Abstract
In this work, isotactic polypropylene (iPP) nanocomposites were prepared containing silver nanoparticles (Ag-NPs) with a novel and easy method, using polyethylene glycol (PEG) as reducing agent and surface modifier. Ag-NPs were prepared using different amounts in weight of silver nitrate into PEG to induce the formation of Ag-NPs. PP/Ag nano compounds were prepared by melt blend method: single-screw extruder and internal Brabender mixer. The effects of Ag-NPs and PEG on the crystallization, morphology, thermal, and mechanical properties were evaluated. Ag-NPs with a particle size of 80 nm and typical growth of the β-form in iPP were observed. The presence of PEG in samples of PP/Ag-NPs was detected by infrared spectrometry and the peak characteristic of Ag-NPs by ultraviolet–visible analysis. X-Ray diffraction patterns and differential scanning calorimetry thermograms showed the β-phase formation in both of the dispersion methods, but Brabender mixer showed higher percentages of crystallinity (31% of β-phase). The elongation at break was increased and it was directly dependent on the relative amount of crystalline β-phase. PEG is an excellent precursor to get Ag-NPs and a good interface modifier of iPP.
Introduction
The growing demand for nanocomposite materials is strongly influencing the industry of automotive, aerospace, food, medicine and pharmacy, as well as materials for engineering applications. 1 Polymer nanocomposites with enhanced performance in the physical, chemical, and mechanical properties have generated interest in the past decade. Silver nanoparticles (Ag-NPs) have been extensively used in various application fields such as spectrally selective coatings for solar energy absorption, intercalation material for electrical batteries, optical receivers, and catalysts in chemical reactions and antibacterial agents. 2 –5 Ag-NPs can be synthesized by microemulsion, X-ray, ultraviolet (UV) irradiation, microwave radiation, spray pyrolysis, laser ablation, organic and inorganic precursors, among other methods. 3,5 –9 Chemical reduction is the most often applied method for the preparations of Ag-NPs, 5 and the successful synthesis involves three steps: nucleation, growth, and termination by the capping agent or ligand. 9 The most commonly used polymers in the synthesis of Ag-NPs are poly(vinyl pyrrolidone), 10 poly(ethylene glycol) (PEG), 11 poly(vinyl alcohol), 12 and so on.
Polypropylene (PP) is a thermoplastic material commonly used in industrial and commercial applications such as injection, blow molding, film and extrusion. PP has a relatively low cost compared to other polyolefins and has good physical, chemical, and mechanical properties by altering the chain regularity (tacticity). 13 However, PP has a disadvantage of being nonpolar due to the absence of functional groups in the structure; hence, it has low surface energy resulting in difficulty in dyeing, adhesion, or coating and poor compatibility with polar polymers or filler. 14 –16 PP exhibits three crystalline forms, the monoclinic (α), being the most common and stable; trigonal (β); and orthorhombic (γ) forms. 17 –19 The β-phase or hexagonal form is less common and stable, 13 depending on the conditions of crystallization of PP, such as nucleating agents, 19,20 shear effect in the extrusion and the injection process, 18,20,21 fillers in micro- and nanoscale, 20,22 –25 and using temperature gradients. 26,27 β-Phase in high proportions usually leads to improved mechanical toughness properties such as impact strength and elongation, which is of considerable importance from the viewpoint of industrial application.19,20,22–24,27
Some authors have published studies on the preparation of nanocomposites using PP and Ag-NPs. 22,23,28,29 These authors have used nanoparticles synthesized with precursors that are released into the environment or provided by companies.
In this article, a novel and easy method was performed in the preparation of PP/Ag nanocomposites, studying the effect of PEG as reducing agent and stabilizer in the synthesis of Ag-NPs, as well as surface modifier of PP during the preparation by melt blending. The dispersion method into PP matrix by melt blending was examined focusing on the β-phase formation during PP crystallization. Nanocomposites were prepared using two methods: internal Brabender mixer and single-screw extruder. β-Phase was examined using X-ray diffraction (XRD) and differential scanning calorimetry (DSC). Polarized light microscopy (PLM) and atomic force microscopy (AFM) were used to examine the growth of β-spherulites into the iPP matrix and size of Ag-NPs. Fourier transform infrared (FTIR) and ultraviolet–visible (UV-Vis) spectrometry were used to analyze the interaction between PP-PEG and to obtain the peak characteristic of Ag-NPs, respectively. Mechanical properties of isotactic PP (iPP)/Ag nanocomposites were also evaluated.
Experimental
Materials
The material used in this study was iPP (Pro-Fax™, XH1760) supplied by Indelpro (Mexico). Its melt flow index was 3 g/10 min (230°C/2.16 kg) calculated according to the ASTM D1238 standard. Silver nitrate (AgNO3) crystals (weight-average molecular weight (
Synthesis of Ag-NPs
Table 1 shows the preparation of the different amounts of AgNO3 (3, 5, and 10 wt%) dissolved in PEG at 80°C. The resulting solution was stirred at this temperature for 1 h and then cooled at 27°C to solidify. 11 The obtained PEG was diluted with 200 ml of DCM, and this solvent was evaporated at room temperature (27°C).
Different amounts of AgNO3 dissolved in PEG to obtain Ag-NPs.
