Abstract
Poly(amic acid) (PAA) was prepared by the reaction of 4,4′-(hexafluoro isopropylidene) diphthalic anhydride with 2,2′-bis(trifluoromethyl) benzidine in N, N-dimethylacetamide. Hybrid films were obtained from blended solutions of the precursor polymer and saponite (SPT) or organically modified hectorite (STN) clays, and the clay content was varied from 0 to 40 wt%. The cast film of PAA was heat treated at different temperatures to create polyimide (PI) hybrid films. The nanostructure of the hybrid films was observed using transmission electron microscopy, which showed that the clay layers were well dispersed into the matrix polymer, although some clusters or agglomerated particles were also detected. The addition of SPT was more effective than the addition of STN with regard to improving the thermal properties, whereas the addition of STN was more effective with regard to improving the optical transparency and gas barrier characteristics of the PI matrix.
Introduction
Polymeric nanocomposites are a class of composites consisting of ultrafine inorganic particles with sizes in the order of nanometers that are homogeneously dispersed in a polymer matrix. The nanometer scale size of the particles means that nanocomposites possess thermomechanical properties that are superior to those of conventional composites, because the interfacial adhesion is maximized. 1 –5
Clays have sandwich-type structures that typically consist of one octahedral aluminum sheet and tetrahedral silicate (Si) sheets; clays with this typical structure are called phyllosilicates. 6 –8 There are many types of phyllosilicates such as kaolinite, montmorillonite, hectorite, saponite (SPT), and synthetic mica. SPT and organically modified hectorite (STN) were chosen in our study for the synthesis of clay/polymer nanocomposites. SPT has a high swelling capacity, which is essential for efficient intercalation of the polymer, and is composed of stacked Si sheets that provide good thermal, tensile, and molecular barrier properties. Hybrid materials of SPT with various polymers provide the same good properties. 9 –11 STN is a different kind of clay that has been widely used as reinforcing filler in polymeric matrices because of its excellent mechanical, electrical, and thermal properties. 12
In the present study, we prepared hybrid films composed of a polyimide (PI) and an excess of clay (≤40 wt%) and examined their properties as a function of the clay content. We also investigated the effects of clay loading on the optical transparency of the PI hybrid films. The thermal and oxygen barrier properties of the hybrids were also studied, as well as the form of the film as a function of the clay content in the PI matrix polymer.
We also prepared hybrid films composed of a PI and clays and examined their properties as a function of the clay content. The colorless PI was synthesized by performing a thermal cyclization of an aromatic precursor polymer bearing trifluoromethyl-substituted benzene in the side groups. Our approach is based on the reaction of 4,4′-(hexafluoro-isopropylidene) diphthalic anhydride (6FDA) and 2,2′-bis(trifluoromethyl) benzidine (TFB), which are the monomers of a colorless and transparent PI.
To obtain hybrids exhibiting no agglomeration during the in situ intercalation processing, we used the two different clays, SPT and STN. This article examines the effects of these clays on the thermal stability, optical transparency, morphology, and gas barrier of the hybrid films. The properties of the SPT/PI hybrid films were compared with those of the STN/PI hybrid films.
Experimental
Materials
The source clays, SPT and STN, were obtained from Kunimine Industries Co. (Tokyo, Japan). By screening the clays with a 325-mesh sieve to remove impurities, we obtained SPT clay with a cationic exchange capacity of 100 meq/100 g and STN clay with a cationic exchange capacity of 78 meq/100 g. SPT and STN consist of stacked Si sheets with lengths of about 165 nm and 46 nm, respectively. They have the same sheet thickness of 1 nm. 13 –15
All reagents were purchased from either Tokyo Chemical Industry (Tokyo, Japan) or Aldrich Chemical Co. (Seoul, Korea). 6FDA and TFB were obtained from TCI and were used as received. N, N-Dimethylacetamide (DMAc) was purified and dried over molecular sieves before use. Commercially available solvents were purified by distillation, and common reagents were used without further purification.
Preparation of the colorless PI hybrid films
Poly(amic acid) (PAA) was synthesized from 6FDA and TFB in DMAc at a low temperature. TFB (3.8 g; 1.18 × 10–2mol) and DMAc (60 mL) were placed in a 100 mL three-necked flask, and the mixture was stirred at 0°C for 30 min under nitrogen (N2) atmosphere. 6FDA (5.2 g; 1.18 × 10–2mol) in DMAc (50 mL) was then added. The resulting solution was stirred vigorously at 0°C for 1 h and then at room temperature for 14 h, yielding a 13 wt% DMAc solution of PAA.
In the chemical imidization procedure for the PAA solution, pyridine (1.71 g; 2.16 × 10–2mol) and acetic anhydride (2.25 g; 2.16 × 10–2mol) were added to the PAA solution, and the mixture was stirred at 25°C for 30 min and at 80°C for 2 h under a steady stream of N2 gas. Then, 200 mL of methanol was added to the PAA solution to obtain the precipitate.
Because the synthetic procedures used to produce the polymer nanocomposites were the same for all clay contents, we only describe the preparation of PI/SPT with 10 wt% clay as a representative example. SPT (1.0 g) and DMAc (30 mL) were placed in a 100 mL three-necked flask, and the mixture was stirred at 25°C for 2 h. The resulting mixture was ultrasonicated for 5 h to obtain a homogeneously dispersed clay solution. In a separate beaker, PI powder (9 g; 1.18 mol) and DMAc (50 mL) were mixed together and then added dropwise to the SPT/DMAc system under vigorous stirring at 30°C for 1 h to obtain a homogeneously dispersed system. The resulting mixture was subjected to ultrasonication 6 times for 5 min each to obtain a homogeneous composite solution.
The composite solution was cast onto glass plates, and then the solution was stabilized at 25°C for 2 h under air atmosphere and at 50°C for 1 h under a steady stream of N2 gas. To remove the solvent, heat treatments were performed at 80°C for 1 h and then at 100, 130, and 160°C for 30 min at each temperature at a pressure of 1 Torr. The film was further imidized on the glass plate by sequential heating at 190, 220, 250, 280, and 300°C for 30 min at each temperature. Table 1 summarizes the heat treatment conditions employed for the preparation of each hybrid film.
Heat treatment conditions for PAA and PI hybrid films.
