Abstract
The thermo-oxidative decomposition behavior of polypropylene (PP) and polyethylene (PE) filled with carbon nanotube (CNT) and organo montmorillonite (OMMT) as a heat stabilizing filler was comparatively investigated using nonisothermal- and isothermal–thermogravimetric (TG) analyses. The isoconversional method was employed to evaluate the kinetic parameters (Ea , lnA, and n) under dynamic heating in air. OMMT and CNT exhibited respective lowest and highest thermo-oxidative stability as revealed from the lowest and highest T onset values, respectively, whereas thermo-oxidative stability of PE was better than PP. The TG profiles and calculated kinetic parameters strongly depended on the sorts of fillers and polyolefins. From TG data and calculated kinetic parameters, the incorporation of CNT and OMMT into PP and PE significantly improved the thermo-oxidative stability of the composites. The highest relative Ea value was observed for CNT-containing composites in the conversion extent region of 0.10–0.45. Simultaneous differential scanning calorimetry thermograms revealed that the degradation processes for the neat polyolefins and their composites were exothermic in air. By adding the same filler, the thermo-oxidative stability of the PE-containing composite system under isothermal heating at 320°C was much higher than that of PP composite systems. The obtained results of nonisothermal and isothermal investigations and morphological observation of the samples after heating suggested the improved thermo-oxidative stability of polyolefins modified by CNT over that by OMMT.
Introduction
During the last few decades, the nanocomposites based on thermoplastic polymers and clay, especially with organically modified montmorillonite (OMMT), are a group of materials that have been extensively investigated in order to find their promising alternatives to traditional composites, though mainly focused on general mechanical and multifunctional properties and filler dispersion. The clay-containing nanocomposite has been of great interest to both academic and industrial researchers due to their ability to display synergistically advanced characteristics with small amount of clay loading (e.g. 3 wt%). 1 The typical improved properties include barrier properties, flame retardancy, thermal stability, chemical and dimensional stability, and mechanical properties when compared to their micro- and macrocomposite counterparts and their neat polymer matrices. 2 –4 Recently, polymer nanocomposites emerged as one of the most promising developments in the area of flame retardancy 5,6 and heat stabilization, 7 –9 offering significant advantages over conventional formulations.
In the case of polymer/nanocomposites, one of the most used fillers is carbon nanotubes (CNTs). CNTs display exceptional properties that can potentially be used in many applications ranging from macroscopic material composites down to nanodevices. The properties and applications of CNTs and related materials have been very active research fields over the last decade. 10 –12 CNTs possess high flexibility, low mass density, and large aspect ratio (typically >1000), whereas predicted and some experimental data indicate extremely high tensile moduli and strengths for these materials. Due to their high aspect ratio, CNTs percolate to form a network at very low loading in the polymer matrix and lead to substantial enhancement of several functional properties such as mechanical, rheological, and flame-retardant properties. 13 –16 The thermal properties of CNT/polymer composites also have attracted many attentions from polymer scientists because CNTs have very high thermal stability. 17 –21 Most of the previous studies suggested that both promoting and hindering effects of CNTs on the thermal stability of polymer are found. Moreover, the dispersion of CNT, the structure of the polymer matrix, and the interaction between CNTs and the matrix have been found as the key factors for the thermal degradation behavior of CNTs-filled polymer composites. Recently, the thermal stability of styrene–(ethylene butylene)–styrene (SEBS)-based elastomer composites modified by OMMT and CNT compared to those modified by liquid crystalline polymer has been investigated. 22 From the kinetic parameters calculated by isoconversional methods reported in our previous work, it is indicated that the incorporations of LCP, OMMT, and CNT into SEBS significantly improve the thermal stability of the composites. Especially, the thermal stability of SEBS is greatly improved by the addition of OMMT and CNT. However, it is known that the improved thermal stability of the polymer composites is contributed not only from the fillers but also due to the inherent characteristics of polymer matrices. Especially for polypropylene (PP) and polyethylene (PE) commodity plastics, these polymers are widely used in many cases for production of polymeric materials. To the authors’ knowledge, very limited information is available with regard to a direct comparison of thermal stabilizing ability, promoted from OMMT and CNT, as a minor blending component for PP or PE thermoplastic composites.
