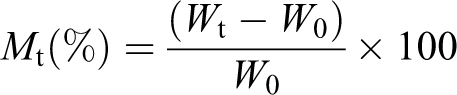Abstract
Leather shavings, the by-products of leather industry, are usually treated by the landfill, which probably makes them hazardous to the human health and the environment due to the oxidation of the containing chromium (Cr) (III) to toxic Cr (VI). Therefore, efficient and environment-friendly reusing of leather shavings is not only of great interest but is also a huge challenge. This article reported a reuse method of leather shavings by combing two novel technologies, that is, the solid state shear milling (S3M) to mechanochemically pulverize and activate leather shavings, and the thermal processing of poly (vinyl alcohol) (PVA) to make them filled with PVA. The related mechanism, the structure, and properties of the obtained composites were investigated. The results showed that S3M method could efficiently pulverize and activate leather shavings through very strong shearing and compressing forces, promoting the formation of more hydrogen bonds and the chelation in the interfaces between PVA and leather shavings, and consequently enhancing their compatibility. In this way, PVA/leather shaving composites with pretty good performances, for example, better mechanical properties, thermal stability, and water resistance as compared with neat PVA, were obtained. This is a convenient, cost-efficient, and environment-friendly technology to recycle hazardous chrome-containing leather shavings.
Introduction
Leathers, made from animal skins by the tanning industry, are extensively applied and demanded in human daily life. As the by-products of the tanning industry, the leather shavings are also produced in a great amount. It was reported that processing 1 ton raw hide would generate about 200 kg final leather products, 250 kg nontanned solid wastes, and 200 kg chrome-tanned wastes with 3 kg chrome, 1 which are very difficult to be dealt with. The leather shavings are genuine leather but can hardly be reused due to their shapes and the cross-linking features and usually have to be disposed by the landfill, which not only squanders the luxury leather material but also very hazardous to the human health and the environment due to the oxidation of their contained chromium (Cr) (III) to toxic Cr (VI). 2 Therefore, efficient and environment-friendly recycling of leather shavings is not only of great importance but is also a huge challenge worldwide.
Great efforts have been devoted to treat the solid chrome-tanned leather shavings. Taylor and colleagues 3 –6 separated the protein-bound chrome from each other and reused the protein and chrome fractions by a multistep process: first, to get a gelable protein by alkali enzymes and then, to get a collagen hydrolysate and chrome cake. However, this process consumed a lot of chemicals and water and inevitably caused a second pollution. El-sabbagh and Mohamed 7 and Przepiorkowska and colleagues 8 –12 used the solid chrome-tanned leather waste as the filler for rubbers to improve their biodegradable property, thermal aging property, and mechanical properties. Santana and colleagues 13,14 and Tarantili and colleagues 15,16 used the solid-tanned leather waste to improve the properties of poly (vinyl chloride) (PVC)-based composites after surface chemical treatment. Mohamed et al. 17 mixed it with polyamide to enhance the properties of the materials. Obviously, using the leather shavings as the organic fillers for polymers is a direct and physical utilization method, more convenient, and cleaner than the chemical methods. The key issue is the good dispersion of the leather shavings in the polymer matrix, which demands the fine particle size and narrow particle size distribution, as well as the good compatibility between the shavings and the polymers.
In order to effectively and environment-friendly reuse the leather shavings, this article reported a new leather like composites, that is, leather shavings-filled poly (vinyl alcohol) (PVA) by combining of two novel technologies that were developed by our research group, that is, solid state shear milling (S3M) and thermal processing of PVA. The S3M is based on our self-designed mechanochemical reactor, 18 which acts as the three-dimensional scissors, offering strong shear force in both radial and tangential directions and squeezing force in normal direction, has excellent pulverizing, mixing, and activating effect on the materials in-between. 19,20 Taking the advantage of the S3M, the leather shavings can be effectively pulverized and activated, promoting the hydrogen bonding and chelating the leather shavings with PVA, therefore enhancing their compatibility.
PVA is a special commodity polymer with multihydroxyl groups, which endows PVA with polarity and good compatibility with other organic and/or inorganic materials, such as cellulose, clay, and silica. 21 –23 Therefore, theoretically, solid chrome-tanned wastes can be filled in PVA to form a compatible system through the interactions between their polar groups. However, it is difficult to prepare PVA-based composites with high performances because the melting point of PVA is very close to its decomposition temperature. The conventional methods of processing PVA are based on the solution method, which limits the application of PVA. Through the molecular complexation and the plasticization, our research group has successfully realized the thermal processing of PVA and prepared some PVA products accordingly, including melt-spun fibers, thermal blown films, PVA containers, and so on, 24 –26 which provides the possibility to prepare the new leather shavings-filled PVA composites by thermal processing. The pulverization and activation of the leather shavings by solid shear milling, the compatibility between the leather shavings and PVA, the thermal processing of pan-milled leather shavings-filled PVA materials, and the structure and properties of the new leather shaving/PVA composites were investigated in this article.
Experimental
Materials
PVA1799 (polymerization degree: 1750 ± 50, hydrolysis degree: 99.9%) was purchased from the SINOPEC Sichuan Vinylon Works (Chongqing, China). Leather shavings were provided by the National Engineering Laboratory for Clean Technology of Leather Manufacture, Sichuan University, Sichuan, China. Deionized water was used throughout the experiment.
Sample preparation
Leather shavings were first washed with deionized water and then dried at 80°C for 12 h. The dried leather shavings were further pulverized using the pan mill mechanochemical reactor. 18 Figure 1 shows the schematic drawing of the pan mill mechanochemical reactor. The rotation speed of the pan mill was fixed at 30 r min−1. The milled particles were discharged from the brim of the pan and collected for the next milling cycle. The process was conducted at room temperature and the heat generated during pan milling was removed by cooling water. The milled leather shavings were sampled for particle size measurement at every a certain number of milling cycles. The pan-milled leather shavings were named as PMCS. The modified PVA was prepared according to the literature. 25 The modified PVA and PMCS in different component ratios were first mixed in Brabender mixer (Germany) and then blended in a Hakke 90 extruder (Germany) with screw of 20 mm in diameter and length/diameter = 25 at 130°C and 25 r min−1 of screw rotation speed. Then, the extrudates were compression molded into testing samples with 200 mm diameter and 2 mm thickness under pressure of 10 MPa at 130°C for 13 min. Before analysis, the obtained composite samples were dried at 80°C for 6 h and then conditioned in desiccators for 2 weeks so as to prepare the highly dehydrated materials.
