Abstract
The waterborne polyurethane/nano-silica composites (WPU/nano-silica, WPUS) and WPU composites modified by polyethersiloxanediol (WPUPES) were prepared, respectively. The properties of WPUS and WPUPES were investigated by various characterizations. The results showed both WPUS and WPUPES had better waterproof property and thermal stability than neat WPU. However, WPUPES has a lower elongation at break due to the higher micro-phase separation. This is ascribed to migration and aggregation of siloxane segments during the film formation. The tensile strength of WPUS was higher than that of neat WPU. This is attributed to the WPUS chain restriction caused by the network and physical cross-link points of nano-silica particles. Moreover, the glass transition temperature of WPUS shifted to higher temperature region while that of WPUPES shifted to lower temperature region.
Introduction
Polyurethane (PU) is a versatile and commercially important polymer used in some fields such as plastics, foams, elastomers, coatings, and adhesives.
1
–3
In general, PU possesses the structure
Nano-silica and polysiloxane are widely used to modify properties of WPU due to their advantages. Since nano-silica has spherical, amorphous, and isotropic properties, incorporation of it can improve water resistance, heat resistance and radiation resistance properties, and mechanical properties of WPU. 7 Since polysiloxane has good properties, such as low glass transition temperature (T g), low surface energy, good biocompatibility, and high thermal and oxidative stability, incorporation of it can bring out lots of advantages, such as good water and chemical resistance. 8 For WPUS composites, there is a drawback that the nano-silica does not have a good compatibility with PU matrix, 9 so the in situ polymerization method was adopted to increase interaction between nano-silica particle and WPU matrix. 10 For preparation of WPU/polyethersiloxanediol composites (WPUPES), the widely used method is the incorporation of polysioxanediol as soft-segment material. 11 Up to now, numerous literatures were focused on the preparation and the influence of filler 12 ; however, to the best of our knowledge, there are few reports presented about the study of properties comparison of WPUS and WPUPES.
In this work, WPUS composites were prepared via in situ polymerization. The relative small size (5–30 nm) of nano-silicon dioxide was used here due to the reason that the chosen particle size must be equal to or less than the radius of gyration (R g) of the polymer film. 13,14 In the meantime, we also prepare WPUPES composites by the incorporation of a novel polyethersiloxanediol. Polyethersiloxanediol does not contain the OH–Si, so during the polymerization, the hydrolytic Si–O–C structure can be avoided. 15 For comparison, WPU also was prepared. The morphology, water resistance, thermal stability, and mechanical properties of WPU, WPUS, and WPUPES were investigated here. Furthermore, related mechanism was discussed to understand the relationship between structure and properties.
Experimental
Materials
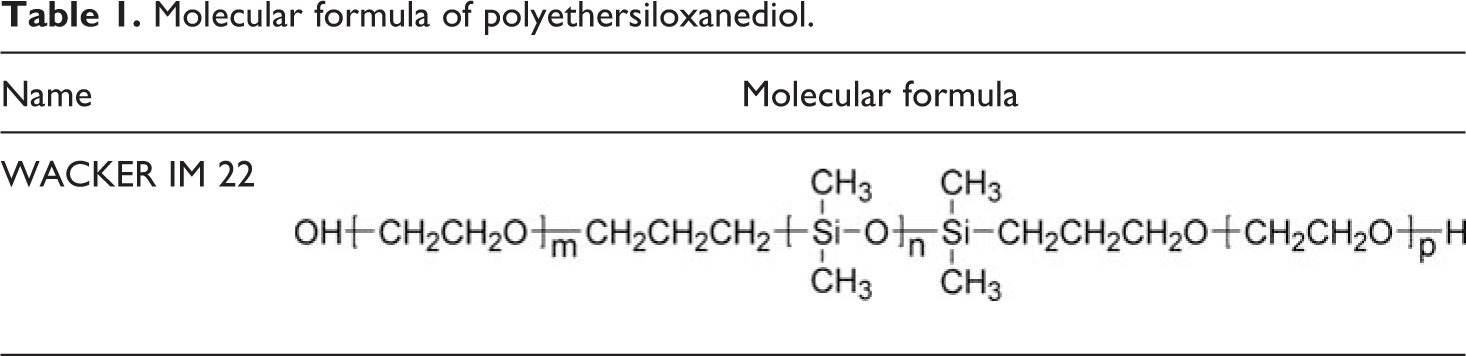
Hexamethylene diisocyanate (HMDI) was obtained from Bayer (Germany). Polytetramethylene glycol (PTMG; 2000 g mol−1) and neopentyl glycol (NPG) were obtained from Mitsubishi Chemical Corporation (Japan). HMDI was vacuum distilled before use. PTMG was dried and degassed at 8°C under vacuum for 3 h. Dimethylol butanoic acid (DMBA) and pentaerythritol (PE) were purchased from Perstorp Specialty Chemicals company (Sweden). NPG, PE, and DMPA were dried at 50°C for 48 h in vacuum oven. Dibutyltin dilaurate (DBTDL), dimethyl formamide (DFM), and triethylamine (TEA) were obtained from Chengdu KeLong Chemical Industry Co., Ltd (China). DBTDL was used as received. DFM and TEA were dried before use. Polyethersiloxanediol (1694 g mol−1; WACKER IM 22, Germany; Table 1) was purchased from Dow Corning (Midland, Michigan, USA). Fumed nano-silica was offered by Wacker Chemie. Polyethersiloxanediol and nano-silica were used directly without further purification. Double-distilled and deionized water was used throughout.
