted based on UL-94, with the dimensions of 130 × 13 × 3.2 mm3. Cone calorimeter test (FFT, UK) was measured with a size of 100 × 100 × 6 mm3; 50 kW m−2 external heat was set. Thermogravimetric (TG) analysis (Q50; TA, USA) was carried out under a nitrogen (N2) atmosphere at a heating rate of 10°C min−1. Fourier transform infrared (FTIR) spectroscopy (Nicolet 6700 [Nicolet Instrument Co., USA]) was conducted via potassium bromide (KBr) pelletes. Differential scanning calorimeter (DSC) test (Q10; TA) was performed at a heating rate of 10°C min−1, and the sample of 5 mg was heated to 250°C to erase thermal history and then cooled to 40°C to record the crystallization curve. After that, the samples were heated again to 250°C to acquire the melting curve. The tensile tests were conducted by Instron WDW-100 tensile tester at a cross-head speed of 50 mm min−1 at room temperature.
Results and discussion
Morphology and size of the flame retardants
Morphology of AP and AP-ethyl was observed by means of SEM as shown in Figure 2(a) and (b), respectively. The AP-ethyl showed irregular shape, and the particles represented a lot of aggregates with the mean size of 30 μm. The AP exhibited the shape of bulk while the mean size was only 8 μm. Obviously, the mean particle size of AP was lower than that of AP-ethyl. The morphology and further the mean particle size of the flame retardants were important for their dispersion degree in the applied polymer, thus had a significant influence in the mechanical properties of polymer. Generally, excellent performances were obtained with better dispersion of additives in polymer matrix. 9,10
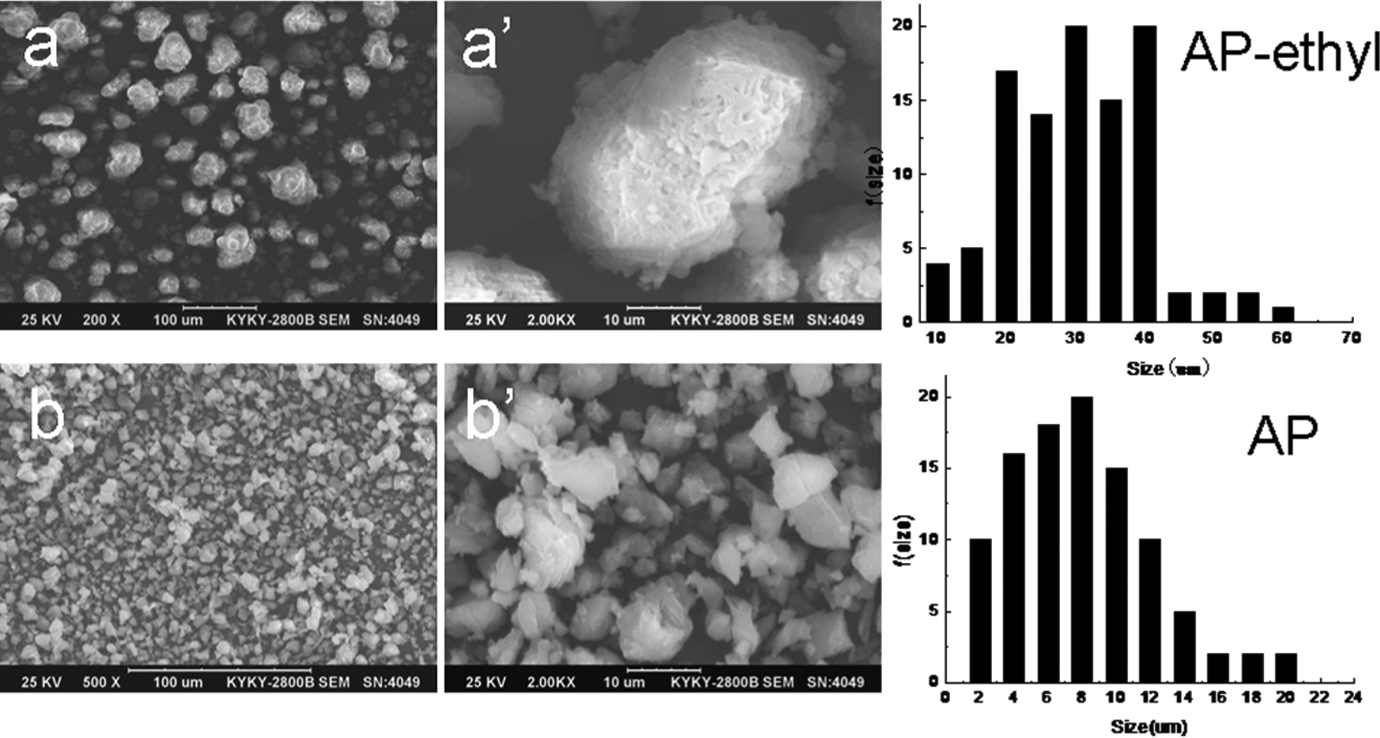
Morphology and f (size) of the flame retardants. SEMs of a, a′ (magnified image)—AP-ethyl; b, b′ (magnified image)—AP; and the corresponding size distribution. SEM: scanning electron micrograph; AP: aluminum hypophosphite.