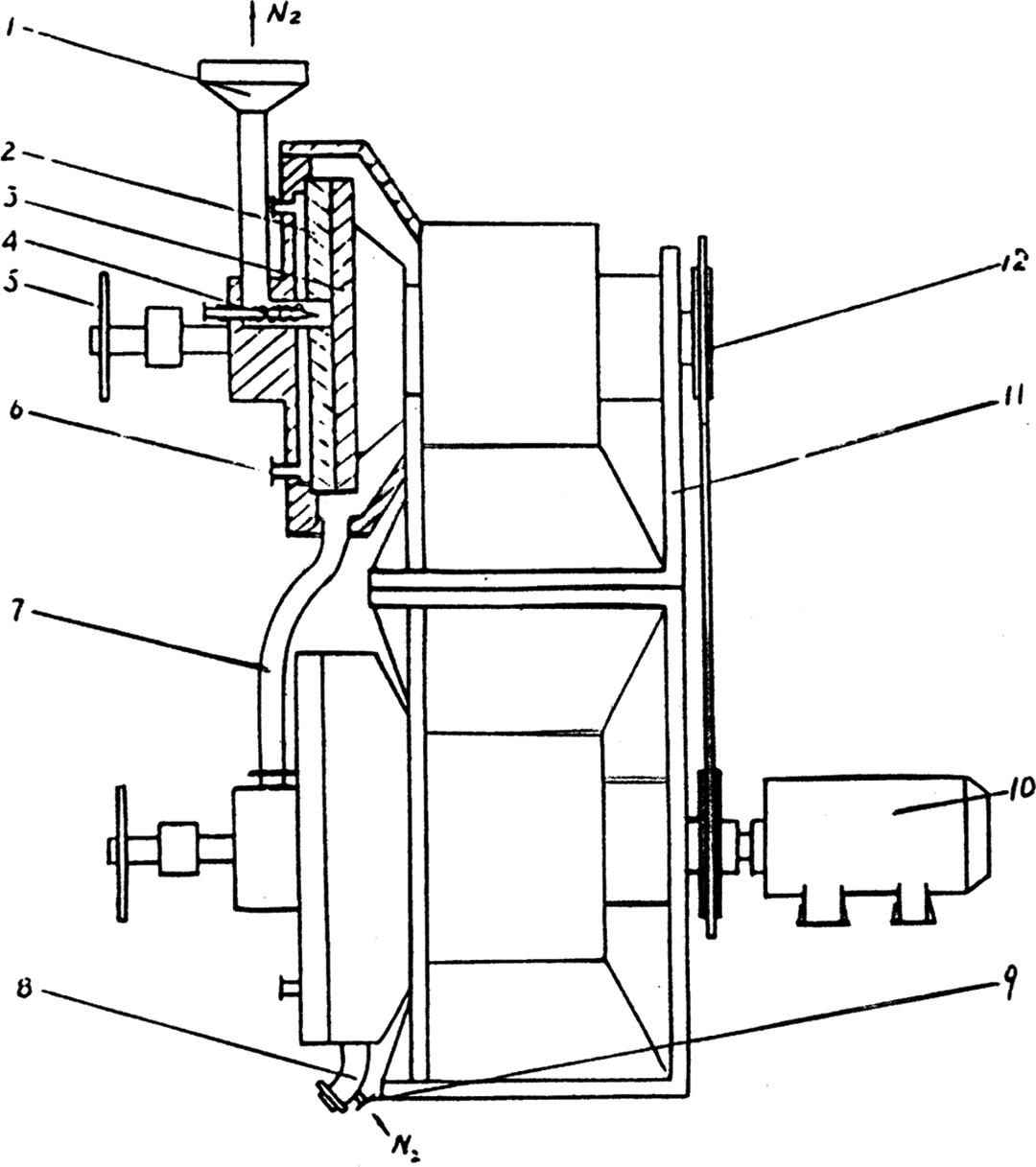
Schematic diagram of the pan mill mechanochemical reactor (1: inlet; 2: stationary pan; 3: moving pan; 4: feeding screw; 5: helve; 6: entrance of medium; 7: soft tube; 8: outlet; 9: entrance of inert gas; 10: motor; 11: stand; 12: drive system).
Size and specific surface area measurement
The average particle size and particle size distribution of PMCS were measured by Masterizer 2000 laser particle analyzer (Malvern Instrument, UK). The sample was first dispersed in alcohol and then stirred at a pumping speed of 2400 r min−1. The data were collected after ultrasonically breaking up. Then, the particle size and particle size distribution were calculated based on the light scattering pattern. Simultaneously, the specific surface area of the samples was estimated.
SEM observation
The morphology of the samples was observed using an Inspect (FEI, Japan) scanning electron microscopy (SEM) instrument. The samples had been cryogenically fractured in liquid nitrogen and then coated with gold before observation.
FTIR analysis
Fourier transform infrared (FTIR) spectra of the samples were recorded using an FTIR spectrometer (Nexus 680, Thermo Nicolet Corporation, Madison, Wisconsin, USA) in the range of 400 to 4000 cm−1.
DSC analysis
The melting behavior of the samples was recorded on a TA-Q20 differential scanning calorimeter (TA instruments, New Castle, Delaware, USA). The samples (approximately 5 mg) were sealed in aluminum pans with lids. The scanning just starts from −20 to 240°C at a rate of 10°C min−1 under nitrogen atmosphere flow of 80 ml min−1. The crystallinity of the samples was calculated by taking 168 J g−1 as melting enthalpy for the 100% crystalline PVA.
Thermogravimetric analysis
The thermogravimetric analysis (TGA) was performed on a TA-Q50 (TA instruments, New Castle, Delaware, USA). The sample (approximately 5 mg) was loaded in a tared platinum pan and heated from 30 to 600°C at a scanning rate of 10°C min−1 under nitrogen atmosphere flow of 100 ml min−1.
Mechanical properties
Tensile strength (TS) and elongation (EL) of each sample were measured using Instron 4302 tensile tester (Canton, Massachusetts, USA) according to ASTM D638 standard. Each reported value was an average of five measurements.
Water absorption test
The samples with dimensions of 20 × 20 × 2 mm3 were used to examine water absorption behavior. First, the samples were immersed in deionized water (25°C) and then periodically taken out of the water. The excess water on the sample surface was wiped by tissue paper and then the specimens were weighed. The water absorption rate (Mt) was calculated using the following equation:
Results and discussion
Structural characteristics of PMCS
The interactions between fillers and polymer matrix and the dispersion of fillers in polymer matrix play an important role in determining the properties of the composites. In order to promote the interactions between leather shavings and PVA and thus improve the dispersion of leather shavings in PVA matrix, it is necessary to change their submicroscopic collagen fiber structure, for example, decreasing their particle size to increase their specific surface area. As mentioned previously, the pan mill mechanochemical reactor has excellent mechanochemical pulverization, dispersion, mixing, and activation effects on polymer materials. 19,20,27,28 Hence, it is expected to be applied in the mechanochemical pulverization and activation of leather shavings. Figure 2 shows the size distribution curves of PMCS, and the corresponding parameters are listed in Table 1. Obviously, the particle size of PMCS reduced under the strong shearing and compressing force field of pan mill mechnochemical reactor. The more the milling cycles, the smaller the PMCS particle size, leading to the shift of the size distribution curves of PMCS to the smaller particle size zone. This indicates that the bundles of collagen fibers are effectively separated and broke into small particles, which could produce the larger surface area at the incision edges. This can also be confirmed using SEM micrographs of PMCS.
