Abstract
Intumescent fire-retardant epoxy (IFR-EP) coatings including ammonium polyphosphate (APP), pentaerythritol (PER), melamine (MEL), magnesium hydroxide (MH), epoxy resin (EP) and polyamide resin (PA) were prepared. Thermal decomposition processes of the pure EP-PA, APP, PER, MEL and MH by thermogravimetric analysis (TGA) technique indicate that the decomposition temperature ranges of EP-PA, APP, PER and MEL are properly consistent with the intumescent mechanism of flame retardants. Thermal decomposition of IFR-EP coatings shows that 15 parts per hundred parts of resin MH is more appropriate to be selected in the formulation of IFR-EP coatings. Kinetic analysis of the TGA data based on the Kissinger methods revealed that activation energy could be used as an important parameter to judge the thermal decomposition process. The conversion dependencies of the activation energy plot show the complex effect of MH content on the thermal decomposition process of IFR-EP coatings.
Introduction
Combustible materials such as polymer materials have been widely used in many fields, such as civil buildings, chemical plants and other applications. The increasing fire risk of these materials has led to the development of various classes of flame retardants. 1 –6 Intumescent fire retardant coatings represent an efficient, environmentally friendly and economic way to provide passive fire protection, which provide flame retardant properties by developing a carbonaceous shield (char) on the surface protecting the underlying material from the radiation of the incident heat flux. There are many researchers who have paid much attention on the applications of the intumescent flame retardants (IFRs). 7 –9 When exposed to the heat flux from a fire, the temperature within these materials rises, causing the thermoplastic to melt. At a critical temperature, an endothermal chemical reaction releases volatiles, which results in swelling of the intumescent layer as many times as its original thickness. Accordingly, the resultant intumescent char layer will form and act as an insulation barrier which can protect the underlying material against the heat generated by combustion process. 10 –14
However, the pyrolysis and combustion process of polymers is a complicated process involving physical and chemical interactions. 15,16 Any modification to the polymer, such as the incorporation of an additive, results in a change of thermophysical properties and will affect the pyrolysis process and combustion process thereafter. So, it is very necessary to investigate the thermal decomposition details of the IFR materials, which influence their flame retardancy to a considerable degree.
Normally, activation energy (Ea) calculated from thermogravimetry analysis (TGA) and differential thermogravimetry (DTG) data is one of the key parameters to understand the thermal decomposition behaviours of intumescent materials. 17 –21 Many kinetic models, 18,21 such as Friedman, Kissinger, Coat–Redfern, Flynn–Wall–Ozawa and Horowitz–Metzger methods, have been developed to calculate the kinetic parameters such as Eas, reaction order (n) and the Arrhenius pre-exponential factor (A), which help us to understand the thermal decomposition processes and mechanisms. From these kinetic parameters, Ea is a very important parameter, which can provide the information on the critical energy needed to initiate the reaction and can also help to understand the thermal stability of the materials. Camino et al. have reviewed the developments of intumescent and chemical–physical mechanisms of IFR systems. 22 Bourbigot et al. have done extensive studies on ammonium polyphosphate (APP)/pentaerythritol (PER)/melamine (MEL) system and completed a review about IFR thermoplastic formulations, synergy and synergistic agents. 23
Magnesium hydroxide (MH) as a fire retardant provides flame retardant properties by absorbing heat when it decomposes and releases water vapour with increase of temperature. Accordingly, char layer decomposed from MH can cover on the surface of materials and act as an insulator barrier to protect the underlying material. The effects of MH content on the fire retardancy of materials such as thermoplastic composite materials have been studied a lot. 24 –29 Huang et al. have studied four kinds of MH with different particle sizes to investigate the effect of particle size on the flame retardancy of ethylene vinyl acetate copolymer (EVA) composites. 24 Fei et al. used MH as a synergist to improve the flame retardancy of the PA6. 25 But how MH affects the thermal decomposition of IFR coatings especially IFR-EP coatings is seldom discussed. In this article, the influences of MH content on thermal decomposition of IFR-EP materials are investigated based on TGA techniques and thermal decomposition kinetics techniques.
Experimental
Materials
PER was supplied by Tianjin Guangfu Co. Ltd (China); APP was made by Qingdao Haida Chemical Co. Ltd (China); EP used as film-forming material was supplied by Baling Petrochemical Co. Ltd (China); PA was manufactured by Zhenjiang Danbao resin Co. Ltd (China); MEL was provided by Shanghai Aibi Chemistry Preparation Co. Ltd (China); MH was prepared by Jinan Taixing Flame Retardant Chemical Co. Ltd (China).
Sample preparation
The materials used to prepare IFR-EP coatings mainly include EP, PER, APP, MEL, PA and MH. At first, EP and PA were heated separately at 60°C in a water bath kettle. At the same time, APP, PER, MEL and MH were mixed and ground for about 1 h in a ball-mill to reach a particle size under 10 μm. The resulting compounds, with the formulations presented in Table 1, were mixed together in a high-speed disperse mixer for 1 h. The unit ‘phr’ in Table 1 means the parts per hundred parts of resin. Then, the mixture containing IFR was poured into aluminum foil boxes to obtain a sheet of specimen with dimensions of 100 × 100 × 2 mm3. The samples were then exposed to fresh air to dry for at least 1 week. After the coatings were entirely dry, they were removed from the aluminum foil boxes and prepared as a powder for TGA experiments.
The formulations of the IFR-EP coatings.
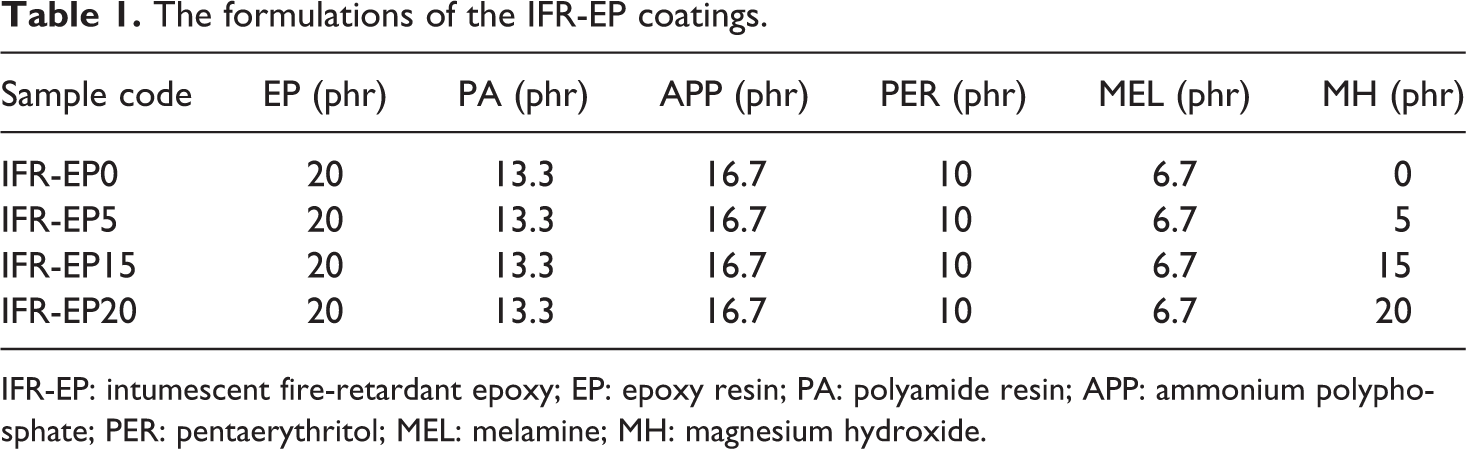
IFR-EP: intumescent fire-retardant epoxy; EP: epoxy resin; PA: polyamide resin; APP: ammonium polyphosphate; PER: pentaerythritol; MEL: melamine; MH: magnesium hydroxide.
Measurements
TGA was examined under nitrogen flow on a DT-50 (Setaram, France) instrument. About 10 mg of sample was put in an alumina crucible and heated from 25°C to 900°C. The heating rate was set as 10, 20 and 40 K min−1, respectively.
Results and discussion
Thermal decomposition of individual components
Thermal decomposition of film-forming material and additives prepared for the intumescent coating was studied using TGA under nitrogen atmosphere. Figure 1 illustrates the TGA curves of the pure EP-PA (EP was mixed with PA with mass ratio of 3:2 and cured at room temperature for at least 7 days), APP, PER, MEL and MH samples, respectively, at a rate of 10 K min−1. TGA technique is commonly used to assess the thermal stability and thermal decomposition behaviour at various temperatures by measuring the onset decomposition temperature (Tonset) at which 5.0% mass loss occurs, the temperature for the maximum degradation rate (Tmax) and char residue. It can be seen from Figure 1 that Tonset of the chemicals MH, APP, EP-PA, MEL and PER are 379, 315, 142, 303 and 244°C, respectively. The char residues (Yc, wt%) at 700°C of the materials MH, APP, EP-PA, MEL and PER are 74, 30, 9, 7 and 7 wt%, respectively.