Thermal stability of the flame retardants
It was found that TG curve of AP under a N2 atmosphere (Figure 3) showed two stages: initial decomposition temperature (defined as the temperature of 5 wt% weight loss, T− 5%) was 339.5°C. The final residue of AP was 75.9%. T− 5% of AP-ethyl was 402.1°C, which was higher than that of AP. AP-ethyl decomposed quickly from 400°C to 500°C, and the final residue was 32.3%, much lower than that of AP (75.9%). The above differences indicated that AP-ethyl could produce more volatile products than did AP. The residues were measured with FTIR and the results are shown in Figure 4. It can be seen that the peaks were similar for AP and AP-ethyl. And the peak appeared at 1126 cm − 1 was attributed to PO3 2 –/PO4 3–. This indicated that the residues of AP and AP-ethyl which played the role of condensed phase flame retardant were similar. Hence, the difference between AP and AP-ethyl was the gaseous phase flame ability.
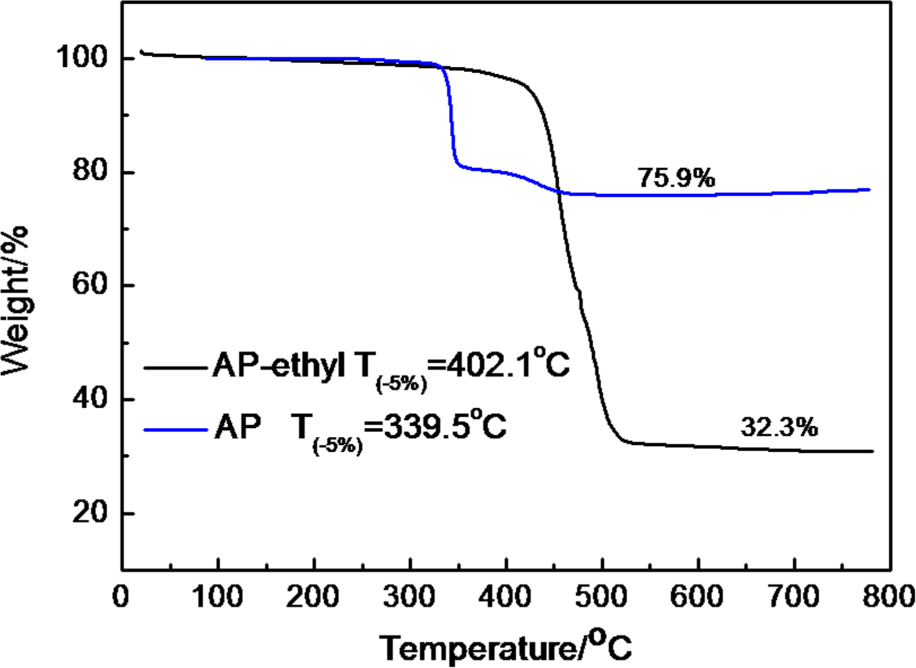
TG curves of AP and AP-ethyl under a N2 atmosphere. TG: thermogravimetric; AP: aluminum hypophosphite; N2: nitrogen.
