Abstract
In order to improve the waste sludge dewaterability of polyacrylamide (PAM), composite conditioner of cationic PAM/montmorillonite (CPAM/MMT) was synthesized by in situ polymerization. The structure and property of the composite were studied in terms of intermolecular hydrogen bonding, melting behavior, intercalation behavior, and dewaterability of the waste sludge. The results revealed that the molecular weight and cationic degree of CPAM/MMT composites decreased with the increase of MMT content. The hydrogen bonding between –OH groups of MMT and –NH groups of CPAM was confirmed by Fourier transform infrared spectroscopic analysis, which led to a relatively high elastic modulus values and low tan δ values at 5 wt% MMT content, indicating the possible formation of a physically cross-linked network in aqueous solution for the composite. The melting temperature of the composites presented decreasing trend first and then became higher as the content of MMT increased. The MMT platelets dispersed well with an intercalated state in CPAM matrix as indicated by x-ray diffraction analysis and transmission electron microscopic observation. The addition of MMT reduced the specific resistance to filtration of the waste sludge, with a minimum value being achieved at 5 wt% MMT content, while the ultraviolet transmittance of the supernatant of the waste sludge was also improved, and the turbidity was reduced. This synergistic effect of the two components in the composite through charge neutrality and bridging action of CPAM as well as the adsorption capacity of MMT resulted in an enhanced dewaterability of waste sludge.
Keywords
Introduction
Sewage sludge production has increased dramatically during the last few decades such that waste sludge has become a current problem due to its huge production and environmental pollution. 1 In order to deal with the waste sludge, dewatering is required as it can reduce the total waste volume and increase the caloric value of the product. 2,3 However, waste sludge is known to be a poor dewaterable system, 4 for which reason it is generally conditioned prior to dewatering with varying flocculants to change the properties of the sludge solid, such as its surface charge and particle size, and as a result a significant improvement in solid–liquid separation can be obtained. 5 The diagram of the process of sewage sludge treatment was shown in Figure 1.

The diagram of the process of sewage sludge treatment.
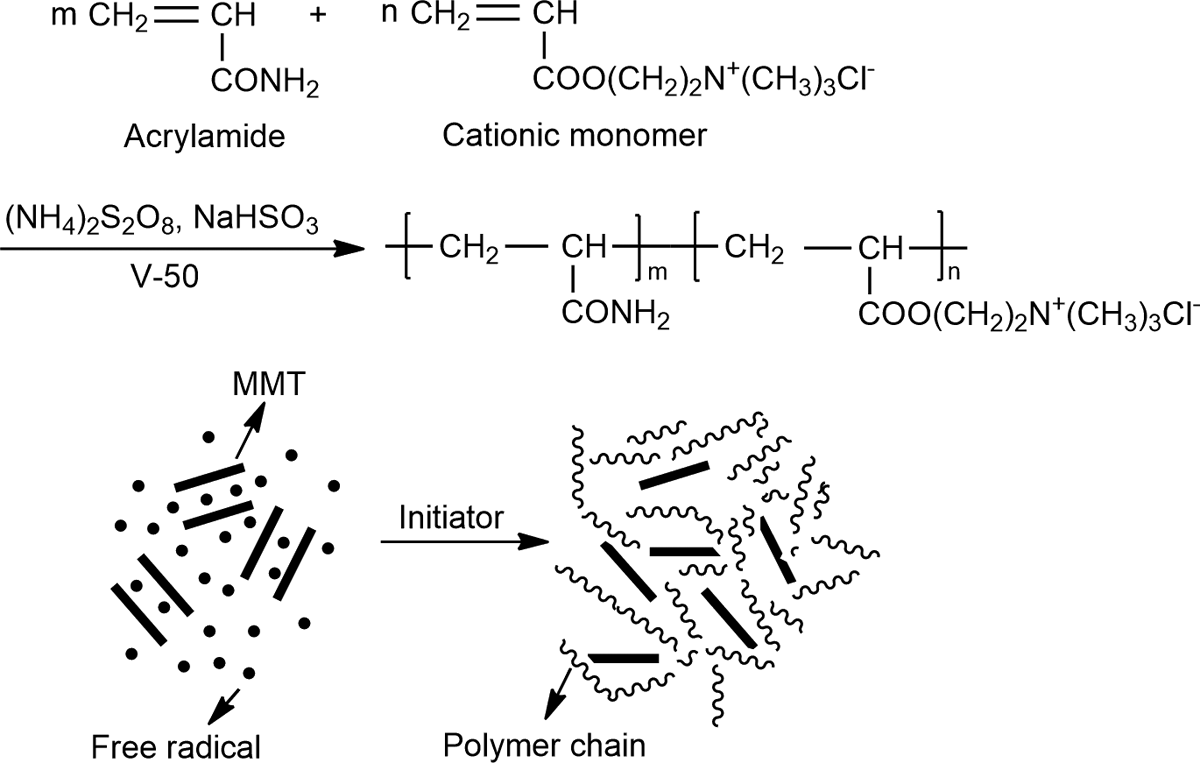
Schematic representation of in situ polymerization of CPAM/MMT composite. CPAM: cationic polyacrylamide; MMT: montmorillonite.
For the dewatering process of waste sludge, the sludge particles are invariably negatively charged and tend to form stable dispersions with poor dewatering characteristics. Therefore, charged organic polymers, also called polyelectrolytes, have gained a large market share over the last decades, since they can be dosed in much lower quantities than inorganic flocculants such as lime and iron chloride. 6 Among these, cationic polyacrylamide (CPAM) is one of the most important water soluble polymeric flocculants, which is available in many different combinations of chemical composition, molecular weight, and charge density. It has been employed in many industries because of its effectiveness as a flocculant and the ability to produce good settling performance at relatively low cost. 7 Addition of CPAM with high-molecular-weight helps to neutralize the surface charge and eliminates the electrostatic repulsion between approaching particles, and the macromolecules can span and reduce the gap between the particles through a bridging mechanism.
Adsorption has been found to be an effective way for wastewater treatment in terms of low cost, simplicity of design, ease of operation, and insensitivity to toxic substances. Montmorillonite (MMT) is often used as the adsorbent due to its excellent chemical stability, high specific surface area, and adsorption capacity. Voorn et al. 8 synthesized polyacrylamide (PAM)/MMT nanocomposite latex particles by inverse Pickering emulsion polymerization. It was found that the latexes were stabilized by hydrophobic clays, and all the clay platelets were incorporated into the composite particles in partially exfoliated state. Long et al. 9 prepared PAM/MMT nacre-like nanocomposites by electrophoretic deposition. The results showed that the nanocomposites had improved mechanical properties and possessed a uniform and well-ordered layer-by-layer structure with MMT sheets as inorganic layers and PAM as interlayer phase.
