Abstract
Composites of acid-functionalized multi-walled carbon nanotubes (MWCNTs) that reinforced poly(amide–imide) (PAI) were developed. The obtained composites containing 5, 10, and 15 wt% MWCNT–COOH exhibited a relatively good dispersion on the macroscopic scale. MWCNT/PAI composite films have been prepared by casting a solution of precursor polymer containing MWCNTs into a thin film, and its tensile properties were examined. The mechanical and thermal properties of the composites were investigated. The incorporation of MWCNTs–COOH improved the mechanical properties of the composites compared with that of neat PAI. An improvement in thermal properties of the MWCNT/PAI composites was also observed. Fourier transform infrared spectroscopy, powder x-ray diffraction, scanning and transmission electron microscopies were also used to evaluate the MWCNT/PAI composite system.
Keywords
Introduction
The field of carbon nanotubes (CNTs) was first started to materials science in 1991. 1 Today, CNTs have greater potential applications than the clay in the industry since they possess unique structure and properties. 2 They are thought to be perfect candidates for polymer reinforcement due to their high aspect ratio, nanoscaled dimensions, low density, stiffness, tensile strength, outstandingly high electrical and thermal conductivity, and damp properties. 3,4 CNT/polymer composites have many exceptional properties such as light weight, good and simple processability, and improved performance at low loadings. 5 However, it is very difficult to obtain a uniform dispersion of CNTs in the polymer matrix due to the naturally present poor compatibility between CNTs and the polymer matrix. To overcome this limitation, one approach is to take advantage of the oxidative formation of carboxyl functionalities and subsequently to graft organic moieties onto the CNTs before using them as fillers in the polymer matrix. 6 –10 However, few studies have been focused on improving the dispersion of CNTs via the synthesis of polymer with pendent hydroxyl groups for the matrix polymer itself.
Currently, four processing methods are in general employed to prepare CNT/polymer composites: direct mixing, in situ polymerization, solution compounding, and melt blending method. 11 The most chief and simple method for producing CNT/polymer composites is solution blending, where the CNTs are dispersed in a solvent and mixed with the polymer matrix through energetic agitation. 12
Preparation of copolymers with the introduction of flexible linkages, such as amide, ether, ester, and sufide linkages between the aromatic rings of the main chain, is an effective way to make polymers more tractable. Poly(amide–imide)s (PAI)s are a well-known class of polymers that possess desirable characteristics with the merits of both polyamides and polyimides, for example, high thermal stability, good mechanical properties, ease of processing, and chemical resistance to harsh solvents. 13,14 Compared with corresponding polyimides, these polymers show good solubility in highly polar solvents, lower glass transition temperatures, and easier processability. They are being used in many engineering applications such as molded parts for the space shuttle, engine parts of world-class racing cars, and many other critical components. 15 –19 PAI can be synthesized from various aromatic monomers containing anhydride, carboxylic acid, and aromatic diamine by condensation reaction. A common synthetic route to PAIs is the amide-forming reaction from imide-containing monomers such as dicarboxylic acids or diamines. 20 –25
In this study, acid-functionalized multi-walled CNT (MWCNT)-reinforced PAI composites have been made from dispersion by ultrasonication and conventional solvent casting. We have investigated a series of dopamine-bearing PAI composites containing a range of concentration of MWCNTs aiming to develop composite films and understand the reinforcement contributed by the MWCNTs. The thermal and mechanical properties, including the elastic modulus and tensile strength, as a function of MWCNT content have been tested. The microstructure of the composites has also been observed.
Experimental
Materials
A carboxyl-modified MWCNT (diameter 8–15 nm, length approximately 50 μm, and purity > 95 wt%), manufactured by a thermal chemical vapor deposition process, was purchased from Neutrino Co. (Iran). Other chemicals used in this study were purchased from commercial sources: Fluka Chemical Co. (Switzerland), Aldrich Chemical Co. (Milwaukee, Wisconsin, USA), and Merck Chemical Co. (Germany). Dopamine hydrochloride (3-hydroxytyraminium chloride), 3,5-dinitrobenzoylchloride, N,N′-dimethylacetamide (DMAc), N-methyl-2-pyrrolidone, and propylene oxide from Merck were used for the synthesis of mediators. Propylene oxide was used as acid scavenger. N,N′-dimethylformamide (DMF) (d = 0.94 g cm−3 at 20°C) and DMAc as solvent (d = 0.94 g cm−3 at 20°C) were distilled over barium oxide under reduced pressure. Other reagents were used without further purification.
Methods
Fourier transform infrared (FTIR) spectra of the composites were recorded with a Jasco-680 (Tokyo, Japan) spectrometer at a resolution of 4 cm−1, and they were scanned at a wave number range of 400–4000 cm−1. Band intensities are assigned as weak (w), medium (m), strong (s), and broad (br). Vibration bands were reported as wave number (per centimeter). Proton nuclear magnetic resonance ( 1 H NMR) spectra were recorded on a Bruker (Rheinstetten, Germany) Avance 500 instrument at room temperature in deuterated dimethylsulfoxide (DMSO-d6). Multiplicities of proton resonance were designated as doublet (d) and mult (m). The chemical shifts were reported in parts per million with respect to the references. Elemental analysis was performed in an Elementar Analysensysteme GmbH (Hanau, Germany). Inherent viscosity was measured by using a Cannon Fenske Routine Viscometer (Mainz, Germany) at the concentration of 0.5 g dL−1 in DMF at 25°C. Optical specific rotation was measured at the concentration of 0.5 g dL−1 in DMF at 25°C using a quartz cell (1.0 cm) with a Jasco polarimeter (JASCO Co. Ltd, Tokyo, Japan). Thermal stability of the MWCNT/PAI composites was evaluated by recording thermogravimetric analysis (TGA)/derivative thermogravimetric (DTG) traces (STA503 win TA TGA, Bahr-Thermoanalyse GmbH, Hüllhorst, Germany) under nitrogen atmosphere (flow rate 60 cm3 min−1). A heating rate of 20°C min−1 and a sample size of 10 ± 2 mg were used in each experiment. The x-ray diffraction (XRD) patterns of the polymer and composites were recorded using an x-ray diffractometer (Bruker, D8ADVANCE, Rheinstetten, Germany) with a copper target at 40 kV and 35 mA and copper Kα λ = 1.54 Å in the range 10–80° at the speed of 0.05° min−1. The dispersion morphology of the MWCNTs on the PAI matrix was observed using field-emission scanning electron microscopy (FESEM, HITACHI S-4160, Tokyo, Japan). Transmission electron microscopy (TEM) images were obtained using a Philips CM 120 (The Netherlands) microscope with an accelerating voltage of 100 kV. The mechanical properties were carried out on a universal testing machine M350/500 (Testometric, Mainz, Germany), according to ASTM D 882 standards, at RT and the tensile rate of 12.5 mm min−1 until/to a deformation of 20% and then at a speed of 50 mm min−1 at break. The tensile strength and tensile modulus were obtained from this measurement. Preparation of MWCNT/PAI composites was carried out on an ultrasonic XL-2000 SERIES (Misonix, Raleigh, North Carolina, USA). Ultrasonic irradiation was performed with the probe of the ultrasonic horn immersed directly in the mixture solution system with the frequency of 2.25 × 104 Hz and power 100 W.
