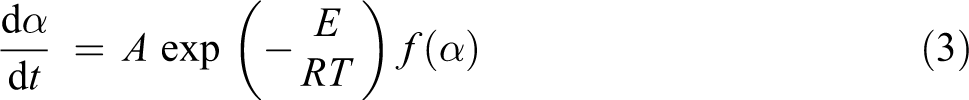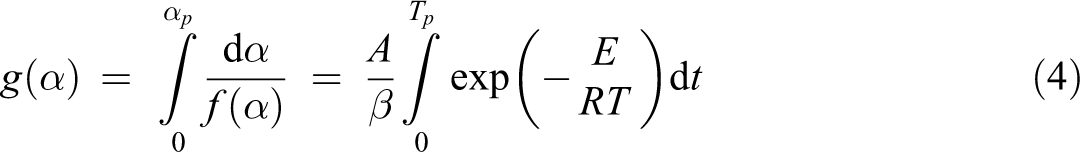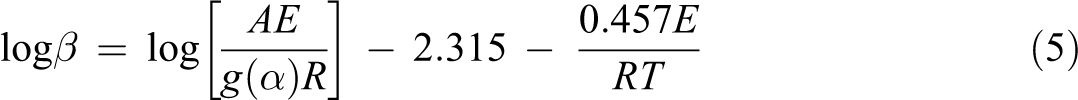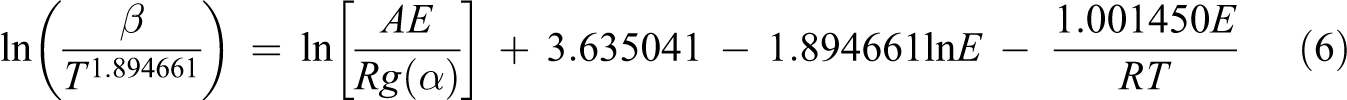Abstract
In this article, coumarin-based random copolymer which can be utilized for atom transfer radical polymerization was investigated both theoretically and experimentally. The thermal degradation mechanism and the activation energies (E a) were obtained by means of the Coats–Redfern (CR), Tang, and Flynn–Wall–Ozawa (FWO) methods. The thermal degradation reaction mechanism for random copolymer obeyed the phase boundary model (contracting volume, R 3) of solid-state mechanism. The thermodynamic properties in the range from 100 K to 500 K have been obtained. The calculated HOMO–LUMO energy gap and electronic properties, such as chemical hardness (η), electron affinity (A), chemical potential (μ 0), global softness (ζ), electronegativity (χ), global electrophilicity (ω), and dipole moment (μ), were investigated and discussed. The molecular electrostatic potential analysis of the random copolymer was also computed.
Introduction
Coumarin and derivatives play an important role as versatile, because of their applications in photochemistry, medicinal chemistry, and synthetic chemistry. Many of these compounds have found to be active as excellent laser dyes at near-ultraviolet to green wavelength, 1 fluorescent probes, 2 bactericides, antibacterial agent, anti-allergy compound, anti-diabetic, cardiovascular agents, control of anti-aging, diagnostic probes for amyloid accumulation disease, therapeutic agents for fibrosis and their chances as skin-whitening compounds, fluorescent brighteners, 3 antimicrobial, 4 antifungal, 5 anti-inflammatory, 6 anticoagulant, 7 antitumor, 8 and free radical scavenging activity especially the superoxide anions generated by activated neutrophils and to investigate neuronal circuits in tissues. 9
In recent years, the polymers containing coumarin have attracted great attention as a consequence of these significant properties. The polymers containing coumarin have also been incorporated in a wide variety of functional materials, such as a two-photon optical memory, 10 photoinduced bendable actuator, 11 and reversible cluster formation. 12 The thermal degradation study of polymers, which are used to determine the upper temperature limit of use for a polymeric material, is very important for polymer science in many cases. Therefore, the use of thermogravimetric data for the determination of kinetic parameters has been under great attention. For this purpose, thermogravimetric analysis (TGA) is a technique largely used due to the information provided by a simple thermogram and its simplicity. 13 To forecast the lifetime as thermal of materials, the thermal decomposition analysis which is especially useful for the study of polymeric materials can be utilized. 14 –16 This analysis can ensure important information relating to critical applications, such as defense, aerospace, and service performance. 15 Currently, quantum mechanical calculations with density functional theory (DFT) are very popular in theoretical modeling due to their accuracy and efficiency with regard to the assessment of molecular properties. 17 Most of the physical and chemical properties of monomer and polymer can be estimated by various computational methods. 18,19 In addition, by adding a monomeric unit to the polymer chain, the thermal properties of a polymer can be significantly modified. 20 This coumarin-based random copolymer can be used for the preparation of various photoresponsive polymers, which has become the focus of interest, as well as thermal decomposition and theoretical studies on designing polymers containing coumarin using the DFT method. So, the main purpose of this work was to obtain the thermal behavior and quantum chemical studies of the structure and reactivity of the coumarin-based random copolymer, which can be used as a macroinitiator for the preparation of designing various polymers.
Experimental and theoretical details
The TGA data of the random copolymer were obtained from our previous study. 21 The activation energy (E a) calculations were made using different methods with the help of this data. Thermogravimetric measurements were obtained from a TGA-50 instrument under flowing nitrogen atmosphere. The thermal stability of random copolymer was determined with the help of TGA at various heating ratios, such as 5, 15, 25, and 35°C min–1. The random copolymer was heated from the room temperature to 500°C. The decomposition E as which are determined using the Coats–Redfern (CR), Tang, and Flynn–Wall–Ozawa (FWO) methods at various heating ratios are summarized below. All the quantum chemical calculations of the random copolymer were carried out using DFT with hybrid Becke–3–Lee–Yang–Parr (B3LYP) 22 –24 combined with 6–311 + G(d, p) basis set using GaussView molecular visualization program 25 and Gaussian 09W package program 26 without any constraint on the molecular geometry.
Thermogravimetric analysis
TGA provides general information about the comprehensive reaction kinetics rather than the particular reactions. The reaction rate (% min−1) for the decomposition reaction is described as shown below 27 :
where k(T) is rate constant and depends on temperature, α is the relative decomposition conversion, t is time, and f(α) is a function of the conversion depend on the mechanism. The reaction rate constant is stated by the Arrhenius equation:
where A (min−1) is the pre-exponential factor, E (J mol−1) is the E a, and R is the gas constant (8.314 J mol−1 K−1).
If the temperature of the sample is changed by a controlled and constant heating rate (β = dT/dt), equations (1) and (2) result in:
The integration form of equation (3) is generally expressed as:
where g(α) is the integral form of kinetic model. This equation is well known in the literature and frequently used the reaction mechanisms of solid-state reactions. Different kinetic methods based on this equation were used in this study.
FWO method
FWO method has generally been used in the investigation of the E a of solid-state reactions as shown in the decomposition processes of polymers. 28,29 Equation (4) is integrated by means of the Doyle approximation 30 and is usually described by the following equation:
The E a at different degrees of conversion can be obtained from a plot of log β versus 1000/T.
Tang method
The following equation has been suggested by Tang and can be described as 31 :
The slope of the straight line for the plot of ln (β/T 1.894661) versus 1000/T is used to determine the E a.
CR method
CR method 32 uses an asymptotic approximation for the resolution of equation (4) and it is given by:
The different expressions of integral function of conversion for thermal degradation are given in Table 2. The E a can be obtained from a plot of ln (g(α)/T 2) versus 1000/T.