In this work, the composites of CPAM/MMT were synthesized by in situ polymerization with acrylamide (AM) and acryloxyethyl trimethyl ammonium chloride (DAC) as cationic monomer in the presence of MMT. MMT was expected to be dispersed in CPAM matrix better than that of the composites prepared by direct mixing due to easier intercalation of AM and DAC into MMT layers. Through the charge neutrality and bridging action of CPAM, as well as adsorption capacity of MMT, the composite conditioner could be expected to improve the dewaterability of waste sludge. The structure and intermolecular hydrogen bonding of the composite were studied, and the effect of MMT content and dosage of the composite on the dewaterability of waste sludge was systematically investigated.
Experimental
Materials
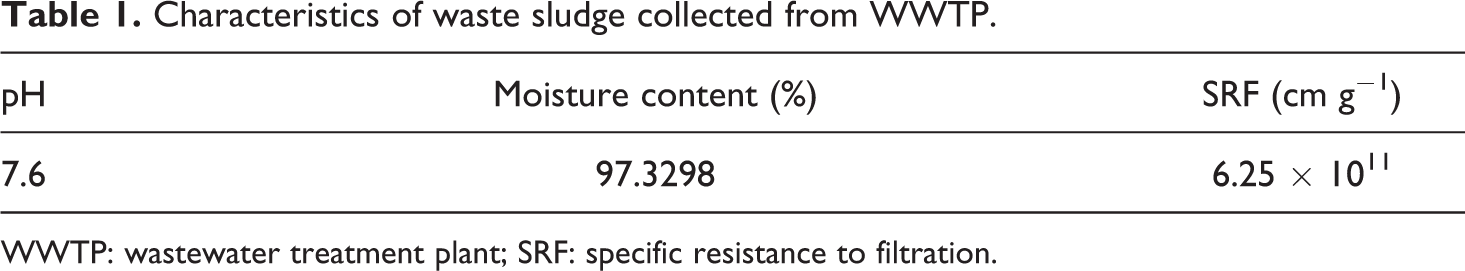
The monomer AM (≥98% purity) was supplied by Bodi Chemical (Tianjin, China) and purified by recrystallization from acetone. Redox initiators, ammonium persulfate ((NH4)2S2O8, ≥98% purity) and sodium bisulfite (NaHSO3, analytical reagent grade) were purchased from Kelong Chemical Reagents (Chengdu, China) and Bodi Chemical, respectively. Cationic monomer DAC and the water-soluble initiator 2,2′-azobis(2-methylpropionamidine)dihydrochloride (V-50) were purchased from Guang Chuang Jing Import and Export Co. Ltd (Shanghai, China). The analytical grade reagents urea (H2NCONH2, Zhiyuan Chemical Reagents, Tianjin, China) and ethylenediaminetetraacetic acid disodium salt (EDTA-2Na, Bodi Chemical) were used as received. MMT modified by trimethyl octadecane hydroxyl ammonium chloride with 90–120 mmol/100 g1 of cation exchange capacity was obtained from Zhejiang Fenghong New Material Co. Ltd (Zhejiang, China). Other chemical agents were all of analytical grade and used as received. Waste sludge samples were collected from the secondary settling tank of a municipal wastewater treatment plant in Chengdu, China. The characteristics of the waste sludge are listed in Table 1.
Characteristics of waste sludge collected from WWTP.
WWTP: wastewater treatment plant; SRF: specific resistance to filtration.
In situ polymerization of CPAM/MMT composites
A series of CPAM/MMT composites with different content of MMT were synthesized according to the following procedure. MMT was first dispersed in water by agitation at 80°C for 2 h and allowed to stand overnight to swell adequately. Then, it was tip sonicated at a 250-kHz frequency for better dispersion in water with a sonicator (model SC-III; Jiuzhou Ultrasonic Technology Co., Chengdu, China) for 15 min before use.
The solution of AM, DAC, urea, EDTA-2Na, and deionized water in the ratio of 10:10:0.02:0.004:32.8 g was charged into a 100-mL three-necked round-bottomed flask equipped with a magnetic stirrer, a nitrogen (N2) inlet, and a thermometer. The mixture was stirred under N2 gas at 5°C. Aqueous solution of V-50 (0.333 g; 0.3 wt%) was added into the reactor. The solution was kept for 10 min, followed by addition of MMT suspension and 2.0 g of aqueous solution of (NH4)2S2O8 (0.28 wt%), and then aqueous solution of NaHSO3 (2.0 g, 0.28 wt%) was added dropwise. The reaction was kept at the constant temperature in water bath for 2 h, and then heated to 70°C and stirred for 3 h. Finally, the product was washed with acetone, filtered, dried under vacuum at 40°C, and stored for the subsequent experiment.
Conditioning experiments
Filterability was measured by a Buchner funnel test, one of the most common methods used in dewaterability measurement,
10
and expressed in terms of specific resistance to filtration (SRF) and moisture of sludge cake. All experiments were performed in triplicate for mean calculation. In each test, the conditioner was added into a 250-mL beaker with 100 mL of the waste sludge, then stirred for 2 min, and then left still for 5 min. The mixed sludge was then poured into the funnel fitted with a filter paper (pore size: 0.45 μm). After 2 min of gravitational drainage, a vacuum of 0.05 MPa was applied and the volume of the filtrate collected at different times recorded. The SRF (in centimeter per gram) of the waste sludge was calculated by:
where P is the filtration pressure (in pascal); A is the filter area (in square centimeter); μ is the filtrate viscosity (in newton second per square meter); w is the weight of cake solids per unit volume of filtrate (in kilogram per cubic meter), and b is the slope of the filtrate discharge curve (in second per hexa meter m−6).
The cake sludge produced from the Buchner funnel process was dried in an oven at 105°C to determine the water content of the cake sludge for evaluating the sludge dewaterability.
Residual turbidity of the supernatant after 30 min of settling was measured by a turbidimeter (Shanghai Xin Rui Instruments Co., China). The ultraviolet (UV) transmittance was measured with an α-1860 UV-visible spectrophotometer (Shanghai Lab-spectrum Instruments Co., China) under room temperature at a wavelength of 660 nm.
