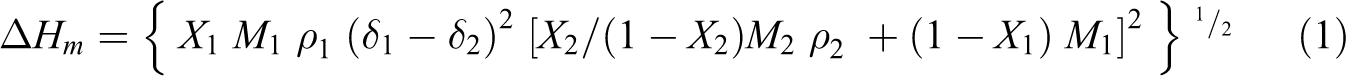Abstract
The main aim is to investigate the effect of adding of 5% hydrolyzed poly (ethylene-co-vinyl acetate) (EVAOH), itaconic anhydride (naturally obtained during fermentation of sugars (It.anhydride)), and mercapto-modified ethylene-co-vinyl acetate (EVASH) as compatibilizers on the mechanical properties before and after thermal oxidative aging at 90°C for different time periods up to 7 days, electrical properties, and morphology of ethylene propylene diene monomer/ethylene vinyl acetate (EPDM/EVA) blends of different compositions (75/25, 60/40, 50/50, and 25/75, respectively). Stress at yield, tensile strength, and elongation at break of EPDM/EVA were improved markedly by the addition of 5% compatibilizers (EVAOH, It.anhydride, and EVASH). The micrographs of fractured surfaces obtained from the scanning electron microscope demonstrated further the enhancement of compatibility between EPDM and EVA by the addition of reactive compatibilizers. Further, the blends permittivity ε′ and dielectric loss ε″ have been measured at specific temperatures. The dielectric properties revealed that It.anhydride and EVASH/EPDM/EVA compositions are promising materials for insulation purposes. Finally, aging resistance was improved by the use of compatibilizers.
Introduction
New materials with desirable physical properties are used for the blending of rubber–plastic. The component polymers (rubber–plastic) and their ratios play an important role in the desired properties of blends. 1 –3 Most of the rubber/thermoplastic blends are incompatible and immiscible. Immiscible blends often have separated phase morphology, poor physical and chemical interactions, and low mechanical properties than those of their individual components. 4 –6 Different techniques can be used to overcome these restrictions. With the aid of a third component “interfacial agent” or “compatibilizer,” interfacial energy between the phases can be reduced, which allowed a finer dispersion at some stage in blending and improved the interfacial interaction. 7 –9
Ethylene propylene diene monomer (EPDM) elastomer is used in electrical and construction industries because of its unique mechanical and chemical properties. 6,10 EPDM exhibits splendid resistance to climate, ozone, acids, and alkalies and maintains acceptable mechanical properties. But, when it is exposed to solvents and oil, it performed poorly. Blending of EPDM with high-diene rubbers draws masses of interest, as such blends may also offer a broader range of applications. 11
Ethylene vinyl acetate (EVA) offers excellent heat resistance, oil resistance, elasticity, good processing technology, and cheap elastomer. The blending of EPDM and EVA leads to a large number of applications. Several papers concerning EVA and rubber blends have been studied. 12
Except polychloroprene/EVA blends, all blends of EVA with rubber are incompatible. This is due to the strong differences in polarity and chemical structure.
Using a small quantity of appropriated mercapto-functionalized copolymer, high-diene rubbers can be compatibilized with other components. 13,14 The usage of mercapto-modified ethylene-co-vinyl acetate copolymer (EVASH) for EVA/rubber blends improved the interfacial adhesion of the blend. This is attributed to the ability of mercapto groups to react with the double bond of the high-diene rubber.
Dielectric properties of polymer blends depend on morphology structure and phase separation. The permittivity is found to increase by increasing the number of dipoles. The dielectric properties of heterogeneous polymer blends are affected by phase separation and interfacial polarization. This type of polarization arises from the difference in conductivity and permittivity of the individual components. 14,15 This article aims to study the mechanical, morphology, and dielectric properties to investigate the compatibility of EPDM/EVA blends of different compositions with hydrolyzed poly (ethylene-co-vinyl acetate) (EVAOH), Itaconic anhydride (It.anhydride), and EVASH as compatibilizers.
Materials and experimental
EPDM, with ethylene content of 55% and density of 0.86 g cm−3, was manufactured by Esso Chemi, Germany.
EVA copolymer in granular (28% and 40% VAc) form was obtained from DuPont Ltd. (USA). 3-mercaptopropionic acid, It.anhydride, and dicumyl peroxide were supplied by Sigma-Aldrich, Germany.
Synthesis of EVAOH and EVASH
Hydrolysis of EVA copolymer was carried out as per procedure reported by Hirata et al. 16 The reaction was carried out in three-necked round bottom flask equipped with a reflux condenser and stirrer. Fifty grams of EVA was dissolved in tetrahydrofuran. To the reaction mixture, measured volume of 0.5 M alcoholic NaOH was added (Figure 1). The reaction mixture was refluxed under a nitrogen atmosphere for 1 h. After completion of the reaction, the medium was neutralized by 1 N HCl solution. The reaction mass was then precipitated in distilled water. The precipitate was washed repeatedly using distilled water to remove inorganic salt and dried at 80°C. Mercapto groups is introduced by esterifying the hydrolyzed EVA copolymer with mercaptoacetic acid in toluene solution under reflux. EVASH sample was obtained using an appropriate amount of the mercapto acid, as shown in Figure 1. 17 The products were characterized by Fourier transform infrared (FTIR) spectroscopy.