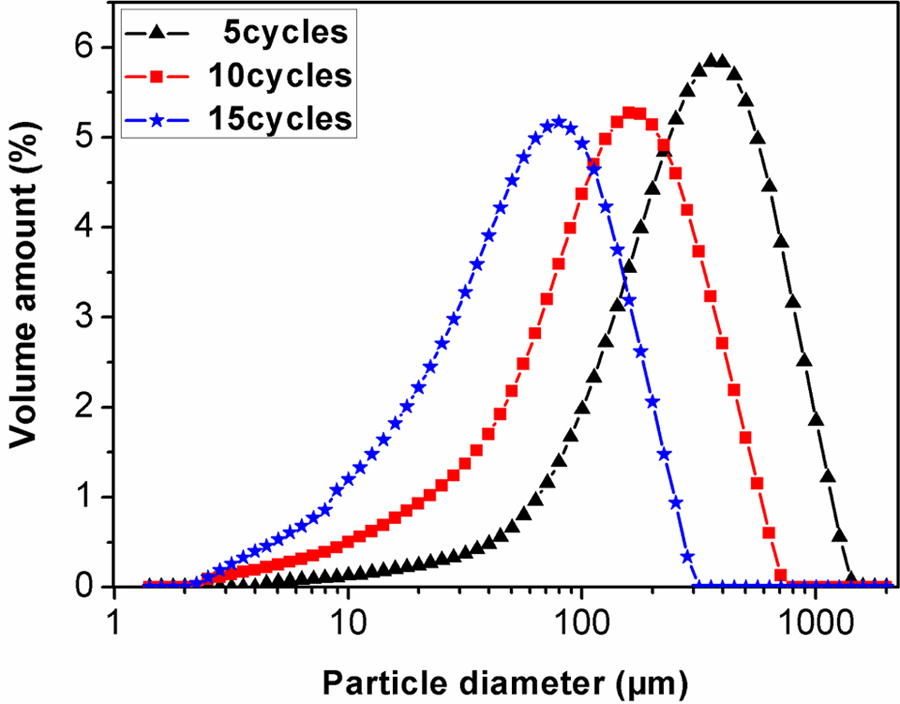
Particle size distributions of PMCS with different milling cycles. PMCS; pan-milled leather shaving.
Parameters of PMCS with different pan milling cycles.
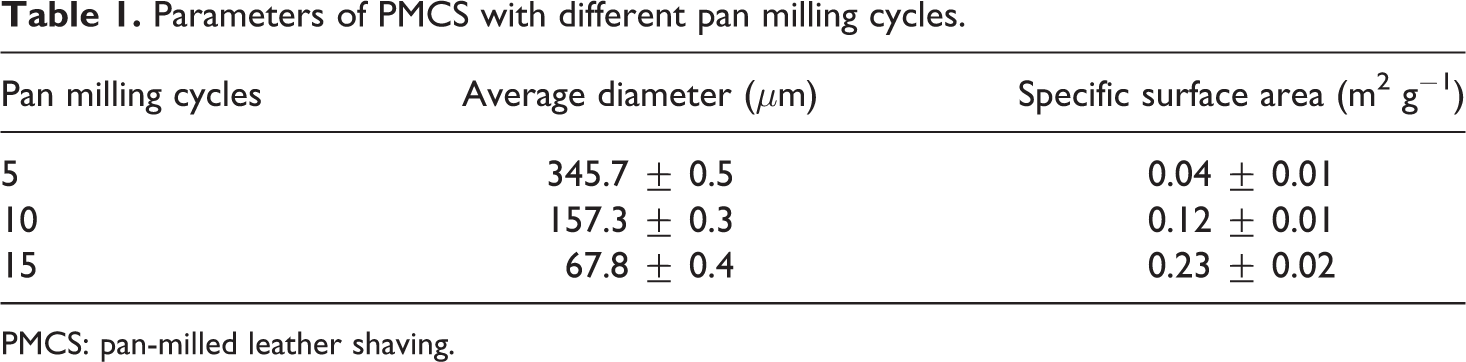
PMCS: pan-milled leather shaving.
Figure 3 illustrates the SEM micrographs of PMCS. It can be clearly seen that the particle size of PMCS reduces with the increase of pan milling cycles. The original leather shavings (Figure 3(a)) consist of entangled collagen fiber bundles with smooth surface. After 5 milling cycles (Figure 3(b)), the collagen fiber bundles of the original leather shavings become loose, and some collagen fibers are broken by the strong squeezing force in normal direction and the shearing force in both radial and tangential directions generated in-between the pan mill. When pan milling cycles increase to 10 (Figure 3(c)), parts of collagen fibers are separated and their surfaces become coarse. After 15 milling cycles (Figure 3(d)), the fibrous structure of leather shavings is almost destroyed and fine powder is obtained. The PMCS with the reduced particle size would undoubtedly provide larger specific surface area and improve their interfacial adhesion in PVA matrix more effectively, which is also beneficial to their dispersion in the matrix.