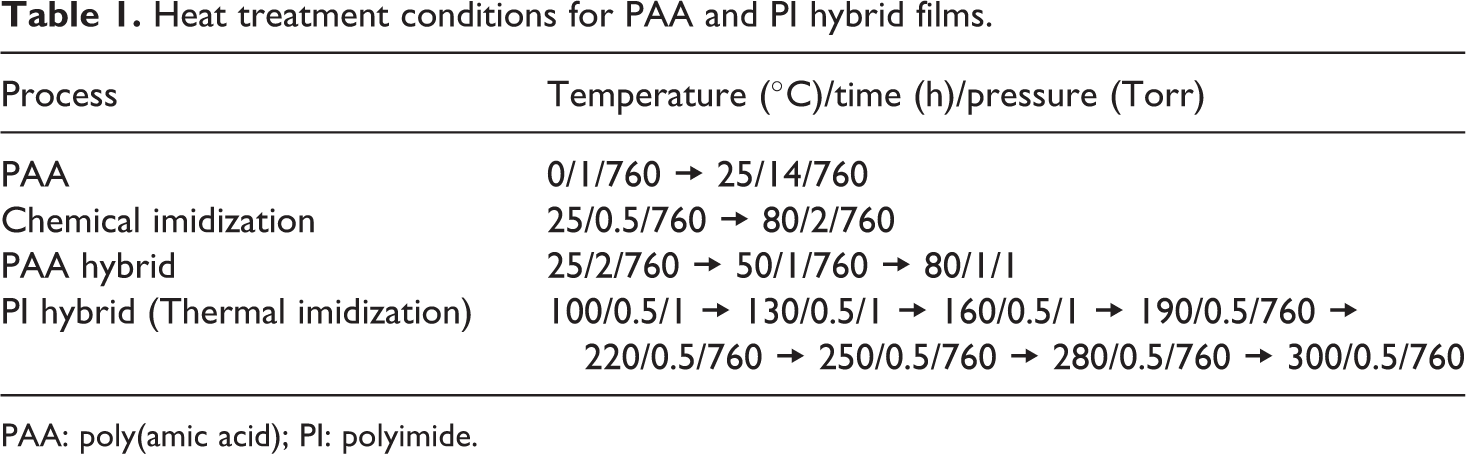
PAA: poly(amic acid); PI: polyimide.
No fixed tools were used to avoid inducing an orientation on the glass plate during heat treatment because orientation can influence some characteristics of film specimens, such as their tensile properties and morphologies. The chemical structures relevant to the synthetic route are shown in Figure 1.
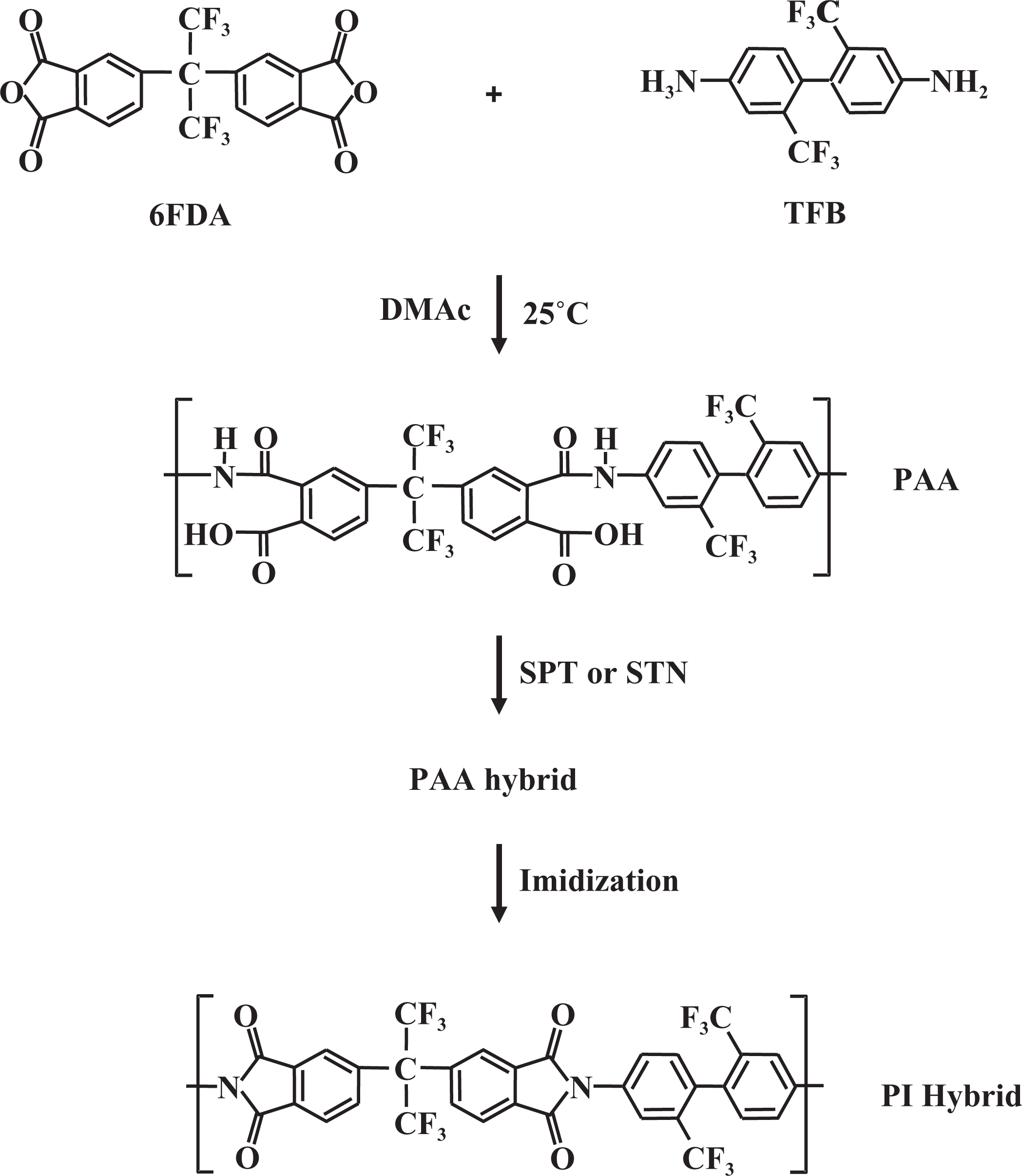
Synthetic route to PI nanocomposite. PI: polyimide.
Characterization
Wide angle x-ray diffraction (XRD) measurements were performed at room temperature on a Rigaku (D/Max-IIIB) x-ray diffractometer using nickel-filtered copper Kα radiation. The scanning rate was 2° min−1 over the range 2θ = 2°–17°. A differential scanning calorimeter (DSC 200F3) was used on a NETZSCH instrument (Germany) and a thermogravimetric analyzer (AutoTGA 1000) was used on a TA instrument (New Castle, Delaware, USA) with a heating rate of 20°C min−1 under N2 flow. Measurement of the coefficient of thermal expansion (CTE) of the samples was performed with a macroexpansion probe (TMA-2940) that applied a 0.1 N expansion force on the film at a heating rate of 5°C min−1 in the temperature range of 40–200°C. Transmission electron microscopy (TEM) photographs of ultrathin sections of the PI hybrid films were obtained with a Leo 912 OMEGA (Finland) transmission electron microscope using an acceleration voltage of 120 kV.
The color intensities of the polymer films were evaluated with a Minolta spectrophotometer (Model CM-3500d, Tokyo, Japan). Measurements were taken using 74- to 87-μm-thick films with an observational angle of 10° and an International Commission on Illumination (CIE-D) illuminant. A CIELAB color difference equation was used. Ultraviolet–visible (UV-Vis) spectra of the polymer films were recorded on a Shimadzu UV-3600 instrument (Tokyo, Japan). The oxygen (O2) permeabilities of the films were measured according to ASTM E96 standard using a Mocon DL 100. O2 transmission rates were obtained at 23°C and a pressure of 1 atm.