Results and discussion
Kinetic analysis
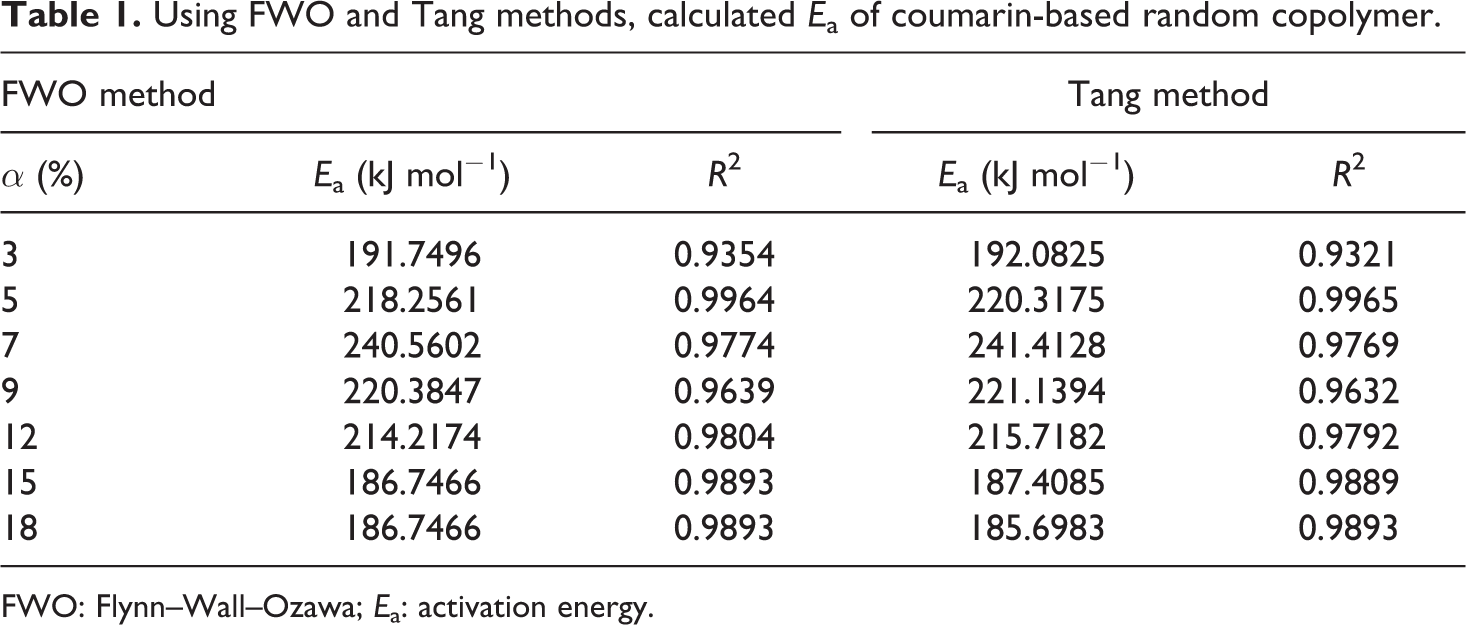
Thermal degradation behavior of random copolymer was determined by way of TGA at several heating rates (5, 15, 25, and 35°C min−1). To abstain from the overlapping of inflection point temperatures, we used 10°C min−1 intervals between measurements. 33 With the help of equation (5) or the FWO method, the decomposition E a can be obtained from a slope of log β versus 1000/T at different degrees of conversion. This equation was derived from the Doyle approach, therefore we have worked at the low degrees of conversion such as intervals from 3% to 18%. The straight lines in Figure 1 are almost parallel, this indicates that this method can be applied to the experimental values of random copolymer at conversion degrees from 3 to 18%. The E a related to the different degrees of conversion was determined using the FWO method and given in Table 1. As it can be seen in Table 1, the average value of the E a for random copolymer was found to be 208.38 kJ mol−1.
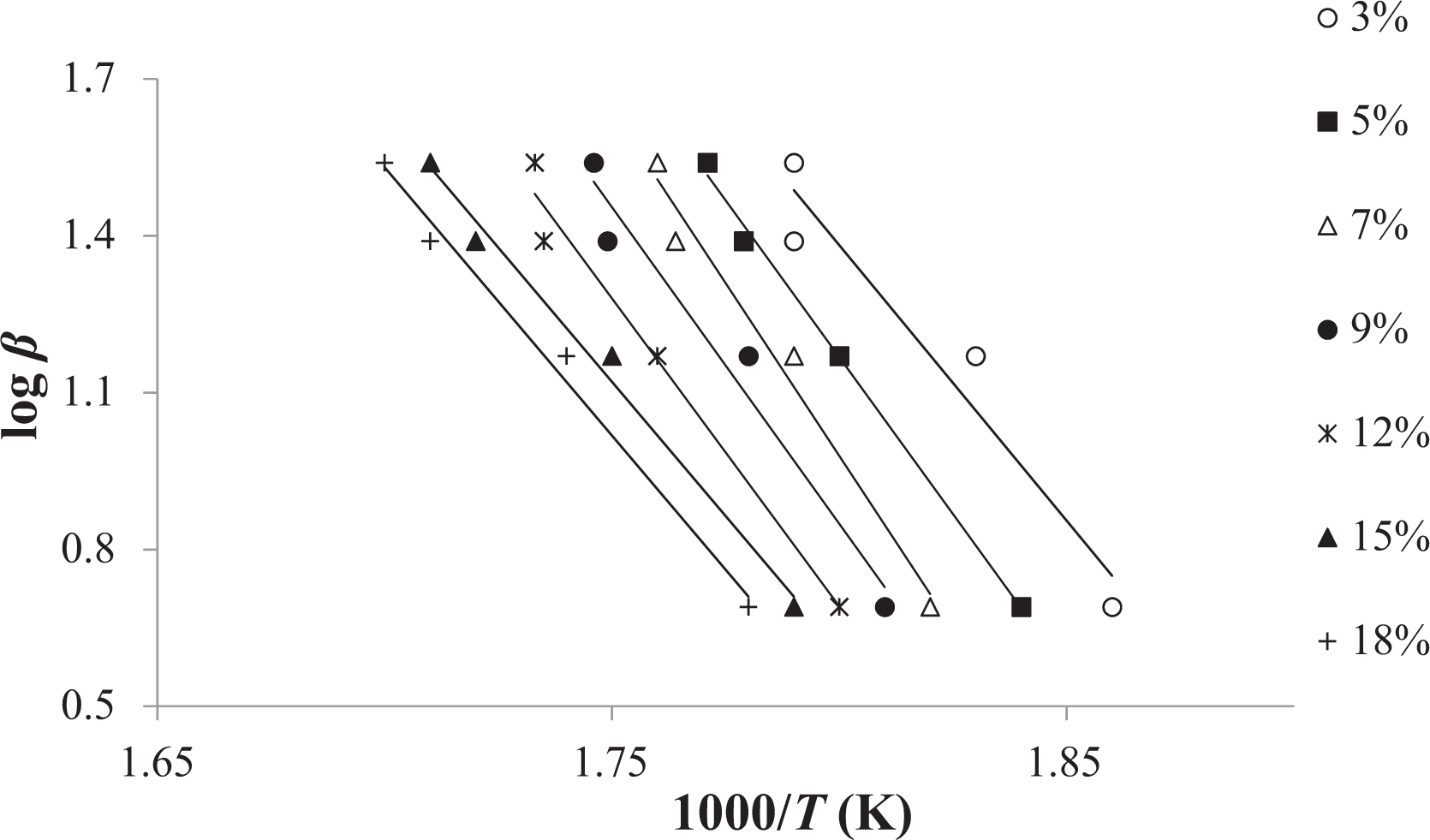
FWO plots of coumarin-based random copolymer at different degrees of conversion. FWO: Flynn–Wall–Ozawa.
Using FWO and Tang methods, calculated E a of coumarin-based random copolymer.
FWO: Flynn–Wall–Ozawa; E a: activation energy.
