Abstract
The aim of the work is to prepare the halogenated methacrylate-based polymer nano-aluminum oxide (Al2O3) composites and to study the effect of incorporated nano-Al2O3 on morphological and thermal studies. A functional 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres (poly(TBPMA-co-GMA); pTG) by emulsion solvent evaporation techniques, whereas (2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate)-Al2O3 nanocomposites (pTG-Al2O3 nanocomposites) were prepared by solution mixing techniques. The pTG and its Al2O3 nanocomposites were structurally characterized by Fourier transform infrared (FTIR) spectroscopy. Thermal studies of pTG and its Al2O3 nanocomposites were carried out by thermogravimetric analysis and differential scanning calorimetry . The molecular weight of the pTG was determined by gel permeation chromatography. The size distribution and morphology of the pTG and its Al2O3 nanocomposites were determined by scanning electron microscopy (SEM). The FTIR results reveal that there is no significant interaction between the polymer matrix and Al2O3 nanoparticle. The significant increase in the initial decomposition temperature and glass transition temperature of pTG-Al2O3 nanocomposites compared to its polymer was due to the incorporation of nano-Al2O3 in the polymer matrix. The SEM observation provides the information about the morphological changes that arise in polymer matrix due to the incorporation of nano-Al2O3.
Introduction
Polymer chemistry is a promising field of research in the current world. The field has high potential in discovering several novel features regarding production and application of polymers. It is a well-known fact that the performance of materials depends on their properties. The properties in turn depend on the composition, atomic structure, microstructure, defects, and so on. Based on the properties, the polymer can find its application in diversified fields of interest. One way of modifying the properties of the polymer is by making composite in which metals, 1 and inorganic nanofillers, 2 which includes layered silicates (such as montmorillonite), nanotubes (mainly carbon nanotubes, CNTs), fullerenes, silicon dioxide, metal oxides (e.g. titanium dioxide, ferric oxide, aluminum oxide (Al2O3)), and nanoparticles of metals 3 –5 (e.g. copper (Cu)) are used as a reinforcing element. Microspheres have attracted increased attention due to their unique properties and have numerous applications depending upon their material and size. Polymer microspheres with various functional groups have many applications in different fields such as solid carriers for the immobilization of biological substrates including enzymes, antibodies, and so on, 6 –8 synthetic polymer microspheres find application in controlled drug delivery, 9 –12 naturally occurring polymer hyaluronan microsphere was used for sustained gene delivery at specific site of interest, 13 and the hollow hydroxyapatite microsphere was used as a device for controlled delivery of proteins. 14 The effective removal of Cu2+ ions from polluted water is by using functionalized chitosan microspheres. 15 Recently, the incorporation of nanoparticles in the microspheres becomes the active and challenging field of research, 16 –18 which is due to the significant change in the properties of nanocomposites. Chen et al. developed Al2O3 nanocomposites with good electrical conductivity, heat-resistant, and mechanical properties. 19 In the field of corrosion control, polymer microspheres were known as micro-containers to encapsulate the anticorrosive materials. The purpose of micro-containers is to release the core material slowly if the coating is damaged mechanically or environmentally. 20 –24 Hence, it is important to enhance the effectiveness of coating by improving the thermal and mechanical properties of it. The incorporation of Al2O3 nanoparticle in the poly(tetra fluoroethylene) matrix improves the wear-resistant properties of materials up to three orders of magnitude. 25 Wang et al. proved that the coating containing Al2O3 nanoparticle showed improvement in scratch and abrasive resistance compared with that of polymer coating. 26 Hence, literature supports the use of nano-Al2O3 that exhibits excellent thermal and mechanical properties 17,18 and that stimulates to select nano-Al2O3 as a dispersed phase element of the composites. The glycidyl methacrylate (GMA) is hydrophobic and water-insoluble monomer with good adhesive property. 27
Thermal property is also a useful one for the application where high temperature is involved because the thermal stability of the polymer decides the application of it in the working temperature. The polymer should be stable enough to sustain in the operating condition like high temperature and mechanical stress, otherwise the polymer loses its properties. Glass transition temperature (Tg) is a very important property of a polymer, and it plays a prominent role in corrosion-coating efficiency. 27 The monomer phenyl methacrylate is hydrophobic in nature and also possesses high Tg. Functionalizing the phenyl methacrylate with bromine improves the thermal property of the monomer. Saric et al. discussed the thermal stabilities of brominated acrylates with styrene and observed that tribromophenyl acrylate-based copolymers are more thermally stable than other acrylates. 28 Literature reported the incorporation of nano-Al2O3 in the polymer by melt intercalation. 17,18 Likewise, few literature deals with the homopolymer nanocomposites that are prepared by solution mixing. 29 In this article, the nano-Al2O3 was incorporated in the halogenated copolymer matrix by solution mixing method. The main theme of this work is to prepare the halogenated methacrylate-based polymer nano-Al2O3 composites and to study the effect of incorporated nano-Al2O3 on morphological and thermal studies. This stimulates to design and develop a thermally stable copolymer of 2,4,6-tribromophenyl methacrylate with GMA microspheres (poly(TBPMA-co-GMA); pTG) and its Al2O3 nanocomposites.
