Abstract
An investigation was carried out on the noncross-linked low-density polyethylene (LDPE) nanocomposite foams with polyethylene graft maleic anhydride (PE-g-MA) compatibilizer. Nanocomposite foams with similar densities were produced by extrusion method, and the effect of organophilic montmorillonite (OMMT) nanoclay (closite20A) content on cell microstructure, mechanical properties, thermal conductivity, and flame retardancy of samples were studied. X-ray diffraction (XRD) was used to investigate the nanoclay dispersion in LDPE matrix. In XRD patterns of LDPE nanocomposite foams, closite20A characteristic peak was not observed as an evidence of nanoclay intercalation–exfoliation in the polymer matrix. For increasing the compatibility of PE and nanoclay, PE-g-MA compatibilizer was used. Due to the presence of compatibilizer in the nanocomposite foams and decreasing the cell nucleation energy around the nanoclay, the average cell size was reduced, but the cell density and microstructure uniformity increased. Furthermore, the compatibilized nanocomposites showed lower thermal conductivity and burning rate than that of the nanocomposites without PE-g-MA compatibilizer and neat LDPE foams, which can be attributed to the cell size reduction as well as distribution of cells of narrow size in nanocomposite foams.
Keywords
Introduction
Many attempts have been conducted to develop foams that exhibit reduced cell size and distribution of cells of narrow size for many applications, such as packaging, textile, agriculture, and construction, due to the enhanced mechanical, physical, thermal, and morphological properties. 1 –5 There are many types of polymer foaming processes, such as extrusion, injection molding, compression molding, and batch foaming. Extrusion method has been known as an efficient and economical procedure to produce polymeric foams by blending blowing agent into molten polymer to create an equally high-pressure solution for instantaneous free expansion at the outside. 6,7 The basic steps in the process involve melting of the solid polymer, injection and dissolution of the blowing agent into the polymer melt, cooling down of the blowing agent saturated melt, foaming through cell nucleation, and finally stabilization of the resultant cell structure. The density of cells in the polyethylene (PE) foam can be controlled by incorporating the nucleating agent such as talc. The nucleating agent used in foam processing plays an important role in determining the foam’s cellular structure. Usually the nucleating agents are fine powders, which serve as solid surfaces for heterogeneous cell nucleation; they remain solid or thermally stable during the foaming process. The talc content is known to significantly affect the cell density of low-density polyethylene (LDPE) foam. According to the spontaneous nucleation theory, this situation is quite reasonable: the formation of a critical nucleus in the cavities of the talc particles is smaller than that in the bulk of polymer matrix. 6
In the past decade, polymer nanocomposites have been an extensively studied class of materials for a variety of applications due to their enhanced properties and performance. organophilic montmorillonite (OMMT) nanoclay structure has a dramatic performance in mechanical, thermal, and burning properties of polymer nanocomposites. 8,9 Burning properties of OMMT can improve LDPE foam flame retardancy; decreasing the cell size can improve thermal properties of the nanocomposites. Several attempts have been made to produce microcellular foams using continuous process and investigate the influence of incorporation of nanoparticles on the foaming process, properties, and cellular structure of the polymeric foams. 2,3 These properties include dimensional stability, cell density, cell size, cell size distribution, microstructure, mechanical properties, thermal behavior, and flame retardancy. 1,2 Incorporating a small amount of nanoclay (closite20A) to PE foam has obviously increased the Young’s modulus and decreased the thermal conductivity and burning rate compared to the neat PE foams. 4 Many studies have shown that the polar interactions of polymer and clay surface play a critical role in achieving nanoclay delamination and dispersion. For nonpolar polymers, such as PE, a polar compatibilizer such as polyethylene graft maleic anhydride (PE-g-MA) is commonly used to improve the compatibility and dispersion of nanoclay in the PE matrix. Intercalated and/or exfoliated structures can be created, according to the compatibilizer content, the blending procedure, and the processing conditions, which results in the improvement of the mechanical properties because of the enhanced interfacial properties. 5
In the literature there are some studies about PE foams and their nanocomposites. 6 –9 Most of these studies are about the cross-linked PE foams and PE nanocomposite foams, and much attention has not shown on noncross-linked PE foams. Noncross-linked PE foams have some advantages and disadvantages compared to the cross-linked PE foams. The most important advantages of noncross-linked PE foams are their recyclability and their production costs compared to the cross-linked foams. But they have some disadvantages such as lower dimensional and thermal stability, lower mechanical properties, coarser cell structure, and more flammability compared to the cross-linked PE foams.
This study tried to improve some properties of noncross-linked foams by incorporating a reinforcement agent such as organically modified MMT nanoclay (closite20A). In reinforced polymers, the dispersion and distribution rates of nanoparticles play a key role in the final properties of the nanocomposites. In this research, a compatibilizer (PE-g-MA) was added to the nanocomposites and its effects on the properties of the noncross-linked PE foams was examined and compared with uncompatibilized PE foams.