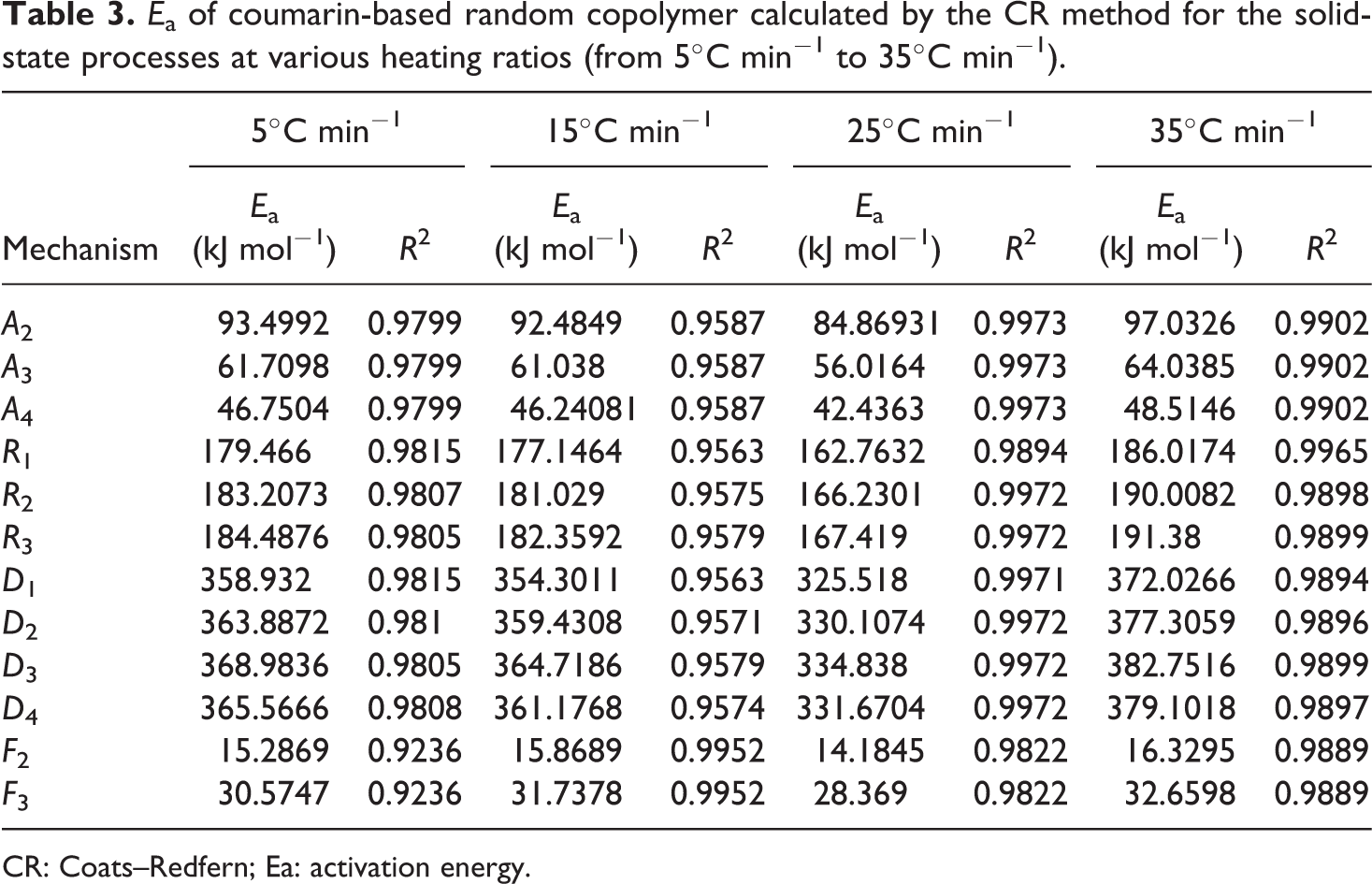
With the help of equation (6) or the Tang method, the decomposition E a can be obtained from the slopes of ln (β/T 1.894661) versus 1000/T at different degrees of conversion. The straight lines in Figure 2 are almost parallel, this indicates that this method can be applied to the experimental values of random copolymer at conversion degrees from 3 to 18%. The E a related to the different degrees of conversion was determined using the Tang method and given in Table 1. As it can be seen in Table 1, the average value of the E a for random copolymer was found to be 209.11 kJ mol−1. Table 1 also indicates that this E a which was obtained by the Tang method was very close to that of 208.38 kJ mol−1 obtained by the FWO method. Compared to other methods, the Tang and FWO methods are more advantageous, but they do not have the basic knowledge of the reaction mechanism for determining the E a. These methods have been utilized for control of a mechanism of thermal degradation models. 34 The different thermal degradation processes listed in Table 2 and the decomposition E a can be determined from a slope of ln (g(α)/T 2) versus 1000/T. The decomposition E as obtained by CR method for random copolymer are listed in Table 3 for each heating rate and mechanism. The kinetic thermal analysis indicated that the E a value (R 3) obtained using the CR method was in good agreement with those obtained using the Tang and FWO methods.
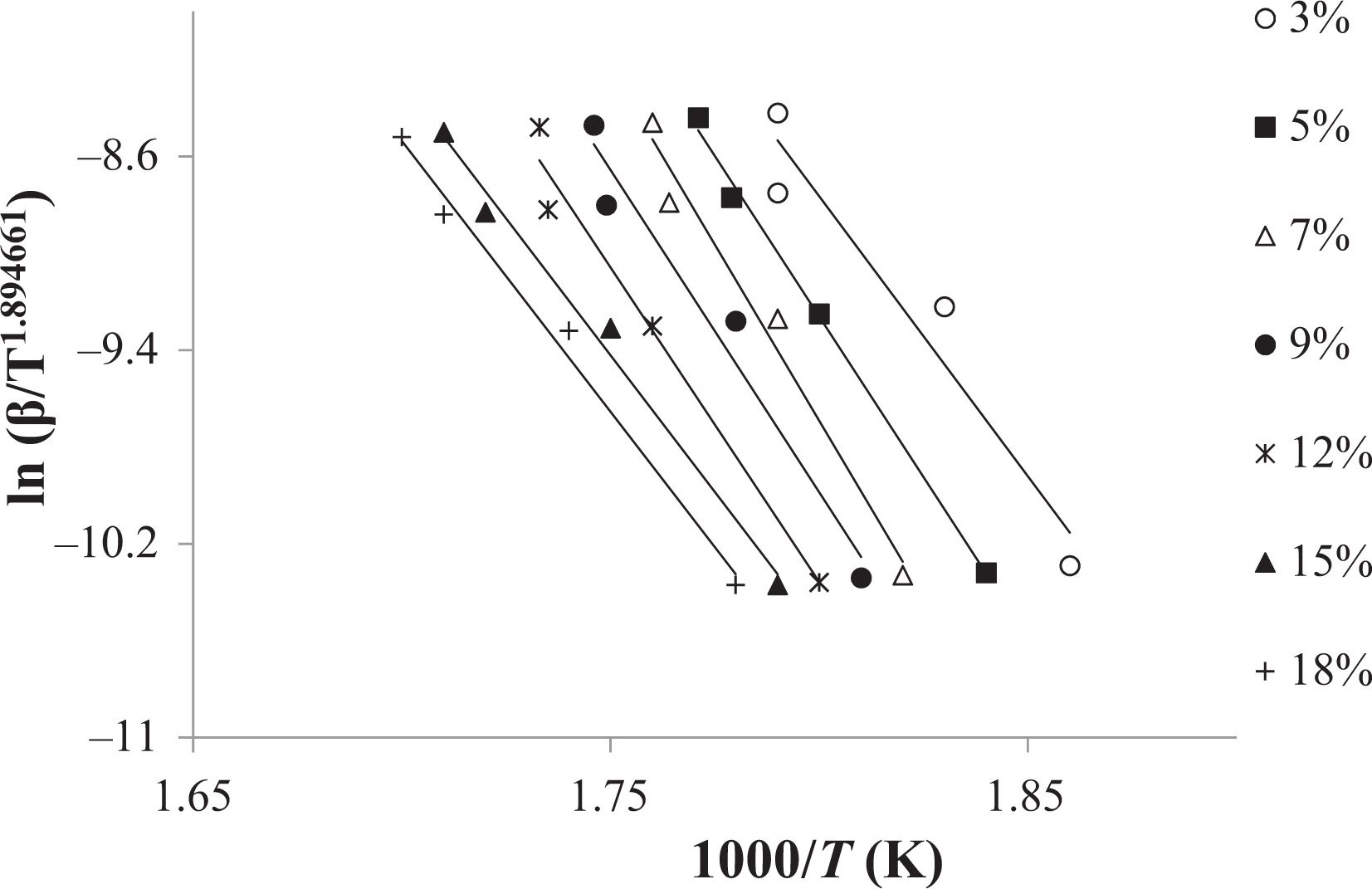
Tang plots of coumarin-based random copolymer at different degrees of conversion.
