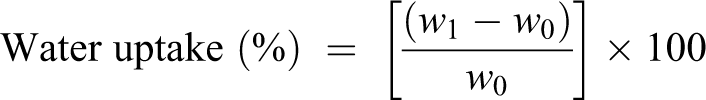Abstract
Agave Americana (agave) and Agave sisalana (sisal) fibres belong to the same family of natural fibres. Both the fibres were treated with alkali. Interestingly, alkali-treated agave fibres displayed a clean and smooth surface, whereas alkali-treated sisal fibres showed a rough surface due to the rupture of alkali-sensitive bonds. This indicated that the sisal fibres are more susceptible to alkali treatment as compared to agave fibres. The experimental studies of chemical composition, Fourier transform infrared spectroscopy and X-ray diffraction were also carried out. Both alkali-treated agave and sisal fibres were used to reinforce polyfurfuryl alcohol (PFA) matrix. The effect of these fibres on the mechanical and thermo-mechanical properties of PFA composites was examined. Mechanical, thermal and thermo-mechanical properties of the composites were studied. The results indicated the improvement in mechanical and thermal properties of the reinforced PFA.
Introduction
Natural fibres are naturally occurring composites consisting mainly of cellulose fibrils embedded in lignin matrix. According to John and Thomas, 1 the reinforcing efficiency of natural fibres is related to the nature of cellulose and its crystallinity. Petroleum-based composites are widely used but they do not degrade in a landfill, and thus they are raising serious environmental concerns. 2 Therefore, environmental compatible alternatives such as the use of natural fibres as reinforcement in natural polymers are being explored by researchers all over the world. 1 –6 The benefits of natural fibre -reinforced composites are derived from the attributes of the cellulosic fibres, such as renewability, biodegradability, low cost, low density, acceptable specific mechanical properties, ease of separation and carbon dioxide sequestration. 3
Studies on mercerization of natural fibres such as agave, 4 hemp, 5 flax 6 and coir 7 have been investigated. It has been observed that higher sodium hydroxide (NaOH) concentrations (>5%) alter the fine structure and morphology of the fibres as well as conformation of cellulose chains. NaOH penetrates the cellulose fibre and the material swells thereby inducing a considerable change in crystallinity. 4 The cellulose chains rearrange from native cellulose I to cellulose II. Since it has been reported that the crystallinity index (CI) strongly depends on the alkali concentration, it reaches its highest value for 2 wt% NaOH due to better packing of cellulose chains that give good mechanical and thermal properties for cellulose. 4 Hence, in this research work, we have used 2 wt% NaOH solution for alkali treatment of natural fibres. The improvements in fibre strength by using 2 wt% NaOH solution have also been reported. 5 –7
Natural fibres such as agave and sisal are interesting environmentally friendly alternative to the use of glass fibres as reinforcement in polymer-based engineering composites. 8 Agave fibres are extracted from the leaves of certain agave plants. These natural cellulosic fibres are characterized by a high hydrophilicity, a low density, a high tenacity and a great extensibility in comparison with other textile fibres. 9 Agave and sisal belong to the same family of lignocellulosic fibres. Traditionally, agave fibres have been used for commercial (rope, fibres, mescal and tequila), ornamental (yucca, century plant and mother-in-law’s tongue) and medicinal applications (steroid extraction and pre-Columbian antibacterial salves). 10 On the other hand, sisal fibre has short renewal times and grows wild in the hedges of fields and railway tracks. 11 The length of sisal fibre is between 1.0 and 1.5 m and the diameter is about 100–300 μm. 12 The chemical compositions of sisal fibres have been reported by several groups of researchers. 13,14 Wilson 13 showed that sisal fibre contains 78% cellulose, 8% lignin, 10% hemicelluloses, 2% waxes and about 1% ash by weight. Nonetheless, Rowell et al. 15 found that sisal contains 43–56% cellulose, 7–9% lignin, 21–24% pentosan and 0.6–1.1% ash. More recently, Joseph et al. 16 reported that sisal contains 85–88% cellulose. These large variations in chemical compositions of sisal fibre are a result of its different source, age and measurement methods. Chand and Hashmi 14 confirmed that the cellulose and lignin contents of sisal vary from 49.62% to 60.95% and 3.75% and 4.40%, respectively, depending on the age of the plant. Sisal fibres are widely used for many applications including ropes for the marine and agriculture industry as well as composites. 17,18 In this study, variations in NaOH treatment with respect to time duration are examined. CI for both agave and sisal fibres was evaluated.
Polyfurfuryl alcohol (PFA) is a hydrophobic biopolymer with improved thermal stability. 19 It has not been explored intensively in composite materials. In this study, we investigate agave and sisal fibre-reinforced PFA composites.
Experimental
Materials
Agave Americana (agave) and Agave sisalana (sisal) were supplied by a farm in Graff Reinet near Port Elizabeth, South Africa. NaOH pellets, potassium hydroxide and sulphuric acid of 99.9%, 85% and 98% purity, respectively, were obtained from Minema Chemicals (South Africa), and sodium chlorite with 80% purity was obtained from Sigma Aldrich (South Africa). Acid-catalysed PFA was prepared according to the method mentioned in the provisional patent filed by Kumar and Anandjiwala. 19 The acid catalyst p-toluenesulfonic acid (PTSA) of 98.5% purity was purchased from Sigma Aldrich and was used for the in situ polymerization of furfuryl alcohol (FA). FA, 97% purity, was purchased from Sigma Aldrich. All chemicals were used as received without further purification.
Methods
Preparation of agave and sisal fibres
The fibres were treated with 2 wt% aqueous NaOH for 1 h in order to remove lignin and hemicellulose. After an hour, the alkali-treated fibres were subsequently washed several times with running tap water followed by deionized water to pH neutral. They were dried at 50°C for overnight and cut to equal lengths of 0.5 cm.
Following the above procedure, the chemical composition of agave and sisal fibres was optimized. Variations in alkali treatment conditions such as time duration were examined, and comparable fibre composition and CI were obtained after sisal fibres.
Preparation of PFA-based composites
PFA-based composites were prepared in a silicon pan rubber-based container. PTSA catalyst (0.3 phr) was added drop wise to the 200 ml FA monomer which was previously kept in the fridge (−4°C) overnight. Two hundred millilitres of PFA charged with acidic catalyst was added to the fibres as described in a patent. 19 In the first stage, the fibres were laid manually in a silicon rubber-based container to obtain uniform dispersion. In the second stage, FA charged with acid catalyst was poured slowly in the silicon rubber-based container. The resulting mixture was then left undisturbed at room temperature for 24 h for a polymerization of PFA. The composites were then dried in an oven at 50°C for 5 days. This was followed by further drying at 100°C for 1 h. The temperature was then increased to 160°C for curing, and the composites were dried further for 30 min in the oven and were allowed to cool down to room temperature before characterization.
Chemical composition of agave and sisal fibres
Untreated and alkali-treated fibres were used to determine chemical compositions. The lignin content was analysed by reaction with sulphuric acid using a standard method recommended in TAPPI-T222 om-88, and the hemicellulose content was obtained as described in TAPPI T19m-54.
Characterization
Scanning electron microscopy
Scanning electron microscopy (SEM) measurements were carried out using an FEI Quanta 200 (FEI Co., Eindhoven, the Netherlands) electron microscope and operated at an accelerating voltage of 20/10 kV. Composites were prepared by freezing them in liquid nitrogen before fracturing to expose the cross-sectional area for a visibility of fibre distribution and morphology.
Dynamic mechanical thermal analysis
Dynamic mechanical thermal analysis was carried out on a dynamic mechanical analyser (DMA8000; PerkinElmer, Waltham, Massachusetts, USA) in dual cantilever mode at a frequency of 1 Hz. The dimensions of the composites tested were 5 and 1 cm in length and width, respectively. The test temperature ranged from 22°C to 240°C, with the heating rate of 2°C min−1.
Tensile and flexural measurements
The tensile strength, elongation at break and Young’s modulus of the composite samples were measured according to ASTM methods D882 (E) using an Instron 3369 testing machine (Instron, Norwood, Massachusetts, USA) at a strain rate of 10 mm min−1. Three point bending tests to determine flexural strength were also carried out according to ASTM D-790 sing a universal testing machine (model 3369).
Water uptake
The water uptake experiments were measured according to the standard test method for water absorption of plastics, ASTM (designation: D 570-98, reapproved 2005). The composite materials were dried in the oven at 50°C for 24 h, cooled in a desiccator, and immediately weighed (w 0) to the nearest 0.001 g. The specimens were immersed in deionized water for 144 h at room temperature. After 24 h, the specimens were removed from water, wiped with a paper towel to remove water on the surface, and immediately weighed (w 1) to the nearest 0.001 g. The samples were immersed in deionized water once again for further investigations. After 48 h, a second measurement was taken following the same procedure as above, and finally the third measurement was taken after 144 h.
