Abstract
Continuous glass fiber (GF)-reinforced polymerized cyclic butylene terephthalate (pCBT) composites were prepared via vacuum-assisted resin transfer molding using butyltin tris(2-ethylhexanoate) as the catalyst. The relationship between melt viscosity and polymerization time was examined in the ring-opening polymerization of CBT resin. The effects of polymerization conditions such as catalyst content and polymerization temperature on viscosity average molar mass (Mv ), crystallization, mechanical properties, and microstructure of GF/pCBT composites were also investigated in detail. It is found that both high molecular weight and high degree of crystallinity of resin matrix can lead to high mechanical properties of composites. The composites prepared with 0.5% catalyst at 190°C show the best mechanical properties with tensile strength of 549 MPa, flexural strength of 585.2 MPa, and interlaminar shear strength of 47.1 MPa. The scanning electron microscopy analysis also demonstrates that good interfacial adhesion exists between fiber and resin, which agrees very well with experimental results.
Keywords
Introduction
Continuous fiber-reinforced thermoplastic composites have been successfully applied in many fields such as aerospace, automotive, construction, and so on. Thermoplastic composites offer many advantages over thermoset composites, including superior impact resistance, unlimited shelf life, short production cycle, and recyclability. However, the melt viscosity of thermoplastic resin is in the range of 100–10,000 Pa·s, which is very difficult for resin impregnation in fiber reinforcement. Compared to thermosetting composites, high temperature and pressure are usually required during the processing of high-performance thermoplastic composites, increasing production cost greatly.
Liquid composite molding (LCM) is one of the popular technologies for the manufacture of thermosetting composites due to high productivity, reduced styrene emissions, and automation friendliness, which involves injecting a liquid resin through a fibrous reinforcement in a closed mold. It represents a family of composite manufacturing processes such as resin transfer molding (RTM), vacuum-assisted RTM (VARTM), and structural reaction injection molding. One of the key requirements in LCM is the low viscosity of resin, that is, <1 Pa·s. 1,2 Recently, low viscous monomers (e.g. lauryllactam 3 and caprolactam 4 –8 ) or cyclic oligomers (e.g. cyclic butylene terephthalate (CBT) 9,10 ) have been reported and can form linear polymers via in situ ring-opening polymerization in the presence of a catalyst, which provides a new method to prepare continuous fiber-reinforced thermoplastic composites by LCM. The CBT resin is made of cyclic oligoesters and polymerizes to form linear thermoplastic polymerized cyclic butylene terephthalate (pCBT) via an in situ ring-opening polymerization with tin or titanium catalysts. 11 –14 The CBT resin presents low melt viscosity (as low as 0.01 Pa·s) and can impregnate fiber reinforcement very well to form thermoplastic composites with high toughness and impact resistance. Continuous fiber-reinforced pCBT composites have been extensively investigated by many researchers using different processes. Parton et al. 9,10 prepared glass fiber (GF)/pCBT composites by RTM process using butyltin chloride dihydroxide as the catalyst and found that the high reaction speed of the cyclic oligoesters resulted in the small time window for fiber impregnation. Ishak et al. 15 –17 studied the thermal and rheological behaviors of CBT containing catalyst and prepared GF/pCBT composites using compression molding technique. Mader et al. 18 investigated the effects of the sizing on the interfacial properties and microscopic structure of GF/pCBT composites. Baets et al. 19,20 used polycaprolacton to toughen GF/pCBT composites and also developed a nonisothermal production process to improve the toughness of composites. Balogh et al. 21 prepared carbon fiber-reinforced pCBT matrix (CF/pCBT) composites with a low-pressure method and examined the effects of low fiber volume fraction on mechanical properties. Agirregomezkorta et al. 22 prepared the CF/pCBT composites by vacuum infusion and studied the effects of vacuum infusion processing parameters on the impact behavior. According to our earlier work, the time window for impregnation is about 2 min at 180°C and less than 1 min at 190°C when using butyltin chloride dihydroxide as the catalyst during the preparation of GF/pCBT composites. 23 However, controllable time window in minutes (e.g. 5 min) for impregnation is necessary to manufacture large-scale composite parts by LCM processes. Here, butytin tri(2-ethyhexoate) with low catalytic activity is chosen as the catalyst to expand the time window. To date, the GF/pCBT composites prepared via ring-opening polymerization of CBT resin using butytin tri(2-ethyhexoate) as the catalyst have not been reported in detail.
In this article, chemical rheological properties of pCBT resin during the in situ polymerization process were studied using a rheometer. The effects of polymerization conditions such as catalyst content and polymerization temperature on viscosity average molar mass (Mv ), crystallization, mechanical properties, and microstructure of GF/pCBT composites were also investigated in detail.
Experimental
Materials
The CBT resin used in this study was supplied by Cyclics Corporation (Schwarzheide, Germany). The resin was dried in a vacuum oven at 100°C for at least 12 h before using. The tin-based catalyst (butyltin tris(2-ethylhexanoate), 98%) was obtained from Energy Chemical (Shanghai, China). Plain woven fabrics of glass fiber (GF) (S-glass SW100A-90a, 400 g/m2) were supplied by Jiangsu Jiuding New Material Co., Ltd. (Jiangsu, China). The fabrics were dried in a vacuum oven at 120°C for 2 h before using.
Preparation of GF/pCBT composites
The GF/pCBT composites were prepared via ring-opening polymerization of CBT resin using butytin tri(2-ethyhexoate) as the catalyst by VARTM. Eleven layers of GF fabrics were positioned along a steel cast and ready for resin impregnation. The CBT resin was melted under a nitrogen flowing atmosphere at 190°C in an oil bath, and then uniformly mixed with a certain amount of butytin tri(2-ethyhexoate) by a mechanical stirrer in a 1000 mL three-neck round bottom flask. The stirrer stopped for about 1 min, and then, the mixture was drawn into the stainless steel mold and impregnated the GF fabrics by the negative pressure using a vacuum pump. The diagram of the VARTM process setup for GF/PCBT composites is shown in Figure 1.
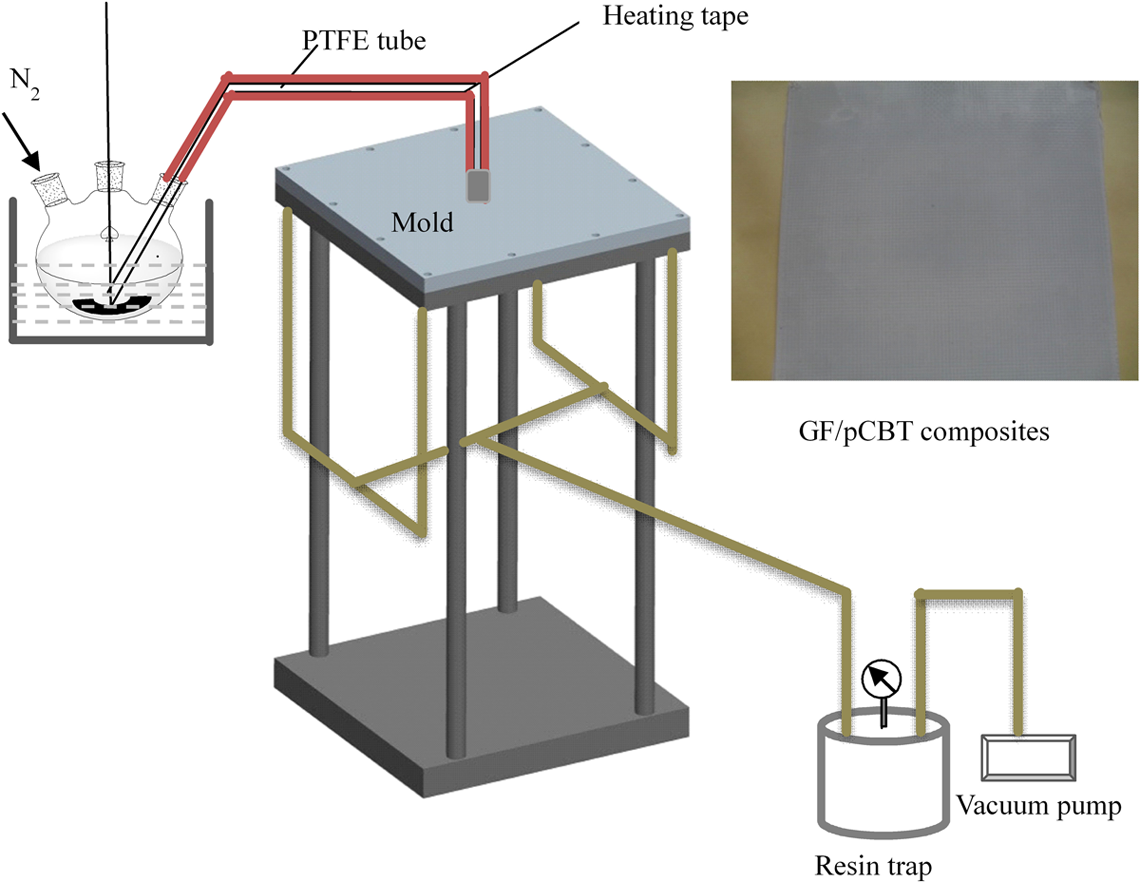
Diagram of the VARTM process setup for GF/pCBT composites. VARTM: vacuum-assisted resin transfer molding; pCBT: polymerized cyclic butylene terephthalate; GF: glass fiber.