Experimental
Materials
The following materials were used for producing LDPE and LDPE/LDPE-g-MA compatibilized noncross-linked foams. LDPE 0020 (ρ = 920 kg/m3, melt flow index (MFI) = 2.0 g/10 min) was supplied by Bandar Imam Petrochemical Co. (Iran). Talc powder (silicon dioxide, magnesium oxide, calcium oxide, ferric oxide, aluminium oxide) as the main nucleating agent was supplied by Azarbaijan Co. (Iran); glycerol monostearate (GMS-Atmer129, melting temperature (Tm) = 69°C) as the cell stabilizer was prepared by FACI (Italy); normal butane and propane mixture (weight ratio: 70/30) as the blowing agent was provided by Tabriz Petrochemical Co. (Iran); modified organoclay (closite20A) as an extra nucleating agent was provided by Fenghong Clay Chemical Co. (China), and LDPE-g-MA (1.0 wt% of MA, MFI = 3 g/10 min) as a coupling agent was provided by Kimia Bespare Asia Co. (Iran).
Preparation and characterization of LDPE foams
LDPE/clay nanocomposites were prepared using a single-screw extruder (Polycell Korea Corp., machine model: PKC 105 M/M) of a length–diameter (L/D) ratio of 38. LDPE granules were mixed with 0.2 wt% talc and various amounts of nanoclay, which were then fed into the drum over the hopper and melted totally by the screw movement in the extruder. Certain amount of blowing agent (including 70% n-butane and 30% propane) was injected into two detached spot on the extrusion drum by two positive displacement pumps and mixed with the polymer melt in the drum and totally dissolved in the melt. Table 1 shows the formulation of materials used for producing noncross-linked LDPE foams.
Materials formulation used to produce LDPE neat and nanocomposite foams.a
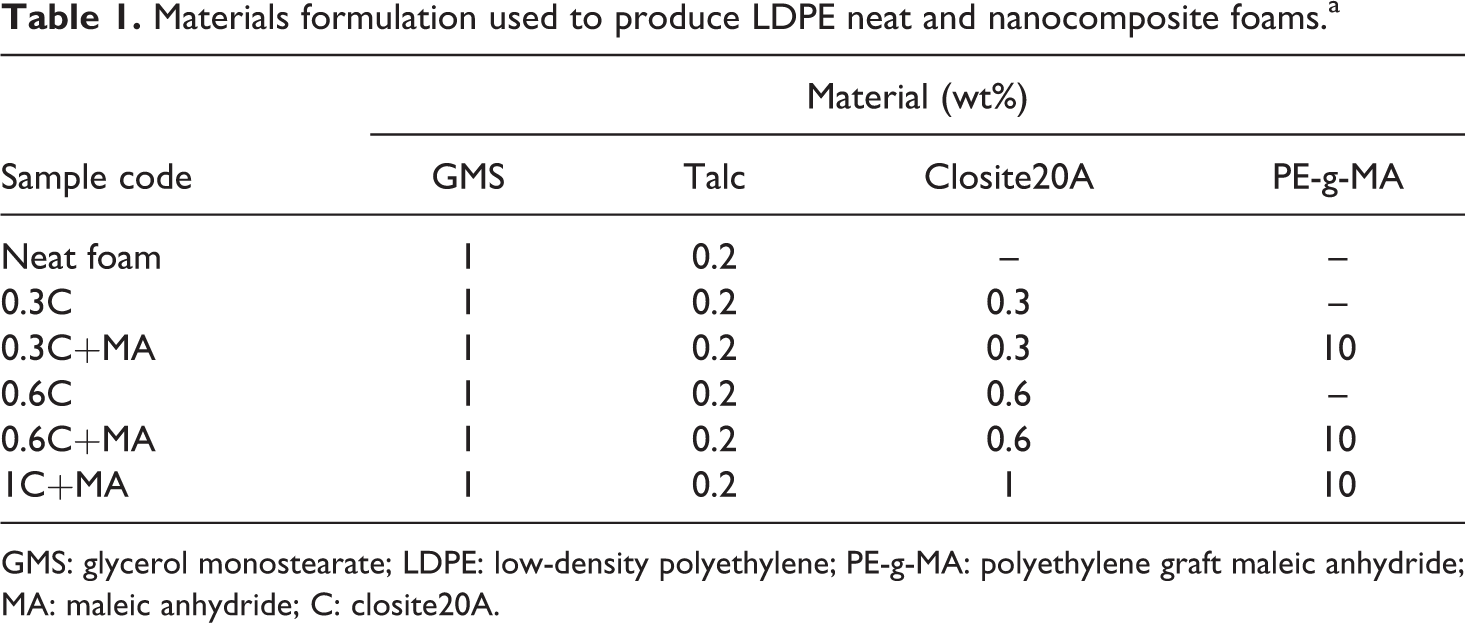
GMS: glycerol monostearate; LDPE: low-density polyethylene; PE-g-MA: polyethylene graft maleic anhydride; MA: maleic anhydride; C: closite20A.