Molecular formula of polyethersiloxanediol.
Preparation of WPU, WPU/nano-silica composites, and WPU composites modified by polyethersiloxanediol
The synthesis of neat WPU, WPUS, and WUPES was adopted from literature procedures, 16 and some of the reaction conditions were modified for our needs. The preparation route is shown in Figure 1. A 500-ml round-bottomed, four-necked separable flask with a mechanical stirrer, thermometer, and condenser with drying tube was used as a reactor. The reaction was carried out in a constant temperature oil bath. The reaction mixture of PTMG and HMDI was stirred in the presence of Dibutyltin dilaurate (DBTAL) for 2 h at 80°C. Then DMBA and NPG dissolved in DFM were added to the flask and reacted till the free –NCO reached the theoretical content. 17 Then polyethersiloxanediol (PESD) (amounting 14 wt% of WPUPES) dissolved in DFM was then added to the reaction mixture to get the prepolymer of WPUPES. To get the prepolymer of WPUS, the nano-silica (amounting 4 wt% of WPUS) dispersed in DFM was added instead. Both the methods were allowed to proceed at 80°C till the free –NCO reached the theoretical content. Next, the PE was added drop by drop to the mixture with adequate DFM and DBTDL. When the content of free –NCO reached its theoretical end point, the mixture was cooled down to 60°C. Then TEA was added and mixed thoroughly for 30 min, and distilled water was added to the polymer at a constant flow rate to the flask under vigorous magnetic stirring (2000 r min−1) at room temperature over 5 min. Finally, the solid content is 35%.
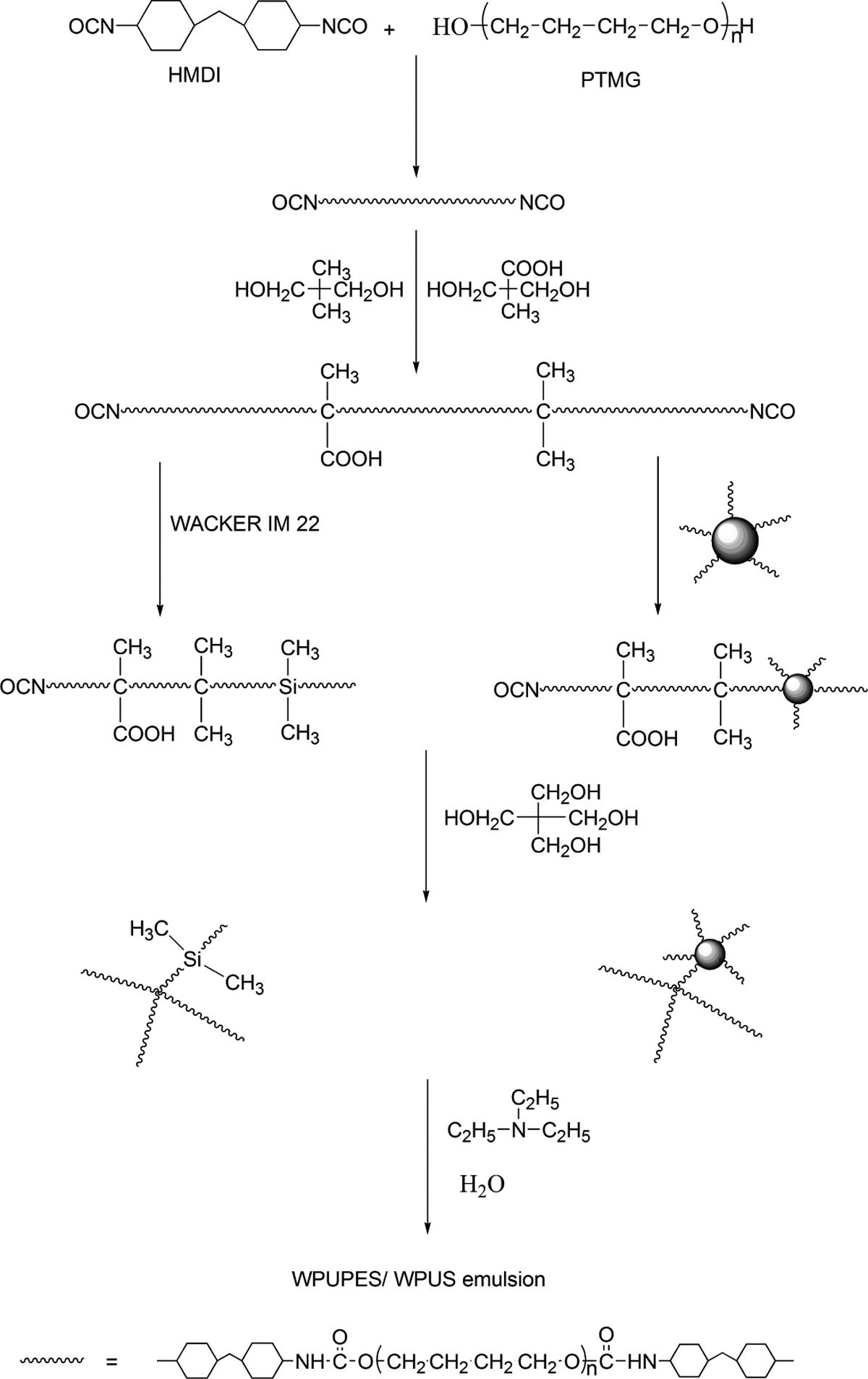
Preparation route of WPUS and WPUPES. WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Preparation of film