Results and discussion
Wide angle XRD
The x-ray diffractograms of pure PI, the clays, and clay/PI hybrid films are shown in Figure 2(a) and (b). The d001 reflection for the SPT occurred at 2θ = 7.56°, which corresponds to an interlayer spacing (d) of 11.71 Å (Figure 2(a)). Only a slight peak at d = 14.17 Å was found in the XRD patterns for the PI films with 5 wt% SPT. Substantial increases in the intensities of the XRD peaks were observed for an increase in the clay loading from 5 wt% to 40 wt%, which suggests that dispersion is more effective at lower clay loadings than at higher clay loadings. Higher clay loadings are expected to result in easier agglomeration of some portion of the clay within the PI matrix. 16,17 The presence of the clay was found, however, to have no effect on the location of the peak, which indicates that no exfoliation of the clay layer structure occurs in this PI system.
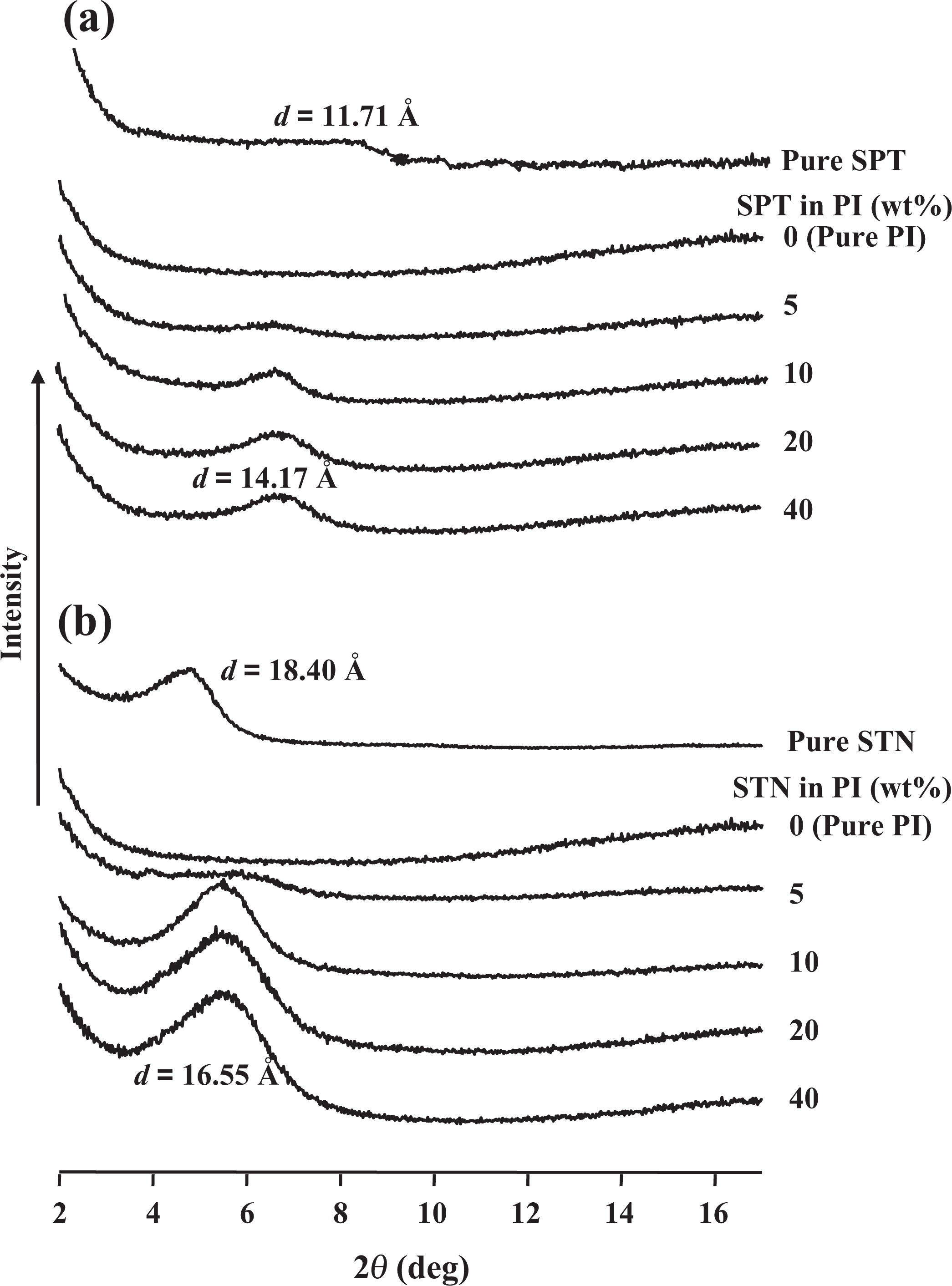
XRD patterns of pure clays and PI hybrids with various clay contents. XRD: x-ray diffraction; PI: polyimide.
`In the case of STN (Figure 2(b)), the d001 reflection occurred at 2θ = 4.74°, which corresponds to a d of 18.40 Å. Similar results were observed for the STN hybrid films; obvious clay peaks appeared in the XRD curves of the PI hybrid films with 5–40 wt% STN, which indicates that the clay layers were intercalated (not exfoliated) and not homogeneously dispersed in the PI matrix. In contrast to the hybrids with low clay contents, clays with higher loadings (≥10 wt%) in the PI matrix exhibited strong diffraction peaks corresponding to the basal spacing at 2θ = 5.48°, corresponding to an interlayer distance of 16.55 Å. This suggests that the degree of clay aggregation increases with increasing clay content.
Yano et al. 14 reported that organic molecules in the clay interlayers that interact only weakly with the clay might detach from the clay surface. This detachment by heat treatment occurs during the cyclization process from PAA to PI and causes a reduction in the interlayer spacing from the basal spacing of the clay (from 18.40 Å to 16.55 Å). This squeezing mechanism should also serve to inhibit exfoliation of the clay in the PI, causing the clay to have an intercalated layer structure.
Morphology
Although XRD allows for precise, routine measurements of clay layer spacing (1–4 nm), little can be said about the spatial distribution of the clay layers or any structural inhomogeneities in the hybrids. Additionally, some layered clays do not initially exhibit well-defined basal reflections. Thus, peak broadening and intensity decreases are difficult to follow systematically. Therefore, conclusions concerning mechanisms of hybrid formation and hybrid microstructure based solely on XRD results are only tentative. Hence, we extended this analysis using TEM to evaluate the degree of intercalation and the amount of aggregation of the clay clusters. 18,19
More direct evidence for the formation of a true nanocomposite can be provided by TEM analysis of ultramicrotomed sections. Figures 3 and 4 show the micrographs of PI hybrid films with the same contents of the two different clays. The dark lines in the photographs are the intersections between the 1-nm-thick clay sheets, while the spaces between the dark lines are the interlayer spaces. Figure 3(a) shows the morphology of a PI hybrid containing 20 wt% SPT. In this sample, each layer in the polymer matrix consists of well-dispersed individual clay layers, apparent as dark lines, although some of the clays agglomerated to thicknesses of around 10 nm. As in the case of the hybrid with 40 wt% SPT (see Figure 3(b)), these clays were for the most part agglomerated in the polymer matrix, but the average particle size was found to be below 20 nm in thickness, as calculated from the TEM photographs.

