Abstract
A novel high solids acrylic resin (HSARs) with high solid content (78%) was synthesized from co-polymerization of the methyl methacrylate, butyl acrylate, acrylate acid, hydroxyl propyl acrylate, and hyper-branched macromonomer (HBM) in this study. The HBM was prepared by an “A2 + B3 + CA2” reaction system that covers four steps. The progress of each step of the reaction was monitored by real-time analysis for acid value and hydroxyl value of the reactants. Fourier transform infrared spectroscopy (FTIR) and 1H nuclear magnetic resonance (NMR) was employed to characterize the structure of HBM. The viscosity, mechanical properties, solvent resistance, and volatile organic compounds (VOC) of the HSARs were also assessed. Compared with conventional commercial polyacrylate resin with a 42% solid content, solid content of HSARs was increased by 78% and VOC was decreased by 40% due to the addition of HBM. Moreover, the HSARs resin possesses good solvent resistance and lower viscosity, which provides the possibility of decreasing the organic solvent when using them to make operational coatings.
Introduction
The preparation and characterization of novel three-dimensional polymeric coatings is not only fascinating for academic research because of their complexity in structure and challenge for synthesis 1,2 but also because of their industrial applications. 3,4 It is well known that dendritic polymers have been especially attractive for industrial purposes because of their high functionality, high solubility, and unique environmentally friendly behavior. During the preparation of dendritic polymers, however, many steps were needed, leading to a relatively high cost and limiting their broad application. 5,6
Hyper-branched polymers could effectively increase the solid content and decrease the emission of volatile organic compounds (VOC). 3,7 They therefore have been generally recognized as potentially economical and more feasible counterparts for dendritic polymers. 8 –10 Since they can be manufactured more quickly and easily in a one-step or quasi-one-step polymerization procedure, their significantly lower manufacturing cost puts them in a much more favorable position for industrial applications, especially as coating resins. However, from more elaborate building blocks, most of the hyper-branched polymers reported are synthesized with so-called “A + B2” monomers. In the case of the “A + B2” model, A and B represent different functional groups, which could theoretically react only with the other one and not among themselves. 11 Generally, reaction of A-groups among themselves would produce low-molecular-weight products instead of polymers, while reaction of B-groups among themselves would cause gel formation. 12 Condensation polymerization of the two types of monomers will lead to a highly branched tree-like structure with one A-group and n B-groups (n ≥ 2) in each molecule. 12,13 In principle, the molecular weight would reach infinity when the degree of poly-condensation reaches infinity. It is therefore difficult to control the synthesis process, and this defect hampers its application. To improve this problem, co-polymerization of A2 and B3 or other mono-functional monomers which can also produce hyper-branched structures was developed. During the improved process, the polymerization is kept below the gel point by either limiting the degree of polymerization or changing the stoichiometry of these two reactants.
In this study, a novel hyper-branched macromonomer (HBM) with the structure “A2 + B3 + CA2,” consisting of phthalic anhydride (A2), glycerol (B3), and maleic anhydride (CA2), was prepared by means of successive esterification (where “A” represents –COOH, “B” represents –OH, and “C” represents –C=C–). Furthermore, to decrease the glass transition temperature of this HBM, stearic acid and benzoic acid (mono-functional monomer, A1) were grafted to its structure. After that, high solids acrylic resin (HSAR-1 resin) was prepared through co-polymerization of this HBM with acrylate monomers. In addition, the structure of HBM was characterized by fourier transform infrared spectroscopy (FTIR), acid value (Av) analysis, and hydroxyl value (Hv) analysis; the mechanical and chemical properties of HBM resin were analyzed and compared with conventional acrylate resins.
Experimental
Materials
O-Phthalic anhydride, maleic anhydride, and tri-(hydroxymethyl)-propane were obtained from Gaobo Chemical Co. (Tianjing, China). Glycerol was purchased from the Shanghai Chemical Reagent Corporation (Shanghai, China). All acrylate reagents used in this work were supplied by Kelong Chemical Reagent Corporation (Chengdu, China). Stearic acid and benzoic acid were obtained from Silong Chemical Co. (China). Three commercial acrylate resins, BS-965, BS-963, and SM5717 were kindly provided by Sanmu Coating Corp. Ltd (Jiangsu, China).
Synthesis of HBM
The HBM was prepared via successive esterification. First, 43.0 g solvent (xylene) and 13.1 g (0.1422 mole) glycerol were added to a 100-mL three-necked flask fitted with a mechanical stirrer and reflux condenser. The solution was heated to about 100°C, and then 38.0 g (0.2566 mole) phthalic anhydride, 14.6 g (0.1490 mole) maleic anhydride, 60.0 g (0.4471 mole) tri-(hydroxymethyl)-propane, 22.4 g (0.2003 mole) benzene carbonic acid, and 28.6 g (0.1001 mole) stearic acid were slowly added into the flask via a constant pressure funnel.