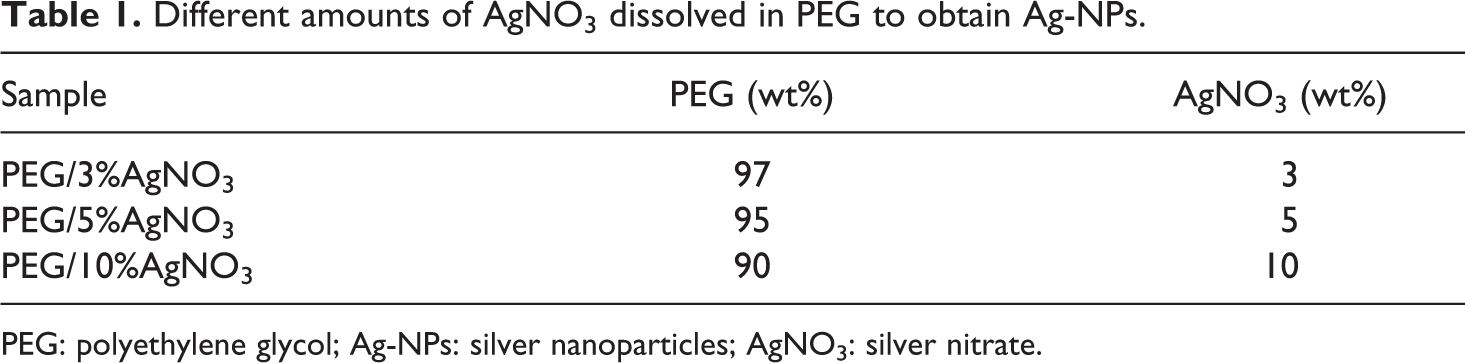
PEG: polyethylene glycol; Ag-NPs: silver nanoparticles; AgNO3: silver nitrate.
Preparation of iPP/Ag-NP nanocomposites
Samples were prepared by mixing 10 wt% PEG/AgNO3 into PP using the three different amounts (3, 5, and 10% of AgNO3) mentioned in Table 1. The melt blends containing Ag-NPs were prepared using two methods, via internal Brabender mixer and single-screw extruder, as follows (see Table 2):
In the first method, blends of iPP and Ag-NPs were prepared with an internal mixer (Brabender) type double rotor for 7 min at 190°C at a rotor speed of 60 r min−1. In the second processing method, a 19-mm single-screw extruder (Beutelspacher, Mexico) was used to compound the nanocomposites. The temperature profile ranged from 190°C in the feed section to 210°C at the die, and the screw speed was 50 r min−1.
Designation of iPP/Ag nano compounds in terms of their composition and dispersion method.

iPP: isotactic polypropylene; Ag: silver; AgNO3: silver nitrate.
Characterization methods
Atomic force microscopy
The iPP/Ag-NPs films were analyzed by AFM in tapping mode using an Innova Vecco microscope (Bruker, Massachusetts, USA). Thin films of about 50 µm thickness of the nano compounds were prepared using compression molding at 200°C.
UV-Vis spectrometry
A Cintra 303 spectrophotometer (GBC Scientific Equipment, Australia) was employed for the UV-vis analysis of the Ag-NPs at room temperature in the range of 200–900 nm in wavelength.
Fourier transformed infrared
A Perkin Elmer FTIR (Waltham, Massachusetts, USA), coupled with diamond attenuated total reflection cell, model Spectrum 100 device was used to see any change in the polymeric structure.
Differential scanning calorimetry
The melting and crystallization behavior of the samples were determined using a Perkin Elmer Diamond differential scanning calorimeter (model NS36-0022). The extruded pellets (about 5–10 mg for sample) were first quickly heated from 50°C to 200°C at a heating rate of 10°C min−1 and then cooled down and finally heated again at the same rate.
X-ray diffraction
Diffraction patterns of the iPP/Ag-NPs nanocomposites were performed with a Bruker AXS D8 Advance diffractometer using copper
Polarized light microscopy
The crystal morphology of the iPP/Ag-NPs nanocomposites were observed on a polarized light microscope (standard GFL model 473059-9901, Carl Seiz Co. Ltd, Germany) equipped with a digital camera.
Tensile properties
Specimens for mechanical characterization were prepared with a molded compression machine (Carver model 3912) at 200°C. Young’s modulus, tensile strength, and elongation at break were obtained in accordance with ASTM D638 standard 30 using an universal testing machine (AutoGraph model Ags-20kNG, Shimadzu, Japan).
Results and discussion
Morphological properties of iPP/Ag nanocomposites
Figure 1 shows the AFM topography of iPP/Ag-NPs nanocomposites. The obtained particle size by AFM showed an average diameter of 80 nm with a standard deviation of 41 nm according to the measurement by ImageJ software (see Figure 1(a)). According to Luo et al., the reaction temperature plays a key role in the dimensions of the particle because the average particle size is enlarged from 10 nm to 80 nm when the temperature is raised to 120°C. 11 In this study, the temperature required to melt the iPP nanocomposites was 190°C. Moreover, Ag-NPs tend to link together to form small clusters 22 into the PP, which may be the reason for the very dispersive size of NPs. The surface of iPP filled with Ag-NPs shows spherical NP with different sizes and good random dispersion on the surface of iPP matrix (Figure 1(b)). Furthermore, Figure 1(c) shows the formation of typical β-spherulites in the sample B-10/10 which was attributed to the nucleating effect of Ag-NPs into the iPP matrix.22,28,31 The sample E-10/10 (Figure 1(d)) by extrusion process also shows the nucleating effect but is less notorious compared with the sample prepared in Brabender mixer.