Films specimens were prepared by casting emulsion onto the glass plates and were firstly dried at an ambient temperature for 5 days and then at 60°C for 24 h. The thickness of the obtained films was about 0.4 mm. Finally, the films specimens were stored in a desiccator to avoid moisture.
Characterization
Fourier transform infrared
Fourier transform infrared (FTIR) spectra of the films were performed on a NEXUS 670 spectrophotometer (Thermo Fisher Scientific, Waltham, Massachusetts, USA) in transmission mode. The spectra were obtained from 4000 cm−1 to 400 cm−1 at a resolution of 2 cm−1. The samples were dried into films with 0.1 mm thickness for measurement.
Water absorption
Dried films (30 × 30 mm2), original weight designated as m 0, were immersed in water for 24 h at 25°C. After the residual water wiped from the films using filter paper, the weight (m 1) was measured immediately. It was calculated, as follows:
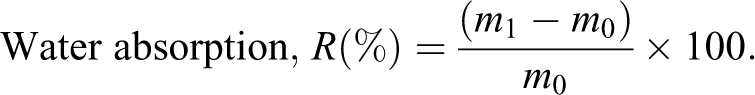
Contact angle measurement
The static water contact angles of the films were determined with the sessile drop method using the OCAH200 high-speed video contact angle measurer (DataPhysics Instruments GmbH, Germany). A 5-μl double-distilled water droplet was used for each point, and the readings were taken when the droplet on the film was stabilized. The contact angle values reported are the average of five measurements taken at five different locations.
Atom force microscopy
Atom force microscopy (AFM) was performed on the surface of the film with a scanning area of 5 × 5 μm2 using the SPA-400 atomic force microscope (Seiko Instruments Inc., Japan). The images were obtained under ambient conditions in tapping mode using a silicon nitride tip. Roughness analysis of the surface was carried out using NanoScope Analysis software-v1.40r1.