Algebraic expressions of g(α) for the most frequently used mechanisms of the solid-state processes.

E a of coumarin-based random copolymer calculated by the CR method for the solid-state processes at various heating ratios (from 5°C min−1 to 35°C min−1).
CR: Coats–Redfern; Ea: activation energy.
HOMO and LUMO analysis
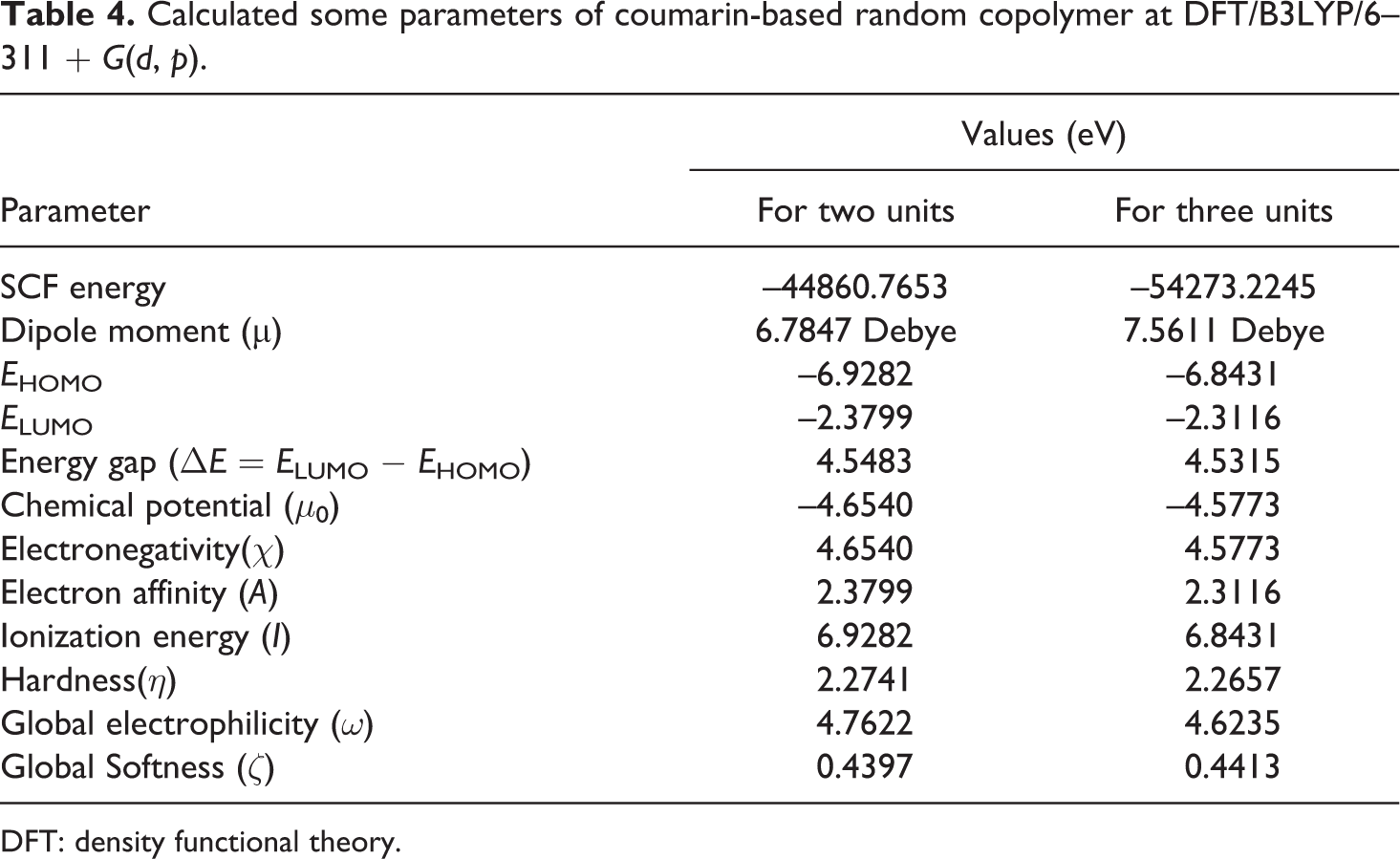
The optimized structure of random copolymer is demonstrated in Figure 3 with proper atomic labeling. The molecular orbitals are calculated with the optimized geometry. HOMO and LUMO are the main orbitals playing a part in chemical reactions and very useful for chemists and physicists. These orbitals indicate the path the molecule interacts with other species and are very important parameters for quantum chemistry that helps to illustrate the kinetic stability and chemical reactivity of the molecules. 35,36 The energy of LUMO relevant to electron affinity and the related orbital acts as an electron acceptor, while the energy of HOMO directly relevant to the ionization potential and the related orbital acts as an electron donor. 37 The 3-D drawing of MOs (HOMO and LUMO) mapped with molecular orbitals for both two and three units is demonstrated in Figure 4. As it can be seen in Figure 4 for both, the positive phase is bluish green and the negative one is greenish. Moreover, it can be seen from Figure 4, the energy gap of HOMO–LUMO decreases from two units to three units and is 4.5483 eV and 4.5315 eV, respectively. The energy gap between HOMO and LUMO, dipole moment, electronegativity, softness, chemical hardness, electrophilicity index, electron affinity, and ionization potential has been calculated with B3LYP/6–311 + G(d, p) level in the gas phase. The obtained data are listed in Table 4. The quantum chemical descriptors have been used in the DFT theory of chemical reactivity for defining the reactive sites of the molecular systems and are connected with the electronic structure and the mechanism which play a part in the covalent bond formation as a result of reaction between the electrophiles and the nucleophiles. 38 Energies of LUMO and HOMO have been used to calculate global chemical reactivity descriptors, such as electronegativity, chemical hardness, chemical potential, global softness, and global electrophilicity index. 39 –41
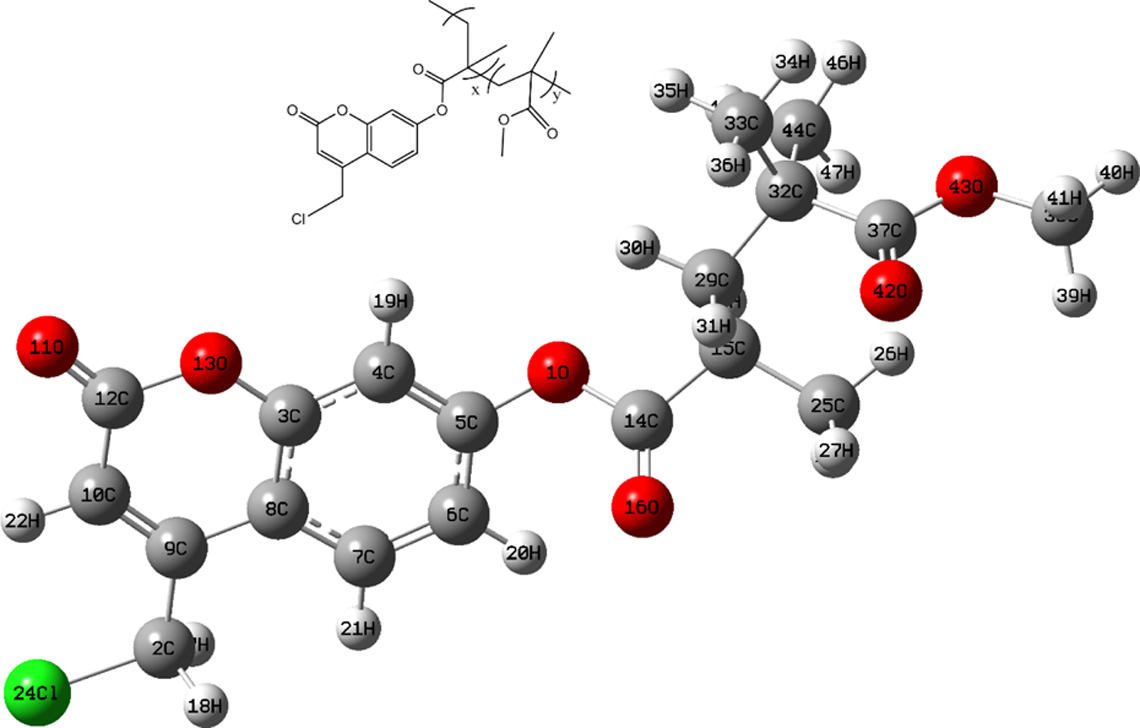
Optimized molecular structure of the coumarin-based random copolymer assuming x and y = 1. The red, gray, white, and green spheres correspond to oxygen, carbon, hydrogen, and chlorine atoms, respectively.