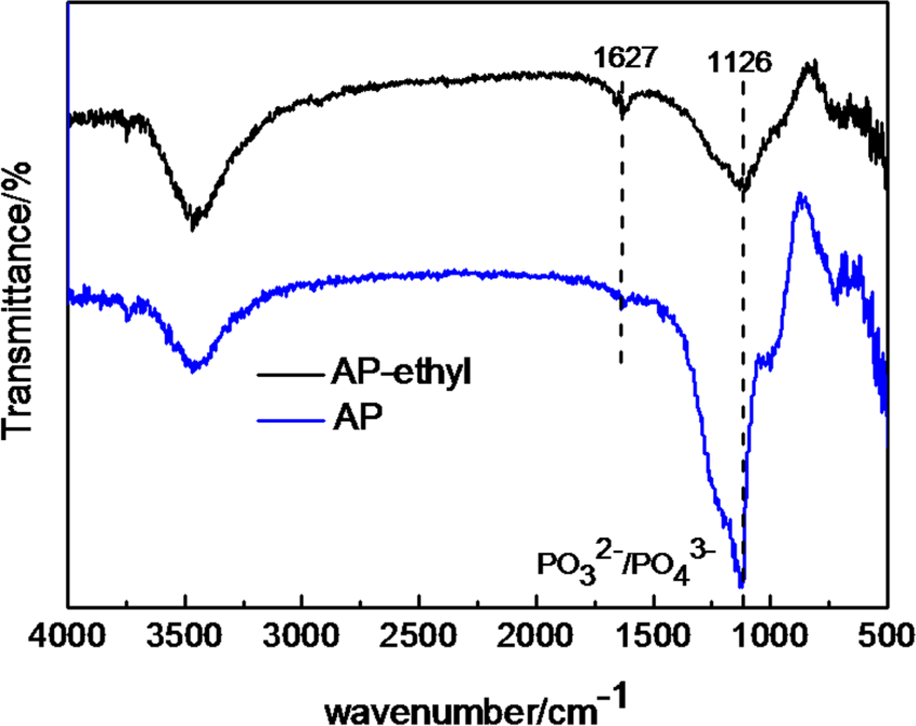
FTIR of the residue after TG test for AP and AP-ethyl. TG: thermogravimetric; AP: aluminum hypophosphite.
Thermal degradation behavior of the flame-retardant PA6
Figure 5 and Table 1 showed the TG results of PA6 and flame-retardant PA6. Compared with pure PA6, T− 5% of PA6/AP-ethyl and PA6/AP was decreased by 34.1°C and 62.0°C, respectively. The flame-retardant PA6 displayed lower thermal stability than that of pure PA6, which is probably attributed to the catalyze degradation of the flame retardants. And T− 5% of PA6/AP (348.2°C) was lower than that of PA6/AP-ethyl (376.1°C). This might be caused by the lower thermal stability of AP. However, the residue at the end of decomposition of PA6/AP (48.7%) was much higher than that of PA6/AP-ethyl (7.8%).