It must be noted that, during this four-step synthesis procedure, after the second step, subsequent reagents must not be added into the flask until the previous reagents have been consumed. The three critical times were determined by real-time analysis for Av) and Hv of the reactants. Upon completion of the reaction, the products were purified by reduced-pressure distillation (160°C/1 mm Hg) in order to remove low-boiling reactants and intermediates. Figure 1 illustrates the schematic mechanism to prepare the HBM.
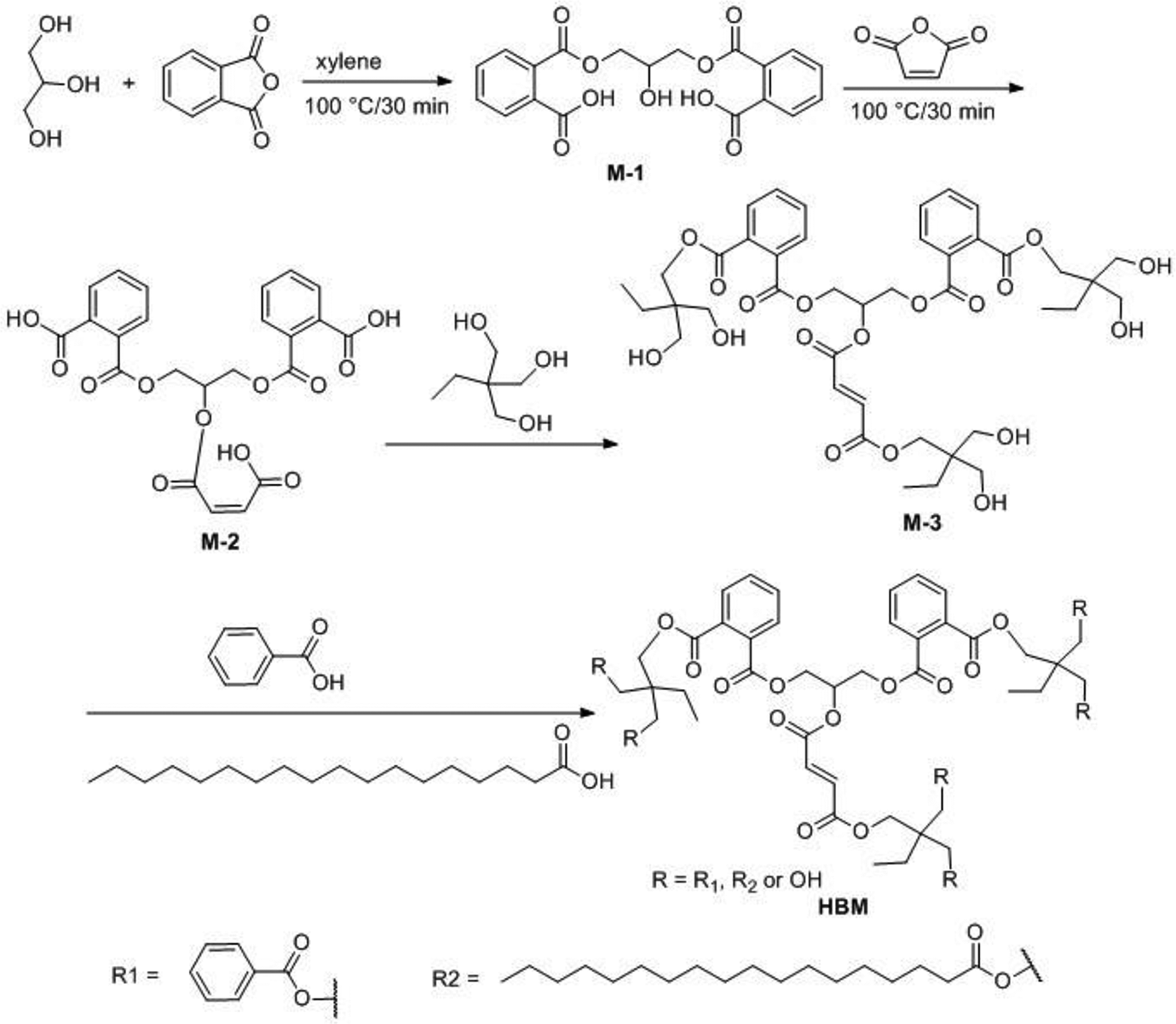
Schematic representation of the synthesis of HBM.
Characterization of HBM
Analysis of acid value
The Av of the intermediates and resin samples was measured by diluting about 1.0 g of the sample to 50-mL neutralized xylene–ethanol mixture (2:1, v/v). The sample was then titrated with 0.2 M KOH in ethanol with phenolphthalein as an indicator. 14 This analysis was done by triplicated for every sample.
