Abstract
A series of polyimides (PIs) was synthesized by reacting a nitrile-containing aromatic diamine, 3,5-diaminobenzonitrile, with various dianhydrides to yield poly(amic acid)s that were then cyclized to yield PIs by a thermal imidization method. The samples were characterized using thermal analysis, tensile testing, optical transparency analysis, and gas permeability analysis. The cast films exhibited good thermal stability with glass transition temperatures of 204–243°C and none exhibited significant decomposition at temperatures below 480°C. These PI films showed coefficient of thermal expansion values in the range 41.68–106.16 ppm/°C. The oxygen transmission rate values of the films containing various dianhydride monomers were in the range 0.93–27.40 cc m−2 day−1. The PI films containing various dianhydride monomers showed tensile strengths of 47–128 MPa, initial moduli of 2.16–3.23, and elongations at break of 1–7%. Moreover, the PI films possessed a transmittance of 58–82% at 500 nm and had a yellowish color with a yellow index of 12.17–59.61.
Introduction
Aromatic polyimides (PIs) have been introduced in most useful and important types of polymers because of their excellent thermomechanical properties, high dimensional stability, good chemical resistance, and good electrical properties. 1 –3 Therefore, they are considered as candidates for microelectronics applications such as the substrates of flexible printed circuit boards, interlayer dielectrics, and insulating layers in multilayer structures and multichip modules. 4 –6 In general, however, PIs are difficult to process because of their non-melting behavior and poor solubility in conventional organic solvents. 7,8 Therefore, PIs can only be used in certain applications.
Much research effort has been devoted to the development of high-performance PI materials with excellent thermomechanical properties and optical transparency. For example, PIs containing trifluoromethyl groups have been synthesized that show a high modulus, low thermal expansion coefficient, and good solubility in conventional organic solvents.
9
–12
The incorporation of heterocyclic units into PI chains increases the glass transition temperature (
As part of our continuing efforts to gain colorless PIs with high thermal stability and mechanical property for advanced polymeric materials, we prepared a series of new PI films through the thermal imidization of an aromatic precursor polymer obtained by the reaction of 3,5-diaminobenzonitrile (DABN) with various dianhydride monomers. In an attempt to understand the structure–property relationships in the PIs, the present article describes the properties of a series of systematically varied PIs derived from the diamine DABN and various structurally different aromatic dianhydrides, such as 4,4′-(hexafluoro-isopropylidene)diphthalic anhydride (6FDA), 4,4′-(4,4′-isopropylidene-diphenoxy)-bis(phthalic anhydride) (BPADA), 3,3′,4,4′-benzophenone-tetracarboxylicdianhydride (BPDA), and 3,3′,4,4′-diphenylsulfone tetracarboxylic dianhydride (DSDA). Moreover, this article discusses the variations in the thermomechanical properties, optical transparency, and oxygen (O2) permeability of the PI films due to different dianhydride monomers.
Experimental
Materials
Dianhydride monomers were purchased from TCI (Tokyo, Japan) and Aldrich Chemical Co. (Yongin, Korea) and used as received.
Preparation of DABN diamine
DABN was synthesized via a palladium on carbon (Pd/C)-catalyzed reduction of 3,5-dinitrobenzonitrile at 90% yield. Further, 20 g (0.1 mol) of dinitrobenzonitrile was dissolved in 1 L of ethylacetate, and the solution was subjected to hydrogenation using 2 g of 10 wt% Pd/C catalyst and hydrogen gas (32 psi). After carrying out the reaction for 24 h, the mixture was filtered to remove the catalyst. The resulting yellow solution was poured into a mixture of diethylether/water (v/v = 1/1.5) to remove unreacted monomers. The organic layer was isolated and rotary evaporated to obtain a diamine monomer. The crude product was recrystallized from ethanol (yield: 17.8 g (90%); melting point: 200°C)).
Fourier transform infrared spectroscopy (FTIR; potassium bromide): –NH2 at 3433–3203 cm−1, –CN at 2223 cm−1, and aromatic C=C at 1625 and 1590 cm−1; proton nuclear magnetic resonance (
1
H NMR; deuterated dimethyl sulfoxide (DMSO)-
Preparation of PI films
Four different PIs (samples I–IV) were prepared from the synthesized DABN and four different dianhydrides, namely, 6FDA, BPADA, BPDA, and DSDA. The chemical structures relevant to the synthetic route are shown in Figure 1.
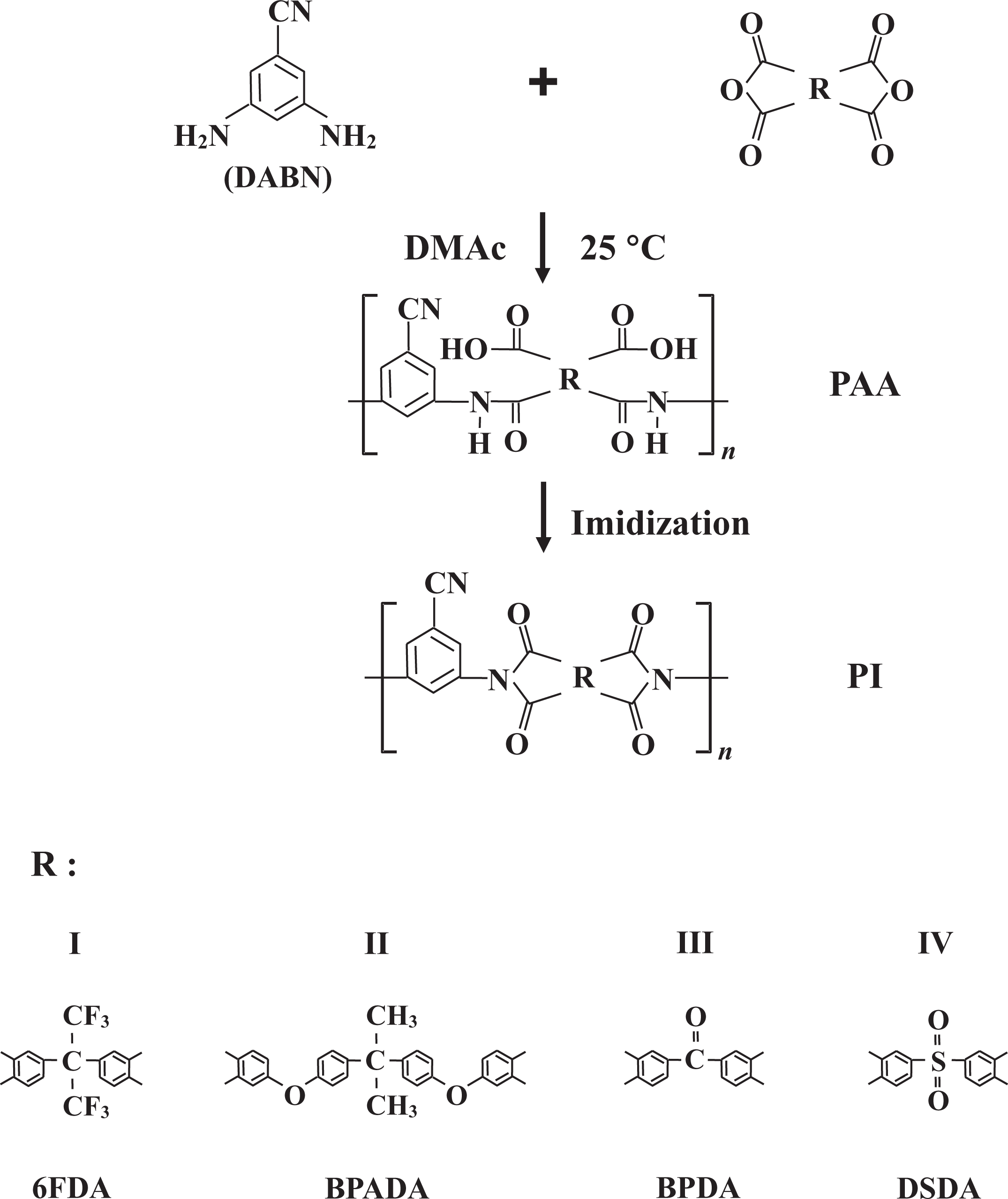
Synthetic routes of polyimide films containing DABN. DABN: 3,5-diaminobenzonitrile.
