Abstract
The C–O, C=O, and C(=O)O groups were introduced onto high-density polyethylene (HDPE) chains by ultraviolet irradiation in air and the groups’ content increased with increase in the irradiation time. When they irradiated for 16 h, gels were formed in the irradiated HDPE, and the content of the gels increased with increase in the irradiation time. Compared with HDPE, the crystal form of the irradiated HDPE did not change and still retained the orthorhombic structure, and its melting temperature decreased, while its crystallinity increased. The irradiated HDPE was added in HDPE/CaCO3 composites as a compatibilizer, and HDPE/irradiated HDPE/CaCO3 composites were obtained. Compared with those in HDPE/CaCO3 composites, the dispersion of the CaCO3 and interfacial interaction between CaCO3 and HDPE matrix in HDPE/irradiated HDPE/CaCO3 composites improved due to compatibilization of the irradiated HDPE, and its mechanical properties (especially impact strength), thus, showed remarkable enhancement. With increase in irradiation time, the tensile strength of HDPE/irradiated HDPE/CaCO3 composites enhanced and its impact strength increased during irradiation time of 16 h and then decreased slightly. In the same irradiation time, the tensile strength and impact strength of HDPE/irradiated HDPE/CaCO3 composites increased with increase in the content of the irradiated HDPE.
Introduction
Polyolefin is a common polymer material, which is inexpensive and simple to process. Through blending and filling, it is possible to prepare polyolefin composites with high performance. However, polyolefin is hydrophobic and inert; so the compatibility between polyolefin and inorganic fillers or engineering plastics is usually poor. Consequently, the polyolefin blends often have even worse mechanical properties than polyolefin themselves. Functionalizing polyolefin to improve its compatibility with inorganic fillers or engineering plastics has been intensively studied to prepare high-performance polyolefin composites. The most common way is by grafting polar monomer containing active functional group onto the molecular chains of the polyolefin through copolymerization. However, the grafting approach is complicated, causes pollution, and may also cause damage to the apparatus. Furthermore, residual graft monomers and other auxiliaries may negatively affect the thermal, electrical, and mechanical properties of the blends.
Some oxygen-containing groups such as C=O, C–O, and C (=O) O were introduced onto polyolefin chains by ultraviolet irradiation in air or O2 atmosphere without the addition of any monomers and auxiliaries, 1 –18 and the polyolefin was functionalized. The compatibility between the functionalized polyolefin and inorganic fillers or polarity polymer improved significantly, thereby obtaining stiffened and toughened polyolefin composite. The technique of functionalizing polyolefin by ultraviolet irradiation is a green chemical method, which is environment friendly and does not cause any chemical pollution.
In this article, high-density polyethylene (HDPE) was first irradiated by ultraviolet light in air and then the irradiated HDPE was added to HDPE/CaCO3 composites as a compatibilizer, and the HDPE/irradiated HDPE/CaCO3 composites were prepared. Compared with those of HDPE/CaCO3 composites, the compatibility, dispersion, and mechanical properties of HDPE/irradiated HDPE/CaCO3 composites increased.
Experiment
Materials
HDPE (DGDA 6098) with a melt flow rate (MFR) of 0.1 g/10 min and an average particle size of 216 µm was manufactured by Qilu Petrochemical Co. Ltd (China). CaCO3 with an average particle size of 1.2 µm was provided by Nanjing University of Technology (China).
Ultraviolet irradiation of HDPE
Ultraviolet irradiation of HDPE was completed using a Ga-I lamp with a wavelength in the range of 365–450 nm at light intensity of 78 W/m 2 and temperature of 40°C in air.
Preparation of HDPE/irradiated HDPE/CaCO3 composite
HDPE, irradiated HDPE, and CaCO3 were blended with a twin roller at 155°C for 10 min to prepare the composites. The composites were melted at 190°C for 6 min pressed to 15 MPa for 4 min in a hot hydraulic press, and then pressed to 15 MPa at room temperature for 10 min in another cold hydraulic press to prepare sheets of 1 or 4 mm thickness.
Measurement and characterization
X-ray photoelectron spectroscopy (XPS) analysis was carried out on an ESCALB MK II spectrometer (UK) with AlKα source (1486.6 eV). The C1S peak was fittingly decomposed into component peaks according to Gaussian distribution.
The gel content included irradiated HDPE which was wrapped by stainless steel net and extracted for 72 h by refluxing xylene in a soxhlet extractor and then dried.