The water uptake of the composites was calculated as follows:
Fourier transform infrared spectroscopy
Fourier transform infrared (FTIR) of the PFA-based composites was carried out on a Spectrum 100 FTIR (PerkinElmer). The range used was between 500 and 4000 cm−1.
XRD analysis
X-ray diffraction (XRD) patterns of the samples were recorded using Philips PW 1830 X-ray diffractometer with copper K
α radiation (λ = 0.154 nm) (Karlsruhe, Baden-Württemberg, Germany). The CI was determined using two methods: The XRD peak height method: In this method, CI is calculated from the height of the 002 peak (I
002) and the height of the minimum (I
AM) between the 002 peak and the 001 peaks. In the second method, the sample crystallinity was determined using XRD deconvolution method. This method is done by taking the amorphous and crystalline contribution to the diffraction intensity.
20
I 002 is the counter reading at peak intensity at 2θ angle close to 26° representing crystalline material, and I AM is the counter reading at peak intensity at a 2θ angle close to 18° representing amorphous material in cellulose fibres.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was carried out with a PerkinElmer Pyris 1 TGA thermogravimetric analyser using nitrogen as a purge gas and a heating rate of 10°C min−1. The TGA programme was conditioned to ramp the temperature linearly from room temperature to 700°C under a flow of nitrogen. The temperature of the sample was monitored, and the loss of weight of the sample was expressed in terms of percentage weight loss.
Results and discussion
Chemical composition of agave and sisal fibres
Standard method
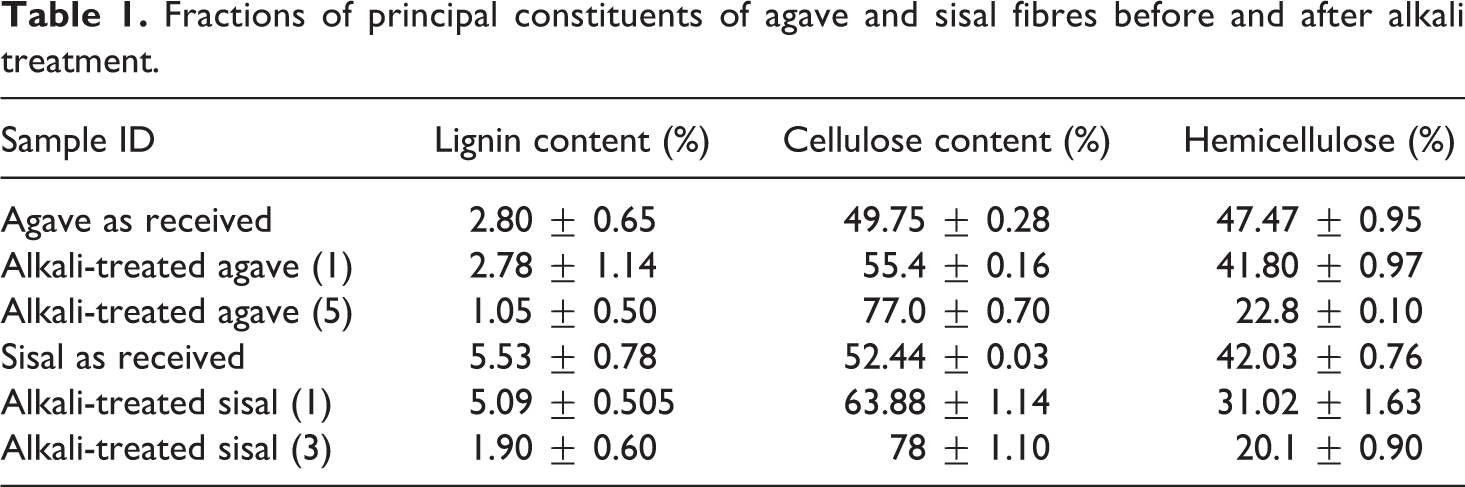
Agave fibres revealed the highest total hemicellulose content, whilst sisal fibres showed the most lignin. On the other hand, sisal fibres showed the highest cellulose content, that is, about 11% higher than agave fibres. The changes in chemical composition of these fibres are attributed to differences in fibre growth time. 21 Furthermore, it is apparent from Table 1 that when agave and sisal fibres were subjected to alkali treatment once, the values of fibre components vary significantly (Table 1). This could be due to susceptibility of sisal fibres in NaOH. However, equivalence in principal components of agave and sisal fibres was observed when sisal fibres were alkali treated three times and agave fibres were subjected to alkali treatment five times, respectively.
Fractions of principal constituents of agave and sisal fibres before and after alkali treatment.
Structural characterizations of agave and sisal fibres
FTIR spectroscopy
Figure 1 and 2 show IR spectra of agave and sisal fibers before and after alkali treatment. Hydroxyl (–OH) bands are represented at 3333 cm−1. The bands at 2903 cm−1 represent C–H stretching vibrations of methyl groups while bands at 1373 cm−1 represent C–H bending vibrations in natural fibers. The intensities of all these bands increased with alkali treatment due to removal of lignin and hemicellulose (Figure 1). However, those attributed to aromatic vibrations from structures of both lignin and hemicellulose at 1510 and 1452 cm−1 are either not observed or less intense 4 –7 . The same observation was sustained for different treatments of the fibres as shown in Figure 2.
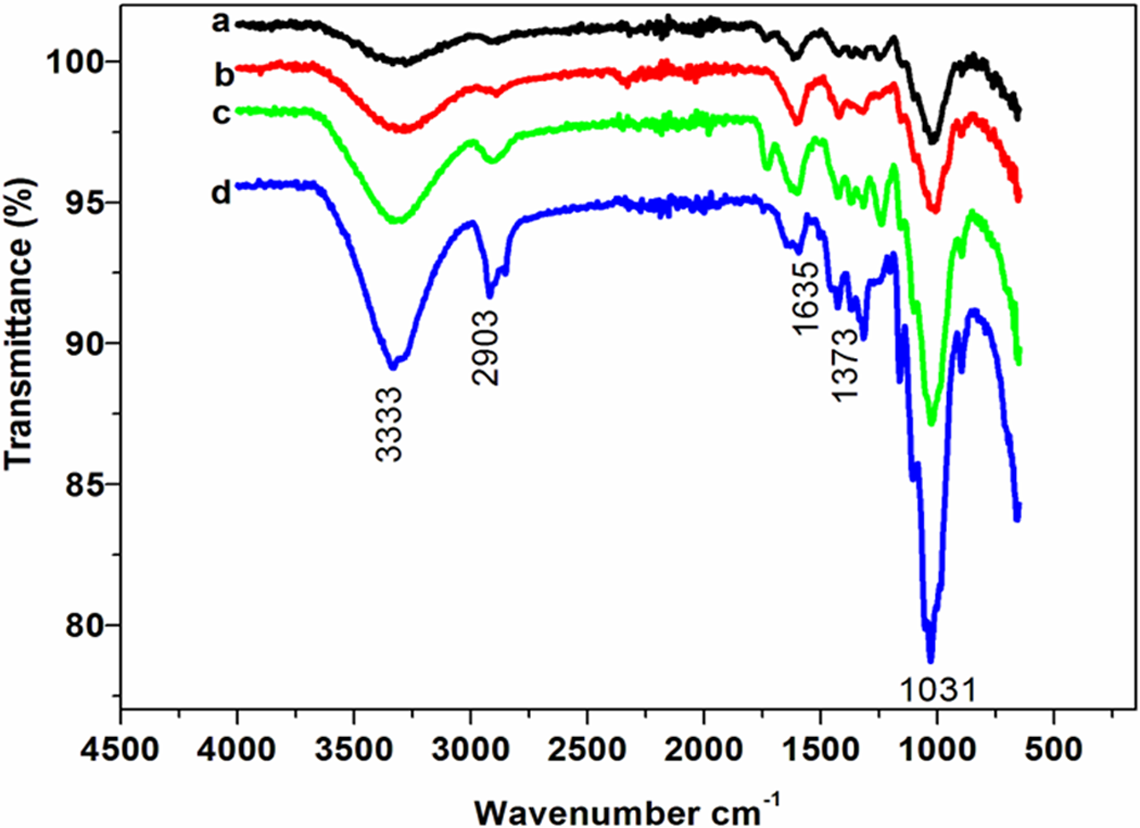
Fourier transform infrared spectra of agave and sisal fibres before and after alkali treatment: (a) agave as received, (b) sisal as received, (c) alkali-treated agave fibres (1) and (d) alkali-treated sisal fibres (1).
