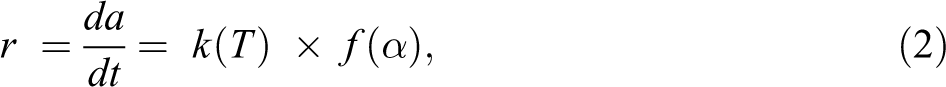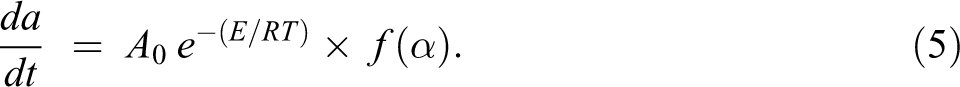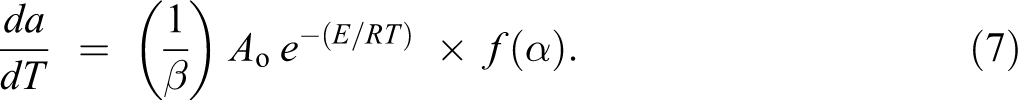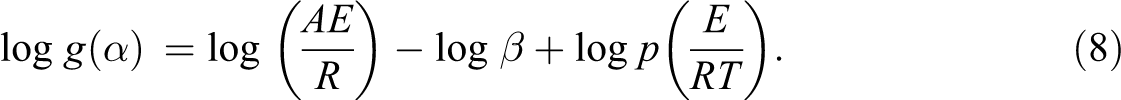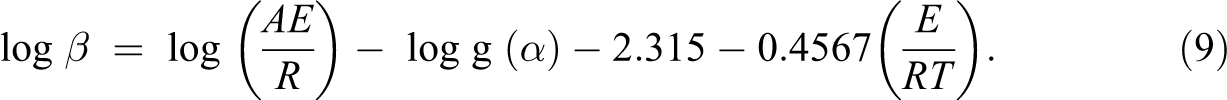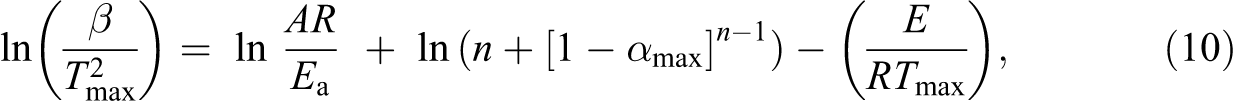Abstract
This study demonstrated that poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane (POSS)) (P(S-co-NMIP)) was successfully prepared using free radical polymerization. For this purpose, firstly, N-maleimide isobutyl POSS (NMIP) was synthesized using aminopropyllsobutyl POSS (POSS-NH2) and maleic anhydride. Secondly, P(S-co-NMIP) was synthesized using styrene, NMIP, and 2,2-azobis(isobutyronitrile) as initiator in tetrahydrofuran for 24 h at 75°C to give copolymer. The synthesized polymer and compounds were characterized by proton nuclear magnetic resonance, gel permeation chromatography, and Fourier transform infrared spectroscopy. Thermal behaviors of P(S-co-NMIP) were analyzed using thermogravimetric and differential scanning calorimetric analyses. The apparent activation energies (Es) for thermal degradation of P(S-co-NMIP) were obtained by integral methods (Flynn–Wall–Ozawa (FWO) and Kissinger). P(S-co-NMIP) was heated thermogravimetrically under various heating rates such as 5, 10, 15, and 20°C min−1 at a temperature range of 30–1000°C to determine their thermal degradation mechanisms. The values of E for P(S-co-NMIP) were found to be 127.5 ± 2.3 and 134.4 ± 14.8 kJ mol−1 for FWO and Kissinger methods, respectively. Also, the values of E of synthesized copolymer (P(S-co-NMIP)) were compared with styrene-co-maleic anhydride copolymer in literature.
Introduction
Nowadays, developing technology that can respond to the growing needs of the pursuit of superior materials continues to increase. Hybrid materials are very important in these related studies because they carry the properties of both organic and inorganic materials. Being used as organic fraction in the hybrid materials, polymers bring flexibility, lightness, easy processability, and durability to products. Using advanced technology, polymers, such as poly(methyl methacrylate), poly(hydroxyethyl methacrylate), and polybenzoxazines, give thermal, mechanical, and chemical resistance to the product. 1 –4 Metal, ceramic, or silicate structures can be used as inorganic matrix. However, the use of ceramic-derived materials as matrix cause low thermal conductivity, low oxidation resistance, low density, and many features like these. Studies on polyhedral oligosilsesquioxane (POSS) gain much importance because POSS distinguishes its superior character as hybrid materials from ceramics. POSS is one of many kinds of silsesquioxane molecules. The term silsesquioxane refers to the molecules whose chemical structure follows the basic composition of RnSi n O1.5n. Here, the R group, also called the vertex group for polyhedral molecules, may be hydrogen, alkyl, alkene, and aryl arylene. 5 –9 POSSs, which have properties between silica and silicon, are spherical hybrid structures with the molecular size of about 1–3 nm. Nonetheless, adversely to silica, silicon, and other packing materials, POSS contains organic groups that align the structure with polymer, biological systems, and surfaces. Adversely to regular organic composition, they do not release volatile organic compounds and they are regarded as scentless and environment friendly. These primal properties make POSS hybrid molecule a superior material, which lead to synthesize composites with polymers or molecules, nanocreatures, and graft materials by using copolymerization. 10 –16 Also, for many years, copolymerization has been one of the most useful methods for modifying and improving polymer properties. Copolymers containing N-substituted maleimide moieties show enhanced thermal and mechanical properties, representing a class of highly stable materials. Various N-substituted maleimide copolymers have been studied in several fields of applications related to high temperature and photoresistance. 17 –22
In this work, we directly synthesized a POSS-containing poly(styrene-co-N-maleimide isobutyl POSS) (P(S-co-NMIP) alternating copolymer (as seen Figure 1) using maleimide isobutyl POSS (NMIP) via free radical polymerization. We could not find any thermal behavior details of that polymer in literature. Also, we saw that kinetic studies of POSS-containing polymer are not sufficient in literature, so we felt it is worth studying the thermal behavior of synthesized polymer and thermal degradation kinetics of P(S-co-NMIP). The apparent values of E for thermal degradation of the copolymer were obtained using Flynn–Wall–Ozawa (FWO) and Kissinger methods.