The mole ratio of diamine/dianhydride was maintained at 1:1. Because the synthetic procedures used to prepare the PI films were the same for all the samples, we describe, in this section, only the preparation procedure of 6FDA/DABN (sample I) as a representative example. Poly(amic acid) (PAA) was synthesized from DABN and 6FDA in DMAc at a low temperature. DABN (2.3 g; 1.72 × 10−2 mol) and DMAc (35 mL) were put in a 200-mL three-necked flask, and the mixture was stirred at 0°C for 30 min under nitrogen (N2) atmosphere. A solution of 6FDA (7.70 g; 1.72 × 10−2 mol) in DMAc (35 mL) was then added to this mixture, which was stirred vigorously at 0°C for 1 h and then at room temperature for 14 h, yielding a 15 wt% solution of PAA in DMAc.
The resulting solution was cast onto glass plates, and the solvent was then evaporated at 50°C for 1 h. These films were dried in a vacuum oven at 80°C for 1 h. The PAA film was further imidized on the glass plate by sequential heating at 110, 140, 170, 200, 230, and 250°C for 30 min at each temperature. Table 1 lists the heat treatment conditions employed for the preparation of the PI films. The PI films obtained in this study were insoluble in organic solvents such as
Heat treatment conditions for PAA and PI films based on DABN.
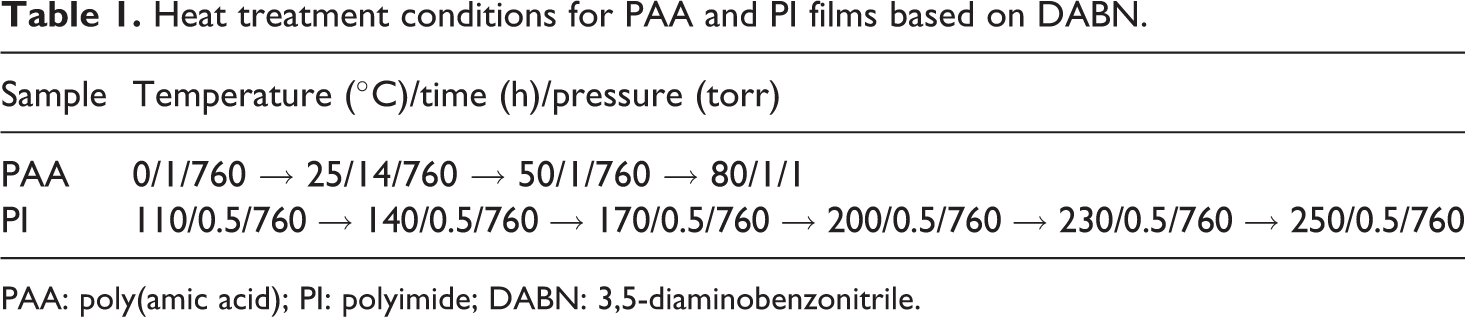
PAA: poly(amic acid); PI: polyimide; DABN: 3,5-diaminobenzonitrile.
No fixing tools were used to orientate the glass plate during the heat treatment because orientation is known to influence some characteristics of the films, such as their tensile properties and morphology.
Characterization
FTIR spectra of the samples were recorded by a Nicolet 360 IR spectrometer (Madison, Wisconsin, USA). The 1 H-NMR spectra were obtained with a Bruker DPX 400 MHz NMR spectrometer (Billerica, Massachusetts, USA) and were referenced to tetramethylsilane. The 13 C cross-polarization magic angle spinning (CP/MAS) NMR was performed at the Larmor frequencies of 100.61 MHz. The sample was placed in the 4 mm CP/MAS probe as powders, and the magic angle spinning rate was set at 7 kHz for 13 C CP/MAS to minimize the spinning sideband overlap.
The thermomechanical properties of the samples were measured with an NETZSCH differential scanning calorimeter (DSC 200F3; Germany) and a TA Instruments thermogravimetric analyzer (Auto TGA 1000; New Castle, Delaware, USA) at a heating rate of 20°C min−1 under N2 flow. The coefficients of thermal expansion (CTEs) of the samples were measured with a macroexpansion probe (TMA-2940), which was used to apply an expansion force of 0.1 N to the films at a heating rate of 5°C min−1 in the temperature range 50–150°C.
The tensile properties of the solution cast films were determined using a universal testing machine (model 5564; Instron, Norwood, Massachusetts, USA) at a crosshead speed of 2 mm min−1. The specimens were prepared by cutting strips with dimensions of 5 × 70 mm2. The reported data represent the mean of at least 10 individual values. The experimental uncertainties in the values of tensile strength and modulus were ±1 MPa and ±0.05 GPa, respectively.
The color intensity of the polymer films was evaluated using a Minolta spectrophotometer (model CM-3500d, Tokyo, Japan). Measurements were taken using films having thickness in the range 76–100 µm at an observational angle of 10° and using a CIE-D illuminant. A CIELAB color difference equation was used. The ultraviolet–visible (UV-Vis) spectra of the polymer films were recorded by a Shimadzu UV-1601 UV-Vis spectrophotometer (Japan).
The O2 permeability of the films was measured according to ASTM E-96 standard with a Mocon DL 100 (Brooklyn Park, Minneapolis, USA). The values of the O2TR were obtained at a temperature of 23°C, relative humidity of 0%, and pressure of 1 atm. The Chemdraw Office program was used to produce the three-dimensional (3D) chemical structures.