X-ray diffraction (XRD) analysis was performed with a D/Max II diffractometer (Rigaku, Japan). The sample was scanned from 10° to 50° at a scan rate of 4°/min.
Differential scanning calorimetry (DSC) analysis was performed on a Pyres 1 thermal analyzer (Perkin Elmer, USA). Sample was heated from room temperature to 180°C at a heating rate of 10°C/min. The crystallinity (C) was determined from C = ΔHm/ΔHm o×100%, where ΔHm is the melt enthalpy of the HDPE sample and ΔHm o is the melt enthalpy at 100% crystallinity of polyethylene, which is 285.9 J/g.
Contact angle with water was measured using a Model RAMHART-100 optical bench-type contact angle goniometer (KINO Industry Co, USA).
MFR was measured using an XNR-400A Melt index tester (Desheng, China) at 190°C and at a load of 2160 g.
In Molau test, the HDPE/irradiated HDPE/CaCO3 composites (1 g) were dissolved in hot xylene (19 g). The suspension was laid for 72 h at 90°C and then the suspension was carefully observed.
SEM photograph was obtained using a LEO-15300VP Scanning Electron Microscope (LEO, Germany).
Tensile test was carried out on an Instron 4466 all-purpose tester (Instron, USA).
Notched Izod impact strength was measured using an XJ-40A apparatus (Wuzhong, China).
Results and discussion
Structure and performance of irradiated HDPE
XPS analysis of irradiated HDPE
The XPS spectra of HDPE and irradiated HDPE are shown in Figure 1. The XPS analysis data of HDPE and irradiated HDPE are listed in Table 1. Compared to the HDPE, the content of C–H in the irradiated HDPE decreased and the content of C–O, C=O, and C(=O)O groups increased. This indicated that the C–O, C=O, and C(=O)O groups were introduced onto HDPE chains through ultraviolet irradiation in air. The content of the groups increased with increase in the irradiation time.
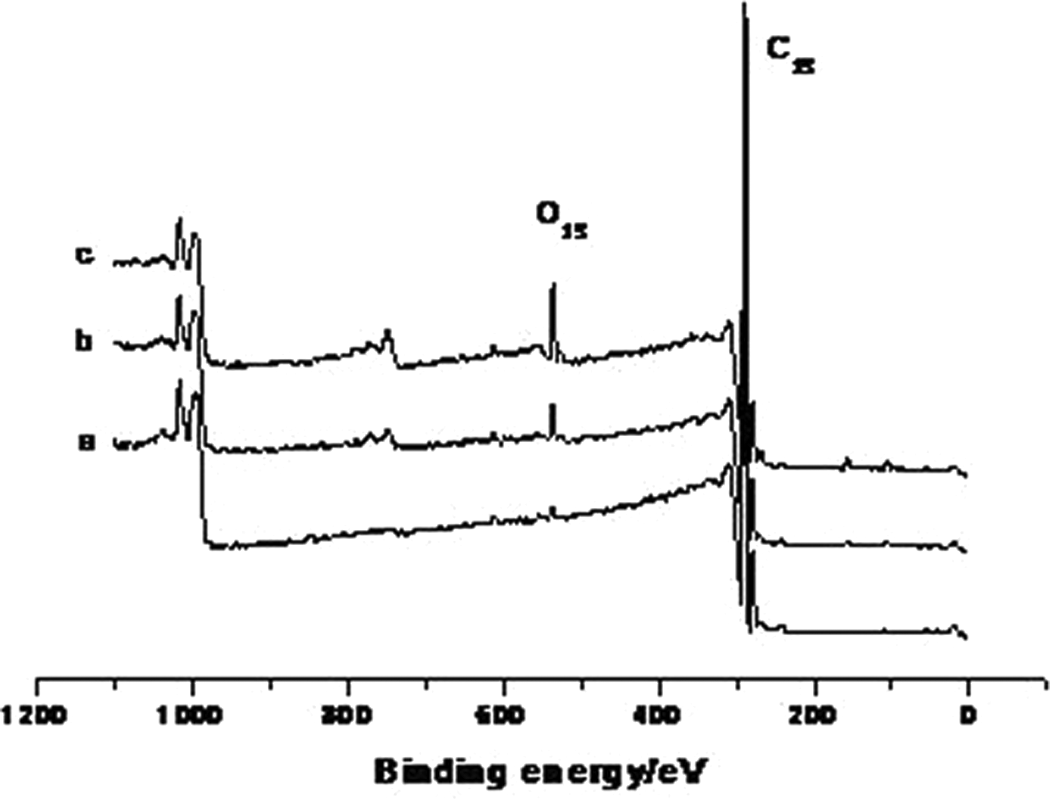
XPS spectra of HDPE (a) and irradiated HDPE for 24 (b) and 36 h (c). XPS: X-ray photoelectron spectroscopy; HDPE: high-density polyethylene.