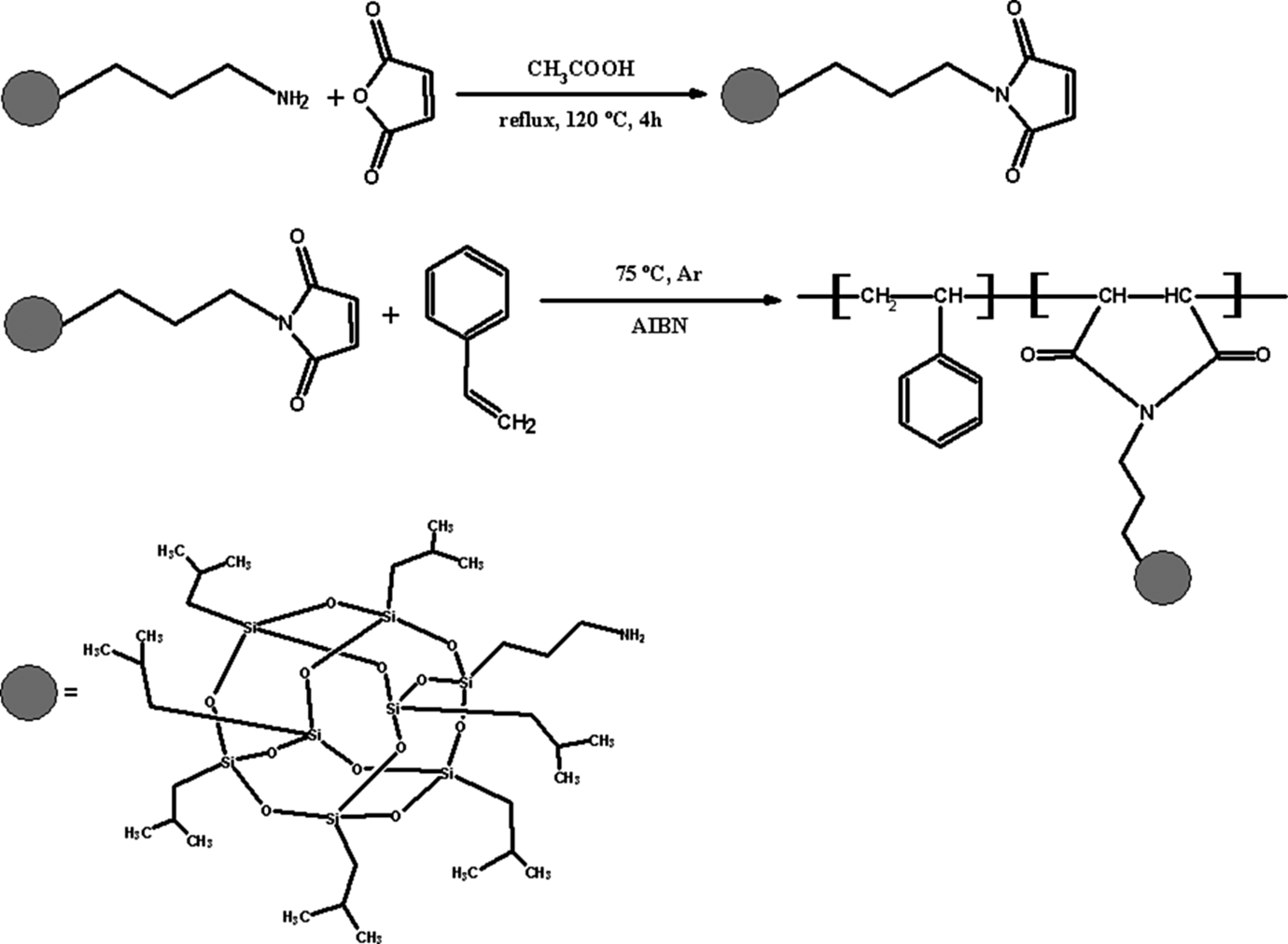
Synthesis of NMIP and P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; NMIP: N-maleimide isobutyl polyhedral oligosilsesquioxane.
Kinetic analysis
Thermogravimetric (TG) analysis can be used for determining the degradation kinetics of a lot of polymers. 23 –26 In general, the thermal degradation reaction of a solid polymer can be shown as:
where A is the starting material and Bsolid and Cgas are the solid residue and the gas product, respectively.
The thermal degradation kinetics of the polymers is generally expressed by the following typical kinetic equation:
where T is the absolute temperature (K), r is the conversion per unit time (t), and f(α) is the conversion function that represents the reaction model. The degree of conversion (α) is calculated by equation (3) where mo, mt, and mf are the weights of sample before degradation, at time t, and after complete degradation, respectively.
where k is the reaction constant which can be expressed by the Arrhenius equation:
where A is called pre-exponential factor, E is the activation energy, and R is the gas constant.
By combining the equations (2) and (4), the following equation is obtained:
According to the kinetic theory for the non-isothermal decomposition reactions, the fractional conversion α is expressed as a function of temperature that depends on the time of heating. Thus the heating rate (β) can be described as:
Then, equation (5) is modified as follows:
Equations (5) and (7) are the bases for many equations derived to evaluate thermal analysis data.