Results and discussion
FTIR spectroscopy
The formation of PAA and completion of imide formation from amic acid were confirmed by examining the FTIR spectra; the spectra of PAA and PIs are shown in Figure 2. Only the 6FDA/DABN system is described here as a representative example. It was observed that PAA exhibited a broad absorption band around 2400–3400 cm−1 because of the acid (COOH) group of PAA. The C=O stretching peaks at 1718 and 1680 cm−1 were due to the acid and amide groups of PAA, respectively. These peaks shifted to higher frequencies in the imides, specifically to approximately 1787 cm−1 (C=O, in phase) and 1729 cm−1 (C=O, out of phase), respectively. In addition, the presence of a feature at 1352 cm−1 corresponding to the C–N–C stretching confirmed the formation of the imides. 17
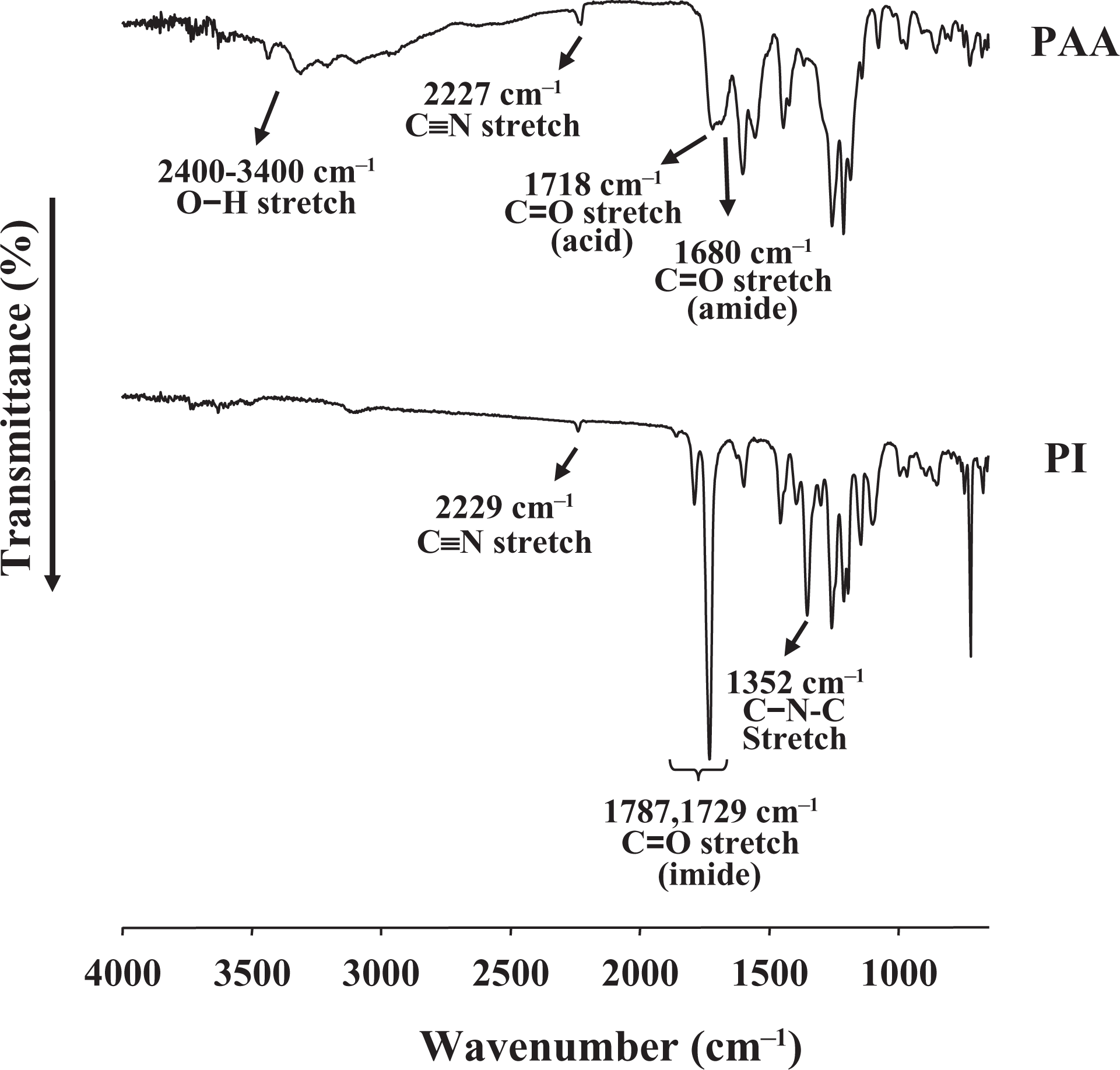
FTIR spectra of PAA and PIs. FTIR: Fourier transform infrared; PAA: poly(amic acid); PI: polyimide.