TEM micrographs of PI hybrid films containing (a) 20 wt% and (b) 40 wt% SPT. TEM: transmission electron microscopic; PI: polyimide; SPT: saponite.

TEM micrographs of PI hybrid films containing (a) 20 wt% and (b) 40 wt% STN. TEM: transmission electron microscopic; PI: polyimide; STN: organically modified hectorite.
Typical TEM photographs of the PI hybrid films containing 20 and 40 wt% STN are shown in Figure 4(a) and (b), respectively. Figure 4 shows that the clay was well dispersed in the polymer matrix at all magnification levels and some particles were observed in agglomerations larger than approximately 20 nm. The presence of the peaks in the XRD patterns of these samples can be attributed to these agglomerated layers (see Figure 2(b)). For the 40 wt% hybrid films (Figure 4(b)), all of the clay appeared in agglomerations larger than approximately 20 nm. Unlike those in the hybrids containing SPT, the clay layers of the STN hybrids were agglomerated inside the matrix polymer. This was also confirmed by XRD, as shown in Figure 2. This difference in the clay dispersion might be attributed to the similarity between the chemical structure of the modified clay and the chemical structure of the polymer matrix and to the interactions between the clay and the polymer matrix.
The XRD and TEM results indicate that the clay particles were well dispersed throughout the PI matrix at low clay contents, whereas agglomerated structures were evident at higher clay contents. The unusual thermo-optical properties of these nanocomposites are discussed in the following sections with reference to the homogeneous dispersion of the nanoscale Si particles.
Thermal behavior
Comparative DSC results for pure PI and the PI hybrids with 0–40 wt% SPT are listed in Table 2. The glass transition temperatures (Tg) of the PI hybrids increase from 291 to 305°C with increasing clay loadings from 0 wt% to 20 wt%. The increase in the Tg of these hybrids could be the result of two factors. First, the effect of small amounts of dispersed clay layers on the free volume of PI is significant and does influence the Tg of PI hybrids. The second factor is ascribed to the confinement of the intercalated polymer chains within the clay galleries, which prevents segmental motions of the polymer chains. 7,20,21 However, the maximum transition peaks of the PI hybrids increase upon the addition of clay up to a critical content and then decrease with a further increase in the loading. For example, when the SPT content in PI reaches 40 wt%, Tg is 293°C. This decrease in Tg seems to be the result of the clay agglomeration that occurs upon the addition of clay into the polymer matrix above the critical clay loading. The presence of clay agglomeration in PI was already confirmed using XRD (see Figure 2) and TEM (see Figure 3). Otherwise, the Tg values of the PI hybrid films with STN decreases gradually from 291 to 260°C with increasing clay loadings from 0 wt% to 40 wt%. This decrease in Tg is due to the low heat resistance of the alkyl chains in the organic content in the chemically modified STN.
Thermal properties of PI hybrid films.
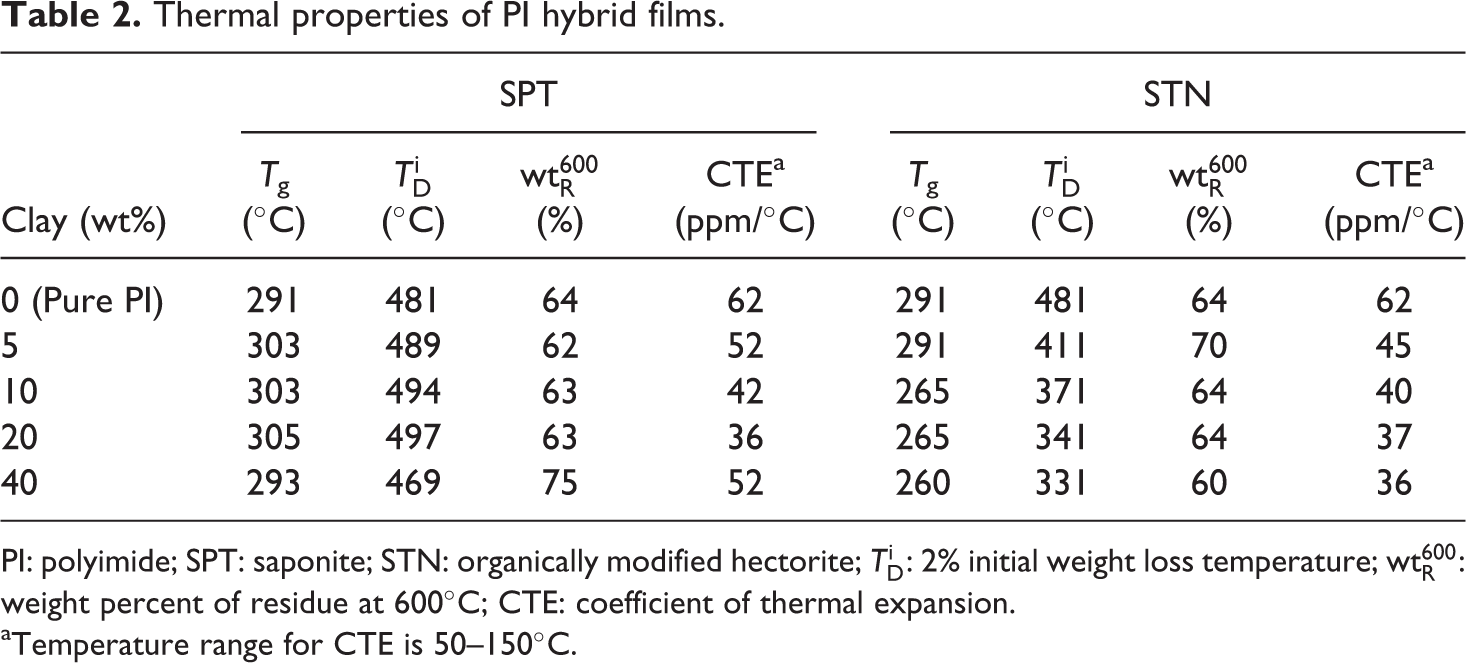
PI: polyimide; SPT: saponite; STN: organically modified hectorite;
aTemperature range for CTE is 50–150°C.