FWO method
The value of E can be determined by this method with no need of any knowledge for the reaction mechanism. 27,28 The pre-exponential factor (A) and the activation energy (E) are not dependent on the fraction of degradation, but they are dependent on the temperature. This method uses the following equation:
Equation (9) is obtained by means of the Doyle approximation:
Hence, from the slope −E/R of the linear plot of log β versus 1000/T, E is readily obtained.
Kissinger method
The value of E can be calculated using equation (10) with Kissinger method without knowing the solid-state degradation reaction mechanism. 29
where b is the heating rate, Tmax is the temperature related to maximum reaction rate, A is the pre-exponential factor, amax is the maximum degradation fraction, and n is the reaction order. Plotting
Experimental
Materials
Styrene (S, 99%), maleic anhydride puriss (NT, ≥99.0%), acetic acid reagent (ACS, ≥99.7%), and tetrahydrofuran (THF) anhydrous (≥99.9%) were purchased from Sigma-Aldrich (St Louis, Missouri, USA). 2,2-Azobis(isobutyronitrile) (AIBN, 98%; Sigma-Aldrich) was recrystallized from ethanol. Aminopropylisobutyl POSS (≥97.0%) was purchased from Hybrid Plastics Inc. (Hattiesburg, Mississippi, USA).
Instrumentation
Proton nuclear magnetic resonance (1H NMR) measurements were recorded using deuterated chloroform (CDCl3) with Si(CH3)4 as internal standard, using Varian AS-400 instrument (400 MHz; Palo Alto, California, USA). (The sample weights were taken in the range of 10–15 mg in 2 mL CDCl3). Fourier transform infrared (FTIR) spectra were recorded on a PerkinElmer FTIR Spectrum One-B spectrometer (Waltham, Massachusetts, USA). Potassium bromide (KBr) pellet was used for FTIR analysis. A small amount of powder sample (about of 0.1–2% of the KBr amount or just enough to cover the tip of spatula) was mixed with the KBr powder. Subsequently, the mixture was ground for 3–5 min. Molecular weights were determined by gel permeation chromatography (GPC) instrument, equipped with a Waters styragel column (HR series 2, 3, and 5E; Milford, Massachusetts, USA), with THF as the eluent at a flow rate of 1.0 mL min−1 and a Waters 410 differential refractometer detector. (Sample for GPC analysis was prepared as follows: 10 mg sample was dissolved in 3 mL of THF). To obtain the TG and differential scanning calorimetric (DSC) measurements of powders, polymer samples were obtained on PerkinElmer Diamond TA/TGA from 25°C to 1000°C with different rates (5, 10, 15, and 20°C) at a constant flow rate of 100 mL min−1 under nitrogen (N2) atmosphere. The sample weights for all the experiments were taken in the range of 8–10 mg.
Synthesis of NMIP
Acetic acid (5 mL), aminopropyllsobutyl POSS (0.1 g, 1143 × 10−1 mmol), and maleic anhydride (0.0112 g, 1143 × 10−1 mmol) were charged in a 25 mL flask with a magnetic stirring bar. The reactive mixture was degassed via three pump-freeze–thaw cycles and then immersed in a thermostatic oil bath at 120°C for 4 h. After the reaction mixture was cooled to room temperature, the obtained solution was dropped into an excessive amount of water. The product was dried under vacuum overnight with a yield of 80%. 1H NMR (400 MHz, CDCl3, δ ppm): 6.60–6.70 (2H, d), 3.40–3.50 (2H, t), 1.80–1.90 (7 H, m), 0.95–1.0 (42 H, –CH2CH (C
Synthesis of P(S-co-NMIP)
A typical polymerization procedure for the synthesis of a P(S-NMIP) copolymer is as follows: a 10-mL glass flask containing a stirring bar was charged with NMIP (0.25 g, 2.62 × 10−1 mmol), styrene (0.35 mL, 3045 mmol), AIBN (0.0163 g, 0.09921 mmol), and 5.0 mL of THF. The solution in the glass flask was degassed via three pump-freeze–thaw cycles, and then the glass tube was sealed under a vacuum. The polymerization was carried out in an oil bath at 75°C for 24 h. The solution was precipitated into the excess methanol. The precipitate was filtered, and the product was dried at room temperature in a vacuum overnight with a yield of 55%. Mn = 6700; Mw = 12,300; and PDI = 1.83. 1H NMR (400 MHz, CDCl3, δ ppm): 7.0–7.1 (m, Ar–H in PS), 6.5–6.6 (m, Ar–H in PS), 3.0–3.25(m, –NCH2CH2), 1.88 (m, –CH2CH (CH3)2), 1.00–1.55 (m, polymer chain –CH2–, –CH–), 0.90–0.1 (m, –CH2CH (CH3)2), 0.5–0.60 (m, –CH2CH (CH3)2). FTIR %T (cm−1): 3400, 3100, 3000, 1800–1950, 1750.
Results and discussion
P(S-co-NMIP) alternating copolymer is synthesized by radical polymerization. It is well known that the copolymerization of styrene with maleic anhydride or maleimide could produce a well-defined alternating structure through a charge-transfer complex. Therefore, the POSS-NH2 with a maleimide group should also have a strong alternating tendency of copolymerization with styrene to prepare the POSS-containing hybrid alternating copolymer via the formation of a charge-transfer complex. Polymerization was carried out in THF at 75°C for 24 h using AIBN as an initiator.
The FTIR spectrums of POSS, maleic anhydrite, NMIP, and P(S-co-NMIP) are shown in Figure 2. Firstly, maleic anhydride was reacted with POSS-NH2 to get NMIP. The appearance of N–H and –CH2 stretching of NMIP can be seen at around 3400 cm−1 and 2850 cm−1 in the spectra, respectively. On the other hand, it is also supported by CH2=CH2 stretch around 1640 cm−1. This shows successful synthesis of NMIP. Otherwise, this figure shows that after polymerization of NMIP with styrene, the appearance of aromatic ring stretch belongs to styrene is clear at around 3100 and 1900 cm−1. As well as, this is also supported by the disappearance of the CH2=CH2 stretching around 1640 cm−1.