Thermal behavior
The DSC and TGA curves of PIs with various dianhydrides are shown in Figures 3 and 4. The thermal behavior data for PIs with various dianhydrides are listed in Table 2. The
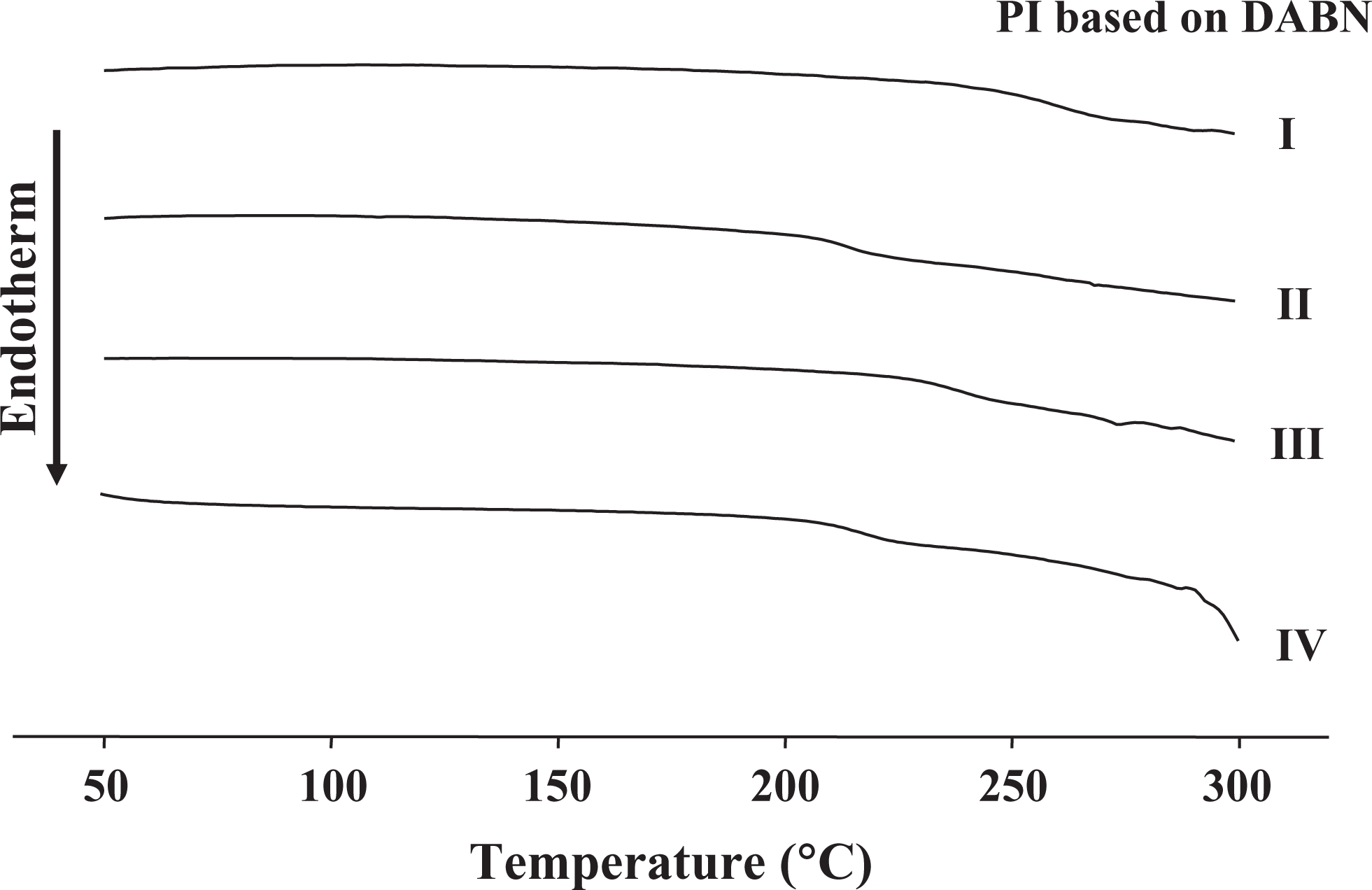
DSC thermograms of PI films based on DABN. DSC: differential scanning calorimetric; PI: polyimide; DABN: 3,5-diaminobenzonitrile.
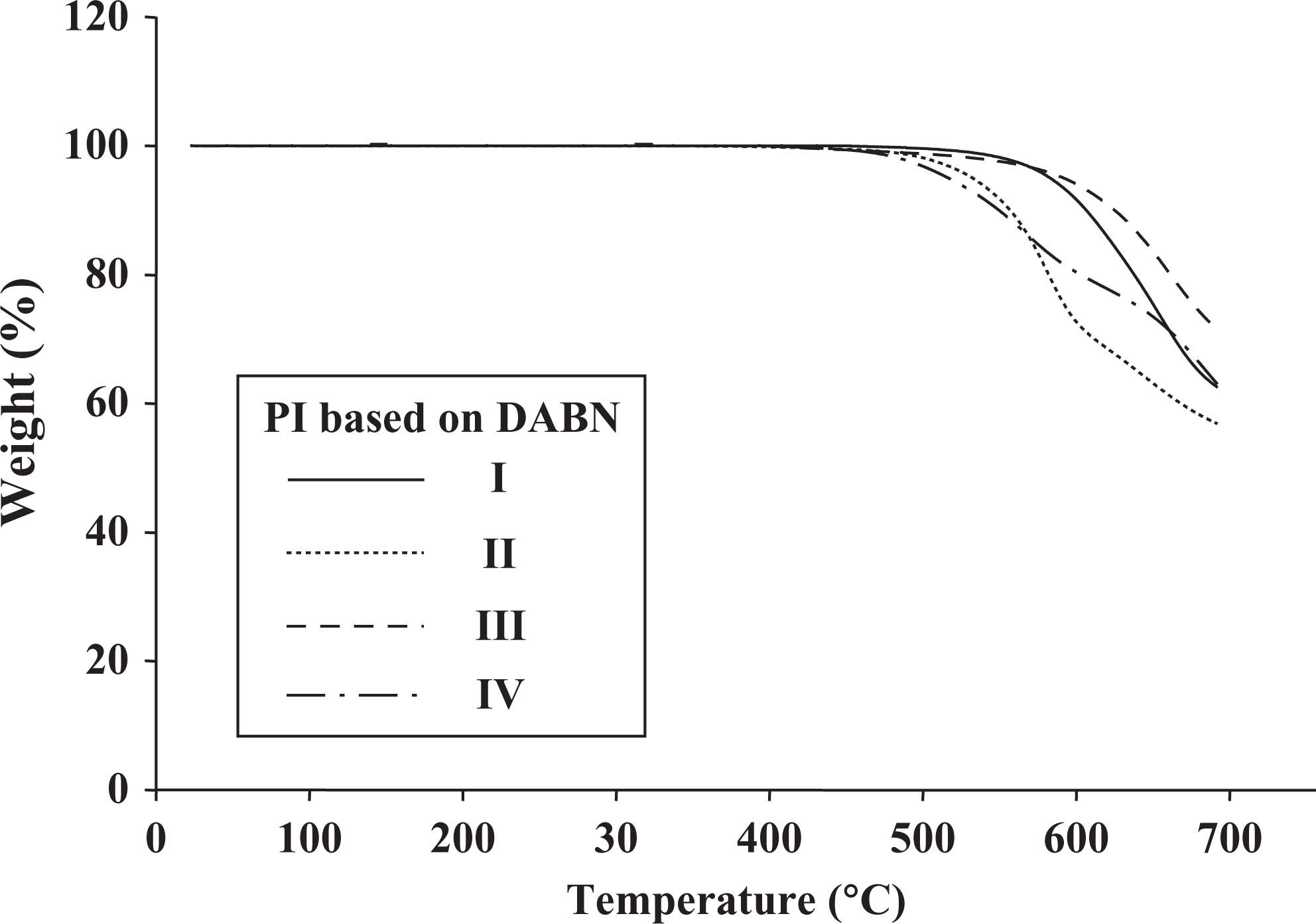
TGA thermograms of PI films based on DABN. TGA: thermogravimetric analysis; PI: polyimide; DABN: 3,5-diaminobenzonitrile.
General properties of PI films based on DABN.