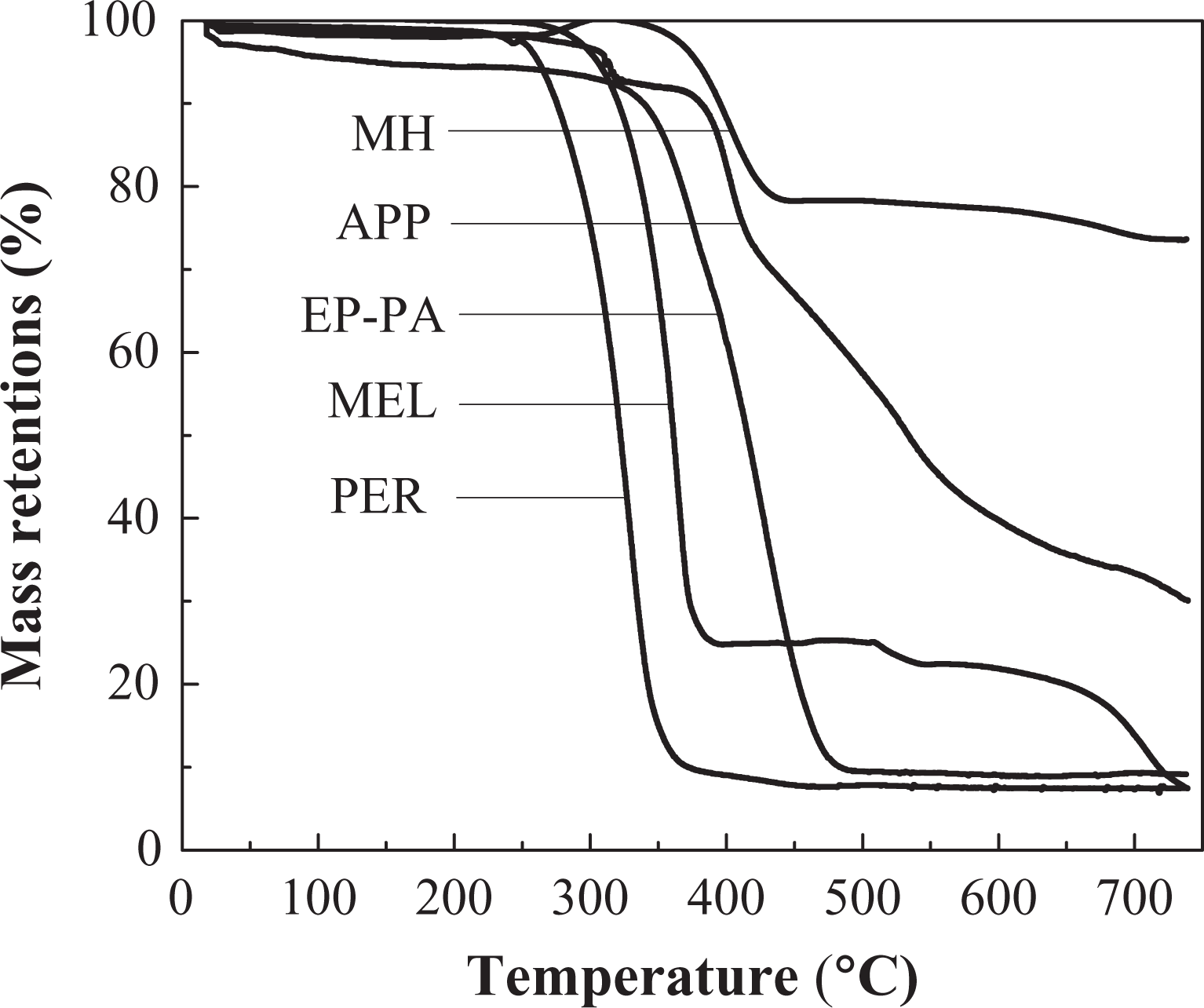
The TGA curve of single component at heating rate of 10°C min−1. TGA: thermogravimetric analysis.
The temperatures at which the 5% weight loss for APP under nitrogen atmosphere occurs are around 315°C. Commonly APP decomposes following the reactions as below
12
:
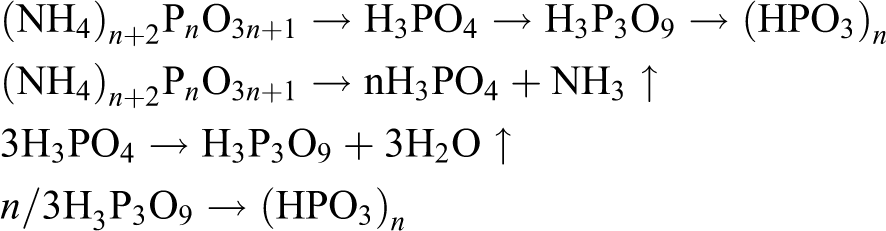
However, APP will react with PER when APP is blended with PER as the components of IFR-EP coating and heated. The resulting reaction equation can be formulated as follows (Figure 2) 12 :
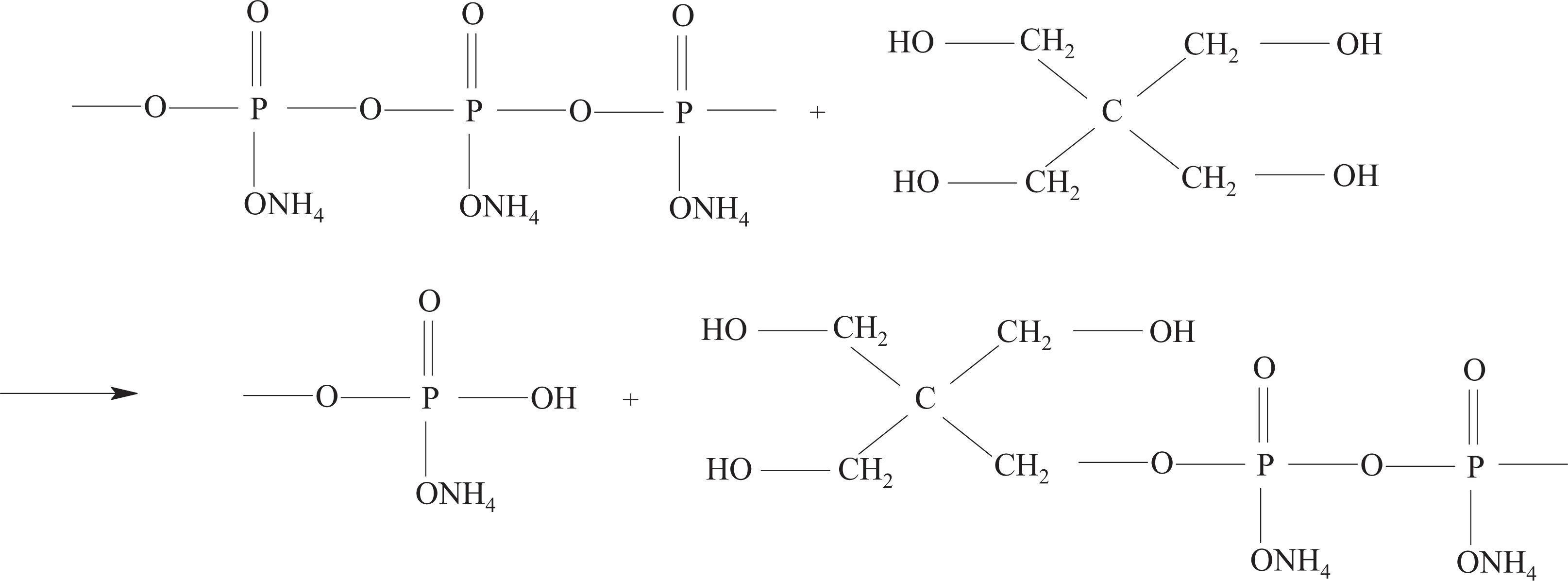
The reaction equation of APP and PER.
The main mass loss for the film-forming material EP-PA is in the range of 326–479°C, which indicates that its thermal stability is good. Thermal decomposition of the pure PER began at 244°C and was followed by a sharp mass loss up to 350°C (see Figure 1). It is known that such a mass loss is attributed to the elimination of water molecules in early stages of the degradation process and intramolecular dehydrogenization of the PER chains. The main mass loss process for MEL occurs in the stage 283–387°C, which mainly depends on the release of ammonia. The mass loss is about 75% when the temperature of MEL approaches 369°C.
From the thermal decomposition analysis of the components above, we can deduce that the decomposition temperature ranges of EP-PA, APP, PER and MEL are properly consistent with the intumescent mechanism of flame retardants.
Effects of MH content on thermal decomposition of IFR-EP coatings
Figures 3 and 4 illustrate the TGA and differential thermogravimetry (DTG) curves of the IFR-EP5, IFR-EP15 and IFR-EP20 samples, respectively, at a rate of 20°C min−1 under nitrogen atmosphere. The Yc (wt%) at 700°C of the IFR-EP15 is 32 wt%, which is higher than that of IFR-EP5 and IFR-EP20. The above results indicate that addition of 15 phr MH into IFR-EP coatings (IFR-EP15) have more char residue and less mass loss accordingly. The synergistic flame retardant effect for MH is that a large amount of heat was absorbed when it decomposes and releases water vapour, which can suppress the rapid temperature rise of IFR-EP coatings. At the same time, magnesium oxide decomposed from MH can cover on the surface of materials and act as an insulator. The decomposed water vapour can dilute the concentration of the mixed combustible gas and weaken the flame intensity if the coatings are ignited in a fire. However, the compatibility of MH and resin matrix is not very good. The mechanical and processing performance of the IFR-EP coatings will worsen if MH is added too much. From above analysis, 15 phr MH is more appropriate to be selected in the formulation of IFR-EP coatings.