In this study, the composites of PP and high-density PE filled with OMMT and CNTs were prepared. Thermo-oxidative decomposition behavior and decomposition kinetics of the composites containing different fillers and polymer matrices were comparatively elucidated. The performance of CNT and OMMT as a thermo-oxidative stabilizing modifier for the polyolefins-based thermoplastic composites was compared.
Materials and experimental procedure
Materials
Polyolefins used as the matrices in the present study were PP and high-density PE. PP was purchased from CCC Chemical Commerce Co., Ltd., Bangkok, Thailand. The specific gravity and melt flow rate of PP were 0.910 (ASTM D1505) and 2.4 g/10 min (ASTM D1238). High-density PE grade 5000S with a melt flow index (MFI) of 0.80 g/10 min was purchased from Bangkok Polyethylene (Thailand). OMMT which is commercially available under the trade name “Claytone® HY” produced by Southern Clay Products, USA, was purchased from a local distributor and used as received. According to product literature, the organic modifier is bis(hydrogenated tallow alkyl) dimethyl ammonium. This particular grade was chosen for this study as it is recommended for low polarity systems and does not require activator. CNT used in the present work was a hydroxyl functionalized multiwall CNTs purchased from Chengdu Organic Chemicals Co., Ltd. (China). This material has a bulk density of 0.28 g cm−3 with a functionalized content of 1.76 mass%. The outer diameter, length, specific surface area, and purity were reported to be 20–30 nm, 20–30 μm, >110 m2 g−1, and >95%, respectively.
Preparation of composites
The PP/CNT, PP/OMMT, PE/CNT, and PE/OMMT composites were prepared by melt blending in an internal mixer (Haake Rheomex, Thermo Electron (Karlsruhe) GmbH, Karlsruhe, Germany) at a temperature of 165°C and a rotor speed of 100 rpm for 10 min. The composites were then compression molded into a sheet of about 1 mm thick. The molding condition was 170°C under a pressure of 20 tons for 10 min. The sample codes of PP- and PE-based composites are designated as PP-CNTX, PP-OMMTX, PE-CNTX, and PE-CNTX, where X depicts the content of the CNT or OMMT in phr. In the present work, in order to absolutely compare the effect of different types of fillers and matrices on the thermal stabilizing ability, the fixed filler content (X) of 5 phr was used for composite preparation without using any compatibilizer.
Thermo-oxidative decomposition analysis by simultaneous TG and DSC measurements
Thermogravimetric (TG) analysis was carried out using TA instruments, SDT Q600 (Luken’s drive, New Castle, DE). The neat and composite samples of 8–10 mg were loaded in alumina crucible and then nonisothermally heated from ambient temperature to 1000°C at four different heating rates of 5, 10, 15, and 20°C/min. The TG was performed in air with the flow rate of 100 mL/min. The TG and differential scanning calorimetry (DSC) data were simultaneously recorded online in TA instrument’s Q series explorer software. The analyses of simultaneous TG and DSC data were done using TA Instrument’s Universal Analysis 2000 software (version 3.3B). The isothermal tests were measured at 320°C for 100 min. Before isothermal heating, the sample was heated at a rate of 20°C/min from ambient temperature to the selected temperature of isothermal degradation. As soon as the system reached the selected temperature, the variations in sample mass with times were registered. The isothermal tests were performed in air at a flow rate of 100 mL/min. The analyses of isothermal TG data were carried out using the same software as those of the nonisothermal TG data.
Morphological characterization
The morphology of the polymer matrices and their composites after isothermal heating at 320°C for 20 min were observed under the digital micro-measure camera (BM-400x, Shenzhen, China). Prior to examination, the rectangular sample with the dimension of 3 × 3 × 1.5 mm3 was prepared. The heating process was carried out using the TA instrument under the same conditions as those of isothermal measurement.
Viscosity measurements
Rheological behavior in the molten state for PP, PE, and their composites containing 5 phr OMMT was characterized with a plate-and-plate rheometer (Physica Anton Paar, MCR5000; Physica Messtechnik GmbH, Stuttgart, Germany). The samples were compression-molded at 160°C into a sheet of about 1.5 mm thick. The sheet was then punched into a disk 25 mm in diameter. The complex viscosity (η*) of all specimens was measured in the oscillatory shear mode with the strain amplitude of 5% within the angular frequency (ω) range from 0.6 to 500 rad/s. The measuring temperature was set at 165°C. The gap between the two plates was set at 0.9 mm.