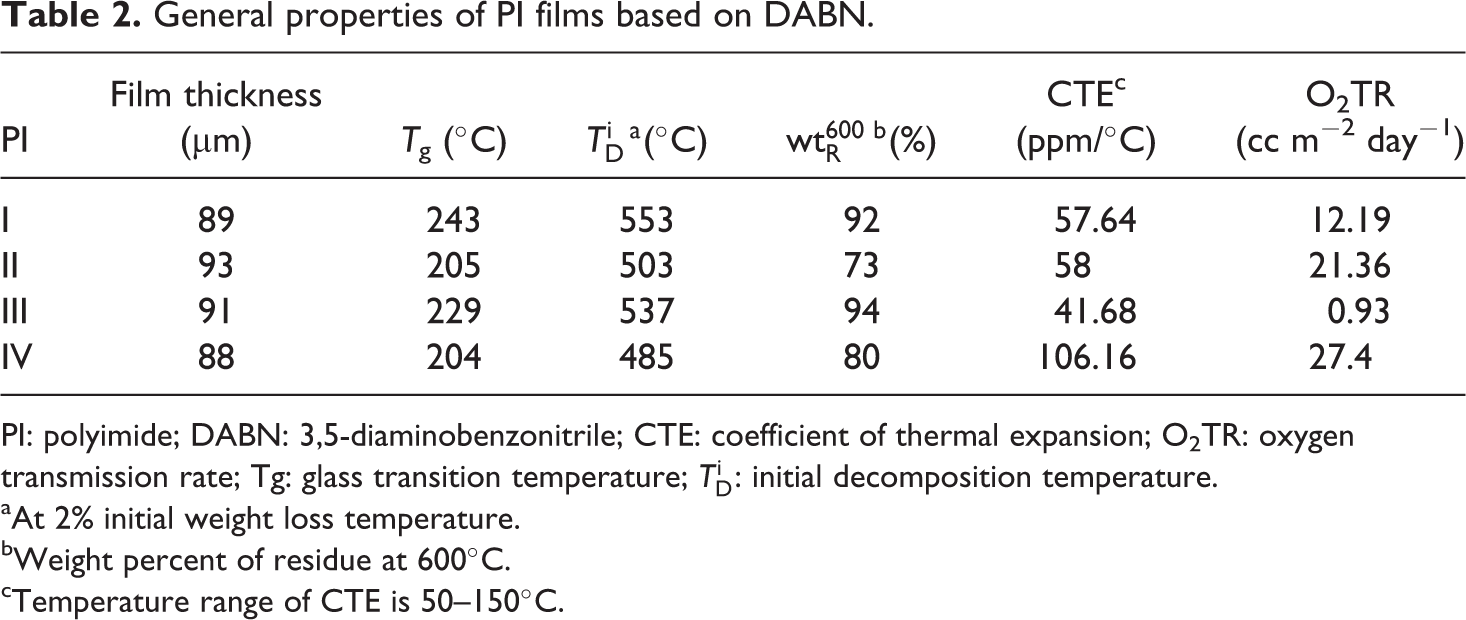
PI: polyimide; DABN: 3,5-diaminobenzonitrile; CTE: coefficient of thermal expansion; O2TR: oxygen transmission rate; Tg: glass transition temperature;
aAt 2% initial weight loss temperature.
bWeight percent of residue at 600°C.
cTemperature range of CTE is 50–150°C.
The thermal properties of sample II had lower values than those of samples I and III because of the presence of free rotational ether (–O–) linkages and kinked structures, which degraded on heating. In an SO2 group DSDA-containing polymer (IV), there was no conformational freedom because the bulky SO2 group retarded free rotation even on its own axis, and this gave rise to high torsional strain. The higher steric and conformational energy could be released by the loss of SO2 group. This led to an easy dissociation of SO2 radicals. Hence, PIs with SO2 groups exhibited low thermal stability. This was also indicated by the weight residue, that is, the SO2-based PIs exhibited low values (80%). PIs obtained from BPADA (II) also exhibited low weight residue because of the presence of a flexible ether linkage between the phthalimide units. 18,19
CTE is a function of molecular orientation. A high molecular orientation gives rise to a low CTE, and hence the residual stress is low. In the case of different types of PIs, PI orientation was greatly reduced because of the presence of bulky groups in the polymer chains, and this led to high CTE values. 20,21 The CTE values of PIs are listed in Table 2 and are in the range of 41.68–106.16 ppm/°C for various dianhydride structures. PIs containing BPDA show the lowest CTE, that is, 41.68 ppm/°C. This may be attributed to the presence of a small group, C=O, which induces close packing and chain–chain interaction, thereby lowering the CTE. However, PIs obtained from DSDA (IV) show the highest CTE, that is, 106.16 ppm/°C, because of the bulky SO2 group and a high torsional strain, which reduces the close packing. The CTE values for the PI films prepared with various dianhydride monomers are shown in Figure 5.
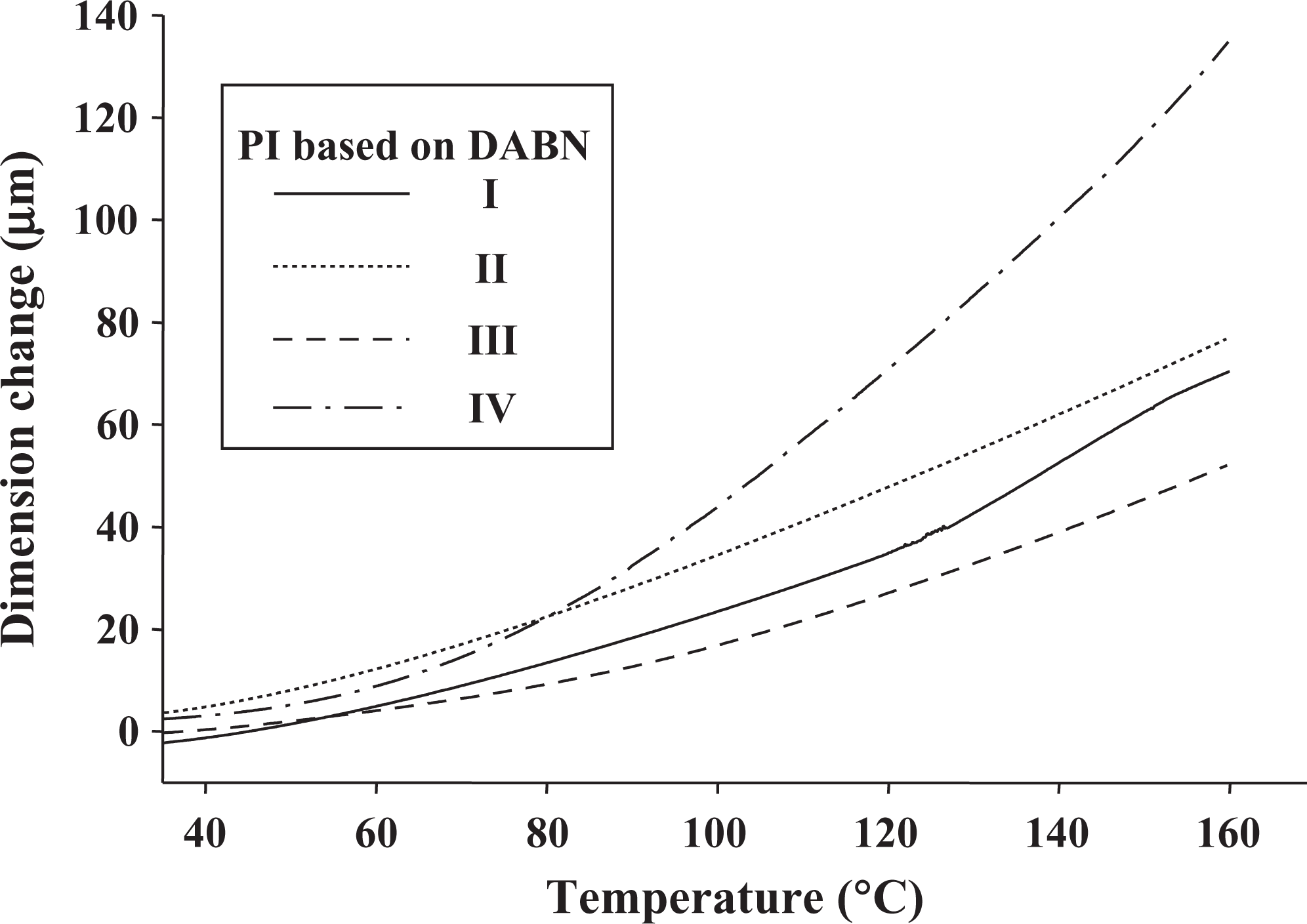
TMA thermograms of PI films based on DABN. TMA: thermomechanical analysis; PI: polyimide; DABN: 3,5-diaminobenzonitrile.