Experimental section
Materials
GMA purchased from Sigma-Aldrich (St Louis, Missouri, USA) it was freed from inhibitor by distillation at 60–78°C under reduced pressure. Benzoyl peroxide (BPO) was recrystallized from chloroform–methanol (1:1). 2,4,6-Tribromophenol and nano-Al2O3 were purchased from Sigma-Aldrich and used as such. The analytical reagent-grade benzene, ethyl methyl ketone (EMK), hexane, chloroform, and methanol were distilled before use.
Synthesis of TBPMA
Methacryloyl chloride (MAC) was prepared following the procedure adopted by Stampel et al. 30 For the synthesis of TBPMA (Figure 1), required amount of 2,4,6-tribromophenol and triethylamine were dissolved in EMK in a round-bottomed flask with stirring placed in an ice bath. MAC dissolved in EMK was added to the flask at 0–5°C. The reaction was allowed to proceed for 6 h with constant stirring. The reaction mixture was washed and dried. The resultant product was recrystallized using chloroform.
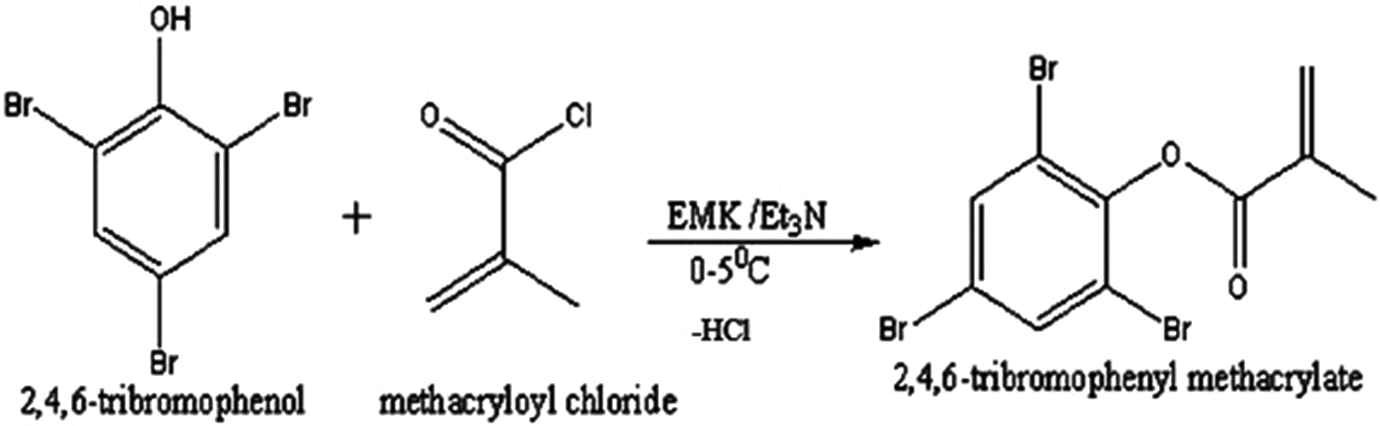
Synthesis of 2,4,6-tribromophenyl methacrylate.
Copolymerization of pTG
Required quantities of the monomers TBPMA and GMA along with BPO were dissolved in benzene. The mixture was flushed with dry nitrogen (N2) gas. The reaction vessel was immersed in a thermostatic water bath maintained at 70 ± 1°C. The copolymerization reaction was allowed to proceed for an appropriate duration (Figure 2). The solution was poured into excess methanol to precipitate the copolymer. The copolymers were purified by repeated precipitation using methanol from the solution present in chloroform. It was then dried in vacuum oven at 40°C for 1 day.