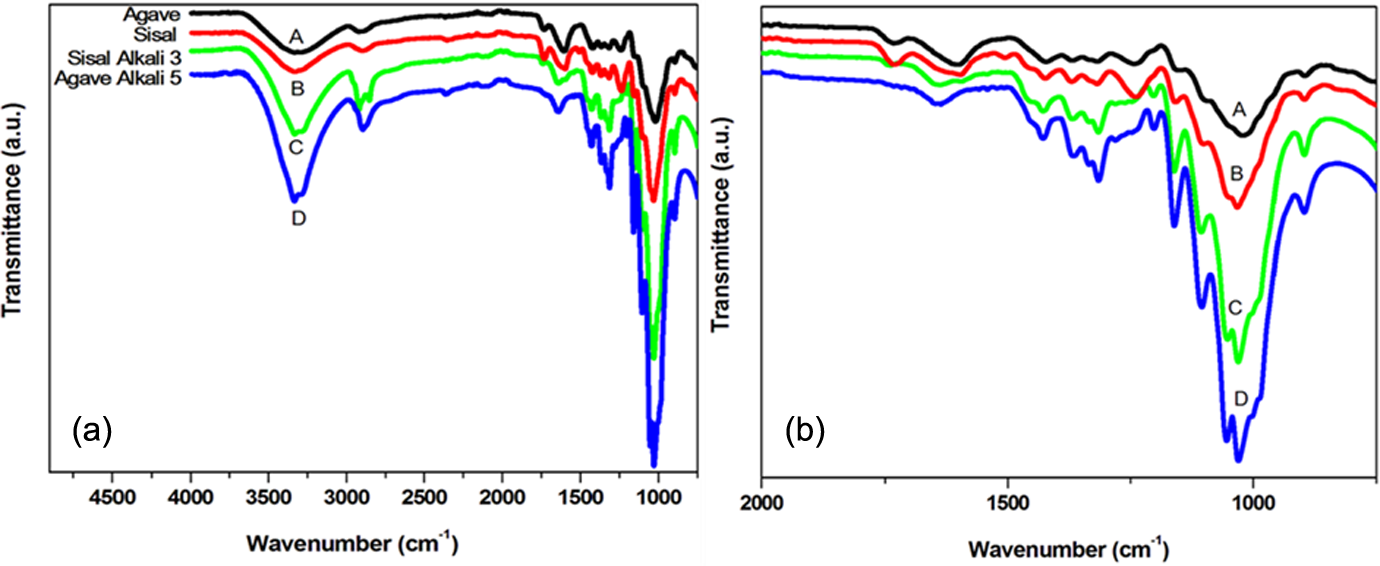
FTIR spectra of agave and sisal fibers before and after alkali treatment: Agave fibers were alkali treated for 5 times and sisal fibers were alkali treated for 3 times.
X-ray diffraction
Figure 3 shows XRD patterns of agave and sisal fibres before and after alkali treatment. The diffraction lines pertain to cellulose I type, as there was no doublet in the main peak at 2θ = 22.6°. 22 The diffractograms of both untreated (as received) and alkali-treated samples display a well-defined main peak around 2θ = 22.6°, associated with the diffraction plane (002) of cellulose I. 23 A broad unresolved diffraction peak in the range of 13–18° (2θ) corresponds to (101) crystallographic planes of cellulose. In general, when the crystalline cellulose content is high, these two peaks are sharp and when the fibre contains large amounts of amorphous material (such as lignin, hemicellulose, pectins and amorphous cellulose), these two diffraction peaks appear as one broad peak. 24 The diffractograms of untreated samples exhibit large amounts of lignin, hemicellulose, pectins and amorphous cellulose. However, alkali treatment removed some of the amorphous material from the fibres. This is indicated by more defined diffraction peaks attributed to (002) and (101) crystallographic planes of cellulose (Figure 3). It is apparent from Figure 3 that sisal fibres are more crystalline than agave fibres; the intensities of both peaks (22.6° and 16°) have increased. However, at point of equivalence, the crystallinity of agave and sisal was reasonably similar (Table 2). Table 2 shows the crystalline size of cellulose obtained before and after alkali treatment. The values of the particle size of cellulose crystallites were reasonably similar (Table 3). XRD deconvolution and XRD peak height methods were used to determine CI. The results showed that the CI of the alkali-treated sisal fibres (1) is higher as compared to that of the agave fibres (1) in both cases (Table 2). However, variations in alkali treatment conditions (duration) gave rise to relatively similar CI values for both agave and sisal fibres (Table 2).
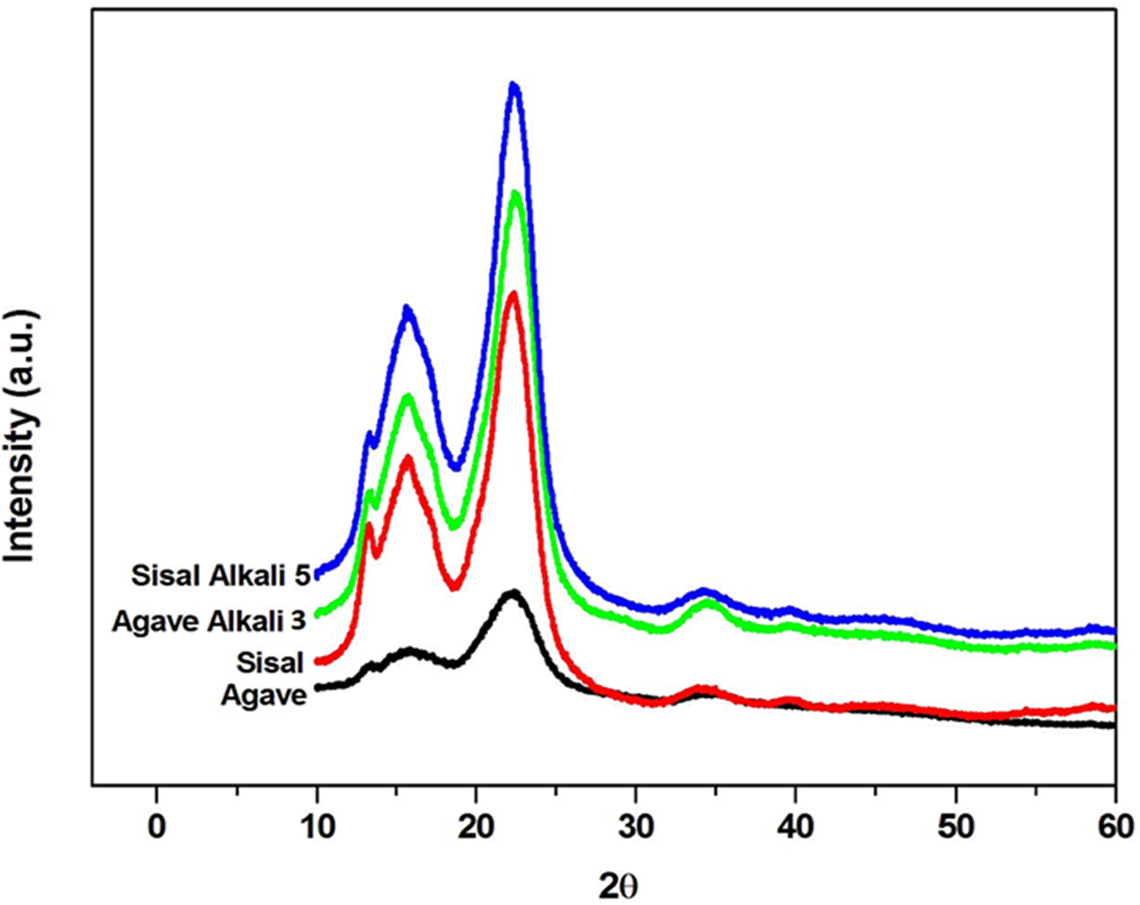
X-ray diffraction profiles of agave and sisal fibres before and after alkali treatment.
Crystallinity index (%) of agave and sisal fibres before and after alkali treatment.
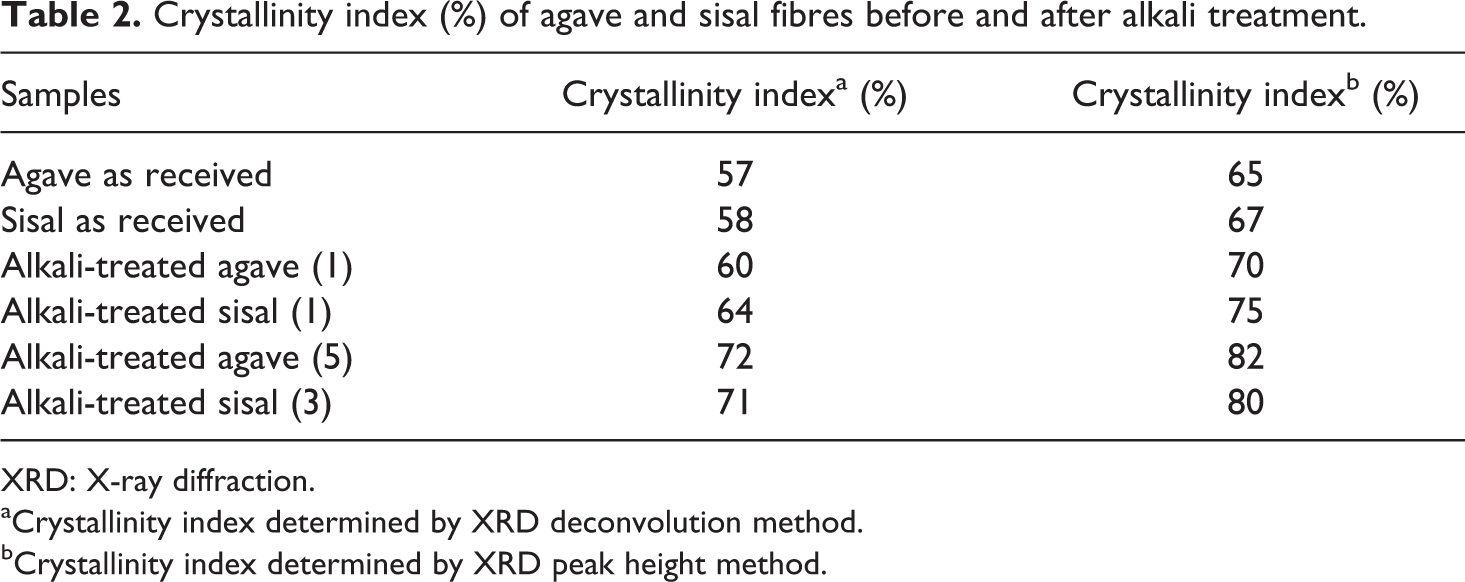
XRD: X-ray diffraction.