Analysis of hydroxyl value
The Hv of the intermediates and resin samples was determined by acetylation of the hydroxide groups with acetic anhydride. The excess acetic acid and free acid groups in the reaction media were titrated with 0.2 M KOH. The Hv was determined based on the Av as described in the literature. 15 This analysis was done by triplicated for every sample.
Nuclear Magnetic Resonance (NMR) and FTIR analysis
1H NMR and 13C NMR were recorded on a AVANCE 300 MHz UltrashieldTM Nuclear Magnetic Resonance (Bruker, USA) using methanol-d4 as the solvent and TMS as the internal standard. FTIR was obtained in the absorbance mode using a Nicolet MX-1E FTIR spectrometer (Nicolet, Japan). Samples were directly placed between two KBr plates about 2 mm in diameter. Typically, 32 scans over the range of 400–4000 cm–1 were utilized for each sample with a resolution of 4 cm–1.
Preparation of HSAR-1 with HBM
The HSAR was prepared by radical polymerization. Xylene solvent (150 g) was added to a 250-mL four-necked flask fitted with a mechanical stirrer, nitrogen inlet, reflex condenser, and constant pressure funnel. After de-aerating with nitrogen gas for 20 min, the acrylic monomers mixture (80%, w/w) including HBM (20%, w/w), dicumyl peroxide (initiator, 3.0%, w/w), β-mercaptoethanol or n-dodecanethiol (chain transfer reagent, 2.0%, w/w) was added. To the 80% (w/w) acrylic monomers mixture, methyl methacrylate (44%, mole ratio), butyl acrylate (36%, mole ratio), acrylic acid (11%, mole ratio), and hydroxyl propyl acrylate or hydroxyl ethyl acrylate (9%, mole ratio) were slowly added into the flask step by step via a constant pressure funnel. The polymerization was then conducted at 140°C for 6 h; after this, the remaining solvent and initiator were added within 1 h and then, conducted the reactants at 140°C for another 1 h. Finally, the products were cooled and purified by pressure-reduced distillation (160°C/1 mmHg), with 95% conversion. The solids content of the resin was adjusted to 75%, and the product was named HSAR-1.
Film formation of HSAR-1 resin
A cleaned steel panel was coated with HSAR-1 solution with a solid content of 75% and dried at room temperature. The steel panel was then placed in a vacuum oven (DZF-6050, Shanghai, China) and baked at 140°C for 30 min to remove the solvent and form a film.
Measurement of physical and mechanical properties of resins
Measurement of viscosity
Two methods were utilized to measure the viscosity of resin samples. The first and simpler method was performed using the TU-4 CUP method at 25°C (Chinese standard GB/T1723-1993), in which the time required for a sample to flow from the top to the bottom of the cup was measured in seconds (s). The second and more complex method, providing more accurate results reported in mPa s, was performed using a BrookField LVDV-III Ultra (Middleboro, MA, USA) at 25°C. 16
Measurement of mechanical properties
The adhesion property of HSAR-1 resin samples was measured according to ASTM D3359-17. A cleaned steel panel was coated with HSAR-1 solution with a solid content of 75% and dried at room temperature. Employ the x-cut tape test and adhesion is assessed qualitatively on the 0–5 scale. The flexibility of resin samples was measured according to Chinese standard GB 1731-1993 and indicated by the diameter of test axles (0.5 mm, 1.0 mm, 1.5 mm, 2.0 mm, 2.5 mm, 5.0 mm, 7.5 mm); the smaller the diameter, the better the flexibility. The impact resistance of resin samples was measured according to the ASTM D6905-03 (2012) and indicated by the weight of hammer times the height (kg cm); the larger the value, the better the impact resistance. The hardness of resin samples was measured according to ASTM D2134-93 (2012) with a Sward-Type Hardness Rocker. The larger the value, the better was the hardness.
Measurement of solvent resistance
The solvent resistance of resin for hydrochloric acid, sodium hydroxide, and 70# petrol was measured. For these analyses, the HSARs films with 52 mm thickness were inserted inside each solvent at room temperature.
Results and discussion
Synthesis of HBM
The HBM was prepared via successive esterification as illustrated in Figure 1. For the synthesis of intermediate M-1, the mole amount of phthalic anhydride added was slightly lower than the theoretical mole amount (2:1) according to the reaction formula. Intermediate M-1 could be obtained by semi-esterification via different activities of the three hydroxyl groups of glycerol due to electronic and steric effects. The synthesis of intermediate M-2 was straightforward using a slight excess mole amount of maleic anhydride over intermediate M-1 and proper reaction time, because there was only one hydroxyl group in intermediate M-1. Intermediate M-3 was achieved via esterification of M-2 using tri-(hydroxymethyl)-propane. During the synthesis of HBM, the mole amount of monomer mixtures (benzoic acid to stearic acid is 2:1, mole ratio) added was 33% lower than the mole amount of hydroxyl groups in M-3. This was in order to reserve some hydroxyl groups in the HBM so that the high solid content resin with HBM could have a better compatibility with other acrylic resins.