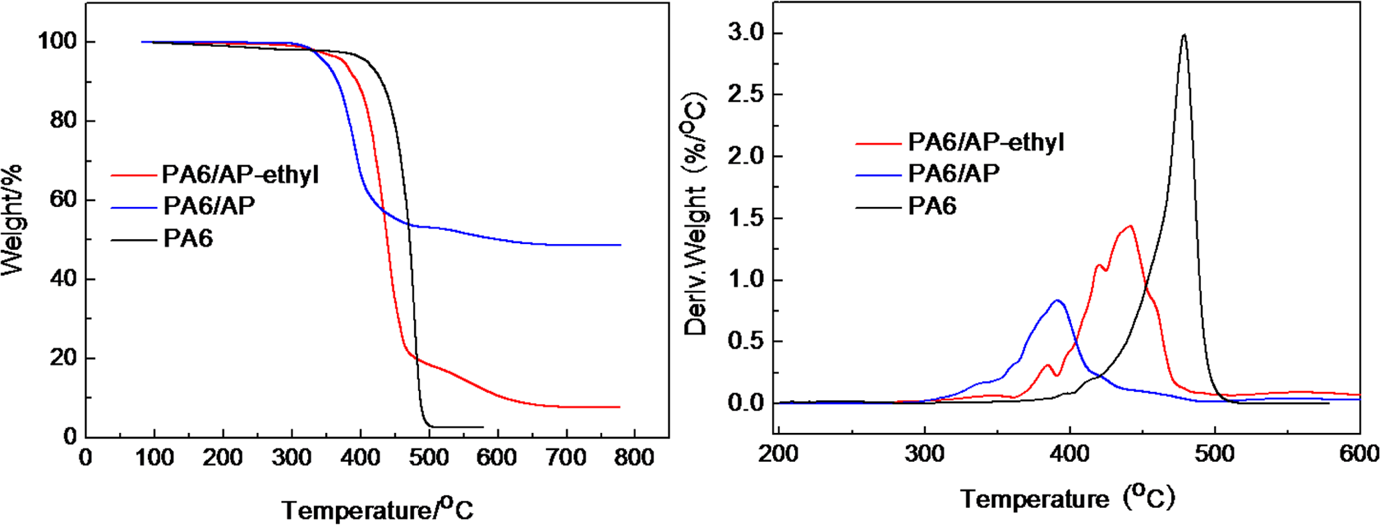
TG curves of PA6 and flame-retardant PA6 with AP and AP-ethyl under a N2 atmosphere. TG: thermogravimetric; PA6: polyamide 6; AP: aluminum hypophosphite; N2: nitrogen.
TG data of PA6 and flame-retardant PA6.

AP: aluminum hypophosphite; TG: thermogravimetric; PA6: polyamide 6; T− 5%: temperature of 5 wt% weight loss.
Forced combustion behavior of the flame-retardant PA6
The forced combustion of PA6 and PA6 composites was conducted by cone calorimeter test (CCT) and the results were shown in Figure 6 and Table 2. As shown in Figure 6(a) and Table 2, the HRR curve of PA6 showed a significant sharp PHRR at 658 kW m−2, while the PHRR value of PA6/AP and PA6/AP-ethyl was 323 and 237 kW m−2, respectively. Obviously, the PHRR value of flame-retardant PA6 showed a significant reduction. Moreover, the PHRR value of PA6/AP was lower than that of PA6/AP-ethyl. On the contrary, the time to ignition (TTI) of PA6/AP-ethyl showed a prolong of 28 s compared with pure PA6 (21 s). In addition, TTI value of PA6/AP was 17 s lower than that of PA6/AP-ethyl. This might be resulting from the lower thermal stability of AP. The mass loss rate of PA6/AP was the lowest (Figure 6(b)). Moreover, the total heat release (THR) curves were shown in Figure 6(c). Similar to PHRR results, the THR value of PA6/AP (114 MJ m−2) was lower than that of PA6/AP-ethyl (121 MJ m−2) and both were decreased in comparison to pure PA6 (155 MJ m−2). Together with PHRR result, the efficiency of AP was higher than AP-ethyl. However, the PA6/AP could only achieve a V-1 rating in the UL-94 test, while PA6/AP-ethyl can reach the V-0 rating at the same addition of 15 wt%.
