Abstract
Nanocopper (nano-Cu)/poly(acetoacetoxyethyl methacrylate (AAEM)–styrene (St)) (P(AAEM-St)) composites were synthesized by reducing copper acetate solution in AAEM and St monomer by ultrasonic technique without adding emulsifier. The morphology, structure, thermal stability, and the interaction between Cu nanoparticles (NPs) and P(AAEM-St) matrix of the composites were characterized using ultraviolet–visible spectroscopy, X-ray diffractometer, transmission electron microscopy, X-ray photoelectron spectroscopy (XPS), Fourier transform infrared spectroscopy (FTIR), and thermogravimetric analysis (TGA), respectively. The results show that Cu NPs possessed face-centered cubic structure, and they were homogeneously dispersed in P(AAEM-St) matrix. XPS and FTIR analyses reveal the interaction between Cu NPs and C=O of the acetoacetoxy in the copolymer of AAEM and St. TGA proves that the thermal stability of the pure P(AAEM-St) is higher than that of the nano-Cu/P(AAEM-St) composites.
Introduction
The advanced functional materials of nanocopper (nano-Cu)/polymer composites continue to be a productive research area due to their high electrical conductivity, excellent processing properties as well as their promising applications applied in catalysis, 1 –3 electrics, 4,5 electrochemistry, 6,7 and biomedical materials, 8,9 which are well established and extensively investigated mainly in colloidal systems. Nevertheless, Cu nanoparticles (NPs) are difficult to be applied because they undergo oxidation easily in ambient air. Cu NPs are often loaded on various substrates and matrices. 1,10,11 For this point, polymer materials may be the best choice due to the specific morphology, chemical and structural nature with the long polymeric chains allowing incorporation and admirable dispersion of NPs. Additionally, the formation principle of polymer-based metal nanocomposites have been regarded as the interaction between polymer and nanometal particles, 12 so the suitable functional groups of polymers can be used as targeted reactive sites for the controlling synthesis of NPs via one step. Acetoacetoxyethyl methacrylate (AAEM) is one of the precursors for preparing metal-based nanocomposites. Besides acting both as a strong chelating agent and a highly reactive methacrylate group, AAEM also can react with the other vinyl monomers readily through chemical bonds. Moreover, it has been demonstrated strongly that the emulsifier-free emulsion polymerization can effectively eliminate the shortcomings of emulsifier which could lead to negative effects on the material properties. Therefore, preparation of core–shell NPs–polymer composites through the emulsifier-free emulsion polymerization is environmentally friendly. By this method, the particles can be protected well when poly(acetoacetoxyethyl methacrylate–styrene (St)) (P(AAEM-St)) matrix is applied as package for synthesis of inorganic NPs. Wang et al. 13 have successfully prepared zirconium dioxide/PAAEM/polystryrene NPs packed with core–shell structure using AAEM and St copolymer as stabilizers.
Up to now, several methods have been established for synthesis of nanometal/polymer composites. 10,14 –16 Most recently, some methods have received people’s great intention, such as ultraviolet (UV) radiation reduction, 17,18 sonochemical synthesis, 19,20 γ-irradiation radiation, 21 and microwave reduction 22 based upon one-step dual in situ preparation of nanometal/polymer composites. In this article, nano-Cu/P(AAEM-St) composites were prepared by reducing copper acetate with vitamin C (Vc) in AAEM and St monomer. In the meantime, the monomers were copolymerized through an emulsifier-free emulsion polymerization approach under ultrasonic radiation. The Cu NPs were also homogeneously dispersed in P(AAEM-St) copolymer. This method is very simple and suitable for large-scale manufacturing of nanometal/polymer composites. The microstructure, composition of the prepared nanocomposites, and the interactions were characterized with various techniques. The thermal stability of the prepared nanocomposites was also evaluated using thermogravimetric analysis (TGA).
Experimental
Preparation of nano-Cu/P(AAEM-St) composites
Vc, 3 mL of copper acetate (analytically pure grade) and 0.009 g of potassium persulfate (analytically pure grade) were dissolved in water under constant stirring, then 1 mL AAEM (99%, Eastman Co., Rochester, New York, USA) and 5 mL St (analytically pure grade) were added under nitrogen (N2) condition. The reaction was controlled at 40°C by ultrasonic (TY98-III ultrasonic generator, Ningbo Scientz Biotechnology Co. Ltd, Ningbo, China; frequency: 20 kHz; output power: approximately 200–1200 W) for 60 min. After extracting with methanol, the reaction system was washed by methanol and water for three times, respectively. Then, reddish brown powder was obtained and dried under vacuum at 100°C for several hours. The sample of pure (AAEM-St) copolymer was also prepared by means of the similar copolymerization approach in the absence of copper acetate.