Comparative TGA results for pure PI and the PI hybrids with 0–40 wt% SPT are also listed in Table 2. These results indicate that the initial thermal degradation temperature
Table 2 also lists the thermal stabilities of the STN hybrid films for various clay contents. The
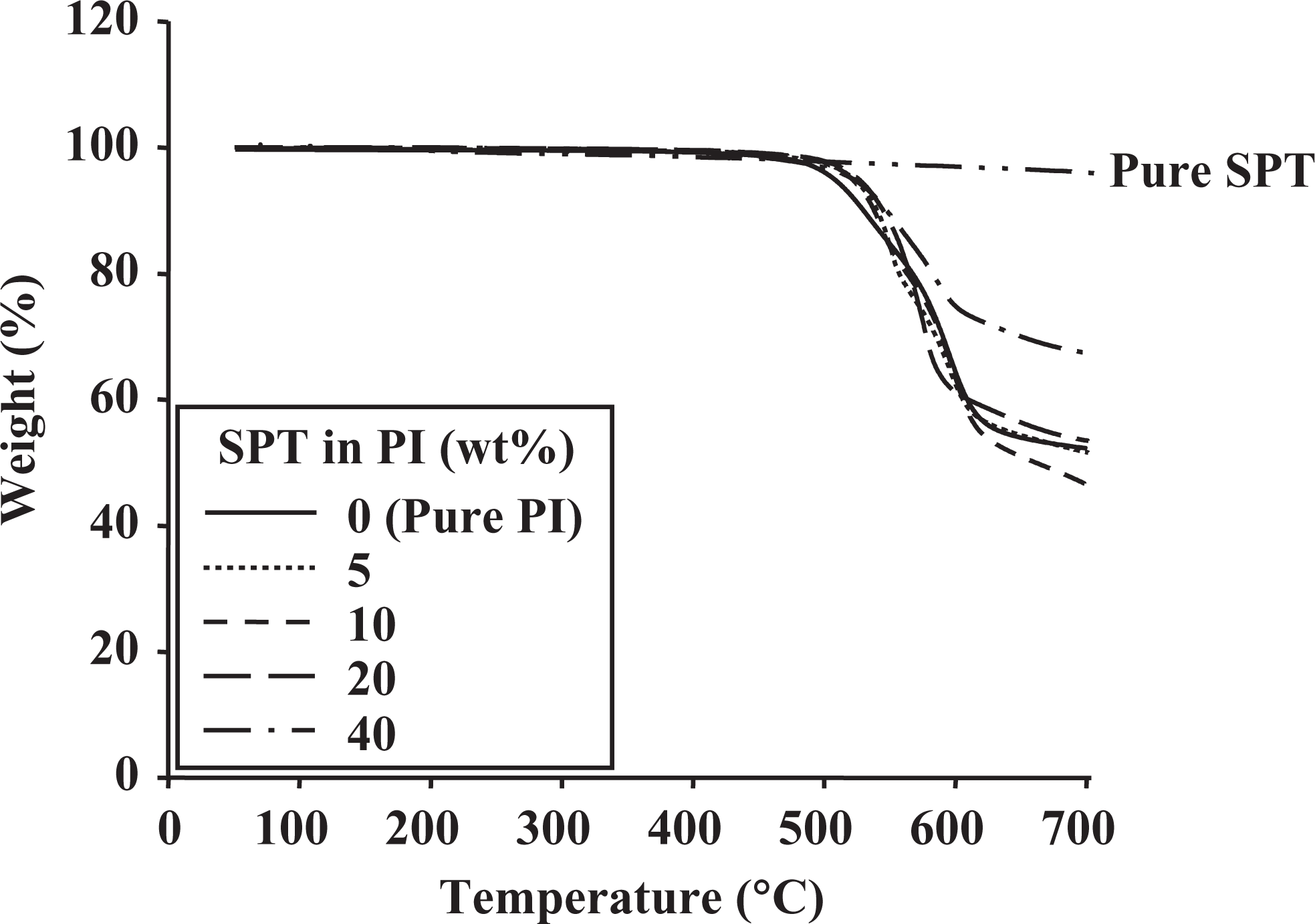
TGA thermograms of pure clay and PI hybrids with various SPT contents. TGA: thermogravimetric analysis; PI: polyimide; SPT: saponite.
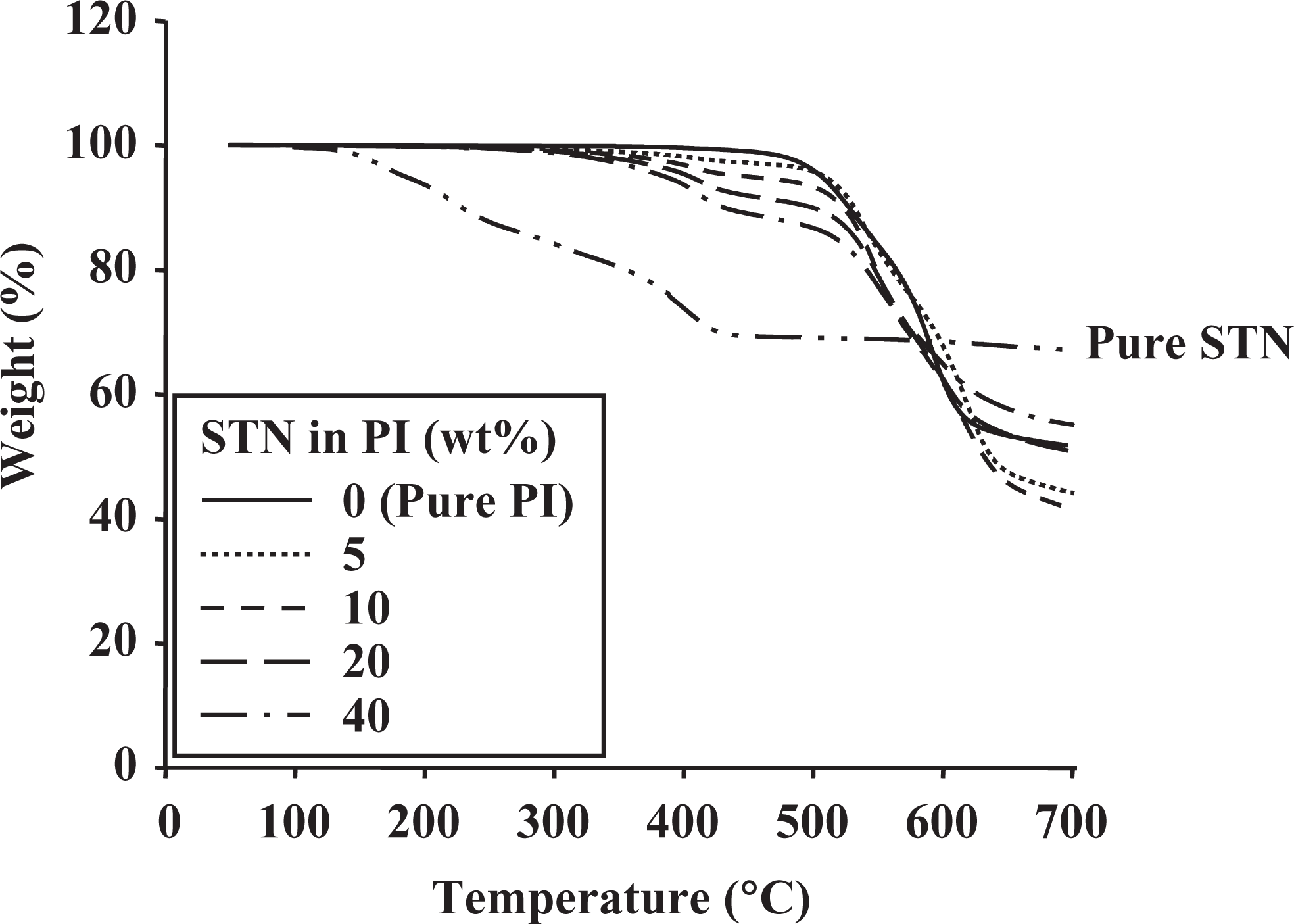
TGA thermograms of pure clay and PI hybrids with various STN contents. TGA: thermogravimetric analysis; PI: polyimide; STN: organically modified hectorite.