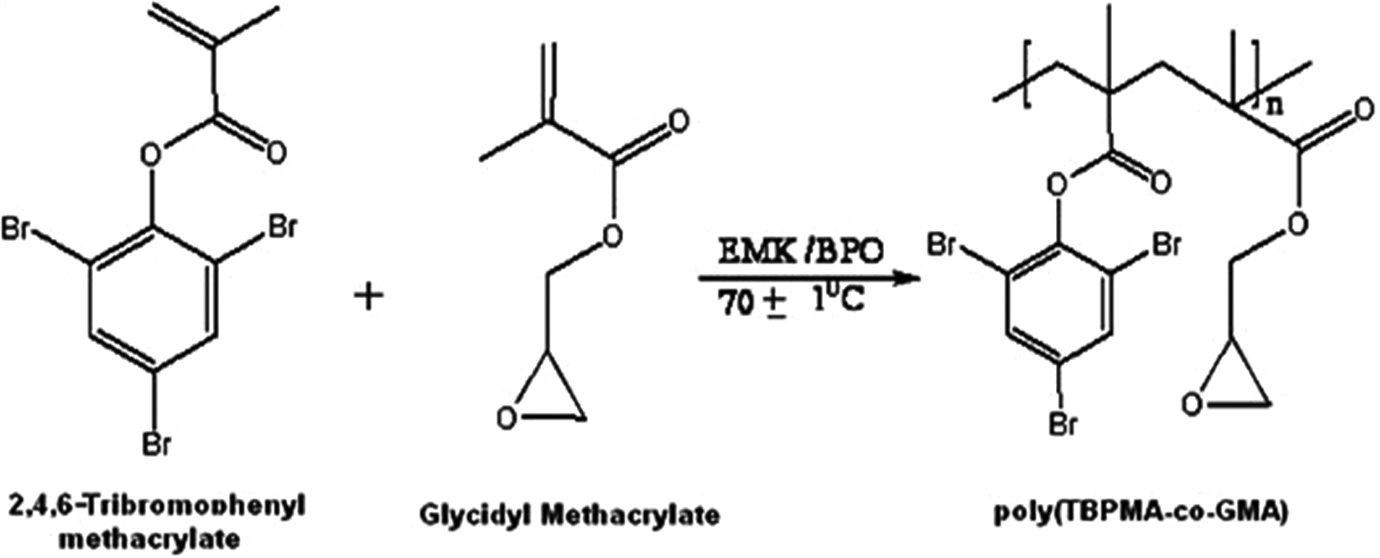
Copolymerization of 2,4,6 tribromophenyl methacrylate-co-glycidyl methacrylate
Formation of pTG
A pTG microsphere was prepared by emulsion solvent evaporation technique. In this method, the synthesized pTG was dissolved in chloroform, and the resulting solution was added slowly drop by drop for 1 hour to the 1% polyvinyl alcohol (PVA) solution. After complete addition, the above mixture was agitated for 4 h at 500 rpm. The microsphere was washed thrice repeatedly with deionized water and dried under vacuum.
Formation of pTG-Al2O3 nanocomposites
Required amount of pTG was dispersed in 100 mL 1% PVA solution in 500 mL beaker under constant stirring at 500 rpm. After 1 hour, predetermined amount (5% mass and 10% mass) of Al2O3 nanoparticle was dispersed in chloroform by ultrasonication and was added slowly into the beaker under stirring at 500 rpm. After 4 h, a colloidal solution was obtained. The resultant pTG-5%Al2O3 nanocomposite (pTG-5) and pTG-10%Al2O3 nanocomposite (pTG-10) were collected by centrifugation.
Characterization
The synthesized pTG microsphere and its Al2O3 nanocomposite were characterized by Fourier transform infrared (FTIR) spectrometer (model paragon 1000; Perkin-Elmer, Waltham, Massachusetts, USA). Thermal stability and Tg of the pTG and Al2O3 nanocomposite were determined by thermogravimetric analysis (TGA; Mettler Toledo 3000 thermal analyzer, Columbus, Ohio, USA) and differential scanning calorimetry, respectively, at a heating rate of 20°C min−1 under dry N2. The weight-average molecular weight (Mw) of the pTG obtained from gel permeation chromatography (GPC; model 501; Waters Corporation, Miliford, Massachusetts, USA). The morphology of the pTG microsphere and its Al2O3 nanocomposite were studied using a scanning electron microscope (Leica Stereo Scan, UK). The histogram was constructed from the SEM micrographs by counting the microspheres using ImageJ program (ImageJ software version 1.50c).