Measurements
Reduced viscosity
The samples of CPAM and CPAM/MMT composites of about 0.05 g were dissolved in 100 mL of sodium chloride (1.00 mol L−1). The time of outflow of the solution was measured by an Ubbelohde viscometer (Shanghai Shenyi glass products Co., China) in a water bath at 30°C, according to GB 12005.1-89 (National Standard of China). Then, The reduced viscosity was calculated using the following equation:
where t is the time of outflow of the CPAM and CPAM/MMT solution (in seconds); t 0 is the time of outflow of the solvent (in seconds), and C is the concentration of the CPAM and CPAM/MMT solution (in gram per milliliter).
Cationic degree
Cationic degree of the samples of CPAM and CPAM/MMT composites was measured according to the Moore method.
11
A dry sample (0.2 g) was dissolved in distilled water, and 1 mL of aqueous solution of potassium chromate was added. The solution was titrated with aqueous solution of silver nitrate (0.1 mol L−1) until the color of the solution turned brick red.
12
The cationic degree of the samples can be calculated from the formula: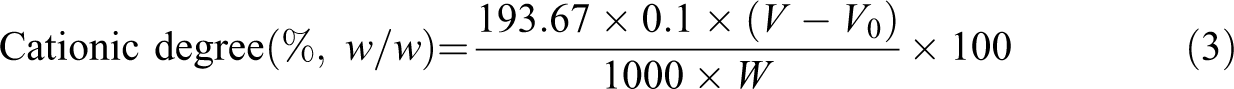
where V and V 0 are the volumes of consumed silver nitrate solution for the CPAM sample and blank sample, respectively, W is the weight of the sample, and the molecular weight of DAC is 193.67 g/mol.
FTIR analysis
The samples of CPAM and CPAM/MMT composites were analyzed with a Nicolet-560 Fourier transform infrared spectrometer (FTIR; Madison, Wisconsin, USA). The scanning rate was 20 scans s−1, and the resolution was 4 cm−1. The pellet sample was prepared by pressing the mixture of CPAM powder sample and potassium bromide powder.
Rheological property
The rheological measurement of the samples of CPAM and CPAM/MMT composites was conducted with a Gemini 200 plate rheometer (Malvern Instruments Co., Worcestershire, UK) at a temperature of 25 ± 0.1°C.
DSC analysis
Differential scanning calorimetry (DSC) analysis of the samples of CPAM and CPAM/MMT composites was conducted with a Netzsch 204 (Phoenix DSC, Germany). The temperature scale of the DSC was calibrated with indium. The samples were scanned at a heating rate of 10 K min−1 from ambient temperature to 250°C under N2 atmosphere. After waiting for 5 min to eliminate the effects of the previous thermal history, the samples were cooled to ambient temperature at a constant rate of 10 K min−1.
WAXD analysis
The interlayer spacing of the samples of MMT and CPAM/MMT composites was measured at room temperature over the scanning range approximately of 2θ = 2–10°with Rigaku D/max III B x-ray diffraction equipment (Japan). Copper (Cu) Kα
radiation (λ = 0.154 nm) was used at a generator voltage of 40 kV, current of 35 mA, and the scanning speed was 2.4 deg min−1. The d-spacing of the MMT layers was calculated with the Bragg equation:
where θ is the diffraction angle; n is the order of diffraction, and λ is the incident wavelength.
TEM observations
Transmission electron microscopy (TEM) analysis was carried out to observe the micromorphology of the sample of CPAM/MMT composite. The aqueous solution of the sample was carefully dropped onto the copper grid and dried with filter paper. The coated Cu grid was then used for TEM observation on a JEM 100CX II TEM equipment (JEOL, Japan).
Results and discussion
Preparation of CPAM/MMT composite by in situ polymerization
CPAM/MMT composites were synthesized by in situ polymerization, and the reaction formula was shown in Figure 2. The cationic degree and reduced viscosity of the CPAM/MMT composites were listed in Table 2. It can be seen that the cationic degree and reduced viscosity of the composites decreased with increasing MMT content. It may be interpreted that MMT acted as monomer absorbers like micelles did, 13 so the local concentrations of AM and DAC near MMT would be elevated. The total amount of AM and DAC adsorbed on MMT would increase as the content of MMT increased, which reduced the concentration of the monomer in the aqueous solution. After the intercalation of MMT, most molecular chains end-tethered on silicate layers encountered with second radicals and terminated by coupling. No further polymerization proceeded inside the silicate layers. Meanwhile, the MMT sheets may act to terminate propagating chains during the polymerization reaction 14 resulting in a relatively low-molecular-weight and cationic degree of the composite.
Characteristics of CPAM/MMT composites synthesized by in situ polymerization.

CPAM: cationic polyacrylamide; MMT: montmorillonite; η: reduced viscosity.
Hydrogen bond formation of CPAM/MMT composite
MMT used in this work was modified by trimethyl octadecane hydroxyl ammonium chloride, which may form hydrogen bonds with –NH groups of CPAM. This was confirmed by FTIR analysis, as shown in Figure 3. For CPAM (curve 1), the strong N–H stretching vibration absorption peaks at 3426 and 3191 cm−1, and C=O stretching vibration absorption peak at 1666 cm−1 were attributed to amide groups of CPAM. The bands at 2931 and 1349 cm−1 were related to C–H and C–N stretching vibrations, respectively. 15 The peaks at wave numbers of 1452 and 952 cm−1 were assigned to the methyl groups of quaternary ammonium cationic group.
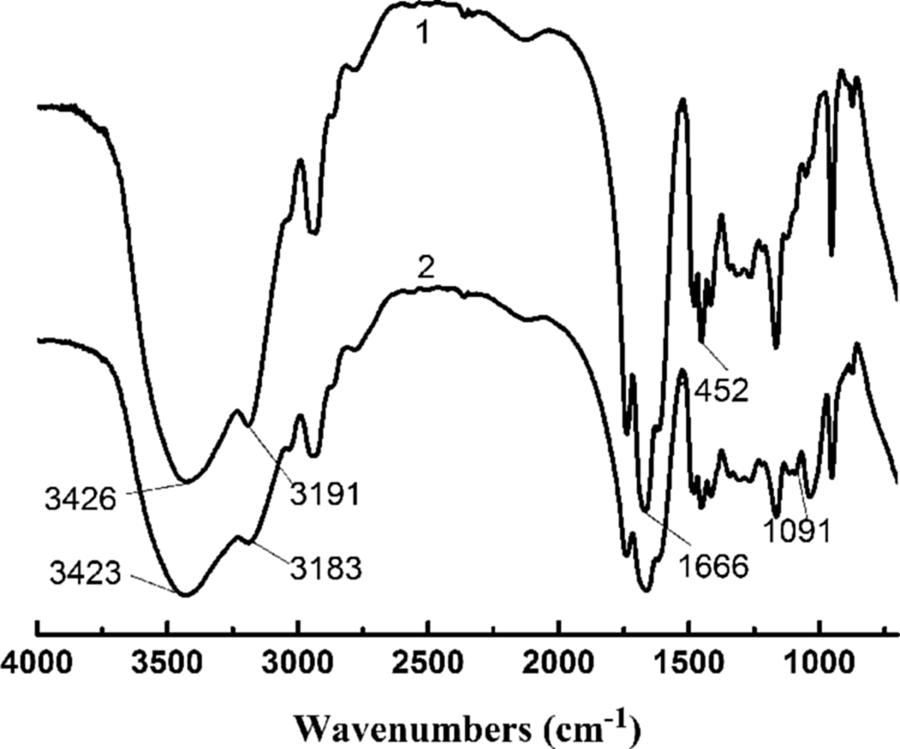
FTIR spectrum of CPAM and CPAM/MMT composite (1) CPAM; (2) CPAM/5 wt% MMT. FTIR: Fourier transform infrared; CPAM: cationic polyacrylamide; MMT: montmorillonite.