The polymerization temperature of pCBT matrix resin in the GF/pCBT composites was the mold temperature, and the composites were prepared at 180, 190, 200, and 210°C, respectively. The mold needed to keep a certain temperature for 1 h and then naturally cooled to room temperature in the air.
Chemical rheology
Rheological measurement was performed using a Physica MCR 301 rheometer (Anton Paar, Austria) with a 25 mm diameter parallel plate fixture. Samples were prepared with CBT resin and a certain amount of catalyst by melt mixing at 190°C. The space between the parallel plates was 0.5 mm. The time sweep was measured at 1% amplitude and 1 Hz frequency under a nitrogen flowing atmosphere.
Viscosity average molar mass
The viscosity average molar mass (Mv ) of pCBT resin was determined using a micro-Ubbelohde Capillary II (SCHOTT AVS 370, Germany). The Mv s of the pure pCBT resin and the pCBT resin matrix in GF/pCBT composites were named Mv 1 and Mv 2, respectively. The GF/pCBT composites were dissolved in an N-methyl-2-pyrrolidone solution at 180°C. GFs were then separated by filtering, and the pCBT polymer was precipitated using acetone from the solution. The obtained polymer was washed repeatedly with acetone until the N-methyl-2-pyrrolidone was removed completely. Then, the polymer was dried at least 24 h at 70°C in a vacuum oven. The dried resin was dissolved in a mixture of phenol/tetrachloroethane (3:2 w/w), and the Mv was obtained by a single-point measurement at a concentration of 0.5 g/dl at 30°C. The inherent viscosity (η inh) was calculated according to Eq. (1)
where t is the flow time of the polymer solution, t 0 is the flow time of the pure solvent, and c is the concentration of the polymer solution. The Mv can be calculated from η inch by the Mark–Houwink equation 24 (Eq. [2])
where K′and α are the Mark–Houwink constants; K′ = 21.5 × 10−3 mL/g and α = 0.82. 25
Crystallization and thermal analysis
The nonisothermal crystallization behaviors of pCBT resin were investigated using a differential scanning calorimetry (DSC) (Perkin-Elmer Pyris Diamond DSC, Norwalk, Connecticut, USA). The samples were first heated from room temperature to 250°C at a scanning rate of 20°C/min in a nitrogen atmosphere, maintained there for 5 min to eliminate the thermal history, and then cooled down to 25°C at different cooling rates of 2.5, 5, 10, 15, 20, 25, 30, and 40°C/min, respectively.
The degree of crystallinity (Xc ) of pCBT matrix resin in GF/pCBT composites was studied by DSC and thermogravimetric analysis (Perkin-Elmer Pyris Diamond TG/DTA, Norwalk, Connecticut, USA). The melting enthalpy (Hm ) of pCBT matrix resin was tested by DSC at a heating rate of 10°C/min over a temperature range of 25–250°C under a nitrogen atmosphere. The matrix resin quantity of GF/pCBT composites was measured by TGA at a heating rate of 20°C/min over a temperature range of 150–800°C under an air atmosphere. Then, the melting enthalpy per unit of mass (ΔH m = H m/m resin) was obtained. The degree of crystallinity was calculated according to Eq. (3)
where ▵H 100 is the melting enthalpy of fully crystalline pCBT: ▵H 100 = 142 J/g. 26
Mechanical testing
The mechanical properties of GF/pCBT composites were measured with an Instron 5985 universal testing machine (Instron corporation, Norwood, Massachusetts, USA). Tensile properties, flexural properties, and interlaminar shear strength (ILSS) were determined according to ASTM D-3039, ASTM D-7264, and ASTM D-2344, respectively. The dimensions of specimens for three-point short beam shear test were 16 × 5.0 × 2.5 mm. The ILSS was calculated according to the following Eq. (4):
where Fm is the maximum force; w and t are the width and thickness of the test specimen, respectively. All specimens were dried in a vacuum oven at 50°C for 12 h before testing.
Morphology observation
The morphologies of fracture surfaces of GF/pCBT composites were observed by a scanning electron microscope (FE-SEM S-4800, Hitachi, Japan). The samples are sputtered with a thin layer of gold before the observation.
Results and discussion
Chemical rheology properties
The relationship between melt viscosity and polymerization time is shown in Figure 2. Figure 2(a) is the curve of complex viscosity versus polymerization time of pCBT resin with different catalyst contents at 190°C. The induction period before the reaction decreased from 25 min to 7 min with increasing the catalyst content from 0.1% to 0.6%, due to the enhanced reaction rate. Apparently, the high catalyst content can shorten the induction period before the reaction.
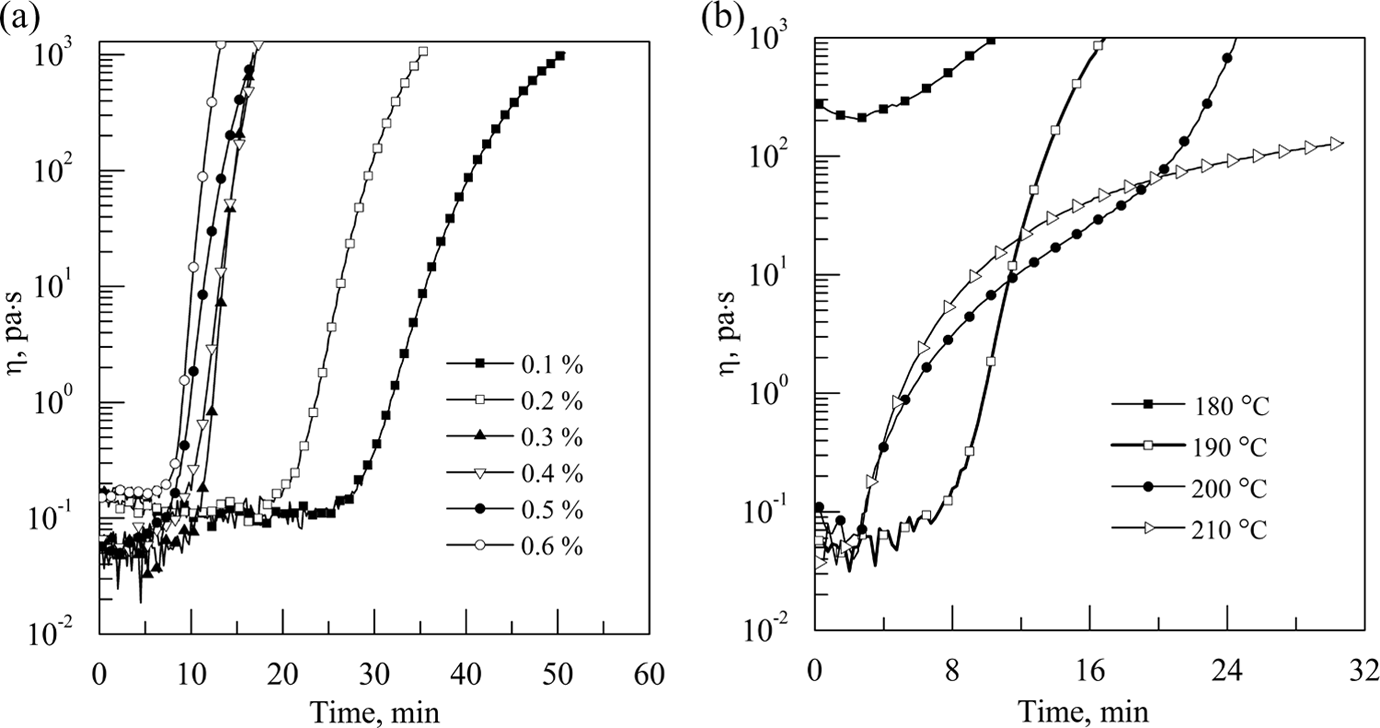
Complex viscosity of pCBT resin as a function of time in different polymerization conditions (a) with different catalyst contents at 190°C; (b) with 0.5% catalyst at different polymerization temperatures. pCBT: polymerized cyclic butylene terephthalate.
Figure 2(b) is the curve of complex viscosity versus polymerization time of pCBT resin with 0.5% catalyst at different polymerization temperatures. It can be seen that the melt viscosity of CBT resin was about 200 Pa·s when the polymerization temperature was 180°C. The reason is that CBT resin containing oligomer rings with two to seven monomers has a melting range of 150–185°C. 15 Some CBT resin with higher molecular weight might not be completely melted, resulting in higher viscosity. The induction period before the reaction is about 8 min when the polymerization temperature is 190°C, which is long enough to manufacture large-scale thermoplastic composite laminates. The molten resin has a very low viscosity of around 0.05 Pa·s when the polymerization temperature is higher than 190°C. In addition, the induction period before the reaction decreases with increasing the polymerization temperature, which is due to the increase in the reaction rates with the polymerization temperature. Thus, the higher polymerization temperature can reduce the induction period before the reaction.