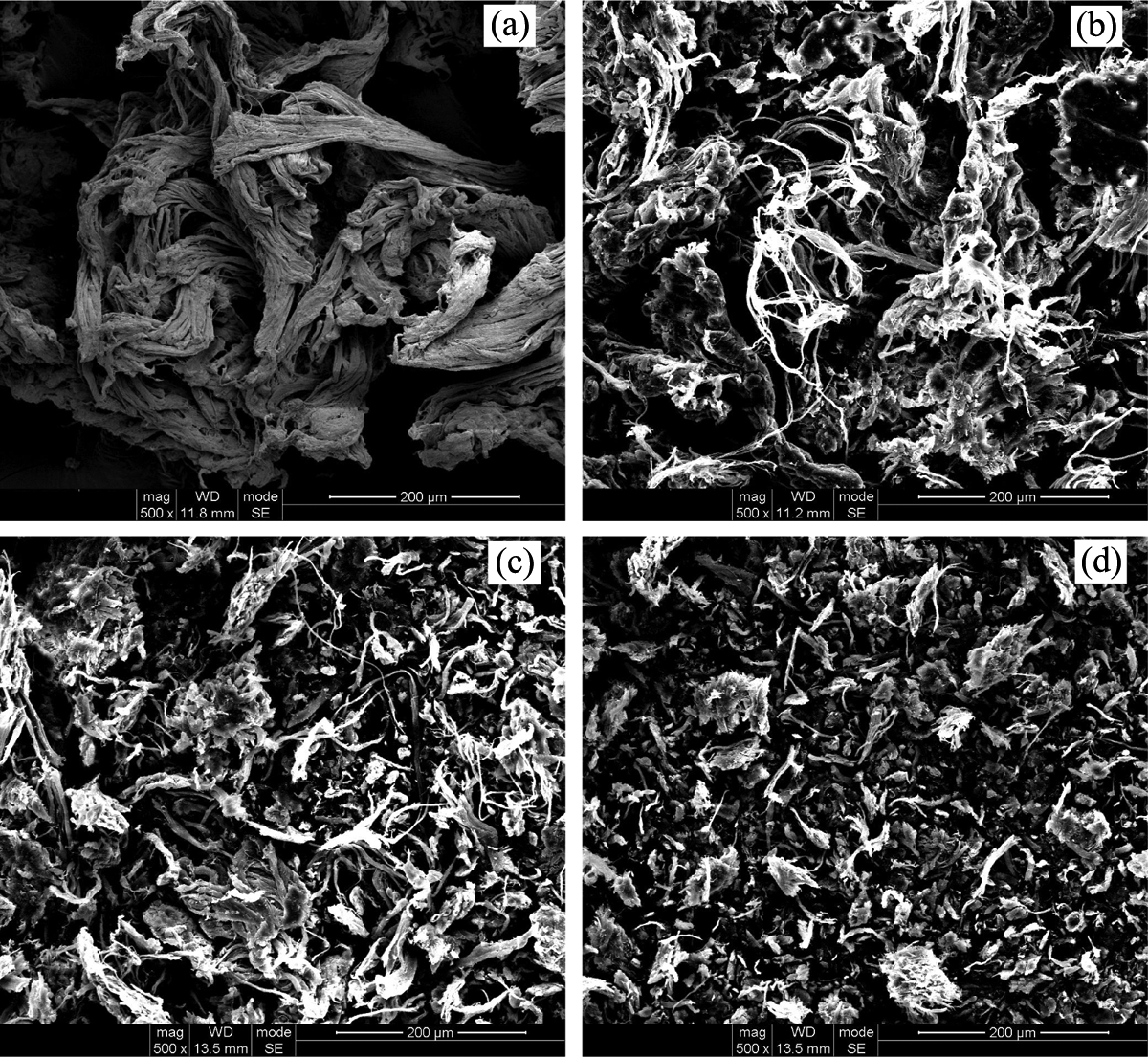
SEM micrographs of PMCS with different milling cycles with ×500 magnification ((a) 0 cycles; (b) 5 cycles; (c) 10 cycles; and (d) 15 cycles). SEM: scanning electron microscopic; PMCS; pan-milled leather shaving.
FTIR characterization is one of the most effective methods widely used to investigate the chemical and physical structure changes in polymers. The FTIR spectra of leather shavings before and after pan milling are shown in Figure 4. The original leather shavings have four characteristic absorption bands (Figure 4(a)), that is, the overlapping stretching band of –NH and –OH (around 3404 cm−1), the C=O stretching vibration (amide I, 1647 cm−1), N–H bending vibration (amide II, 1545 cm−1), 29 and Cr (III) absorptions (around 662 and 556 cm−1). 30 After 15 milling cycles (Figure 4(b)), the characteristic absorption of the –NH/–OH stretching band of leather shavings shifts to a lower wave number compared with the original ones and the shift value reaches 59 cm−1. Simultaneously, the peak of amide I changes from 1647 to 1656 cm−1. All above changes may result from the newly formed carbonyl groups, hydroxyl groups and amide groups of leather shavings during pan milling and also the breakage and the rearrangement of the hydrogen bonding in leather shavings under the strong shearing and compressing force exerted by pan mill mechanochemical reactor. It is further found that the Cr (III) peak at 556 cm−1 shifts to a high wave number (566 cm−1) and the peak at 662 cm−1 almost disappear. Interestingly, due to the new modality of Cr (III), 31 a new peak appears at 532 cm−1. These phenomena indicate that the complexation between Cr (III) and carboxylic groups of collagen is partly broken after pan milling and Cr (III) is mechanochemically activated.
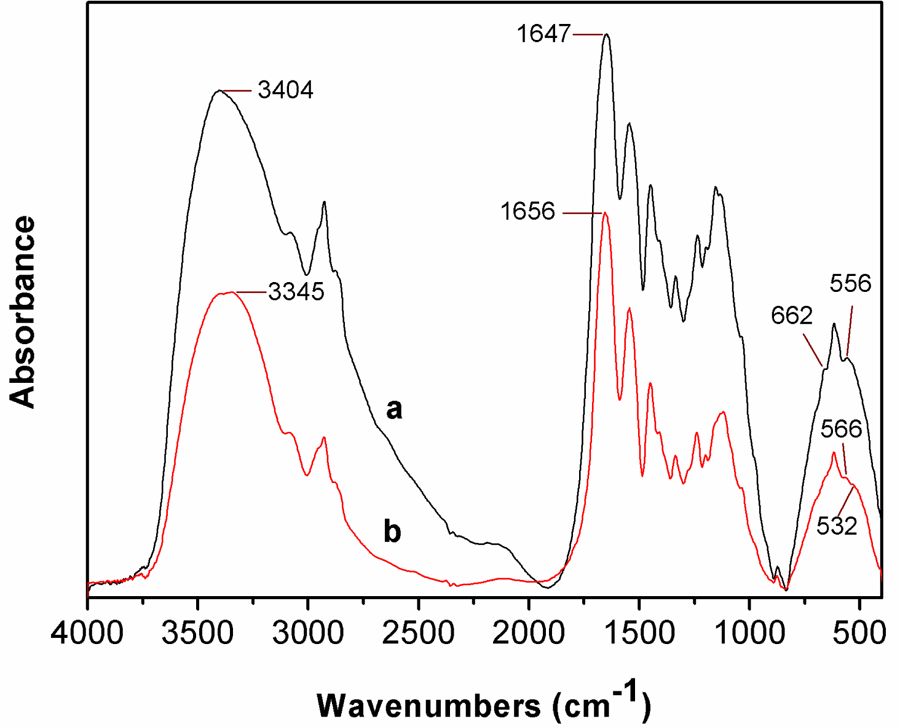
FTIR spectra of leather shavings (a) un-milled and (b) 15 milling cycles. FTIR: Fourier transform infrared.