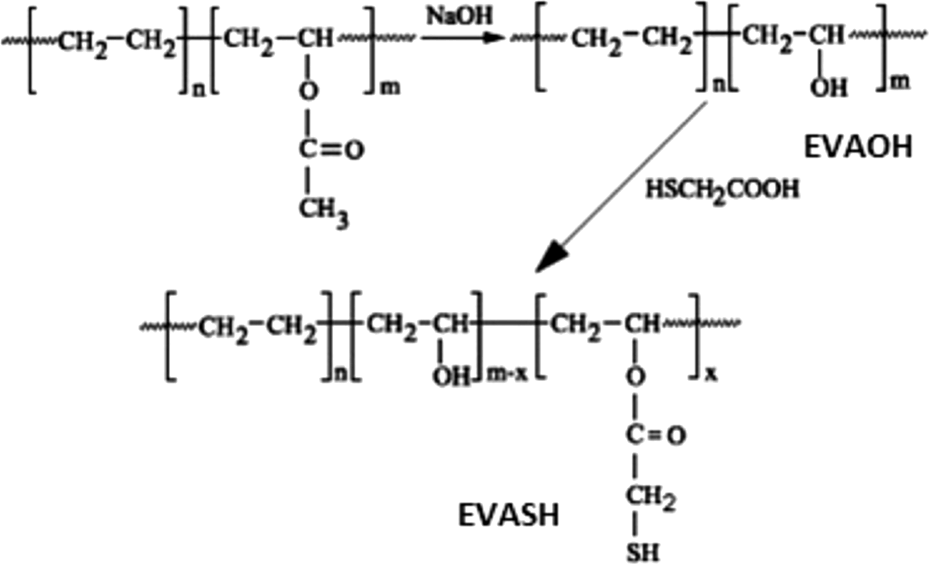
Synthesis of EVAOH and EVASH. EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
Blend preparation
Blends were prepared in the Brabender Plasticorder (C. W. Bra, Instrument, Inc., Hackensack NJ; 230 V, 40 A) at 80°C, 70 r min−1 for 5 min. EPDM was blended with different compatibilizers (EVAOH, It.Anhydride, and EVASH) for 3 min in order to promote interaction between functional groups present in compatibilizers and the double bond of the EPDM. Then EVA was added and blended for 7 min. The peroxide (3 phr) added to the mixture acts as a cross-linking agent on a laboratory two-roll mill of outside diameter 470 mm, working distance 300 mm, speed of the slow roll 24 r min−1, and friction ratio 1.4:1. The compounded blends were vulcanized in a clean polished mold in a hydraulic press under a pressure of about 40 kg cm−2 for 10 min at a temperature of 152 ± 1°C.
Techniques
The FTIR spectra of samples were reordered on a Jasco (Japan) FTIR 430 series infrared spectophotometer equipped with KBr disks.
The surface morphology of the samples was investigated by environmental scanning electron microscope (SEM) (Quanta FEG-250).
The tensile properties were determined at room temperature (25°C ± 1°C) using a Zwick tensile testing machine (model-1425; Munich, Germany) according to ASTM D 412-06a. The samples were cut with a dumbbell-shaped cutter. Cross-sectional areas of the dumbbell specimens were measured accurately with the aid of a thickness gauge. The mean of five readings was taken into consideration.
Thermal oxidative aging was carried out at 90 ± 1°C in an air circulating oven for different time periods up to 7 days according to ASTM D 572-04, 2010. The reported results were averaged from a minimum five specimens.
The dielectric properties and conductivity measurements were carried out by means of high-resolution broadband impedance analyzer (Solartron TM 1260-Schlumberger Ltd, UK) interfaced with a PC. The frequency range of the applied ac electric field was between 0.1 Hz and 1 MHz. Good electromagnetic shielding was implemented to the whole sample holder in order to diminish noise problems that are common especially at low frequencies. The measurements were automated by interfacing the impedance analyzer with a personal computer through a GPIB cable IEE488. A commercial interfacing and automation software LabVIEW [Version 7.1 -NI “National Instruments (c) 2004 ni.com/labview”] was used for acquisition of data. Prior to the sample measurements, the calibration was performed to eliminate the effect of stray capacitance. The percent of error in permittivity ε′ and dielectric loss ε″ amounts to 1% and 3%, respectively. The temperature of the samples was controlled by a temperature regulator with Pt100 sensor. The percent of error in temperature measurements amounts 0.5°C. To avoid moisture, the samples were stored in desiccators in the presence of silica gel. Thereafter, the sample was transferred to the measuring cell and left with P2O5 until the measurements were carried out.
Results and discussion
Characterization of EVAOH and EVASH
Figure 2 shows FTIR spectra of EVAOH and EVASH. From this figure, the absorption bands of acetate and hydroxyl groups were appeared. The intensity of the acetate group is decreased, while OH (3200–3400 cm−1) is increased (Figure 2(a)).On the other hand, there is an increase in the intensity of the carbonyl band at 1736 cm−1 and the decrease of OH absorption intensity at 3800–3500 cm−1 (Figure 2(b)). This investigation may be due to the mercaptoacetic acid incorporation. These results confirmed esterification and take a suitable analytical method to quantify the vinyl mercaptoacetate in EVASH copolymers.
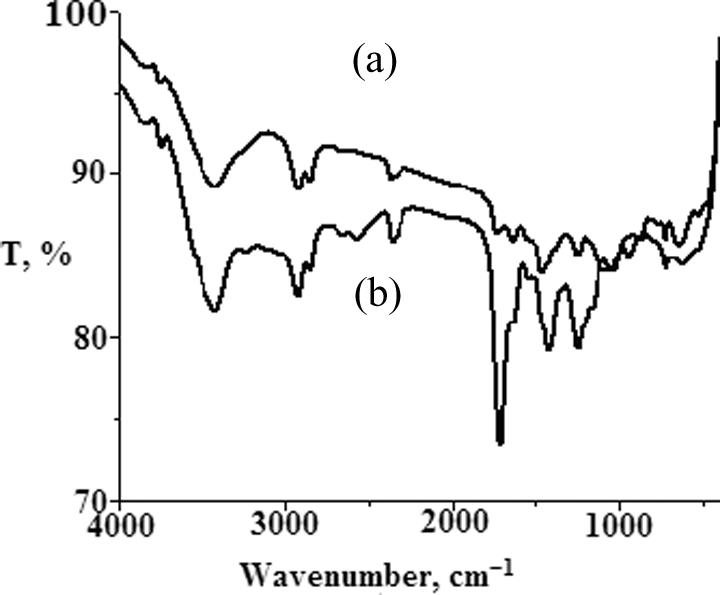
FTIR spectra of (a) EVAOH and (b) EVASH. FTIR: Fourier transform infrared; EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
Heat of mixing
The compatibility of EPDM/EVA blends has been measured by the theoretical calculations of the heat of mixing according to Schneier’s equation, 18,19 which is given as
where X, ρ, and M are the weight fraction of polymer, density, and molecular weight of the monomer unit, respectively, δ is the solubility parameter of the polymer. When the value of the heat of mixing of two polymers is under the value 41.85 × 10−3 J mole−1, they are considered to be compatible, but polymer pairs are considered to be incompatible if the value of heat of mixing is above this limit as in previous work. Figure 3 shows the relation between the composition of EPDM and EVA and values of heat of mixing. From this figure, it is obvious that EPDM/EVA blends are thermodynamically incompatible because the values of heat of mixing for all compositions lie above the limit of compatibility.