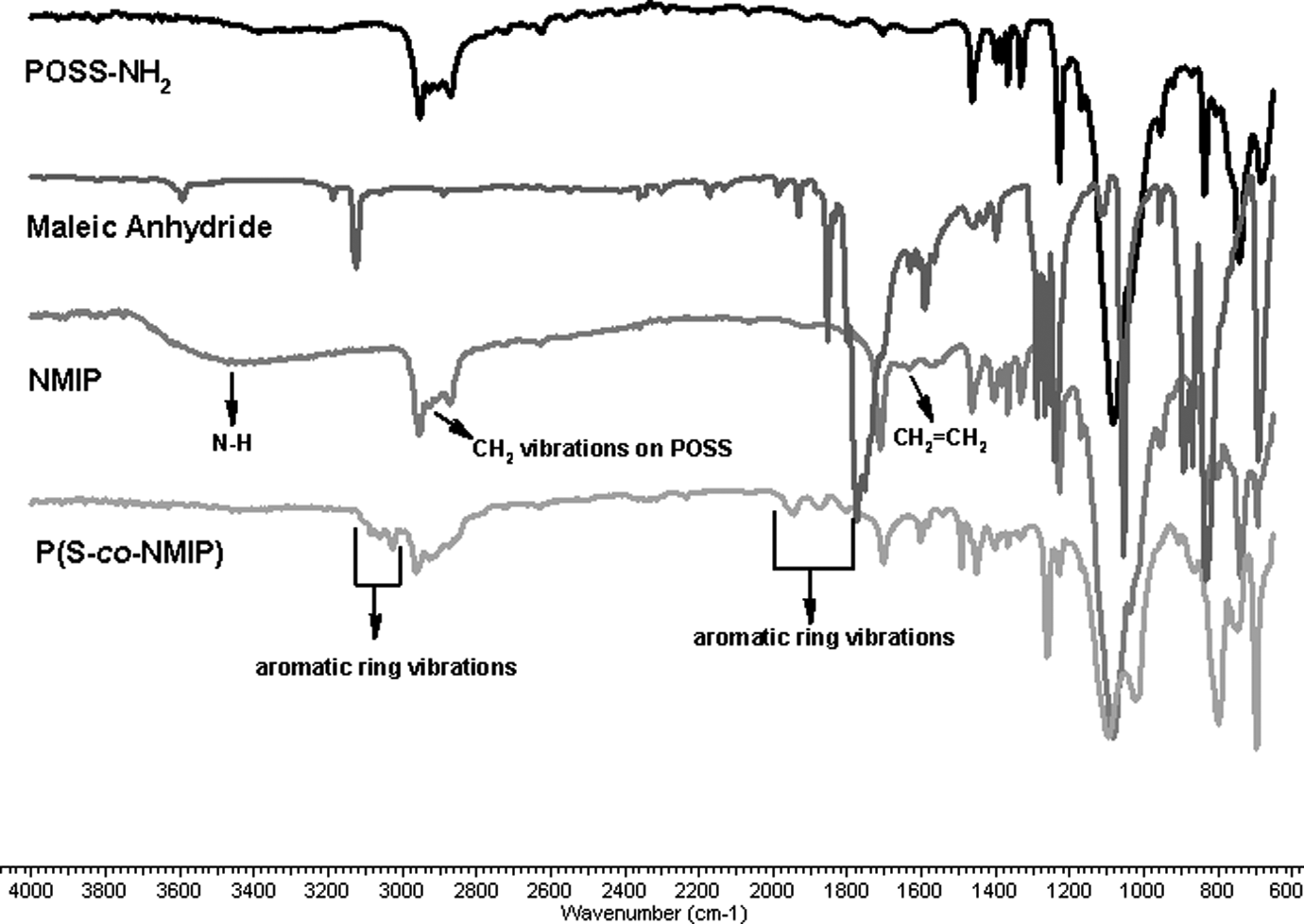
FTIR spectrums of POSS, maleic anhydrite, NMIP and P(S-co-NMIP). FTIR: Fourier transform infrared spectroscopy; P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; NMIP: N-maleimide isobutyl polyhedral oligosilsesquioxane; POSS: polyhedral oligosilsesquioxane.