Effects of polymerization conditions on the viscosity average molar mass
The molecular weight of pCBT resin determines the properties of resin, which in turn affects the properties of GF/pCBT composites. Both catalyst contents and polymerization temperature have substantial effects on the molecular weight of the pCBT resin as shown in Figure 3. Figure 3(a) shows that the values of Mv of pure pCBT resin (Mv 1) and pCBT resin matrix in GF/pCBT composites (Mv 2) increase from 4.99 × 103 mol·g−1 to 3.44 × 104 mol·g−1and from 1.71 × 104 mol·g−1 to 3.05 × 104 mol·g−1 with the catalyst contents, respectively. It is interesting that the Mv 2 reaches the maximum value at the catalyst loading of 0.5%, and then keeps unchanged with further increase in catalyst loading. It can also be seen that the polymerization temperature has similar effects on both Mv 1 and Mv 2 as shown in Figure 3(b). The values of Mv 1 and Mv 2 increase from 1.66 × 104 mol·g−1 to 3.23 × 104 mol·g−1 and from 1.43 × 104 mol·g−1 to 3.05 × 104 mol·g−1 until the polymerization temperature arrives at 190°C, respectively, and then change little with increasing the polymerization temperature. It is obvious that the Mv 2 is smaller than the Mv 1 with the same catalyst content or polymerization temperature. This might be caused by chemical interactions. It has been reported that graphite oxide and SiO2 have large specific functional groups (e.g. acidic groups, hydroxyl groups, and epoxy groups) on their surface, which affect the polymerization of CBT. 27 Therefore, the active groups such as acidic groups and hydroxyl groups on the surface of GF might terminate the polymerization reaction of pCBT, which results in a smaller value of Mv 2.
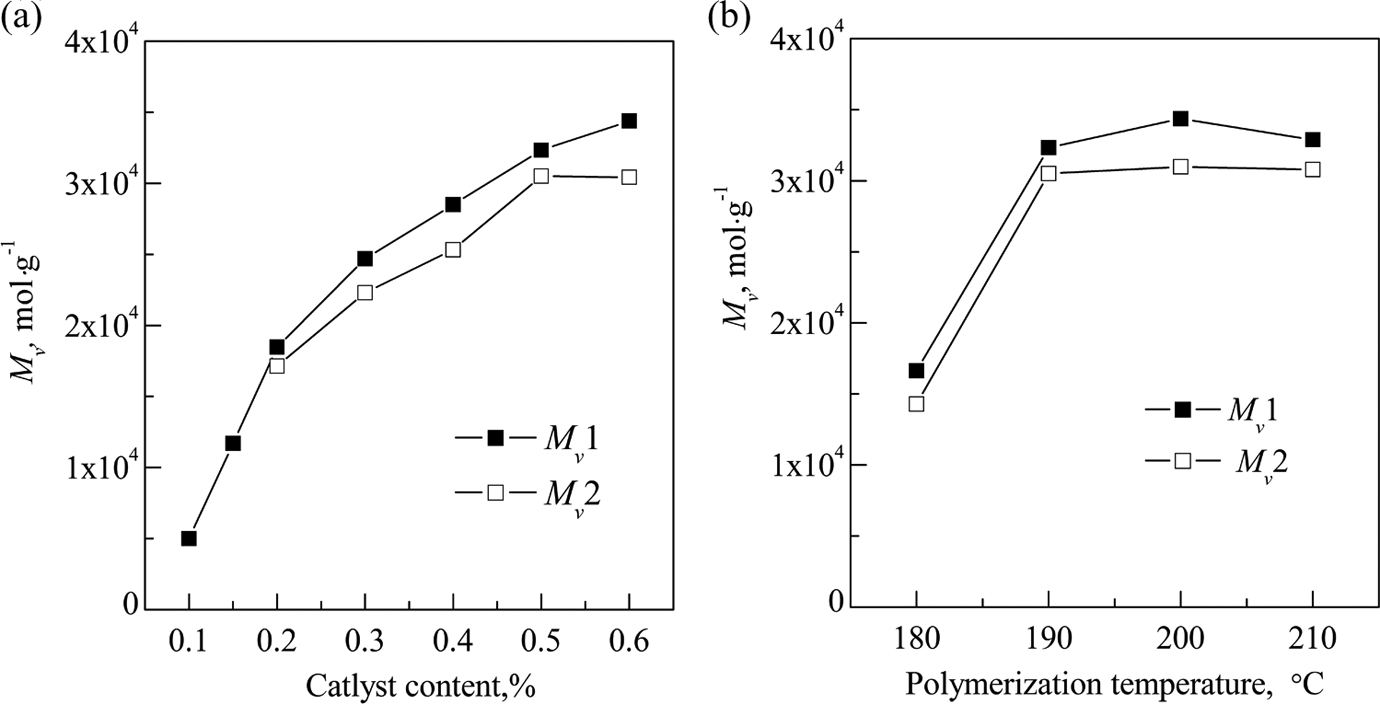
Effects of polymerization conditions on the viscosity average molar mass of pCBT resin (a) polymerization temperature: 190°C and polymerization time: 1 h; (b) catalyst content: 0.5%; polymerization time: 1 h. pCBT: polymerized cyclic butylene terephthalate.
Nonisothermal crystallization kinetics of pCBT with different molecular weights
Nonisothermal crystallization behavior of pCBT resin with different molecular weights
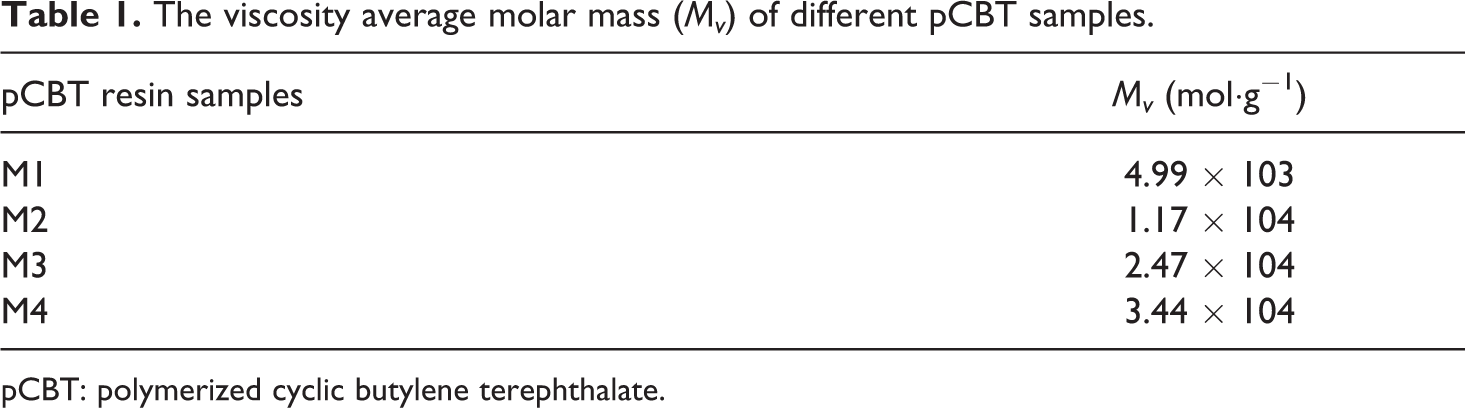
The values of the viscosity average molar mass of different pCBT resin samples are listed in Table 1. The crystallization curves of pCBT resin with different molecular weights at various cooling rates are shown in Figure 4. All samples show a single sharp exotherm in the temperature range of 136–204°C, and the DSC peaks are broadened with increasing the cooling rate. This is because that increasing the cooling rate decreases the rearrangement time of polymer chains, which promotes the formation of small and imperfect crystallites. 28 It is also found that the heat flow rate increases with increasing the cooling rate. The reason is that the crystallization time decreases with increasing the cooling rate.
The viscosity average molar mass (Mv ) of different pCBT samples.
pCBT: polymerized cyclic butylene terephthalate.