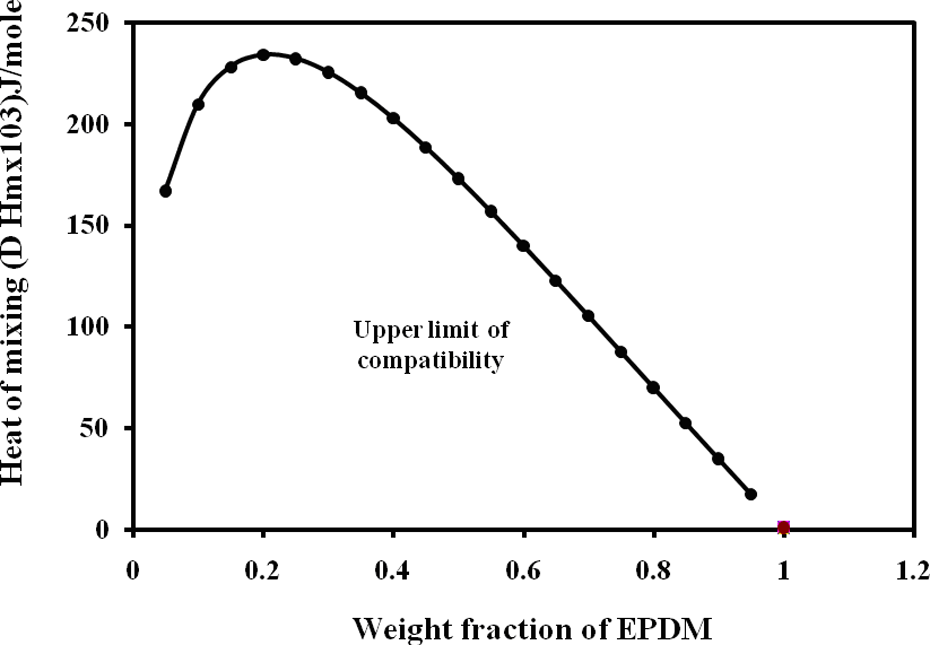
Heat of mixing as a function of composition in blend of EPDM/EVA. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
Morphology of EPDM/EVA blends
Figure 4 shows the micrographs of statically vulcanized dicumyl peroxide (DCP) EPDM/EVA (75/25, 60/40, 50/50, and 25/75) obtained from the SEM. It is seen that the EVA particles dispersed randomly as domains in EPDM (Figure 4(a)). On the other hand, the phase morphology was changed by increasing the EVA content from 40% to 75% (Figure 4(b) to (d)). As the EVA content increases, the domains size of EVA decreases and EPDM morphology increases. 6

Scanning electron micrographs of statically vulcanized (DCP) EPDM/EVA blends. (a) 75/25 EPDM/EVA blend, (b) 60/40 EPDM/EVA blend, (c) 50/50 EPDM/EVA blend, and (d) 25/75 EPDM/EVA blend. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
This observation indicates that these blends are heterogeneous (incompatible).
Mechanical properties of EPDM/EVA blends
The variation of mechanical properties such as stress at yield σ B, tensile strength σ R, and elongation at break ε R as a function of weight percentage of EVA before and after thermal oxidative aging for different time periods was determined and collected in Table 1. It is clear that both σ B and σ R increase with the increasing percent of EVA because ethylene chains of EVA can crystallize in EPDM/EVA blend and increase blend rigidity. Also, the increasing of EVA content in the blends increases the elongation at break due to the decreasing of the stiffness of the blend. After thermal oxidative aging at 90°C, little reduction in mechanical properties compared to the original ones after aging for 3 and 7 days can be observed, indicating rubber degradation, as expected.
Blend compositions and mechanical properties of EPDM/EVA before and after thermal oxidative aging at 90°C.