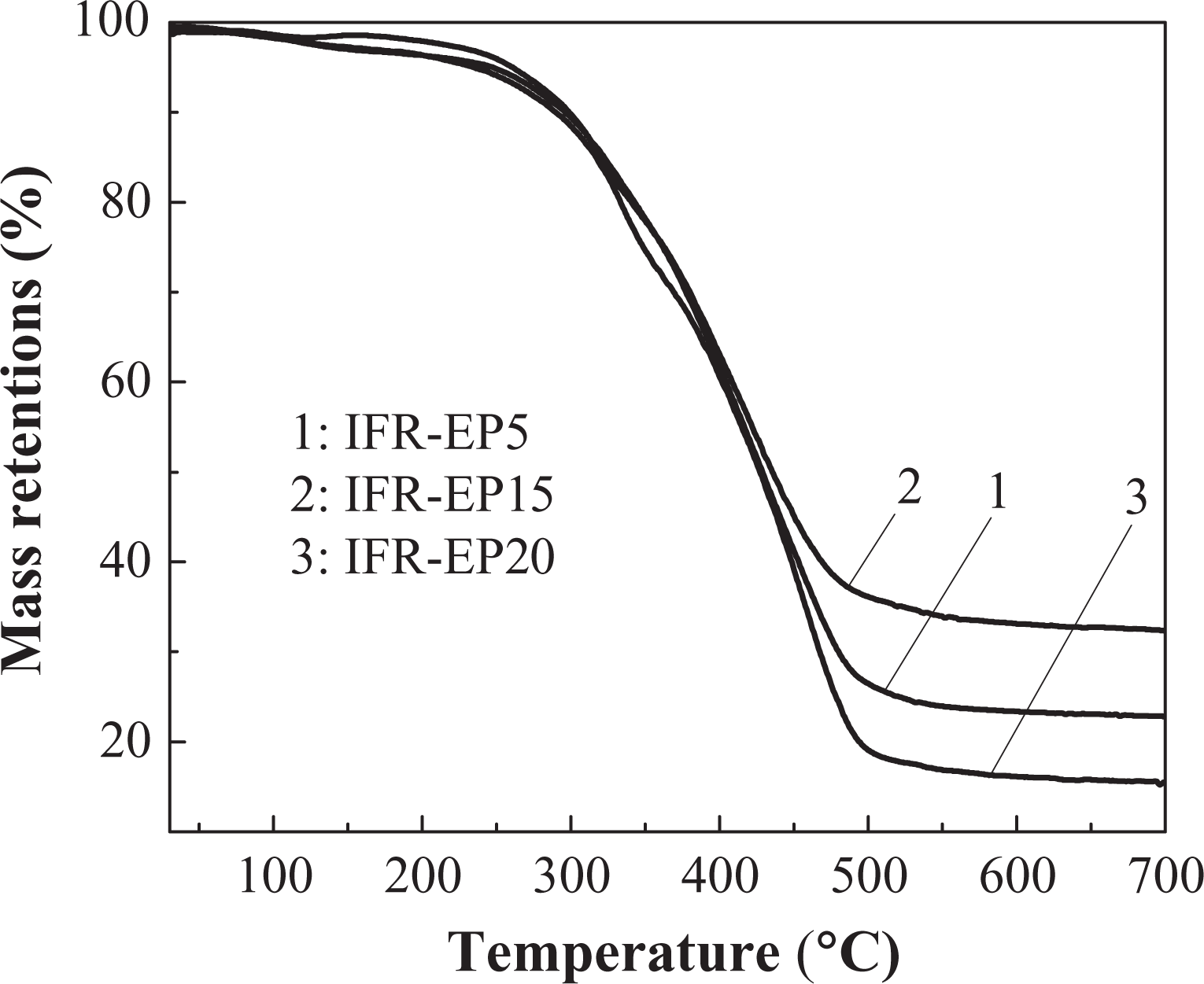
The TGA curve of different coatings at the heating rate of 20°C min−1. TGA: thermogravimetric analysis.
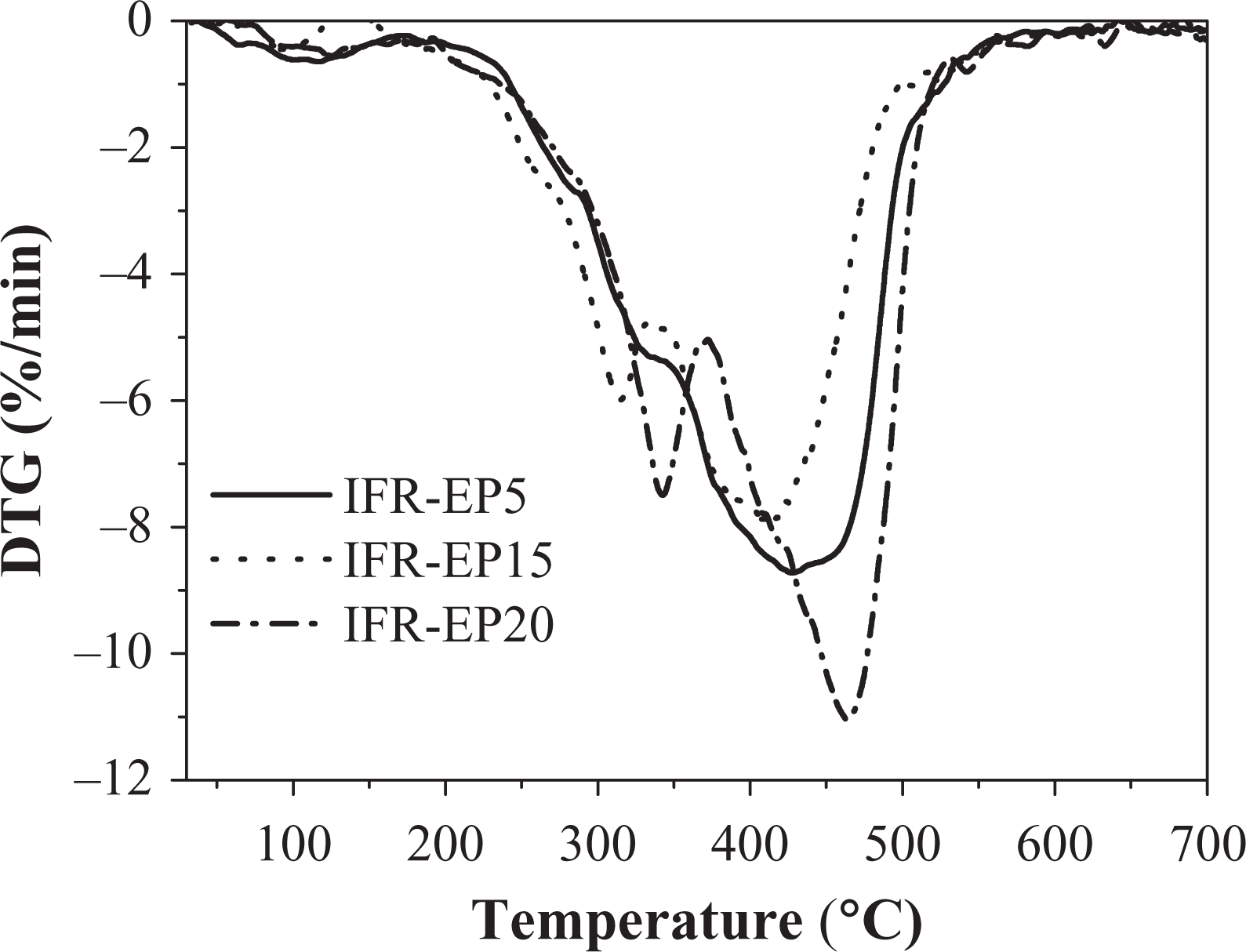
The DTG curve of different coatings at the heating rate of 20°C min−1. DTG: differential thermogravimetry.
Effects of heating rates on thermal decomposition of IFR-EP coatings
Effects of different heating rates on thermal decomposition of IFR-EP coatings under nitrogen atmosphere are presented in Figures 5 to 10. From these figures, similar thermal decomposition behaviour can be found for IFR-EP5 (Figures 5 and 6), IFR-EP15 (Figures 7 and 8) and IFR-EP20 (Figures 9 and 10) with the rise of heating rate. At slower heating rate (10°C min−1) equilibrium state is reached readily with the rise in temperature, whereas faster heating rates (20 and 40°C min−1), may be because of the slow diffusion of heat, and the equilibrium state is reached slowly and thermal decomposition temperature shifts towards higher temperature region and is observed in Figures 5 to 10. The parameters derived from the TGA and DTG curves of IFR-EP coatings are compiled in Tables 2 to 4.