Results and discussions
Infrared spectroscopy
To evaluate the interface affinity between the pTG matrix and Al2O3 nanoparticle, FTIR measurement was carried out. The FTIR spectra of the pTG and pTG-5 are shown in Figure 3. The symmetric and asymmetric stretching of epoxy ring was observed at 1282 and 994 cm−1, respectively. The C–H out-of-plane bending vibration of aromatic ring appeared at 859 and 743 cm−1. The appearance of peak at 907 cm−1 indicates the incorporation of GMA unit in the copolymer. The bands at 1775 and 1725 cm−1 were due to the ester carbonyl of both TBPMA and GMA. Disappearance of C=C stretching frequency peak at 1636 cm−1 confirms the formation of copolymer. Neither shifts nor sharpening of absorption band is observed in pTG-5. This indicates that there is no significant chemical interactions between the functional group of pTG and nano-Al2O3 in the resulting nanocomposites. 29 Whereas, the compatibility exists between pTG and nano-Al2O3 is by non-covalent interaction.
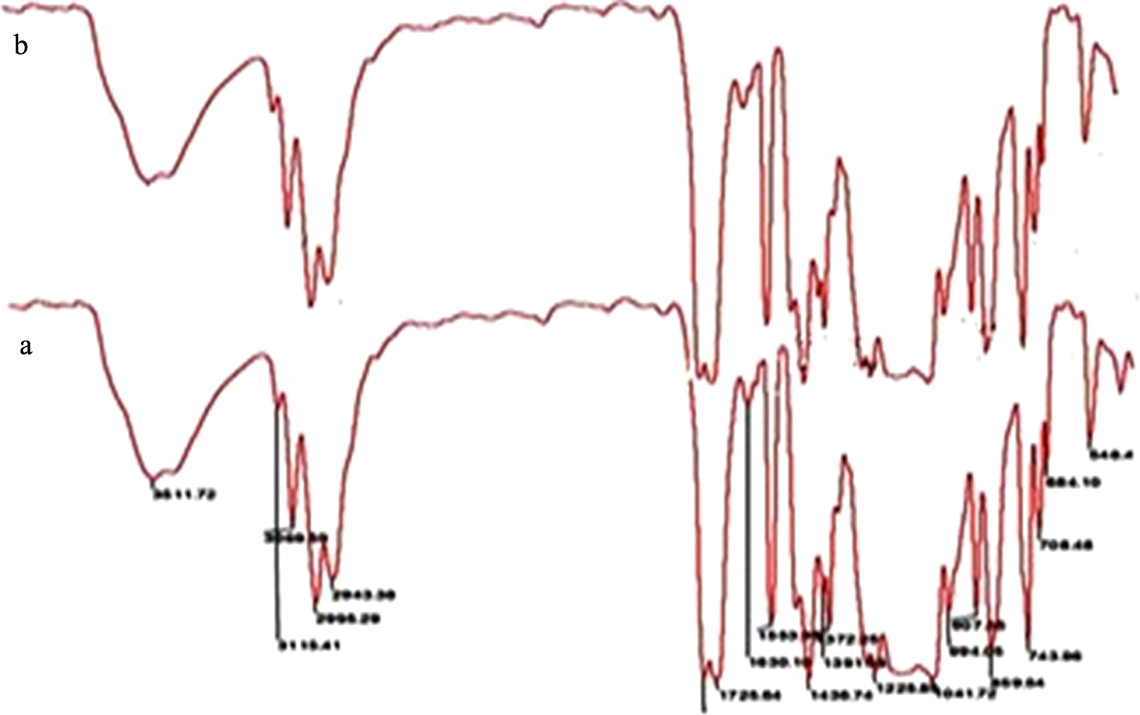
FTIR spectrum of (a) pTG microsphere and (b) pTG-5. FTIR: Fourier transform-infrared; TBPMA-co-GMA; 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres; Al2O3: aluminum oxide; pTG: poly(TBPMA-co-GMA) microsphere; pTG-5: poly(TBPMA-co-GMA)-5% Al2O3 nanocomposite.