O2 permeation
Generally, to improve the gas permeability of PI films, the following factors must be considered: (1) the backbone chains must be stiffened by inhibiting their intrarotational mobility for improving the selectivity and (2) the intersegmental packing of the polymer chains must be simultaneously prevented for achieving a good permeability. 22,23
Comparing BPDA (sample III) and DSDA (sample IV), the connecting sulfonyl (–SO2) group in DSDA is more bulkier than the carbonyl (C=O) linkage group in BPDA. The molecular chains containing carbonyl groups will be highly rigid and stiffer than those containing sulfonyl group linkages, and, correspondingly, the
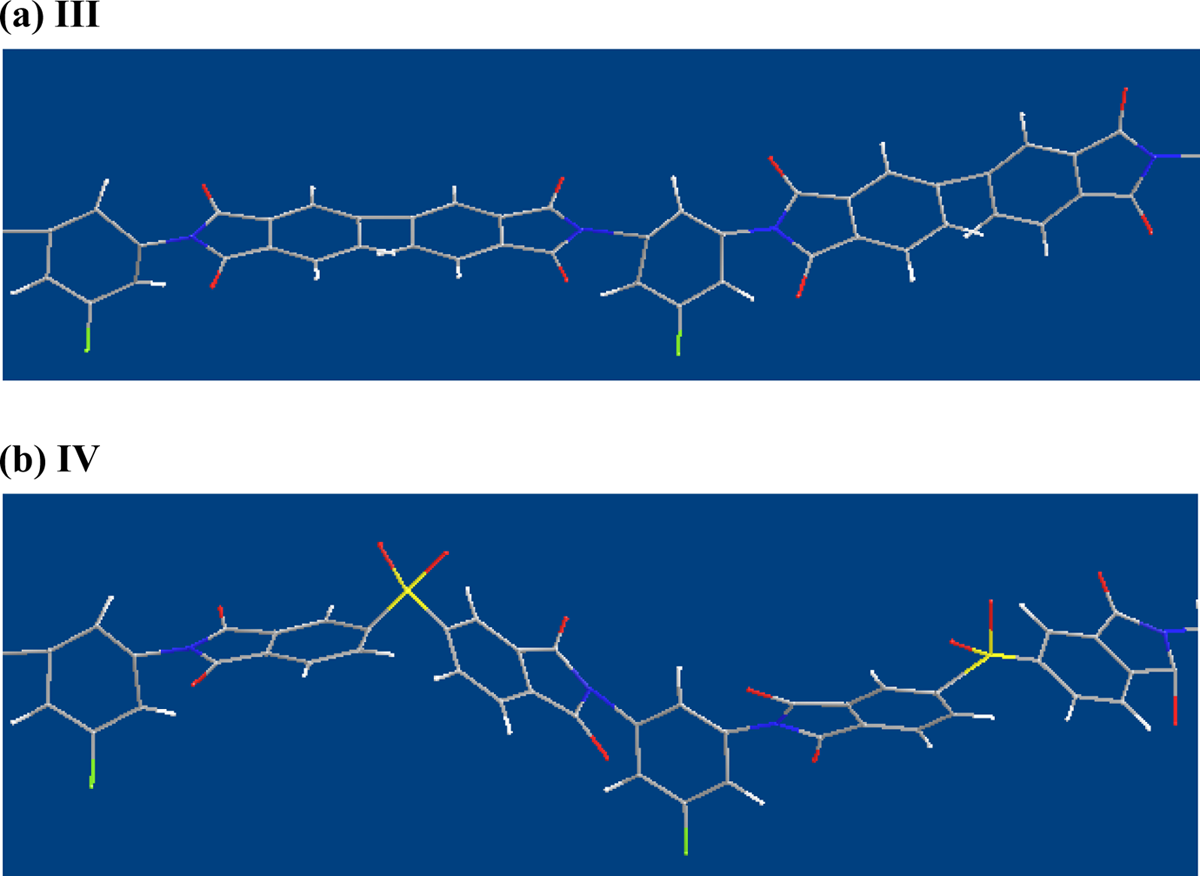
Comparison of three-dimensional chemical structures of samples III and IV.
Mechanical property
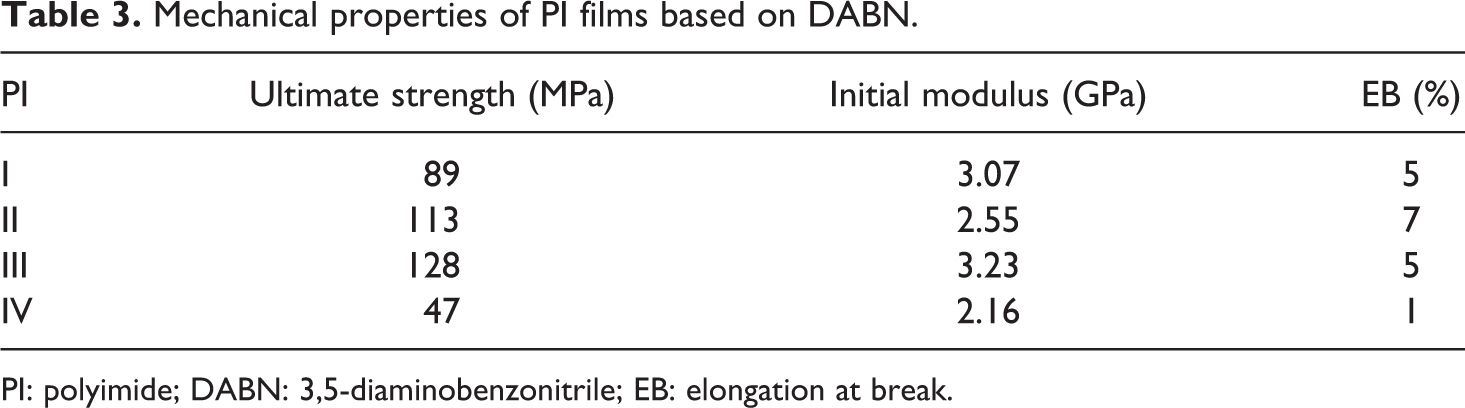
The mechanical properties of all PIs were obtained from the polymers in the form of thin films. The values of these properties are listed in Table 3. The PI samples I–IV showed ultimate tensile strengths of 47–128 MPa, initial moduli of 2.16–3.23 GPa, and elongations at break of 1–7%.