Structural characteristics of PVA/PMCS composites
FTIR spectra of PVA and PVA/PMCS composites are shown in Figure 5. It can be observed that the peak of OH stretching in PVA/PMCS composites shifts to lower wave number compared with that of PVA, indicating that new hydrogen bonding forms between PVA and PMCS. Due to the new chelation between Cr (III) and the hydroxyl groups of PVA, 32 the peak at 614 cm−1 shifts to 583 cm−1. These interactions between PVA and PMCS may contribute to the improvement in their compatibility.
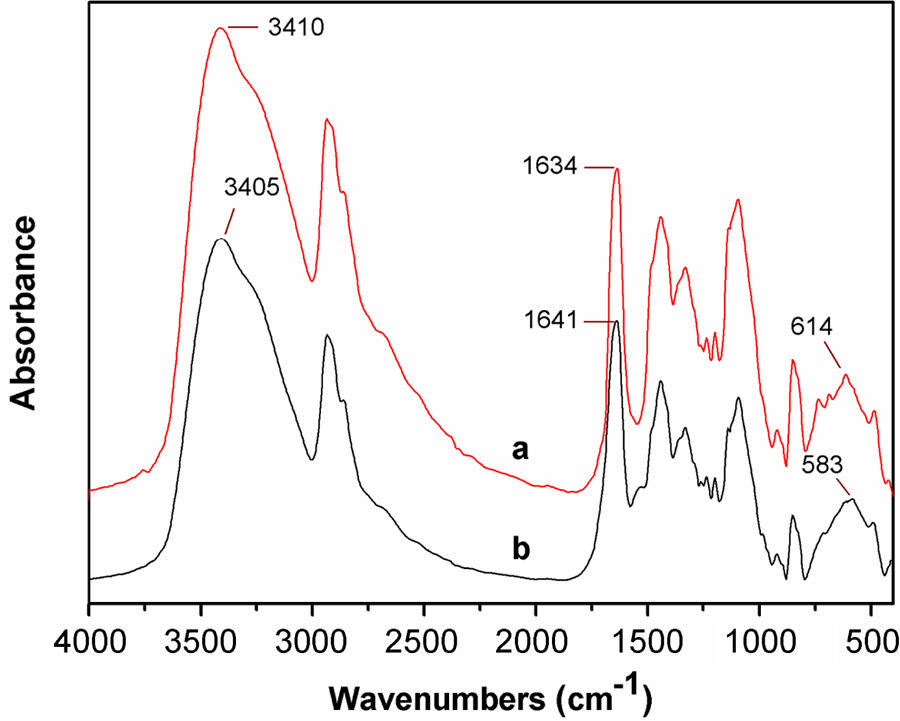
FTIR spectra of PVA (a) and PVA/PMCS composites (b). FTIR: Fourier transform infrared; PMCS; pan-milled leather shaving; PVA: poly (vinyl alcohol).
The dispersion of the fillers in matrix and the adhesion between the matrix and fillers are crucial factors for the efficient stress transfer, which influences the property of the obtained materials to a considerable degree. Figure 6 shows the morphology of the fractured surface of PVA/PMCS composites. As can be seen, there is good adhesion at the interface between PVA and PMCS, indicating that PMCS is compatible with PVA. This can be ascribed to the following factors: (1) more active groups are released under the mechanochemical activation of pan mill reactor, resulting in the possible formation of new hydrogen bonding and the chelation between PMCS and PVA; (2) large specific surface area of PMCS is obtained after pan milling, increasing the interfacial interaction between PVA and PMCS.

SEM micrographs of fractured surface of PVA/PMCS composites with different milling cycles (×5000 magnification; (a) 5 cycles; (b) 10 cycles; and (c) 15 cycles). SEM: scanning electron microscopic; PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Figure 7 shows the effect of PMCS content on its dispersion morphology in PVA matrix. As well known, the long fibers has an intensive tendency to be entangled and thus become very hard to well disperse in polymer matrix during melt processing. 33 After 15 pan milling cycles (Figure 3(d)), the fibrous structure of leather shavings is almost destroyed and the fine powder is produced, which can avoid the entanglement and improve the dispersion of leather shavings at low content (Figure 7(a)). But when PMCS content increases to 10 wt%, the agglomerates appear (Figure 7(b)) and the further increase of PMCS will lead to the formation of more agglomerates (Figure 7(c) and (d)).
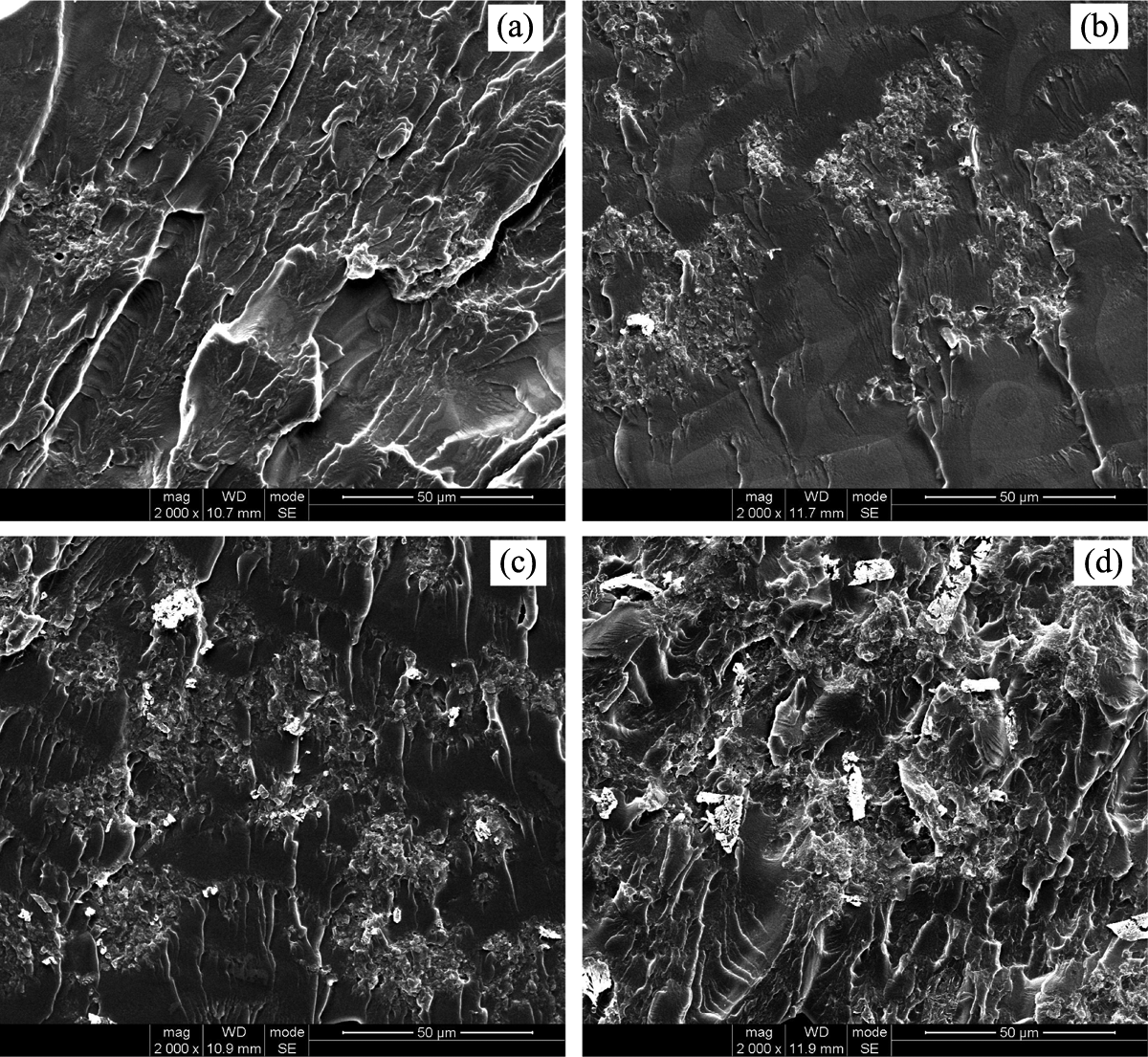