Differential scanning calorimetry
Differential scanning calorimetric (DSC) thermograms were recorded on DSC200 (NETZSCH Instruments, Germany), with a heating rate of 5°C min−1 from −100°C to 150°C under nitrogen atmosphere. The specimens of mass approximately 5 mg were placed in standard aluminum pans for measurement.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was carried out in TG 209 F1 differential thermogravimetric analyzer (NETZSCH Instruments), with a heating rate of 10°C min−1 from 25°C to 550°C under nitrogen atmosphere. The specimens of mass approximately 5 mg were placed in a crucible for measurement.
Mechanical properties
Mechanical properties were measured on the SDL201 tensile testing machine (UK) at a crosshead speed of 100 mm min−1 under ambient conditions. The size of the film specimens is 50 ± 0.02 mm in gauge length, 25 ± 0.02 mm in width, and approximately 1.0 mm in thickness. The results reported are the average values for five replicates.
Results and discussion
FTIR spectroscopy was applied in order to find out the presence of the silica structure in modified PU matrix. The FTIR results of PU, WPUS, and WPUPES are shown in Figure 2. From the spectra of the three samples, it can be found that the absorption peak at around 1727 and 1539 cm−1 is ascribed to the stretching vibration of C=O and deformation vibration of N–H in amide bond, respectively. Another peak at 1240 cm−1 belongs to the stretching vibration peaks of C–O–C structure in –NHCOOR group. That shows the prepared specimens contain the urethane structure, indicating that these PU specimens had been successfully synthesized. In the spectrum of WPUS, the absorption peak at 1100 cm−1 attributes to the Si–O–CH3 characteristic absorption, and the absorption peak at 453 cm−1 is associated to the deformation of Si–O–C and absorption of Si–O–Si, the mentioned peaks indicated that the nano-silica had been covalently grafted into the PU chains. 18 In the spectrum of WPUPES, characteristic absorption peak of Si–O–CH3 can also be found at 1100 cm−1, the absorption peaks at 1020–1100 cm−1 are associated to the stretching vibration of Si–O–Si and C–O–C, and the peak at 804 cm−1 attributes to the rocking vibration of Si–CH3, and these changes illustrate that WPUPES has been incorporated into the PU chain matrix. 19
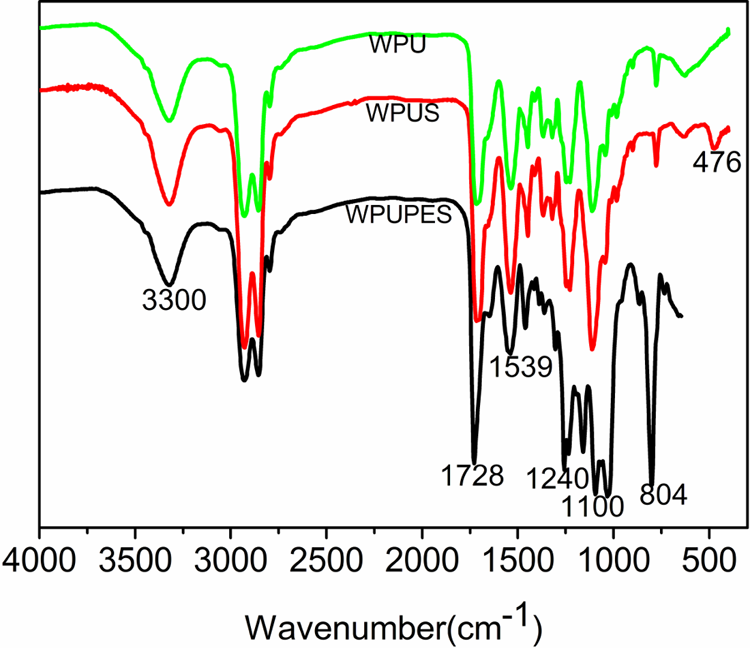
FTIR spectra of WPU, WPUS, and WPUPES. FTIR: Fourier transform infrared; WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Surface morphology
Phase separation occurred during the formation of PU films, this unique structure contributed to the mechanical properties of PU materials. The nano-silica and polyethersiloxanediol are expected to influence the phase separation structure of PU. Hence we can use AFM analyses to figure out the multiphase nature of the PU specimens. 20 Figure 3 shows the height image, phase image, and three-dimensional image of WPU, WPUS, and WPUPES. From the height images, a typical two phase structure can be observed. The comparatively bright part corresponds to the micro-phase of the aggregated hard segments and the dark part corresponds to the micro-phase of the aggregated soft segments. This implies the existence of micro-phase separation in the film structure. From the height images of WPU and WPUS, it can be found that the phenomenon of micro-phase separation of WPUS is more obviously than that of WPU, indicating that the nano-silica has been made the incompatibility of two phases greater. In the height image of WPUPES, the dark spots can be found evenly distributed on the film surface, which is also shown in the phase image, as the dark part corresponds to the soft segments, this can imply that the aggregation of soft segments of WPUPES is more serious than WPU and WPUS. This phenomenon may be due to migration of siloxane chains to the film surface during the course of filming, finally causing the aggregation of soft segments.