Thermal behavior
The thermal stability of pTG, pTG-5, and pTG-10 was evaluated by TGA. Figure 4 shows the TG curve of pTG, pTG-5, and pTG-10. From Figure 4, it is evident that all the three show two different degradation steps. The first at lower temperature (i.e. at temperature < 300°C) is the evolution initiated at the unstable terminal double bond present in the macromolecules as a consequence of the disproportionate termination reaction, while the second follows the random bond scission of the polymeric chain. 31 It is also confirmed from the figure that the initial decomposition temperature of the pTG-Al2O3 nanocomposite shifts significantly toward higher temperature range than that of pTG microsphere, which indicates the enhancement of thermal stability due to the incorporation of Al2O3. Thermal analysis data given in Table 1 (thermal decomposition temperature at 50% and 10% weight loss is T−50% and T−10%, respectively) explain that the pTG-10 is slightly less thermal stable than the pTG-5 because the dominant factor that influences the thermal decomposition temperature of pTG-10 is becoming the effect of the interface between the nano-Al2O3 particle and the pTG matrix. 32 The thermal degradation temperature of pTG-Al2O3 nanocomposite needs less energy due to the large interface and high surface energy of the nano-Al2O3 particle, which leads to decrease in the thermal decomposition temperature of thepTG-10. The Tg of the pTG, pTG-5, and pTG-10 are given in Figure 5. From Table 1, it is also confirmed that Tg increases with the increase of nano-Al2O3 particle content. Typically, filler serves to increase the Tg value by decreasing the mobility of the chains close to the surface of nano-Al2O3 particle content. 29
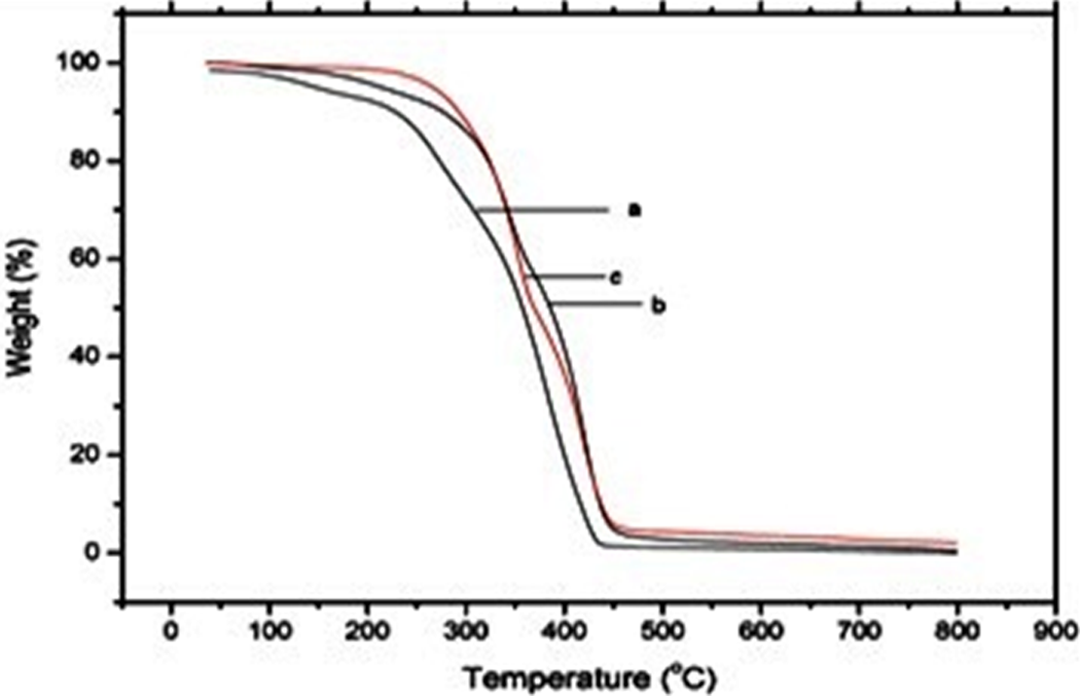
Thermal curves of (a) pTG, (b) pTG-5, and (c) pTG-10. TBPMA-co-GMA; 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres; Al2O3: aluminum oxide; pTG: poly(TBPMA-co-GMA); pTG-5: poly(TBPMA-co-GMA)-5% Al2O3 nanocomposite; pTG-10: poly(TBPMA-co-GMA)-10% Al2O3 nanocomposite.
TG data of pTG microsphere, pTG-5, and pTG-10.