Characterization of nano-Cu/P(AAEM-St) composites
XRD pattern of the prepared nanocomposites was determined at a scanning rate of 0.03°s−1in 2
Results and discussion
The crystal structure of the nano-Cu/P(AAEM-St) composites was investigated by XRD (Figure 1). The XRD pattern of products illuminated that there were three main peaks appeared at 2
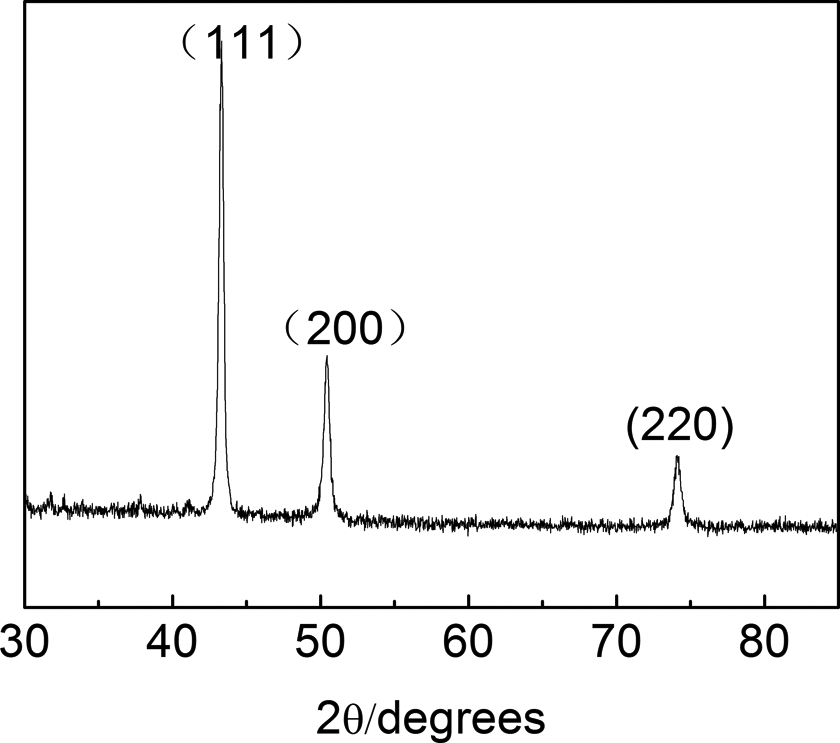
XRD pattern of nano-Cu/P(AAEM-St) composites. XRD: X-ray diffraction; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
Figure 2 shows the TEM image of nano-Cu/P(AAEM-St) composites. It can be seen that the Cu NPs within polymer composites were synthesized with uniform size in the range of 10–30 nm. As shown in Figure 2(a), the synthesized samples were dispersed homogeneously in the polymer matrix. The Cu NPs were of nearly spherical shape or spherical structure. The face-centered cubic polycrystalline structure was also proved by the electron diffraction analysis in Figure 2(b). According to the basic formula of electron diffraction (

(a) TEM image of nano-Cu/P(AAEM-St) composites and (b) electron diffraction photograph of Cu nanoparticles. TEM: transmission electron microscopy; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
Figure 3 shows the ultraviolet–visible (UV-Vis) absorbance spectrum of the dispersion Cu NPs after the sonochemical process. The absorption peaks at 286 and 293 nm are attributed to the P(AAEM-St) copolymer and nano-Cu/P(AAEM-St) composites, respectively. More significantly, there was a new absorption band at 589 nm for nano-Cu/P(AAEM-St) composites, which can be ascribed to the surface plasmon bands for the Cu NPs. It is well known that the colloidal dispersion of metals exhibits an absorption band in the UV-Vis regions due to collective excitations of the free electron (surface plasmon band). Haas et al. 23 observed a similar absorbance value for Cu NPs prepared by a chemical method.