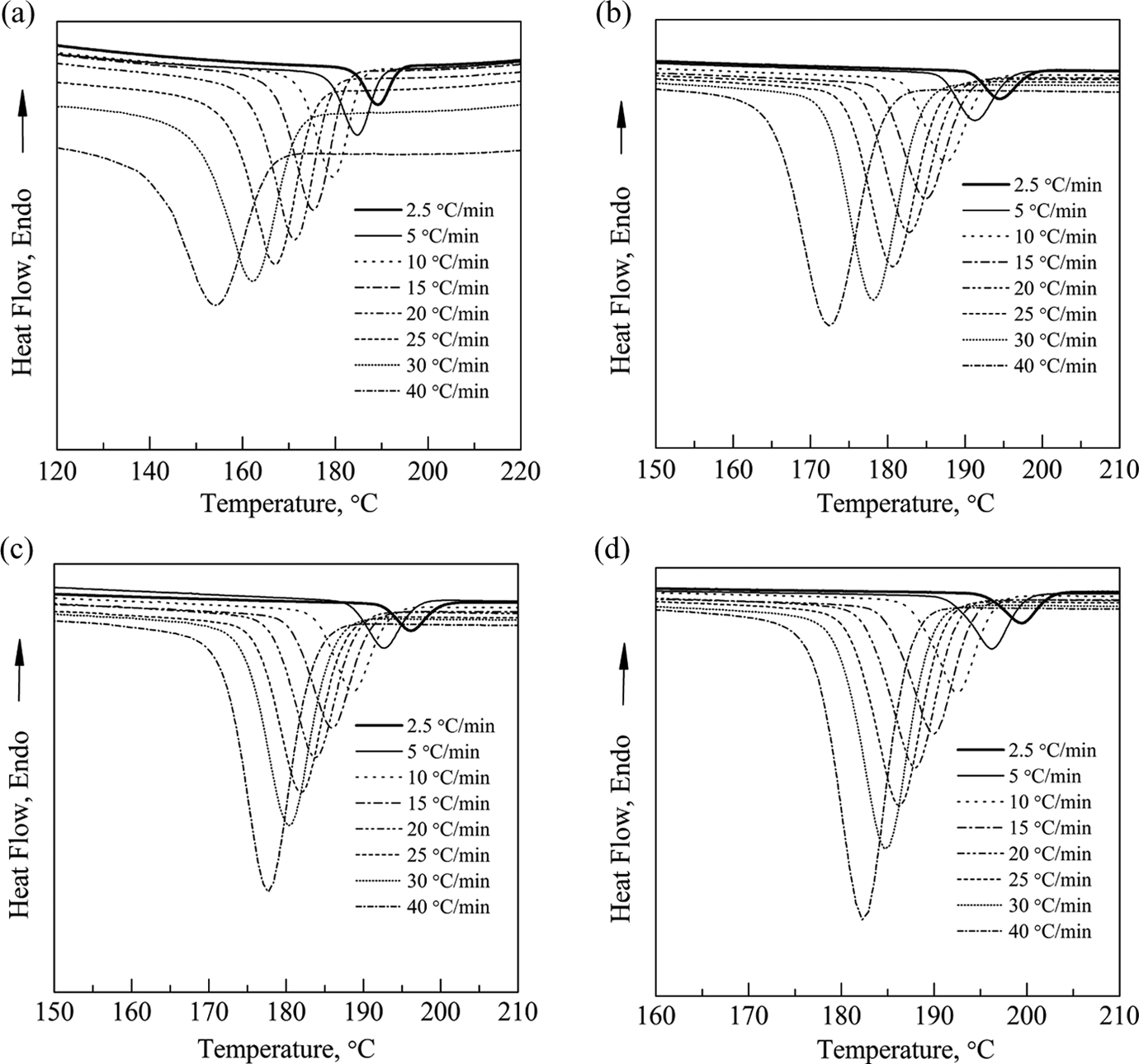
Crystallization curves of pCBT resin with different molecular weights at different cooling rates: (a) sample M1, (b) sample M2, (c) sample M3, and (d) sample M4. pCBT: polymerized cyclic butylene terephthalate.
The values of the crystallization parameters derived from the crystallization curves are summarized in Table 2. T 0, Tc , T 1/2, and t 1/2 are initial crystallization temperature, crystallization peak temperature, crystallization half temperature, and crystallization halftime, respectively. It can be seen that T 0, Tc , T 1/2, and t 1/2 decrease with increasing the cooling rate. Crystallization starting at higher temperature can provide sufficient time to overcome nucleation energy barriers and activate nucleation at a slower cooling rate, while the nuclei becomes active at lower temperature. 29 Therefore, the crystallization rate increases with increasing the cooling rate. In addition, T 0, Tc , and T 1/2 increase with the molecular weight at the same cooling rate, whereas t 1/2 keeps decreasing. These results indicate that the crystallization rate of pCBT resin increases with the molecular weight within the range of this study. The pCBT resin with lower molecular weight may have too many chain end groups and hinder the crystallization.
Crystallization parameters for pCBT resin with different molecular weights.
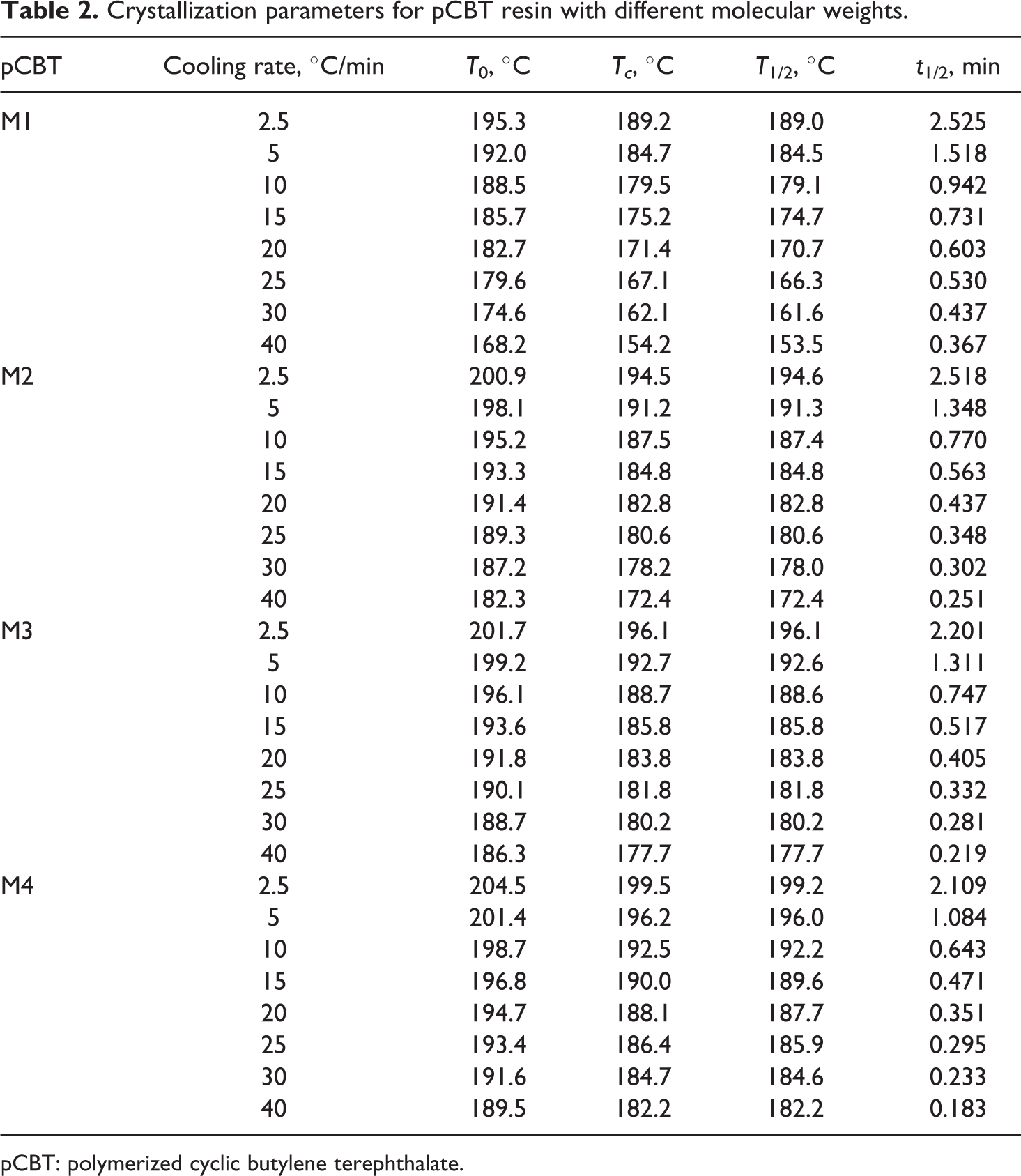
pCBT: polymerized cyclic butylene terephthalate.
Assessment of Ozawa equation
In order to obtain the relationship between crystallization rate of pCBT resin and crystallization temperature, Ozawa model was chosen to analyze the nonisothermal crystallization data. Ozawa 30 properly modified the Avrami equation by considering the effect of cooling rate on dynamic crystallization. The relative crystallinity function of temperature can be represented as

where Xt is the relative crystallinity, φ is the cooling rate, and k 0 and n 0 are Ozawa crystallization rate constant and Ozawa exponent, respectively. The k 0 is related to the overall crystallization rate and indicates how fast crystallization occurs. 31 Equation (5) can be changed to its linear form as follows:
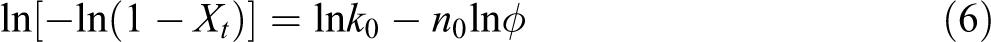
Figure 5 illustrates the plots of ln[−ln(1 − Xt )] as a function of lnφ for a chosen crystallization temperature. Obviously, a series of straight lines of ln[−ln(1 − Xt )] against ln φ can be obtained. The k 0 and the n 0 can be estimated from the antilogarithmic value of the y-intercept and the negative value of the slope. Ozawa kinetic parameters and regression coefficient (R 2) are listed in Table 3.