Monomer synthesis
3,5-Diamino-N-(3,4-dihydroxyphenethyl)benzamide (4) as a diamine monomer was prepared according to our previous work and is shown in Figure 1. 26
N-Trimellitylimido-S-valine (5) was prepared according to our previous work. 27
Polymer synthesis
A mixture of 0.10 g (3.43 × 10−4 mol) of N-trimellitylimido-S-valine (5), 0.098 g (3.43 × 10−4 mol) of diamine 4 and 0.44 g of tetrabutylammonium bromide (TBAB; 13.72 × 10−4 mol) was ground until a powder was formed. After the mixture was completely ground, it was transferred into a 25-mL, round-bottomed flask and then 0.36 mL (13.72 × 10−4 mol) of triphenyl phosphite (TPP) was added to the mixture which was heated until a homogeneous solution was formed. Then, the solution was stirred for 12 h at 120°C, and the viscous solution was precipitated in 15 mL of methanol. The orange solid was filtered off and dried to give 0.162 g (85%) of PAI6. The optical specific rotation was measured
FTIR (potassium bromide (KBr), cm−1): 3416 (m, br, NH and OH stretching), 3114 (w, C–H aromatic), 2963 (w, C–H aliphatic), 2927 (w, C–H aliphatic), 1776 (m, C=O imide, asymmetric stretching), 1717 (s, C=O imide, symmetric stretching), 1647 (m, C=O amide, stretching), 1599 (s), 1550 (m), 1446 (s), 1376 (m, CNC axial stretching), 1198 (m, CNC transverse stretching), 1073 (m), 865 (m), 766 (m), 725 (s, CNC out-of-plane bending), 683 (w).
1 H NMR (400 MHz, DMSO-d6, ppm): 0.91 (t, 3H, CH3, distorted), 1.29 (d, 3H, CH3, J = 7.20 Hz), 1.50 (m, 2H, CH2), 2.59 (m, 1H, CH), 2.87 (t, 2H, CH2, distorted), 3.15 (t, 2H, CH2, distorted), 4.63-4.65 (d, 1H, CH, J = 8.40 Hz), 6.45–6.47 (d, 1H, Ar–H, J = 8.40 Hz), 6.61–6.63 (d, 1H, Ar–H, distorted), 7.66 (s, 1H, Ar–H), 7.71 (s, 1H, Ar–H), 7.94 (s, 1H, Ar–H), 8.00 (s, 1H, Ar–H), 8.10 (d, 1H, Ar–H, distorted), 8.46 (s, 1H, Ar–H), 8.51–8.53 (d, 1H, Ar–H, distorted), 8.76 (s, 1H, NH), 10.15 (s, 1H, OH), 10.23 (s, 1H, OH), 10.78 (s, 1H, NH), 10.85 (s, 1H, NH).
Elemental analysis: calculated for (C30H28N4O7) n : C, 64.74%; H, 5.07%; N, 10.07%. Found: C, 64.35%; H, 4.96%; N, 10.29%.
Fabrication of the MWCNT/polymer composite films
To attain uniformly mixed MWCNT/PAI solutions with desired weight percentages of MWCNT, a two-step pathway was used. First, two stock solutions were prepared: PAI was dissolved and MWCNTs were separately dispersed in a DMAc solution with stirring for 1 day at 30–40°C. Then, two stock solutions were mixed to achieve the desired weight percentages of MWCNTs from 5 to 15 wt%. The MWCNT/PAI solutions were stirred for 1 day at 30–40°C and then ultrasonicated in water bath for 1 h. To remove the DMAc solvent, MWCNT/PAI solutions were poured into uncovered preheated glass petri dishes and uniformly heated at 60°C for 1 day; then, the semidried film was further dried in vacuum at 160°C for 8 h, for the removal of the residue solvent, and a solid film was formed. Curly films formed after evaporation of DMAc and could be easily lifted from the glass petri dishes. Freestanding polymer films 30–40 μm thick were then peeled from the glass plate and were subjected to different tests. Because MWCNTs are black in color, the films containing more MWCNTs look darker: film colors varied from tan (0% MWCNT) to light gray (5% MWCNT) to dark black (10 and 15% MWCNT). Uniform color was observed, which is an indication of good distribution of MWCNTs in the polymer matrix.
Results and discussion
Synthesis of PAI
In the course of our study on the application of green chemistry principles in polymerization, we wish to report a fast, simple, safe, and efficient method for the step-growth polymerization of natural S-valine-based diacid 5 with an aromatic dopamine-based diamine (4) in molten TBAB in the presence of TPP (Figure 1). Molten TBAB salt was selected because it proved to be the most valuable among those employed in our recent works.