X-ray photoelectron spectroscopy analysis data for irradiated HDPE.
Gel analysis of irradiated HDPE
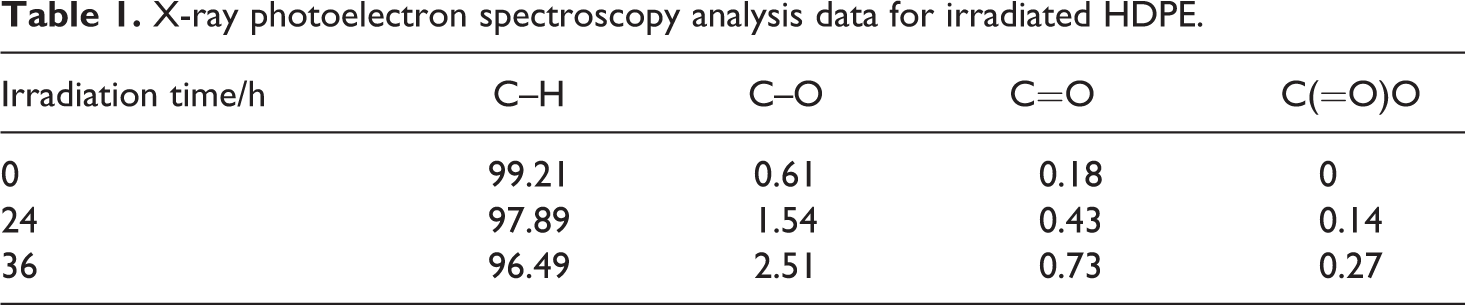
The gel content of HDPE and irradiated HDPE is listed in Table 2. The gel was not found in the irradiated HDPE when it was irradiated for 8 h, while there were gels in the irradiated HDPE when the irradiation time was 16 and 24 h, and the gel content enhanced with increase in the irradiation time.
Gel content of irradiated high-density polyethylene.
XRD and DSC analyses of irradiated HDPE
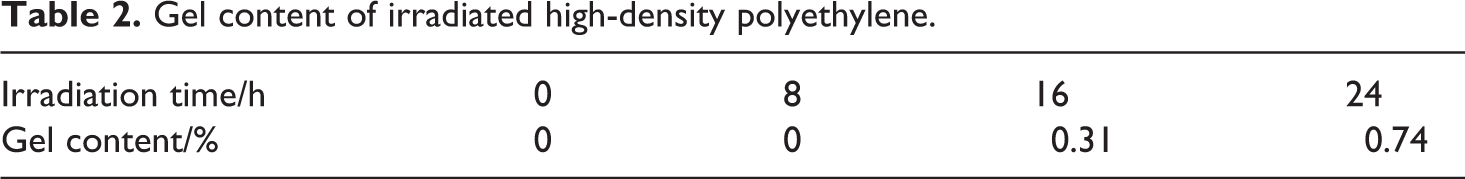
Compared to the HDPE, the crystal form of the irradiated HDPE did not change and still kept an orthorhombic structure, while its melting temperature dropped and its crystallinity advanced (Table 3). The decrease in the melting temperature was due to the crystal lattice defects caused by ultraviolet irradiation and oxidation. 18 The increase in the crystallinity was due to recrystallization of the broken chains of the irradiated HDPE. 18
Melting temperature and crystallinity of irradiated high-density polyethylene.

Hydrophilicity and fluidity of irradiated HDPE
The contact angle with water and MFR of HDPE and irradiated HDPE are listed in Table 4. Compared with that of the HDPE, the hydrophilicity of the irradiated HDPE improved due to the introduction of C–O, C=O, and C (C=O)O groups. The hydrophilicity increased with increase in the irradiation time. The irradiated HDPE was added to HDPE/CaCO3 composites as a compatibilizer which might improve the compatibility of the composites.