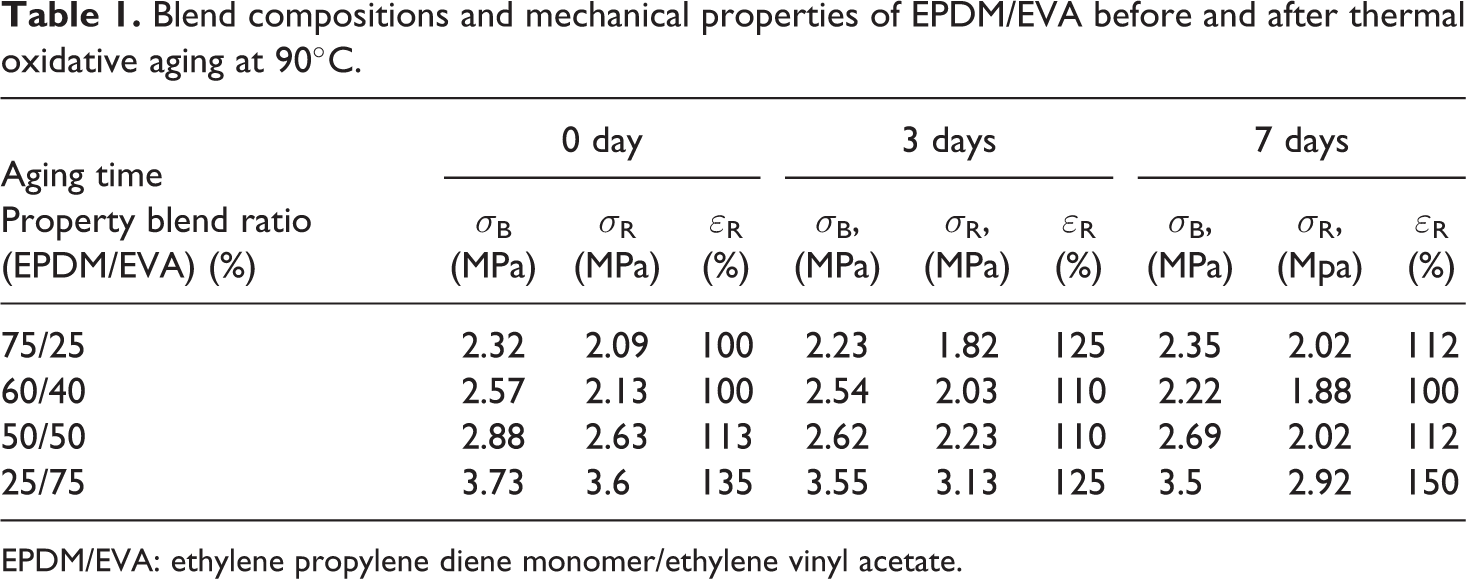
EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
Effect of compatibilizers on the properties of EPDM/EVA blends
Mechanical properties
The effectiveness of the addition of compatibilizers (5% of EVAOH, It.anhydride, and EVASH) on tensile properties (σ B, σ R, and ε R) for the blends with different compositions was studied before and after thermal aging at 90°C for different time periods up to 7 days (Table 2). Figures 5 to 7 represent the dependence of tensile properties of EPDM/EVA blends on the addition of 5% compatibilizers. One can be seen that the compatibilizing agents improve the ultimate tensile properties (σ B, σ R, and ε R) due to the enhancement of interfacial adhesion between EPDM and EVA. The EVAOH and It.anhydride systems show significantly higher σ B, σ R, and ε R for blend which contains 40–75% of EVA. This behavior suggests interactions between EVAOH and It.anhydride and EVA blend component. On the other hand, the EVASH system gives best results for the blends containing 40–75 phr of EPDM. This may be attributed to the presence of double bond in the blend (EPDM and EVA), which reacts with SH (mercapto) groups presented in EVASH during the mixing process. 13
Effect of functionalized EVA (EVAOH, EVASH) and its anhydride on the mechanical properties of EPDM/EVA blends before and after thermal aging at 90°C.
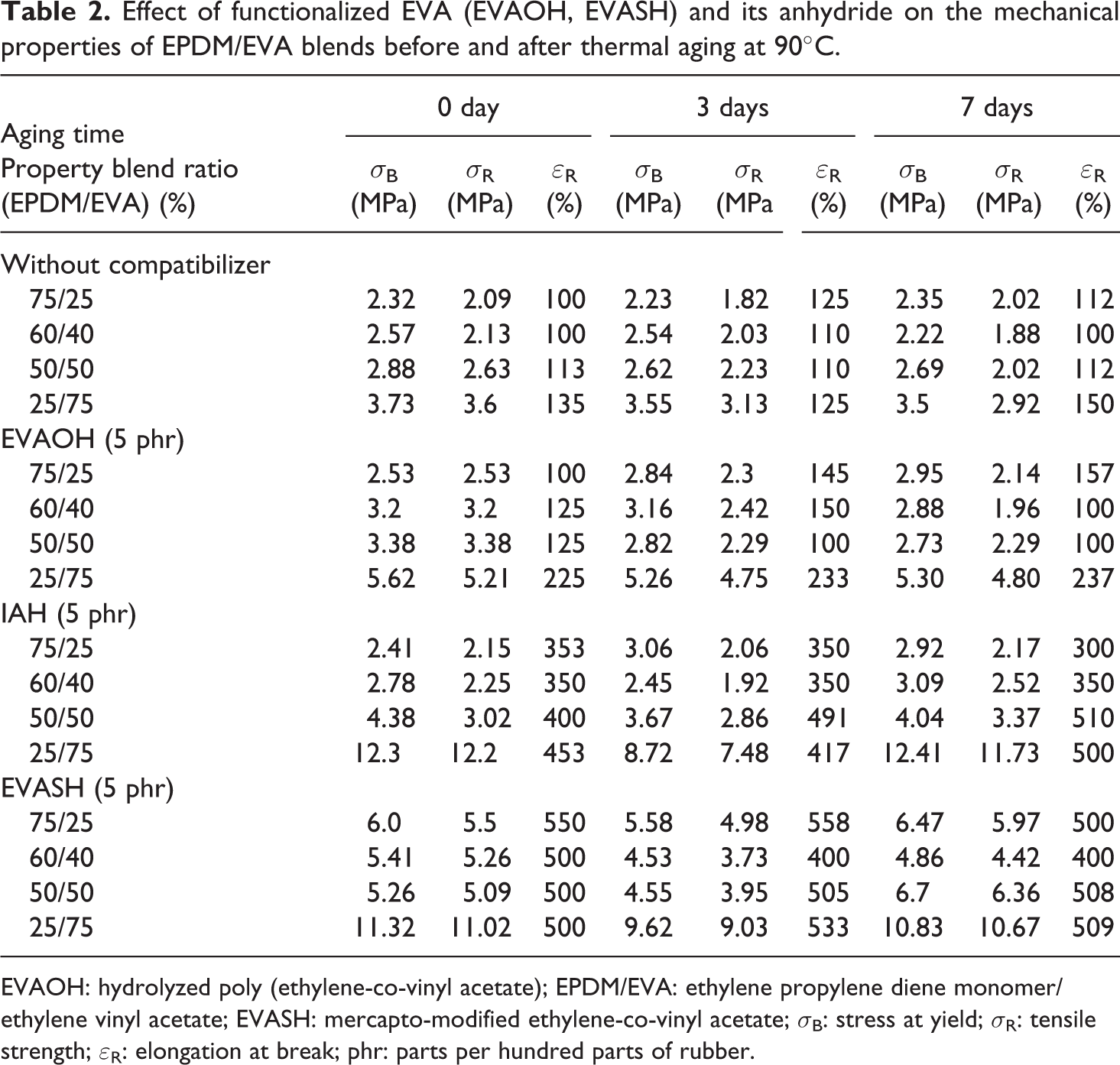
EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate; EVASH: mercapto-modified ethylene-co-vinyl acetate; σ B: stress at yield; σ R: tensile strength; ε R: elongation at break; phr: parts per hundred parts of rubber.