Results and discussion
Thermo-oxidative decomposition behavior of neat materials
Before discussing the effect of CNT in comparison with OMMT on the thermo-oxidative decomposition behavior of polyolefins-based composites, the decomposition behavior of the neat materials is presented. Figure 1 shows the TG (Figure 1(a)) and derivative TG (DTG) (Figure 1(b)) curves of PP, PE, OMMT, and CNT at a fixed heating rate of 10°C/min in air, as a realistic atmosphere for processing and application. It is seen that TG and DTG curves of all samples remarkably reveal the different profiles. PP and CNT seem to degrade in a major single step, whereas the thermo-oxidative degradation of PE and OMMT occurs in at least two mass loss steps. The neat PP exhibits the first major mass loss in the range of 220–400°C, involving random scission followed by radical transfer process. 23 For the neat PE, the nonisothermal TG profile reveals multistep of weight loss at a temperature range of around 250–530°C. The degradation mechanism of PE is responsible for the propene product and the other gives rise to 1-hexane. 23 The degradation mechanism of PE begins at the weak link site along the polymer chain once the thermally induced scission has occurred. The first degradation step of OMMT appears in the range of 200–350°C, which corresponds to the decomposition of organic components. The content of the organic substance is about 30 mass%. Interestingly for CNTs, the single mass loss appears at the highest temperature range (450–650°C) compared with other samples. This is due to the high stability of network structure of CNTs. As we know, CNTs are sheets of graphite rolled into tubes and have excellent properties due to their symmetric structure. For the present work, the hydroxyl functionalized multiwall CNTs were used. The hydroxyl groups are grafted onto the defects