Contact angle and melt flow rate of irradiated high-density polyethylene.
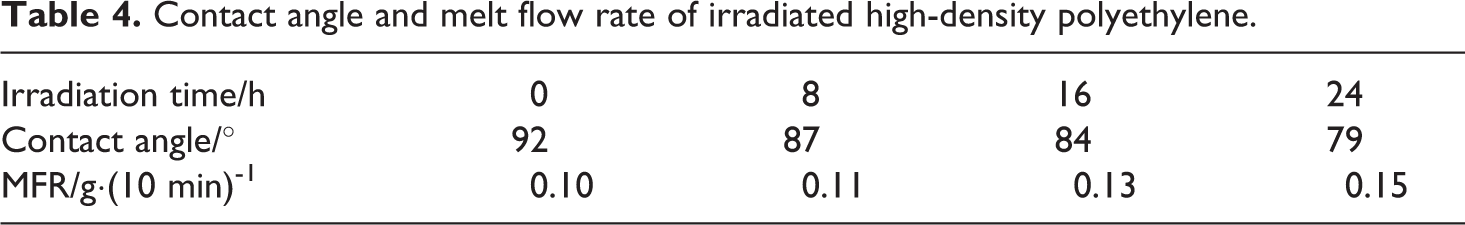
The MFR of the irradiated HDPE was higher than that of the HDPE due to the degradation of the irradiated HDPE chains. The increase of the MFR will be favorable to the subsequent fabrication of HDPE/irradiated HDPE/CaCO3 composites.
Compatibilization of irradiated HDPE
Molau test of HDPE/irradiated HDPE/CaCO3 composites
The xylene suspensions of the HDPE/CaCO3 composites was separated after 72 h into two parts, namely the transparent xylene solution containing HDPE component on the top and CaCO3 on the bottom (Table 5). This indicated that the dispersion of CaCO3 and the interfacial interaction between the HDPE matrix and CaCO3 were poor. Thus, the aggregated CaCO3 was completely separated from the HDPE and deposited on the bottom.
Molau test of high-density polyethylene (HDPE)/irradiated HDPE/CaCO3 (45/15/40) composites.

When the xylene suspensions of the HDPE/irradiated HDPE/CaCO3 composites were allowed for 72 h, the top part consisted of a turbidity suspension containing the CaCO3 component, while partial CaCO3 deposited at the bottom, and the amount of the deposited CaCO3 decreased with increase in the irradiation time. These indicated that the dispersion of CaCO3 and the interfacial interaction between CaCO3 and the HDPE matrix in HDPE/irradiated HDPE/CaCO3 composites were better than those in the HDPE/CaCO3 composites, thus the partial CaCO3 suspended in the xylene solutions containing the HDPE component, while the other CaCO3 depositedat the bottom.
SEM of HDPE/irradiated HDPE/CaCO3 composites
The SEM photographs of liquid nitrogen frozen fractured surface of HDPE/CaCO3 and HDPE/irradiated HDPE/CaCO3 composites are shown in Figure 2. On the frozen fractured surface of HDPE/CaCO3 composite, some bare agglomerated CaCO3 and big holes caused by the fall of the CaCO3 from the HDPE matrix were found, indicating that the dispersion and the interfacial interaction of HDPE/CaCO3 composites were poor. On the frozen fractured surface of HDPE/irradiated HDPE/CaCO3 composites, there was no bare agglomerated CaCO3, and the CaCO3 was coated by the HDPE matrix, indicating that the dispersion and the interfacial interaction of HDPE/irradiated HDPE/CaCO3 composites increased.