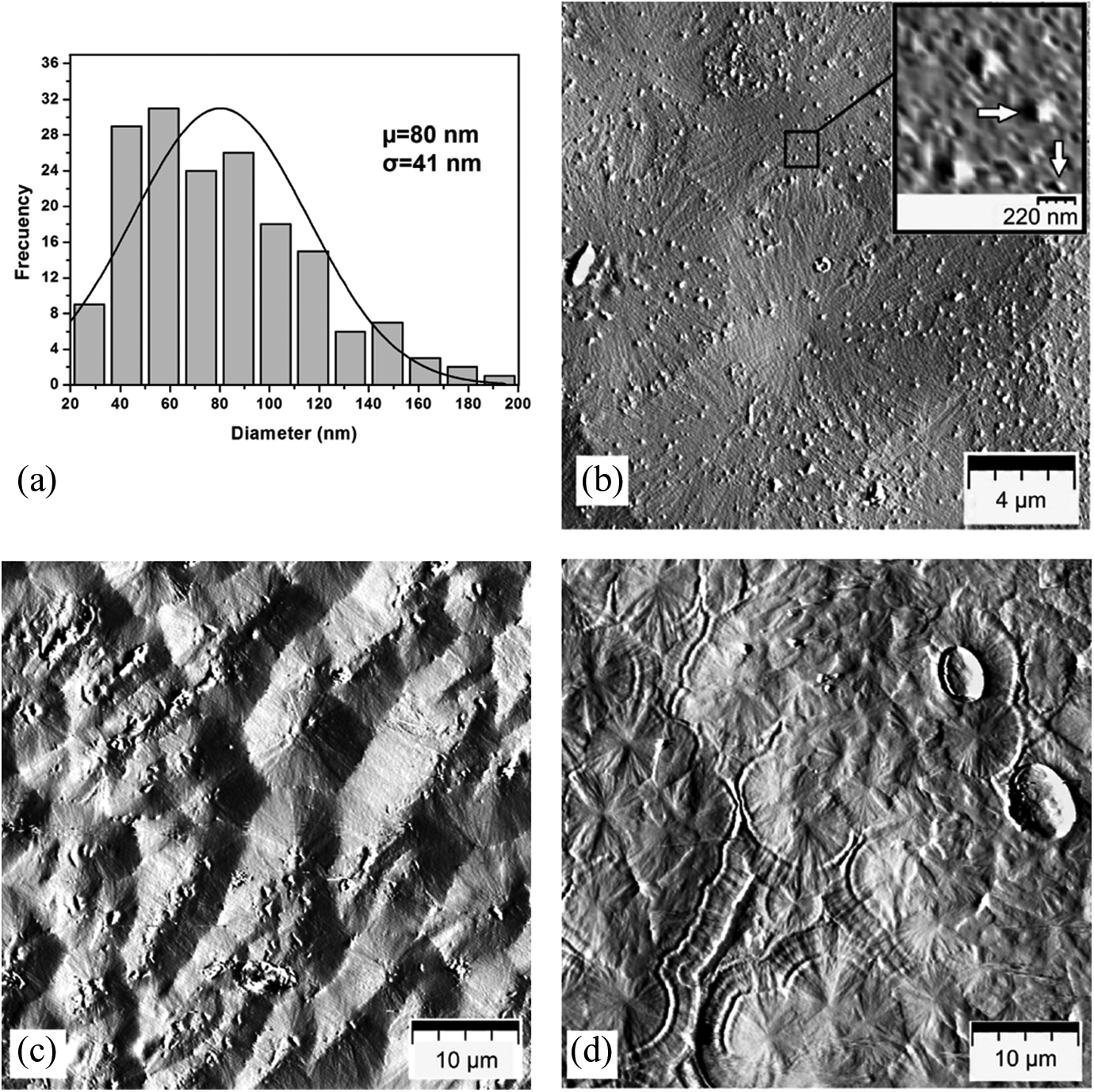
AFM micrographs of iPP/Ag-NPs nanocomposites: (a) histogram of Ag-NPs size, (b) iPP/Ag-NPs surface, (c) sample B-10/10, and (d) sample E-10/10. AFM: atomic force microscopy; iPP: isotactic polypropylene; Ag-NPs: silver nanoparticles.
UV-Vis spectrometry
Figure 2 shows the UV-Vis absorption spectra of the iPP/Ag sample obtained by melt blending. It is well known that the optical absorption spectra of Ag-NPs show an intense peak at approximately 400 nm due to the surface plasmon resonance (SPR). 32 –34 The location and shape of the absorption peak are strongly dependent on the particle size, while the peak width depends on the particle size distribution and its height corresponds to the concentration of the Ag metal particles. 34 Figure 2 reveals the formation of Ag-NPs on the film surface by showing a maximum absorption peak of 410 nm, this wavelength indicates an approximate particle size of less than 100 nm. The wide size of curve indicates a very dispersed NP.