AFM images of WPU, WPUS, and WPUPES. AFM: atom force microscopy; WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
The rougher surface may contribute to the hydrophobicity of films. 21 Thus, the surface roughness of root mean square (R q) and arithmetic average (R a) was calculated and the results are listed in Table 2. It is evident that both the nano-silica and polyethersiloxanediol had significant effect on the surface roughness of PU film. The R q and R a of WPUS and WPUPES are much greater than that of WPU. The R q and R a of WPUS are higher than that of WPUPES, and this is ascribed to the larger size of nano-silica compared to the siloxane groups migrated to the surface of the film.
Roughness values of WPU, WPUS, and WPUPES.
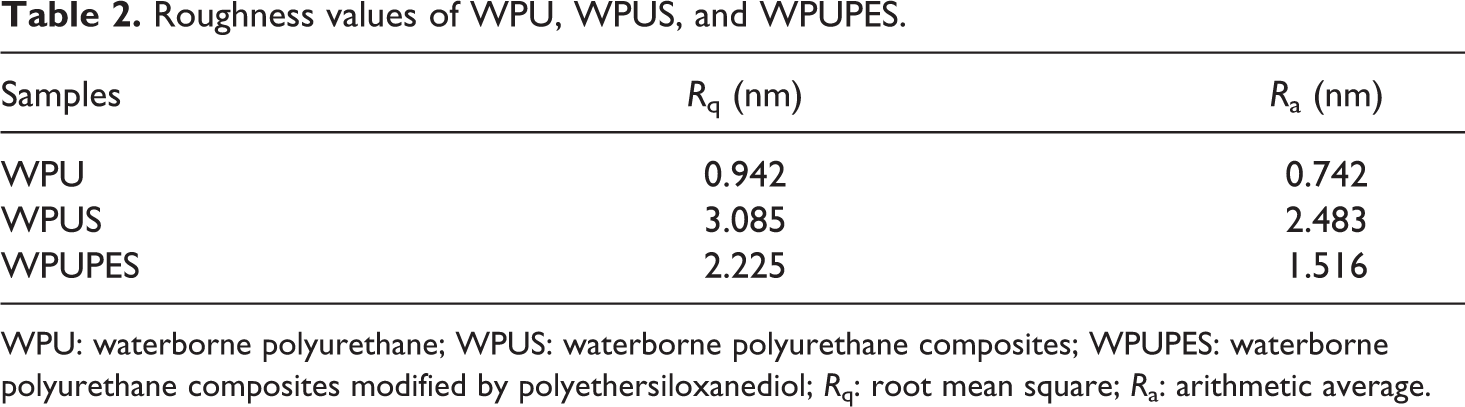
WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol; R q: root mean square; R a: arithmetic average.
Thermal properties
Incorporation of silica particles and siloxane structure to PU matrix is expected to influence the thermal stability of PU. T g is the transition temperature of polymer from glassy state to elastomeric state, the lower T g is assigned to the PU soft segment. 22 The T g values of the soft segment indicate the microstructure and phase change of the polymer. Figure 4 shows that the T g of the WPUS are slightly higher than that of WPU. This indicates the increase in cross-linking of PU matrix. 21 By contrast, the T g of WPUPES is 6.38°C lower than that of WPU, implying that WPUPES exhibit higher degrees of micro-phase separation. The starting and terminal points of the T g windows are listed in the Table 3. ΔT g is defined as the difference between the onset point and the end point of T g value. The ΔT g of the WPUS is close to that of WPU, which means that the nano-silica has little influence on the extent of micro-phase separation in the material. 23 However, the ΔT g of the WPUPES is lower than that of WPU, indicating the increasing extent of micro-phase separation in the material.