We have also supported these results by 1H NMR. Figure 3 shows the 1H NMR spectrum of NMIP and P(S-co-NMIP). According to this figure, it is observed that Si-C
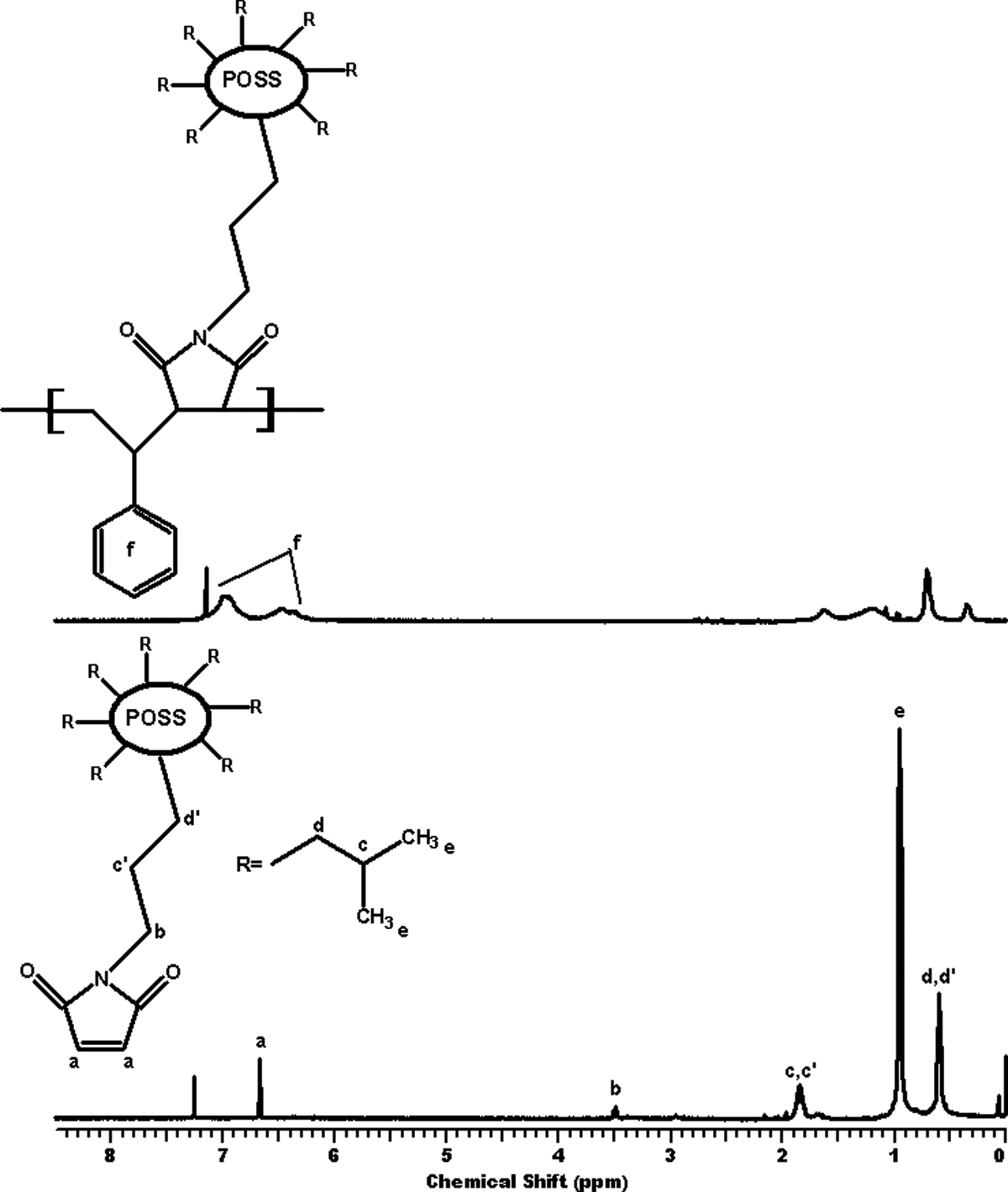
1H NMR spectrum of NMIP and P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; NMIP: N-maleimide isobutyl polyhedral oligosilsesquioxane; 1H NMR: proton nuclear magnetic resonance.
TG and DSC measurements were obtained to study the thermal behavior and thermal stability of the prepared P(S-co-NMIP). TG/derivative TG (DTG) and DSC curves of the polymers are shown in Figures 4 and 5, respectively. According to the data obtained from Figure 4, TG thermograms under N2 atmosphere revealed that P(S-co-NMIP) degradation takes place in one stage that occurs at a temperature around 325°C. Also, Tmax is about 375°C. TG and DTG curves of P(S-co-NMIP) are similar to styrene–maleic anhydride copolymer (st/ma) curves, but due to the presence of POSS molecule in the copolymer, the decomposition temperature is higher than st/ma. 27 Glass temperature of P(S-co-NMIP) is also investigated by DSC (Figure 5). In the DSC curve, the glass transition of the P(S-co-NMIP) is detected at around 175°C. According to data obtained from DSC study, it is clear that the glass transition temperature of the P(S-co-NMIP) is enhanced by POSS when compared with st/ma or cross-linked st/ma. 30,31
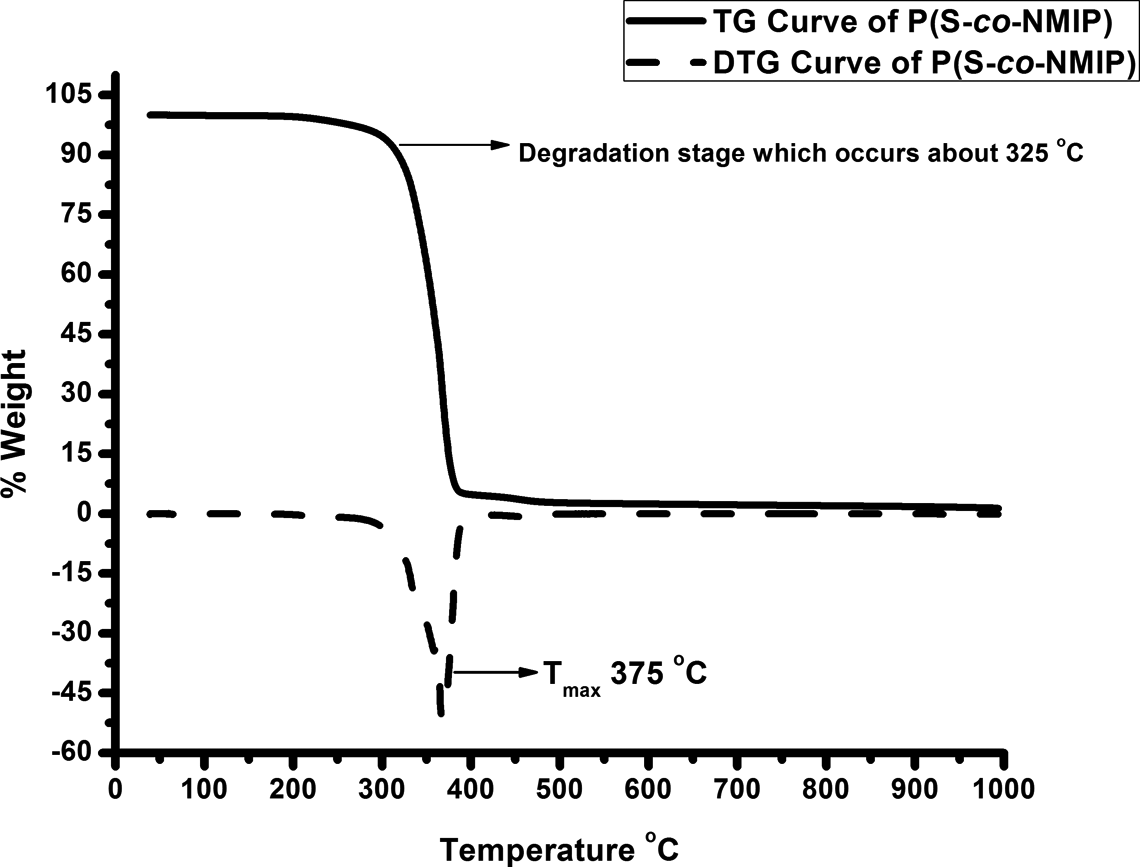
TG-DTG curves of P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; TG: thermogravimetry; DTG: derivative thermogravimetry.