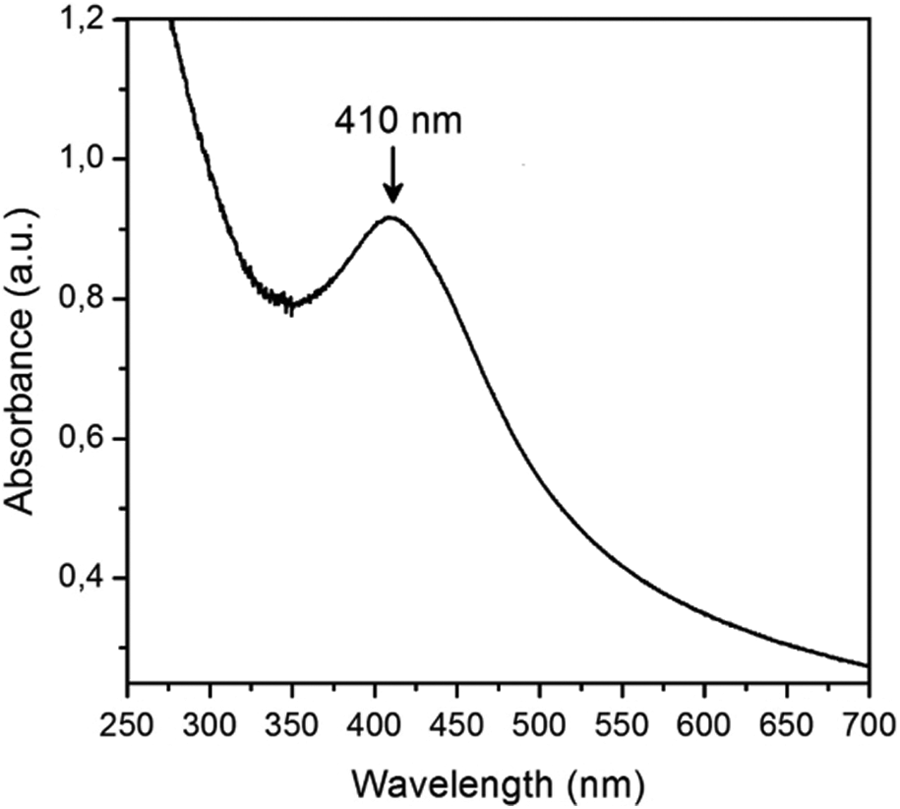
UV-Vis spectra of iPP/Ag sample after the melt blending process. UV-Vis: ultraviolet–visible; iPP: isotactic polypropylene; Ag: silver.
Fourier transformed infrared
Figure 3 shows the spectra of samples PP, PP-PEG, and PP/PEG-Ag nanocomposites. The peaks at 842 and 1169 cm−1 are representative of the helical chains in PP (C–C stretching). Moreover, the intense peaks at 1371 and 1453 cm−1 corresponding to the symmetric C–H bending and asymmetric CH3 bending, respectively. 35 According to Chen et al., the band of PP and PEG at about 1103 cm−1 was assigned to C–C groups in iPP and C–O groups in PEG (see Figure 3), showing the presence of PEG in the samples of PP/Ag nanocomposites. 15,16 PEG remaining is presented in the iPP/Ag-NPs compounds after washing with DCM during the synthesis of Ag-NPs. The presence of PEG in PP, according to Li et al., improved the impact toughness and β-form content adding a small amount of PEG (5%) into iPP. 36 PP is a polyolefin with low adhesion due to its nonpolarity, and PEG is helpful in adhesion between the inorganic nanofiller and iPP matrix because the functional groups of PEG grafted into the PP have the function of interface between PP and Ag-NPs. PEG was used as a reducing agent and stabilizer of the Ag-NPs but also it can take the role of a modifier into the iPP.
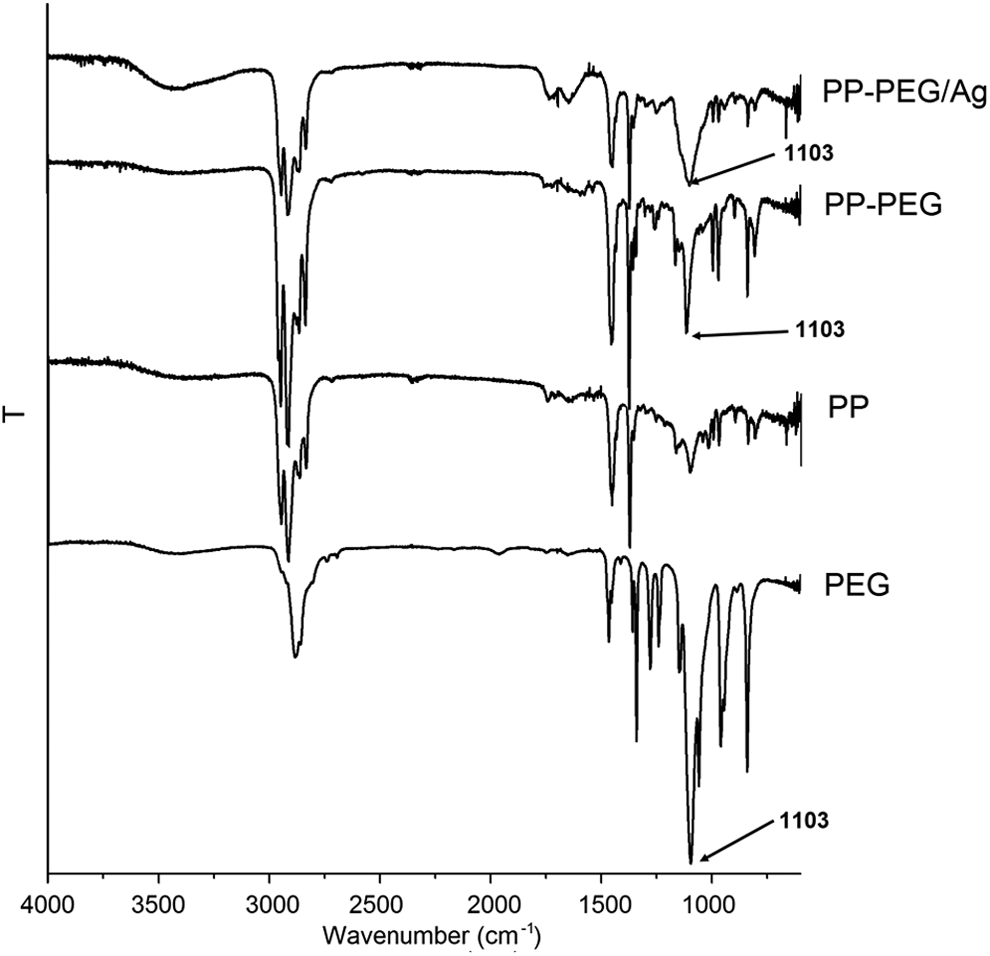
ATR-FTIR spectra of samples iPP, PEG, PP/PEG, and iPP/PEG-AgNO3. PEG: polyethylene glycol; iPP: isotactic polypropylene; ATR-FTIR: attenuated total reflection–Fourier transformed infrared; PP: polypropylene; AgNO3: silver nitrate.