Characterization of HBM
Av and Hv of the intermediate and HBM
It is evident from Figure 1 that HBM was prepared by successive esterification. The completion point for each step was monitored by real-time analysis for Av and Hv of the reactants. The changing trend of Av and Hv during each step played an important role in monitoring whether a reaction was completed or not.
During the synthesis of M-1, if the semi-esterification of glycerol with phthalic anhydride was performed according to the reaction illustrated in Figure 1, the Av and Hv in M-1 should approach 2:1, according to theoretical calculation. Figure 2 shows that, for M-1, Av = 285.2 and Hv = 147.6, which indicated that the Hv exhibited a slightly higher than theoretical value due to the slightly lower amount of phthalic anhydride added, consistent with the expected results as described in “Synthesis of HBM” section. Similarly, for the synthesis of M-2, if hydroxyl groups in M-1 were completely reacted with maleic anhydride, the Hv would decrease to zero, while the Av would increase. Figure 2 shows that a logical change of Hv (4.7) and Av (348.7) occurred. The results of Av and Hv for M-3 were as expected theoretically. Figure 2 also indicates that after the formation of HBM, its Hv decreased from 410.7 (of M-3) to 134.0, while the Av increased from 3.9 (of M-3) to 4.7. These results implied that stearic acid and benzoic acid were successively grafted onto M-3, and that some hydroxyl groups in HBM were preserved as desired.
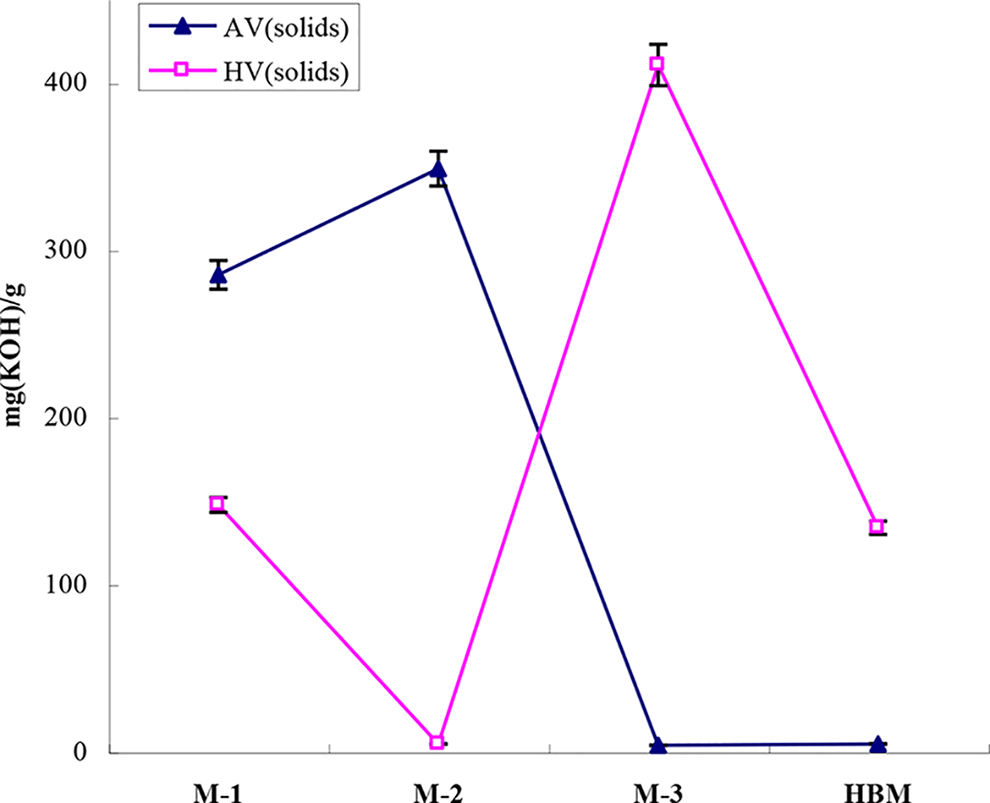
Av and Hv of the intermediate and HBM.