28
Run in molten salt was carried out by thermal heating technique. The entire polycondensation reaction readily proceeds in a homogeneous solution and after work up tough and stringy precipitates was formed. The inherent viscosity of the synthesized PAI was 0.48 dL g−1, and the yield was 85%. The resulting polymer showed optical rotation, which indicated that the polymer is optically active and chirality was introduced into the backbone of the polymer. The optical specific rotation of this polymer was
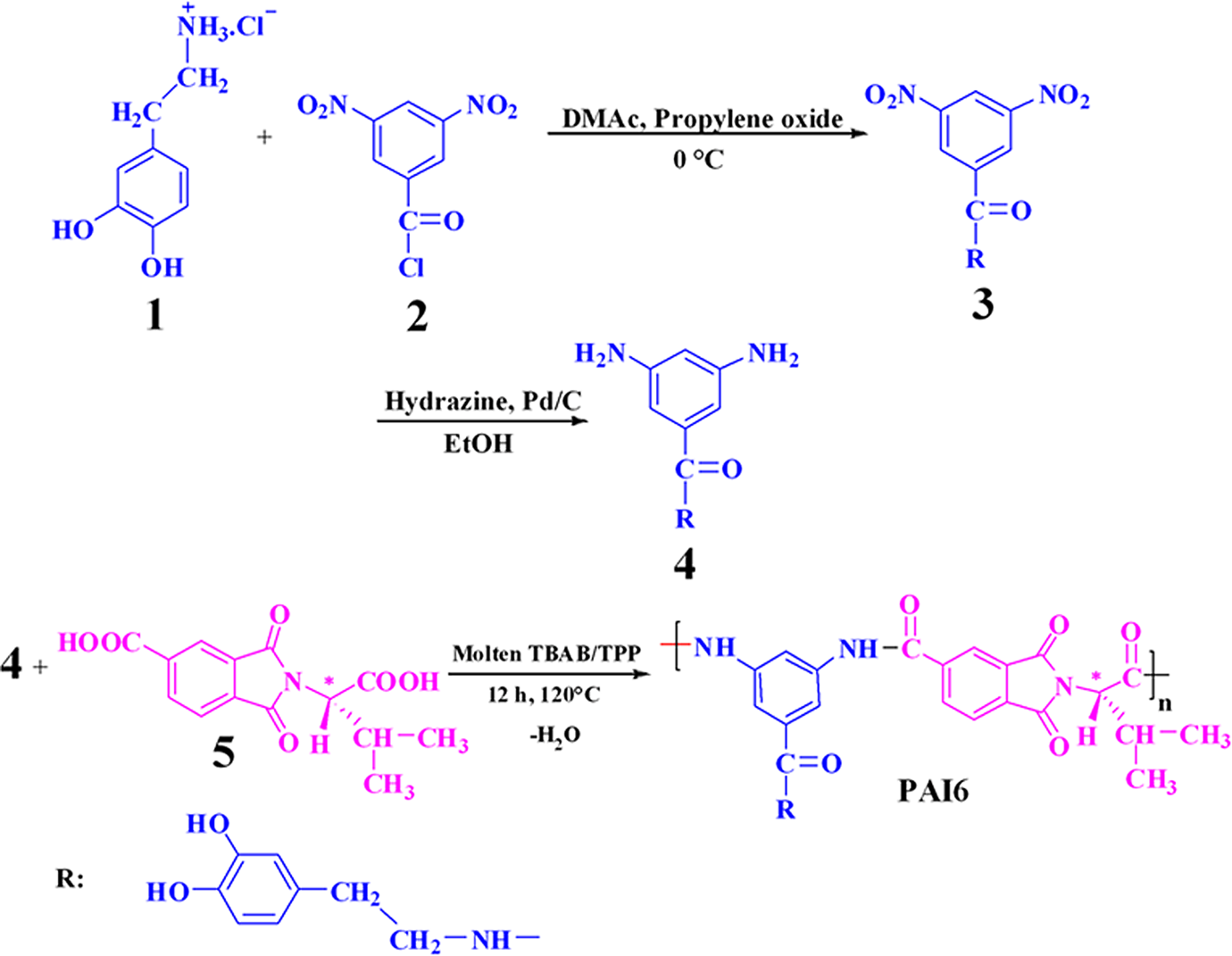
Preparation of diamine 4 and the PAI.
Fabrication of the composite films
The dispersion of the carboxylic acid-modified MWCNTs in the 5, 10, and 15 wt % solutions of PAI in DMAc was achieved by a vigorous stirring with a speed of 15,000 r min−1 for 1 day using a homogenizer, followed by utrasonication process for 1 h to form a new series of composites (Figure 2). The effective use of CNTs in composite applications depends on the ability to disperse the CNTs uniformly throughout the matrix without reducing their aspect ratio. Due to the van der Waals attraction, CNTs are held together as bundles and ropes. Therefore, they have very low solubility in solvents and tend to remain as entangled agglomerates. To overcome the difficulty of dispersion, several mechanical/physical methods such as ultrasonication and chemical modification through functionalization were used. 6 –10 So, carboxylated MWCNTs were used in this study. The lower level of aggregation in the modified CNTs can be attributed not only to the presence of functional groups such as carboxyl groups but to their shorter length and aspect ratio. This transformation should contribute positively to the good dispersion of CNT in the PAI matrix. Moreover, the introduction of hydroxyl functional groups into the side chain of aromatic polymer performs a hydrogen interaction with modified CNTs and a composite based on hydrogen interaction, by which PAI chains were tightly attached to the surface of MWCNT–COOH, can be resulted. The possible interactions between PAI chains and functionalized MWCNTs are described in Figure 3.
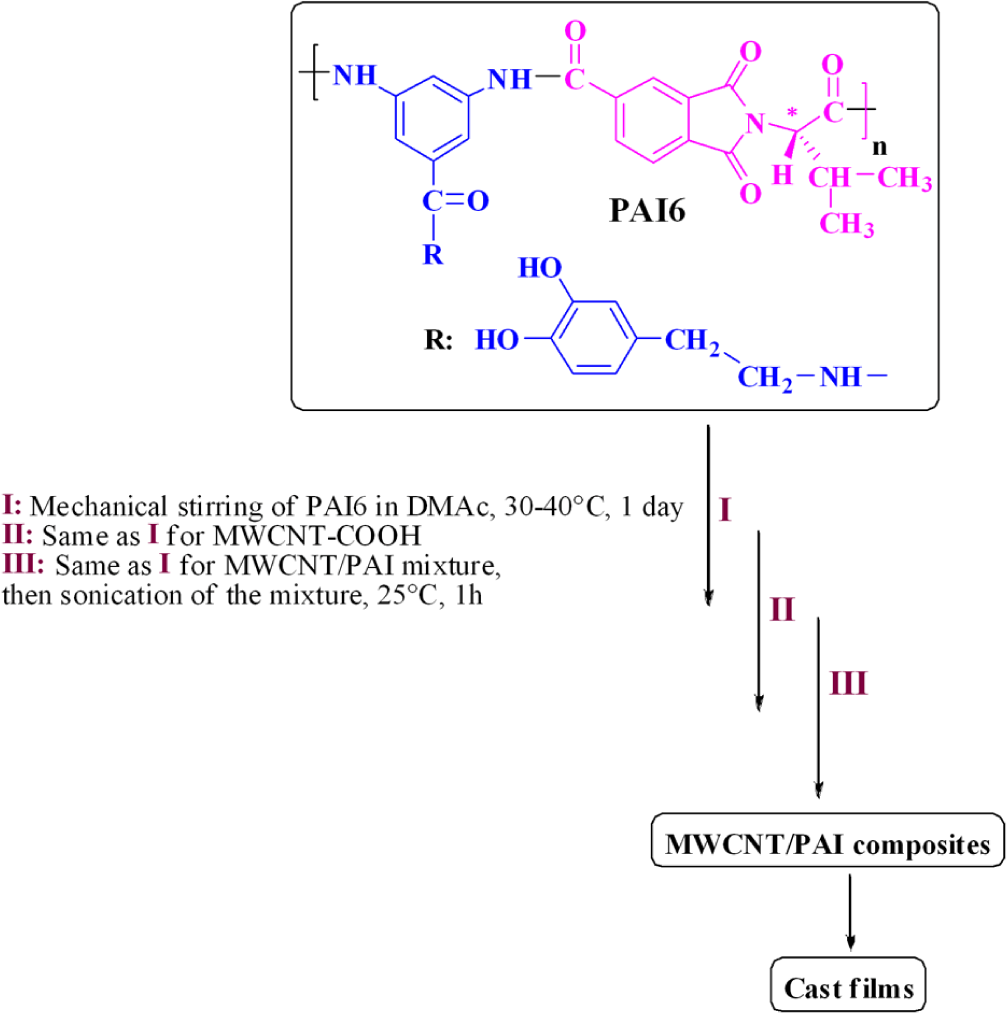
Schematic representation of the preparation process of MWCNT/PAI composites. MWCNT: multi-walled carbon nanotube; PAI: poly(amide–imide).