of multiwall CNT generated at the broken penta-basic, heptatomic ring or at the open ends of multiwall CNTs. Therefore, with the time prolonging or the temperature increasing, more rings around the defects would be broken; finally the nanotube would be cut shorter. Note that after heating, the highest amount of char residues is clearly found for OMMT mainly due to aluminum silicate minerals thermal stability in air. 24,25
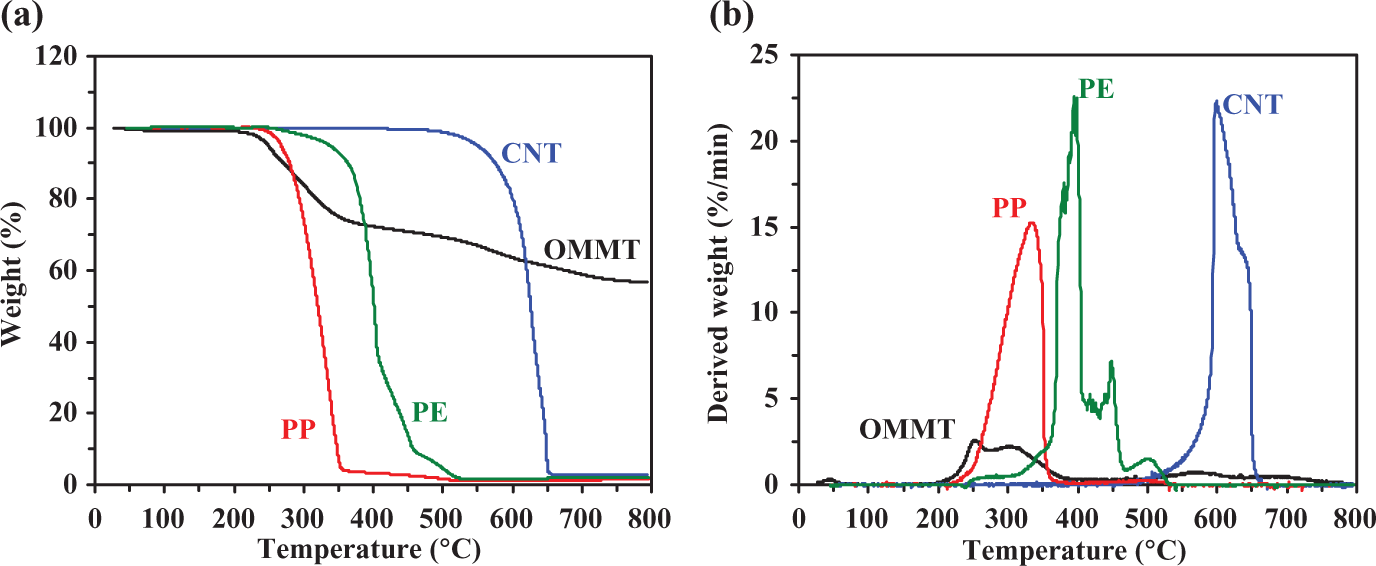
TG (a) and DTG (b) curves of thermo-oxidative decomposition for PP, PE, CNT, and OMMT at a heating rate of 10°C/min in air. TG: thermogravimetric; DTG: derivative TG; PP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
Table 1 shows the summarized data of thermo-oxidative decomposition for the neat materials. T onset represents the onset degradation temperature. T max represents the temperature at the maximum weight-loss rate, (dα/dt)max. It is seen that T onset and T max values of all samples are found in the following order: CNT > PE > PP > OMMT. Note that T onset and T max of OMMT show the lowest values due to the presence of organic compounds possessing low heat stability within OMMT. Based on the obtained results of T onset and T max of the fillers and matrices, it may be expected that CNT can improve the heat stability of the polyolefins but OMMT cannot. However, the improvement in thermal stability of composite materials is generally dependent not only on the inherent properties of the filler but also on the properties of polymer matrix, processing methods, dispersion of filler, interaction between phases, and so on. The effect of CNT and OMMT loadings on thermo-oxidative decomposition of polyolefin-based composites will be discussed in next section. Figure 2 shows the expanded DTG curves of all neat materials. Clearly, OMMT starts to first degrade followed by