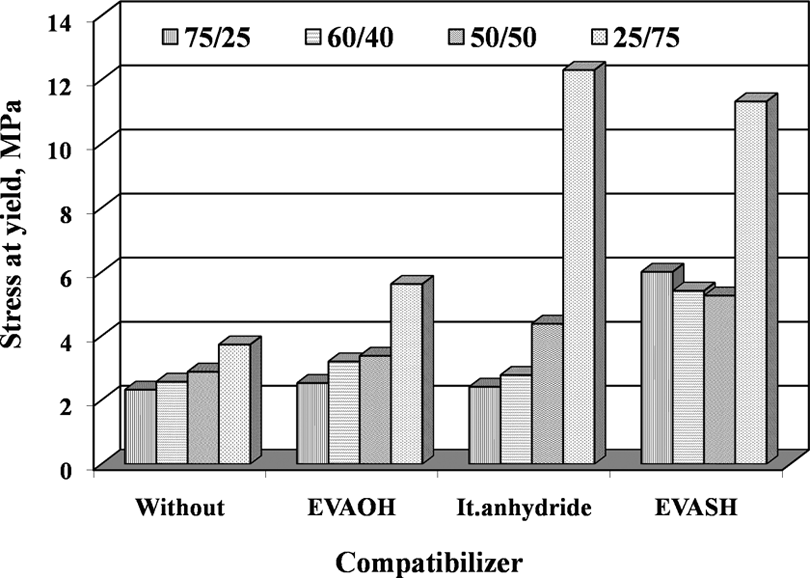
Stress at yield of vulcanized EPDM/EVA blends with and without compatibilizer (EVAOH, It.anhydride, and EVASH). EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate; EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
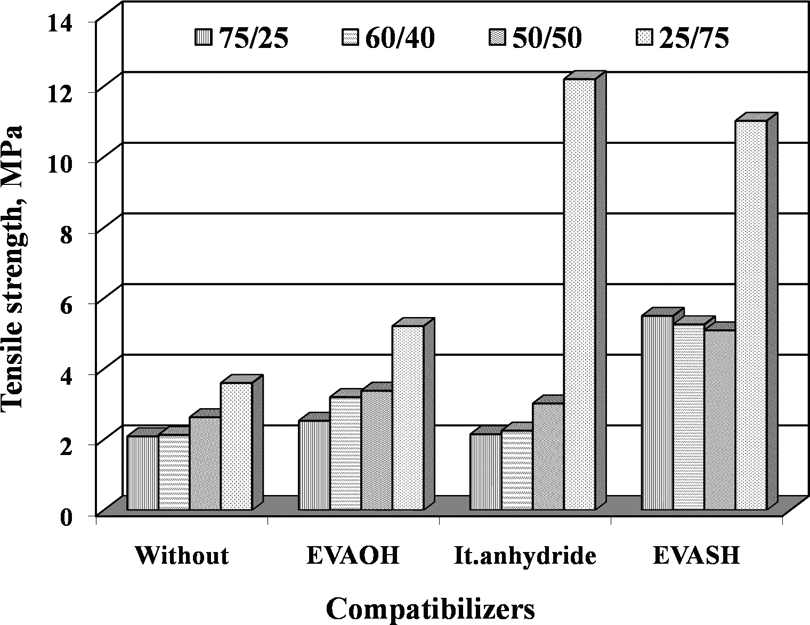
Tensile strength of vulcanized EPDM/EVA blends with and without compatibilizer (EVAOH, It.anhydride, and EVASH). EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate; EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
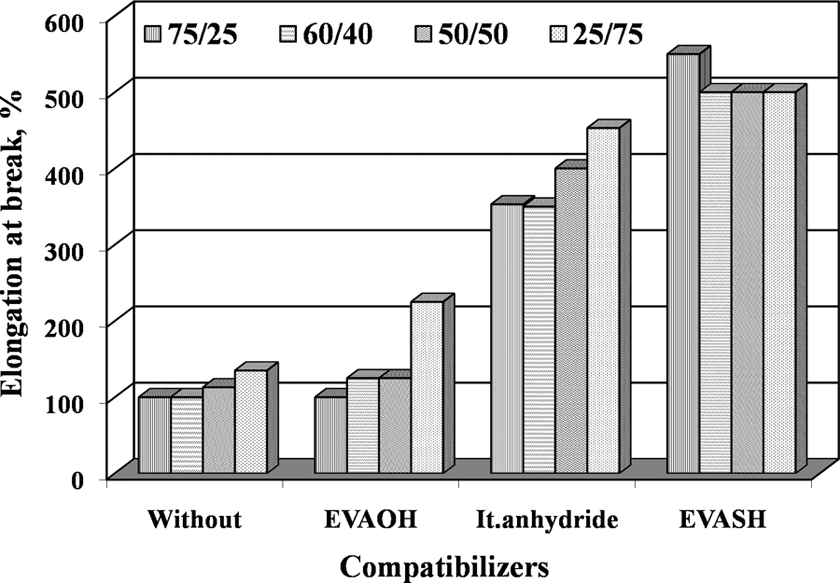
Elongation at break of vulcanized EPDM/EVA blends with and without compatibilizer (EVAOH, It.anhydride, and EVASH). EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate; EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
The tensile properties after thermal aging at 90°C for different period times of EPDM/EVA blends were measured and collected in Table 2. It is obvious the addition of 5% of EVAOH, It.anhydride, and EVASH to these blends leads to a little decrease in mechanical properties after 3 days, whereas after 7 days of aging, the mechanical properties are improved, indicating a post-curing process during aging.