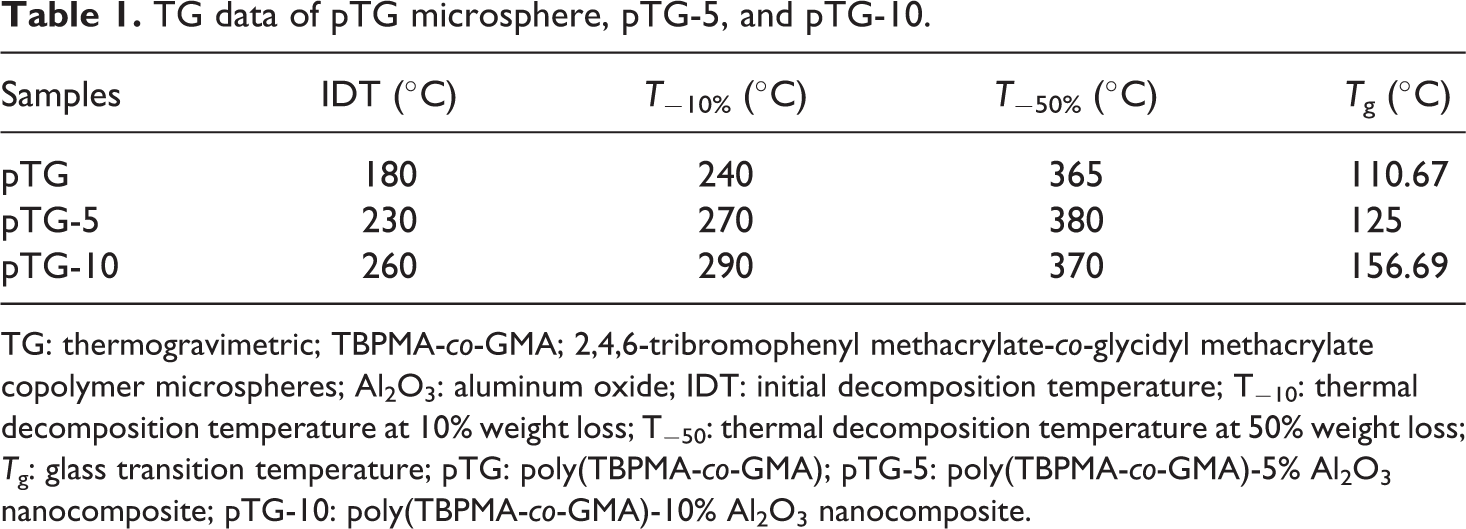
TG: thermogravimetric; TBPMA-co-GMA; 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres; Al2O3: aluminum oxide; IDT: initial decomposition temperature; T−10: thermal decomposition temperature at 10% weight loss; T−50: thermal decomposition temperature at 50% weight loss; Tg: glass transition temperature; pTG: poly(TBPMA-co-GMA); pTG-5: poly(TBPMA-co-GMA)-5% Al2O3 nanocomposite; pTG-10: poly(TBPMA-co-GMA)-10% Al2O3 nanocomposite.
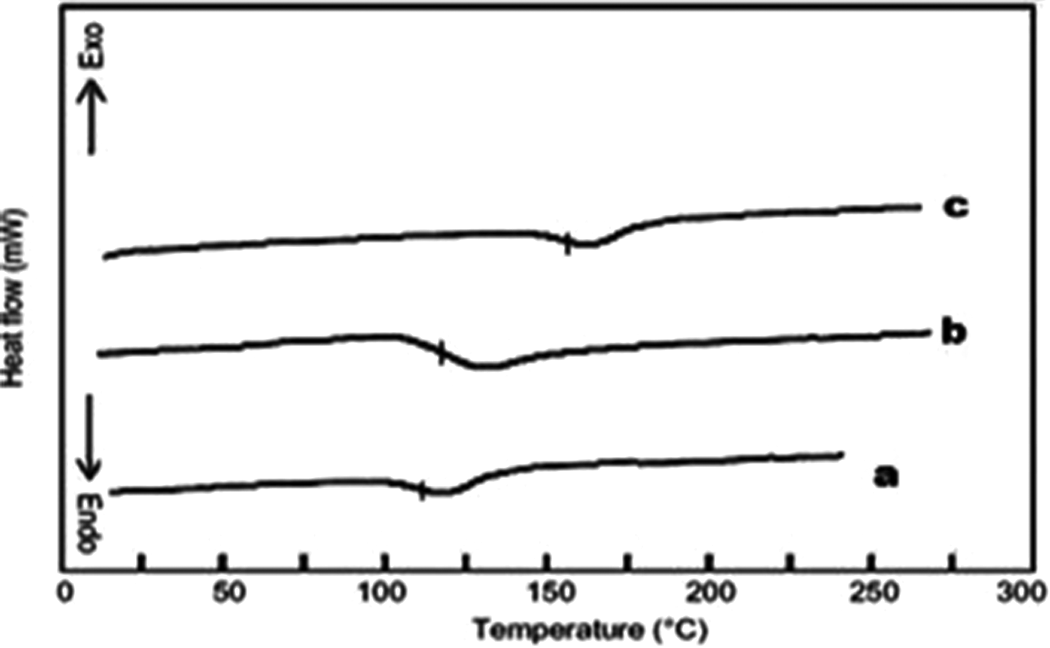
DSC thermogram of (a) pTG, (b) pTG-5, and (c) pTG-10. DSC: differential scanning calorimetry; TBPMA-co-GMA; 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres; Al2O3: aluminum oxide; pTG: poly(TBPMA-co-GMA); pTG-5: poly(TBPMA-co-GMA)-5% Al2O3 nanocomposite; pTG-10: poly(TBPMA-co-GMA)-10% Al2O3 nanocomposite.
Molecular weight determination
The number-average molecular weight (Mn) and Mw of copolymers were determined by GPC. The polydispersity index (Mw/Mn) of the copolymers ranges between 1.92 and 2.16. This value for pTG is close to 2.0, which suggests a strong tendency for chain termination by disproportionation rather than radical combination. 33 Molecular weight has an influence on Tg, which revealed that the polymer’s molecular weight up to around 20,000 g mol−1 has a significant influence in Tg. 34 But the molecular weight of the pTG was 37,806 g mol−1 infers that improved Tg value mainly corresponds to the incorporation to the nano-Al2O3 particle in the pTG matrix.