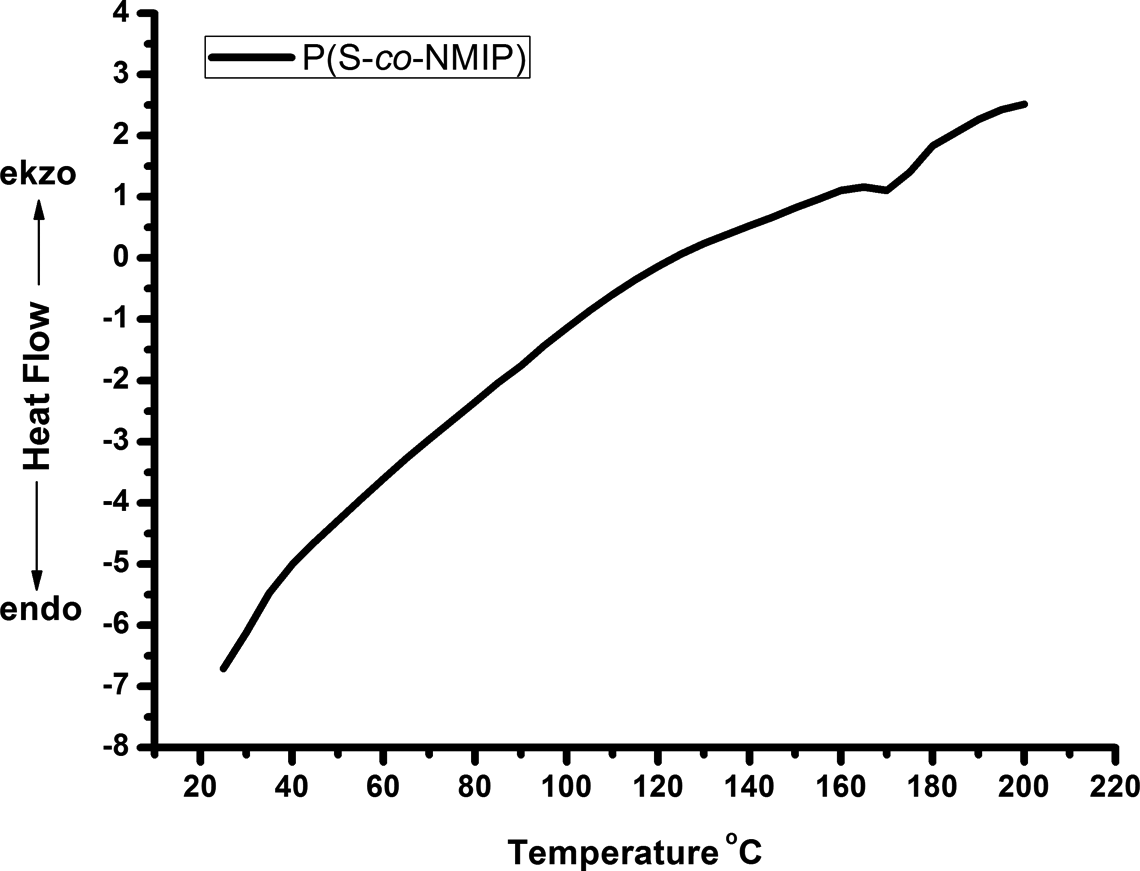
DSC curve of P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; DSC: differential scanning calorimetry.
Thermal degradation kinetics of P(S-co-NMIP)
P(S-co-NMIP) was heated thermogravimetrically under various heating rates such as 5, 10, 15, and 20°C min−1 with a temperature range of 30–1000°C to determine their thermal degradation mechanisms and the values of E. The TG and DTG curves obtained for P(S-co-NMIP) are shown in Figures 6 and 7, respectively. The individual degradation behavior of copolymer was analogous at all heating rates as seen from these figures. The apparent Es and thermal degradation models for the copolymer were estimated by FWO and Kissinger methods. These methods are most widely used as the integral methods that can determine the value of E without the knowledge of reaction mechanism. Pre-exponential factor (A) and activation energy (E) do not depend on degradation fraction, but they depend on the temperature. The calculated values are shown in Tables 1 and 2. FWO and Kissinger methods were first employed to analyze the TG data of P(S-co-NMIP) because they were independent of any thermal degradation mechanism.