Thermal properties
The DSC thermograms of pure iPP and iPP nanocomposites filled with different PEG/Ag-NPs contents are shown in Figure 4, and the values of melting
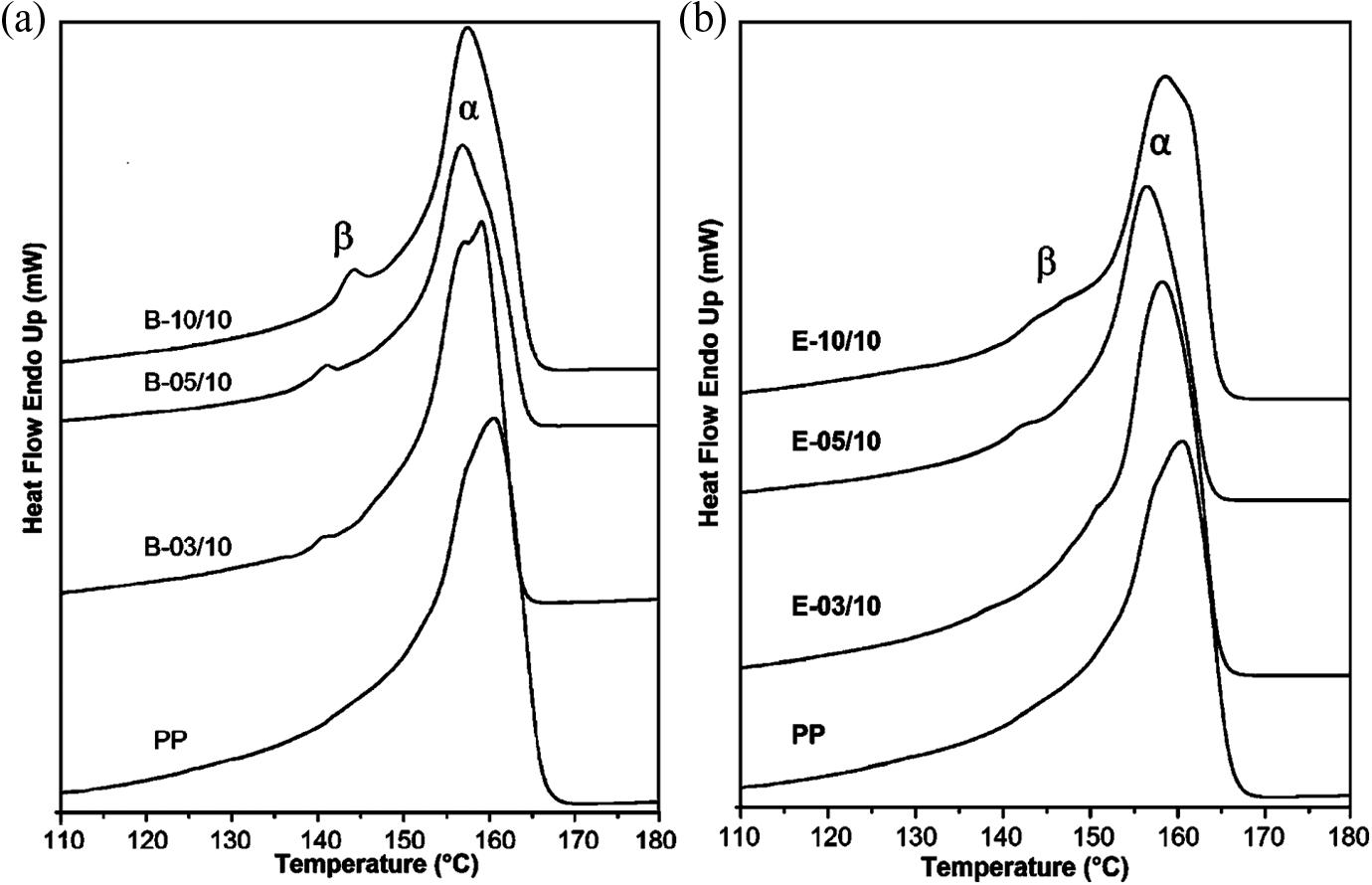
DSC thermogram curves (second heating run) of pure iPP and iPP/Ag nanocomposites by (a) Brabender mixer and (b) single-screw extruder. iPP: isotactic polypropylene; DSC: differential scanning calorimetry; Ag: silver.