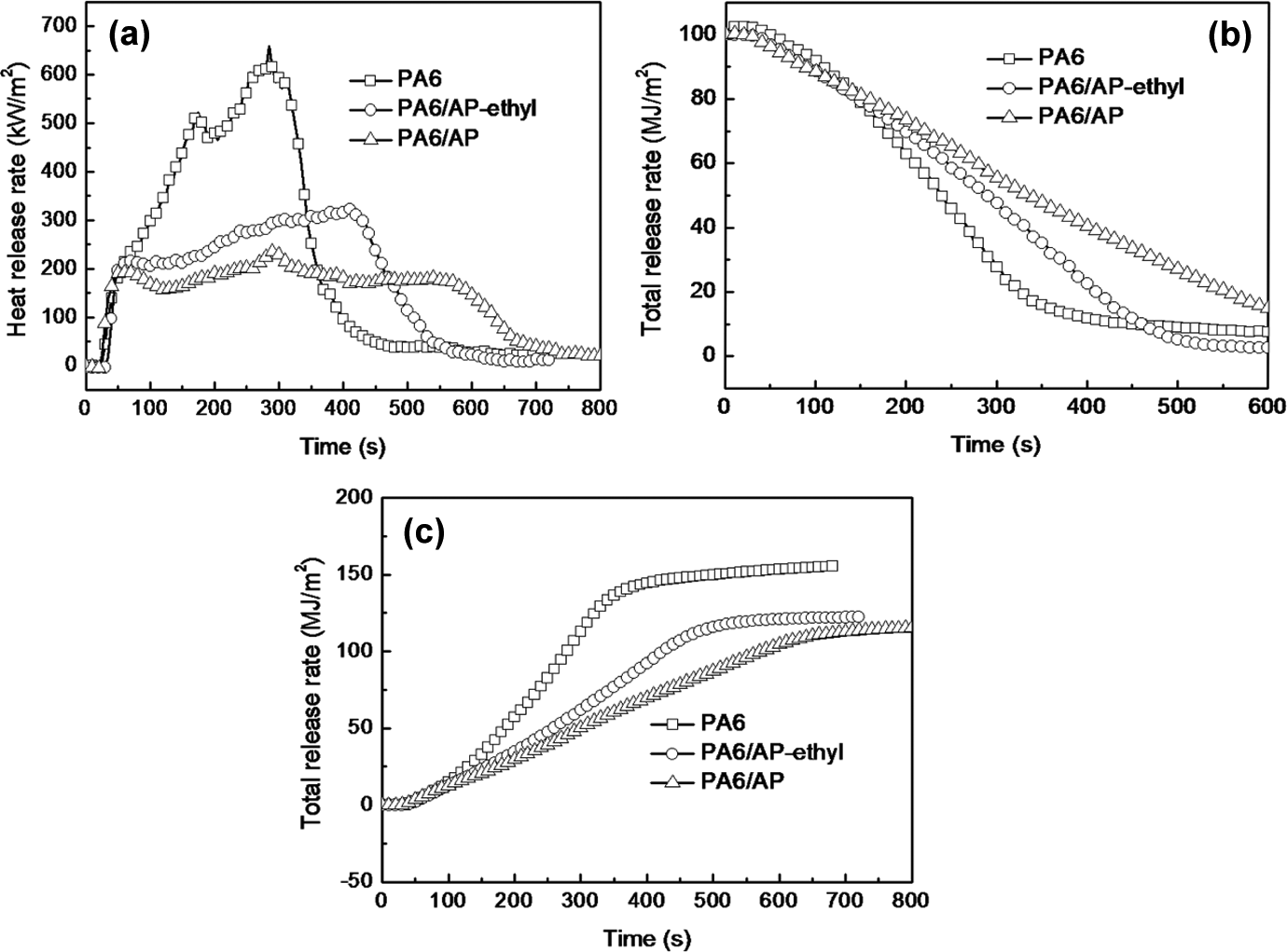
CCT results of PA6 and flame-retardant PA6. (a) HRR, (b) mass loss curves, and (c) THR. PA6: polyamide 6; HRR: heat release rate; THR: total heat release.
CCT data of PA6 and flame-retardant PA6.