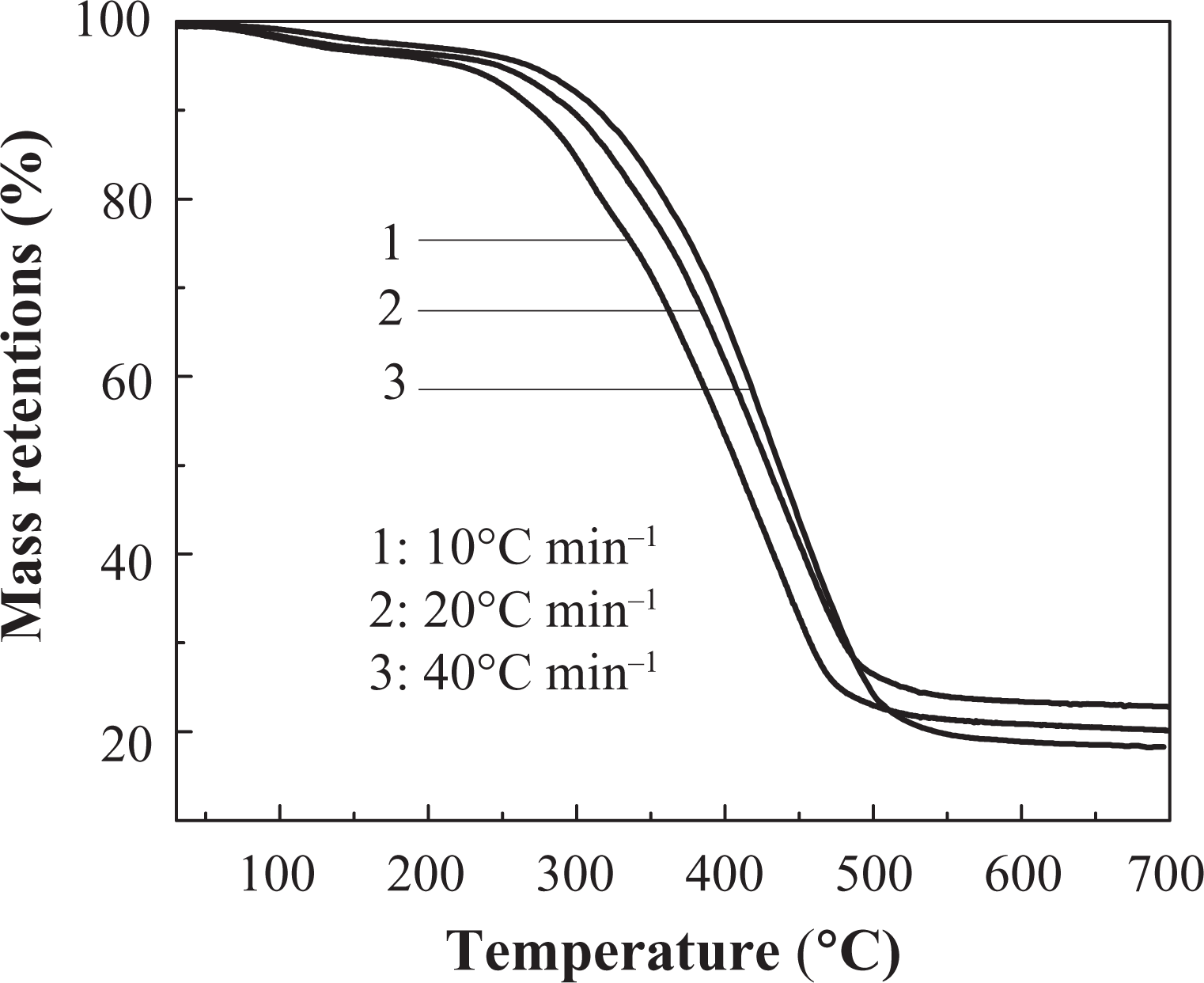
The TGA curve of IFR-EP5 coating in different heating rates. TGA: thermogravimetric analysis.
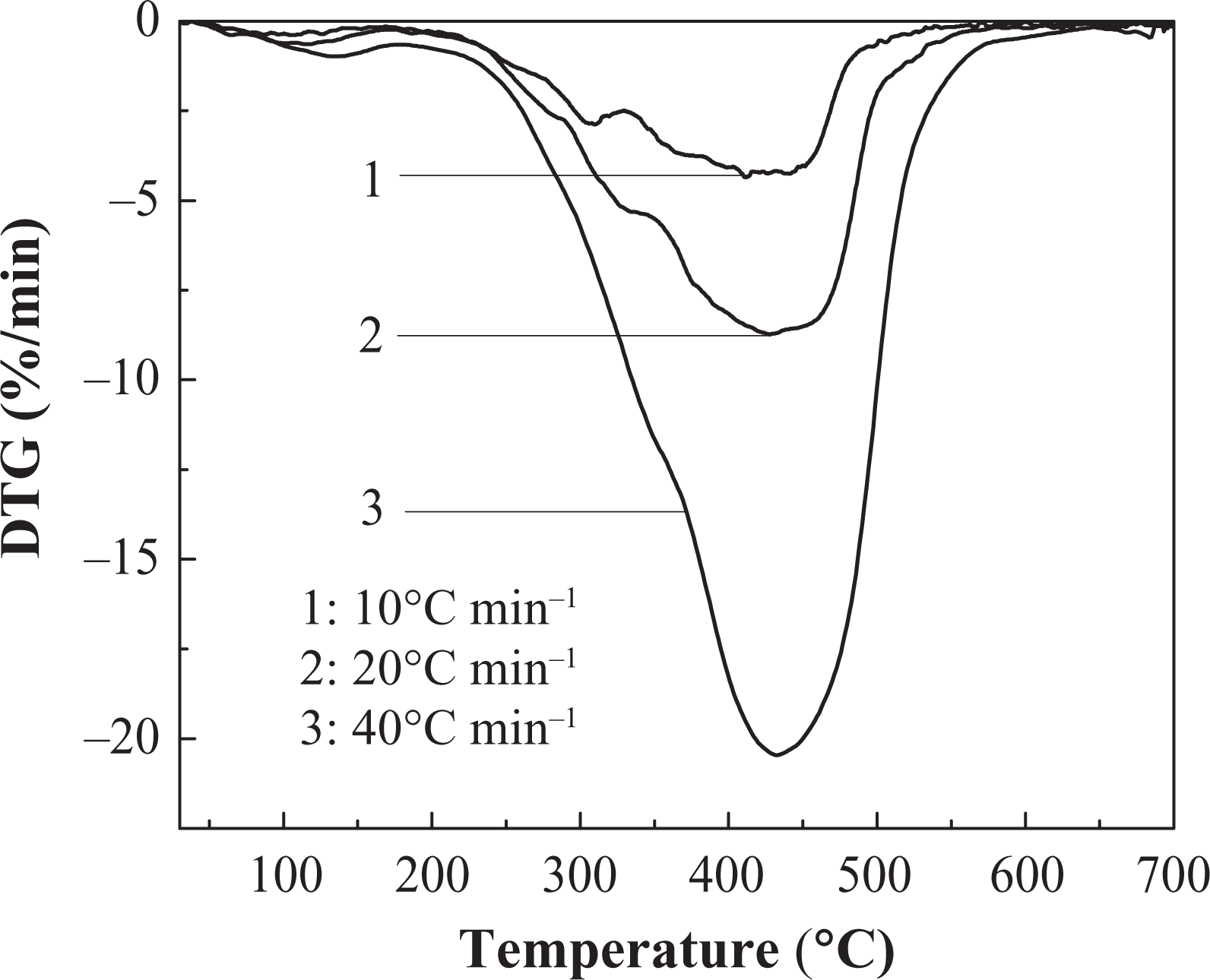
The DTG curve of IFR-EP5 coating in different heating rates. DTG: differential thermogravimetry.
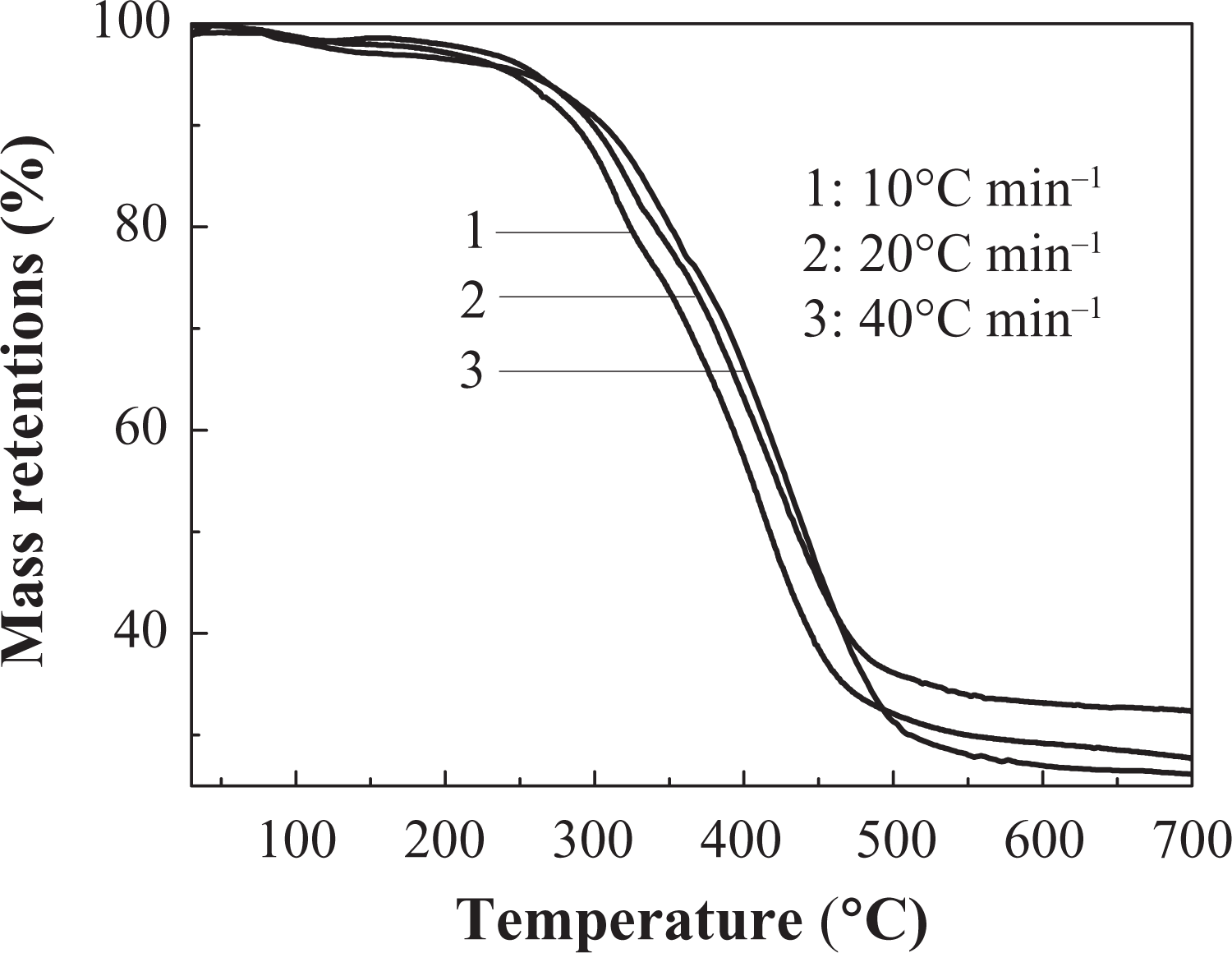
The TGA curve of IFR-EP15 coating in different heating rates. TGA: thermogravimetric analysis; IFR-EP: intumescent fire-retardant epoxy.