Morphology of the compatibilized blends
The effect of the addition of 5% EVAOH, It.anhydride, and EVASH on the surface morphology of EPDM/EVA blends (75/25 and 25/75) were examined (Figure 8). From this figure, it is clear that, upon introduction of compatiblizers, a fine morphology is observed for the blend samples in Figure 8(a), (b), (c), (e), and (f), respectively. The surface appeared to be smooth and uniform indicating the compatibility between EVA and EPDM compared to those without compatibilizer (Figure 4). The addition of small molecules of (EVAOH, It.anhydride, and EVASH) compatibilizers can weaken the hydrogen bond interactions so that the interfacial adhesion is enhanced, indicating improvement in compatibility between EPDM and EVA. In other words, the addition of EVAOH, It.anhydride, and EVASH compatibilizers leads to improvement in the coherency between EPDM and EVA.

Micrographs of EPDM/EVA blends of different concentrations (a) 75/25/5 EVAOH, (b) 75/25/5 It.anhydride, (c) 75/25/5 EVASH, (d) 25/75/5 EVAOH, (e) 25/75/5.5 It.anhydride, and (f) 25/75/5 EVASH obtained from SEM. SEM: scanning electron microscope; EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate; EVAOH: hydrolyzed poly (ethylene-co-vinyl acetate); EVASH: mercapto-modified ethylene-co-vinyl acetate.
Dielectric properties of EPDM/EVA blend
Permittivity
The experimental data of the permittivity ε′ of different EPDM/EVA blend compositions at the fixed frequency (100 Hz) are depicted in Figure 9. It is obvious that ε′ increases with the increase of EVA loading in the blend. This is true for incompatibilized as well as for compatibilized blend compositions. This increase in ε′ is mainly due to the presence of polar EVA. The presence of polar groups enhances the orientation polarization in addition to interfacial polarization. However, the blend becomes more heterogeneous in nature at higher EVA content. This heterogeneity enhances the interfacial polarization and increases ε′ of the blend. 20
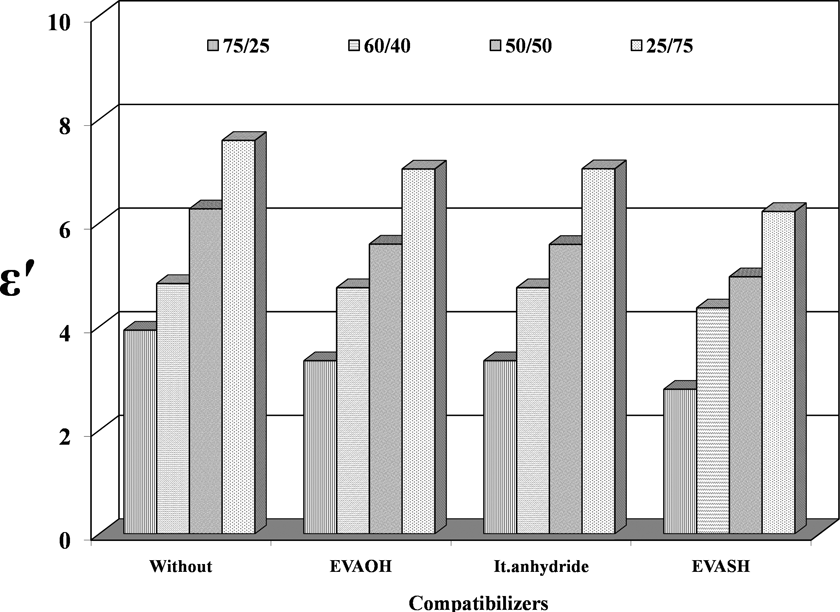
Permittivity (ε′) versus compatibilizers of different EPDM/EVA compositions at 30°C. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
On the other hand, compatibilized samples containing 5% of EVAOH, It.anhydride, and EVASH exhibit lower permittivity compared to incompatibilized blend compositions. This could be due to some sort of interaction between the two phases. These interactions reduce the polar groups of EVA.
Dielectric loss
The dielectric loss ε″ as a function of EVA content for incompatibilized and compatibilized blend compositions is illustrated in Figure 10. It is clear that the values of ε″ increase with the increase of EVA concentration. This is attributed to interfacial polarization and increase in conductivity of the blend. 21 EVASH blends show more insulative behavior compared to the other compatibilizers.
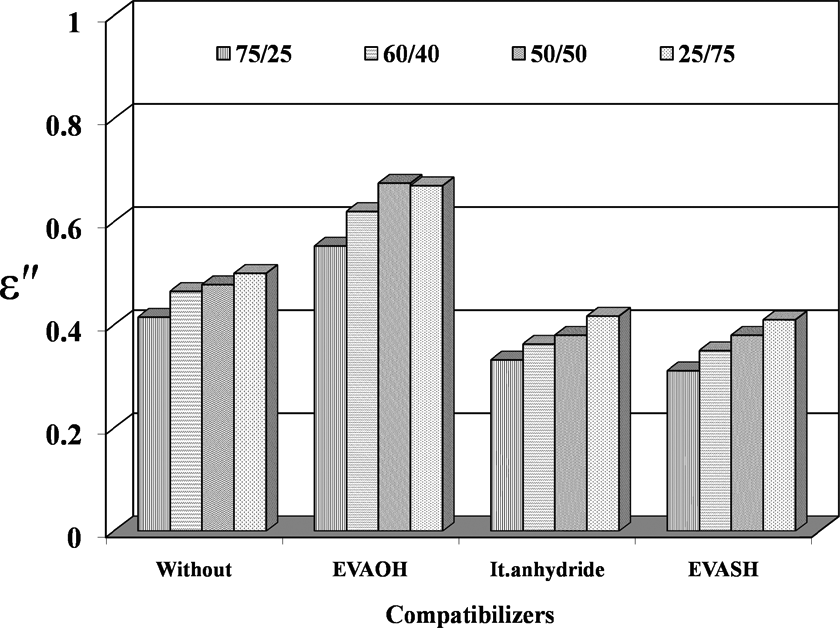
Dielectric loss (ε″) versus compatibilizers of different EPDM/EVA compositions at 30°C. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
Resistivity
The resistivity of the blends (Figure 11) is decreased by incorporation of both EVA and EVAOH as their presence can assist charge tunneling mechanism within the blend. The reduction of the polarity of the system by introducing 5% of It.anhydride and EVASH results in a notable increase in the resistivity of the blend. In this case, charge carriers transport mechanism via hopping or tunneling is unexpected due to the relatively large insulating barrier. The higher values of resistivity are desirable for safe operation of the insulator.