DSC results of melting (α and β phase) and crystallization temperature of iPP and iPP/Ag-NPs.

DSC: differential scanning calorimetry; B: Brabender; E: extruder;
The β-phase temperature (approximately 150°C) usually appears as a peak before the α melting point (approximately 160°C).
22,24,25,31
Furthermore, the temperature at which β-phase appears depends on the mixing conditions. It can be seen that in iPP/Ag-NPs nanocomposites, β-phase was detected in all the samples, while in samples prepared in single-screw extruder the sharp β-phase peak was decreased compared to Brabender mixer (Figure 4(b)). The
The fraction percentage of β form in a sample (Xβ) can be obtained from the crystallinities of the α and β phase according to the following equation:
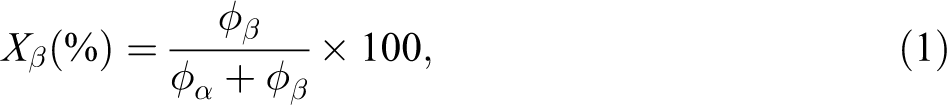
where φα and φβ are the crystallinities of the α and β forms and can be calculated separately using the following equation:

where Δ
The values obtained from DSC analysis of crystallinity reveal that the content of β-crystal is between 1.0% and 3.0% in samples prepared with PEG–Ag-NPs according to equation (1).
X-Ray diffraction
Figure 5 shows the diffraction patterns between 36° and 40° of the crystal phase structure of PP/PEG-Ag nanocomposites. According to Xu et al., the peak at 38.18° is the characteristic of metallic Ag, corresponding to the plane (111). 37 The peak in samples is not so higher due to small amount of Ag in the matrix of PP (between 0.2 and 0.6 wt%).
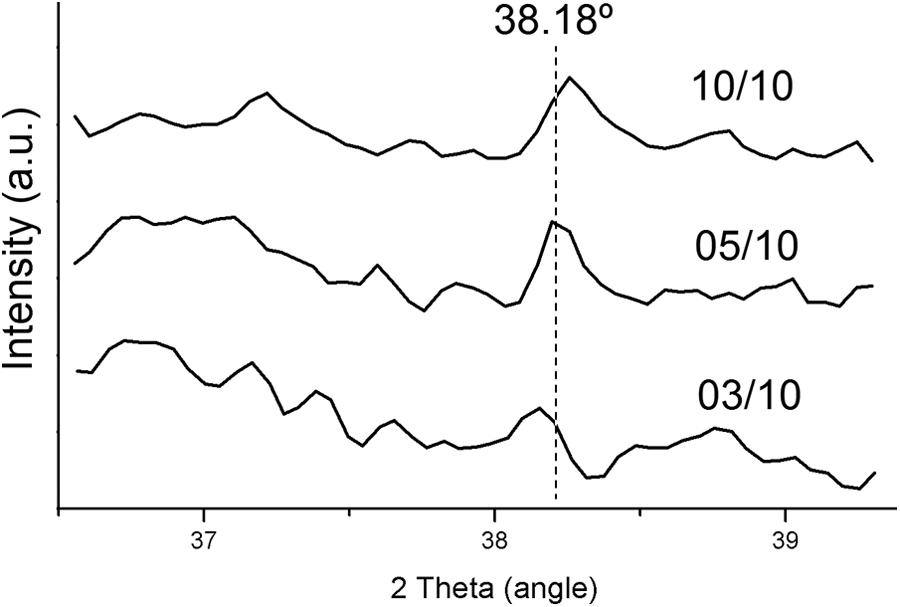
XRD of the PP/PEG-Ag nanocomposites showing the silver peak at 38.18°. XRD: X-ray diffraction; PEG: polyethylene glycol; PP: polypropylene.
Figure 6 shows the XRD patterns of the iPP/Ag-NPs nanocomposites using the two dispersion methods. The monoclinic α-crystal can be identified from XRD measurement of the peaks at 2
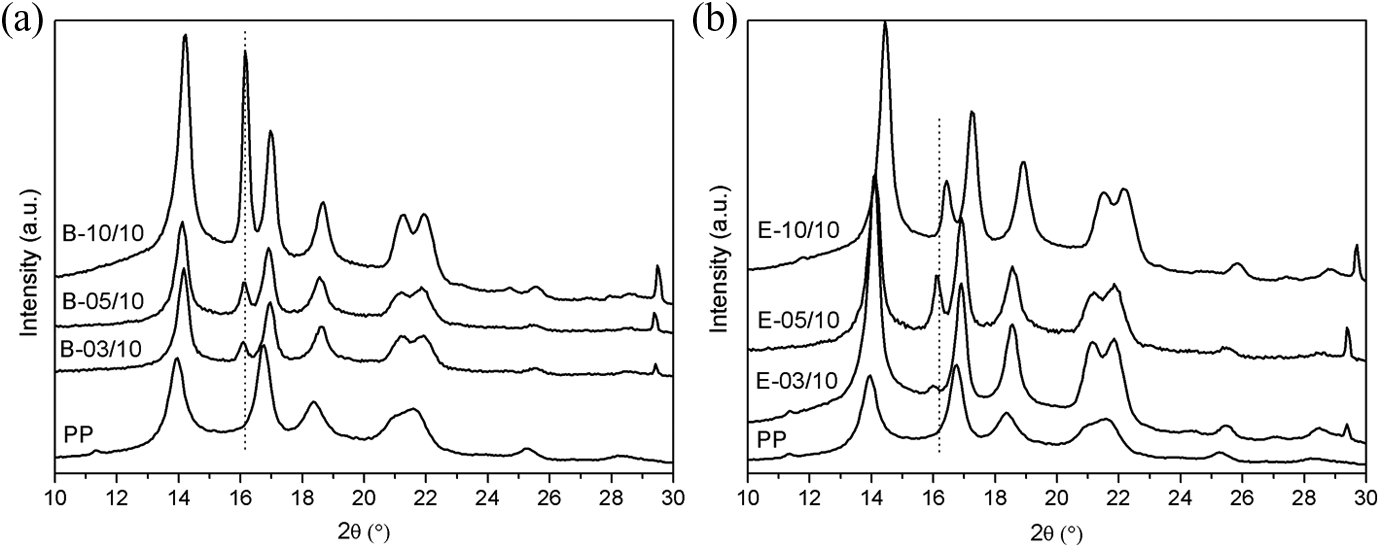
XRD patterns of PP and iPP/Ag-NPs nanocomposites by (a) Brabender mixer and (b) single-screw extruder. XRD: X-ray diffraction; PP: polypropylene; iPP: isotactic polypropylene; Ag-NPs: silver nanoparticles.