aCrystallinity index determined by XRD deconvolution method.
bCrystallinity index determined by XRD peak height method.
Crystallite sizes of agave and sisal fibres before and after alkali treatment.

The crystallite sizes were calculated using Scherrer equation and Table 3 shows comparable values at point of equivalence (Table 3).
Scanning electron microscopy
Figure 4 illustrated more uneven surface morphology of the as-received agave fibres as compared with the as-received sisal fibres. The soft lumps on surface of agave fibres are attributed to waxes and pectins present on the surface (Figure 4). However, when both fibres were alkali treated once, sisal fibres showed susceptibility towards degradation by alkali treatment (Figure 5). Figure 5 illustrated a rougher surface morphology of alkali-treated sisal fibres in comparison with alkali-treated agave fibres. Roughness, shrinkage and entanglement of sisal fibres were due to the rupture of alkali-sensitive bond and dissolution of uronic acid present in hemicellulose in alkali solutions. 25 The results confirmed that sisal fibres are susceptible to degradation whereas agave fibres showed slight resistance (Figure 5). Alkali treatment of sisal fibres gave rise to the breaking down of the cell wall (lignin) thereby exposing the microfibrillated cellulose. The similar observation is common in literature. 26,27

SEM images of agave and sisal fibres before alkali treatment. SEM: scanning electron microscopy.

SEM images of alkali-treated agave and sisal fibres (both alkali treated once). SEM: scanning electron microscopy.
To balance the degree of crystallinity of both fibres, a series of experiments were performed and the experimental results showed that when agave fibres were alkali treated five times, its CI was comparably similar to that of sisal fibres alkali treated three times. At equivalence level, SEM images illustrated that after alkali treatment, both fibres looked cleaner on the surface confirming the removal of lignin and hemicellulose as predicted by FTIR and XRD results. However, at the point of equivalence, the agave fibres treated five times seemed to have the better surface roughness, sharp edges and ridges in comparison to sisal fibres alkali treated three times (Figure 5). On other hand, sisal fibres treated three times seemed to have more loose fibres spirals on the surface than agave fibres. Previous studies also indicated that those competing surface features from the fibres are essential for reinforcement of polymers.1,12,28 Rong et al. 26 argued that the vacancies outside and inside sisal bundles created by alkali treatment facilitate the penetration of polymer matrix. The diameters of the fibres after alkali treatment were ranging from 5 to 21 µm for agave fibres (5) and 5 to 12 µm for sisal fibres (3) (Figure 6). The reduction is known as a result of a breakdown of fibre bundles by alkali treatment. 27

SEM pictures of agave and sisal fibres after alkali treatment. SEM: scanning electron microscopy.
Properties of PFA/agave and PFA/sisal composites
Water uptake
Figure 7 represents water resistance of PFA and its composites. It can be seen that PFA is hydrophobic with very low water uptake value; however, the incorporation of natural fibres increased its tendency to absorb water. Water uptake experiments showed that PFA/agave composites have a high tendency to absorb water as compared to PFA/sisal composites. The percentage water uptake increased with increasing time duration, as illustrated in Figure 7. All natural fibres are hydrophilic due to the presence of hydroxyl groups in cellulose materials. Nevertheless, the degree at which they absorb water depends on the nature of the fibre and fibre chemical composition. The contents of the main constituents (cellulose, hemicellulose and lignin; Table 1) showed that agave fibres exhibited the highest total hemicellulose content whereas sisal fibres achieved a higher lignin content (Table 1). Hemicellulose is very hydrophilic; hence the higher water uptake value for PFA/agave composites was achieved, whereas lignin has been shown to reduce the water absorption and thickness swelling of the composites. 29
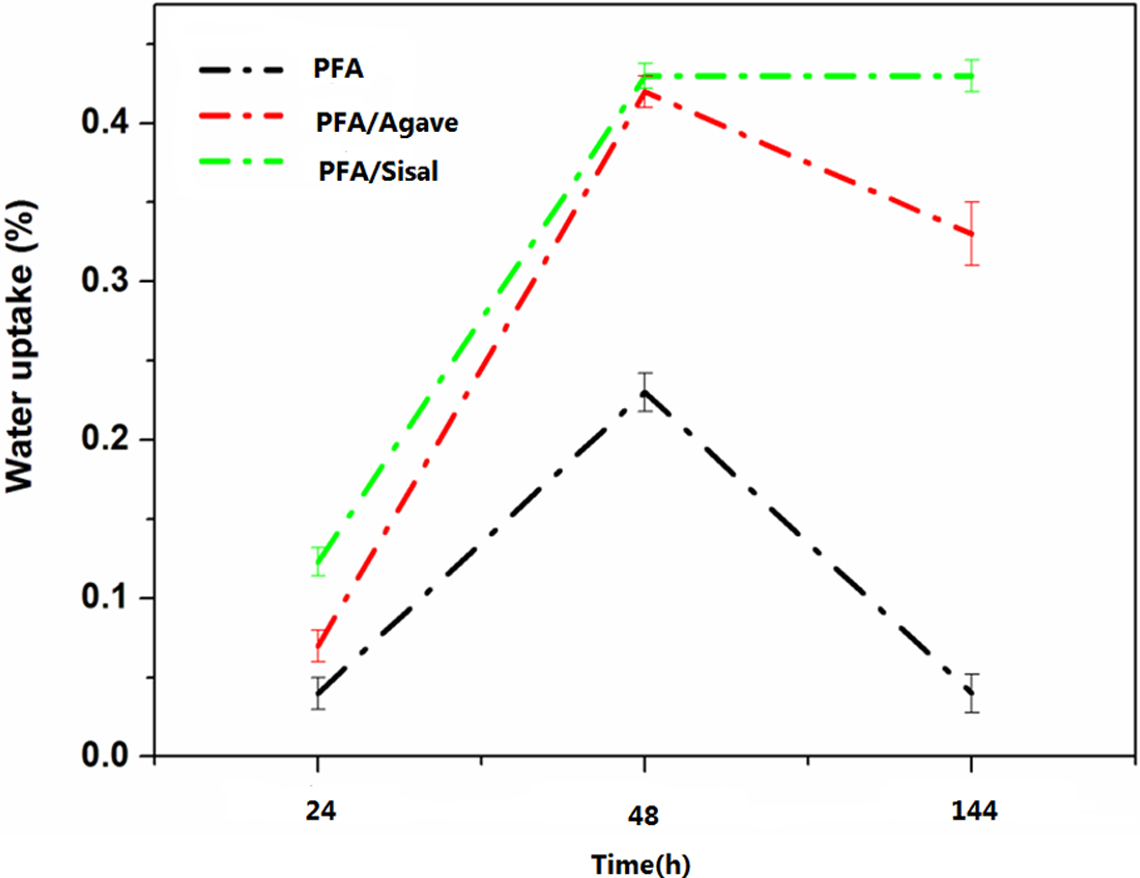
Water uptake properties of the neat PFA and PFA-based composites, agave and sisal fibres alkali treated once. PFA: polyfurfuryl alcohol.
Mechanical properties of PFA-based composites
According to John and Anandjiwala, 30 flexural strength is a combination of the tensile and compressive strengths, which directly varies with the interlaminar shear strength. During flexural testing, tension, compression and shearing mechanisms take place simultaneously. The flexural properties of different composites are compared in Figure 8, and in general, the fibre reinforcement led to increased flexural strength compared to pure PFA which has 47 MPa. This increase in flexural strength of natural fibre-reinforced composites also suggests good impregnation of the polymer into the fibres. In composite materials, good impregnation of polymer proposes enhancement in interfacial bond strength. 31 –33 There was no much difference in flexural modulus observed except for a slight increase after fibre reinforcement.