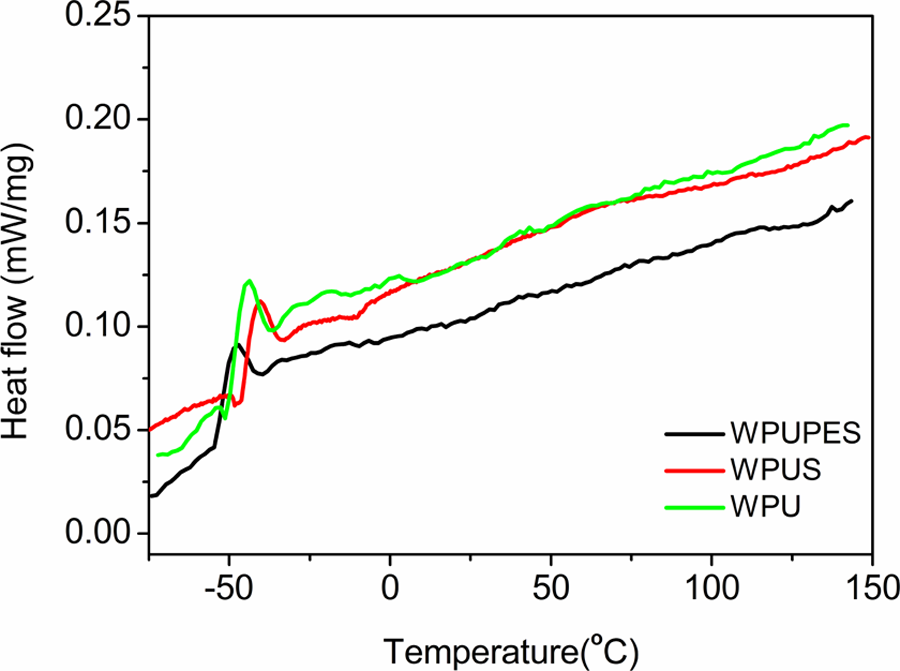
DSC curves of WPU, WPUS, and WPUPES. WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol; DSC: differential scanning calorimetric
The T g and ΔT g values of WPU, WPUS, and WPUPES.

WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol; T g: glass transition temperature; ΔT g: the difference between onset point and end point of T g.
TGA was performed to investigate the thermal stability of WPU, WPUS, and WPUPES. The results of thermal degradation behaviors of the three samples are shown in Figure 5. The weight loss from 100°C to 200°C is attributed to the loss of residual water and organic solvent. The next process, corresponding to the maximum weight loss in the range of 230–500°C, is due to the decomposition of the polymer chain. The temperature that is stemmed from point of intersection between the tangent line of decreasing steep and base line is used to represent the thermal stability of the materials. 24 It is found that the thermal stability temperature of WPUPES is approximately 30°C higher than that of neat PU, indicating that the thermal stability of the WPUPES was remarkably enhanced. This may be attributed to the high bond energy and thermal stability of silane linkages. Compared to neat PU, the thermal stability temperature of WPUS was also increased to 252.1°C. The increase can be ascribed to the fact that the incorporation of nano-silica causes strong interaction between nano-silica particles and macromolecular chains and the formation of physical cross-link network between nano-silica particles and PU matrix. However, it is lower than that of WPUPES. This may be ascribed to the poor dispersion of nano-silica in the PU matrix. It had been found that the nano-silica particles were well dispersed in the PU matrix up to 3 wt%. 18
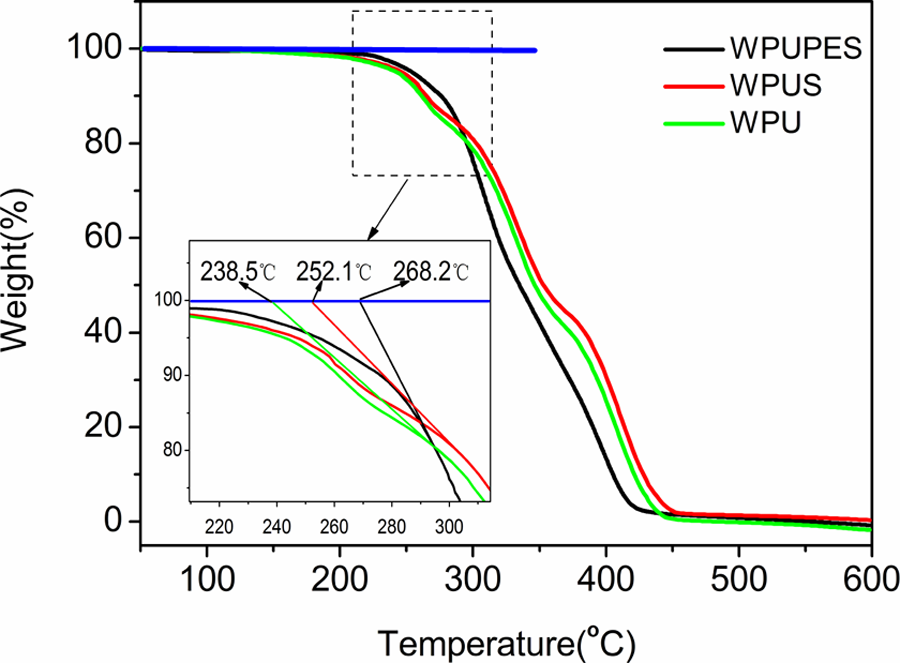
TGA curves of WPU, WPUS, and WPUPES. TGA: thermogravimetric analysis; WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Water resistance of films