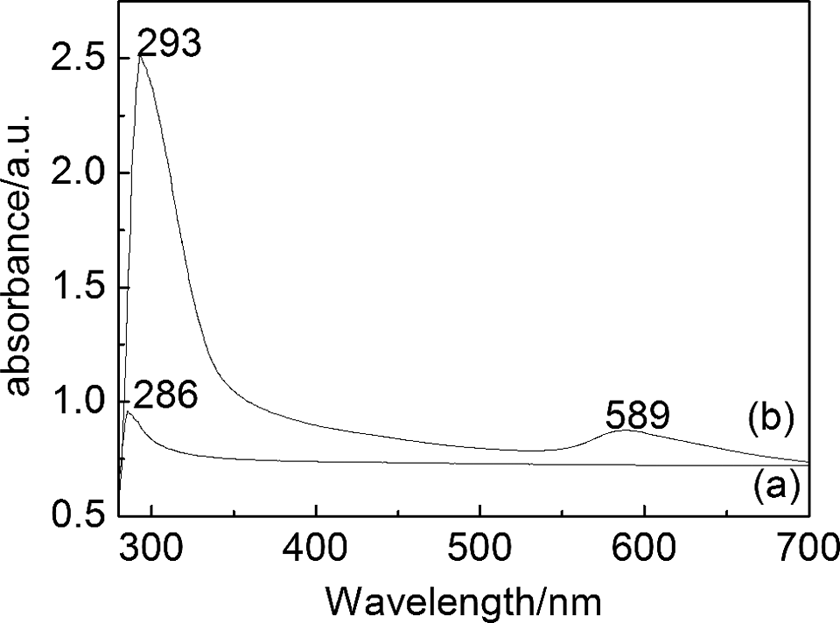
UV-Vis spectra of (a) P(AAEM-St) and (b) nano-Cu/P(AAEM-St) composites. UV-Vis: ultraviolet–visible; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
To investigate the bonding characteristics or the interaction between the inorganic particles and copolymer, the FTIR technique was used to characterize the organic groups among them. Figure 4 shows the typical FTIR spectra of P(AAEM-St) and nano-Cu/P(AAEM-St) composites. The stretching vibration bands centered at 1747 and 1723 cm−1 could be attributed to C=O stretching vibration of the ester group and keto group, respectively. The distortion vibration peaks of CH2 centered at 1600, 1494, and 1452 cm−1, respectively. The vibration peak located at 1650 cm−1 correspond to the v(C=O + C=C) vibration of the enolic form of β-ketoesters. A new absorption peak located at 1515 cm−1 appeared in the FTIR spectrum of the composites, which is the typical asymmetric vibration peak of carbonyl group after complexation on the surface of particle. 24 In addition, the intensity of the characteristic absorption peak of the ester group and keto group of the composites located at 1747 and 1723 cm−1 decreased remarkably in comparison with the copolymer, which convincingly indicating the formation of complexation of the carbonyl group of polymeric matrix on the surface particle. The absorption peak became broad in the low-frequency range and a new absorption peak located at 577 cm−1 appears may be due to the effect of the CH2 vibration of hexatomic ring formed by complexation between the oxygen atom of carbonyl group and Cu NPs.
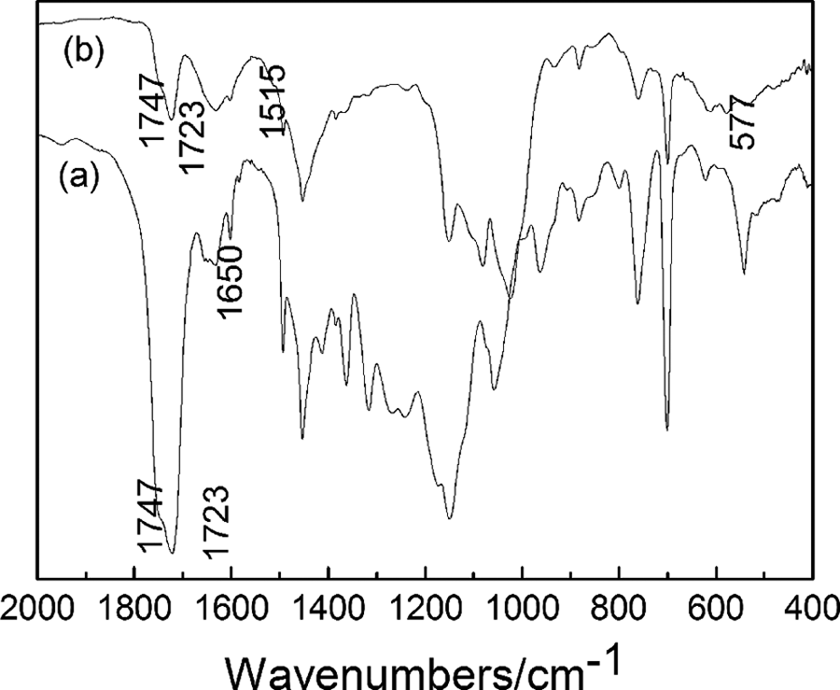
FTIR spectra of (a) P(AAEM-St) and (b) nano-Cu/ P(AAEM-St) composite. FTIR: Fourier transform infrared; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
The schematic diagram of the chemical interaction between Cu NPs and P(AAEM-St) was shown in Figure 5.