In this research of PE foam preparation, a single-screw extruder with 105 mm screw diameter and L/D ratio of 38 was used. This extruder contains 12 heating zone from 165 to 200°C. LDPE granules, PE-g-MA granules, talc powder, and nanoclay powder were premixed in dry form and then were fed to the extruder. The feeding zone temperatures were kept constant at 160–185°C.
After melting the LDPE granules, GMS, as the cell stabilizer in liquid form, was fed in zone 4 at 195°C. After achieving a uniform mixture, certain amount of blowing agent (including 70% n-butane and 30% propane) was injected into two detached spot on the extrusion drum by two positive displacement pumps and mixed with the polymer melt in the drum and totally dissolved in the melt in zone 5 of extruder at 190°C. At zones 6 and 7, after mixing of blowing agent, GMS, LDPE and other additives, a uniform prefoam is achieved. The temperature in zones 8 to 10 is reduced to less than 100°C by cooling water with a flux of 30 m3/h. At the final stage of the extrusion in zones 11 and 12, the temperature is kept at 95°C. For producing LDPE for different applications, different die geometry is used.
Density measurement
The foam density was measured by dividing the weight of the foamed sample in the air to the volume of foamed sample, according to ASTM D3575. All the foam densities were kept constant in range of 22–23 kg/m3.
Morphology and cell microstructure
Morphological studies were carried out by x-ray diffraction (XRD, Siemens model D5000) method at room temperature. All XRD data were collected from 2θ = 0–10°, with a step interval of 0.02°.
Cell microstructure of the foams was studied by a Bell stereo microscope (STMDLX-T-POL, Maximum magnification: ×65) attached with a Bell high-speed digital camera (DV 1300, speed: 12 frame/s) and the images were analyzed by a Bell view image analysis software to determine the average cell size, cell size distribution, and cell density in the foam samples. Cell density, Nf
, was calculated as follows
10
:
where n is the number of cells in the micrograph of area (A) in cm2 with magnification of M, and
Mechanical properties
The mechanical properties of LDPE nanocomposite foams were examined by compression test using a Zwick/Roell tensile–compression machine (model TI-FR010THA50) at room temperature at a test speed of 5 mm/min and the sample size of 50 × 50 × 50 mm3. All experiments were repeated at least three times and the average and the standard deviation of the results were recorded.
Thermal conductivity
The samples' thermal conductivity coefficient was examined using a simple transient thermistor probe method of thermal conductivity meter designed and manufactured in the Institute of Polymeric Materials, Sahand University of Technology, Iran.
Needle probe thermal conductometer is commonly used for measuring thermal conductivity in liquid and solid materials, especially for rock cores, living tissues, and food materials. In this method, the needle probe is placed in the sample and is charged with pulse energy. Then the probe initial energy is decayed within the sample with time and its temperature is reduced with time. The pulse energy diffusion rate is dependent on the sample heat diffusion properties. The governing equations for this method have been presented elsewhere and the reduced temperature, which is a function of time and thermal conductivity coefficient, K, is calculated from the following equation
10,11
: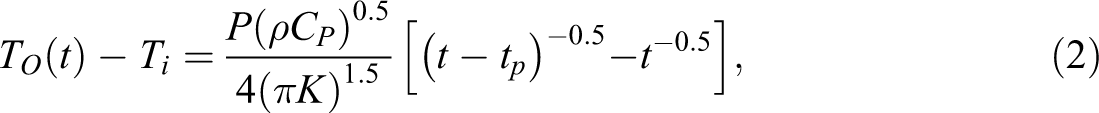
where TO is probe center temperature, Ti is probe initial temperature, P is the electrical power, ρ is sample density, Cp is sample heat capacity, t is the elapsed time (s), and tp is the duration of pulsed energy (s).
It is supposed that the pulse is created from one point. In this research, a negative thermal coefficient (NTC) thermistor was used as a temperature sensor instead of needle probe. In NTC thermistor, the probe resistivity is reduced with temperature increment.
For the LDPE foam thermal conductivity measurement, the cubic samples were prepared with 25 × 25 × 25 mm3 dimension. Before thermal conductivity measurement, the thermal conductometer was calibrated at room temperature to eliminate any effect of ambient temperature variation on the sample thermal conductivity. After calibration, the thermistor probe within the foam sample and pulse energy was charged into the samples and the temperature reduction was recorded against time for the samples. Each experiment was repeated at six different points at various directions of the samples and the average of the results was reported as the sample thermal conductivity coefficient.
Flame retardancy
Due to flammability of blowing agent (including 70% n-butane and 30% propane) in LDPE foam, it was tried to estimate the flammability of LDPE nanocomposite foams, the amount of dripping during burning test, and the burning rate of the samples using FAR 25.853 (b) standard. 12 In this experiment, the specimens (368 × 76 × 10 mm3) were held vertically using a cramp and the flame of the burner was applied at the lower end of the specimen for 12 s; then the flame was removed (ASTM D635– 03) and the burning rate (centimeter per minute) was calculated. In this method, the height of flame was maintained at 38 mm and the distance of sample from the top of the flame was 19 mm.