PP, PE, and CNT, respectively. At 250°C, OMMT degrades with the highest rate, whereas no significant change in the decomposition rate is observed for CNT. Note that, by considering the decomposition throughout the reaction interval of all samples, the highest maximum weight-loss rate is found in the following order: CNT > PE > PP > OMMT.
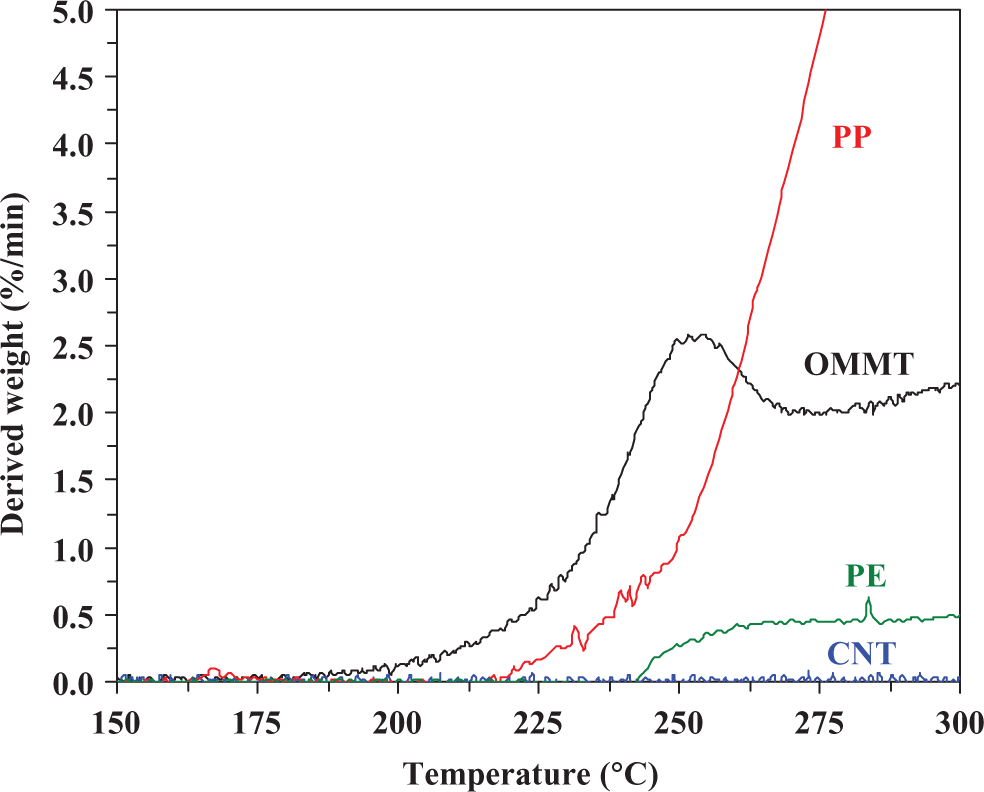
Expanded DTG curves of PP, PE, CNT, and OMMT in the region of 150–300°C. TG: thermogravimetric; DTG: derivative TG; PP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
TG and simultaneous DSC data of thermo-oxidative decomposition for PP and PE composite systems at a heating rate of 10°C/min in air atmosphere.a
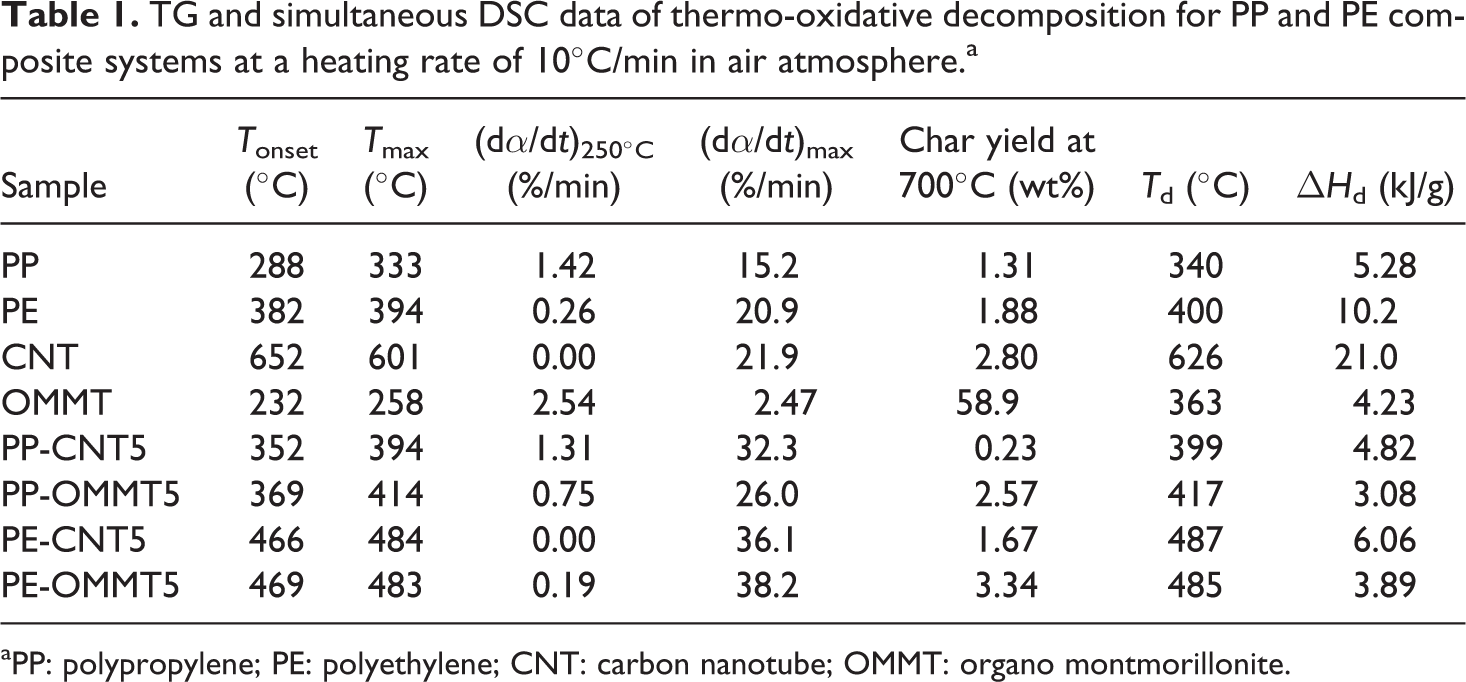
aPP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
The simultaneous DSC traces of thermo-oxidative degradation for the PP, PE, CNT, and OMMT are shown in Figure 3. The corresponding peak temperature (T d) and enthalpy (▵H d) associated with the thermal degradation of all materials are also presented in Table 1. The exothermic degradation process is observed for all the samples due to the fact that the concurrent and further degradation mechanisms in air tend to involve the formation reaction. PP shows a very broad degradation exotherm that stretches from 200 to 360°C with a peak maximum (T d) at 340°C. The DSC trace of PE shows a large exothermic peak stretched from 250 to 550°C whose maximum is 400°C. After this region, the remaining signal of PE is unstable as evident by the appearance of several minor peaks. OMMT exhibits a broad degradation exotherm with a peak maximum at 363°C. In the case of CNT, T d is found at 626°C. Note that the enthalpy of thermal decomposition of CNT shows the highest value (≈ 21 kJ/mol), whereas the enthalpies of PE, PP, and OMMT are about 10, 5, and 4 kJ/mol, respectively.