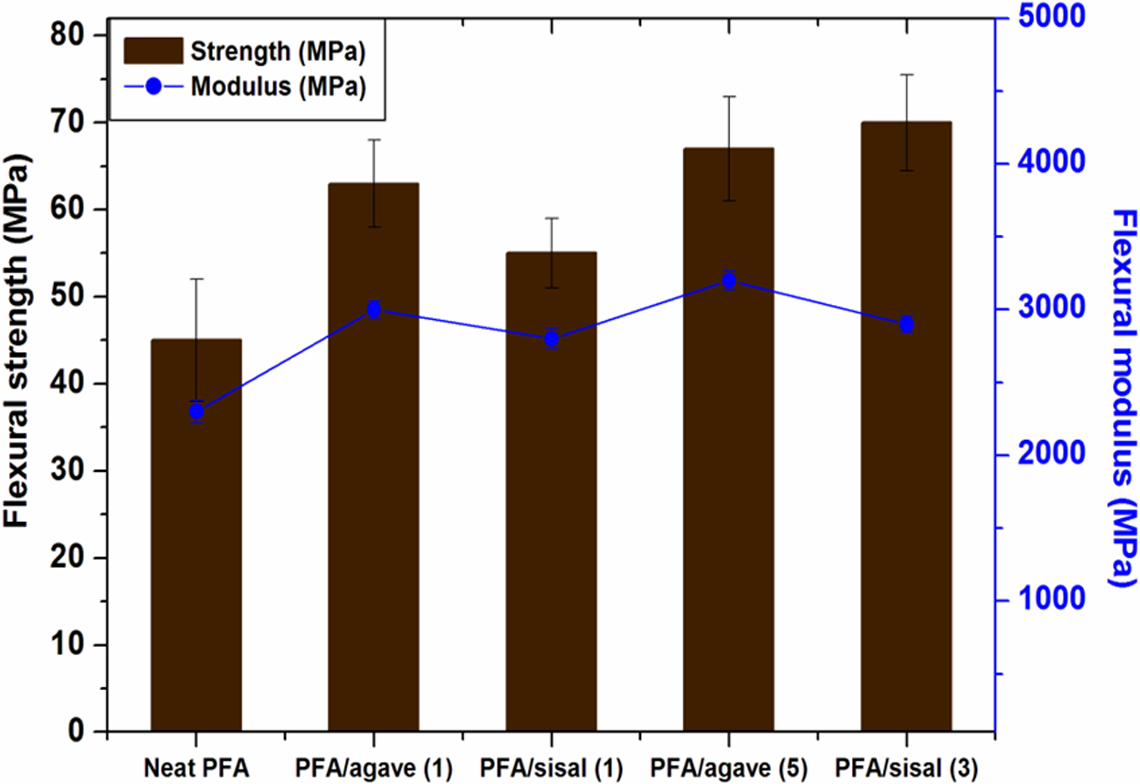
Flexural properties of the neat PFA and PFA-based composites. PFA: polyfurfuryl alcohol.
Further mechanical tests, such as tensile and impact tests, were performed on PFA-based composites. The experimental data revealed that the conditions of alkalization chosen in this study lead to enhancement in tensile strength of PFA composites. It is interesting that composites prepared from alkali-treated sisal fibres (3) showed a significant increase in tensile strength (Figure 9). The overall tensile strength of natural fibre-reinforced PFA composites increased with increasing CI. 26,34–36 Therefore, alkali treatments resulted in greater extensibility and lower stiffness for sisal fibre-reinforced composites due to structural variations in ultimate cells such as swelling and partial removal of lignin and hemicellulose from natural fibres. 36
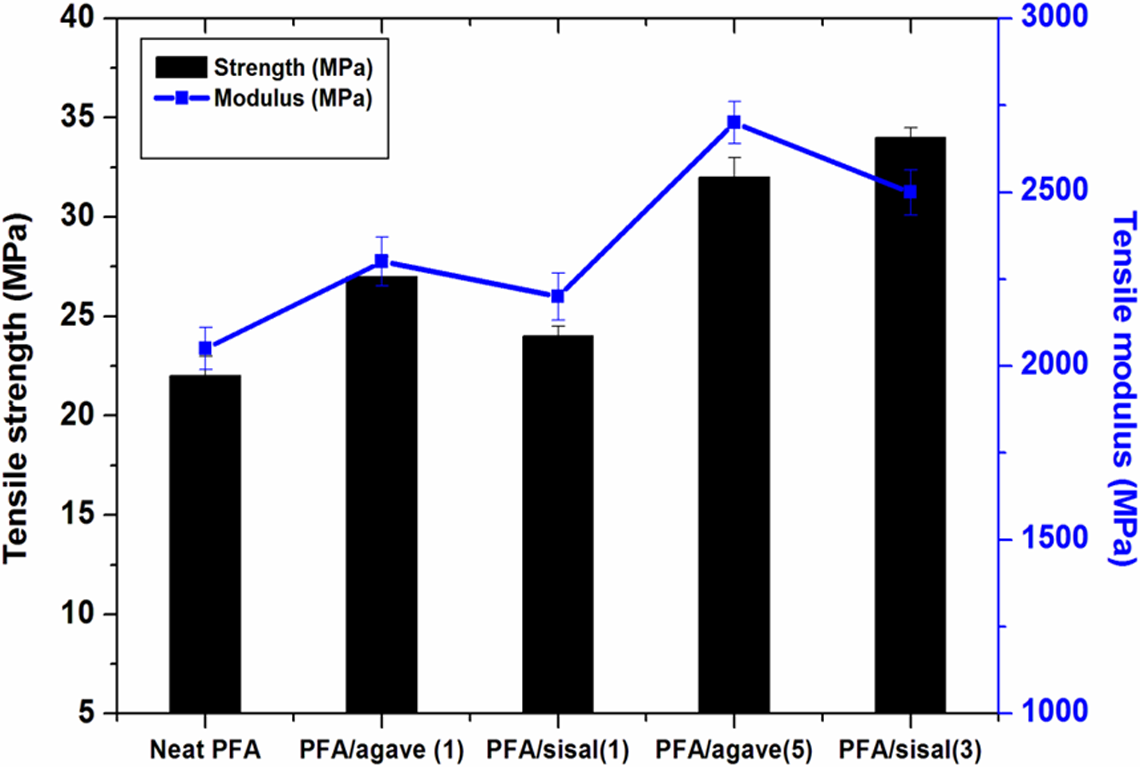
Tensile properties (strength and modulus) of PFA-based composites. PFA: polyfurfuryl alcohol.
The Charpy impact strength results for the tested materials are shown in Figure 10. PFA-based composites registered higher impact energy values as compared to neat PFA due to the presence of fibres. Furthermore, enhancement in impact strength is due to the fact that natural fibres increased the energy required to pull them out during crack propagation in the composites. PFA/agave registered slightly higher impact strength than PFA/sisal and the opposite occurred at the point of equivalence. Those could be attributed to the susceptibility of sisal fibre to NaOH treatment and partial degradation of cellulose, respectively. The two effects seemed to control the toughness of the composites, since it is well known that the impact strength is controlled by material toughness. 37
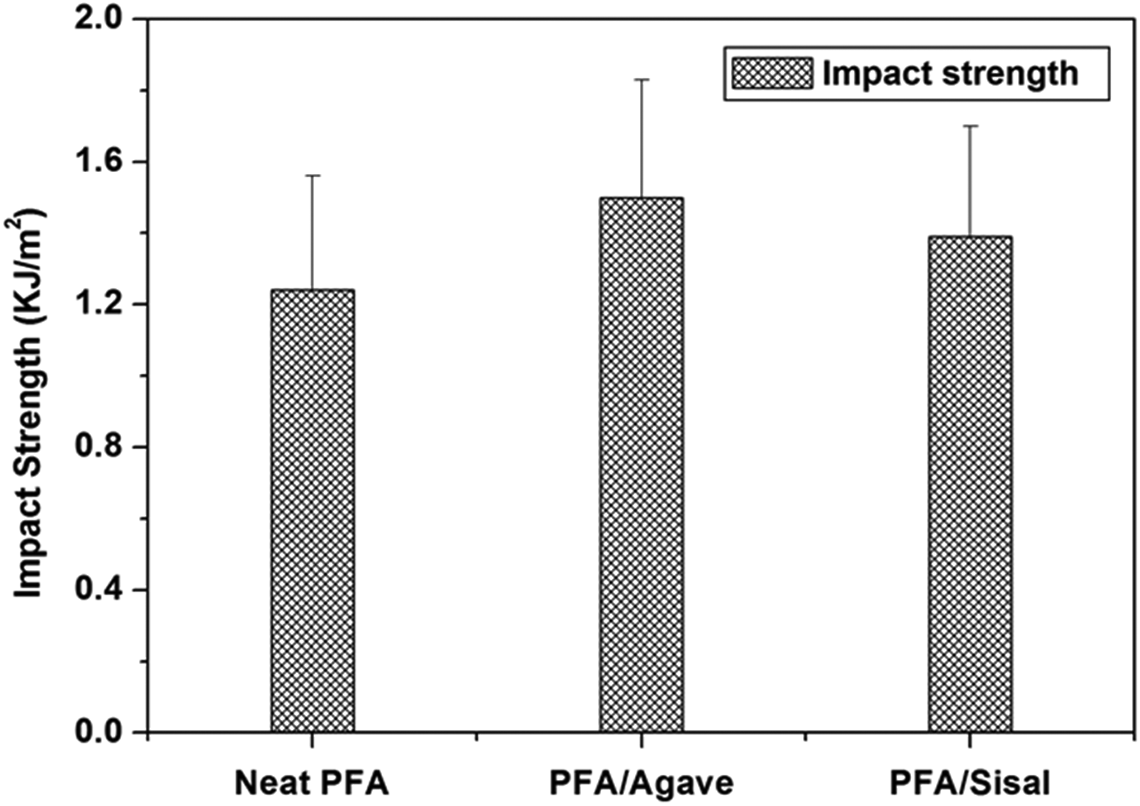
Impact strength of neat PFA and PFA-based composites (agave and sisal fibre alkaline treated once). PFA: polyfurfuryl alcohol.