At the end of experiment, the burnt length of the samples was calculated. The flame extinguish time as the foam self-extinguishing parameter was also determined. Furthermore, the amount of dripping during burning was reported for the samples. Comparison of these parameters for neat LDPE foam, LDPE nanocomposite foam, and PE-g-MA compatibilized LDPE nanocomposite foam with standard values could reveal the samples’ flammability resistance.
Results and discussion
XRD analysis
XRD analysis was used to study the degree of dispersion of the nanoclay into the LDPE matrix. The XRD patterns of cloisite20A nanoclay and LDPE nanocomposite foams are shown in Figure 1. The peak at 2θ = 3.48° for closite20A corresponds to d-spacing of 2.06 nm. In nanocomposite foams with 0.3, 0.6 and 1 wt% closite20A, no such peak is observed within the scan range, indicating that the nanoclay might be properly dispersed in LDPE matrix. More detailed discussion about the state of nanoclay dispersion can be achieved using transmission electron microscope (TEM) micrograph, but the sample preparation for TEM in PE foams is very difficult, and in most of the cases it is not successful.
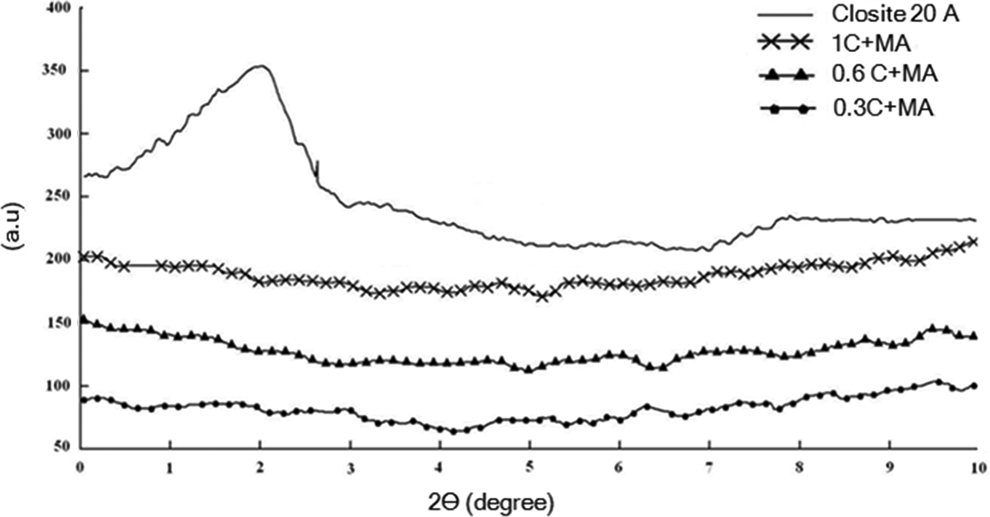
XRD patterns of Closite20A and LDPE/Closite20A nanocomposites. XRD: x-ray diffraction.
Cell microstructure
The micrographs in Figure 2 illustrate the effects of clay content on the cell morphology of LDPE nanocomposite foams without LDPE-g-MA compatibilizer. It can be observed that adding nanoclay to LDPE matrix reduces the cell size and improves cell microstructure in nanocomposite foams. The micrographs of foamed samples after incorporating LDPE-g-MA compatibilizer are presented in Figure 3. The presence of LDPE-g-MA compatibilizer remarkably decreases the average cell size and results in the formation of a more homogenous microstructure than that of the uncompatibilized LDPE nanocomposite foams.