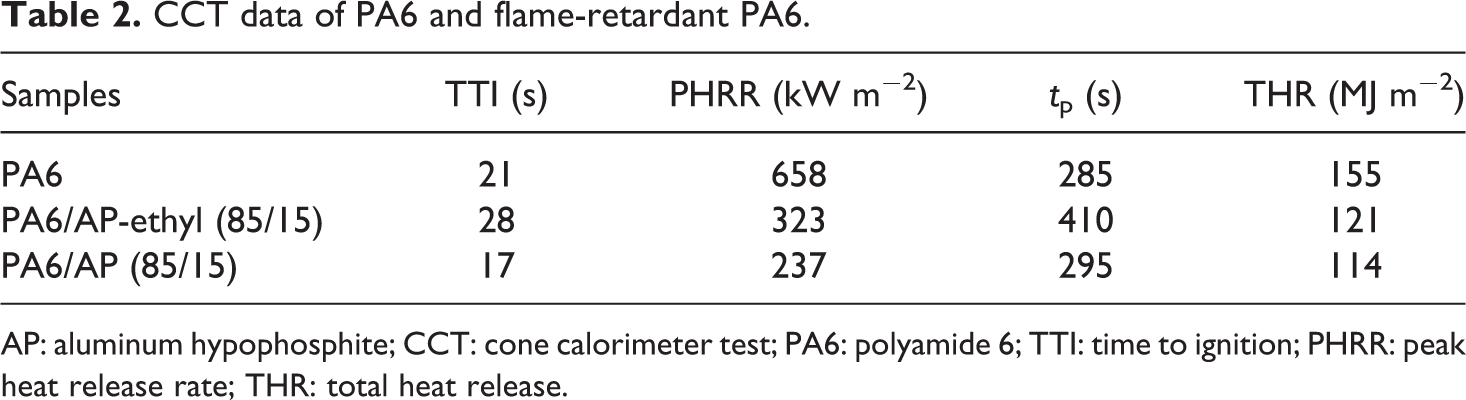
AP: aluminum hypophosphite; CCT: cone calorimeter test; PA6: polyamide 6; TTI: time to ignition; PHRR: peak heat release rate; THR: total heat release.
The pictures of residue after CCT test were shown in Figure 7. It can be seen that the char of PA6/AP was heavy, continuous, and strongly intumescent. Conversely, the char of PA6/AP-ethyl was rare and thin. This may result in the lower PHRR and TSR values of PA6/AP than those of PA6/AP-ethyl.

Digital picture of the char after CCT test for PA6/AP and PA/AP-ethyl. PA6: polyamide 6; AP: aluminum hypophosphite.
Crystallization properties of the flame-retardant PA6
The crystallization properties of PA6 and flame-retardant PA6 were investigated by DSC and the results were shown in Figure 8 and Table 3. From Figure 8 and Table 3, the crystallization temperature, melting temperature, and crystallinity of PA6/AP and PA6/AP-ethyl showed a slight decrease. It can be noticed that a broad but low peak at 195.1°C appeared in the cooling curve of PA6/AP. However, the crystallinity of PA6/AP revealed a slight higher than that of PA6/AP-ethyl. This might be the better dispersion of AP particles in PA6 matrix resulting from its smaller particle size than AP-ethyl.
Data derived from DSC test.
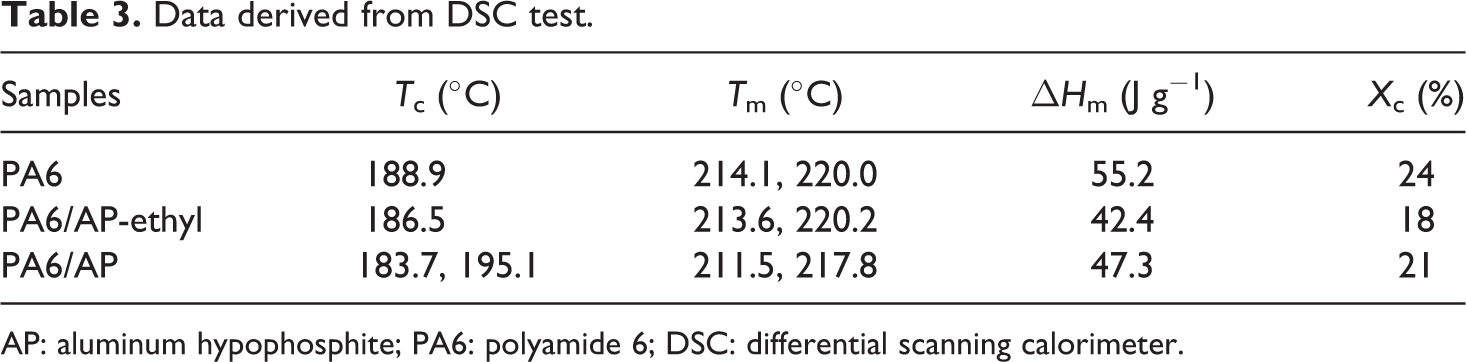
AP: aluminum hypophosphite; PA6: polyamide 6; DSC: differential scanning calorimeter.