The water absorption and water contact angle can reveal waterproofness of PU films. The R ratio of WPU, WPUS, and WPUPES is shown in Figure 6. The results show that the absorption ratio was greatly influenced by the incorporation of the polyestersiloxandiol or nano-silica. For WPUS composite, the absorption ratio of WPUS is 7.2%, which is much lower than that of WPU. This indicates that the hydrophobicity of the film is greatly enhanced, which can be ascribed to the excellent hydrophobicity and filler effect of nano-silica. For WPUPES composites, due to the existent of hydrophobic Si–O–Si, the absorption ratio of WPUPES is lower than that of WPU. However, the R value of WPUPES is a little higher than that of WPUS because of the higher micro-phase separation.
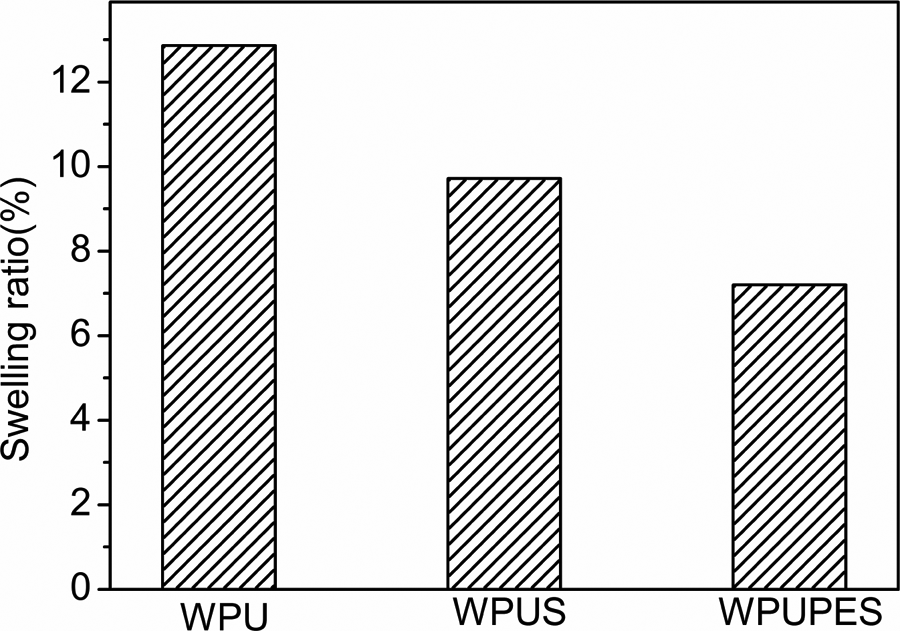
Water absorption of WPU, WPUS, and WPUPES. WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Figure 7 shows the results of the static contact angle of WPU, WPUS, and WPUPES. For the WPU, the contact angle is 79.4°, after modification with nano-silica, the contact angle increased to 101.4°, indicating a water-repellant surface. The contact angle of WPUPES (107.9°) is higher than that of WPUS (101.4°), implying that the water proofing property of WPUPES is better than that of WPUS. This phenomenon may be ascribed to lower surface free energy of silicone. Therefore, it has tendency to migrate to the surface of the film during the film formation and cross-linking. 25,26 Moreover, it seems that the direct contact of probe liquids with nano-silica has been prevented by the adsorbed layer of polymeric matrix on the surface of nano-silica. 27

Water contact angle of WPU, WPUS, and WPUPES. WPU: waterborne polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Mechanical properties
Tensile strength and elongation at break of WPU, WPUS, and WPUPES are shown in Figure 8. The results show that mechanical properties of WPU are significantly affected by the nano-silica or polyestersiloxandiol. The tensile strength of WPU is 4.62 MPa, and the elongation at break is 915% which is higher than that of WPUS and WPUPES. The tensile strength of WPUS is higher than that of neat PU and reaches 10.53 MPa, and its elongation at break decreases to 629%. This could be attributed to the WPUS chain restriction caused by the network and physical cross-link points of nano-silica particles. However, the tensile strength of WPUPES approaches that of WPU and has a lower elongation at break (595%). WPUS is a hard and strong material, while WPUPES is a flexible material with better elastic properties.