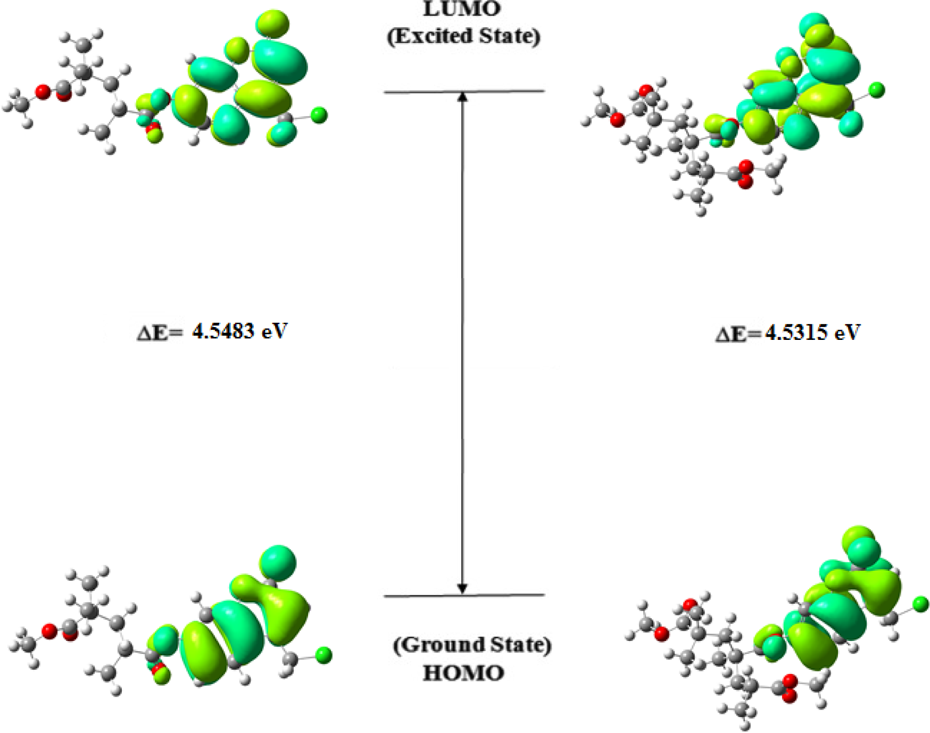
Optimized HOMO and LUMO orbitals of coumarin-based random copolymer (for two and three units, respectively).
Calculated some parameters of coumarin-based random copolymer at DFT/B3LYP/6–311 + G(d, p).
DFT: density functional theory.
Using Koopmans’ theorem, these chemical descriptors are calculated as follows:
Chemical hardness (η) can be expressed as:

Softness (ς) can be expressed as:

Chemical potential (µ) can be expressed as:

Electronegativity (χ) can be expressed as:
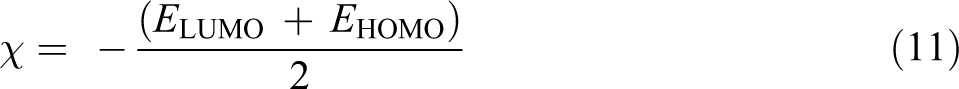
Global electrophilicity (ω) can be expressed as:

The larger energy gap between LUMO and HOMO for the two units (E
LUMO − E
HOMO = 4.5483) as compared to three units (E
LUMO − E
HOMO = 4.5315) suggests that three units is less reactive than two units. Further, from Table 4, it can be seen that higher values of the global electrophilicity index for the two units (
Thermodynamic properties
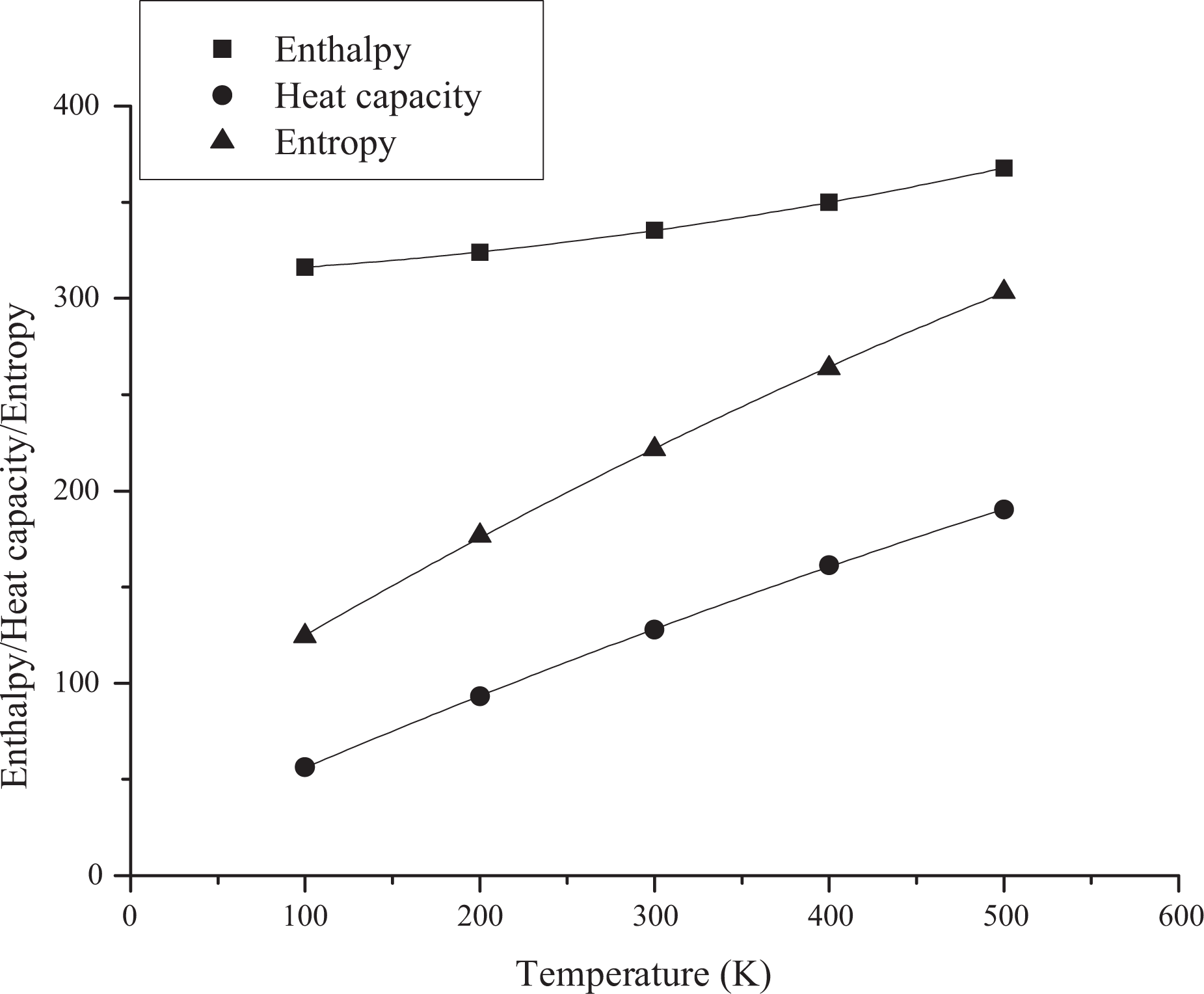