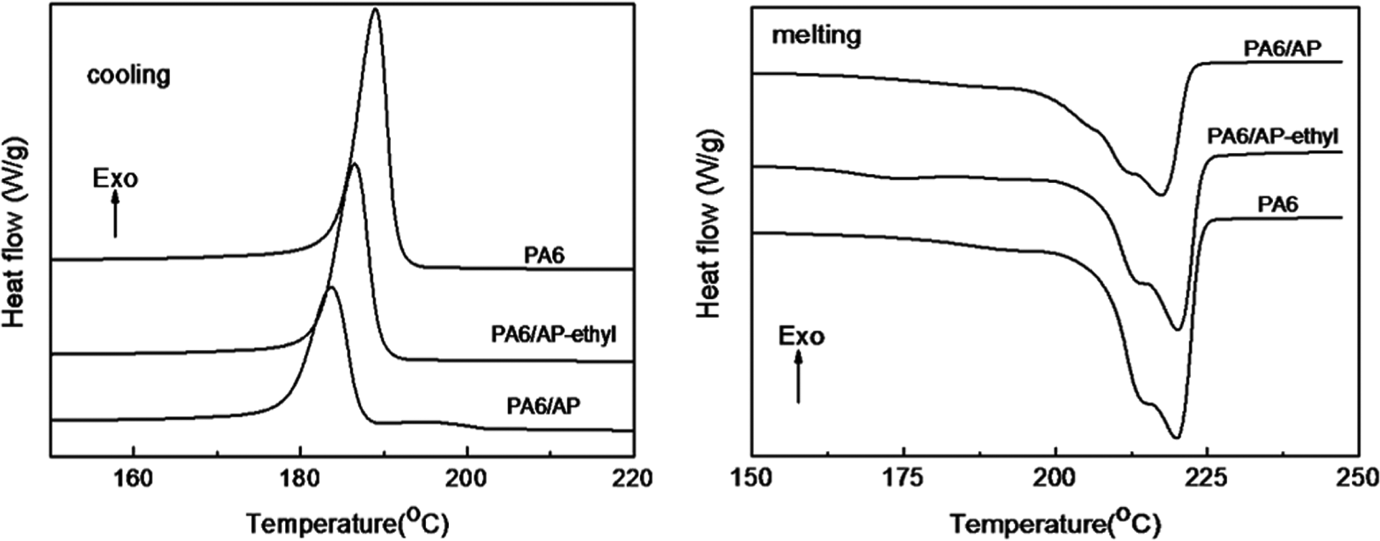
DSC curves of PA6 and flame-retardant PA6. DSC: differential scanning calorimeter; PA6: polyamide 6.
The mechanical properties of flame-retardant PA6 and the fracture surface morphology were shown in Figure 9. As shown in Figure 9, pure PA6 showed ductile stress–strain curve with the tensile strength of 54.8 MPa. However, PA6/AP and PA6/AP-ethyl displayed brittle stress–strain curve and their tensile strength decreased to 52.3 and 47.1 MPa, respectively. The tensile strength of PA6/AP was higher than that of PA6/AP-ethyl. In addition, pure PA6 showed a glossy and tough fracture surface morphology. The rough fracture surface morphologies for PA6/AP and PA6/AP-ethyl were observed, and the interface of PA6/AP was more obscure than that of PA6/AP-ethyl. Consequently, the small particle size of AP had a more uniform dispersion in PA6 matrix.