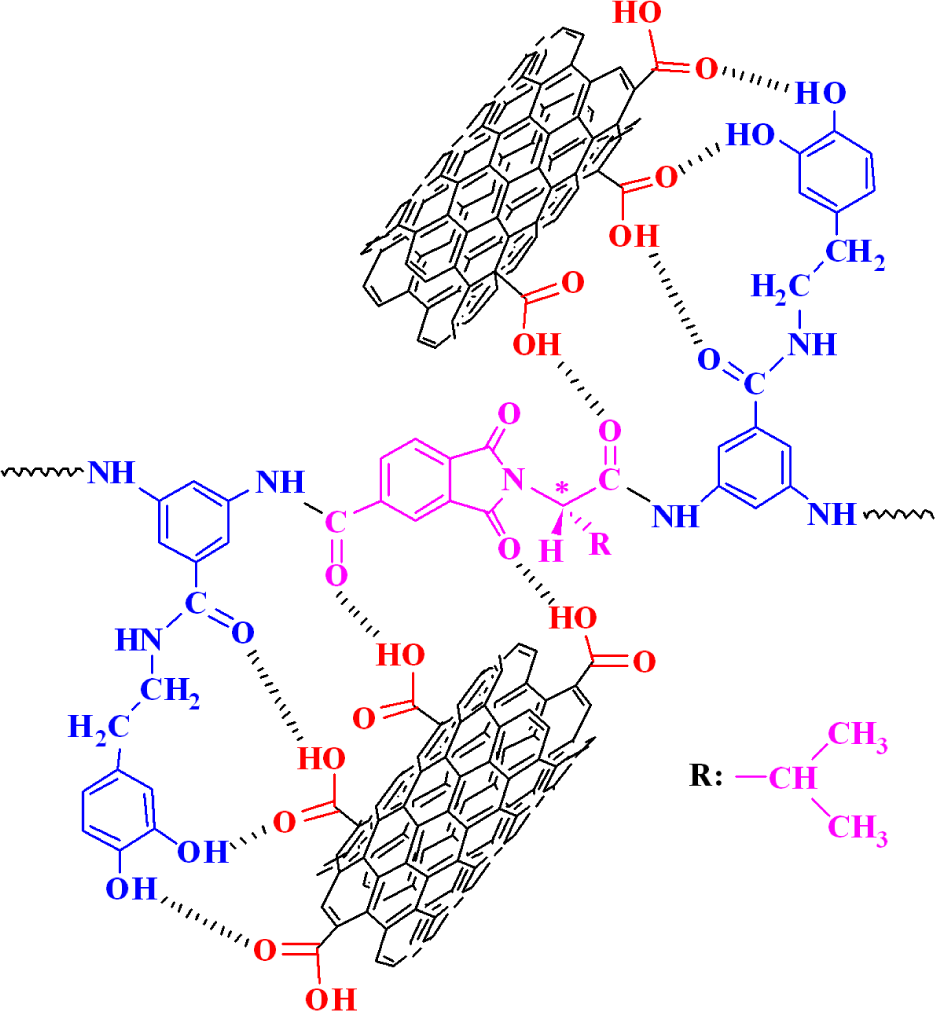
Representation of possible hydrogen bonding between the MWCNTs –COOH and the PAI matrix.
Characterization of the composites
Spectral data
The FTIR spectrum of PAI (Figure 4(a)) showed absorptions around 3392 cm−1 (N–H and O–H), 3101 cm−1 (aromatic C–H stretching), 2962 and 2930 cm−1 (aliphatic C–H stretching) and two overlapped carbonyl (imide and amide’s C=O) absorptions at 1777, 1719, and 1648 cm−1, respectively. Absorption at 1376 and 726 cm−1 was confirmed the presence of the imide heterocycle in this polymer. The functionalized MWCNTs were also characterized by FTIR. The spectrum of a pristine MWCNTs/KBr pellet (Figure 4(e)) shows a strong, broad absorption band centered at 3433 cm−1, which is attributed to the O–H stretching bands of carboxylic acid moieties from the surface of MWCNTs. The small peak around 2923 cm−1 is ascribed to aliphatic sp 3 C–H of MWCNTs. 29 The presence of CNTs in the polymer matrix showed very little changes in the FTIR spectrum, presumably due to the low MWCNT composition and the weak vibration signals of MWCNTs (Figure 4(b) to (d)). The structure of neat PAI was also identified by 1 H NMR spectroscopy. In the 1 H NMR spectrum of this polymer, appearances of the N–H protons of amide groups at 8.75, 10.77, and 10.84 ppm as three singlet peaks, and OH groups at 10.22 and 10.31 ppm as two singlet peaks, indicate the presence of amide groups in the polymer’s side chain as well as main chain and hydroxyl groups in the polymer’s side chain. The resonance of aromatic protons appeared in the range of 6.47–8.65 ppm. The proton of the chiral center appeared as doublet at 4.73 ppm.
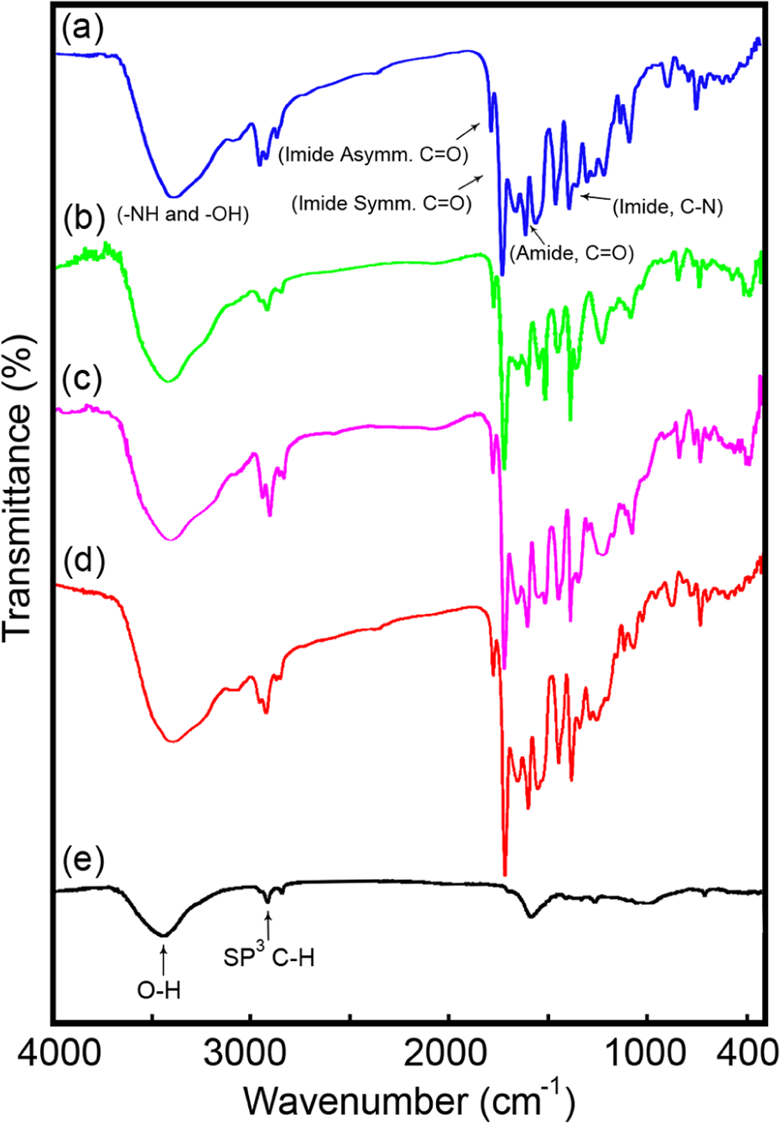
FTIR spectra of PAI and the MWCNT/PAI composites: (a) PAI; (b) MWCNT/PAI 5 wt%; (c) MWCNT/PAI 10 wt%; (d) MWCNT/PAI 15 wt%; and (e) carboxyl-modified MWCNT.