The CTE of the SPT hybrid films shows a trend similar to those of Tg and
The values of the CTE for the STN/PI hybrids in the temperature range of 50–150°C are also listed in Table 2. In contrast to the results for the SPT hybrids, the CTEs of the STN hybrids decreased with increasing clay loadings up to 40 wt%. For example, the CTEs of the PI hybrids decreased from 62 ppm/°C to 36 ppm/°C when the clay loading increased from 0 wt% to 40 wt%. This means that the magnitude of the reduction of the thermal expansion due to the clay layers depends on the orientation of the PI molecules and the rigid nature of the clay layers. Upon heating, the in-plane-oriented PI molecules tend to relax in a direction normal to their original direction and therefore expand mainly in the out-of-plane direction. 27,28 The clay layers are much more rigid than the PI molecules, and they do not deform or relax as easily as the PI molecules. Consequently, the clay layers very effectively retard the thermal expansion of the PI molecules in the out-of-plane direction.
Optical transparency
The color intensities of the hybrid films can be elucidated by measuring the cutoff wavelength (λ0) in the UV-Vis absorption spectra. The color intensities of the PI hybrid films with various clay contents are listed in Table 3. The λ0 values of the PI hybrids were virtually unchanged when the clay loading was varied; specifically, they increased from 362 nm to 369 nm for SPT and from 362 nm to 366 nm for STN as the clay content increased from 0 wt% to 40 wt%. The UV-Vis absorption spectra for the PI hybrid films with various amounts of clays are shown in Figures 7 and 8.
Optical properties of PI hybrid films.
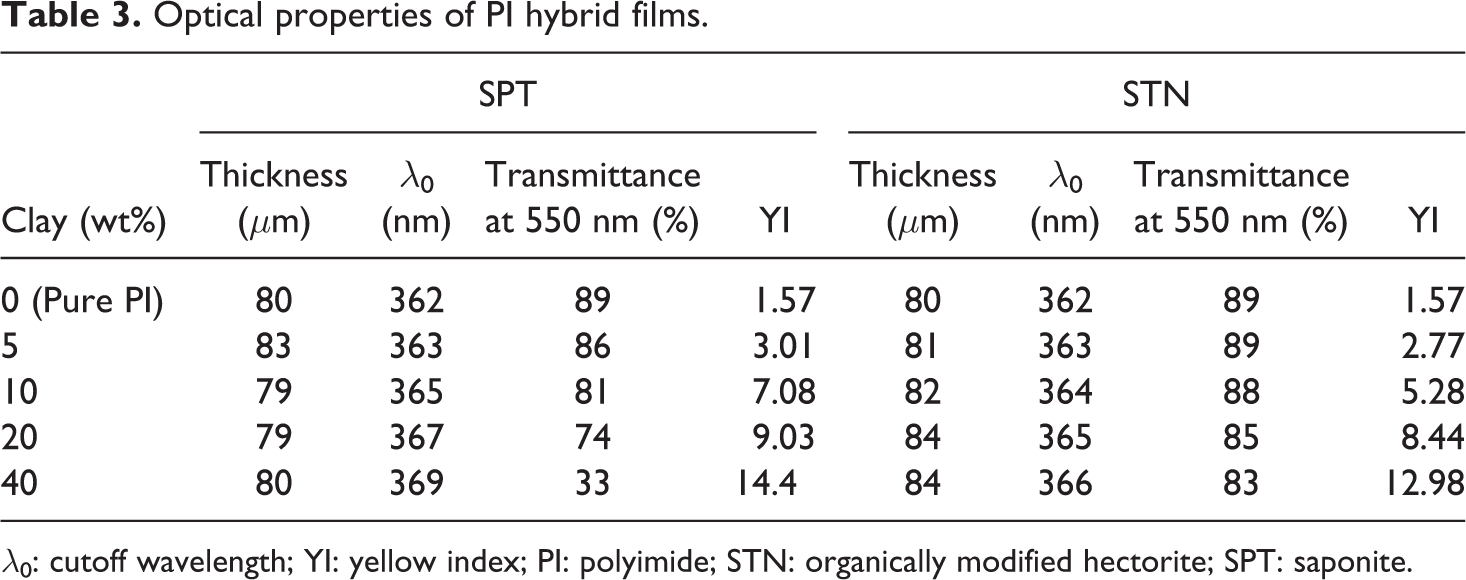
λ0: cutoff wavelength; YI: yellow index; PI: polyimide; STN: organically modified hectorite; SPT: saponite.
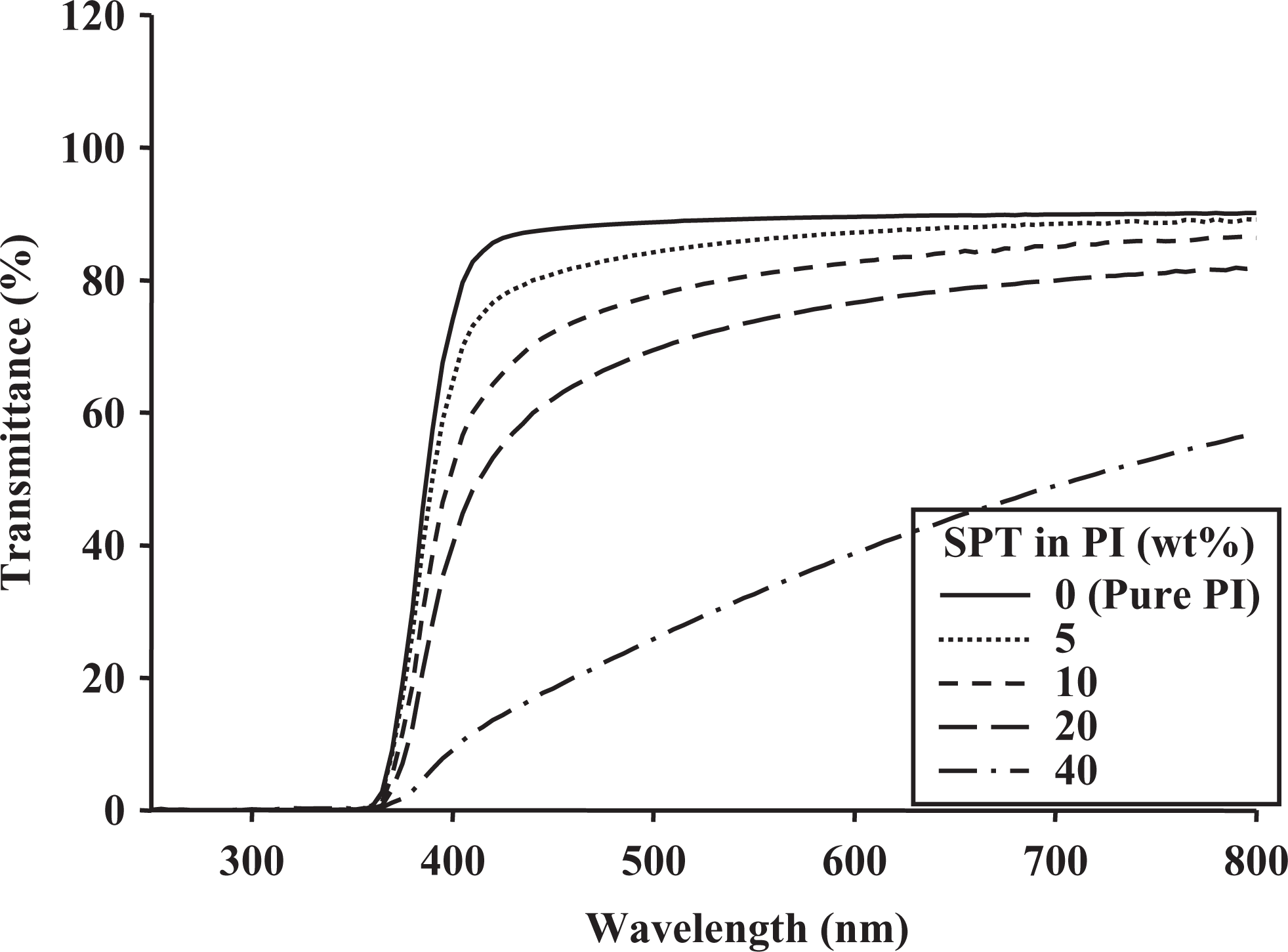
UV-Vis transmittance of PI hybrid films with various SPT contents. UV-Vis: ultraviolet–visible; PI: polyimide; SPT: saponite.