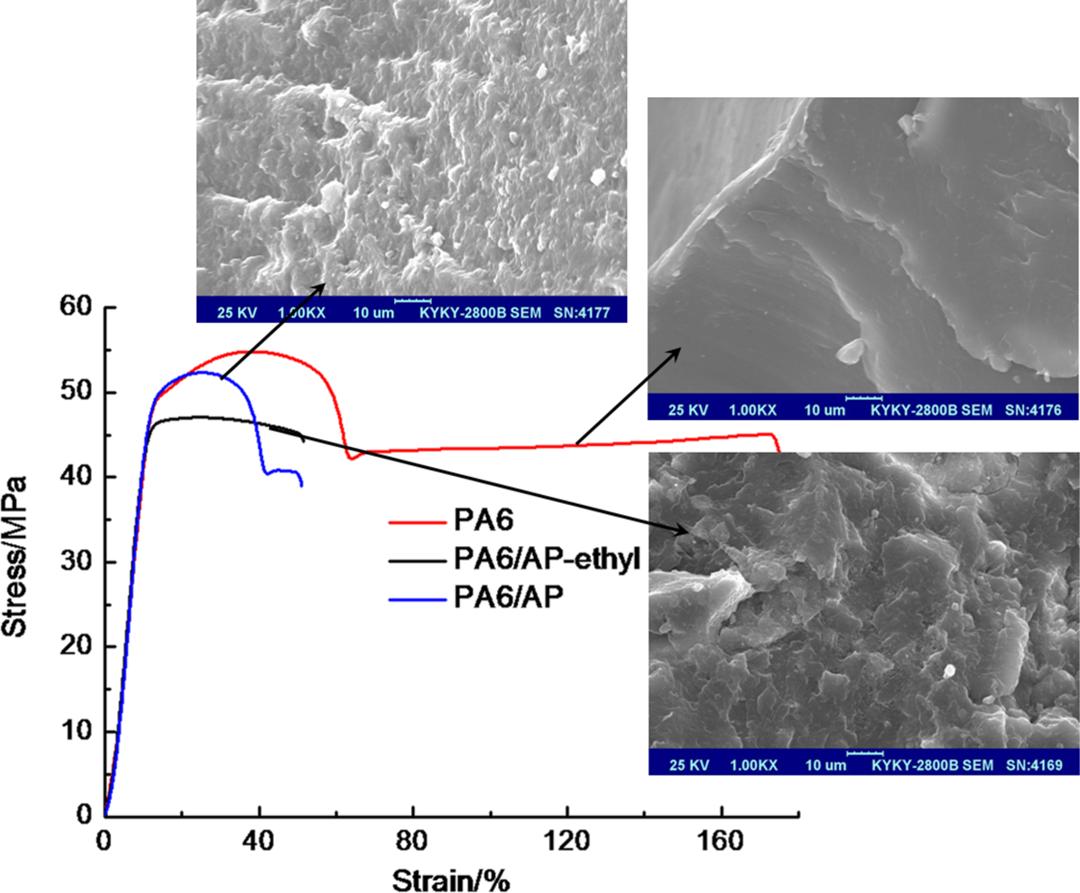
Mechanical test and SEM images of the fracture surfaces. SEM: scanning electron micrograph.
Conclusions
After the comparison of the effects of AP and AP-ethyl on the thermal degradation, flame performance, and mechanical properties of PA6, some significant and meaningful results could be obtained. Although AP had an obvious flammability in PA6 as well as AP-ethyl (CCT result), mechanical results and fracture surface morphology investigated indicated that AP with a smaller particle size had a uniform dispersion in PA6 matrix. Hence, we proposed that decreasing the particle size of flame retardant was an effective method to improve the efficiency of flame retardant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (51263003), industrial key project of Guizhou Province [2015]3008, high level innovative talents training project of Guizhou Province [2015]4039, Guizhou training program of outstanding youth in science and technology [2015]26, and the National Science and Technology Support Program (2013BAB07B03).