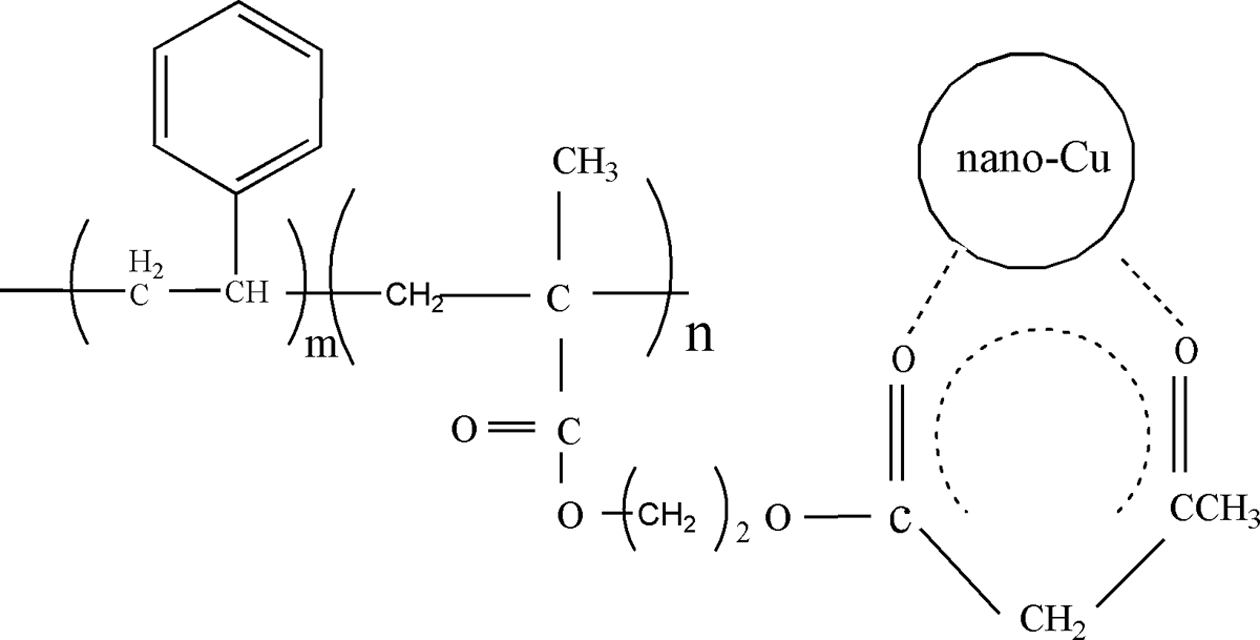
Schematic diagram showing the interaction between nano-Cu and P(AAEM-St). Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
To comprehend the interaction between Cu NPs (dispersing phase) and P(AAEM-St) copolymer (continuous phase), the nano-Cu/P(AAEM-St) composites were examined by XPS. The obtained spectrum of nano-Cu/P(AAEM-St) was shown in Figure 6(a). And carbon, oxygen, and Cu elements were found (Figure 6(a)), which were consistent with the chemical components of the nano-Cu/P(AAEM-St) composites. The binding energies of 284, 533, and 934 eV could be attributed to C1s, O1s, and Cu2p peaks, respectively.