Morphology and size distribution
Figure 6 illustrates the morphologies of pTG microsphere and its Al2O3 nanocomposites. Figure 6(a) and (b) shows that the pTG microsphere surface is smooth and that the microspheres exhibit spherical shape with the majority of it falling in the range of 15 µm size having a standard deviation of 7.9 µm. The smooth and nonporous surface of pTG microsphere is due to the hydrophobicity of it. The micrograph (Figure 6(c) and (e)) of the obtained polymer nanocomposites reveals that the production was achieved successfully with some agglomerates of Al2O3 nanoparticles on the surface. The pTG-5 and pTG-10 surfaces were rough and porous due to the incorporation of Al2O3 nanoparticle leading to hydrophilic moiety. 35 The histogram was constructed from the SEM micrograph (Figure 6(c) and (e)) and is presented in Figure 6(d) and (f). It is noted from the histogram that the mean diameter of pTG-5 and pTG-10 microspheres was 27.5 µm and 48.3 µm, respectively. Taking advantages of the porous and hydrophilic nature of pTG-Al2O3 nanocomposite, it is possible to apply in the medical (like isolation of biological compounds) and industrial (like separation of toxic metals) fields. It is clear from Figure 6(e) that the agglomeration of hydrophilic Al2O3 nanoparticle on the pTG-10 surface is due to the high surface energy of the Al2O3 nanoparticle.