Thermo-mechanical properties of PFA-based composites
The storage modulus, tan δ and loss modulus of the pure PFA and the PFA composites having 1, 3 and 5 wt% of the fibres are reported in Figures 11, 12 and 13, respectively. The presence of fibres in the PFA led to the higher storage modulus compared to PFA which suggests reinforcement effect. The fibres treated once showed less moduli than three and five times treated indicating better stress transfer and/or increased stiffness at the point of equivalence. This observation is relatable to SEM results which showed more defibrillation at the point. Agave and sisal fibre-reinforced composites displayed rather similar storage modulus at equivalent values of CI and fibre composition (Figure 11). This suggests virtually similar load transfer to PFA during sinusoidal stress.
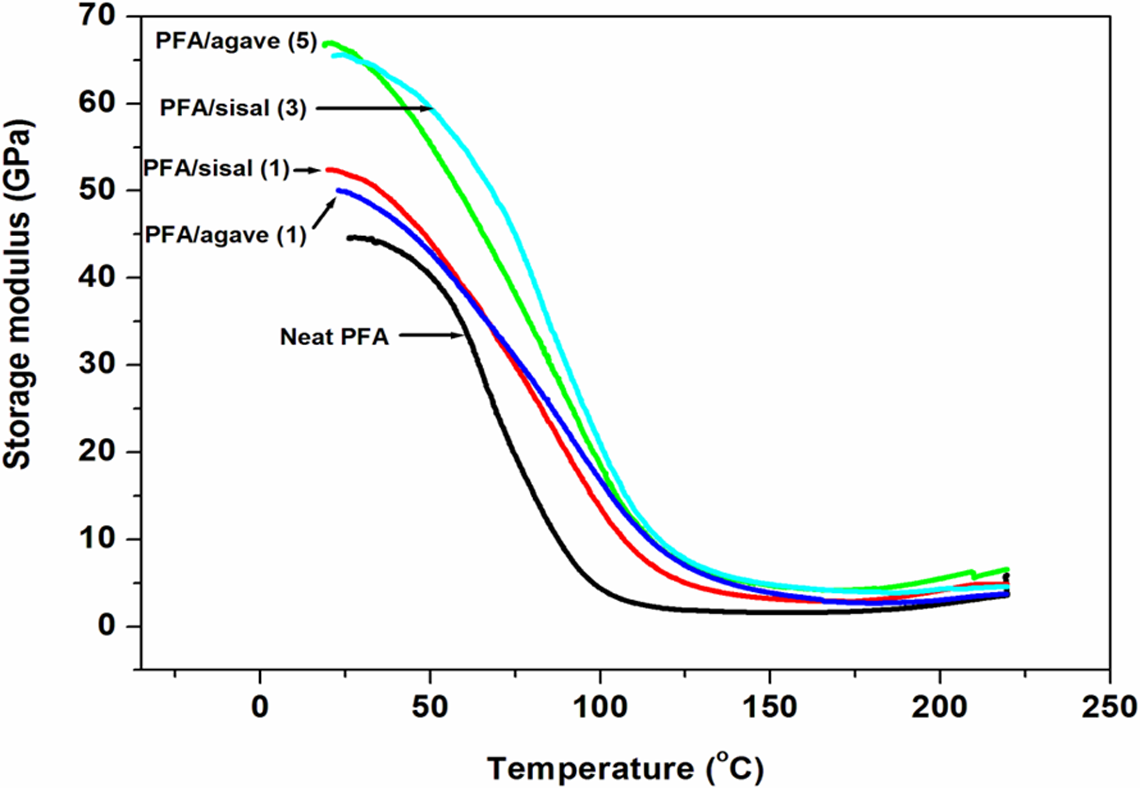
Storage modulus curves of the neat PFA and PFA based composites.
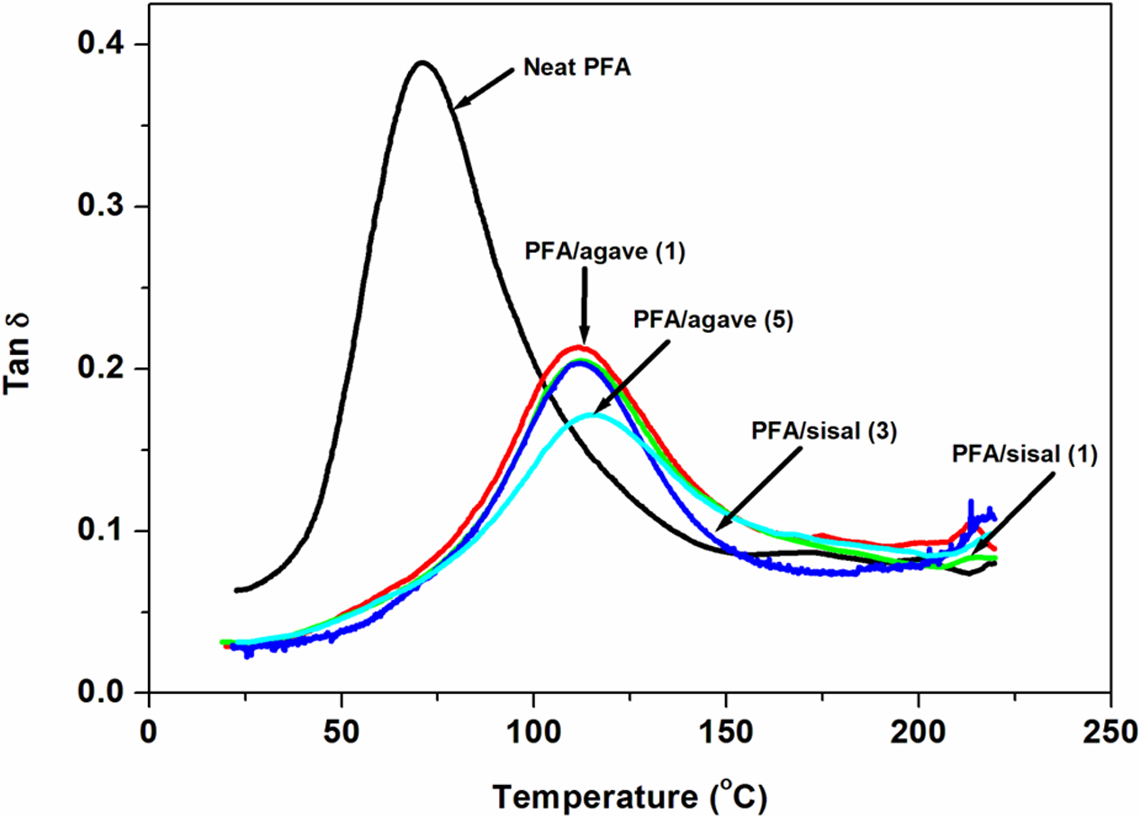
Tan δ curves of the neat PFA and PFA-based composites: agave (1) = alkali treated once, agave (5) = alkali treated five times, sisal (1) = alkali treated once and sisal (3) = alkali treated five times. PFA: polyfurfuryl alcohol.