For the CPAM/MMT composite (curve 2), in addition to the characteristic absorption peaks of CPAM, the strong broadband at 1091 cm−1 was likely due to siloxane (Si–O–Si) bonds. Compared with curve 1, a red shift of N–H stretching vibration absorption peak to 3423 and 3183 cm−1 can be observed, which was attributed to the formation of hydrogen bonds between –NH groups of CPAM and –OH groups of MMT, because the hydrogen bond reduced the electron density of the N–H bond and its frequency. The proposed hydrogen bond formation in the CPAM/MMT composite is shown in Figure 4.
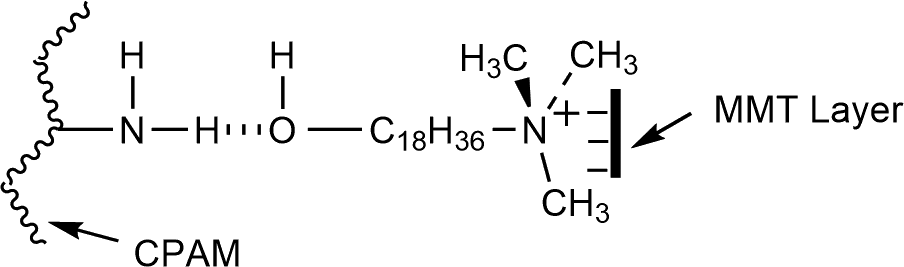
Hydrogen bond formation of CPAM/MMT composite. CPAM: cationic polyacrylamide; MMT: montmorillonite.
The elastic modulus (G′) and loss factor of CPAM and CPAM/MMT composites as a function of shear frequency are shown in Figures 5 and 6. Except for the sample of CPAM/5 wt% MMT, a relatively low G′ values can be observed for CPAM/MMT composites compared with pristine CPAM. The increasing addition of MMT resulted in a decline of the molecular weight of CPAM, leading to a decrease of the viscosity of CPAM solution. However, as for the sample of CPAM/5 wt% MMT, the increase in G′ values suggested the strong intermolecular bonding of CPAM and MMT, and possible formation of a physically cross-linked network in aqueous solution with the MMT serving as the cross-links.
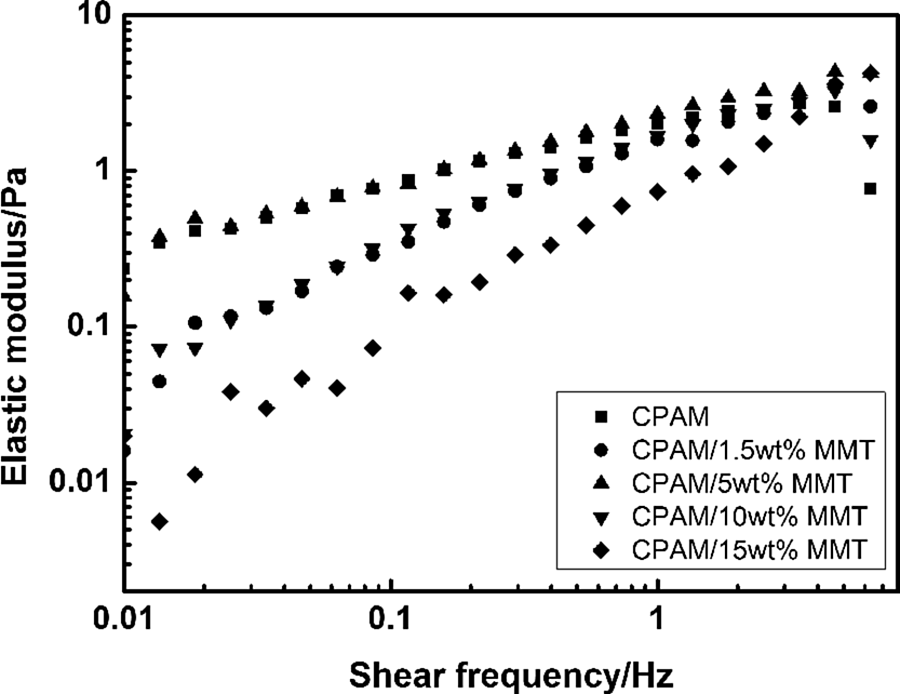
Elastic modulus of CPAM and CPAM/MMT composites at 25 ± 0.1°C. CPAM: cationic polyacrylamide; MMT: montmorillonite.