SEM micrographs of fractured surface of PVA/PMCS composites with different PMCS contents after 15 milling cycles (×2000 magnification; (a) 5 wt%; (b) 10 wt%; (c) 15 wt%; and (d) 20 wt%). SEM: scanning electron microscopic; PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Mechanical properties of PVA/PMCS composites
Figure 8 illustrates the effect of PMCS content and the milling cycles on the TS and EL of PVA/PMCS composites. Very encouragingly, PMCS shows an obviously reinforcing effect on PVA. The TS of PVA is 40 MPa, much higher than that of polypropylene and polyethylene. The addition of PMCS greatly increases the TS of PVA without sacrificing its EL, and the maximum value appears at the 5 wt% PMCS content due to good dispersion of PMCS in PVA matrix, as shown in Figure 7. It is noteworthy that even when PMCS content reaches to 20 wt%, the TS and the EL of the composite are still little higher than or almost the same as that without PMCS.
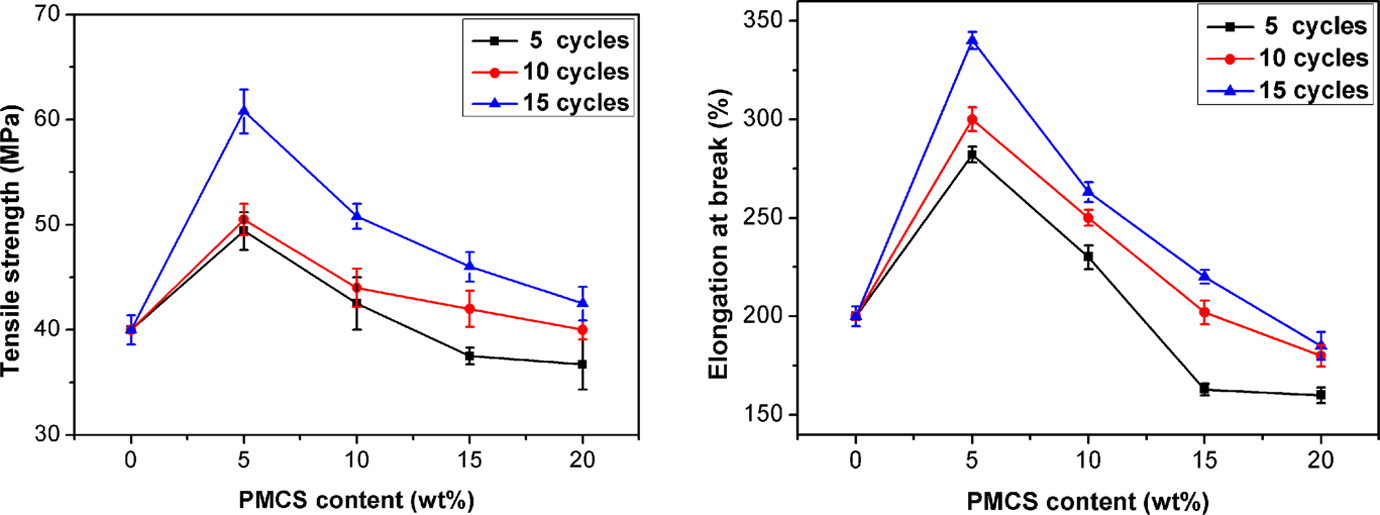
Tensile strength and elongation at break of PVA/PMCS composites. PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Pan milling cycles also apparently affect the mechanical properties of the composites. It is well known that it is very difficult to break the hydrogen bonding networks formed among the carbonyl groups, hydroxyl groups, and amide groups of collagen and the stable complexation formed between the Cr (III) and carboxylic groups of collagen in leather shavings. Therefore, it is hard to prepare high-performance leather shavings-filled materials due to the bad interfacial interaction in composites. However, the strong shearing and compressing force field of our pan mill mechanochemical reactor can effectively destroy the hydrogen bonding in collagen and the complexation between Cr (III) and collagen carboxylic groups to a considerable degree, thus generating a certain number of active groups (as confirmed by FTIR). The specific surface area of PMCS also increases due to the reduced particle size caused by the pulverization effect of pan mill mechanochemical reactor. All the above results are beneficial to enhance the interfacial interactions between PVA and PMCS, which surely contribute to the enhancement in the mechanical properties of the composites. It is noted that with the increase of PMCS content, the effect of the milling cycles on the involved mechanical properties becomes weaker, which can be ascribed to the deteriorated dispersion of PMCS with higher loading in PVA matrix. Due to the better mechanical properties achieved by proper increase of milling cycles, the PVA/PMCS composites prepared at 15 milling cycles are selected as the example for the further investigation.