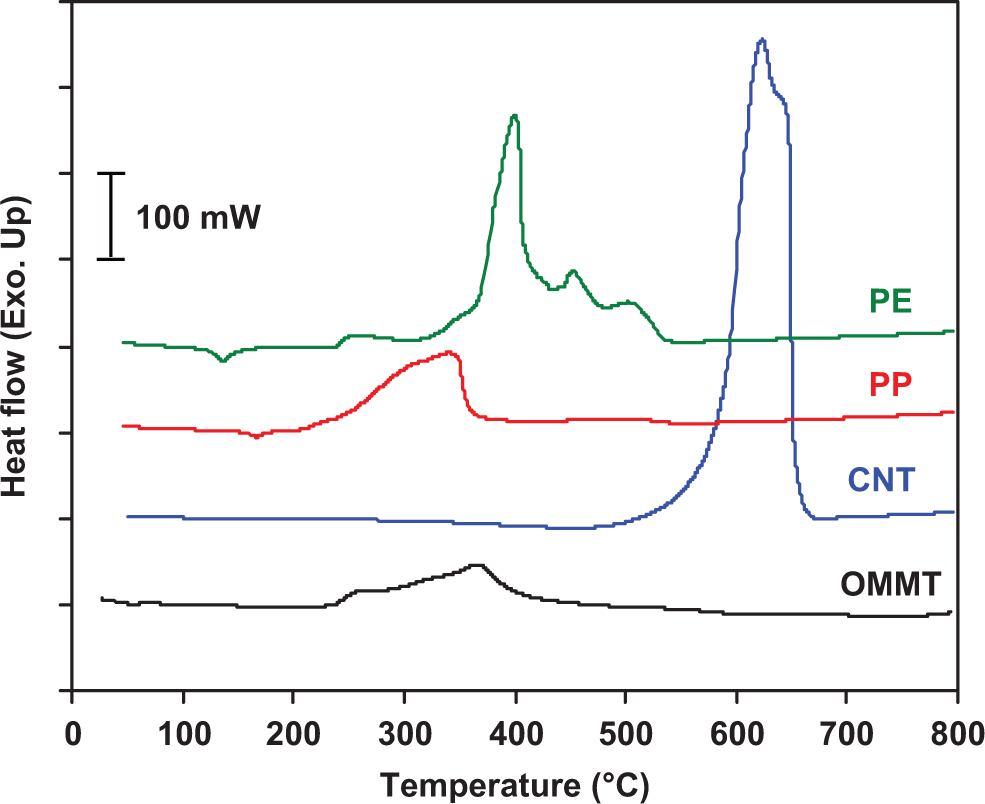
Simultaneous DSC curves of thermo-oxidative decomposition for PP, PE, CNT and OMMT at heating rate of 10°C/min in air. DSC: differential scanning calorimetry; PP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
Thermo-oxidative decomposition behavior of CNT- and OMMT-filled polyolefins
The influence of the CNT or OMMT loadings on nonisothermal decomposition behavior of PP- and PE-based thermoplastic composite system is shown in Figure 4, and the corresponding decomposition data are also presented in Table 1. For the PP composite system (left column of Figure 4), it is seen that the TG curves of PP-CNT5 and PP-OMMT5 clearly shift to higher temperature when compared with that of PP. The remarkable shift of TG curves to high temperature for the composites containing OMMT and CNT indicates the significant improvement in thermo-oxidative stability of the composites. From Figure 4(Ib), it is understood that the shapes of DTG curves for the neat materials and their composites are different. The decomposition rate of PP-CNT5 and PP-OMMT5 gradually increases first and rapidly increases at temperature of 300°C. However, before reaching the maximum rate of degradation, very rapid increase in the decomposition rate is observed for both composite samples. Interestingly, the shapes of DTG curves for PP-CNT5 and PP-OMMT5 are nearly the same. It is seen that, beyond the decomposition rate peak, the dramatic drop is observed in the rate of PP-CNT5 and PP-OMMT5. The change in decomposition rate upon dynamic heating is additionally clarified by expanding DTG curves as shown in Figures 4(Ic). It is seen that OMMT and PP-OMMT5 start to gradually degrade at temperatures of 175 and 200°C, respectively, whereas PP and PP-CNT5 are found to degrade at 220 and 225°C, respectively. However, although CNT exhibits thermally stable material at very high temperature, the rate of decomposition for the CNT-filled PP composite seems to increase sharply similar to that of the neat PP matrix. Interestingly in the case of OMMT composite system, although the neat OMMT