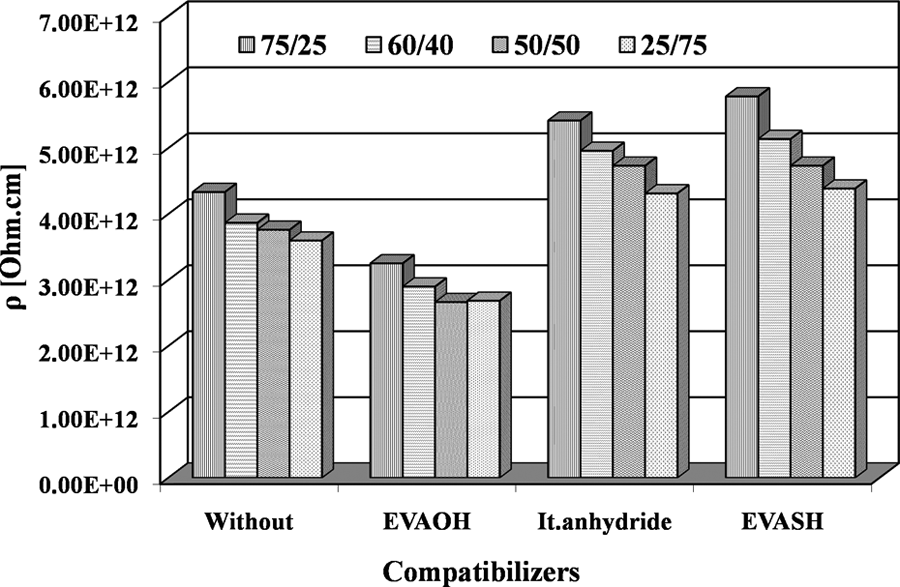
The resistivity (ρ) versus compatibilizers of different EPDM/EVA compositions at 30°C. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
Effect of temperature
The effect of temperature on permittivity ε′ and dielectric loss ε″ of uncompatibilized and compatibilized blend compositions is represented in Figures 12 and 13. It is clear that for all EVA loading, ε′ increases with increasing temperature. The intermolecular forces between polymer chains are broken by increasing temperature. This, in turn, enhances thermal agitation. So, the polar groups could be free to orient, allowing it to maintain up with the electric field change. This gives rise to ε′ values. The EPDM/EVA samples containing 5% of EVAOH, It.anhydride, and EVASH have lower ε′ values for the same reasons mentioned before.
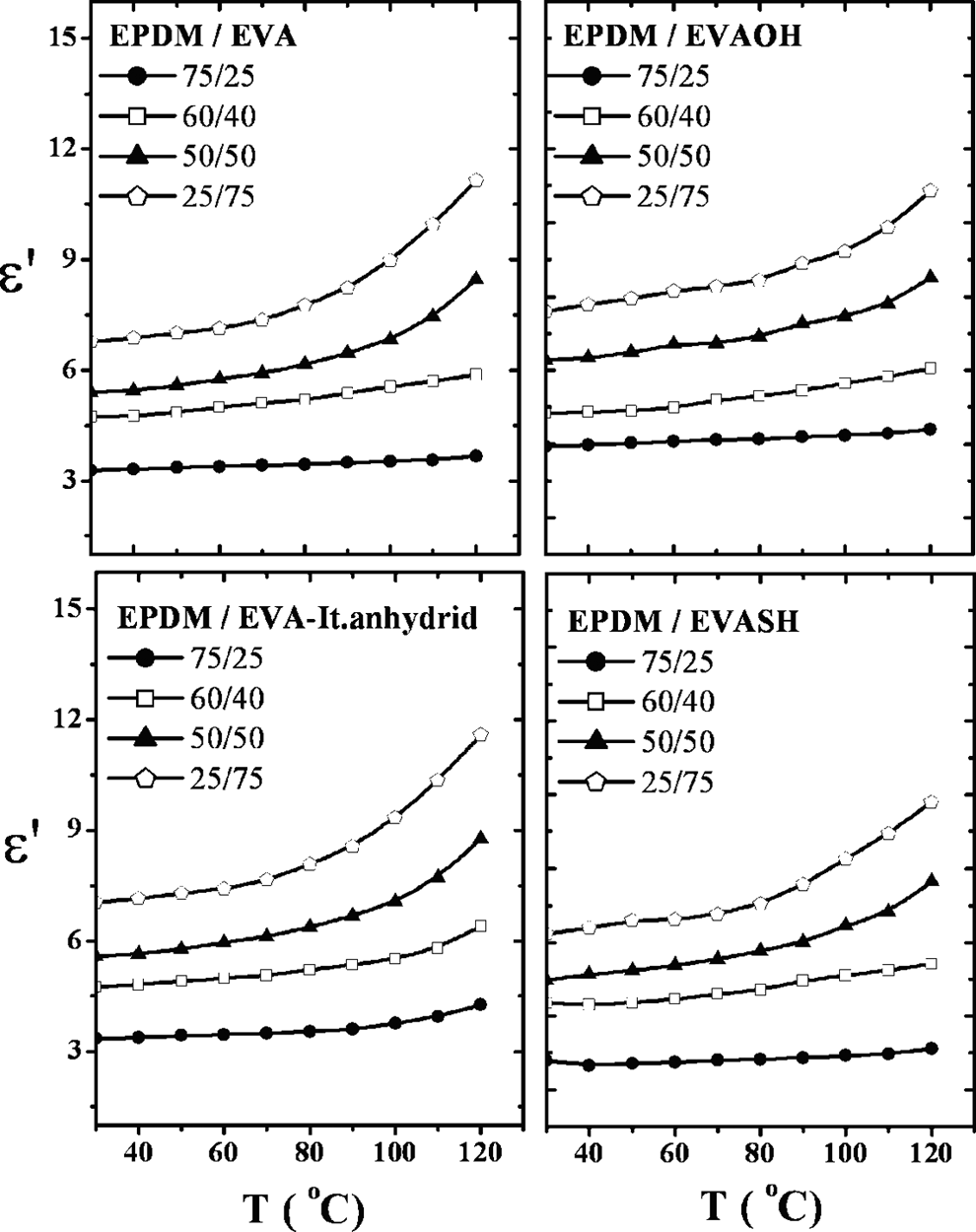
Permittivity (ε′) versus temperature of different EPDM/EVA compositions. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
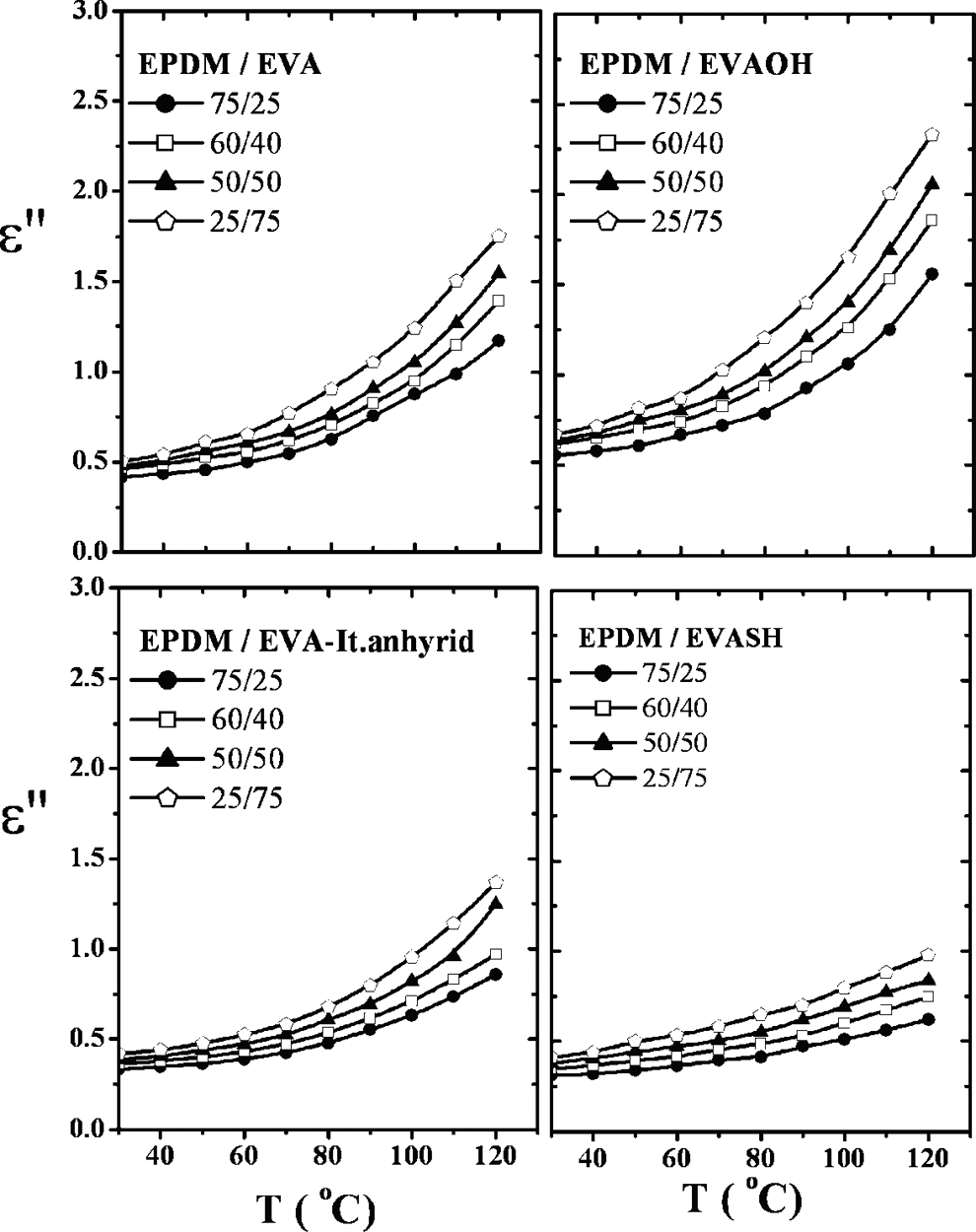
Dielectric loss (ε″) versus temperature of different EPDM/EVA compositions. EPDM/EVA: ethylene propylene diene monomer/ethylene vinyl acetate.
On the other hand, the increase in ε″ is related to the increase in segmental mobility and conductivity due to consequence drop in the viscosity of the EPDM/EVA blends and thermal dissociation of the molecules. 22 Moreover, the values of ε″ for IAH and EVASH blend compositions are lower compared to incompatibilized blends and EVAOH. This may be due to increasing of cross-linking and reduction of the number of charge carriers. 23
However, it is worth to mention that the lower values of ε′ and ε″ lead to lower loss and lesser tendency to heat up under a high voltage gradient.
Conclusions
As the EVA concentrations increase in the blend (EPDM/EVA), the tensile properties (σ B, σ R, and ε R) increase.
There is an improvement in the tensile strength by adding 5% EVAOH, It.anhydride, and EVASH on all blend compositions.
Also, the presence of the compatibilizers shows improvement in mechanical proprieties of blends after thermal oxidative aging.
The incorporation of 5% EVAOH, It.anhydride, and EVASH to blend compositions resulted in an improvement of dielectric properties.
It.anhydride and EVASH/EPDM/EVA compositions are promising materials for insulation purposes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.