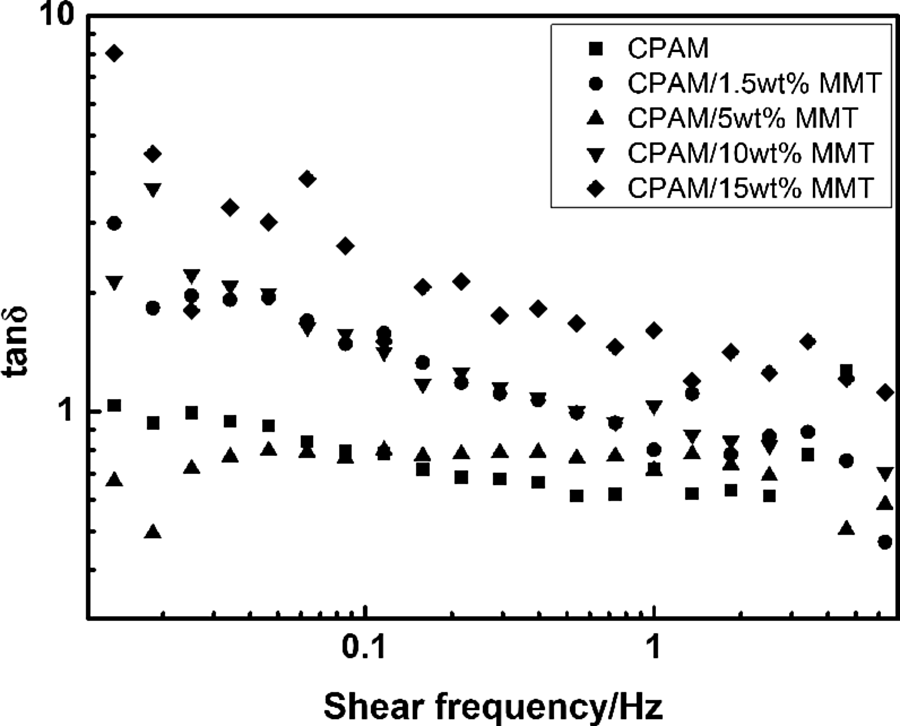
Tan δ of CPAM and CPAM/MMT composites at 25 ± 0.1°C. CPAM: cationic polyacrylamide; MMT: montmorillonite.
The quantity of tan δ represents the ratio of dissipated energy to stored energy during one deformation cycle. Except for the sample of CPAM/5 wt% MMT, tan δ values of the CPAM/MMT composites were higher than that of pristine CPAM (Figure 6), suggesting the molecular chains were more flexible and easier to move because of decrease of CPAM molecular weight. As for the sample of CPAM/5 wt% MMT, the decrease of tan δ values indicated that the mobility of the CPAM chains decreased with the presence of MMT. As suggested by Haraguchi et al., 16 –18 CPAM chains were bound to the surface of MMT particles due to the intermolecular hydrogen bonding as well as complex formation between the metal ions on the MMT surface and carbonyl oxygen of the acrylamide.
Melting behavior of CPAM/MMT composites
The melting curves of CPAM and CPAM/MMT composites are shown in Figure 7, and the melting parameters are listed in Table 3. It can be seen that CPAM exhibited two melting curves. The first melting curve was due to the cationic block of the polymer, and the relatively large and sharp endothermic curve with a peak at 220.6°C was the main melting curve of CPAM. Almost no change in the first melting curve was observed, and the main melting peak of CPAM/MMT composites presented first decreasing trend and then increased slightly with increasing MMT content. The depression of the melting temperature indicated that the ordered association of CPAM chains were reduced with the presence of MMT, 19 when the MMT concentration further went up, it acted as the role of a crystal nucleating agent, which could promote crystallization of CPAM. During the cooling process of DSC measurement, no exothermic peak was observed for the samples of CPAM and the composites indicating the weak crystallization ability of CPAM.
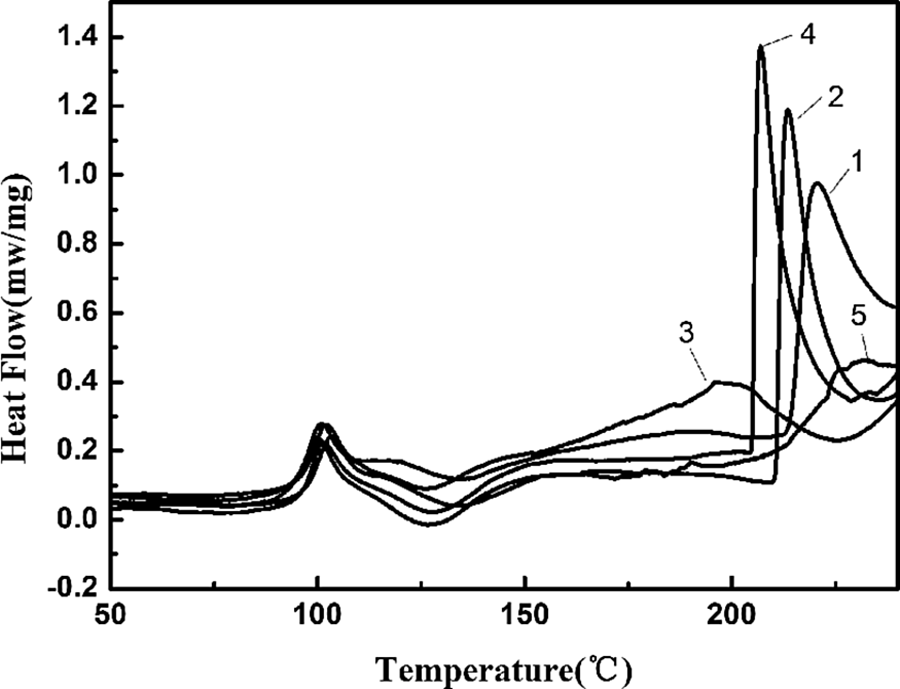
DSC melting curves of CPAM and CPAM/MMT composites (1) CPAM; (2) CPAM/1.5 wt% MMT; (3) CPAM/5 wt% MMT; (4) CPAM/10 wt% MMT; (5) CPAM/15 wt% MMT. DSC: differential scanning calorimeter; CPAM: cationic polyacrylamide; MMT: montmorillonite.
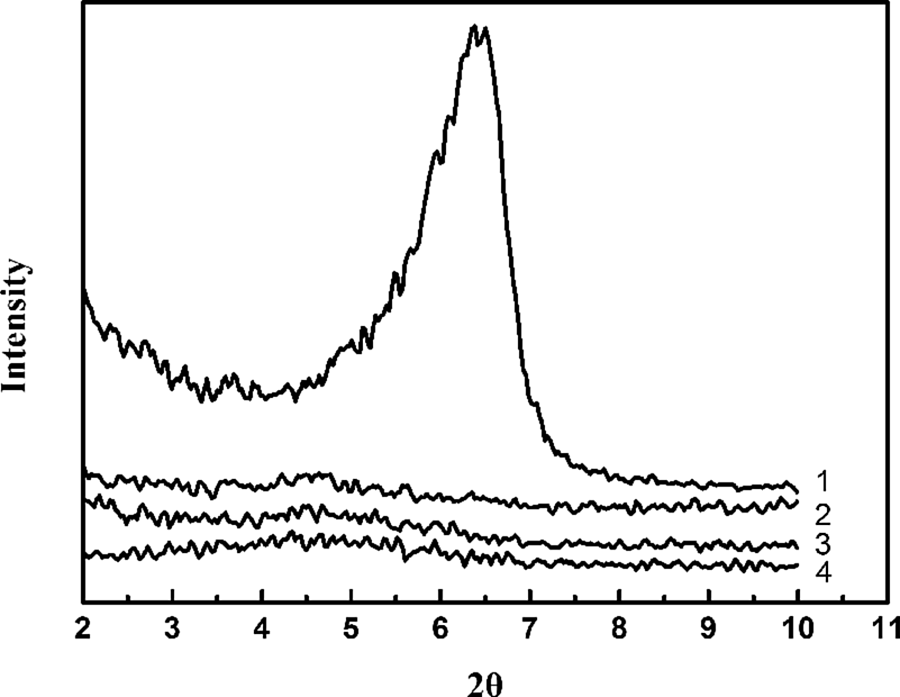
X-Ray diffraction patterns of MMT and CPAM/MMT composites (1) MMT; (2) CPAM/5 wt% MMT; (3) CPAM/10 wt% MMT; (4) CPAM/15 wt% MMT. MMT: montmorillonite; CPAM: cationic polyacrylamide.