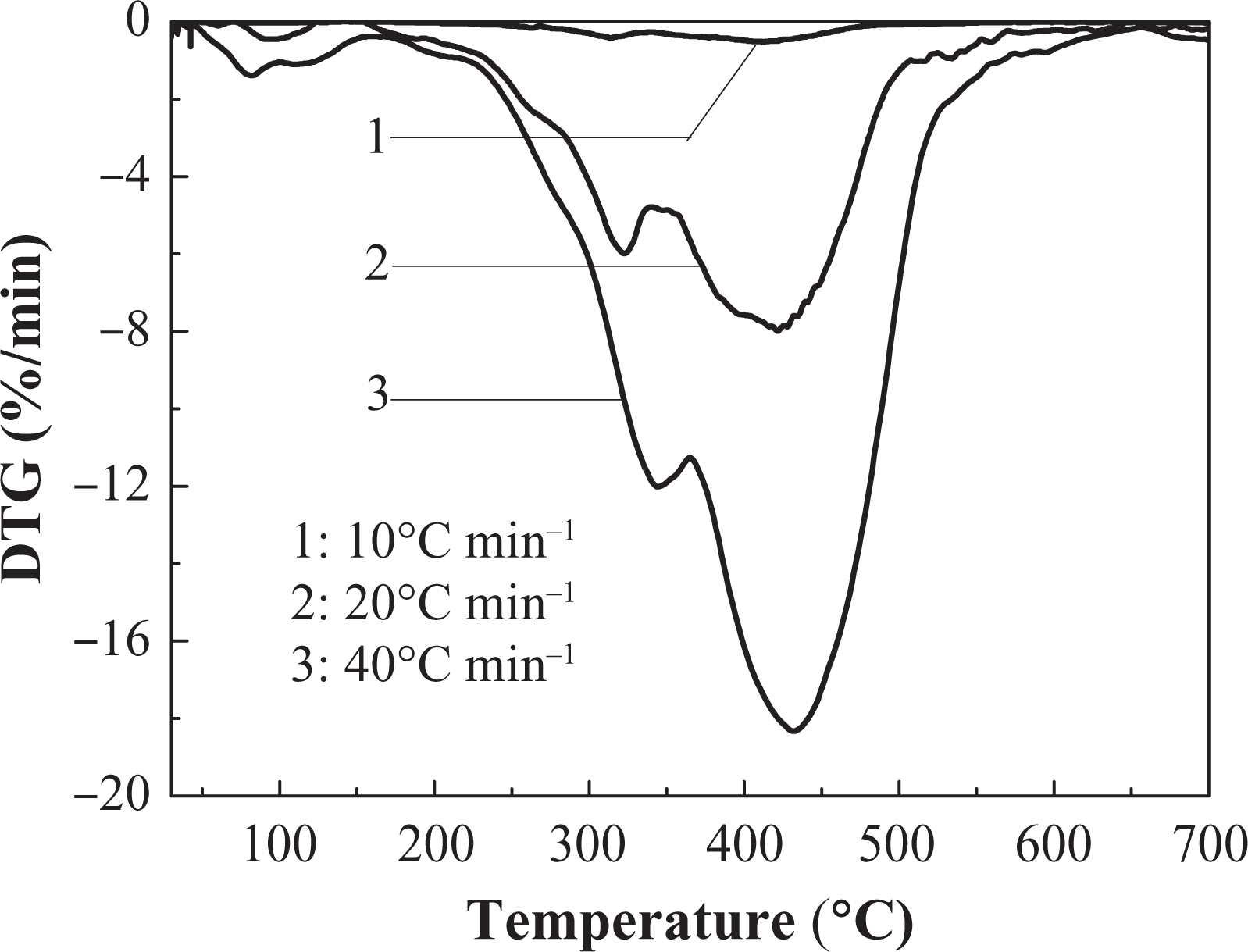
The DTG curve of IFR-EP15 coating in different heating rates. DTG: differential thermogravimetry; IFR-EP: intumescent fire-retardant epoxy.
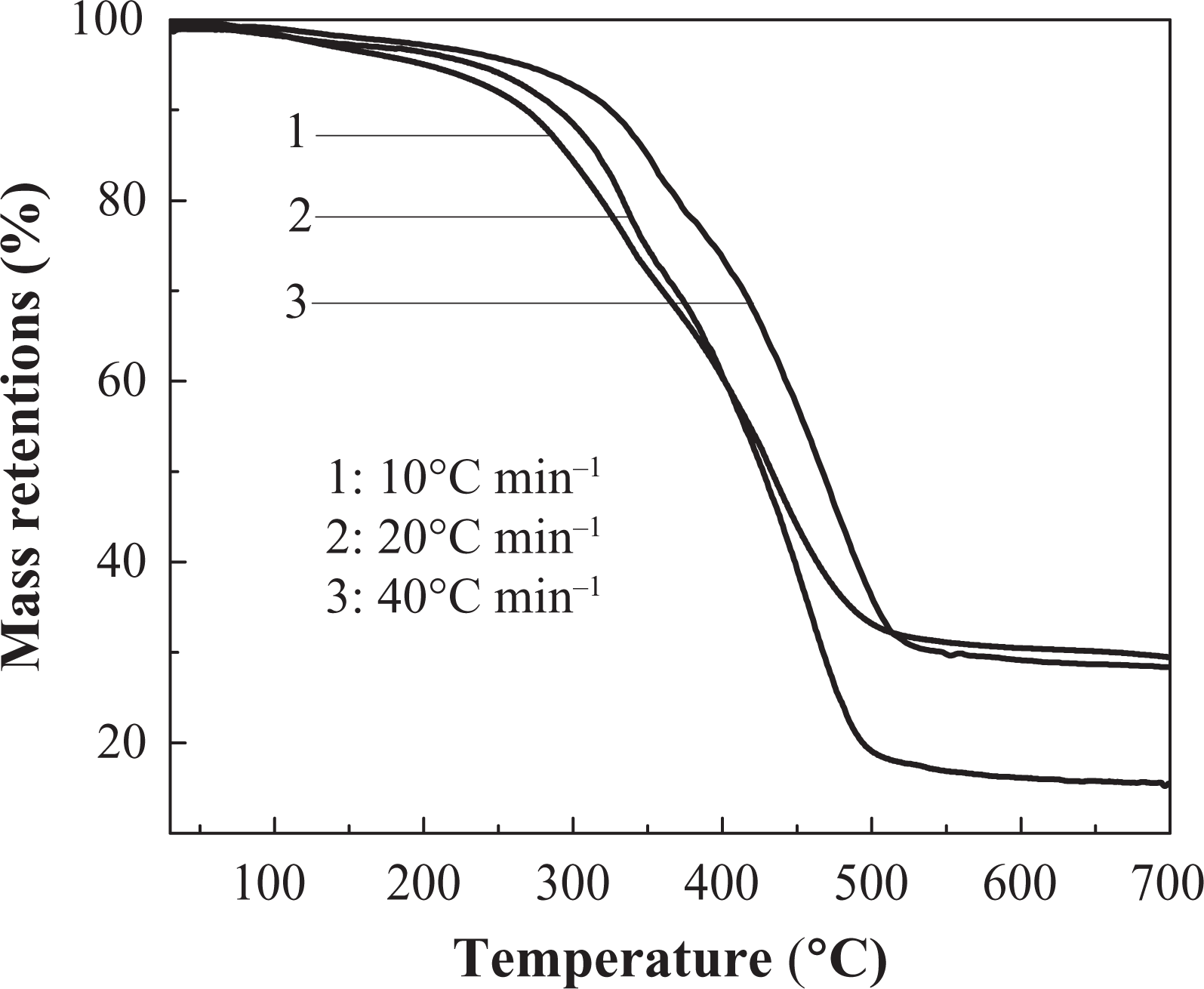
The TGA curve of IFR-EP20 coating in different heating rates. TGA: thermogravimetric analysis; IFR-EP: intumescent fire-retardant epoxy.