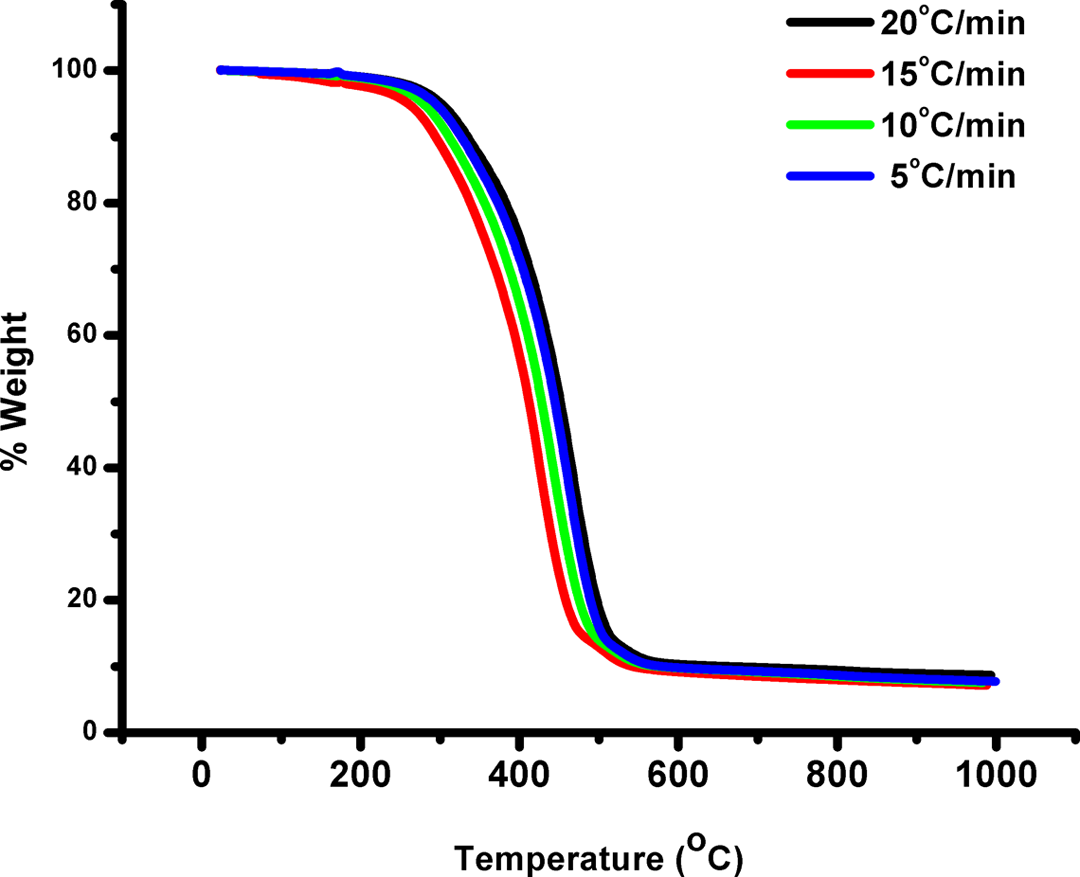
TG curves obtained for the P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; TG: thermogravimetry.
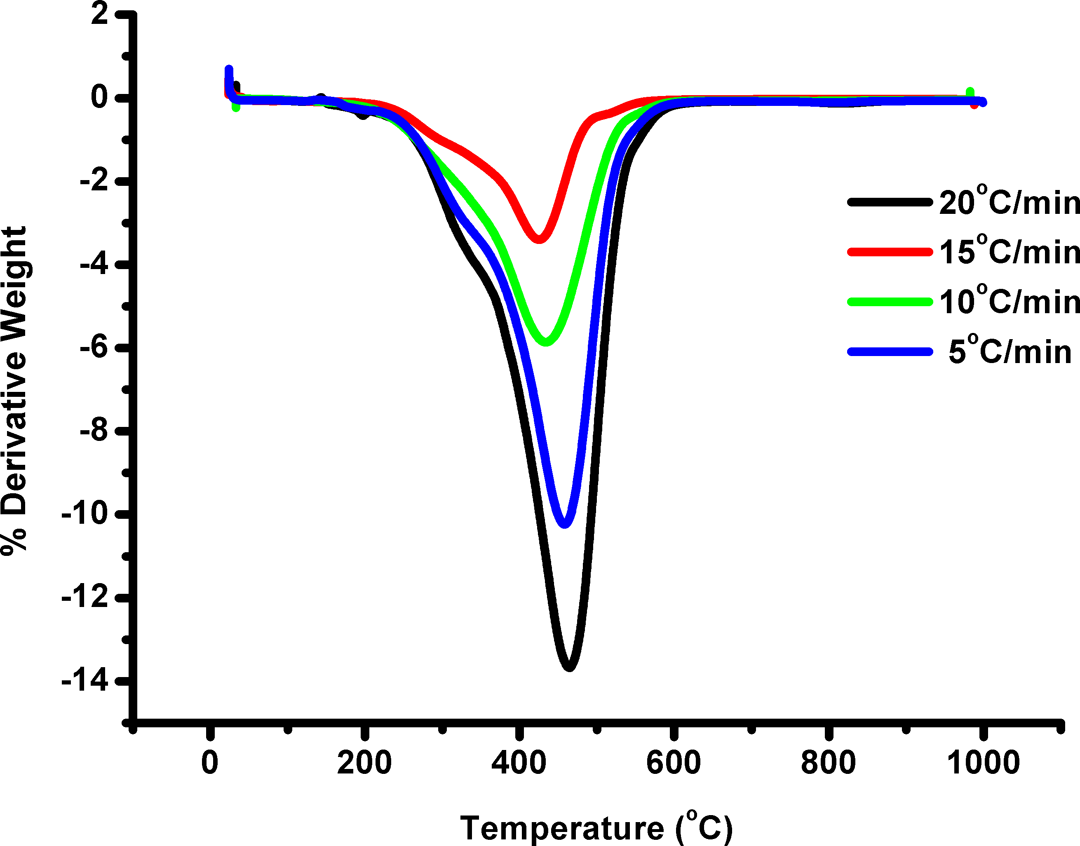
DTG curves obtained for the P(S-co-NMIP). P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane; DTG: derivative thermogravimetry.