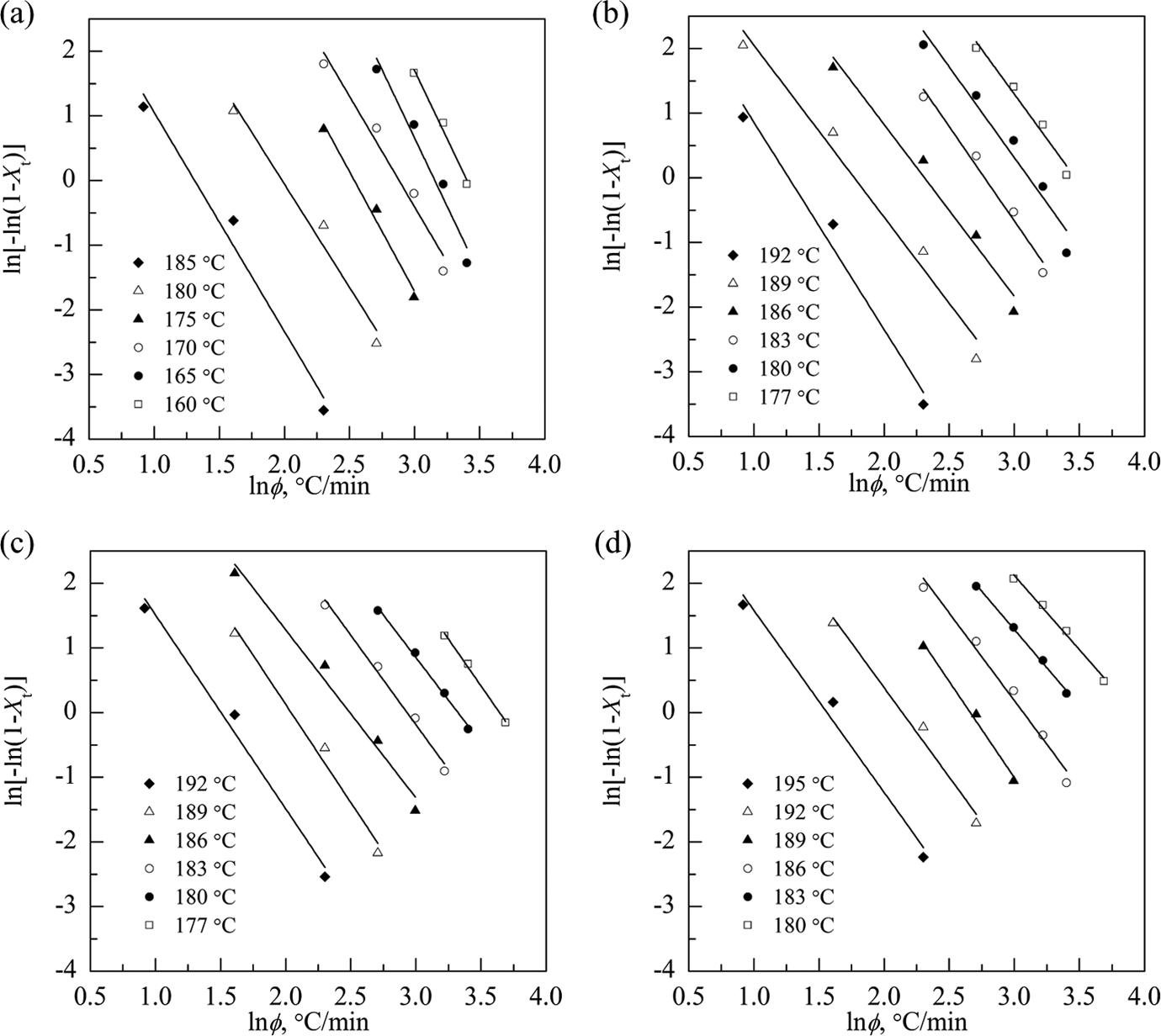
Ozawa analysis based on the nonisothermal crystallization of pCBT resin: (a) sample M1, (b) sample M2, (c) sample M3, and (d) sample M4. pCBT: polymerized cyclic butylene terephthalate.
Nonisothermal melt crystallization kinetics for pCBT resin with different molecular weights based on Ozawa analysis.
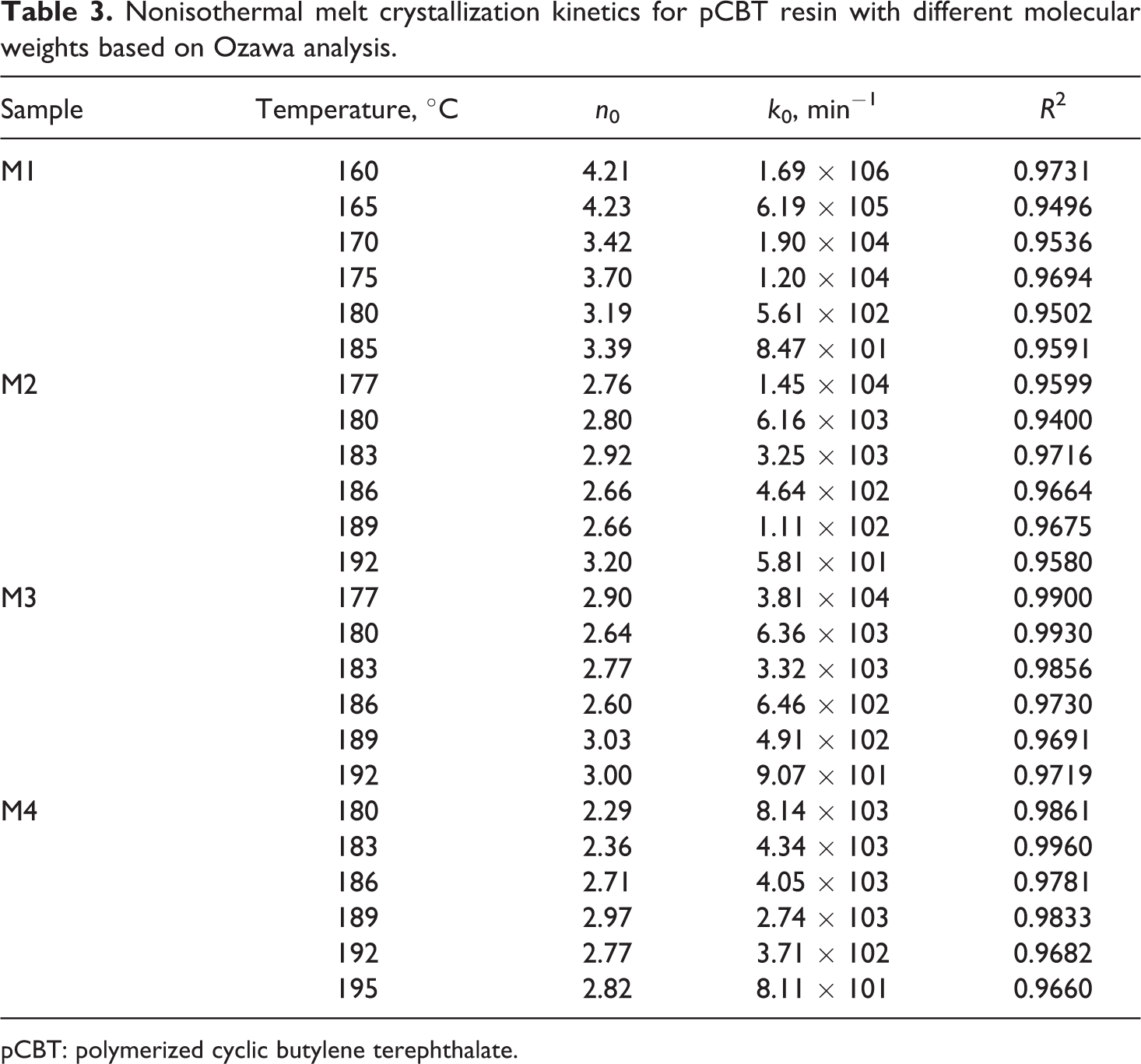
pCBT: polymerized cyclic butylene terephthalate.
The values of R 2 listed in Table 3 are in the range of 0.9400–0.9960, which indicates that Ozawa model fits the experimental data very well. Ozawa exponents (n 0) are found to remain practically constant (3.19–4.23 for sample M1, 2.66–3.20 for sample M2, 2.60–3.03 for sample M3, and 2.29–2.97 for sample M4) with varying temperature and show a decreasing trend with increasing the molecular weight of pCBT resin, which means that the crystallization mechanism should be different 32 and different crystal structures and morphologies form in pCBT resin. It is also found that the k 0 decreases with increasing the temperature and increases with the molecular weight in all samples, suggesting that the crystallization rate of pCBT resin reduces with increasing the temperature, while increases with increasing the molecular weight.