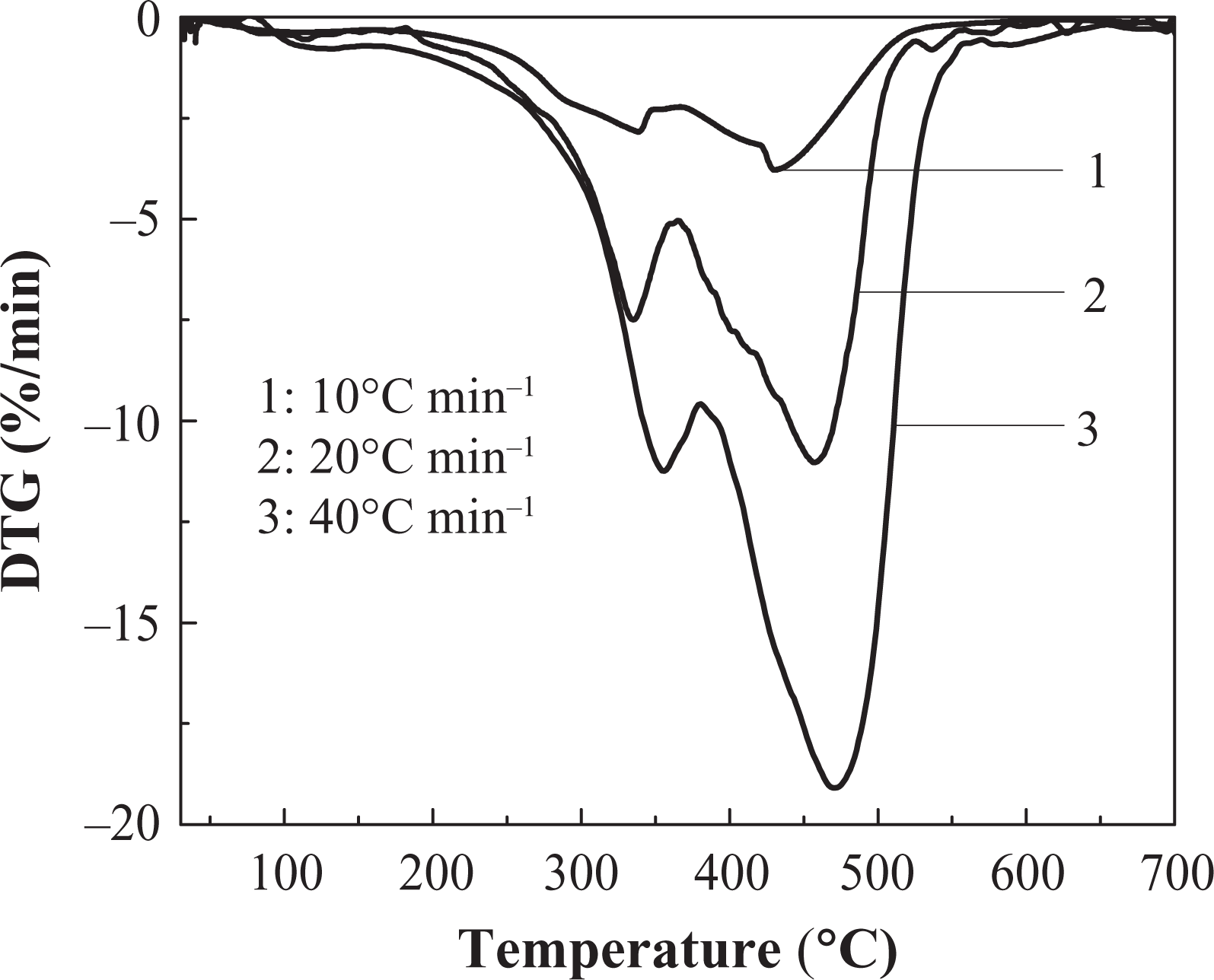
The DTG curve of IFR-EP20 coating in different heating rates. DTG: differential thermogravimetry; IFR-EP: intumescent fire-retardant epoxy.
Characteristic thermal decomposition parameters of IFR-EP5 coating.
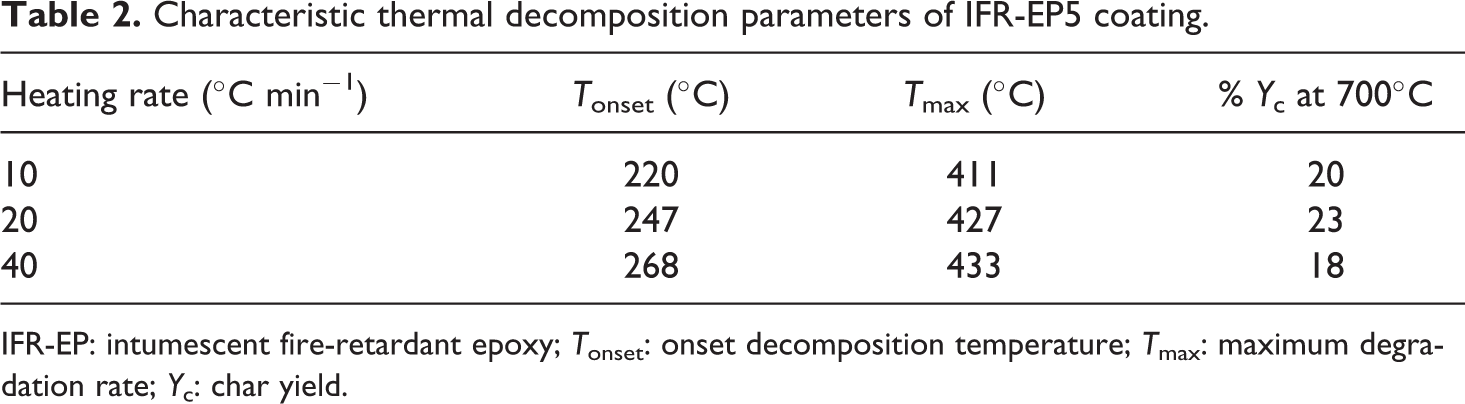
IFR-EP: intumescent fire-retardant epoxy; Tonset: onset decomposition temperature; Tmax: maximum degradation rate; Yc: char yield.
Characteristic thermal decomposition parameters of IFR-EP15 coating.
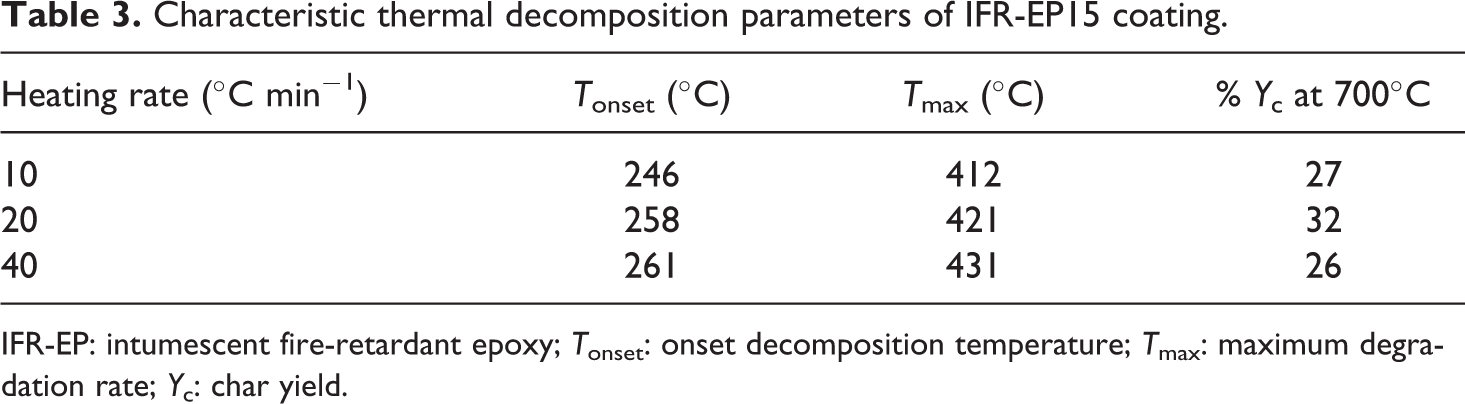
IFR-EP: intumescent fire-retardant epoxy; Tonset: onset decomposition temperature; Tmax: maximum degradation rate; Yc: char yield.
Characteristic thermal decomposition parameters of IFR-EP20 coating.

IFR-EP: intumescent fire-retardant epoxy; Tonset: onset decomposition temperature; Tmax: maximum degradation rate; Yc: char yield.
Thermal decomposition kinetics of IFR-EP coatings
TGA has been used widely to obtain the kinetic parameters of thermal decomposition processes, such as Ea, n, and the A, which can be calculated using various kinetic models such as Friedman, Kissinger, Coats–Redfern, Flynn–Wall–Ozawa and Horowitz–Metzger methods. The thermal decomposition kinetics of IFR-EP coatings based on the Kissinger technique is presented here.