NMR analysis for intermediates and FTIR analysis for HBM
In order to confirm each reaction process, NMR analysis of the three intermediates, M-1, M-2, and M-3, were undertaken, the results were as below:
M-3: 1H NMR, δ: 8.06 (overlapped, 2H), 7.91 (d, J = 8.4 Hz, 2H), 7.72 (overlapped, 4H), 6.63 (overlapped, 2H), 5.70 (m, 1 H), 4.48 (dd, J = 6.5, 4.9 Hz, 2H), 4.40 (s, 4H), 4.33 (s, 2H), 4.18 (dd, J = 6.5, 5.3 Hz, 2H), 3.80 (s, 2H), 3.78 (s, 4H), 3.64 (s, 4H), 3.24 (s, 2H), 1.78 (q, J = 6.9 Hz, 4H), 1.45 (q, J = 7.2 Hz, 2H), 0.96 (t, J = 7.1 Hz, 6H), 0.85 (t, J = 6.7 Hz, 3H). 13C NMR, δ: 169.4, 168.3, 168.1, 168.0, 168.0, 167.6, 132.2, 132.1, 131.4, 131.4, 131.3, 131.3, 130.5, 130.5, 130.4, 130.4, 130.3, 130.3, 130.3, 130.2, 71.5, 69.8, 69.7, 69.4, 66.7, 66.7, 66.7, 66.6, 66.6, 66.5, 65.3, 65.2, 42.7, 42.6, 42.6, 20.5, 20.5, 20.3, 8.9, 8.9, 8.8.
Above results indicated that, intermediates were obtained as described in Figure 1.
The FTIR spectrum for HBM is shown in Figure 3. The strong stretching adsorption at 3515.1 cm–1 indicated the presence of –OH groups in HBM. 15 Absorbance at 3120 cm–1 marked the presence of unsaturated ν (–C–H) of methyl and methylene. The characteristic adsorption at 1646.9 cm–1, 870 cm–1 indicated the presence of C=C 7 and (–C–H) external bending vibration of benzene, respectively. Three adsorption peaks, that is, 1724.8 cm–1, 1268.5 cm–1, and 1070.4 cm−1, resulted from the vibrations of C=O and C–O–C. This information indicated that HBM was prepared as expected via successive esterification.
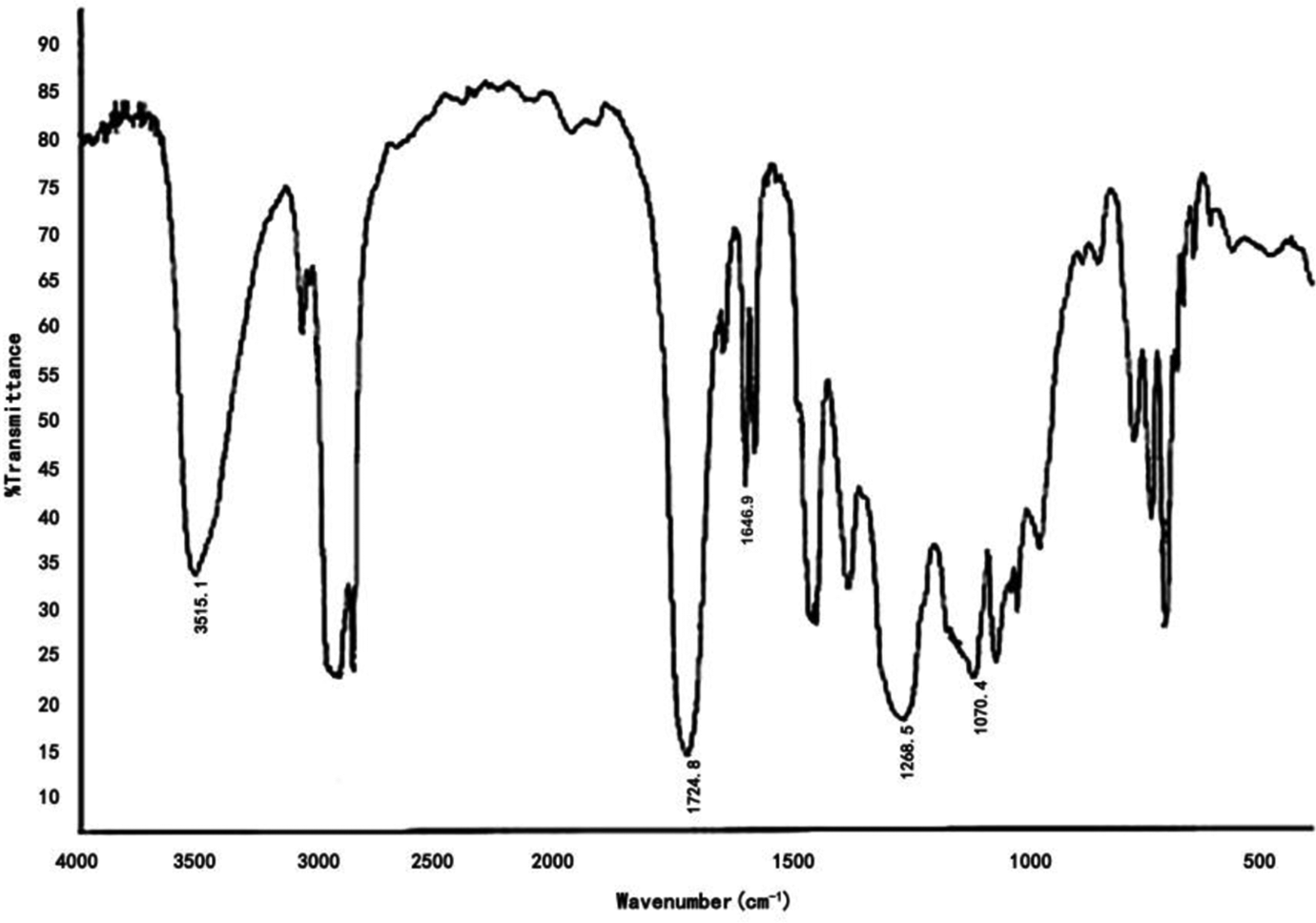
The FTIR spectrum of HBM.