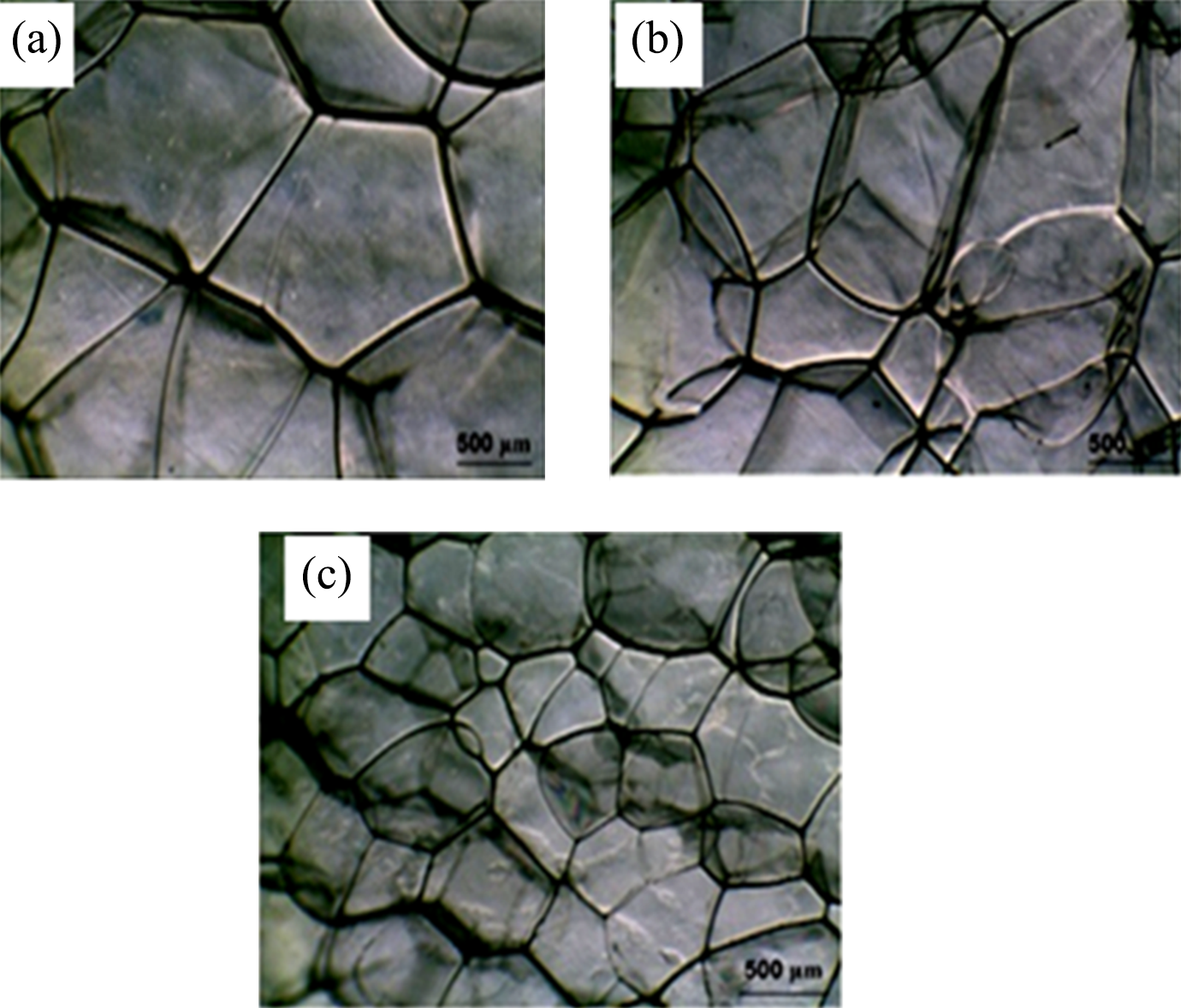
Micrographs of foamed samples without LDPE-g-MA compatibilizer: (a) neat foam, (b) 0.3C, and (c) 0.6C nanocomposite foam samples. LDPE-g-MA: low-density polyethylene graft maleic anhydride; C: closite20A.

Micrographs of foamed samples containing LDPE-g-MA compatibilizer: (a) 0.3C + MA, (b) 0.6C + MA, and (c) 1C + MA nanocomposite foam samples. LDPE-g-MA: low-density polyethylene graft maleic anhydride; C: closite20A; MA: maleic anhydride.
Figure 4 compares the average cell size and cell density of foam samples containing various amounts of nanoclay with and without LDPE-g-MA compatibilizer. To achieve foam cells with a controlled microstructure and uniform cell size distribution, some particles are added as nucleating agents to the polymer matrix to reduce the cell nucleation free energy. In this study, nanoclay and talc powder function as the nucleating agents in the foaming process. Incorporating the nanoclay into the LDPE matrix decreases the formation of the cell nucleus. Furthermore, appropriate dispersion of closite20A nanoclay as a nucleating agent can increase the cell density of LDPE foam and decrease the average cell size. As it is demonstrated in Figure 4(b), the cell density has been increased from 4.12 × 104 (cells/m3) for neat foam to 8.06 × 104 (cells/cm3) for 0.6C nanocomposite foam. It can be concluded that the presence of LDPE-g-MA compatibilizer enhances nanoclay dispersion and thus improves the cell size and density.