Kissinger method is proposed by the following integrated equation for calculating the kinetic parameters:
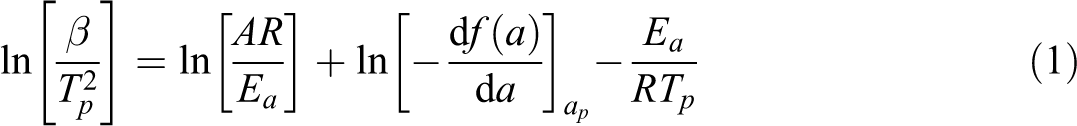
where β is the heating rate (°C min−1), Ea is the conversion dependent activation energy, R is the universal gas constant (8.314 J mol−1 K−1), T is the temperature corresponding to particular conversion percentage, A is the pre-exponential factor or Arrhenius frequency factor (s−1). The linear plot of ln(β/T2 max) against 1000/Tmax (K−1) yields slope (−Ea/(1000R)). From the values of the slope, Ea can be calculated.
The linear plots of TGA data for IFR-EP5, IFR-EP15 and IFR-EP20 coatings are shown in Figures 11, 12 and 13, respectively. Kinetics parameters calculated from the slope values are listed in Table 5.
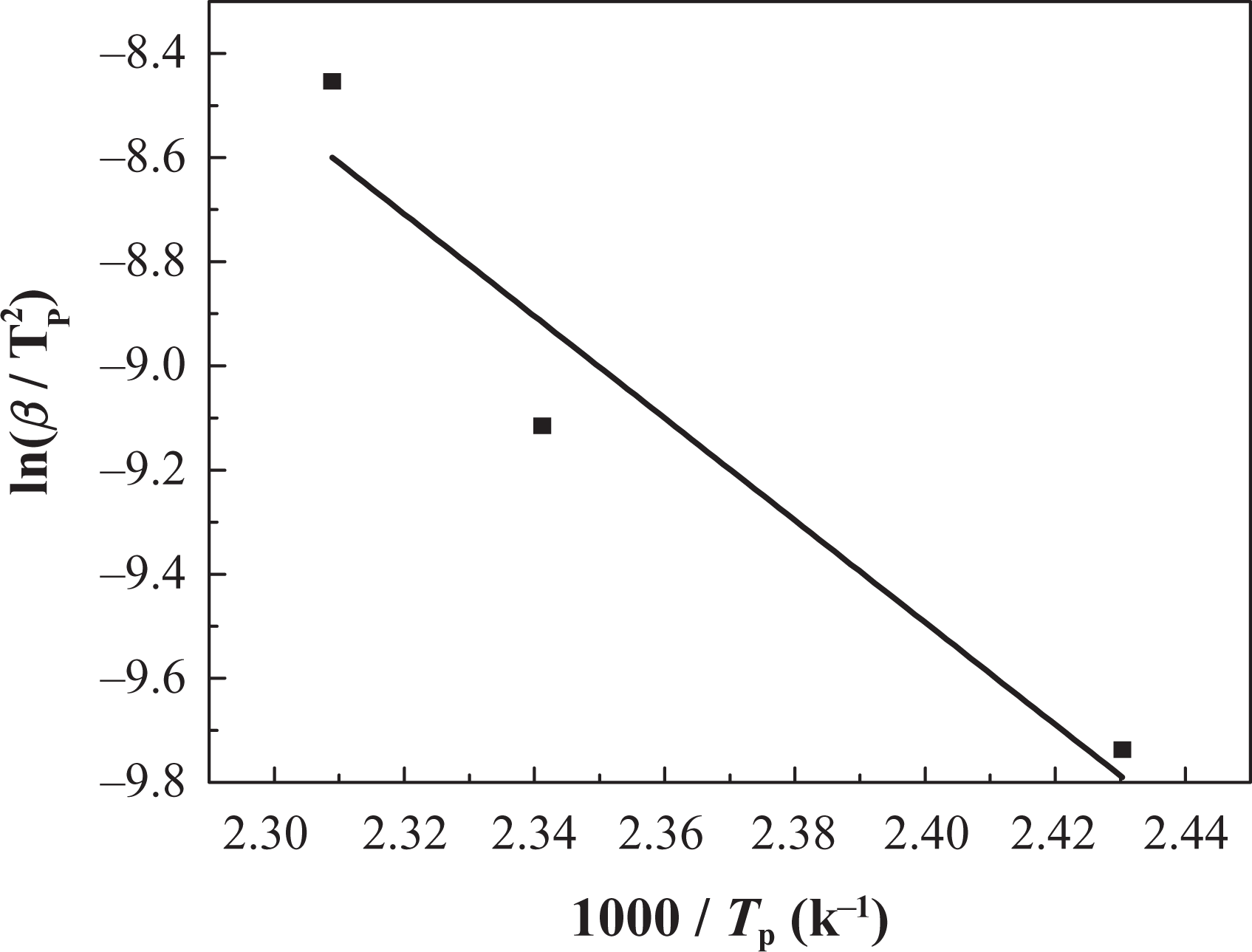
Kissinger method applied to coating IFR-EP5 at different heating rates. IFR-EP: intumescent fire-retardant epoxy.
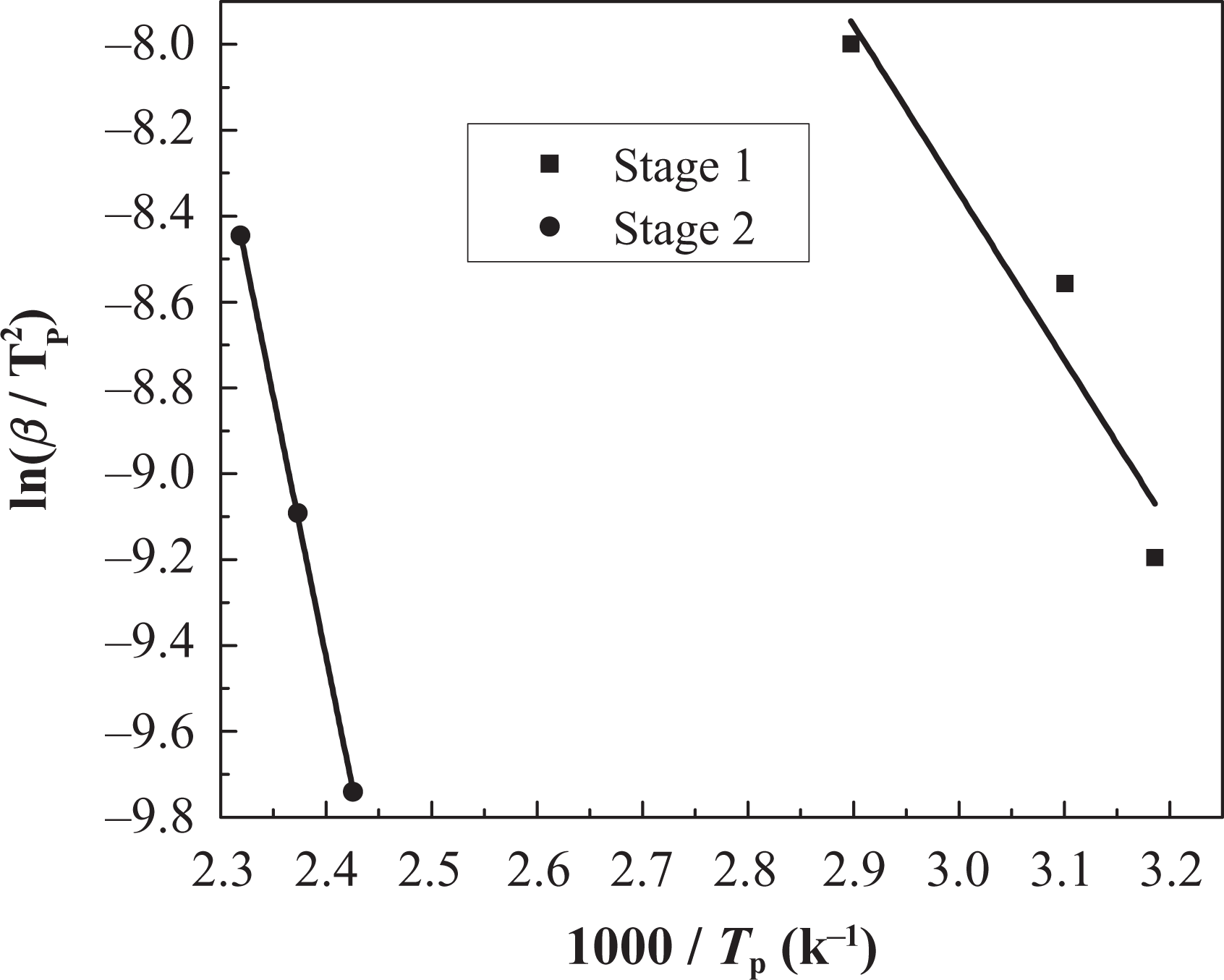
Kissinger method applied to coating IFR-EP15 at different heating rates. IFR-EP: intumescent fire-retardant epoxy.