Melting parameters of CPAM and CPAM/MMT composites.

T m1: temperature of the first melting peak; T m2: temperature of the second melting peak; H m1: enthalpy of the first melting peak; H m2: enthalpy of the second melting peak; CPAM: cationic polyacrylamide; MMT: montmorillonite.
Intercalation of MMT in CPAM matrix
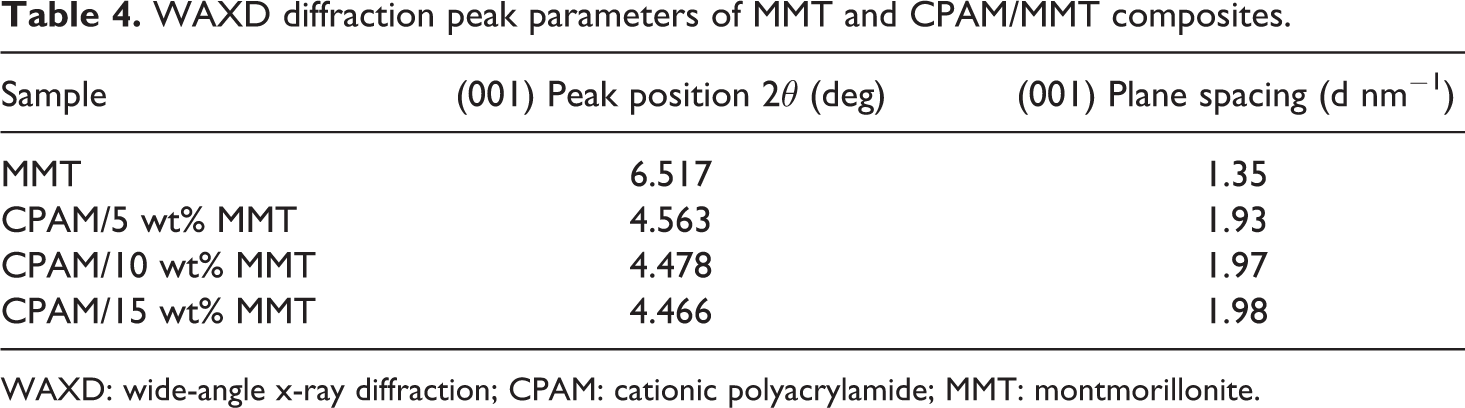
The interlayer spacing of MMT layers in pristine MMT and CPAM/MMT composites were determined using x-ray diffraction (XRD; Figure 10). A strong peak was observed at 2θ = 6.52° for pristine MMT, corresponding to the (001) plane, indicating the interlayer spacing (d-spacing) of MMT was 1.35 nm. In curve 2, only a very weak peak appeared at 2θ = 4.56° (d-spacing = 1.93 nm), which implied that the MMT platelets were in an intercalated state in CPAM matrix. Addition of CPAM increased the interlayer spacing of MMT, but the content of MMT influenced the interlayer spacing slightly. In this experiment, the composite had the intercalation of silicate up to 15 wt% of MMT. The WAXD diffraction peak parameters of MMT and CPAM/MMT composites are listed in Table 4.
WAXD diffraction peak parameters of MMT and CPAM/MMT composites.
WAXD: wide-angle x-ray diffraction; CPAM: cationic polyacrylamide; MMT: montmorillonite.
TEM was used to observe the morphology of the composites with 5–15 wt% MMT, as shown in Figure 9. It can be seen that most of MMT layers, corresponding to the dark stripes, were dispersed in CPAM matrix in an intercalated state, and only few of them aggregated. For the sample of CPAM/1.5 wt% MMT composite, MMT content was too low to be observed.

TEM images of CPAM/MMT composites (a) CPAM/5 wt% MMT; (b) CPAM/10 wt% MMT; (c) CPAM/15 wt% MMT. TEM: transmission electron microscopy; CPAM: cationic polyacrylamide; MMT: montmorillonite.
Waste sludge dewaterability of CPAM/MMT composite
Effect of MMT content on dewaterability of waste sludge
The SRF and the moisture of sludge cake of the waste sludge using CPAM composites with varying MMT contents (approximately 0–15 wt% MMT) were shown in Figure 10. The dosage of the composites was 0.06 g L−1. It revealed that SRF of the waste sludge presented decreasing trend first, then increased rapidly with MMT content, with the minimum value being achieved for a 5 wt% MMT content, and the increasing floc size and aggregation rate can be observed. This synergistic effect through the charge neutrality and bridging action of CPAM as well as the adsorption capacity of MMT provided bridging of primary flocs into larger and stronger ones, resulting in an enhanced dewaterability of waste sludge. Excessive MMT reduced the molecular weight and charge density of CPAM, and the relative content of CPAM became lower, which weakened the bridging ability and the electrostatic charge neutralizing ability of the polymer, resulting in a decreasing dewaterability of waste sludge.