degrades at relatively low temperature, significant improvement in thermo-oxidative stability of the OMMT-containing composites is observed. This would suggest the accumulation and reassembly of silicate layer on the surface of burning material, offering a great barrier effect to hinder the formation of small molecules, resulting from thermal decomposition and simultaneous resisting their movement during desorption from the surface. 26 Meanwhile, the activity of OMMT was increasing barrier properties and insulating properties to heat transport of composite that should improve the thermal stability. Similarly for CNT-filled composites, CNTs have also been reported that can increase thermal stability of thermoplastics. 27 –30 This was because CNT has excellent thermal conductivity. The improvement in thermal stability of the composites was also attributable to the formation and stabilization of CNTs-bonded macroradicals. 31 The stabilization effect of CNT was explained by the barrier effect of the nanotubes and their formed aggregates, which hinder the diffusion of the degradation product from the bulk of the polymer onto the gas phase. The increased thermal stability of CNT-containing composites over that of the neat matrix polymer is likely the result of adsorption, by activated CNTs surface, of free radicals generated during polymer decomposition.
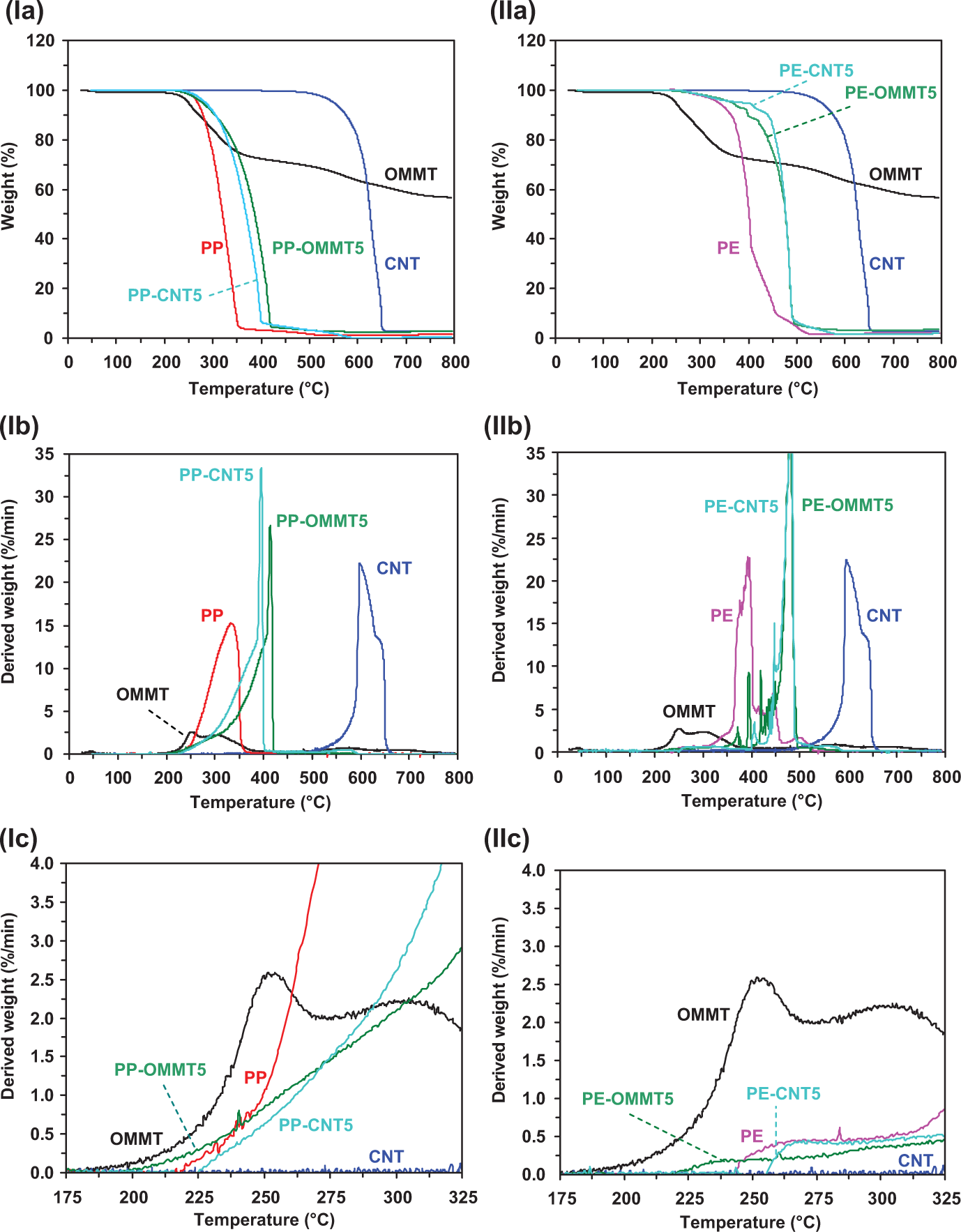
TG (row A), DTG (row B), and expanded DTG (175–325°C) (row C) curves for PP (column I) and PE (column II) composite systems. TG: thermogravimetric; DTG: derivative TG; PP: polypropylene; PE: polyethylene.
For PE-filled composite system (right column of Figure 4), the TG curves of PE-CNT5 and PE-OMMT5 clearly shift to the higher temperature than that of PE. As seen from Figure 4(IIb), T max of decomposition of the two composites appears to be nearly the same. DTG curves of PE composites show unstable trace, indicating that the decomposition mechanism of PE-filled composites is more complex than the PP composite system. In fact, the thermal degradation of PE is normally more complicated than