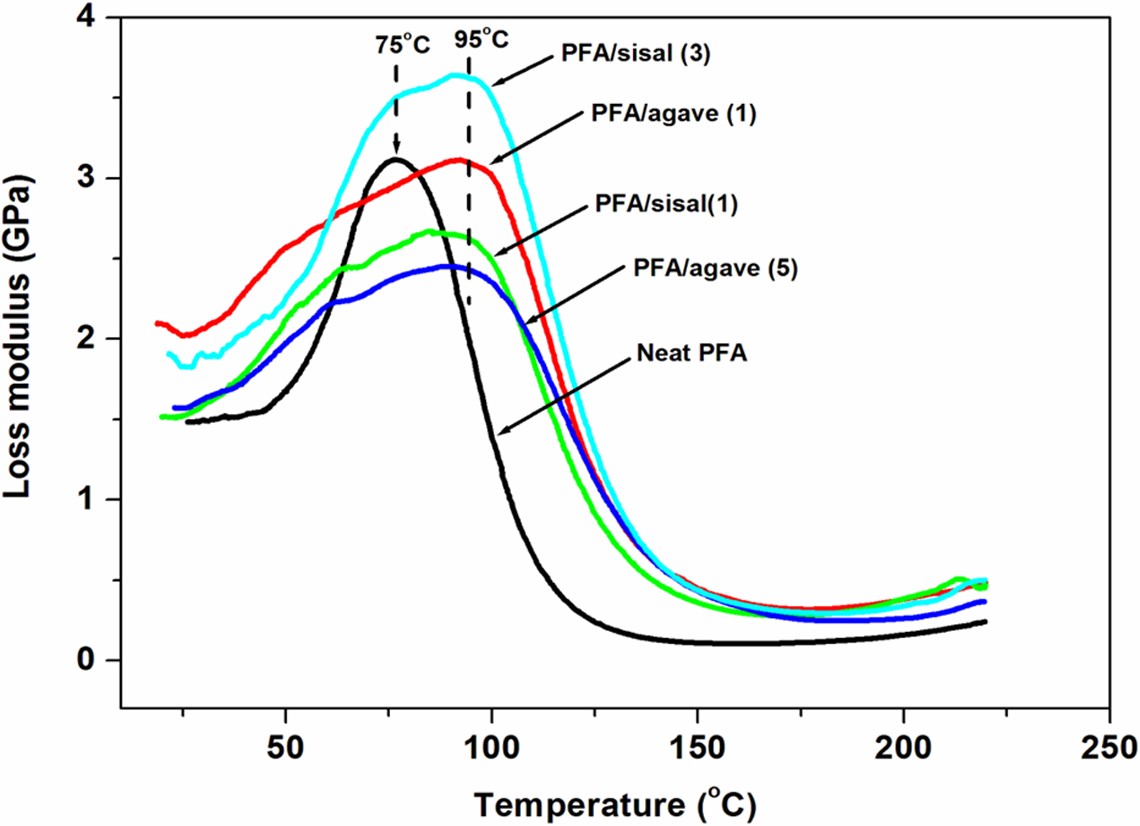
Loss modulus curves of neat PFA and PFA-based composites. PFA: polyfurfuryl alcohol.
A plots of dynamic (tan δ) as a function of temperature is shown in Figure 12. When natural fibres were incorporated, the peak intensity decreased and the peaks were shifted towards higher temperatures. The damping amplitude is reduced due to enhanced interfacial adhesion between the natural fibres and PFA matrix. Enhancement in interfacial strength resulted in improved α-relaxation temperatures of PFA-based composites. PFA/agave (1) and PFA/sisal (1) composites displayed similar effect on damping behaviour (Figure 12). However, at equivalence point, agave-reinforced PFA composites displayed better damping behaviour.
Glass transition temperature (T g) of PFA-based composites was evaluated by loss modulus curves (Figure 13). The neat PFA showed a T g value of 75°C, whilst PFA-based composites showed a higher T g values ∼95°C. The shifting of T g to higher temperatures can be associated to the decreased mobility of the matrix chains, which indicates enhancement in interfacial bond strength between the natural fibres and PFA matrix. Also, in composite materials, an increase in T g values signifies improvements in stiffness in polymers with incorporation of natural fibres. This is in agreement with elastic modulus results (Figure 9).
The thermal stability of natural fibres and the PFA based is shown in Figure 14. As expected, the PFA and its composites showed two degradation steps in the investigated temperature range as confirmed by derivative thermogravimetric (DTG) curves in Figure 15. The source of the step at lower temperature was attributed to the formation of alkylfurans due to scission of both methylene and methyne links, while at higher temperatures it was linked to scission of furanic links to form ketonic volatile compounds. 38 Curves of pure fibres, three times treated sisal, five times treated agave and PFA are shown in Figure 16. All the two fibres revealed clear two-step degradations as seen using DTG thermograms (Figure 15). A very small hump at ∼250°C is attributed to degradation of hemicellulose whilst a broad one at 317°C (agave fibre) and 327°C (sisal fibre) is due to the combination of degradation of lignin and cellulose. 39 –43 At point of equivalence, sisal-reinforced PFA composites displayed the highest thermal stability. This could results from partial cellulose degradation of the Agave as suggested by SEM results.
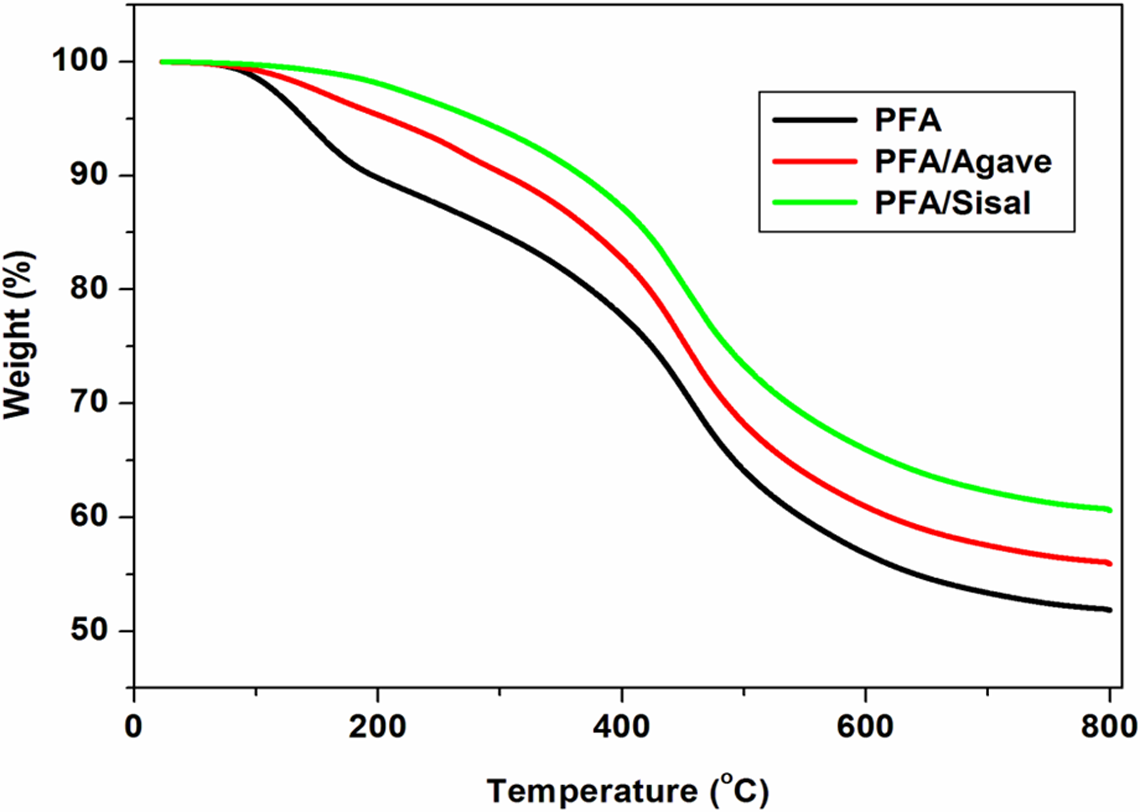
TGA profiles of the neat PFA, PFA/agave and PFA/sisal: agave and sisal fibres, alkaline treated once. PFA: polyfurfuryl alcohol.