Effects of polymerization conditions on the degree of crystallinity of the matrix resin in GF/pCBT composites
The effects of polymerization conditions such as catalyst content and polymerization temperature on the degree of crystallinity of the matrix resin in GF/pCBT composites are shown in Figure 6. As shown in Figure 6(a), the degree of crystallinity increases from 33% to 40% when the catalyst contents change from 0.2% to 0.6%, with a maximum value at the catalyst loading of 0.5%. When the molecular weight increases to a certain degree during the polymerization process of CBT resin, pCBT resin will begin to crystallize. In general, the reaction rate accelerates with increasing the catalyst content, and a relatively full-crystallization time can be obtained. Therefore, the high catalyst content will promote polymer crystallization and lead to a higher degree of crystallinity.
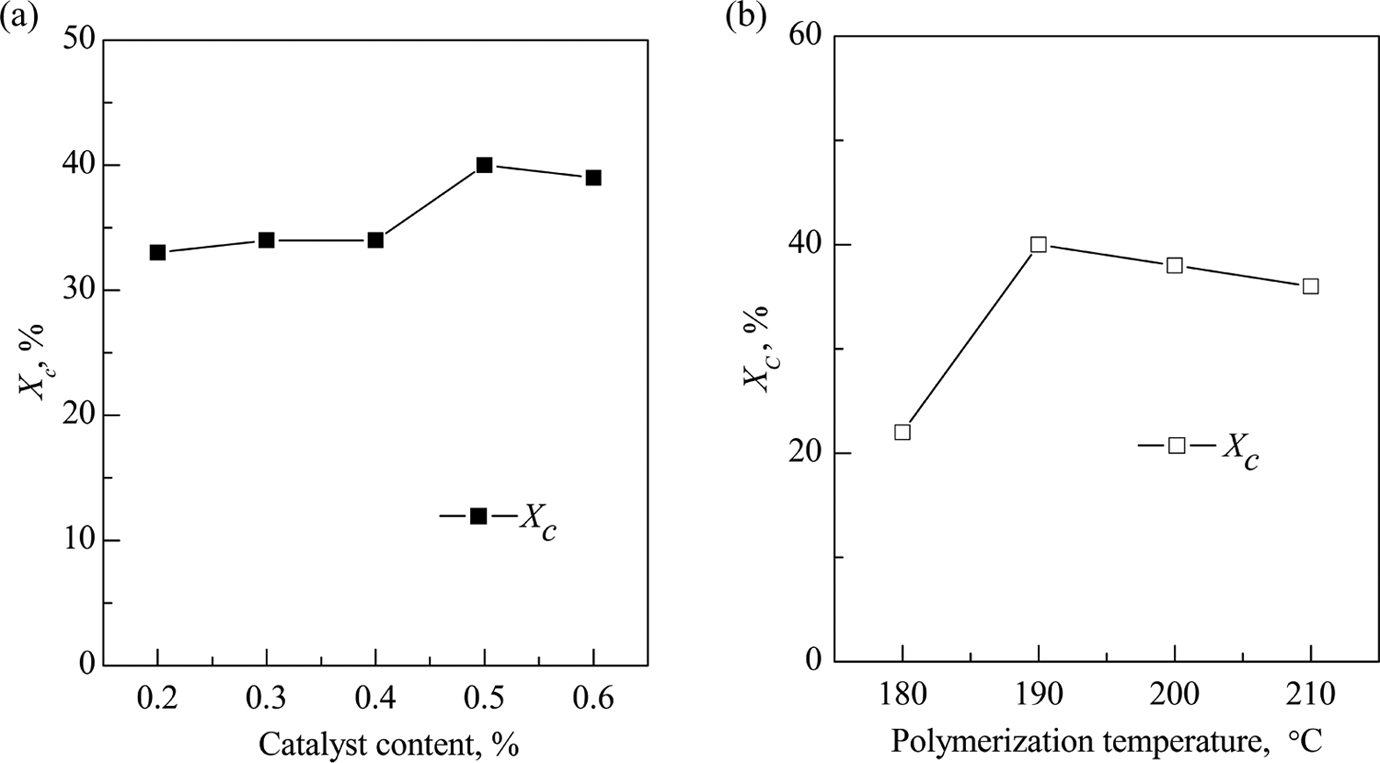
Effects of polymerization conditions on the degree of crystallinity of the matrix resin in GF/pCBT composites (a) polymerization temperature: 190°C and polymerization time: 1 h; (b) catalyst content: 0.5% and polymerization time: 1 h. GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate.
Figure 6(b) shows the effects of polymerization temperature on the degree of crystallinity of the matrix resin in GF/pCBT composites. The degree of crystallinity increases from 22% to 40% with the polymerization temperature until it reaches a maximum value at 190°C, and then slightly decreases when the polymerization temperature further increases. The polymerization temperature of CBT resin is below the melting point of the polymer. Thus, the chain propagation of pCBT resin is accompanied with crystallization when the molecular weight increases to a certain extent. The polymerization reaction rate of CBT resin is relatively low at the low polymerization temperature. As the polymerization reaction proceeds, the increase in the molecular weight of pCBT resin improves the crystallization rate. When the crystallization rate is higher than the polymerization reaction rate, polymerization reaction occurs to terminate because the active ends of the polymer chains are buried in crystals. Therefore, the low molecular weight of pCBT is obtained and retards the further crystallization, which causes a lower degree of crystallization at the low polymerization temperature. As the polymerization temperature is enhanced, both the molecular weight and the melt viscosity of polymer increase with the polymerization reaction rate. The polymerization reaction almost stops when the active ends of the polymer chains are buried in polymer molecular chains. However, the degree of crystallization keeps increasing because the crystallization process is still in progress. Hence, the degree of crystallization of pCBT resin increases with the polymerization temperature. When the polymerization temperature further increases, the molecular weight of pCBT resin becomes higher due to the improved polymerization reaction rate. According to the nonisothermal crystallization kinetics of pCBT with different molecular weights, the crystallization rate increases with the molecular weight, whereas decreases with the polymerization temperature. However, the effect of polymerization temperature on crystallization rate may be much more obvious than that of the molecular weight in this case. Therefore, the degree of crystallization decreases slightly at the high polymerization temperature.
Figure 7 shows the DSC curves of the matrix resin in GF/pCBT composites. As shown in Figures 7(a) and 3(a), it can be seen that the melting temperature and the Mv 2 of the matrix resin in GF/pCBT composites have the same changing tread with different catalyst contents. The melting temperature increases from 216°C to 226°C with the catalyst content from 0.2% to 0.5%. It shows a maximum value at the loading of 0.5%, and then remains constant when the catalyst content further increases. The lower Mv 2 at the lower catalyst content (Figure 3(a)) results in lower melting temperature. The Mv 2 reaches the maximum value when the catalyst content is more than 0.5%. Therefore, the melting temperature also achieves the maximum value at the catalyst loading of 0.5%.
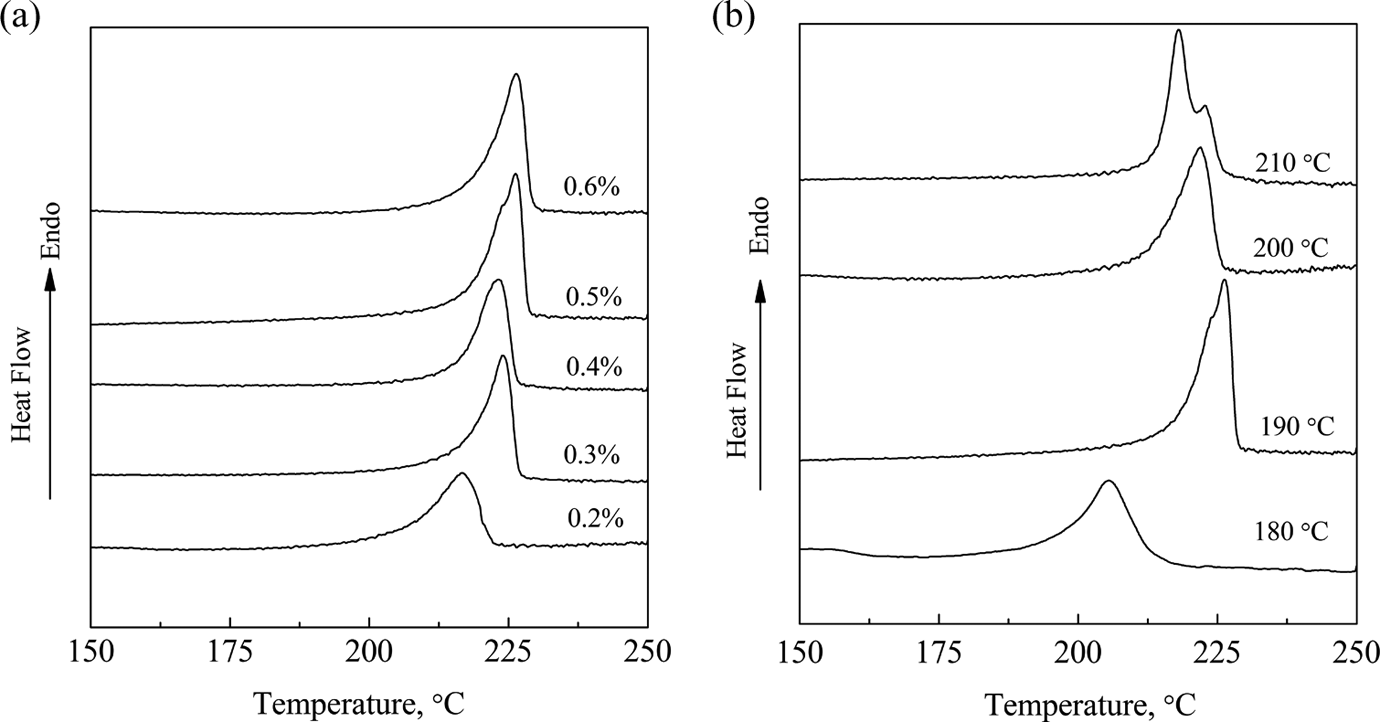
DSC curves of matrix resin in GF/pCBT composites (a) polymerization temperature: 190°C and polymerization time: 1 h; (b) catalyst content: 0.5% and polymerization time: 1 h. GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate.