Thermal properties of PVA/PMCS composites
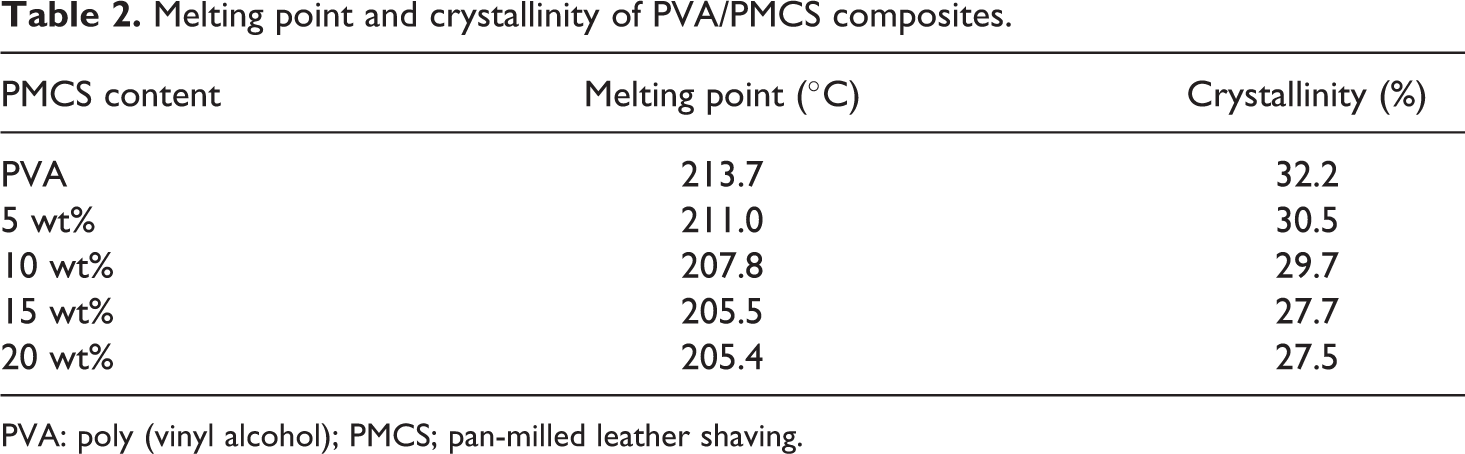
The DSC thermograms of PVA/PMCS composites are presented in Figure 9 and the corresponding melting temperature (Tm) and crystallinity are listed in Table 2. Again, it is interesting to find that the PMCS improves the thermal property of PVA. PVA exhibits a relatively large and sharp melting peak at 213.7°C (Figure 9(a)). For PVA/PMCS composites, their melting peaks become broader and also shift to lower temperature region (Figure 9(b) to (e)), which is beneficial to their thermal processing. This may attribute to the decrease in the ordered association of PVA molecules highly constrained by PMCS and the chelation between PVA and PMCS.
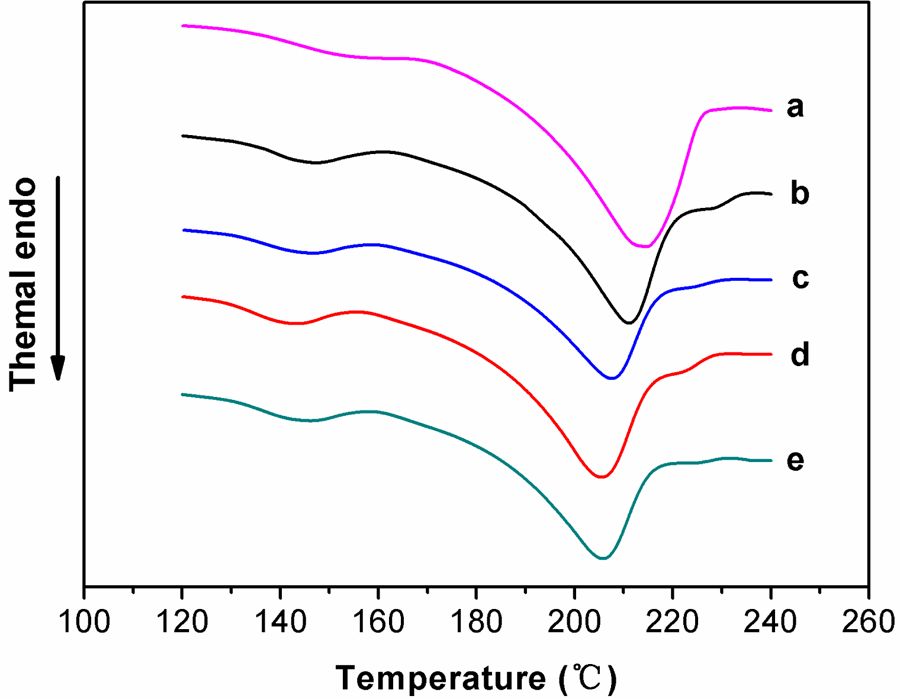
Differential scanning calorimetry curves for PVA/PMCS composites with different PMCS contents after 15 milling cycles (a) PVA; (b) 5 wt%; (c) 10 wt%; (d) 15 wt%; and (e) 20 wt%. PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Melting point and crystallinity of PVA/PMCS composites.
PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
The thermal stabilities of PVA, PMCS, and PVA/PMCS composites were compared based on TGA analysis, as shown in Figure 10. Both PVA and PVA/PMCS composites have three degradation steps and finally remain no more than 15 wt% of residues at 600°C under nitrogen atmosphere. In the case of PVA, the evaporation of water and other volatile components dominate the first weight loss (4–9 wt%), which the second and third steps describe the sequence of pyrolytic reactions. 34 A major degradation peak at 261°C, due to the decomposition of side chain of PVA, mainly involves dehydration and formation of some volatile products. The latter one appearing at 421°C can be ascribed to the decomposition of main chain of PVA, which could yield carbon and hydrocarbon. For PMCS, the initial weight loss below 200°C can be attributed to the loss of volatile components and there is only one major decomposition peak occurring at 327°C. For PVA/PMCS composites, the decomposition temperature of both side chain and main chain of PVA increases with the additive amount of PMCS content, but the decomposition temperature of side chain is almost the same when PMCS content is above 15 wt%. The change in decomposition temperature of side chain is smaller than that of main chain, indicating the inhibition of the thermal motion of PVA molecules due to the easier movement of the side chain of PVA than that of the main chain in the composites. 35 These results suggest that the introduction of PMCS into PVA matrix promotes the thermal stability of PVA/PMCS materials, which may be attributed to the high thermal stability of PMCS and the chelation between PMCS and PVA.