XRD analysis
XRD patterns of the MWCNT/PAI composite materials are presented in Figure 5. MWCNT and pristine PAI were also investigated and compared. For the MWCNTs, two peaks appear at 2θ = 26 and 43°, which correspond to the interlayer spacing d(002) and d(100) reflection of the CNT. 30 For the neat PAI, the weak reflection centered at a 2θ value around 20° is characteristic of the amorphous polymer. Composite samples showed similar XRD patterns to the pure polymer when the MWCNT composition was 5 wt%. For the MWCNT content higher than 5 wt%, the MWCNT/PAI composites exhibit peaks of PAI and MWCNT, as shown in Figure 5. The reflections at 2θ = 20 and 27° become slightly increased, when the MWCNT composition is increased to 15 wt%.
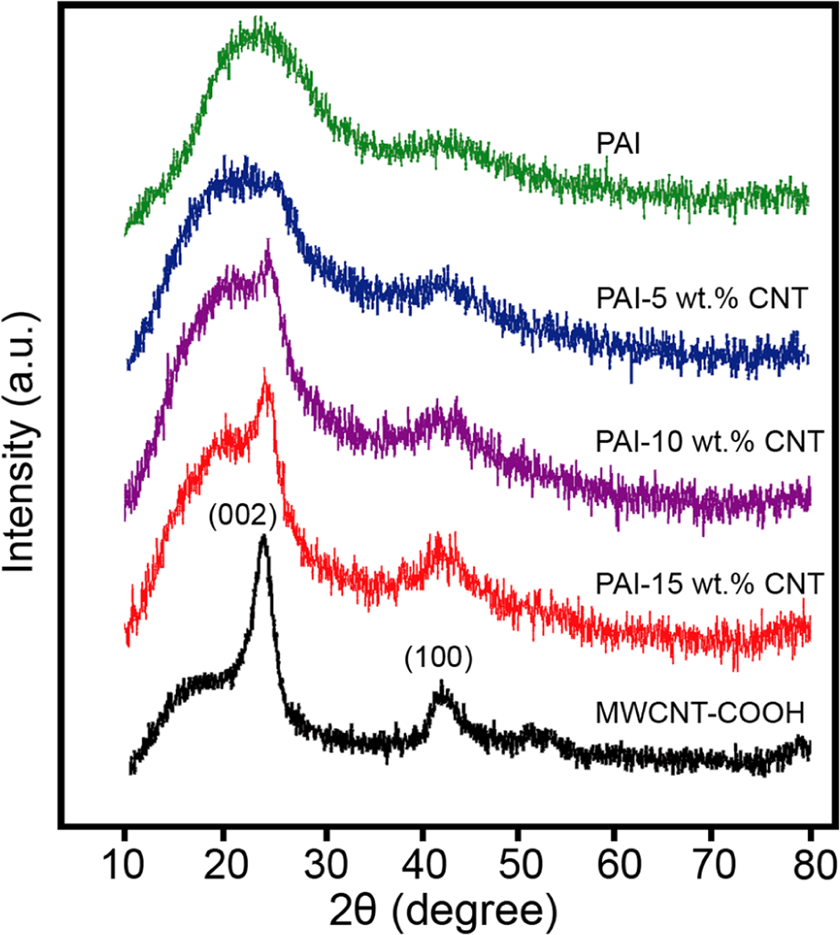
XRD spectra of MWCNT and MWCNT/PAI composites.
Thermal analysis
Figure 6 shows the thermal decomposition of PAI and series of composites at a rate of 20°C min−1 under nitrogen atmosphere. The thermal stability of the polymer and composites was studied on the basis of 5 and 10% weight losses (Td5 and Td10, respectively) of the samples and the residue at 800°C. As can be seen from TGA curves and their analysis results in Table 1, the onset of decomposition temperature of the composites is higher than that of pure PAI and it shifts toward higher temperatures as the amount of MWCNT is increased. The Td10 of pure PAI and the MWCNT/PAI composites with increasing amount of MWCNT such as 5, 10, and 15 wt% are 350, 377, 388, and 423°C, respectively, as listed in Table 1. It is clear that the MWCNT/PAI materials improve thermal stability due to the incorporation of MWCNTs. The end temperature of decomposition is also retarded with increasing MWCNT content. The masses remaining at 800°C are almost entirely due to the remaining MWCNTs and are consistent with initial MWCNT loading. The weight percentage remaining after major degradation at 800°C was higher for composites than for neat PAI. This indicates that MWCNT reduces the degradation of PAI at high temperature as the effect was clearly seen in the curves. Therefore, it could be demonstrated that a small amount of MWCNT acts as effective thermal degradation resistant reinforcement in the PAI matrix, increasing the thermal stability of the MWCNT/PAI composites.