Based on vibration analysis, the statistical thermodynamic functions such as entropy (S), specific heat capacity (C), and enthalpy (H) and some thermodynamic parameters such as rotational constants, the zero–point vibrational energies, and rotational temperatures, which obtained theoretical harmonic frequencies of random copolymer (for two and three units) were calculated by B3LYP/6–311 + G(d, p) and the results at 298.15 K in ground state were presented in Tables 5 and 6. According to Table 6, because of the molecular vibrational intensities increase with temperature, the thermodynamic functions are increasing with temperature ranging from 100 K to 500 K. The correlation equations between heat capacity, entropy changes, enthalpy, and temperatures are fitted by quadratic formulas. The relevant correlation graphs for two units of random copolymer are as demonstrated in Figure 5 and the corresponding fitting equations are as follows:
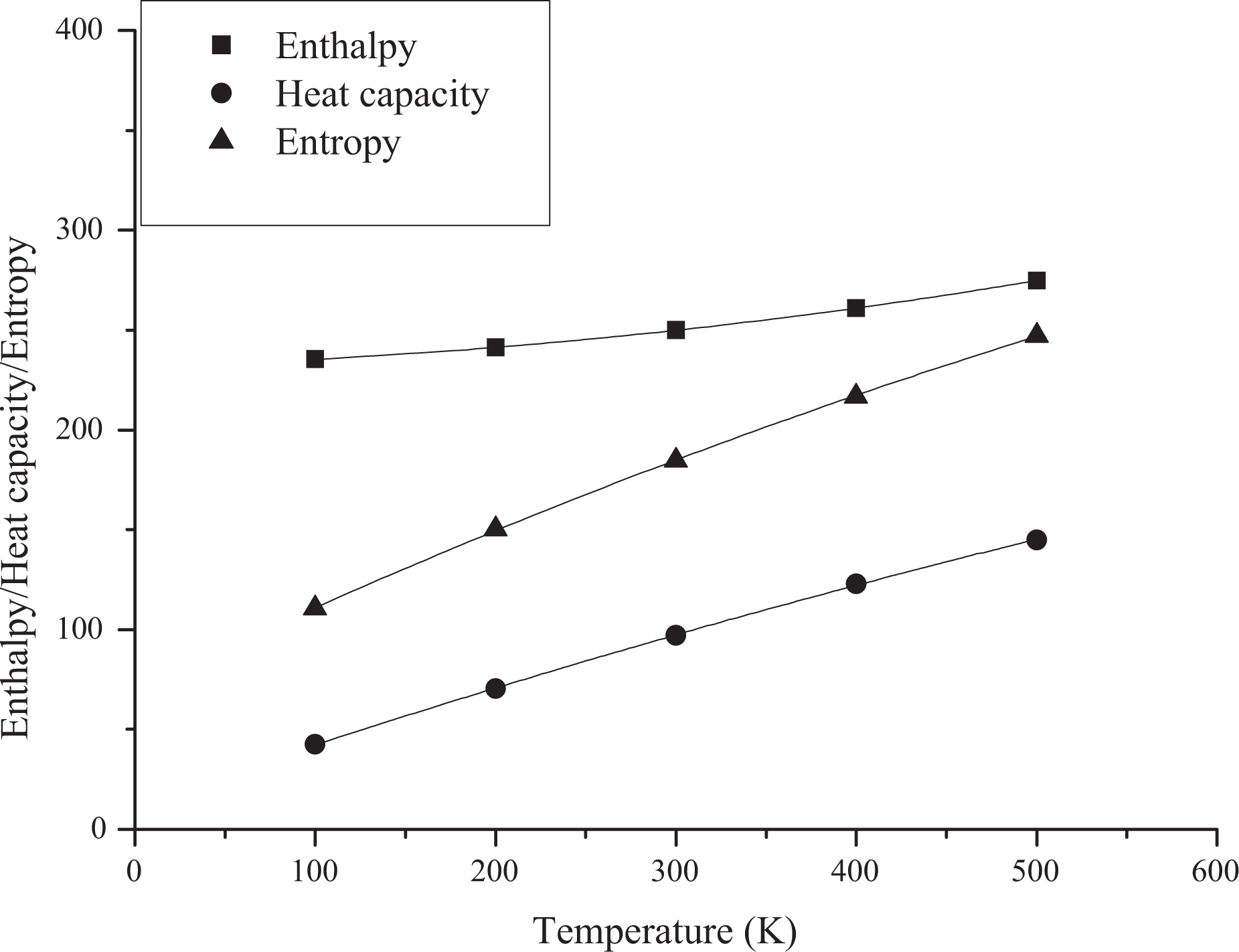
Correlation graphic of thermodynamic parameters and temperature for coumarin-based random copolymer (for two units).