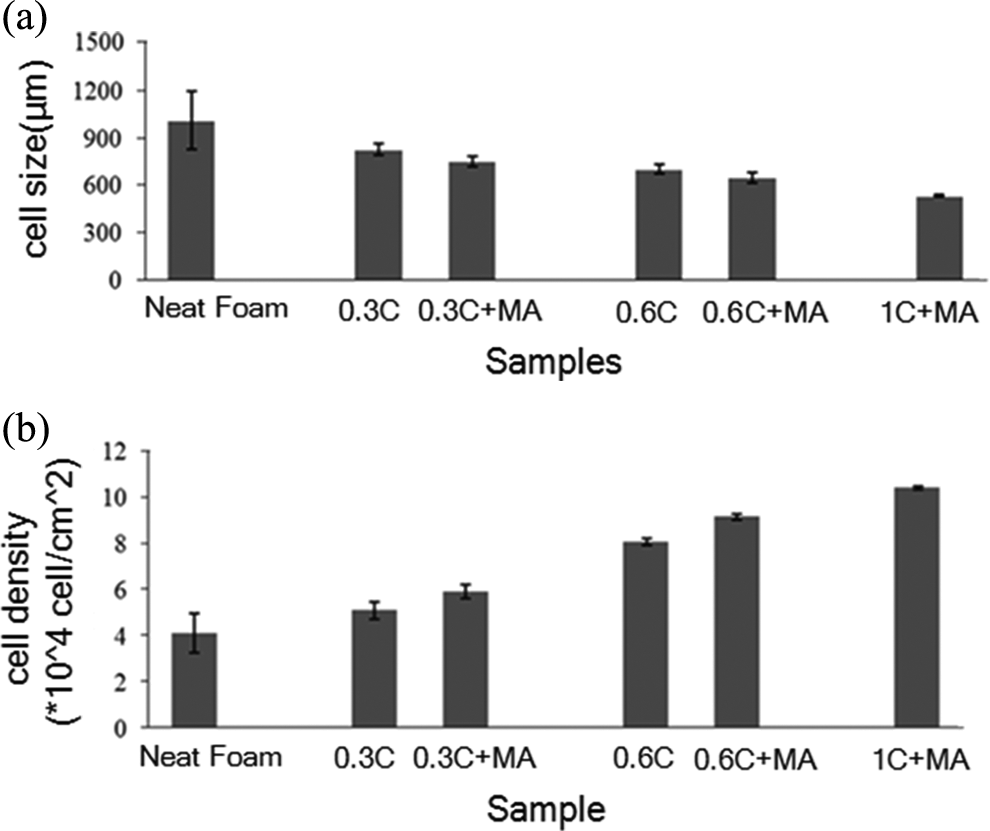
The effect of clay content and LDPE-g-MA on the (a) cell size, and (b) cell density of neat and nanocomposite foams. LDPE-g-MA: low-density polyethylene graft maleic anhydride.
Figure 5 demonstrates the normal distribution cell size of LDPE neat and nanocomposite foams. Due to the proper dispersion of nanoclay in pre-foamed LDPE matrix, it can be concluded that with increase in nanoclay content, the cell size of the LDPE nanocomposite foams also decreased and the distribution of cell size is homogeneously narrowed. Furthermore, the addition of LDPE-g-MA compatibilizer decreases the cell size due to the improved compatibility of LDPE and nanoclay; but no significant effect on cell size distribution is observed.
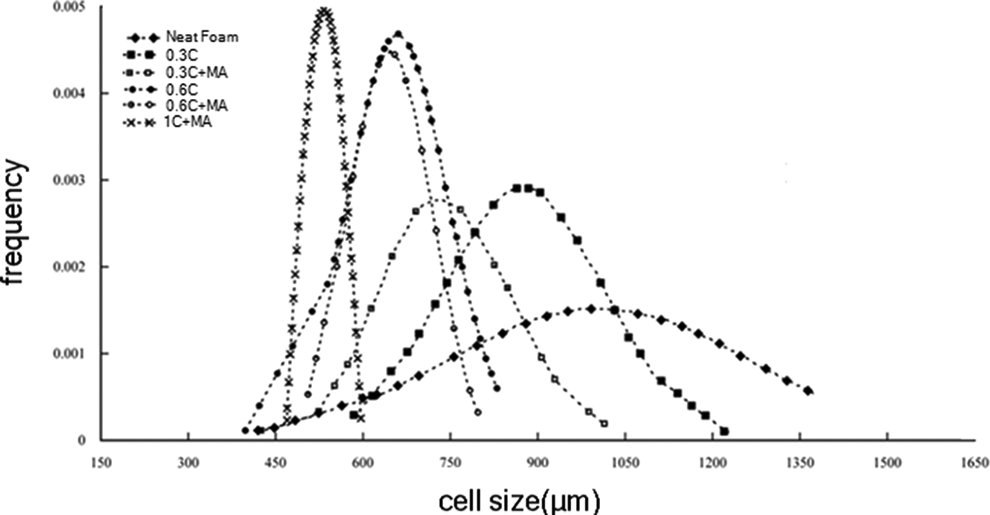
The effect of clay content and LDPE-g-MA on the cell size distribution of neat and nanocomposite foams. LDPE-g-MA: low-density polyethylene graft maleic anhydride.
Mechanical properties
The high aspect ratio and large surface area of nanoparticles offer the potential for high reinforcing efficiency, good barrier properties, and improved dimensional and thermal stability. The nanometer dimension is especially beneficial for reinforcing foam materials, considering the micron and sub-micron thickness size of cell walls. It is conventional to use nanoparticles to reinforce microstructures in order to improve the properties of final product. 13 Incorporating closite20A nanoclay to LDPE foam could affect the microstructure and produce foams with enhanced mechanical properties. As it can be observed in Figure 6, by incorporating nanoclay to LDPE matrix, Young’s modulus increased in LDPE nanocomposite foams compared to neat foam, as well as further improvement in mechanical properties is observed by adding LDPE-g-MA compatibilizer to the nanocomposite foams. This behavior can be attributed to the nanoclay dispersion states in nanocomposite foams with different clay contents in the presence of compatibilizer.