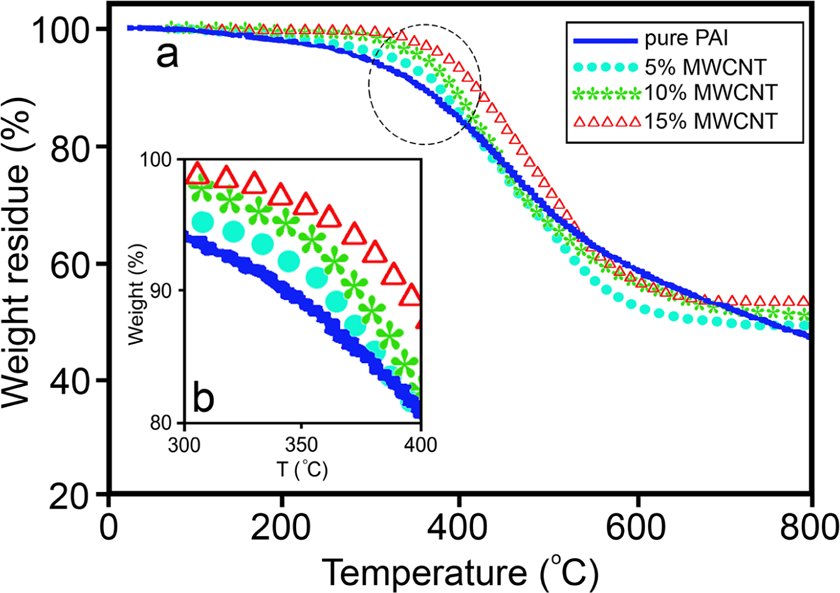
(a) TGA thermograms for PAI and the MWCNT/PAI composites under nitrogen atmosphere at a heating rate of 20°C min−1; (b) magnification of circled area in (a).
Thermal properties of PAI and the composites.
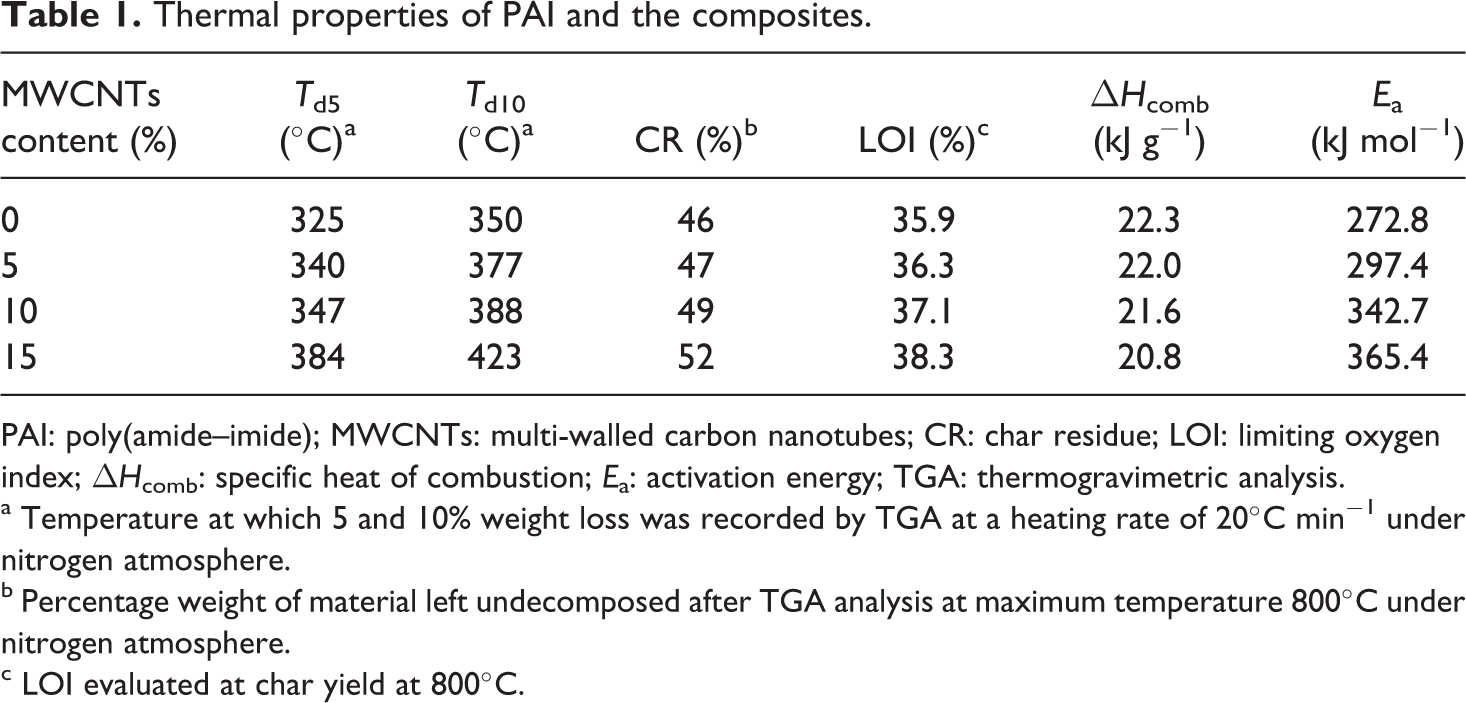
PAI: poly(amide–imide); MWCNTs: multi-walled carbon nanotubes; CR: char residue; LOI: limiting oxygen index; ΔHcomb: specific heat of combustion; Ea: activation energy; TGA: thermogravimetric analysis.
a Temperature at which 5 and 10% weight loss was recorded by TGA at a heating rate of 20°C min−1 under nitrogen atmosphere.
b Percentage weight of material left undecomposed after TGA analysis at maximum temperature 800°C under nitrogen atmosphere.
c LOI evaluated at char yield at 800°C.
Two interesting relationships have been found between the limiting oxygen index (LOI) and the parameters of the combustion process: char yield or char residue (CR) and heat of combustion. LOI also called critical oxygen index (COI) or oxygen index (OI), and it is defined as the minimum fraction of oxygen in a mixture of oxygen and nitrogen that will just support combustion (after ignition). On the other hand, OI methods describe the tendency of a material to sustain a flame.
According to van Krevelen,
31
there is a linear relationship between LOI and CR only for halogen-free polymers (equation (1)):

From this equation, a higher char yield will increase flame retardance. PAI and composites containing 5, 10, and 15 wt% had LOI values 35.9, 36.3, 37.1, and 38.3, respectively, which were calculated from their char yield. On the basis of the LOI values, such materials can be classified as self-extinguishing materials.
According to Johnson,
32
the LOI values of many common materials can be logically well predicted by the following expression (equation (2)):

where ΔHcomb is the specific heat of combustion in joules per gram. So, in the case of this polymer (PAI6) and MWCNT/PAI composites (5, 10, and 15 wt%) ΔHcomb is 22.3, 22.0, 21.6, and 20.8 kJ g−1, respectively.