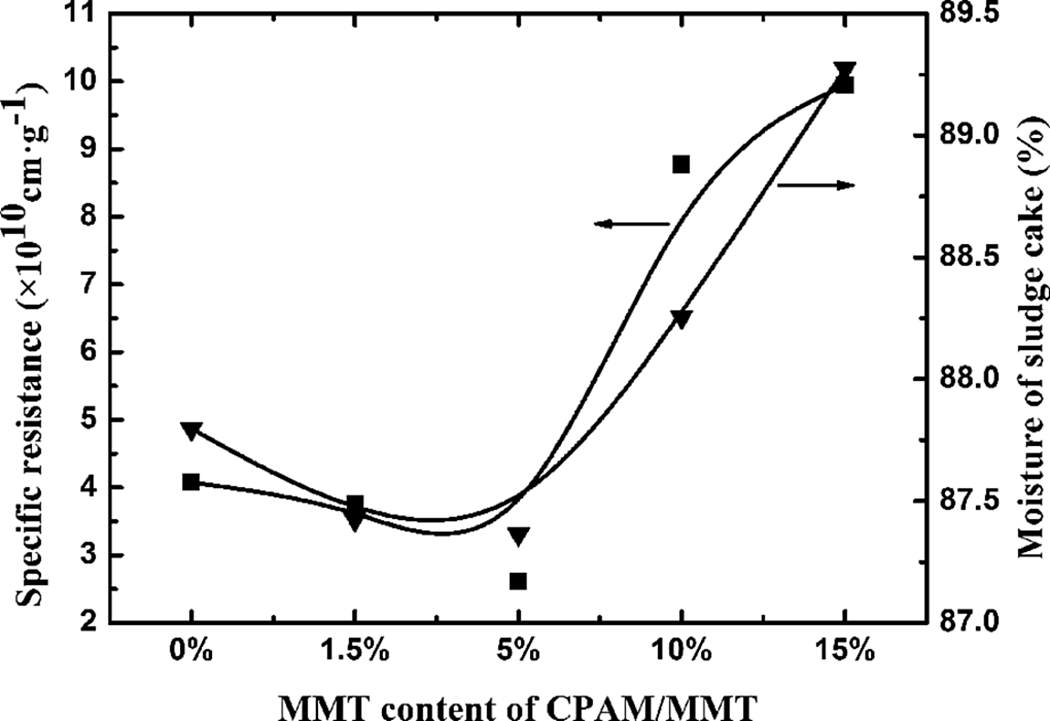
SRF of waste sludge and moisture of sludge cake by using CPAM composite with different MMT content. SRF: specific resistance to filtration; CPAM: cationic polyacrylamide; MMT: montmorillonite.
The moisture of sludge cake by CPAM composites with different MMT content displayed the same trend with SRF.
The UV transmittance and residual turbidity of the supernatant of the waste sludge settling with CPAM composite with different MMT content are shown in Figure 11. The UV transmittance of the supernatant of the waste sludge initially increased to a maximum at 5 wt% MMT content and then decreased slightly with increasing MMT content. The turbidity of supernatant of waste sludge displayed the opposite trend with MMT content.
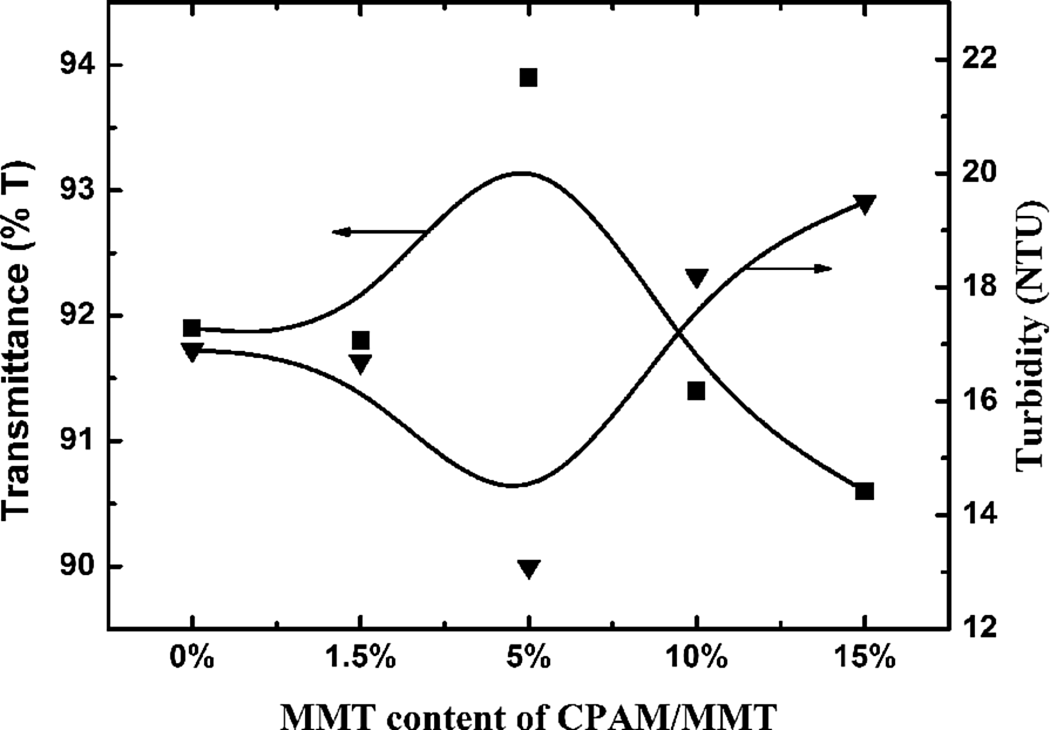
UV transmittance and residual turbidity of supernatant of waste sludge settling for 30 min with different MMT content. UV: ultraviolet; MMT: montmorillonite.
MMT had a very large specific surface area, which could adsorb the suspended sludge particles and bridging the organic materials in the out layer of the waste sludge. At high dosage of MMT, the attachment sites of sludge particles were used up and the nonadsorbing MMT remained in the aqueous phase, resulting in a relatively low UV transmittance and high turbidity.
Effect of CPAM/MMT dosage on dewaterability of waste sludge
Using the sample of CPAM/5 wt% MMT, the effect of the dosage of CPAM/MMT composite on the dewaterability of the waste sludge was investigated (Figure 12). The result showed that SRF and the moisture values of sludge cake decreased with increasing dosage of CPAM/MMT composite. A deficient dosage of the CPAM/MMT composite cannot make all the sludge particles flocculate to compact sludge flocs, and most of the restricted water still remained in the interior of the small flocs. With increasing dosage of CPAM/MMT composite, the flocs became less porous, with less internal gaps, and hence the space for water decreased, so the filter cake structure would be more compact.