The relative content of β crystal form was measured in terms of the
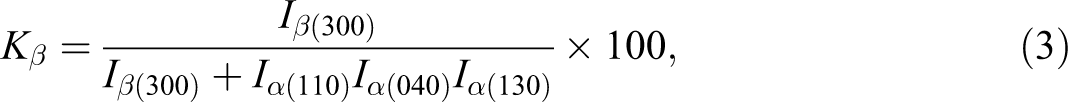
where
The β-phase content (
The

XRD: X-ray diffraction;
Tensile properties
Figure 7 shows the stress–strain curves of PP and PP/PEG-Ag nanocomposites to compare the effect of the incorporation of PEG and Ag-NPs in the matrix of iPP using the internal mixer and extrusion process. In Table 5, the main results of tensile properties (tensile strength, Young’s modulus, and elongation at break) of each sample according to the standard test method (ASTM D638) is reported.
30
It can be seen that elongation at break is higher for samples prepared by Brabender mixer (samples with the best relative amount
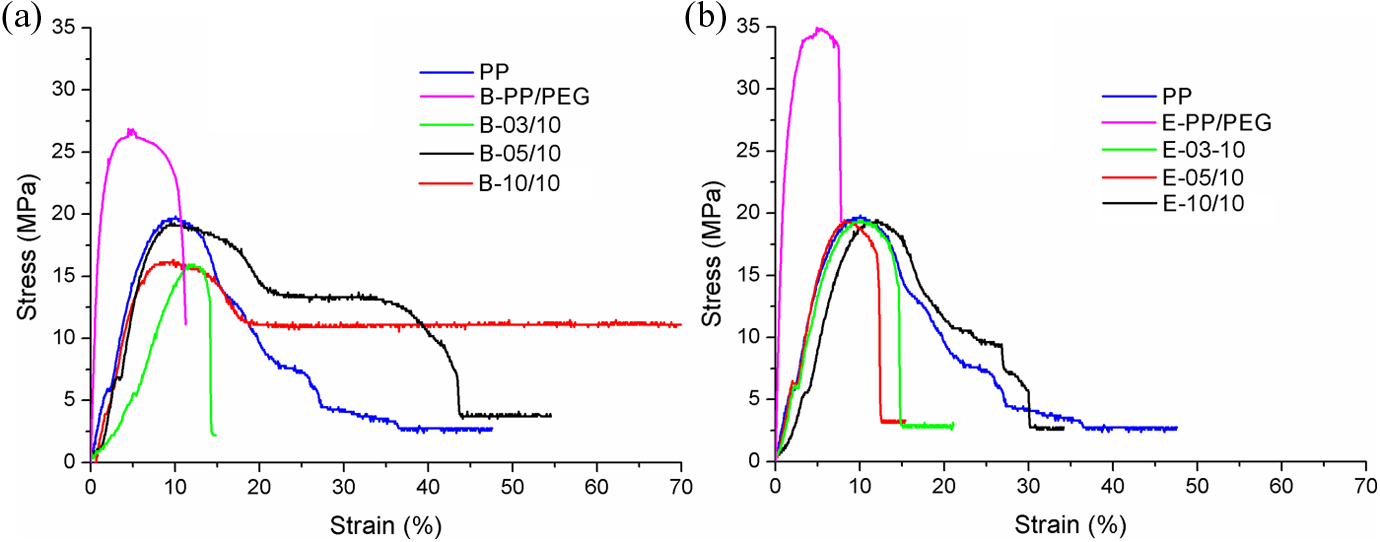
Stress–strain curve of samples PP, PP-PEG, and PP/Ag nanocomposites by (a) Brabender mixer and (b) single-screw extruder. PP: polypropylene; PEG: polyethylene glycol; Ag: silver.
Tensile properties of iPP/Ag-NPs nanocomposites.