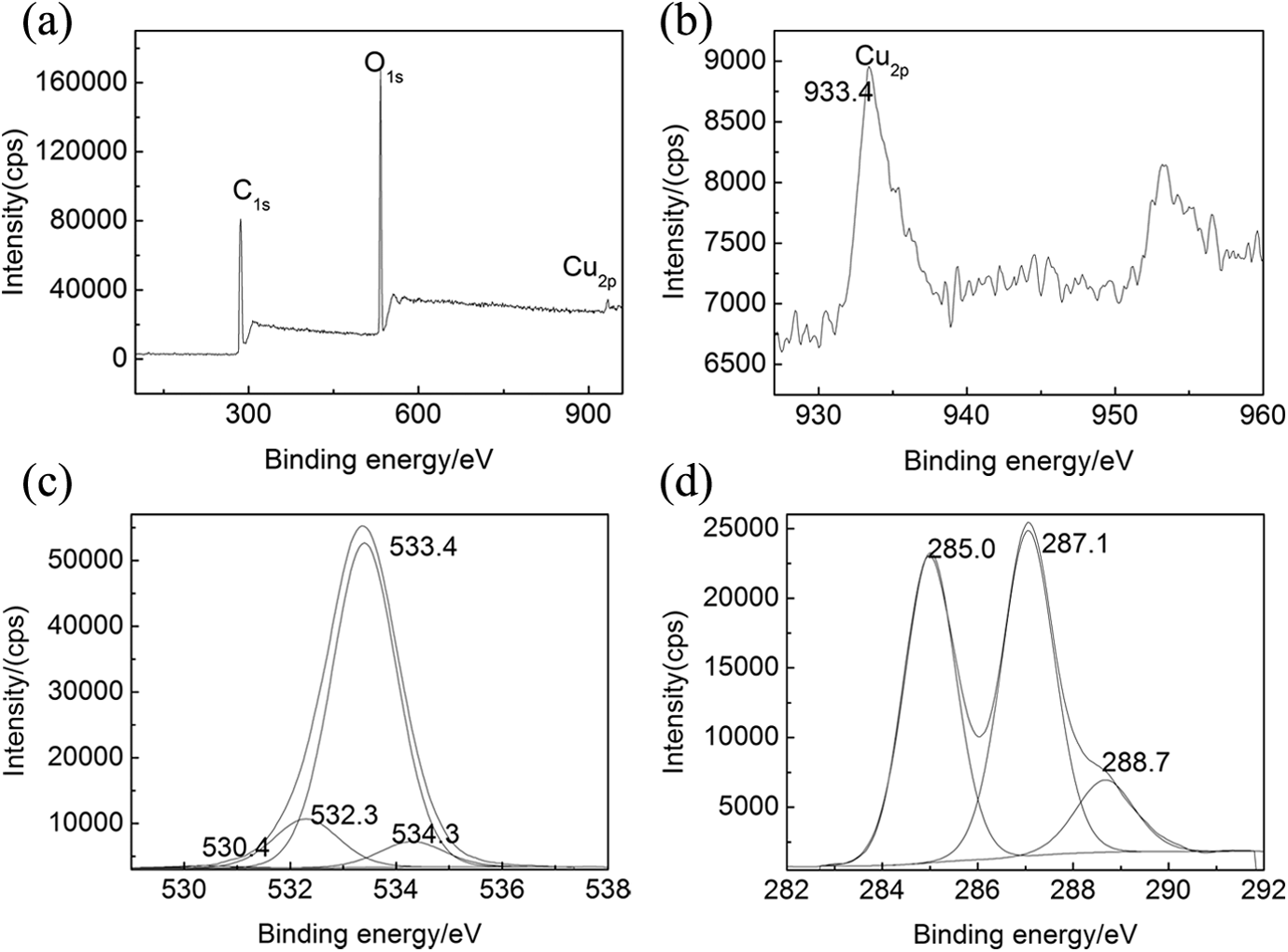
XPS spectra of nano-Cu/P(AAEM-St) composite. (a) survey XPS of nano-Cu/P(AAEM-St) powder, (b) XPS of Cu2p, (c) XPS of O1s, and (d) XPS of C1s. XPS: X-ray photoelectron spectroscopy; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
In Figure 6(b), the energy spectrum peaks of Cu2p electrons positioned at 933.4 eV, which is higher than the standard energy spectrum peaks of Cu2p electrons that positioned at 932.7 eV. The energy spectrum peaks of Cu2p electrons within nanocomposites moved to the high-binding energy, which indicated that the chemical environment of Cu NPs was changed already may be due to the coordination interaction between certain atoms of polymer and Cu NPs. The binding energy within the electronic shell was increased because of decreasing of valence electron density and the shielding effects of domestic electronic shell.