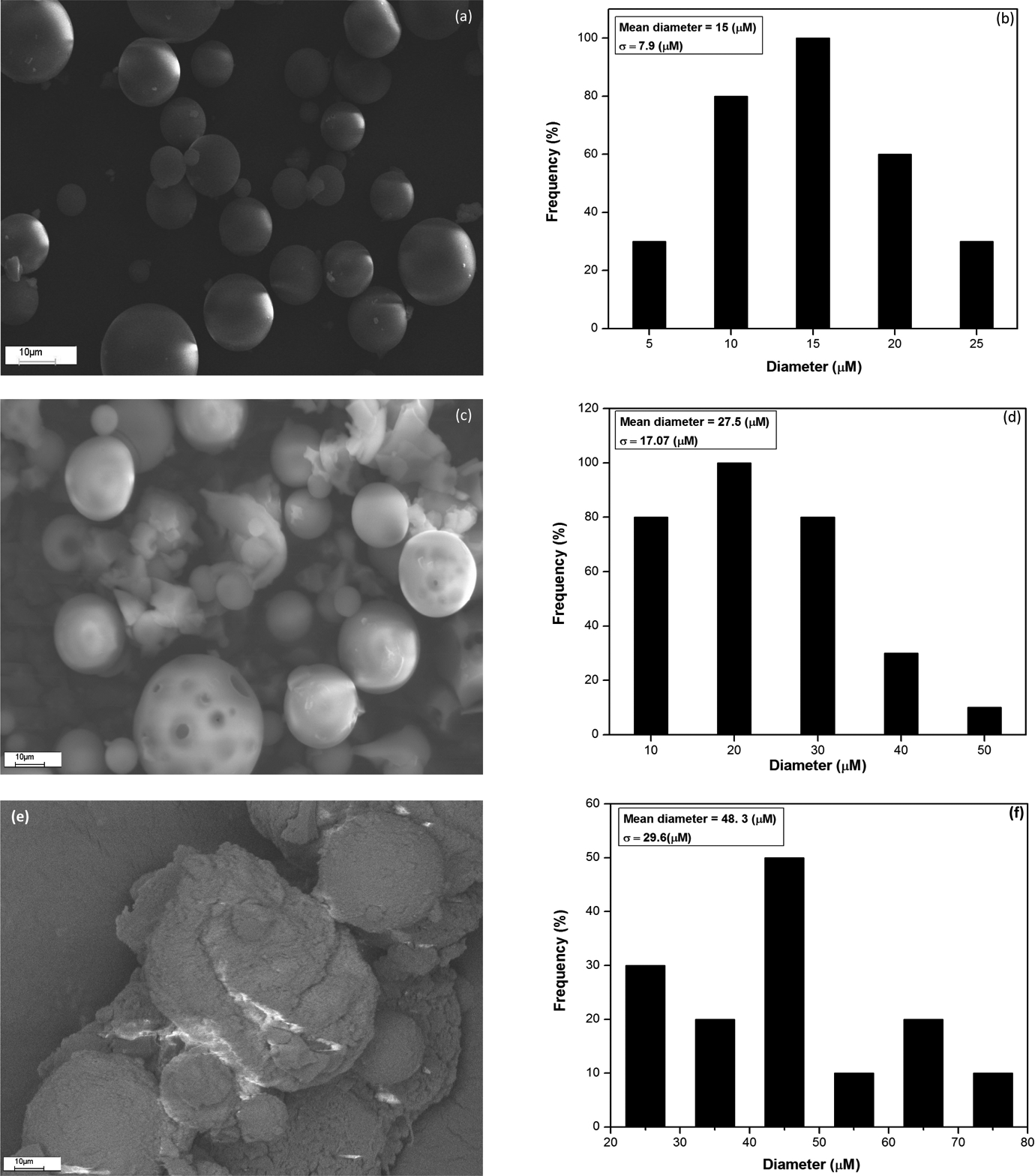
SEM images of (a) pTG, (c) pTG-5, and (e) pTG-10. Size distribution histogram of (b) pTG, (d) pTG-5, and (f) pTG-10. SEM: scanning electron microscope; TBPMA-co-GMA; 2,4,6-tribromophenyl methacrylate-co-glycidyl methacrylate copolymer microspheres; Al2O3: aluminum oxide; pTG: poly(TBPMA-co-GMA); pTG-5: poly(TBPMA-co-GMA)-5% Al2O3 nanocomposite; pTG-10: poly(TBPMA-co-GMA)-10% Al2O3 nanocomposite.
Conclusion
A novel functional halogenated methacrylate-based microsphere and its Al2O3 nanocomposites have been synthesized successfully by emulsion solvent evaporation and solution mixing technique, respectively. The FTIR spectroscopy shows that there is no significant interaction between the pTG and nano-Al2O3 particle in the resulting nanocomposite, but the compatibility between the nano-Al2O3 and the pTG matrix is by non-covalent interaction. The molecular weight of the pTG confirms that the enhancement in the Tg was due to the addition of nano-Al2O3 particle in the pTG. Thermal studies reveal that the incorporation of nano-Al2O3 particle in the pTG matrix enhances the thermal stability and Tg than its microsphere. The SEM observation indicated that the surface of pTG nanocomposites were rough due to the incorporation of nano-Al2O3 particle, which in turn increases the surface area, and also confirms that the incorporation of nano-Al2O3 induces the hydrophilicity in the pTG matrix, which finds application in diversified fields.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