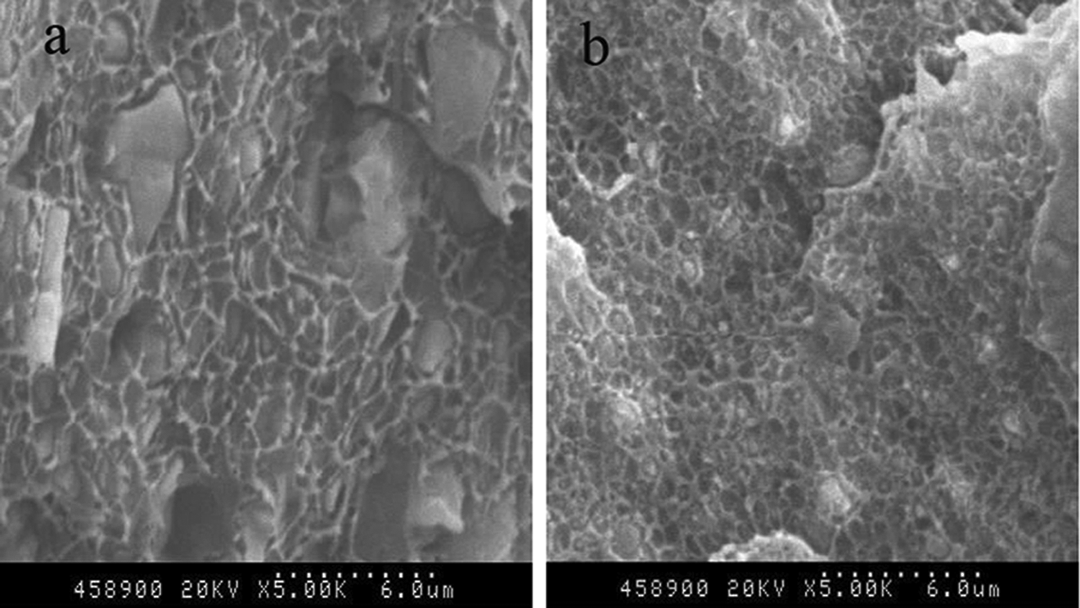
SEM photographs of liquid nitrogen frozen fractured surface of HDPE/CaCO3 (60/40) (a) and HDPE/irradiated HDPE (16 h)/CaCO3 (45/15/40) composites (b). SEM: scanning electron microscope; HDPE: high-density polyethylene.
The SEM photographs of the impact fractured surface of HDPE/CaCO3 and HDPE/irradiated HDPE/CaCO3 composites are shown in Figure 3. The impact fractured surface of HDPE/CaCO3 composites was smooth, indicating that HDPE/CaCO3 composites displayed a brittle fracture during the impact process. The impact fractured surface of HDPE/irradiated HDPE/CaCO3 composites was accidented and had some fibriform HDPE component, indicating that the HDPE/irradiated HDPE/CaCO3 composites showed a rough fracture during the impact process.

SEM photographs of impact fractured surface of HDPE/CaCO3 (60/40) (a) and HDPE/irradiated HDPE (16 h)/CaCO3 (45/15/40) composites (b). SEM: scanning electron microscope; HDPE: high-density polyethylene.
Mechanical properties of HDPE/irradiated HDPE/CaCO3 composites
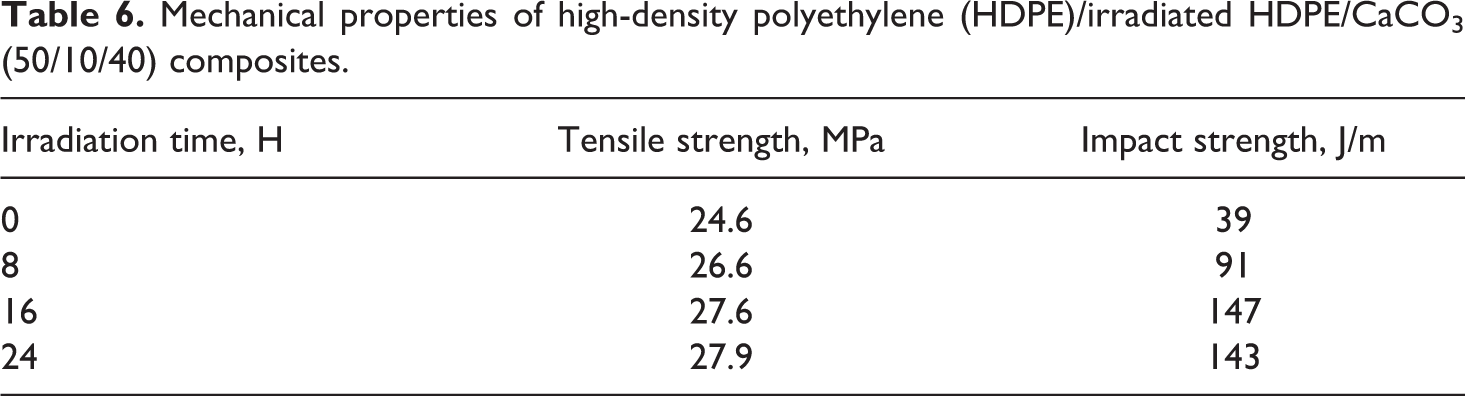
The mechanical properties of HDPE/CaCO3 and HDPE/irradiated HDPE/CaCO3 composites are listed in Tables 6 and 7. The HDPE/CaCO3 composites showed low tensile strength and notched impact strength due to poor dispersion of CaCO3 and weak interfacial interaction between CaCO3 and the HDPE matrix.