Activation energy for the thermal decomposition, Ea, of the PAI and the composites can be estimated from the corresponding TGA curves using the integral method proposed by Horowitz and Metzger (equation (3))
33

where α is the decomposed fraction, and Ea is the activation energy of decomposition in kilojoules per mole. Tdm is the temperature at maximum rate of weight loss, and θ is the variable auxiliary temperature defined as
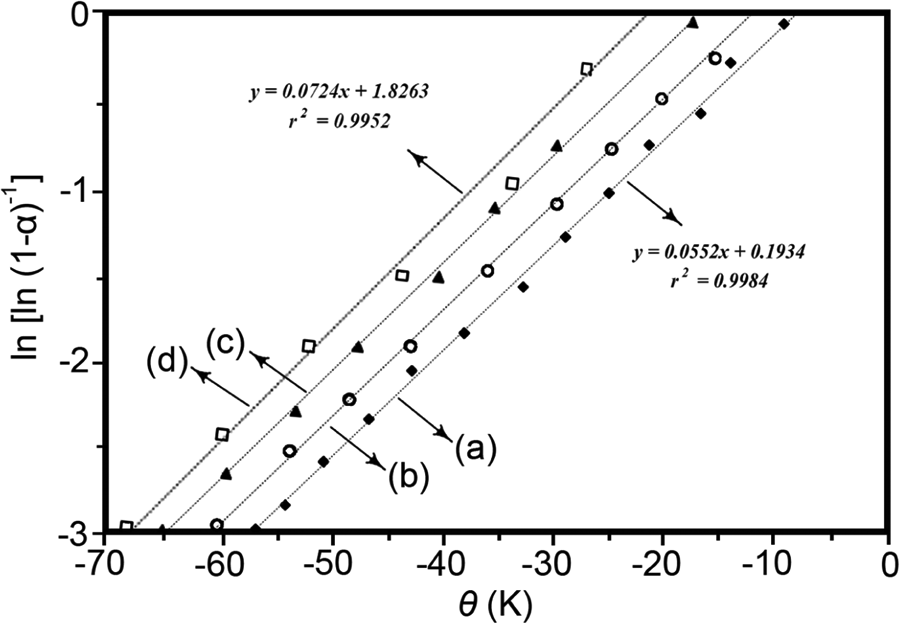
Plots of
Microscopic characterization
To characterize the morphology and dispersity of MWCNT in the composites, FESEM images of fracture surfaces of neat PAI and the composites were examined as displayed in Figure 8. The neat PAI copolymer showed a smooth fracture surface morphology (Figure 8(a)). FESEM observation revealed that PAI particles self-organized into nanopatterns. As can be seen from these images, the average diameter of polymeric particles is approximately 39 nm and shape of them is spherical. For MWCNT/PAI composites, MWCNTs are well dispersed and embedded in the PAI matrix without showing noticeable MWCNT aggregates (Figure 8(b) to (d)). In addition, the fracture surface of the MWCNT/PAI composites is relatively smooth without exhibiting the pullout of MWCNTs from the PAI matrix, and the boundary between MWCNT and the PAI matrix is not discernible clearly. These results strongly support that due to hydrogen bonding between MWCNT–COOH and hydroxyl groups in the side chains of the PAI, the MWCNTs are wrapped around PAI particles and MWCNTs are also mixed well with the PAI matrix, eventually leading to good dispersity and compatibility of the MWCNTs in the PAI matrix. The representative TEM images of the MWCNT/PAI composites are shown in Figure 9 at different scales. In general, the drawbacks related to the homogeneous dispersion of the CNTs in the polymer matrix resulted from intrinsic van der Waals attractions between the individual CNTs in combination with high aspect ratio and large surface area, making it difficult for the CNTs to disperse in the polymer matrix. The interfacial adhesion between the CNTs and the polymer matrix plays an important role in improving the properties of the composites. As shown in Figure 9(a) to (d), the MWCNT was dispersed well in the composites, which was explained by the fact that the MWCNT stabilizes their dispersion by good interactions with the PAI matrix, resulting from the increased polarity by the functional groups formed on the surfaces of the MWCNT as well as good interactions of the carboxyl groups with the hydroxyl groups of the PAI matrix.

FESEM images of a fracture surface of the produced materials: (a) pristine PAI and the composites containing (b) 5 wt%-, (c) 10 wt%-, and (d) 15 wt%-modified MWCNTs at different magnifications.
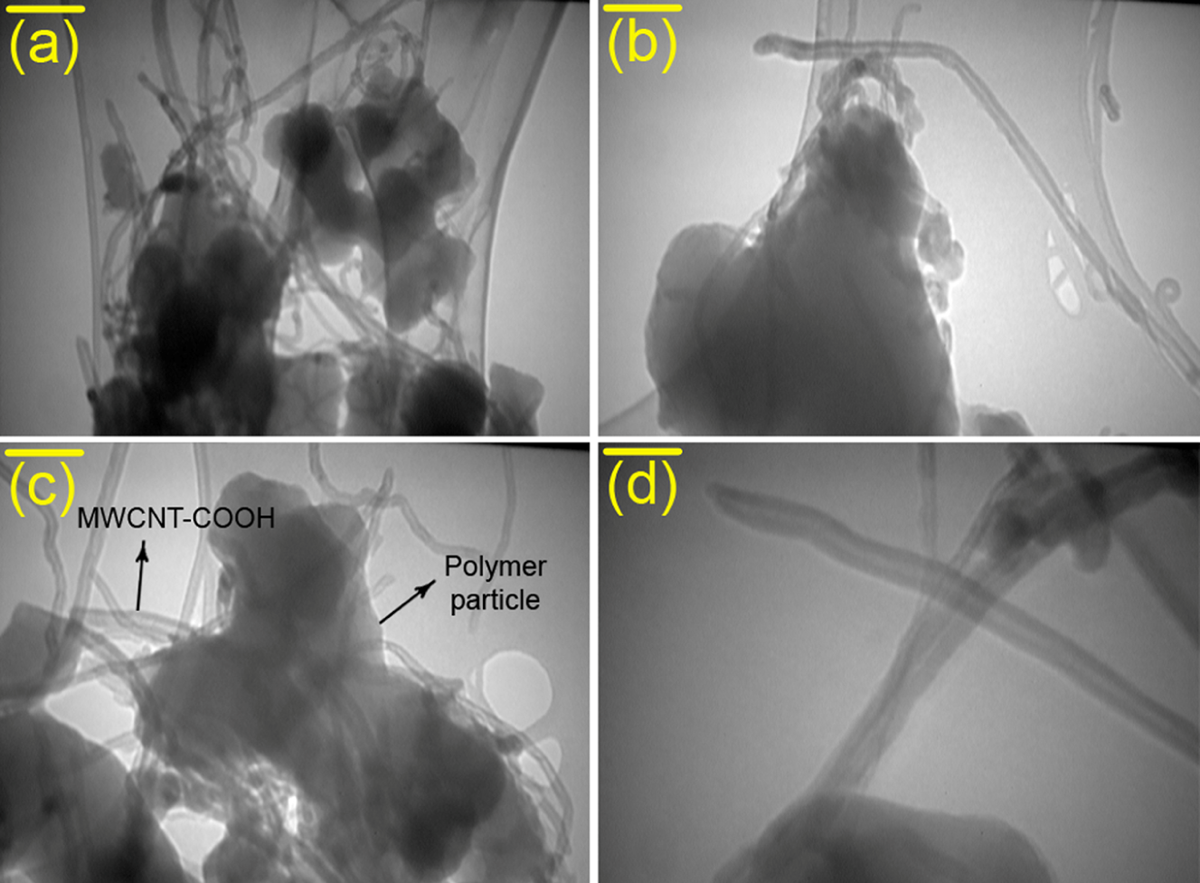
TEM micrographs of MWCNTs dispersed in composites containing 5 wt% of MWCNTs: (a) scale bar = 120 nm, (b) scale bar = 70 nm, (c) scale bar = 70 nm, and (d) scale bar = 40 nm.