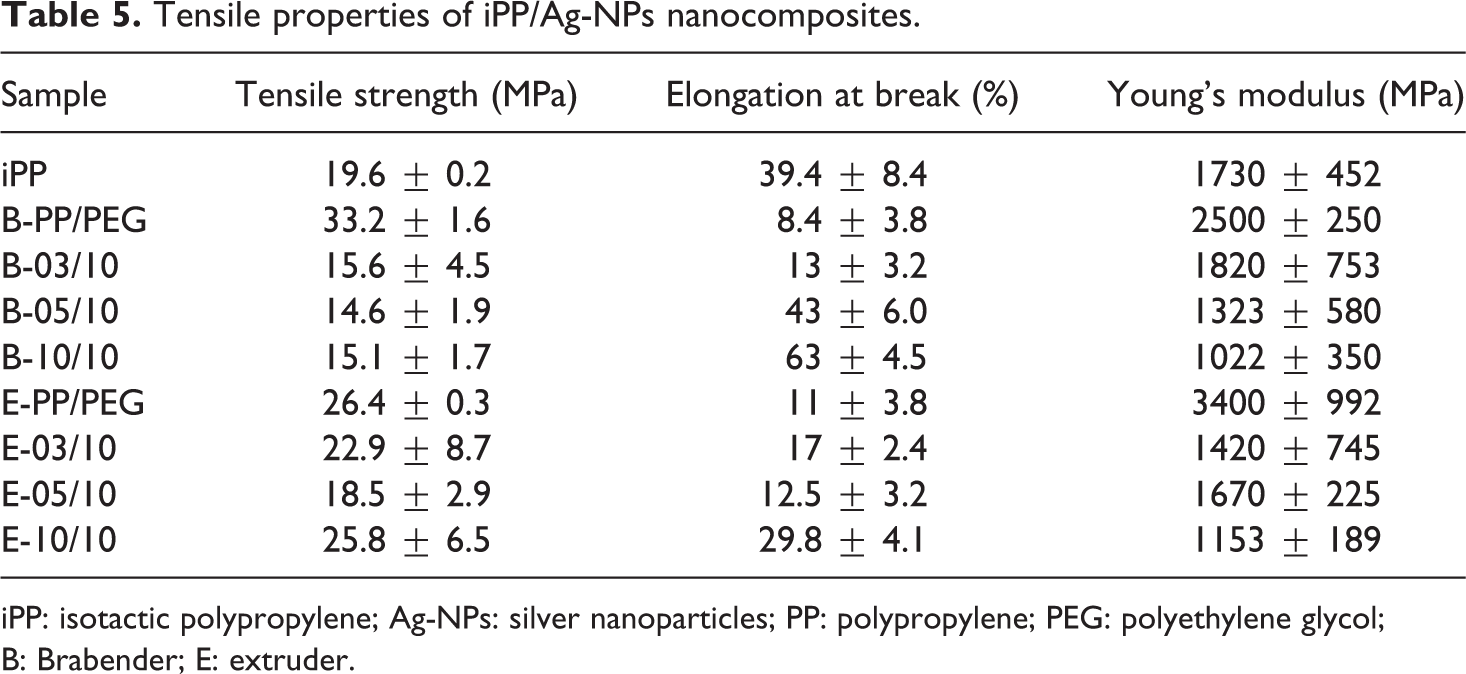
iPP: isotactic polypropylene; Ag-NPs: silver nanoparticles; PP: polypropylene; PEG: polyethylene glycol; B: Brabender; E: extruder.
Polarized light microscopy
Figure 8 shows the PLM micrographs of iPP/Ag-NPs nanocomposites prepared by both the processing methods. β-Spherulites were identified by their negative birefringence color typical of β crystallization. 39 Ag-NPs promote the formation of spherulites with a diameter of about 21.6 ± 7.1 µm, according to the micrographs obtained for these nanocomposites. Figure 8(a) shows the spherulites growth in β-form like little white circles (β-spherulites are indicated by arrows). Samples processed in the extruder presented spherulites growth in minor proportion compared with Brabender mixer (see Figure 8(b)). The Ag-NPs resulted in a large number of nuclei and induced a large number of β-spherulites in the limited space of the matrix.

Polarizing optical micrographs of iPP/Ag-NPs nanocomposites with β-spherulite: (a) B-10/10 and (b) E-10/10. iPP: isotactic polypropylene; Ag-NPs: silver nanoparticles.
Conclusions
In conclusion, we have presented a novel and easy method for the preparation of iPP/Ag nanocomposites via melt blending using PEG as precursor of Ag-NPs and surface modifier in the iPP matrix. Ag-NPs with a particle size of 80 nm using PEG as a reducing agent and stabilizer were obtained. The nanocomposites were prepared via melt compounding by two different methods of dispersion, which had a significant effect on their properties as well as the final Ag content. PEG remaining is presented in nanocomposites according to FTIR analysis. Typical growth and morphology of β-spherulites in iPP were observed by AFM and PLM. According to DSC and XRD analysis, the β-phase induction in iPP/Ag nanocomposites were observed, but samples prepared by Brabender mixer obtained higher values of crystallinity, 31.1% relative amount of β-phase, higher than those reported with similar techniques. The elongation at break was directly dependent on the preparation method and the relative amount of crystalline β phase due to the good dispersion of Ag-NPs in the iPP matrix. PEG is an excellent precursor to obtain Ag-NPs and a good surface modifier between iPP and Ag-NPs. This method is advantageous over other methods in other studies because it requires no additives, compatibilizer, surfactant or reducing agent, and stabilizer are needed in the procedure.
Footnotes
Acknowledgments
The authors are thankful to Engineer Sebastian Pacheco from CICATA (Altamira) for technical assistance with X-ray diffraction samples and Indelpro (Altamira) for their kind donation of polypropylene (XH1760).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by CONACYT (grant numbers 491405, 237583 and 220989) and PROMEP.