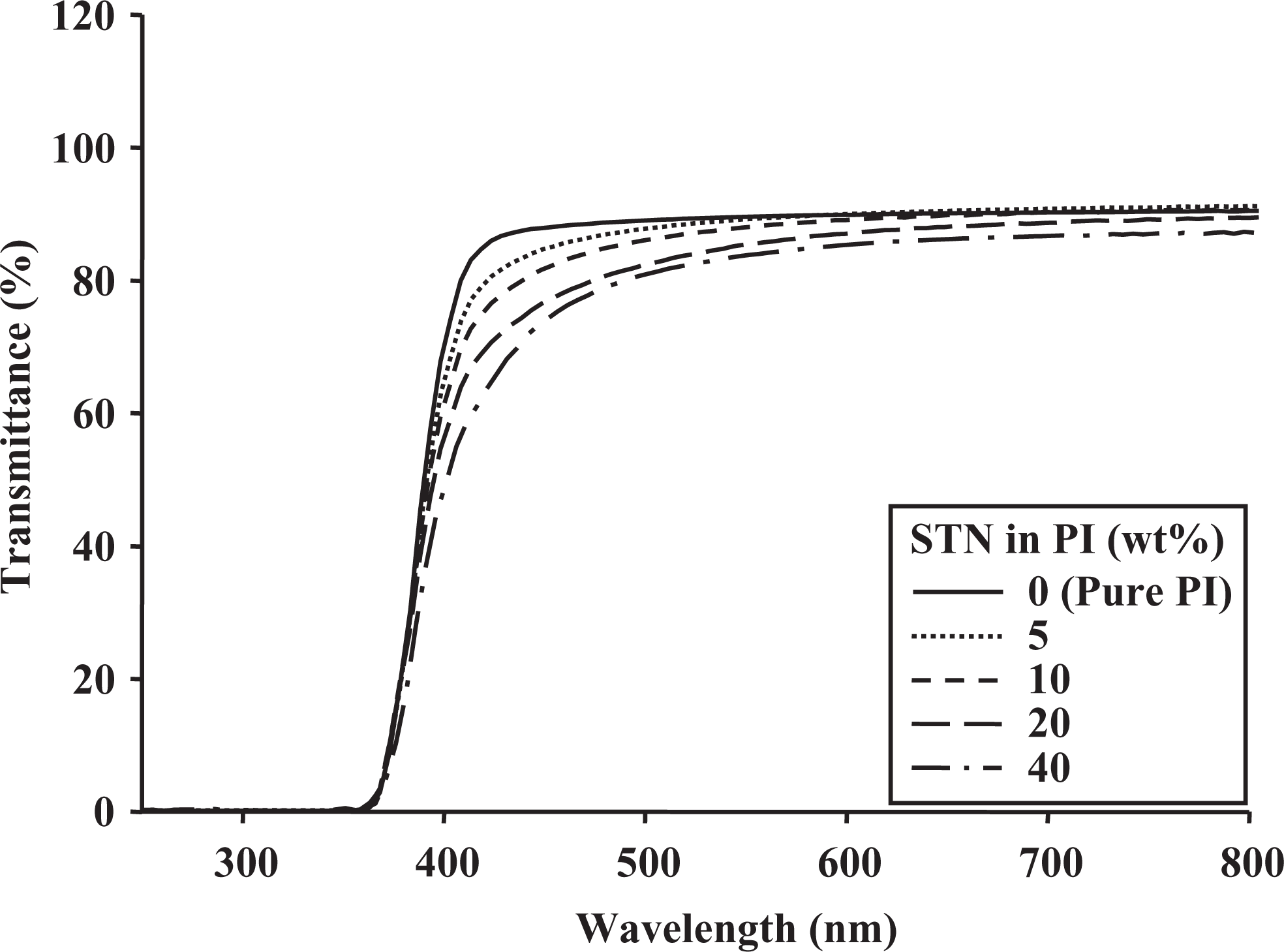
UV-Vis transmittance of PI hybrid films with various STN contents. UV-Vis: ultraviolet–visible; PI: polyimide; STN: organically modified hectorite.
The transmittances of the PI hybrid films decreased when the clay content was increased because of the agglomeration of the clay particles. The transmittance at 550 nm decreased significantly from 89% to 33% as the SPT content increased from 0 wt% to 40 wt% (Table 3). The evidence for clay agglomeration was obtained using TEM, as shown in Figures 3 and 4. Compared with those of the SPT hybrid films, the transmittances of the STN hybrid films at 550 nm showed excellent optical transparency, as shown in Table 3. These colorless STN hybrid films had maximum UV transmittances of almost 80% at 550 nm and excellent optical properties.
Table 3 also shows that the color intensity of the PI hybrid films was affected by the clay content. The PI hybrid films with lower clay contents had lower yellow index (YI) values than did the corresponding hybrids with higher clay contents. The YI value of the PI hybrid with 0 wt% SPT (pure PI) was 1.57, and its degree of colorlessness was almost the same as that of aliphatic poly(methyl methacrylate) (YI = approximately 1.50). 29 When the clay loading was increased to 5 wt% and then to 40 wt%, significant increase in the YI value to 3.01 and then to 14.40, respectively, were observed because of the agglomeration of the clay particles. This increase in the YI suggests that the clay particles are better dispersed in the polymer matrix at lower clay loadings.
Table 3 also shows that the YI values of the PI hybrid films were affected by their STN content and that the PI hybrid films with lower STN contents have lower YI values than the hybrids with higher STN contents. The YI value of the PI hybrid with 5 wt% STN was 2.77; when the clay loading was increased to 20 wt% and then to 40 wt%, the YI value increased to 8.44 and then to 12.98, respectively. However, these values of YI were much lower than those of conventional aromatic PI films produced by Dupont, such as Kapton® 200KN (YI = 97.50). 30
All the solvent-cast hybrid films with clay contents in the range of 0–40 wt% were almost colorless, as shown in Figures 9 and 10. However, their optical transparencies decreased slightly when the clay content was increased. The clay hybrid film containing 20 wt% clay was slightly cloudier than the films containing 5–10 wt% clay, but the optical transparency was still good, as there was no problem in reading a letter through the film. The levels of transparency were not significantly affected by increase in the clay content from 5 wt% to 40 wt%, as shown in Figures 9 and 10. Therefore, there were only small differences between the optical transparencies of the samples.