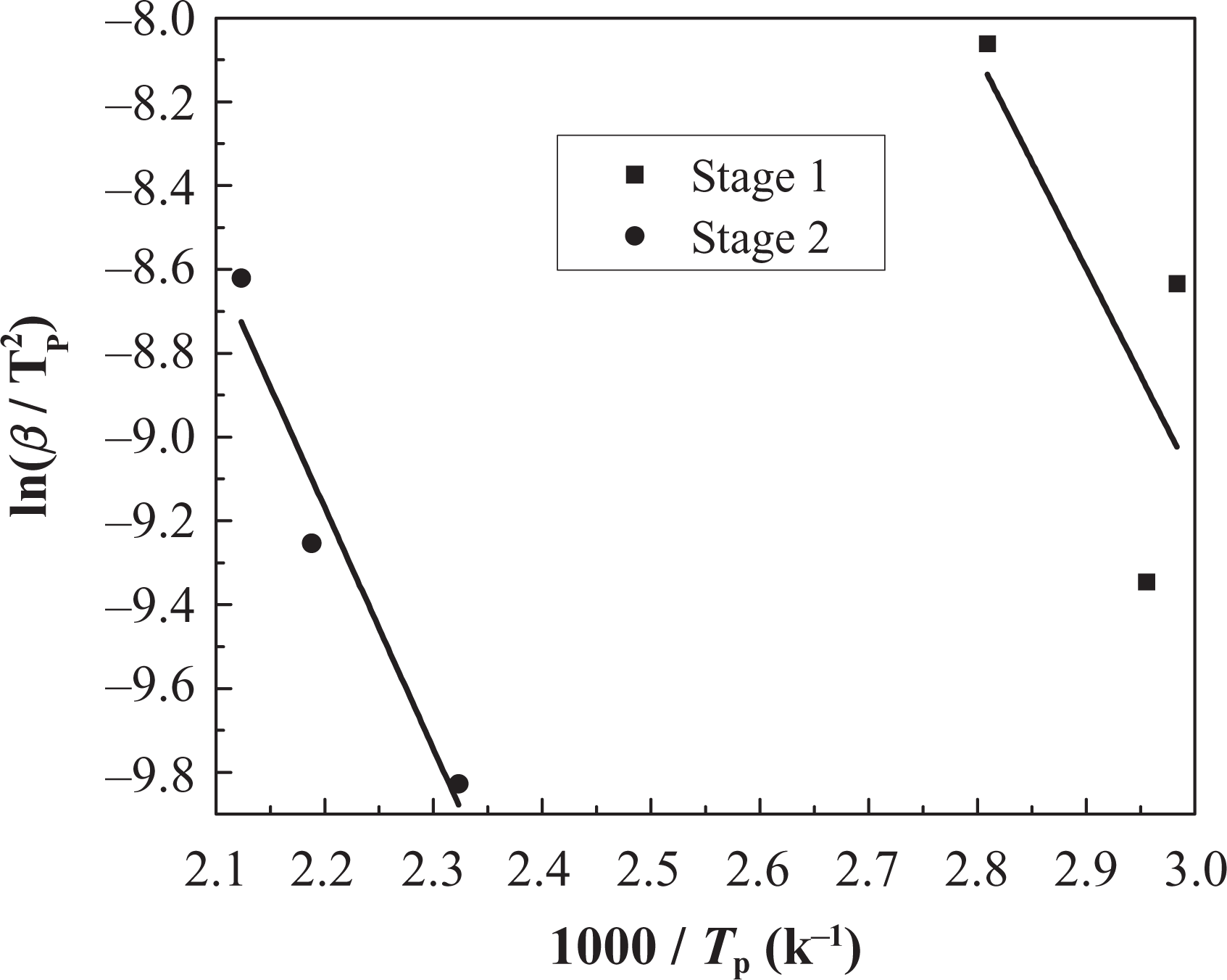
Kissinger method applied to coating IFR-EP20 at different heating rates. IFR-EP: intumescent fire-retardant epoxy.
Kinetic parameters for four coatings at different heating rates.
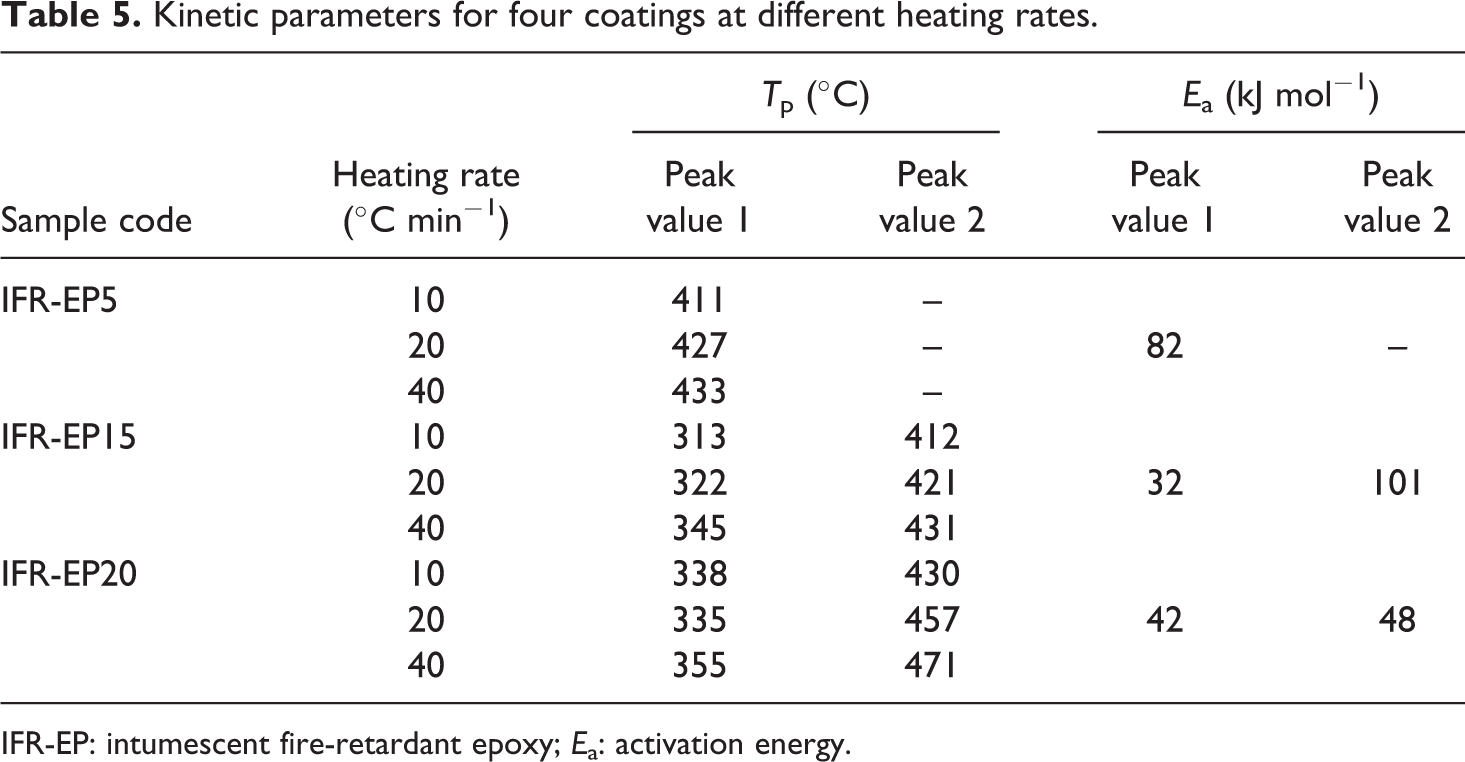
IFR-EP: intumescent fire-retardant epoxy; Ea: activation energy.
As shown in Figures 11 and 13 and Table 5, there is only one mass loss peak in the DTG of IFR-EP5 coating (5 phr MH). Accordingly, only one Ea (82 kJ mol−1) appears in Table 5. However, the number of mass loss peak in the DTG of IFR-EP15 and IFR-EP20 coatings is different compared with that of IFR-EP5 coating. They both have two mass loss peak values, respectively. Thus, two Ea values can be calculated for IFR-EP15 or IFR-EP20. For the coating IFR-EP15, the first Ea is 32 kJ mol−1 corresponding to the first mass loss peak. Accordingly the second Ea is 101 kJ mol−1. For the coating IFR-EP20, the similar Ea can be calculated, as shown in Table 5. Although the temperature corresponding to the second mass loss peak value for the IFR-EP20 is higher than that of IFR-EP15, the corresponding Ea is lower for IFR-EP20 and is lower than that of IFR-EP15. The results illustrate that incorporation of MH into IFR-EP coating too much will worsen the mechanical and processing performance and weaken the thermal stability of IFR-EP coatings, which validate above analysis results from Figures 3 and 4.
Conclusions
TGA and DTG traces of the pure EP-PA, APP, PER, MEL and MH prepared for the intumescent coating reveal that the decomposition temperature ranges of EP-PA, APP, PER and MEL are properly consistent with the intumescent mechanism of flame retardants. Thermal decomposition of IFR-EP coatings shows that 15 phr MH is more appropriate to be selected in the formulation of IFR-EP coatings. Thermal decomposition temperature shifts towards higher temperature region based on the analysis of effects of heating rates on thermal decomposition of IFR-EP coatings. Using the TGA curves obtained from the multiple heating measurements, the Ea for the decomposition of IFR-EP coatings was computed using Kissinger method. The conversion dependencies of the Ea plot bring out the complex effect of MH content on the thermal degradation behaviour of IFR-EP coatings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support of China NSFC Funds (no. 51006054).