Mechanical properties of PI films based on DABN.
PI: polyimide; DABN: 3,5-diaminobenzonitrile; EB: elongation at break.
PI sample III-containing BPDA showed the highest tensile property values, with ultimate strength of 128 MPa and initial modulus of 3.23 GPa. The modulus, which is a mechanical characteristic in the film plane, might result from chain rigidity and in-plane orientation rather than chain ordering. Higher chain rigidity, which causes higher molecular in-plane orientation, produces higher in-plane modulus. This result indicates that both the high chain ordering and limited chain flexibility are reflected directly in the mechanical properties.
However, PI sample IV containing DSDA showed the lowest tensile property values, with ultimate strength of 47 MPa and initial modulus of 2.16 GPa. This result too might be related to the monomer structures mentioned previously. PI sample II showed the largest elongation at break (7%) in this series, indicating high toughness. This result suggests that the introduction of ether linkages into the imide molecules increased the strength and flexibility of the PI films.
Optical property
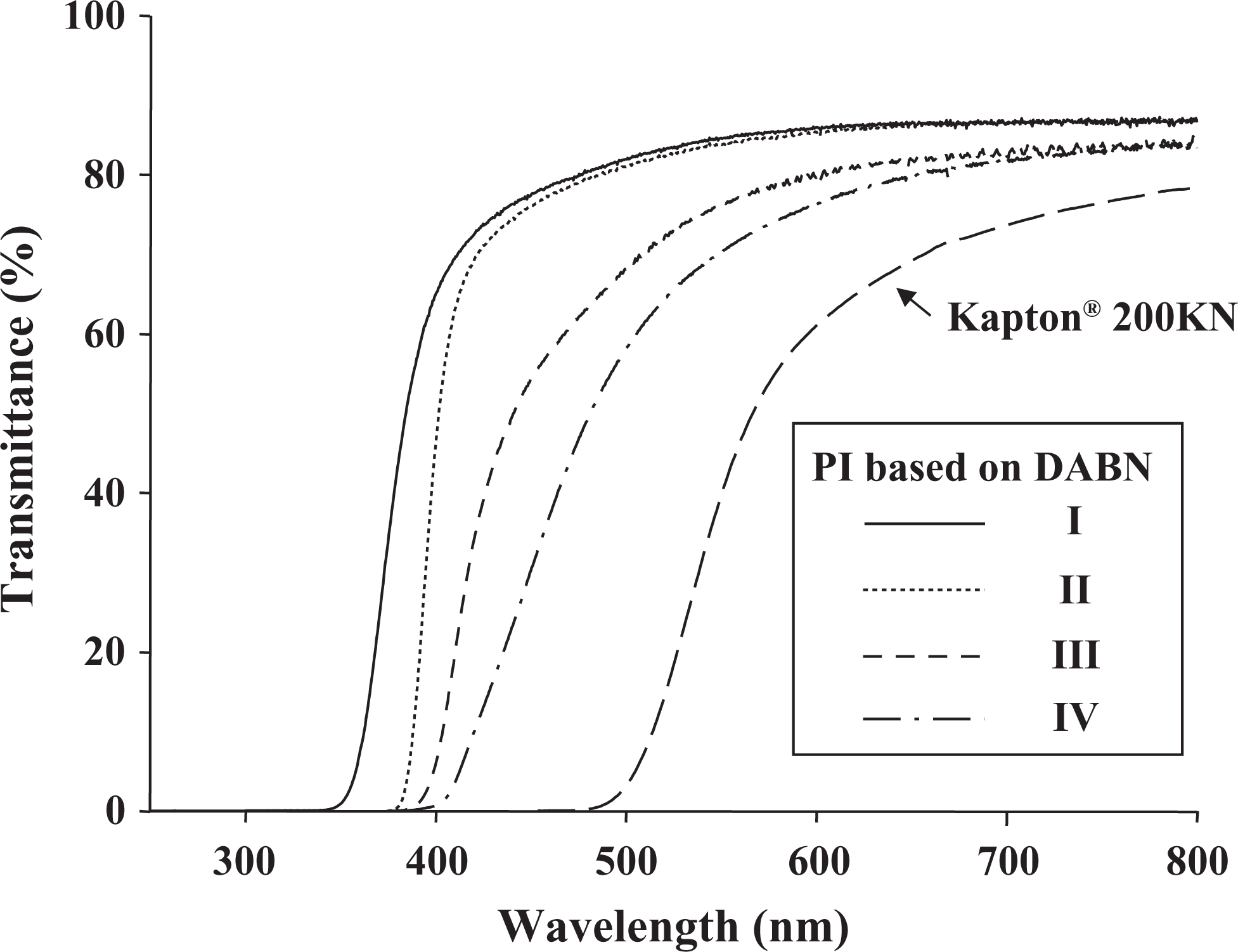
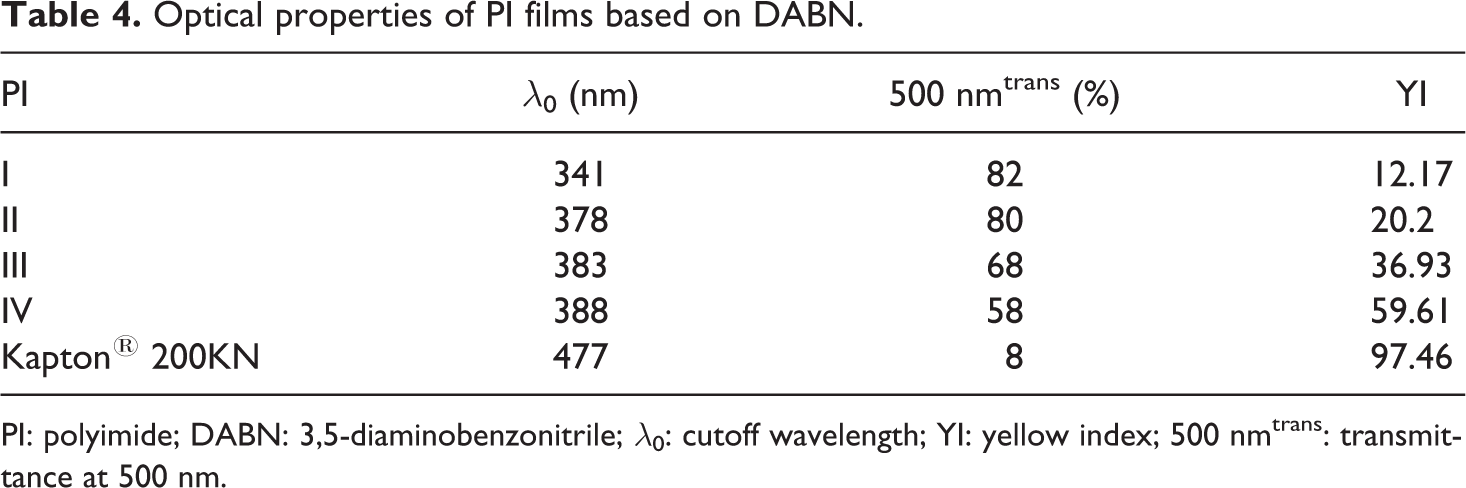
UV-Vis spectra of all the thin PI films were measured. The typical UV-Vis spectra of some representative PI films prepared with various dianhydride monomers are shown in Figure 7. The optical properties of the PIs were elucidated from the cutoff wavelengths (λo), transmittance at 500 nm, and yellow index (YI), listed in Table 4.