Photographs of PI hybrid films containing (a) 0 wt% SPT (pure PI), (b) 5 wt% SPT, (c) 10 wt% SPT, (d) 20 wt% SPT, and (e) 40 wt% SPT. PI: polyimide; SPT: saponite.
Photographs of PI hybrid films containing. (a) 0 wt% SPT (pure PI), (b) 5 wt% SPT, (c) 10 wt% SPT, (d) 20 wt% SPT, and (e) 40 wt% STN. PI: polyimide; STN: organically modified hectorite.
Gas permeability
The introduction of an organic material into the polymer films affects their gas permeabilities. In general, the gas permeabilities of hybrid films are lower than those of polymer films and are independent of the type of gas. This behavior is attributed to the high aspect ratio and rigidity of the clay platelets in the polymer matrix. 31 –34
Models for the gas permeability of polymer composites must consider the shape and spatial orientation of the dispersed phase. The permeabilities of composites consisting of filler particles dispersed in a polymer matrix can be predicted by the Maxwell model. 35 The platelet morphology of the clays in nanocomposites significantly reduces the gas permeability, more so than the spherical droplet morphology. In this case, the clay is a mineral consisting of stacked Si sheets measuring 1 nm in thickness and about 100–2000 nm in length. It possesses a high aspect ratio and a platy morphology.
The mobility of the polymer chain segments in the polymer hybrid is obviously different from that in the pure polymer because of the confined geometry, which affects the gas permeability. There are two main factors responsible for the reduction in permeability, namely, the reduction in polymer chain segment mobility and the detour ratio, which is defined as the ratio of the film thickness in the nominal diffusion flow direction to the average length of the tortuous diffusion distance between the clay layers. 36,37
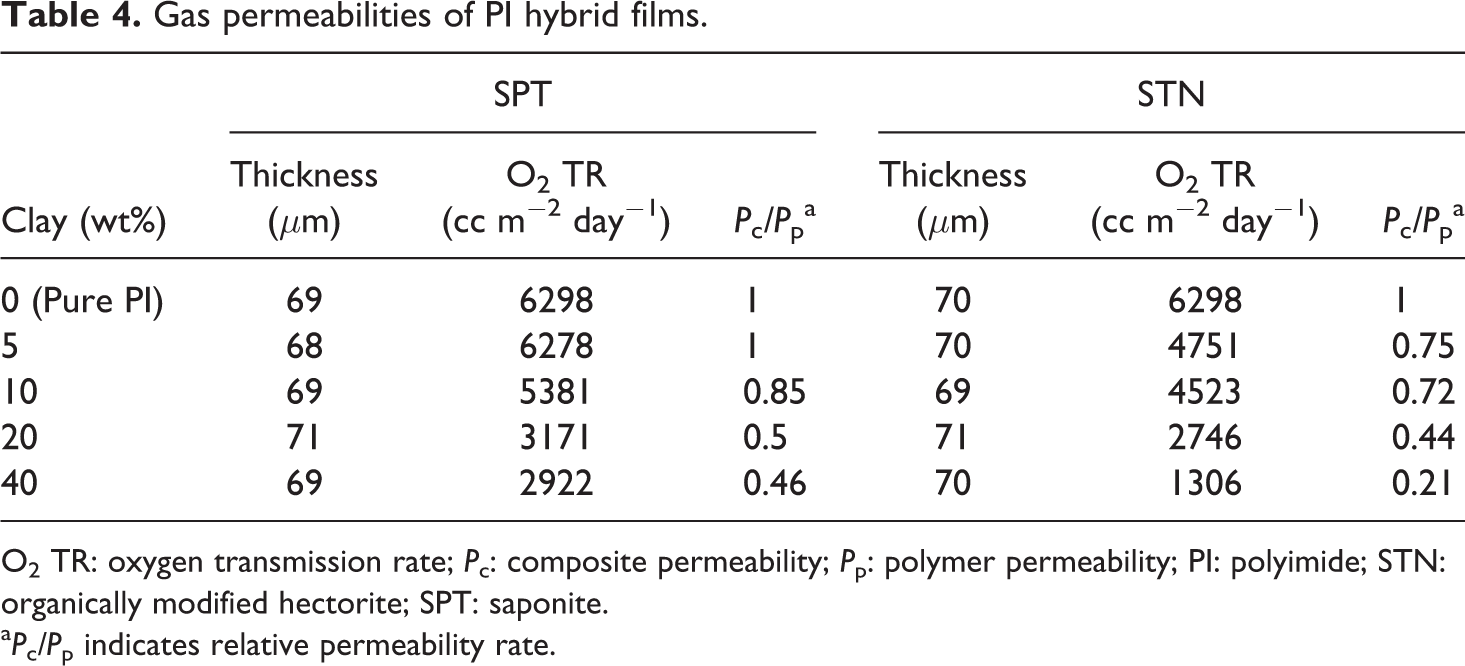
To further characterize the barrier properties of the PI/clay hybrids fabricated by intercalating polymer chains in the galleries of SPT and STN, the permeability of the resultant PI/clay hybrids to O2 was evaluated. The clay loading levels ranged from 0 wt% to 40 wt%. The measured values are summarized in Table 4. The permeability rates of SPT and STN decreased monotonically with increasing clay loading from 0 wt% to 40 wt%: from 6298 cc m−2 day−1 to 2922 cc m−2 day−1 for SPT and from 6298 cc m−2 day−1 to 1306 cc m−2 day−1 for STN. For example, the addition of 20 wt% SPT brings the relative permeability rate of O2 to a value that is half that of pure PI. When 20 wt% STN was loaded, the O2 permeability of the nanocomposite also decreased to less than half of that of the pure PI; at all clay loadings, the O2 permeability of the STN hybrid films was lower than that of the SPT hybrid films. This result is due to the increase in the lengths of the tortuous paths followed by the gas molecules and to the interaction between the oxygen gas and the organically modified clay molecules. 38 –40 Furthermore, films containing higher amounts of clay seem to be much more rigid, and this contributes to a decrease in their gas permeability.
Gas permeabilities of PI hybrid films.
O2 TR: oxygen transmission rate; Pc: composite permeability; Pp: polymer permeability; PI: polyimide; STN: organically modified hectorite; SPT: saponite.
aPc/Pp indicates relative permeability rate.
Conclusion
The thermo-optical properties and morphologies were determined for colorless and transparent PI hybrid films comprising two types of clay, SPT and STN. The clay contents of the hybrid films were varied from 5 wt% to 40 wt%, and the effects on the thermo-optical properties, morphologies, and gas permeabilities of the films were examined. We found that the thermo-optical properties and gas permeabilities were dependent on both the type and amount of clay in the PI matrix.
The thermal properties (Tg,
Footnotes
Declaration of Conflicting Interests
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