A broad and complex band for O1s was observed in the XPS spectrum for the composites (Figure 6(c)) and the better subdivision of the spectrum along with baseline-corrected raw data revealed the convolution of four different groups. The O1 and O2 energy bands located at around 530.4 and 532.3 eV could be assigned to the O1s in C=O associated with ketone carbonyl and ester carbonyl of the conjugate ring, respectively. As the carbonyl oxygen atoms could accept electrons that come from Cu2p, the binding energy for the O1 and O2 was higher than the standard energy spectrum peaks. The O3 and O4 energy bands located at around 533.4 and 534.3 eV could be assigned to the O1s in C–O–C and C=O associated with aliphatic chain, respectively. It is concluded that the interaction between Cu NPs and polymeric matrix was brought mainly through the complexation of the carbonyl oxygen atoms in copolymer with Cu atoms. The conclusion demonstrated the chemical interaction between Cu NPs and P(AAEM-St) as shown in the schematic diagram (Figure 5). Moreover, the C1s peak was also subdivided and five bands were obtained as shown in Figure 6(d). The peaks located at 285.0, 287.1, and 288.7 eV could be ascribed to the C–H, C–O, and C=O of the polymers, respectively.
Figure 7 shows the TGA data for pure P(AAEM-St) and nano-Cu/P(AAEM-St) composites. The decomposition temperature of nano-Cu/P(AAEM-St) composites was lower than that of pure P(AAEM-St), and the composites have two distinct transitions in the mass loss curve, while the pure P(AAEM-St) has only one. There are two opposing reasons for this phenomenon. On the one hand, Cu NPs that dispersed homogeneously in P(AAEM-St) matrix could rapidly transmit heating effect in the heating process, thereby promoting decomposition of P(AAEM-St) polymer. On the other hand, there is a stable hexatomic ring structure between Cu NPs and P(AAEM-St), and this interaction hinders the movement of molecular chain of polymer. It enhances the amount of energy of the molecular chain rupture required, which restrains decomposition of P(AAEM-St) polymer. Therefore, we can conclude that the thermal stability of nano-Cu/P(AAEM-St) composites decreased with Cu NPs in the composites, and the existence of stable hexatomic ring structure produces certain effect on the thermal properties of the complex.
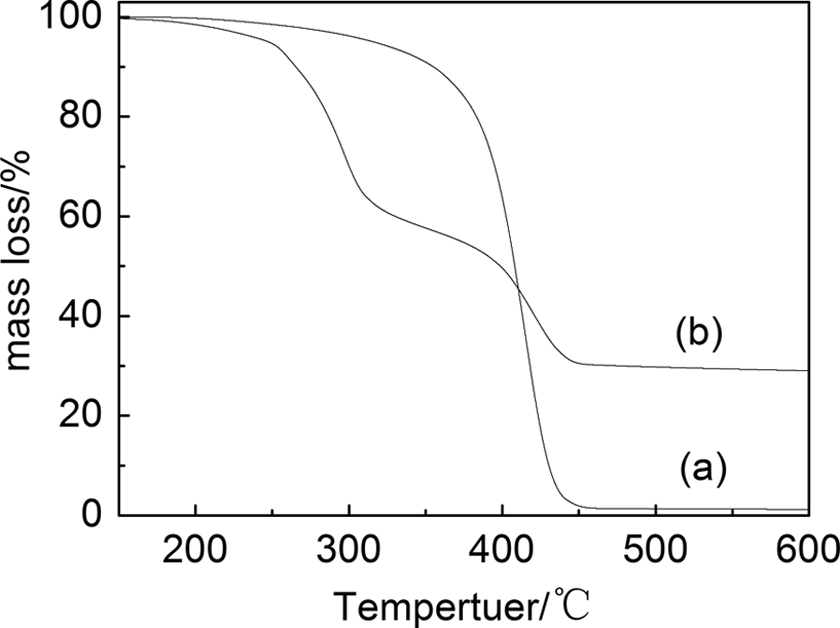
TG curves of (a) P(AAEM-St) and (b) nano-Cu/P(AAEM-St) composites. TG: thermogravimetric; Cu: copper; P(AAEM-St): poly(acetoacetoxyethyl methacrylate–styrene).
Conclusion
The nano-Cu/P(AAEM-St) composites were prepared using an emulsifier-free emulsion polymerization method. TEM displays that Cu NPs were homogeneously dispersed in polymer matrix. UV-Vis, XPS, and FTIR analyses indicated that there was chemical action between Cu NPs and C=O associated with acetoacetoxy from AAEM and St copolymer. TGA proved that the thermal stability of the nano-Cu/P(AAEM-St) composites was lower than that of the pure P(AAEM-St).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Grant No. 51173002), the Natural Science Foundation of Anhui Province, China (Grant No.090414183), and the Doctoral Project of Anhui University of Science and Technology, China.