UV-Vis transmittance of PI films based on DABN. UV-Vis: ultraviolet–visible; PI: polyimide; DABN: 3,5-diaminobenzonitrile.
Optical properties of PI films based on DABN.
PI: polyimide; DABN: 3,5-diaminobenzonitrile;
All PI films exhibited
For comparison, the values of
The solvent-casted PI films containing 6FDA and BPADA monomers were slightly yellowish and transparent, as shown in Figures 8(a) and (b). The PI films containing BPDA (sample III) and DSDA (sample IV) appeared deep yellow in color; however, these films had good optical transparency, as there was no problem in reading a letter through the films (Figure 8(c) and (d)). In contrast to Kapton 200KN from DuPont (Wilmington, Delaware, USA; Figure 8(e) and Table 4), these PI films prepared in our study showed very good optical properties.
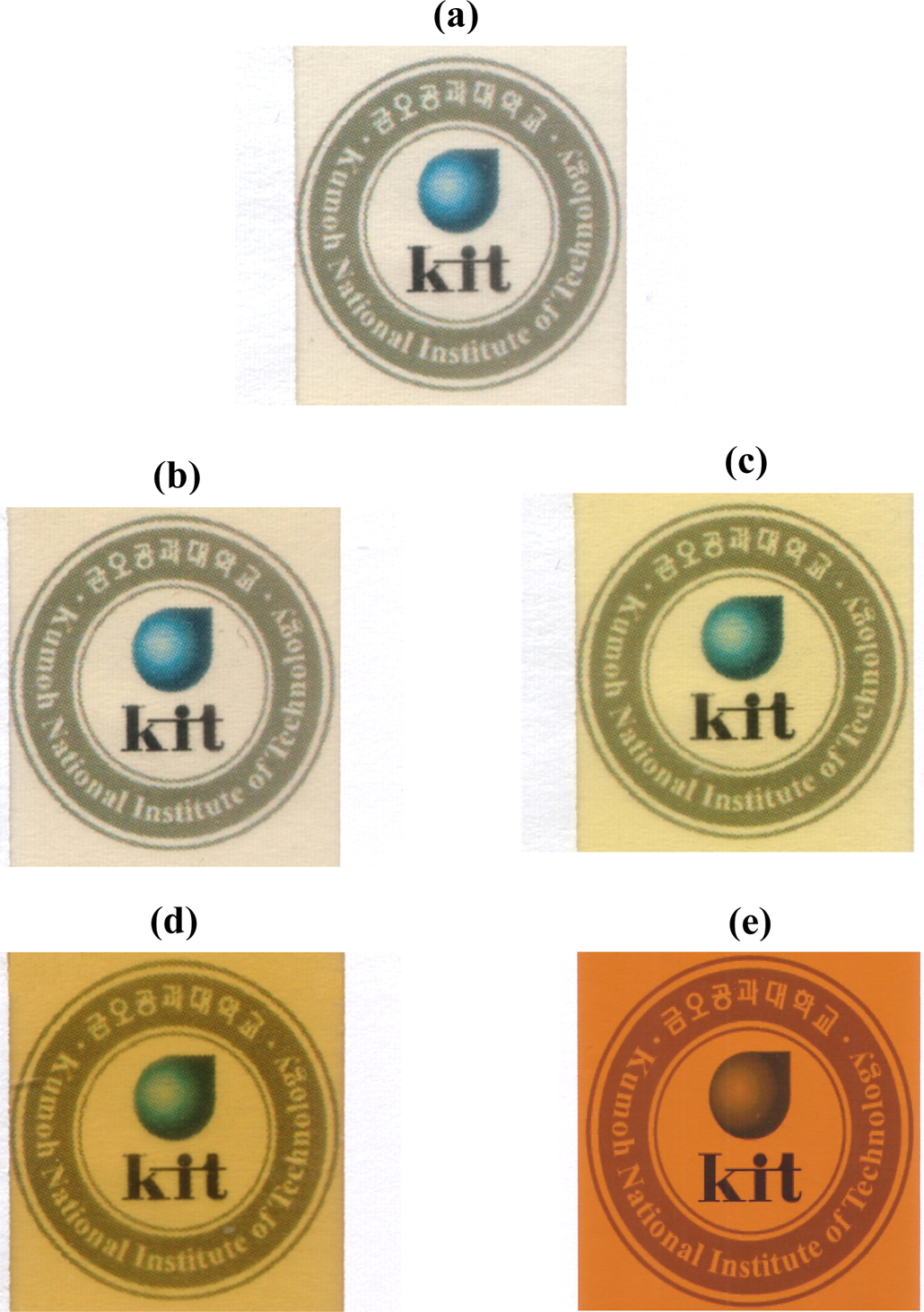
Photographs of PI films based on DABN: (a) I, (b) II, (c) III, and (d) IV. Photograph of Kapton® 200KN is shown in (e) for reference. PI: polyimide; DABN: 3,5-diaminobenzonitrile.
Conclusions
A diamine monomer, DABN, was successfully prepared and a series of PIs was synthesized from DABN with various aromatic dianhydrides via thermal imidization. PI sample I, which was synthesized from 6FDA, showed higher thermal (
In summary, it appears that the structure of the dianhydride monomers influences the determination of the thermomechanical properties, gas permeation, and optical transparency of PI polymer films.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