Tensile properties
It is known that the nanoparticles or nanomaterials benefit the mechanical properties when they are well distributed in the polymer matrix. 37 In this study, MWCNTs were found to greatly improve the mechanical properties of PAI due to the nanoreinforcing effect of MWCNT with high aspect ratio. The resultant stress–strain curves are shown in Figure 10. It can be seen that, in comparison with neat PAI film, the MWCNT/PAI composite films exhibit higher Young’s modulus and tensile strength. The Young’s modulus and tensile strength increased from 1.3 ± 0.2 GPa and 72.6 ± 2.7 MPa for neat PAI film to 2.8 ± 0.5 GPa and 108.3 ± 1.2 MPa, 3.0 ± 0.4 GPa and 114.4 ± 2.8 MPa, and 3.2 ± 0.1 GPa and 120.5 ± 1.4 MPa for MWCNT/PAI composite films with increasing the MWCNT feeding content from 5 to 10 and 15 wt%, respectively. This increasing effect of MWCNT on the tensile strength and tensile modulus of the composites is more significant at low MWCNT content when compared with high MWCNT content. As the loading of MWCNTs becomes higher (wt% = 10 and 15%), they began to agglomerate to a little extent (Figure 8(c) and (d)), causing the stress concentration phenomenon and preventing efficient load transfer to the polymer matrix. The value in 108.3 MPa for the sample with 5 wt% MWCNT loading is almost 36% higher than that of the pure PAI. In this study, the reinforcing efficiency of MWCNT–COOH is defined as the normalized mechanical properties of the composites with respect to those of pure PAI. The improvement in the mechanical properties of the MWCNT/PAI composites is attributed to the better interfacial interaction between the MWCNT and the PAI matrix as well as the better dispersion of the MWCNTs in the PAI matrix. The incorporation of the MWCNT–COOH and hydroxyl groups into the PAI matrix resulted in the good interfacial adhesion between the MWCNT–COOH and the PAI matrix. This interaction between the MWCNT–COOH and the PAI matrix is crucial for improving the mechanical properties of the composites. The elongation at break of the composite films was decreased with the introduction of MWCNT, indicating that the composites became somewhat brittle when compared with pure PAI because of the increased stiffness of the composites.
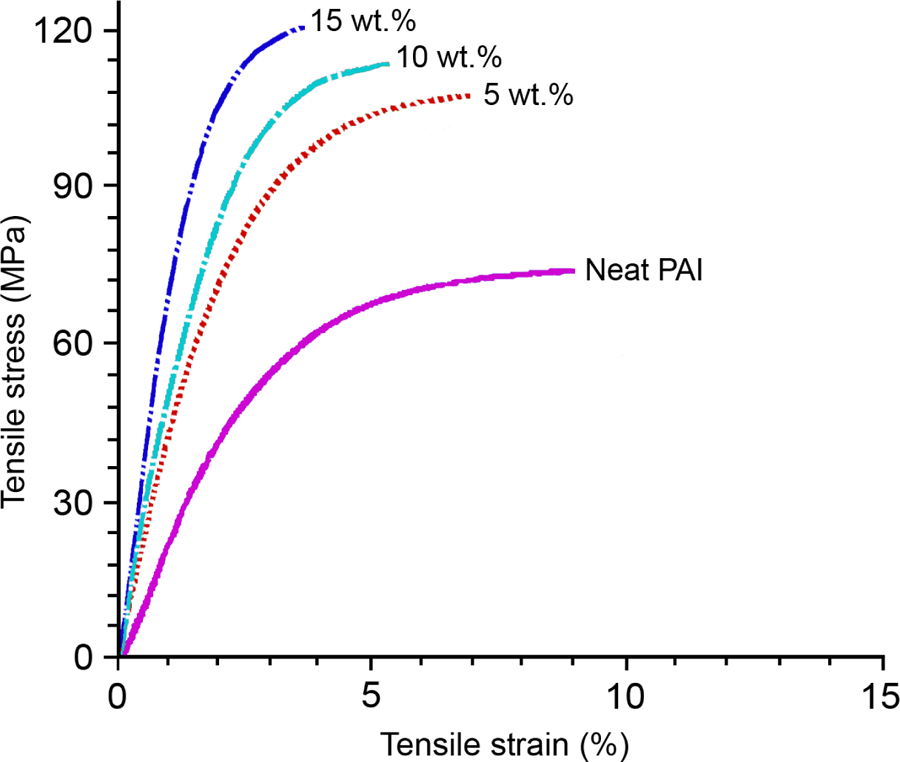
Variation in the tensile strength and Young’s modulus of the MWCNT/PAI composites with increasing amount of MWCNTs.
Conclusions
As an approach to the preparation of MWCNT/PAI composites, ultrasonically assisted solution compounding of a hydroxyl-containing PAI was carried out in the presence of MWCNT. The homogeneous dispersion of MWCNTs in the PAI matrix was visually confirmed by detailed microscopic observation. Due to the state of dispersion and the relative amount of MWCNT in the polymer matrix, composites displayed much better thermal stability compared with the pure PAI. Also, the Ea for thermal decomposition of the composites reflected that the incorporation of very small quantity of the MWCNT into the PAI matrix increased the thermal stability of the composites. The mechanical properties of the composites were significantly improved with the introduction of MWCNTs. For the MWCNT-reinforced composites, strong interfacial adhesion and uniform dispersion are more crucial factors for improving the mechanical properties of the composites. The contribution of amino acid as a biocompatible natural segment in the main chain of optically active PAI makes the obtained composites more susceptible for better biodegradation process. This study demonstrated that the thermal and mechanical properties of the composites are strongly dependent on the uniform dispersion of CNTs and the interactions between CNT and PAI, which can be improved using modified CNTs and the introduction of several functional groups in the polymer chains, providing a design guide of CNT-reinforced composites with a great potential for industrial applications. Addition of the MWCNTs to other polymers for improved composites properties is currently in progress in our laboratory.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the Research Affairs Division of Isfahan University of Technology (IUT), Isfahan, Islamic Republic of Iran, for the financial support of this work. Further financial support from National Elite Foundation (NEF) and Center of Excellency in Sensors and Green Chemistry Research (IUT) is gratefully acknowledged.