The effect of chain transfer reagent on the property of resin
During the preparation of high solids resins, polymers consisting of low molecular weight and narrow molecular weight distribution are generally employed so that a proper viscosity adequate for spraying may be achieved. However, lower molecular weight would lead to diminution of the properties of the cured film. The balance between molecular weight of polymer and the vital properties of the coating, such as glass transition temperature, melt viscosity, and so on, is therefore very important. In the case of HSAR prepared in this work, there are clear advantages in adopting HBM. On the one hand, the presence of hydroxyls in HBM increased the activity and shortened the time for film formation; on the other hand, the control of molecular weight was made more convenient by changing the type and dosage of chain transfer reagent, due to its radical polymerization mechanism, rather than employing poly-condensations during the synthesis process. Table 1 shows the effect of two chain transfer agents on the properties of resins, indicating that β-mercaptoethanol was a better chain transfer agent than n-dodecanethiol.
Effect of chain transfer agents on the property of resins.
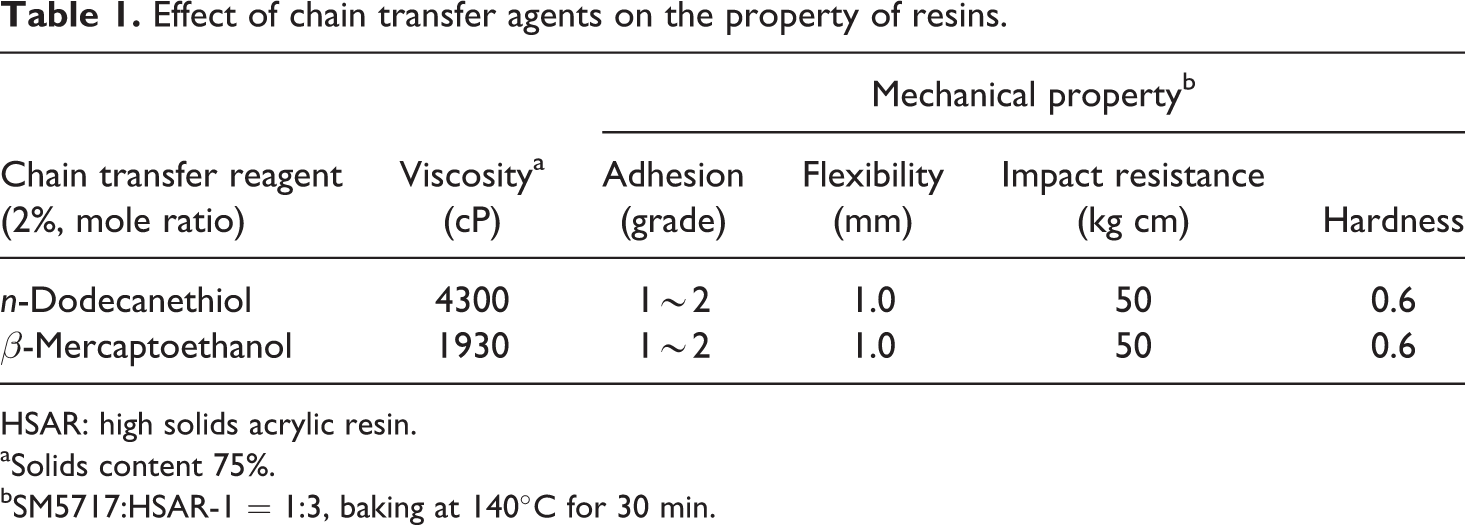
HSAR: high solids acrylic resin.
aSolids content 75%.
bSM5717:HSAR-1 = 1:3, baking at 140°C for 30 min.