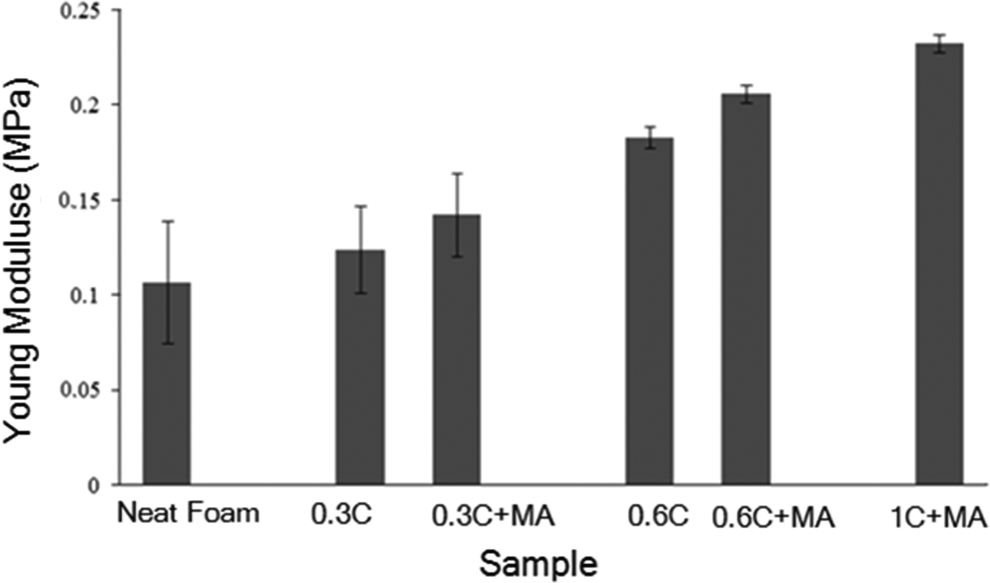
The effect of clay content and LDPE-g-MA on Young’s modulus of LDPE neat and nanocomposite foams. LDPE-g-MA: low-density polyethylene graft maleic anhydride; LPDE: low density polyethylene.
Thermal conductivity
Some studies emphasize that the control of density, chemical composition in the cell walls and especially the mean cell size affect thermal conductivity of polymeric foams. 14 –18 Figure 7 demonstrates the effect of the modified closite20A content on the thermal conductivity of LDPE foam. Considering the cell microstructure and the fact that reduction in cell size may lead to increase in the number of cell walls and reduce the radiation contribution of thermal conductivity, it can be observed that LDPE nanocomposite foam with smaller cell size shows lower thermal conductivity compared to neat LDPE foam. It can be concluded that by adding nanoclay to LDPE foam and due to the proper nanoclay dispersion in prefoamed material by incorporating LDPE-g-MA compatibilzer, the thermal conductivity coefficient will be decreased and consequently its insulating properties will be improved.
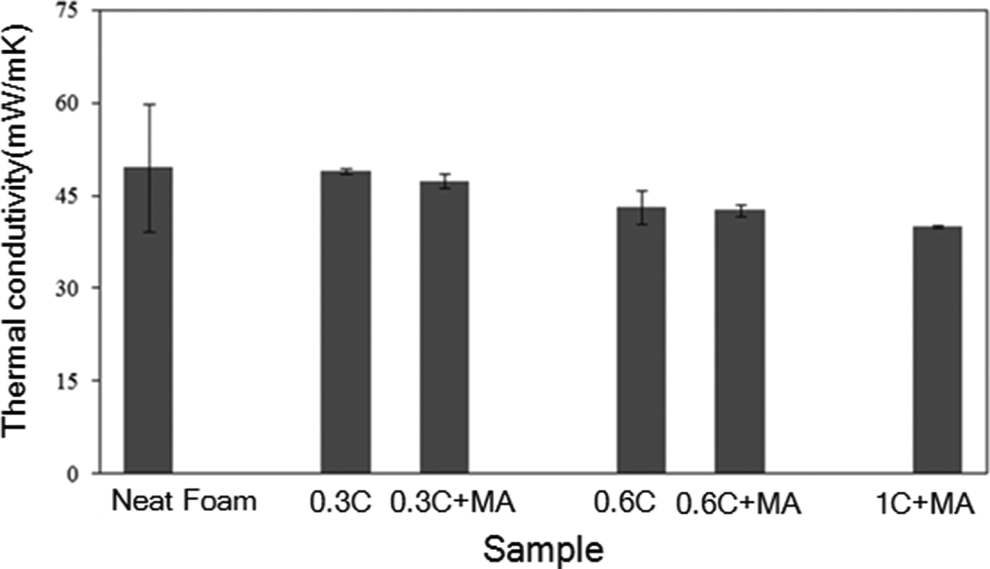
The effect of clay content and LDPE-g-MA on thermal conductivity coefficient of LDPE neat and nanocomposite foams. LDPE-g-MA: low-density polyethylene graft maleic anhydride; LPDE: low density polyethylene.