Mechanical properties of high-density polyethylene (HDPE)/irradiated HDPE/CaCO3 (50/10/40) composites.
Mechanical properties of high-density polyethylene (HDPE)/irradiated HDPE/CaCO3 (45/15/40) composites.
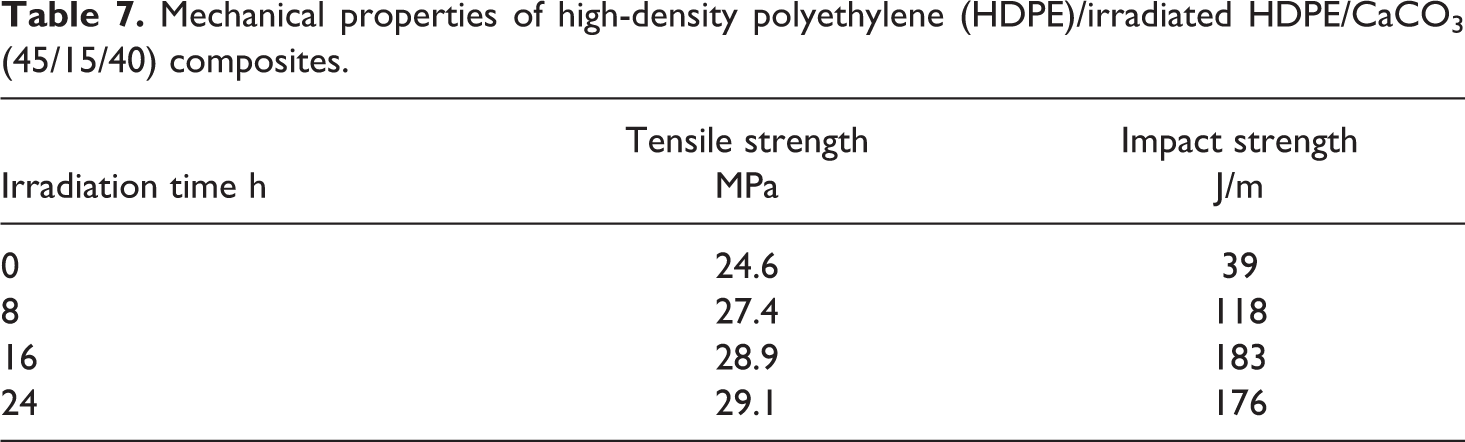
With increase in the irradiation time, the tensile strength of HDPE/irradiated HDPE/CaCO3 composites advanced, and its notch impact strength heightened within the irradiation time of 16 h and then decreased. With the same irradiation time, the tensile strength and notched impact strength of HDPE/irradiated HDPE/CaCO3 composites increased with increase in the content of the irradiated HDPE. Compared with that of the HDPE/CaCO3 (60/40) composites, the tensile strength and notched impact strength of the HDPE/irradiated (16 h) HDPE/CaCO3 (45/15/40) composites enhanced by 17.5% and 369.2%, respectively.
Conclusions
The groups of C–O, C=O, and C(=O)O were introduced onto HDPE chains by ultraviolet irradiation in air. The content of the groups increased with an increase in the irradiation time. There were gels in the irradiated HDPE during the irradiation time of up to 16 h, and the content of the gels increased with increase in the irradiation time. Compared with that of the HDPE, the crystal form of the irradiated HDPE did not change in an orthorhombic structure and its melting temperature decreased, while its crystallinity increased. With an increase in the irradiation time, the degree of dispersion of CaCO3 and the interfacial interaction between CaCO3 and HDPE in HDPE/irradiated HDPE/CaCO3 composites improved, and the tensile strength of HDPE/irradiated HDPE/CaCO3 composites enhanced. The impact strength of HDPE/irradiated HDPE/CaCO3 composites increased during the irradiation time of 16 h and then decreased slightly. The tensile strength and impact strength of the composites enhanced with an increase in the content of the irradiated HDPE, which was irradiated for the same duration of time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