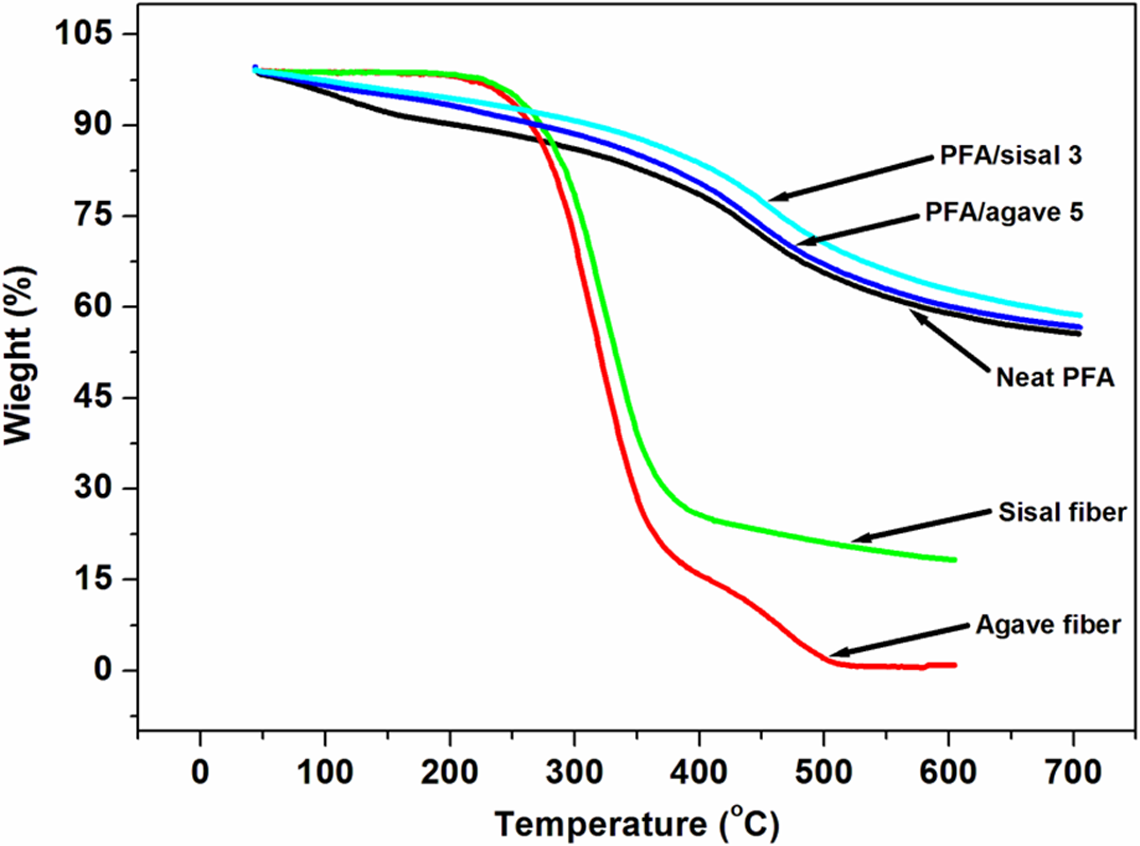
TGA profiles of agave and sisal fibres, neat PFA, PFA/agave (5) and PFA/sisal (3) composites. PFA: polyfurfuryl alcohol; TGA: thermogravimetric analysis.
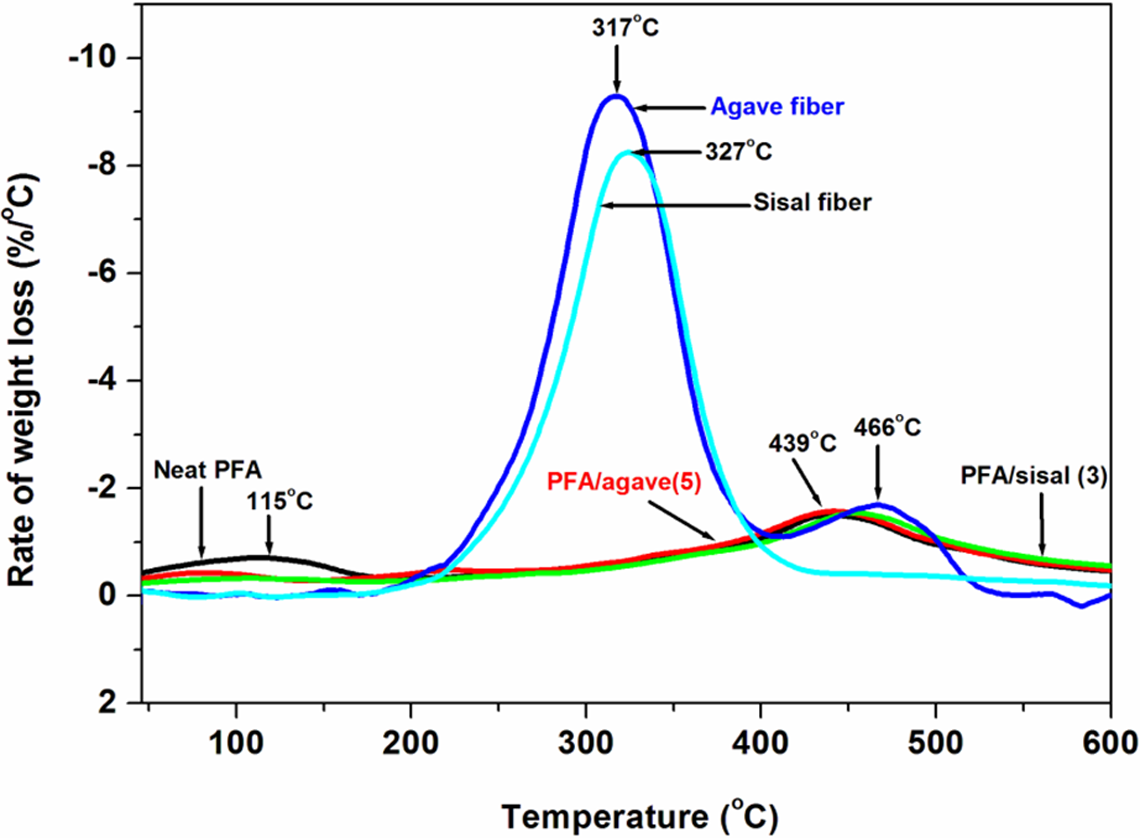
DTG of alkali-treated agave (5), alkali-treated sisal (3), neat PFA, PFA/agave (5) and PFA/sisal (3) samples. PFA: polyfurfuryl alcohol; DTG: derivative thermogravimetry.
The incorporation of agave and sisal fibres to PFA bioresin improved the overall thermal stability of PFA (Figure 14), which suggested a fairly good interfacial interaction. Sisal-reinforced composites displayed better thermal stability as compared to agave (1)-reinforced PFA composites (Figure 14). Differences in thermal stability may be due to differences in chemical compositions and better interfacial interaction (Tables 1 and 2). SEM results showed different morphologies of the fibres which could account for different interfacial interactions, since it is well known that enhancement in thermal stability of composites to natural fibres is influenced by fibre–matrix interaction. 41 The other important fact illustrated by the FTIR results is that at point of equivalence, new functional groups were introduced on the surface of both agave and sisal fibres. These functional groups also play a crucial role in enhancing the thermal stability of cellulosic materials. 25,27 It is worth to note that the TGA curves confirmed the retention of ∼45% char residue at 700°C. The same observation was observed by many researchers. 39
Conclusions
In this study, PFA-based composites were prepared from A. americana and A. sisalana fibres. Agave and sisal fibres were used to reinforce PFA. An experimental study of chemical composition, FTIR spectroscopy and XRD results indicated that to a large extent the hemicellulose and lignin were removed from the fibres. Furthermore, chemical composition experiments and XRD results showed differences in fibre composition. Quantitative analyses of XRD results investigated that alkali-treated sisal fibres have a higher CI than alkali-treated agave fibres. SEM analysis suggested that sisal fibres are more susceptible to alkali treatment than agave fibres, the surface of the alkali-treated sisal fibres appeared rough whereas the surface of agave fibres remained smooth after chemical treatment. Roughness, shrinkage and entanglement of sisal fibres were due to the rupture of alkali-sensitive bond (uronic acid in the hemicellulose).
Mechanical tests such as flexural, tensile, impact and water uptake experiments were carried out and the experimental data showed that both fibres improved the mechanical properties of the neat PFA. However, when agave fibres were used as reinforcement, the mechanical and thermo-mechanical properties improved to a greater extent. The alterations in mechanical and thermo-mechanical properties were estimated to be due to differences in fibre composition and crystallinity indices. Water uptake experiments showed that PFA/agave composites absorb to some extent higher amount of water whereas PFA/sisal composites showed a slight resistance towards water absorption. Hemicellulose is very hydrophilic; the high content of hemicellulose in alkali-treated agave fibres resulted to considerable increase in water uptake value.
Thermo-mechanical properties of PFA/agave composites were better than those of PFA/sisal composite materials. The storage modulus of PFA/agave composite is higher than that of PFA/sisal composite material and T g value increased further when agave fibres were used as reinforcement. This suggests that when agave fibres were used as reinforcing elements, the stiffness of PFA polymer improved and interfacial bond strength between agave fibres and PFA matrix enhanced. Since both sisal fibres and agave fibres belong to the same family of natural fibres, the differences in thermo-mechanical behaviour may be attributed to differences in fibre composition.
At point of equivalence, agave and sisal fibre-reinforced composites exhibited relatively similar thermo-mechanical properties. However, sisal fibres proved to have a better thermal stability as compared to agave fibres.
Footnotes
Acknowledgements
This article forms part of a research project, ‘Greener Cities in South Africa’, funded by the Green Fund, an environmental finance mechanism implemented by the Development Bank of Southern Africa (DBSA) on behalf of the Department of Environmental Affairs (DEA) and CSIR. Opinions expressed and conclusions arrived at are those of the author and are not necessarily to be attributed to the Green Fund, DBSA or DEA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.