Flame retardancy
Latest studies suggested that the presence of nanoclay can improve the flammability properties compared to neat polymeric material. In this study, by utilizing n-butane and propane as blowing agent, flame retardancy becomes quite low. Cloisite20A nanoclay can improve LDPE foam flame retardancy by attending in the cell walls. Actually, nanoclay could act as a flame retardant to LDPE foams. The nanoclay plates in the LDPE foam walls and struts act as a barrier to heat diffusion inside the samples and improve the flammability resistance of the nanocomposite foams compared to neat LDPE foams.
Furthermore, the proper nanoclay dispersion in the polymer matrix will affect the foam flammability performance. Incorporation of PE-g-MA compatibilizer has improved the LDPE foam flame retardancy; in other words, burn rate as well as dripping content decreased in nanocomposite foams containing PE-g-MA compared to those without PE-g-MA and neat LDPE foam, as illustrated in Figure 8. Compatibilization of LDPE nanocomposite foam may lead to the dispersion of nanoclay in the foam walls and struts, finally leading to flame retardancy of the compatibilized foams compared to uncompatibilized noncross-linked LDPE nanocomposite foams.
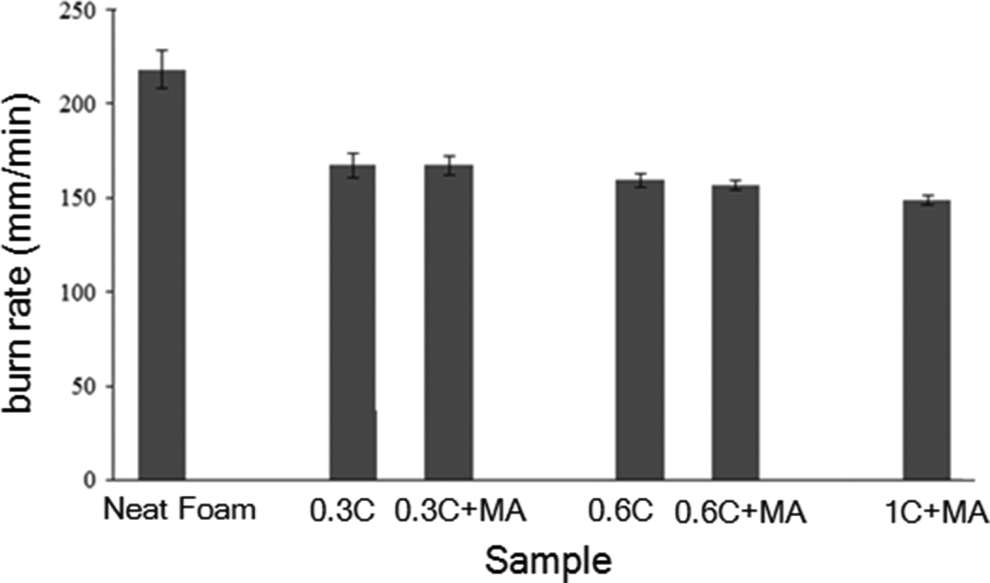
Burning rate of LDPE and nanocomposite foams with and without LDPE-g-MA compatibilizer. LDPE-g-MA: low-density polyethylene graft maleic anhydride; LPDE: low density polyethylene.
Conclusion
Noncross-linked LDPE nanocomposite foams containing LDPE-g-MA compatibilizer and OMMT nanoclay (closite20A) with similar densities were prepared by extrusion method, and the effects of nanoclay content and LDPE-g-MA on morphology, mechanical, and thermal properties of foam samples were studied. XRD patterns did not show any closite20A characteristic peak, as an evidence of intercalation/exfoliation of the nanoclay in the polymer matrix. Due to proper nanoclay dispersion in prefoamed materials in the presence of compatibilizer and decreasing the work of cell nucleation around the nanoclay, the average cell diameter decreased and the cell density increased. Achieving the proper dispersion of closite20A nanoclay in the foams led to the production of foams with homogenous microstructure and improved thermal and mechanical properties. Furthermore, due to the presence of nanoclay on the cell walls and its appropriate dispersion, the foams’ flame retardancy improved. Finally, the results of this study revealed that compatibilized LDPE nanocomposite foams with 1 wt% closite20A nanoclay content showed the most appropriate physical and mechanical properties with narrow distribution of cell size and homogenous cell microstructure.
Footnotes
Acknowledgements
The authors wish to acknowledge Mr. Naser Harasi, for supporting, his kind attention and consulting in the production of PE foams in Kimiagaran Polymer Faravardeh Co, Iran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