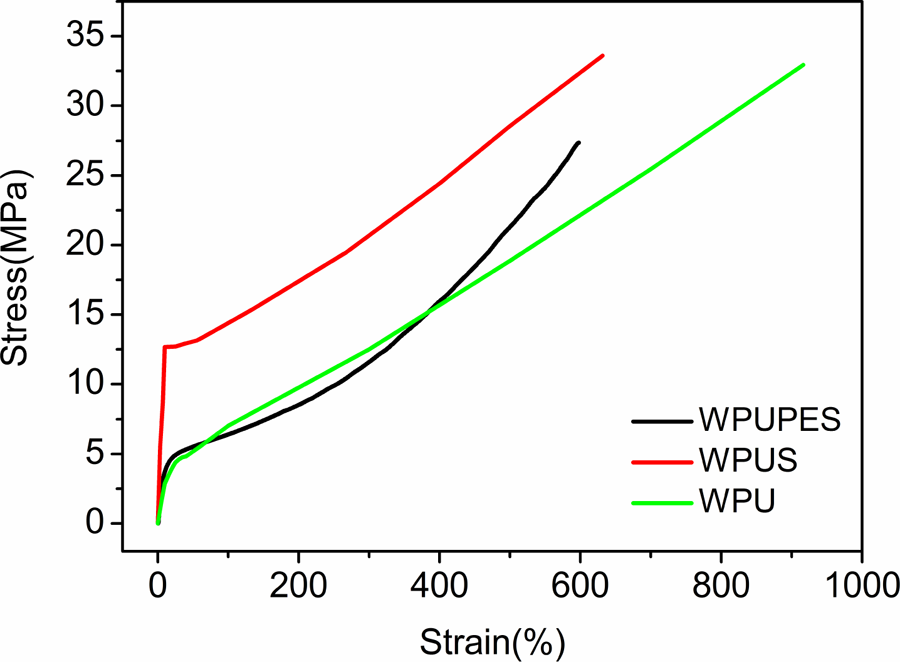
Stress–strain plots of neat PU, WPUS, and WPUPES. PU: polyurethane; WPUS: waterborne polyurethane composites; WPUPES: waterborne polyurethane composites modified by polyethersiloxanediol.
Conclusions
In this study, the WPU/nano-silica composites and WPUPES were prepared, respectively. The results of FTIR spectra confirmed that nano-silica and polyethersiloxanediol were covalently grafted into the PU macromolecular chains. AFM analyses demonstrated the aggregation of siloxane segments of WPUPES and rougher surface of film. This may contributed to its water-repellant surface. The WPUS also had a hydrophobic surface, and its tensile strength is higher than that of neat PU and reached 10.53 MPa. This could be attributed to the restriction of WPUS chain caused by the network and physical cross-link points of nano-silica particles. TGA measurement showed the enhanced thermal stability of WPUS and WPUPES; moreover, the thermal stability temperature of WPUPES was approximately 16°C higher than that of WPUS. This was mostly due to the high thermal stability of siloxane linkages and poor dispersion of nano-silica in PU matrix. The DSC analyses showed that T g of the soft segment of WPUPES shifted to lower temperature region while that of WPUS shifted to higher temperature region. WPUPES exhibit higher degrees of micro-phase separation that is consistent with the results of AFM.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