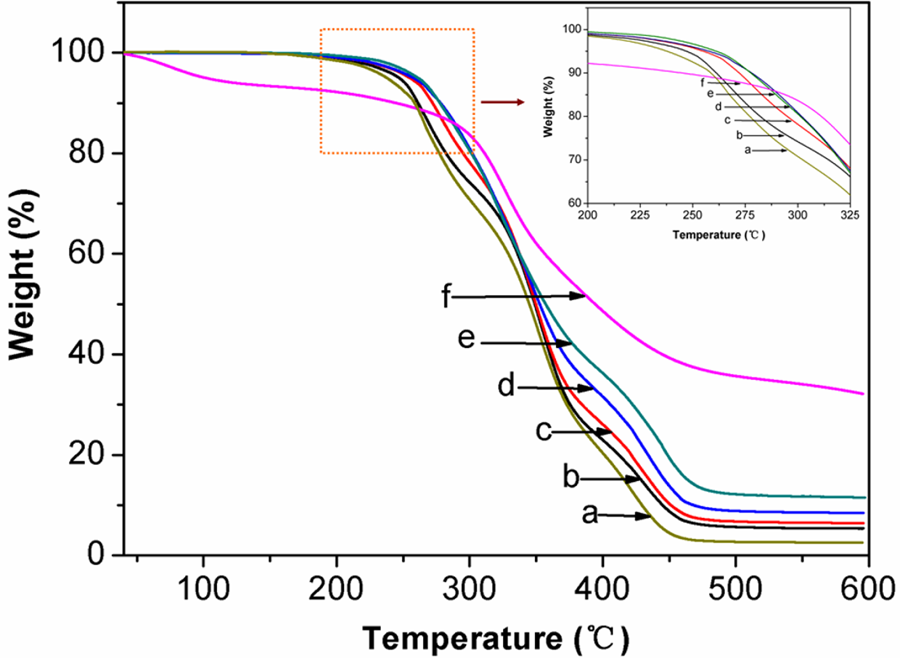
Thermogravimetric traces of PVA/PMCS composites (a) PVA; (b)5 wt%; (c) 10 wt%; (d) 15 wt%; (e) 20 wt%; and (f) PMCS). PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Water absorbability of PVA/PMCS composites
Composites based on PVA and the renewable fillers are known to be water sensitive, which restricts their application. 36 Due to the chelation between PVA and PMCS via Cr (III), this water-sensitive disadvantage of PVA/PMCS composites may be weakened. Water absorption behavior of PVA-based composites is presented in Figure 11. As the illustrated information, the water absorption ratio of PVA and PVA/PMCS composites increases quickly at the initial absorption stage, but then turns to be a slight increase, and finally reaches equilibrium state after 10–12 h. This phenomenon can be ascribed to the different osmotic pressure of water molecule at different phase. At the initial phase, the water molecules have great osmotic pressure that promotes the pervasion of water into inner part of the samples, thereby, accelerating the water absorption. With the progress of the absorption process, more and more water molecules enter the samples, thus leading to the reduction in the osmotic pressure of the sample and the decrease of the absorption rate with the absorption time prolonging. Finally, the osmotic pressure of the sample reaches the equilibrium, no water will permeate into the samples, and the water absorption ratio of the composites tends to be constant. It can also be seen from Figure 11 that with increasing PMCS content, the water equilibrium absorption ratio of the composites decreases, indicating that the addition of PMCS can reduce the water sensitivity of PVA. As reported elsewhere, 37 the increase of cross-linking degree and crystallinity would restrict the movement of polymer chains as well as the transformation of the macromolecular chain configuration. As a result, the water molecules can hardly penetrate into the hydrogen bonding network of the composites and hence decreasing the water equilibrium absorption ratio can be obtained. However, in our system, the addition of PMCS depresses the crystallinity of PVA, and the more the PMCS, the lower the crystallinity of PVA, as shown in Table 2. Therefore, the decrease in the water equilibrium absorption ratio of the composites with the increase of PMCS content may be mainly caused by the chelation between PVA and PMCS, that is, the cross-linking between PVA and PMCS plays a main role in decreasing the water absorption ratio of the composites.
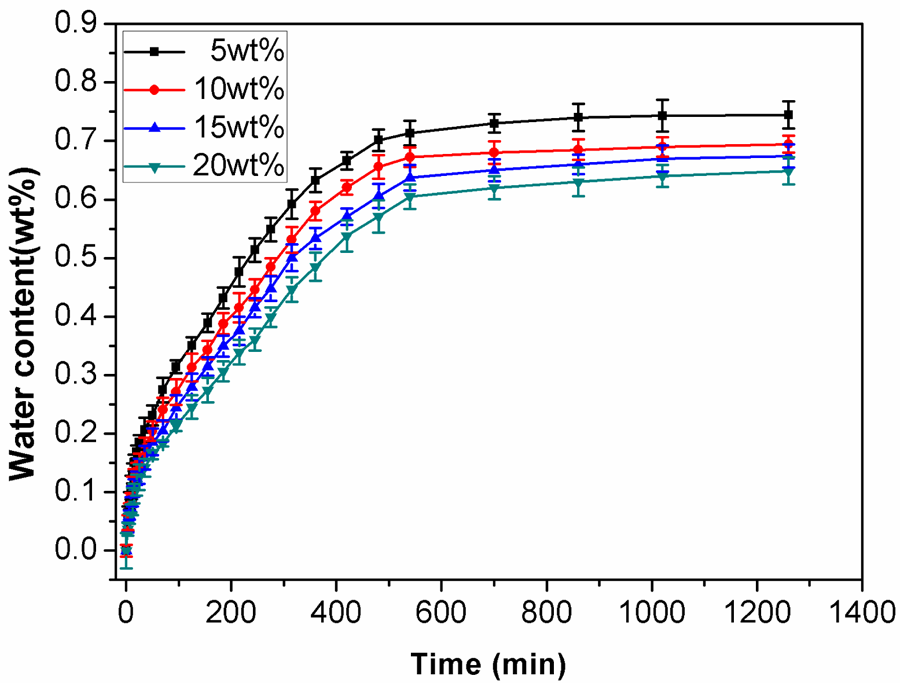
Water absorption property of PVA/PMCS composites with different PMCS content after 15 milling cycles. PVA: poly (vinyl alcohol); PMCS; pan-milled leather shaving.
Conclusions
The recycling of chrome-containing leather shavings is of great importance and a huge challenge worldwide. This article reported a novel recycle technology of chrome-containing leather shavings by combining the S3M with the thermal processing of PVA and successfully obtained a new leather-containing material with potential application as packaging material, tribological material, shoe-making material, and so on. The experimental results showed that under the strong shearing and compressing forces of pan milling, the leather shaving particles with smaller size and larger specific surface area could be obtained. Meanwhile, more reactive carbonyl groups/hydroxyl groups/amide groups and Cr (III) were continuously exposed on the surface of leather shavings, leading to good compatibility and good dispersion of leather shavings in PVA matrix. As a result, the PVA/leather shaving composites with pretty good performances, for example, better mechanical properties, thermal stability, and water resistance than neat PVA were obtained. Especially, the TS and EL of the composites reached the maximum at 5 wt% PMCS content and 15 milling cycles. Compared with the conventional chemical methods of recycling leather shavings, the technology reported in this article saved the resources and did not cause the second pollution and could also avoid the disadvantages of the solution method for preparing PVA-based composite. It can be concluded that this is a convenient, cost-efficient and environment-friendly technology to deal with the tough problem of recycling the chrome-containing leather shavings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to express their thanks to the National Natural Science Foundation of China (50833003, 51010004) and the international collaboration program from the Ministry of Science and Technology of China (2010 DFA54460) for their financial support throughout this work.