As shown in Figure 7(b), it can be seen that the melting temperature increases from 205°C to 226°C with the polymerization temperature from 180°C to 190°C. It reaches the maximum value at 190°C, and then decreases when the polymerization temperature further increases. The melting temperature is much lower when the polymerization temperature is 180°C, which also proves that the molecular weight is lower. The melting temperature of polymer decreases when the temperature exceeds 190°C. The double melting peaks appear when the polymerization temperature is 210°C, which demonstrates that the imperfect crystals are obtained. 27
Effects of polymerization conditions on mechanical properties and microstructure of GF/pCBT composites
Effect of catalyst content
Catalyst content has an important effect on both polymerization rate and molecular weight. Polymerization rate is fast when catalyst content is high, which causes a short infusion time to impregnate fiber reinforcements difficultly. In this study, the infusion time is enough to prepare a 300 × 300 × 2.5 mm composite laminate.
The effects of catalyst content on the mechanical properties of GF/pCBT composites are shown in Figure 8. As shown in Figure 8, we can see that the mechanical properties (tensile strength, flexural strength, and ILSS) increase with increasing the catalyst contents until it reaches a maximum value (549 MPa for tensile strength, 585.2 MPa for flexural strength, and 47.1 MPa for ILSS) at 0.5% catalyst, and then decrease slightly when the catalyst content further increases. However, it seems that the catalyst content has little effect on both tensile modulus and flexural modulus. As discussed before, the catalyst content has a great effect on the molecular weight and the degree of crystallinity of pCBT resin. Another point is that the molecular weight and the degree of crystallinity also play an important role in the mechanical properties of composites. Therefore, the maximum mechanical properties of GF/pCBT composites are achieved at the catalyst loading of 0.5%, since the molecular weight and the degree of crystallinity of pCBT resin in GF/pCBT composites reach the maximum values at 0.5% catalyst.
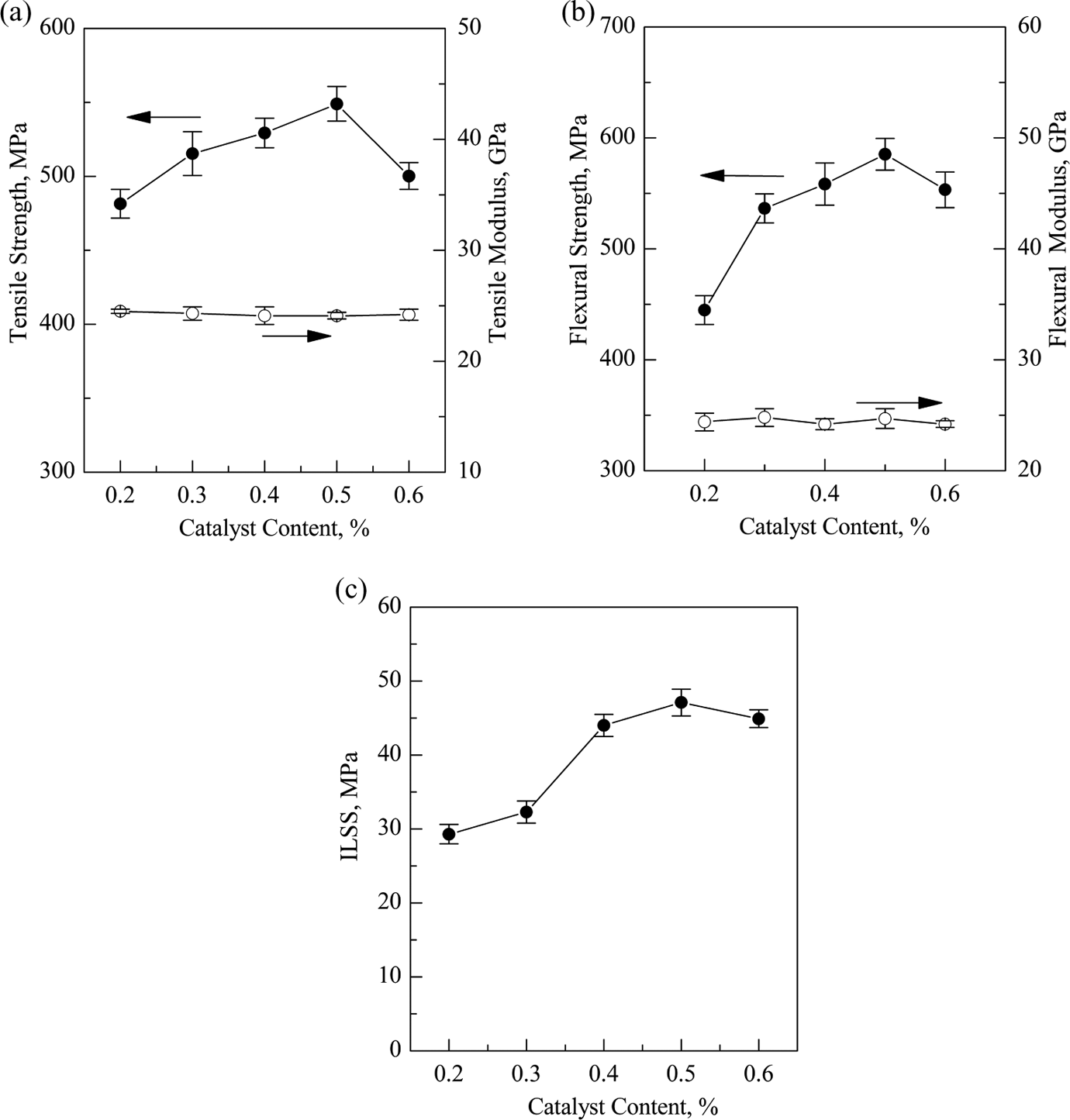
Effect of catalyst content on the mechanical properties of GF/pCBT composites (a) tensile strength, (b) flexural strength, and (c) ILSS. GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate; ILSS: interlaminar shear strength.
Figure 9 shows the scanning electron microscopy (SEM) micrographs of the tensile fracture surfaces of GF/pCBT composites with different catalyst contents. It can be seen that most fibers are pulled out in an accumulating failure mode and some resin adheres to the surface of the fibers. There are a large number of resins on the surface of fibers, which reveals the good interfacial properties between the fiber and the resin. Also, the fracture surface of pCBT resin appears rough and irregular. The fracture irregular strips increase with catalyst content, which indicates that the toughness of matrix resin is improved due to the increase in molecular weight of matrix resin with catalyst content.

SEM micrographs for fracture surfaces of GF/pCBT composites with different catalyst contents: (a) 0.2%, (b) 0.3%, (c) 0.4%, and (d) 0.5%. SEM: scanning electron microscopy; GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate.
Effect of polymerization temperature
The effects of polymerization temperature on the mechanical properties of GF/pCBT composites are shown in Figure 10. It can be seen that the mechanical properties of GF/pCBT composites also increase with the polymerization temperature until they reach a maximum value (549 MPa for tensile strength, 585.2 MPa for flexural strength, and 47.1 MPa for ILSS) at 190°C, and then remains unchanged when the polymerization temperature further increases. Since the molecular weight of pCBT resin increases with increasing the polymerization temperature, the higher polymerization temperature can improve the mechanical properties of composites.