The degradation activation energies of P(S-co-NMIP) calculated by FWO method.
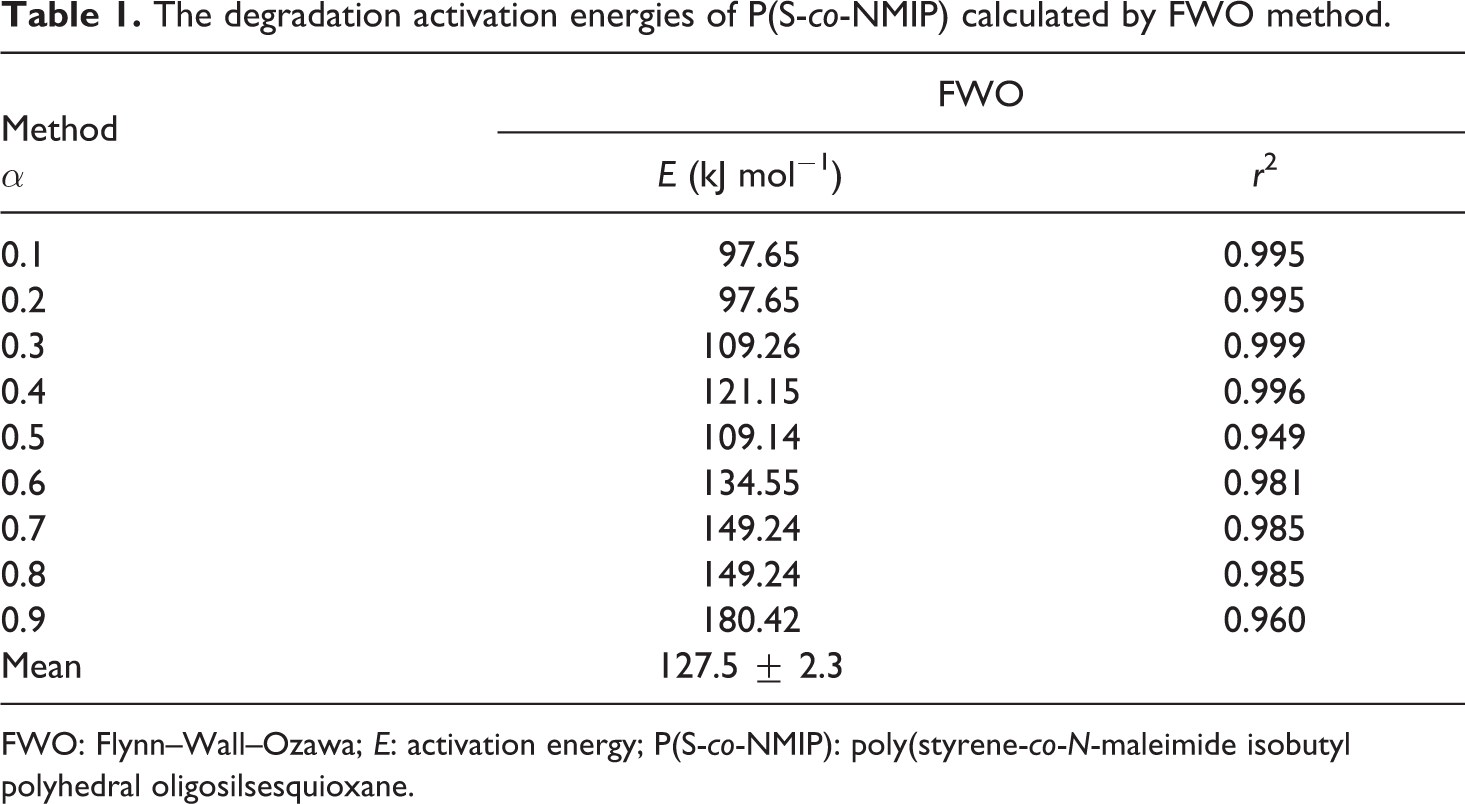
FWO: Flynn–Wall–Ozawa; E: activation energy; P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane.
Tmax and α values of P(S-co-NMIP) at the different heating rates.

Tmax: maximum degradation temperature; α: residual mass; β: heating rate; P(S-co-NMIP): poly(styrene-co-N-maleimide isobutyl polyhedral oligosilsesquioxane.
The E value of P(S-co-NMIP) was found to be 127.5 ± 2.3 kJ mol−1 for main stage by means of the classical FWO method. The average E values of P(S-co-NMIP) were calculated by Kissinger method as 134.4 ± 14.8 kJ mol−1 for main stage. The calculated E values for P(S-co-NMIP) seem to be pretty higher than that of st/ma, which was found to be 64.7 kJ mol−1. 32
Conclusion
In conclusion, P(S-co-NMIP) was prepared by using free radical polymerization method. The synthesis of this polymer was supported by FTIR spectroscopy and 1H NMR. Thermal properties and degradation were investigated by DSC and TG analyses. According to DSC and TG curves, POSS hybrid molecule could affect the thermal stability and the kinetics of thermal degradation of st/ma. The values of E calculated by FWO method for P(S-co-NMIP) were found to be 127.5 ± 2.3 kJ mol−1, while it was 134.4 ± 14.8 kJ mol−1 for P(S-co-NMIP) by Kissinger method. The synthesized copolymer (P(S-co-NMIP)) has significantly higher E value, glass transition temperature, and thermal stability than st/ma. The reason should be advantageous performance in mechanical and thermal properties of POSS hybrid molecule which can increase the temperature dependence of st/ma.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.