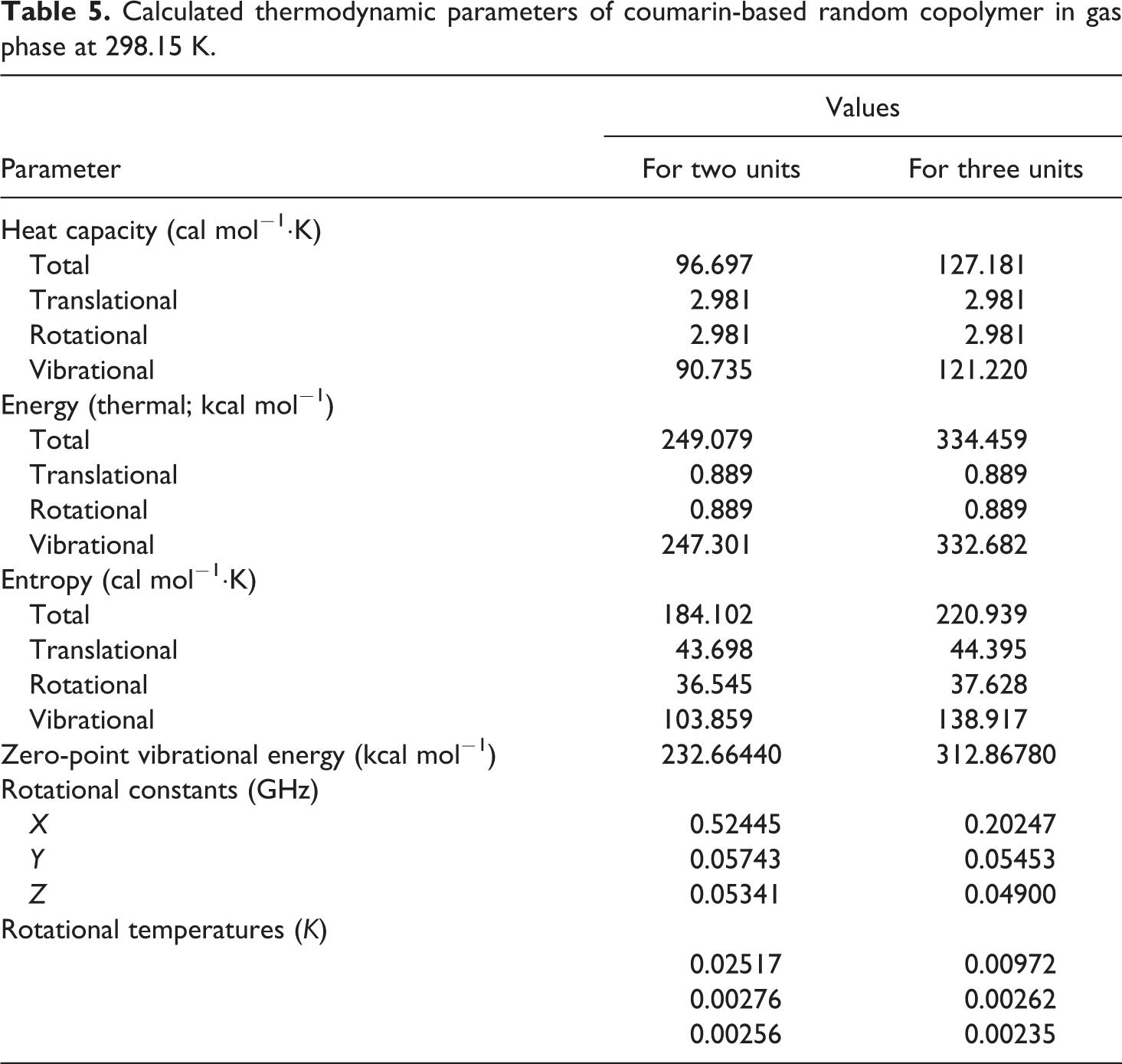
Calculated thermodynamic parameters of coumarin-based random copolymer in gas phase at 298.15 K.
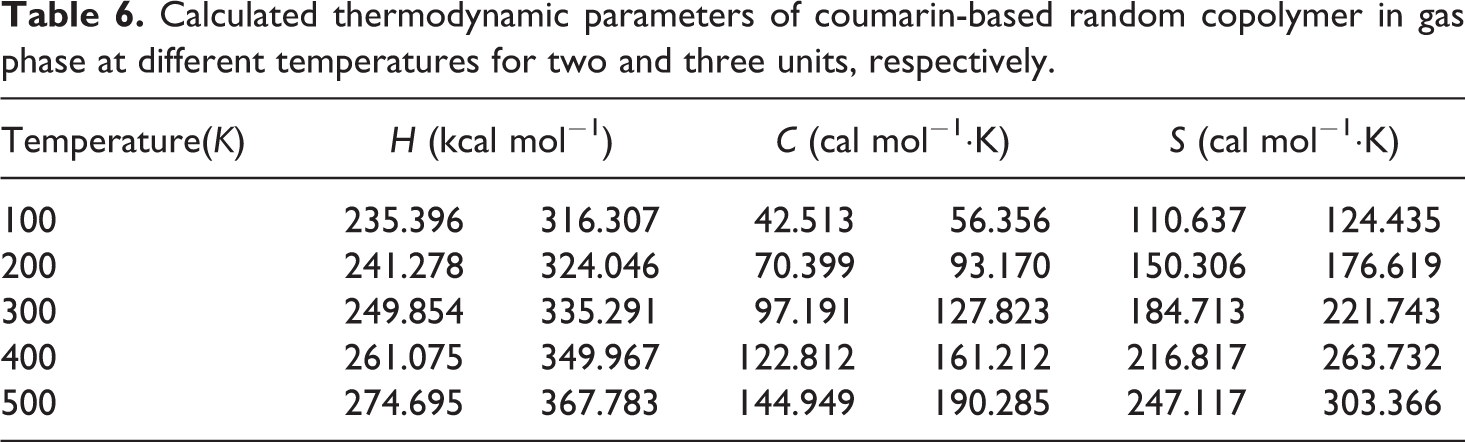
Calculated thermodynamic parameters of coumarin-based random copolymer in gas phase at different temperatures for two and three units, respectively.
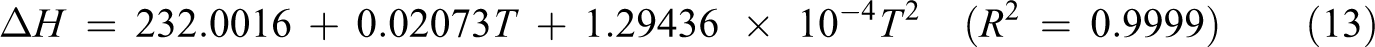
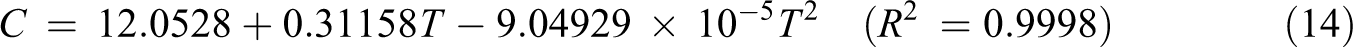
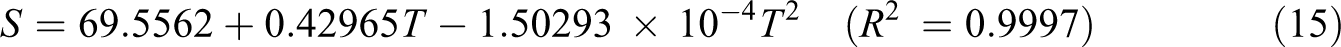
The relevant correlation graphs for three units of random copolymer are as demonstrated in Figure 6 and the corresponding fitting equations are as follows:
Correlation graphic of thermodynamic parameters and temperature for coumarin-based random copolymer (for three units).
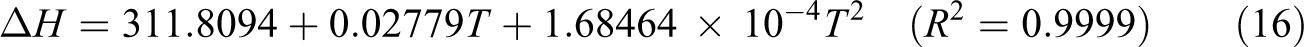

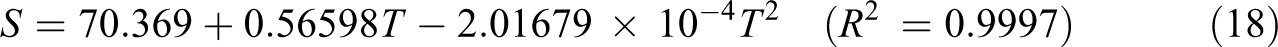
The related fitting factors (R 2) of entropy, heat capacity, and enthalpy for both two and three units (random copolymer) are 0.9998, 0.9997, and 0.9999, respectively. It is observed from Table 6 and the related fitting equations that the value of these thermodynamic properties is increased with both temperature and when one passes from two to three units. All the thermodynamic data provide useful information for further research on the coumarin-based random copolymer. These parameters can be utilized to predict reaction mechanism and compute the other thermodynamic energies related to thermodynamic functions.
Molecular electrostatic potential surfaces analysis
The molecular electrostatic potential (MEP) is a very important feature and used to guess molecular reactive regions and widely used as a reactivity surface exhibiting most possible regions for the electrophilic attack. A 3-D plot of MEP and electrostatic potential of random copolymer is demonstrated in Figure 7. The potential has been especially helpful as an indicator of the sites or regions of a molecule. Electrostatic potential is connected with electronegativity, dipole moment, partial charges, and region of chemical reactivity of the molecule. Such surfaces indicate the size, shape, region, and charge density of chemical reactivity of the molecules.