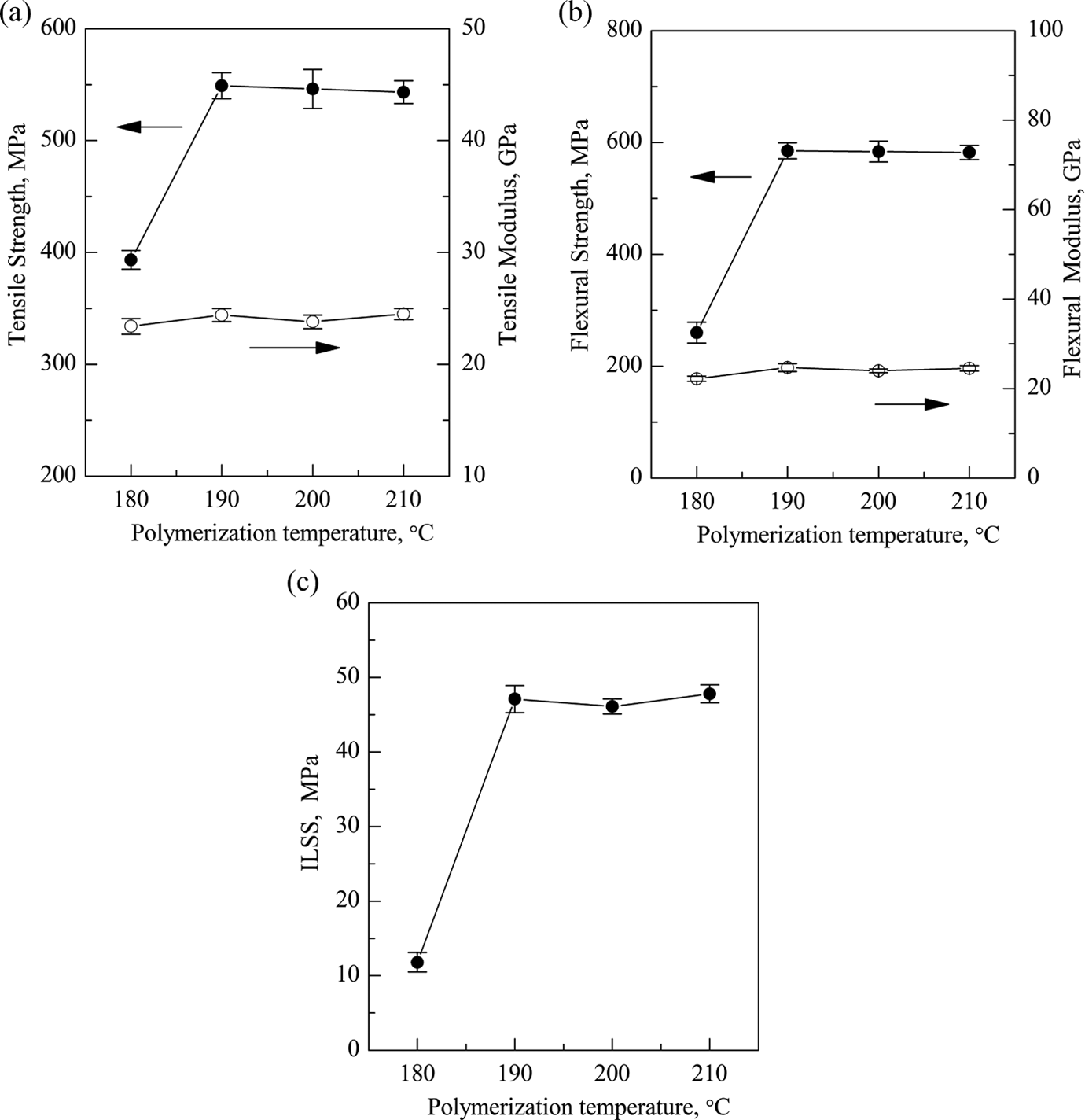
Effect of polymerization temperature on the mechanical properties of GF/pCBT composites (a) tensile strength, (b) flexural strength, and (c) ILSS. GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate; ILSS: interlaminar shear strength.
Figure 11 shows the SEM micrographs of the tensile fracture surfaces of GF/pCBT composites with different polymerization temperatures. The resin of GF surface in Figure 11(a) exhibits brittle fracture because of low molecular weight at 180°C. Also, little resin adheres to the surface of fibers, which indicates the poor interfacial properties between the fiber and the resin. However, it can be seen from the fracture shape of composites in Figure 11(b) to (d) that some resins adhere to the surface of fibers, which suggests the good interfacial properties between the fiber and the resin. Moreover, the matrix resin with many irregular strips has good toughness due to the high molecular weight at the high polymerization temperature.

SEM micrographs for fracture surfaces of GF/pCBT composites with different polymerization temperatures: (a) 180°C, (b) 190°C, (c) 200°C, and (d) 210°C. SEM: scanning electron microscopy; GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate.
In summary, both the viscosity average molar mass and the degree of crystallinity of pCBT resin matrix in GF/pCBT composites reached the maximum value at the catalyst loading of 0.5% at 190°C. Also, the pCBT resin can impregnate GF well according to the SEM micrographs of the tensile fracture surfaces of composites under this condition. Therefore, the GF/pCBT composites prepared with 0.5% catalyst at 190°C have the best mechanical properties with the tensile strength of 549 MPa, the flexural strength of 585.2 MPa, and ILSS of 47.1 MPa.
Effects of the cooling treatment on the mechanical properties of GF/pCBT composites
The GF/pCBT composites prepared by VARTM were heated up to 250°C above the melting point of pCBT resin and kept for 5 min, which made the matrix resin melt completely. Then, the quenching process and the annealing process were carried out in the conditions of rapid cooling using ice water and slow cooling in the air, respectively. The mechanical properties of the composites with different cooling methods were listed in Table 4. It can be seen that the annealing treatment improves the mechanical properties of untreated composites, while the quenching treatment reduces the mechanical properties of untreated composites. Compared to the quenching process, the tensile strength, flexural strength, and ILSS of the composites with the annealing treatment increase by 9.8%, 17.3%, and 8.9%, respectively. The modulus changes slightly. The main reason is that the crystal size of matrix resin increases and the crystallization becomes more complete in the annealing process, which results in higher degree of crystallization of polymer and better mechanical properties of the GF/pCBT composites. However, the crystallization of pCBT resin is not completed, and the amorphous components increase in the quenching process, which leads to a reduction in the mechanical properties of GF/pCBT composites.
Mechanical properties of the GF/pCBT composites with different thermal treatments.
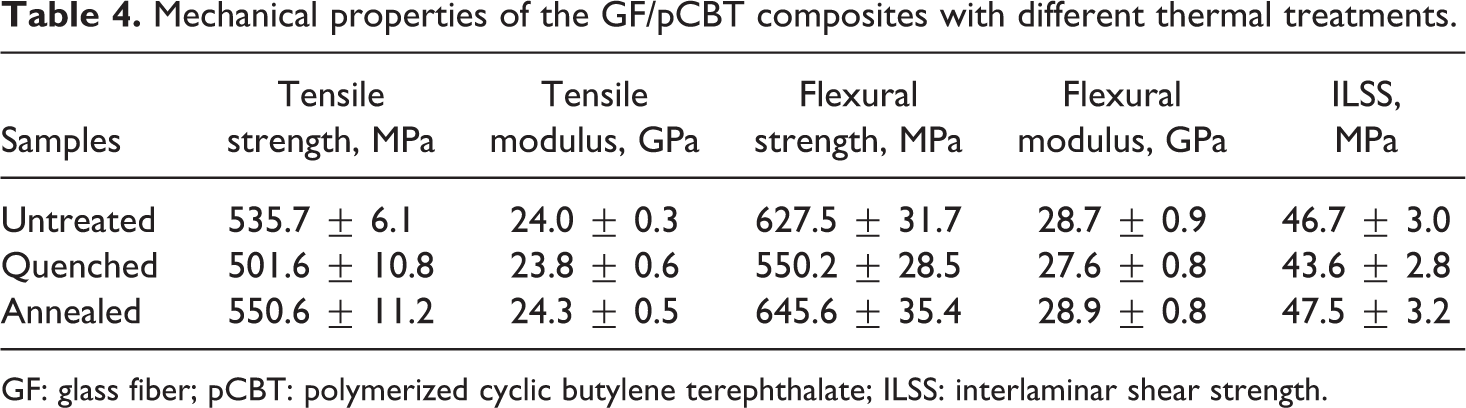
GF: glass fiber; pCBT: polymerized cyclic butylene terephthalate; ILSS: interlaminar shear strength.
Conclusion
The GF/pCBT composites were prepared via VARTM. The chemical rheological properties of pCBT resin in the in situ polymerization process suggest that the injection time is enough to manufacture large-scale thermoplastic composite laminate at 190°C using butytin tri(2-ethyhexoate) as the catalyst. Both catalyst content and polymerization temperature have significant effects on the molecular weight and the degree of crystallinity of pCBT resin in GF/pCBT composites. The viscosity average molar mass of pCBT resin obtained from the GF/pCBT composites is lower than that of the pure pCBT resin because the active groups (the acidic groups and hydroxyl groups) on the surface of GF might terminate the polymerization reaction of pCBT. The Ozawa analysis shows that the crystallization rate of pCBT resin increases with the molecular weight, while decreases with the temperature. The composites prepared with 0.5% catalyst at 190°C have the best mechanical properties with the tensile strength of 549 MPa, the flexural strength of 585.2 MPa, and ILSS of 47.1 MPa. Microstructure analysis of SEM images also shows good agreement with the mechanical properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from the Key Project of Chinese National Programs (2016YFB0101704), Chinese Academy of Sciences and Technology Services Network program (KFJ-SW-STS-150), Ningbo Key Project (2015S1004), Zhejiang Provincial Natural Science Foundation (LQ16E030001), Ningbo Natural Science Foundation (2016A610257), and China Scholarship Council (201404910243).