The effect of Hv and Av on the property of resins
It is well known that hydroxyl groups and carboxyl groups play a crucial role in affecting the film properties of high solids coatings by affecting the formation of hydrogen bonds in the products. As a result, the content of hydroxyl groups and carboxyl groups influences the chemical, physical, and mechanical properties of high solids coatings, such as viscosity and adhesion. 17,18
As shown in Table 2, as Hv increased, the viscosity of the resin increased, while adhesion and impact resistance decreased. As mentioned previously, due to the formation of hydrogen bonds, an increase in the number of hydroxyl groups increased the brittleness of HBM coating so as to decrease its adhesion property and impact resistance.
Effect of Hv on the properties of resins.

HSAR: high solids acrylic resin; Hv: hydroxyl value.
aSolids content 75%.
bSM5717:HSAR-1 = 1:3, baking at 140°C for 30 min.
Table 3 indicates that the content of carboxyl groups had a similar effect on the properties of resins as that of the content of hydroxyl groups.
Effect of Av on the properties of resins.

Av: acid value; HSAR: high solids acrylic resin.
aSolids content 75%.
bSM5717:HSAR-1 = 1:3, baking at 140°C for 30 min.
The relationship between the viscosity and concentration of HSAR-1 and commercial acrylic resins
Figure 4 indicates that the viscosity of both HSAR-1 and commercial acrylic resins increased as their concentrations increased. However, the viscosity of commercial acrylic resins increased faster than that of HSAR-1. The explanation for this observation is that, compared with commercial acrylic resins, which have a linear chain structure, the molecular structure of HSAR-1 does not tend to coil with each other, and it has a relatively smaller fluid mechanical radius and a better fluidity. 7 Thereafter, HSAR-1 maintains a relatively small viscosity while its concentration increased, and this is the most important characteristic of hyper-branched polymers. 19
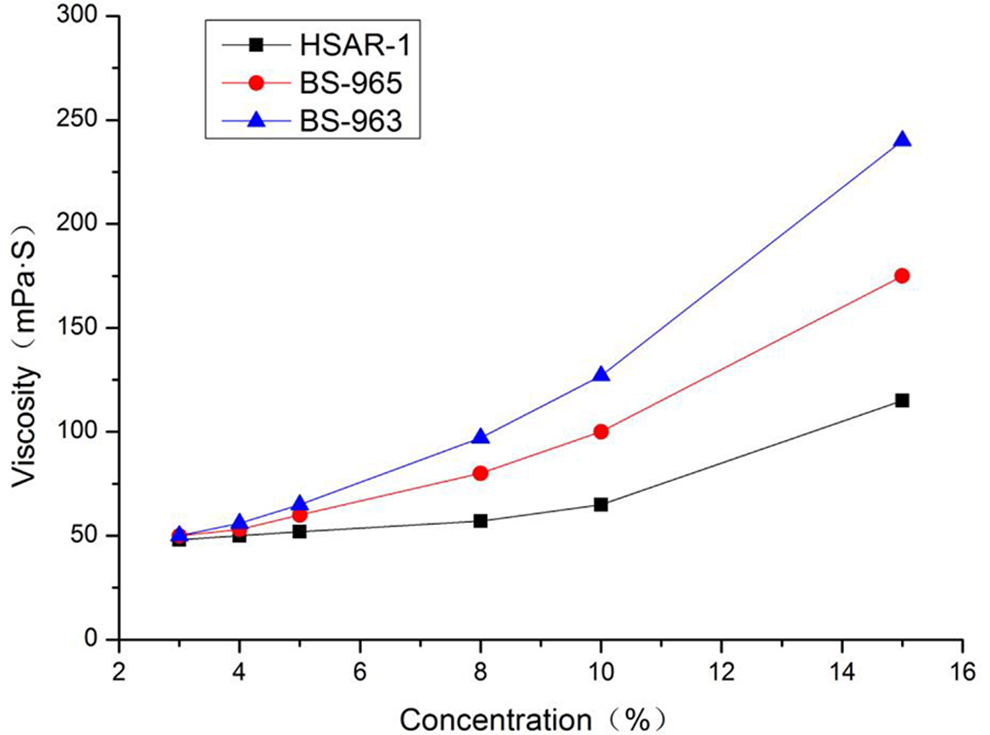
The relationship between the viscosity and concentration of the three acrylic resins.
Properties of resins
To explore the advantages of HSAR-1, its film properties were measured and compared with those of conventional acrylic resins. Table 4 indicates that HSAR-1 had a higher solids content and lower VOC emission than BS-965, mainly due to the addition of HBM. The presence of HBM in HSAR-1 therefore decreased its viscosity and improved the solids content and decreased the emission of VOC. In addition, Table 4 indicates that other important properties, such as impact resistance, adhesion, and petrol resistance, were not weakened due to the addition of HBM.
Properties comparison of HSAR-1 and commercial acrylic resin.