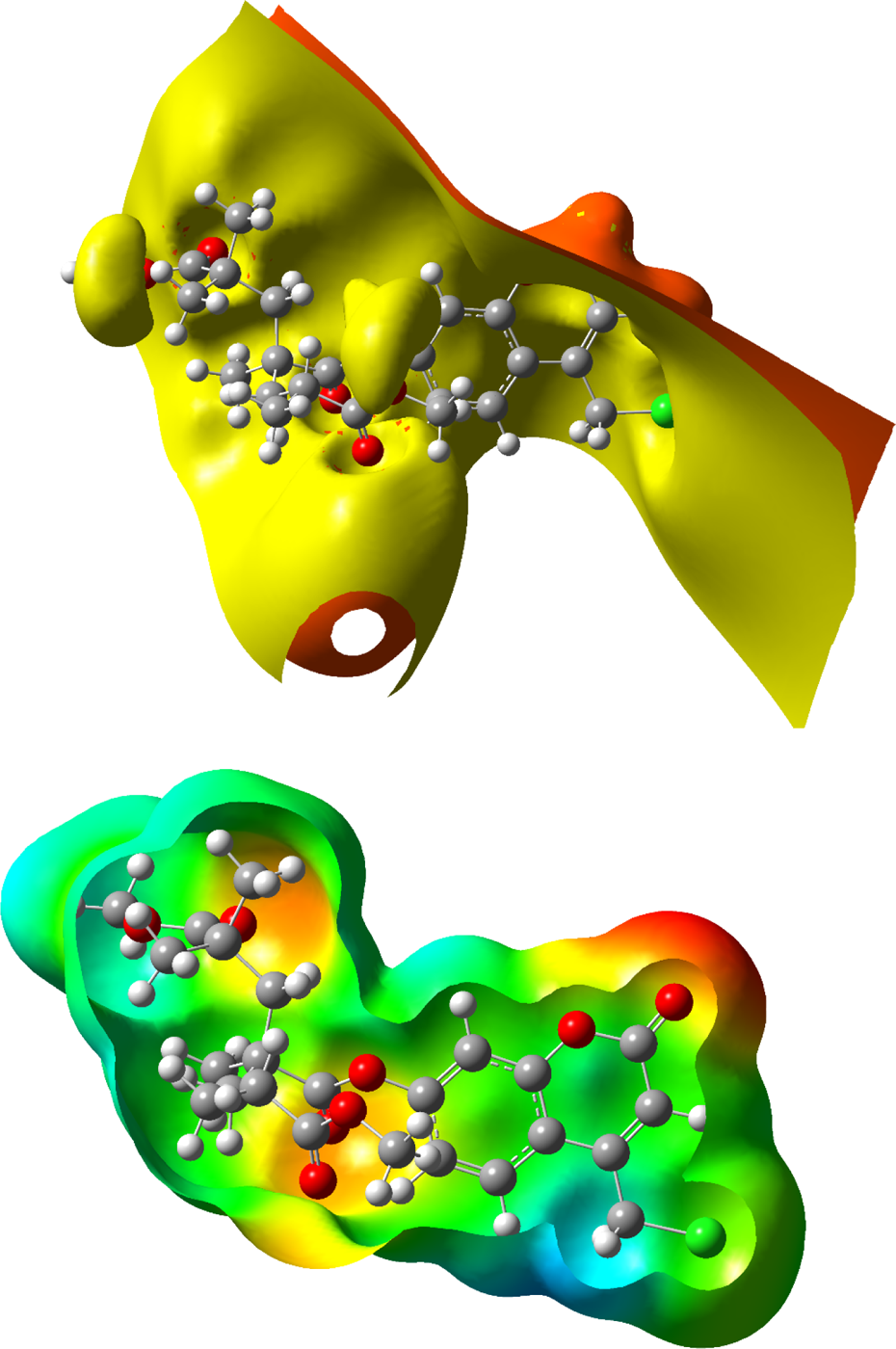
Electrostatic potential surfaces of coumarin-based random copolymer (for three units).
The different colors on the map result from different values of the electrostatic potential. The electrostatic potential increases in the order red (electron rich region and partly negative charge) < orange (less electron rich region) < yellow (slightly electron rich region) < green (neutral) < blue (electron deficient and partly positive charge). The maximum positive regions are nucleophilic regions and seen as blue color, and the maximum negative regions are electrophilic regions and seen as red color. Therefore, molecular electrostatic potential (Figure 7, below) clearly indicates the presence of high negative charge on the oxygen atoms. Furthermore, we can say that the negative electrostatic potential (ESP) is seen as a reddish or a yellowy blob and is located more over the oxygen atoms (Figure 7, top). The contour maps such as positive, negative, and both charges of random copolymer are demonstrated in Figure 8. As shown in Figure 8, the contour map supports the negative and positive potential regions of the random copolymer in accordance with the molecular electrostatic potential surfaces. MEP, V(r) at a given point r(x, y, z) in the vicinity of a molecule, is described as the interaction energy between a positive test charge (a proton) located at r and the electrical charge generated from the molecule nuclei and electrons. For the system worked, the V(r) values are computed by the following equation 42 :
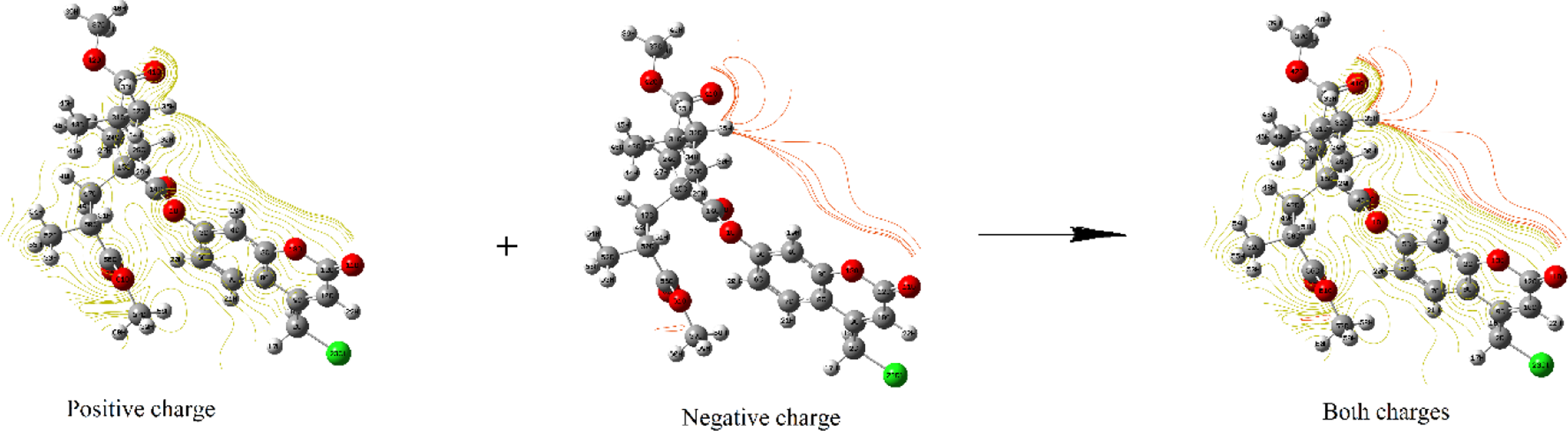
Molecular electrostatic potential contour surface of coumarin-based random copolymer (for three units).
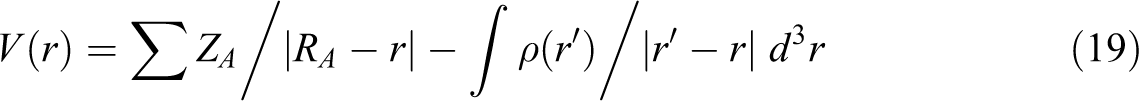
where
Conclusions
DFT calculations were used for the two and three units which can take part as repeating units in the random copolymer. Firstly, the molecular structure of random copolymer was determined theoretically using the DFT calculations. Molecular electrostatic potential, electrostatic potential, and contour surfaces, which help in the determination of electrophilic and nucleophilic sites in the molecule as well as possible reaction path, are determined. The electronic and thermodynamic properties for two and three units of random copolymer were calculated. These quantum chemical calculations may help to understand the activity and properties of the coumarin-based random copolymer. Secondly, the thermal decomposition kinetic analyses were performed using the CR, Tang, and FWO methods. The thermal decomposition mechanisms for random copolymer obeyed a deceleration R 3 type, which is a solid-state process based on phase boundary model (contracting volume).
Footnotes
Acknowledgements
The author is grateful to Prof. Dr. Kadir Demirelli and Bitlis Eren University for supporting the Gaussian software of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.