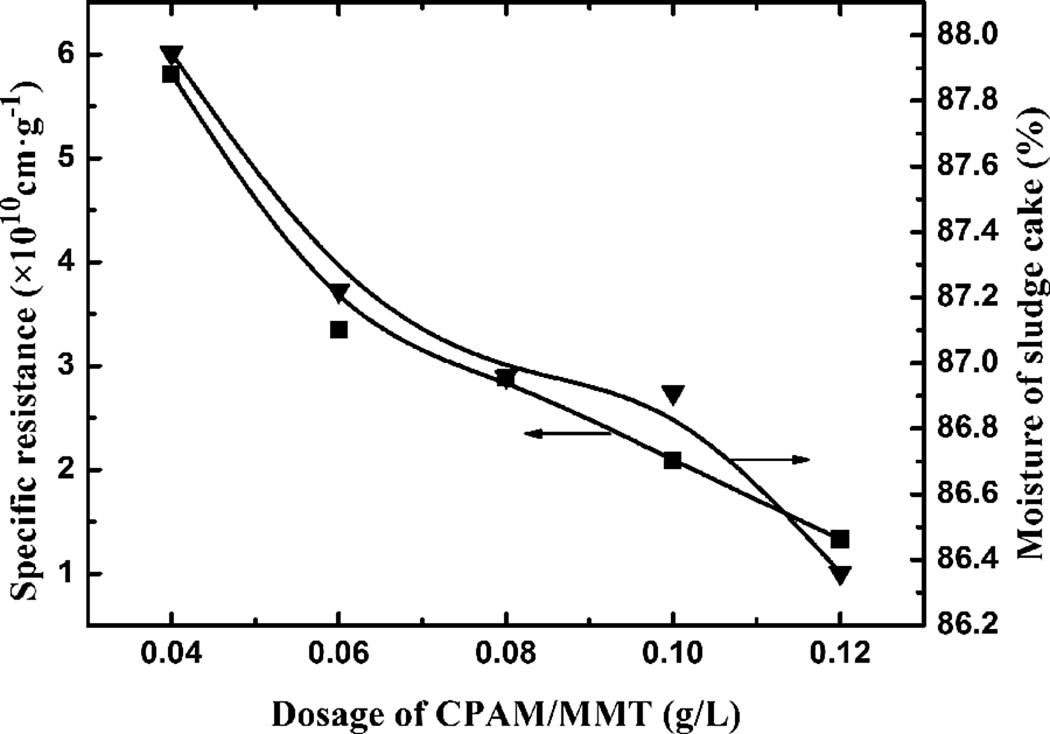
SRF of waste sludge and moisture of sludge cake using CPAM composite with 5 wt% MMT and different dosages. SRF: specific resistance to filtration; CPAM: cationic polyacrylamide; MMT: montmorillonite.
The UV transmittance and residual turbidity of supernatant of waste sludge settling with different dosages of CPAM/MMT (Figure 13) revealed that the turbidity of the supernatant substantially increased with increasing dosage of CPAM/MMT composite. For high dosage of MMT, excessive MMT remained in the liquid phase, resulting in a relatively low UV transmittance and high turbidity.
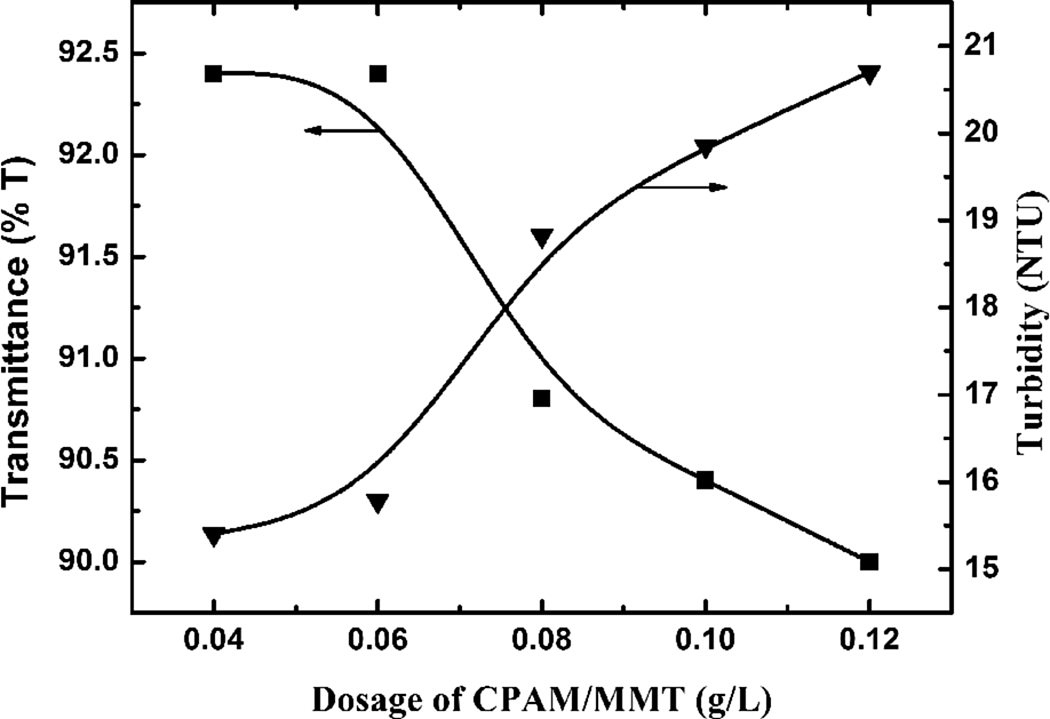
UV transmittance and residual turbidity of supernatant of waste sludge settling for 30 min with 5 wt% MMT and different dosages. UV: ultraviolet; MMT: montmorillonite.
Conclusions
The composites of CPAM/MMT were synthesized by in situ polymerization to be applied in dewatering of waste sludge. The molecular weight and cationic degree of the composite decreased with increasing MMT content. The hydrogen bonding between –OH groups of MMT and –NH groups of CPAM was confirmed using FTIR analysis. Compared with CPAM, a relatively high G′ value and low tan δ values can be achieved for CPAM/5 wt% MMT composite. At lower MMT content, the melting temperature of the composite decreased, and as the content of MMT increased, the melting temperature of the composite became higher. The XRD peak intensity of MMT at 2θ = 6.52° became weak and d-spacing increased, implying that the MMT platelets may be in an intercalated state in CPAM matrix, which was also confirmed by TEM analysis. Addition of MMT reduced SRF of waste sludge and the moisture of sludge cake, and the minimum value can be achieved at 5 wt% MMT content, while the UV transmittance of the supernatant of the waste sludge was also improved, and the turbidity was reduced. This synergistic effect of the two components in the composite provided charge neutrality, adsorption capacity, and bridging of primary flocs into larger and stronger ones, resulting in an enhanced dewaterability of the waste sludge.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Support Program of Sichuan Province (China) (2012FZ0012).