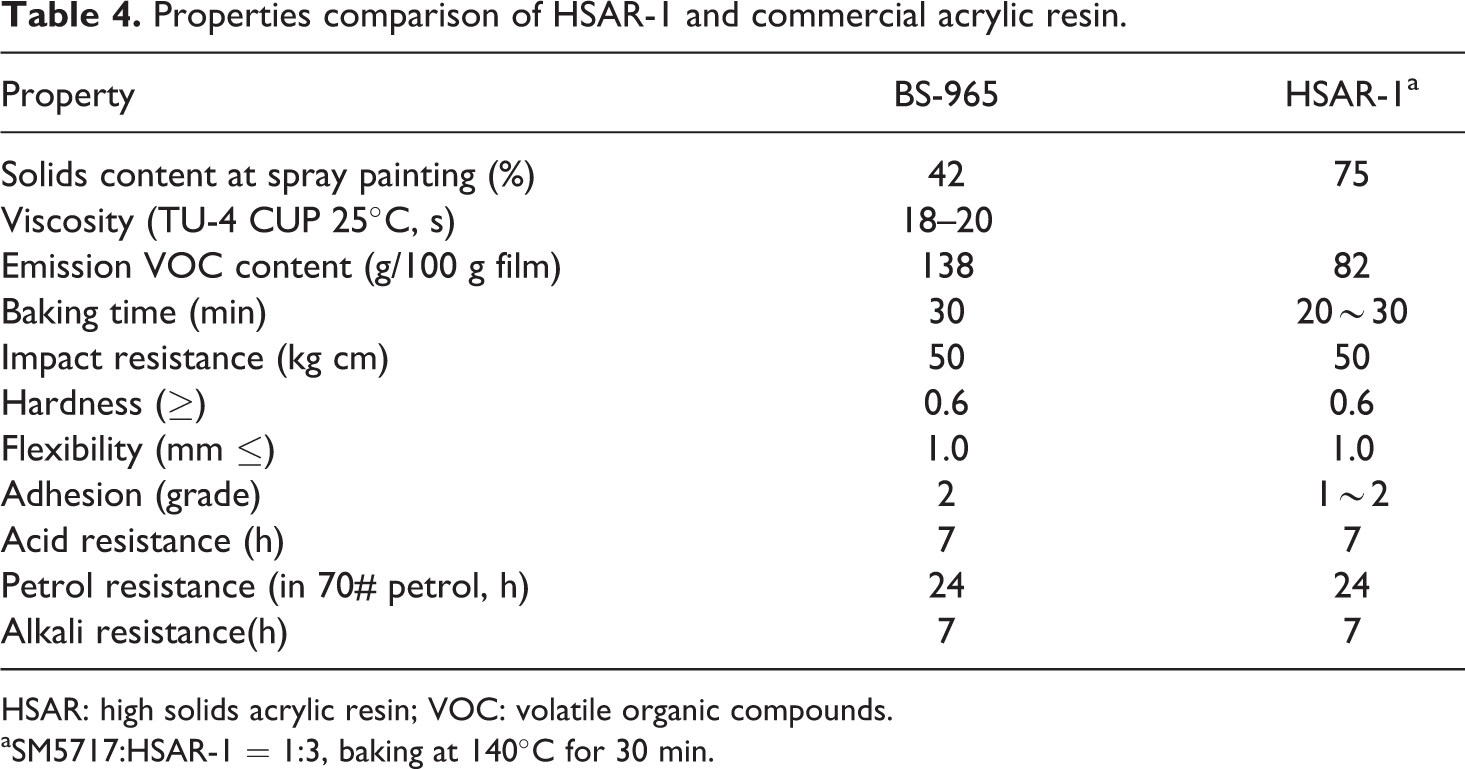
HSAR: high solids acrylic resin; VOC: volatile organic compounds.
aSM5717:HSAR-1 = 1:3, baking at 140°C for 30 min.
Conclusions
This study demonstrated that a novel HSAR-1 with 75% solids content was prepared by radical polymerization of HBM with other acrylic monomers. The HBM consisting of glycerol, phthalic anhydride, and maleic anhydride with an “A2 + B3 + CA2” structure could be prepared by a four-step successive esterification. The completion point for each step was determined by real-time analysis for Av and Hv of the reactants, and FTIR analysis confirmed the formation of HBM.
Meanwhile, the physical and mechanical properties of HSAR-1 were investigated and compared with those of conventional acrylic resins. The results indicated that HSAR-1 not only exhibited improved properties over conventional resins but also increased the solids content and effectively decreased VOC emission as expected. In addition, other important properties of HSAR-1, such as impact resistance, adhesion, and petrol resistance, did not decline due to the addition of HBM.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support from the National Natural Science Foundation of China (Project No.21474114), Application and Industrialization of Hyperbranched Polymer Technology in Environmentally Friendly High Solid Coatings (BY2016042-01), and Youth Scientific Innovation Research Group of Sichuan (2017TD0024).