that of PP. By considering the expanded DTG curves (Figure 4IIc), the PE-OMMT5 seems to first degrade at ≈220°C, whereas PE-CNT5 begins to degrade at ≈255°C. Interestingly, PE-CNT5 and PE-OMMT5 begin to degrade with a low rate in the temperature range of 200–300°C ((dα/dt)max < 0.5%/min). This behavior is different from that of the CNT- or OMMT-filled PP. For instance, at temperature >250°C, the degradation of the PP composites rapidly occurs at higher rate, that is, (dα/dt)max > 0.5%/min. Although the neat OMMT exhibits the lowest heat stability as seen from degradation starting at the lowest temperature (Figure 4IIc), the decomposition rate, especially at temperature >260°C, for the OMMT-filled PE composites shows the relatively lowest value compared with PE and PE-CNT5. This means that the thermal decomposition of OMMT alone and as the filler for the PE composites is significantly different. As discussed above, the presence of OMMT in the polymer matrix normally improves the barrier to diffusion of oxygen gas into the material. Another possible reason is that the clay increases the viscosity, which in turn will suppress the diffusion of the degradation by-products from the sample, resulting in low rate of thermal decomposition. 31 The latter reason can be confirmed by the results of viscosity measurement. As seen from Figure 5, the viscosity of PP and PE matrices clearly increases with OMMT loading over the whole range of shear frequency investigated. Additionally, amount of the char left for OMMT-filled systems is higher than those for CNT-containing composites. It can be accepted that in the presence of OMMT layers acting as a barrier to heat and mass transport, first more extensive random scission of polymer chain occurs due to superheated conditions in the condensed phase. Then chemical species, trapped between OMMT layers, have more opportunity to undergo further intermolecular reactions, such as radical recombination. The intermolecular reactions lead to the formation of complex compounds, thus lowering the volatilization rate and favor the char formation process. 9
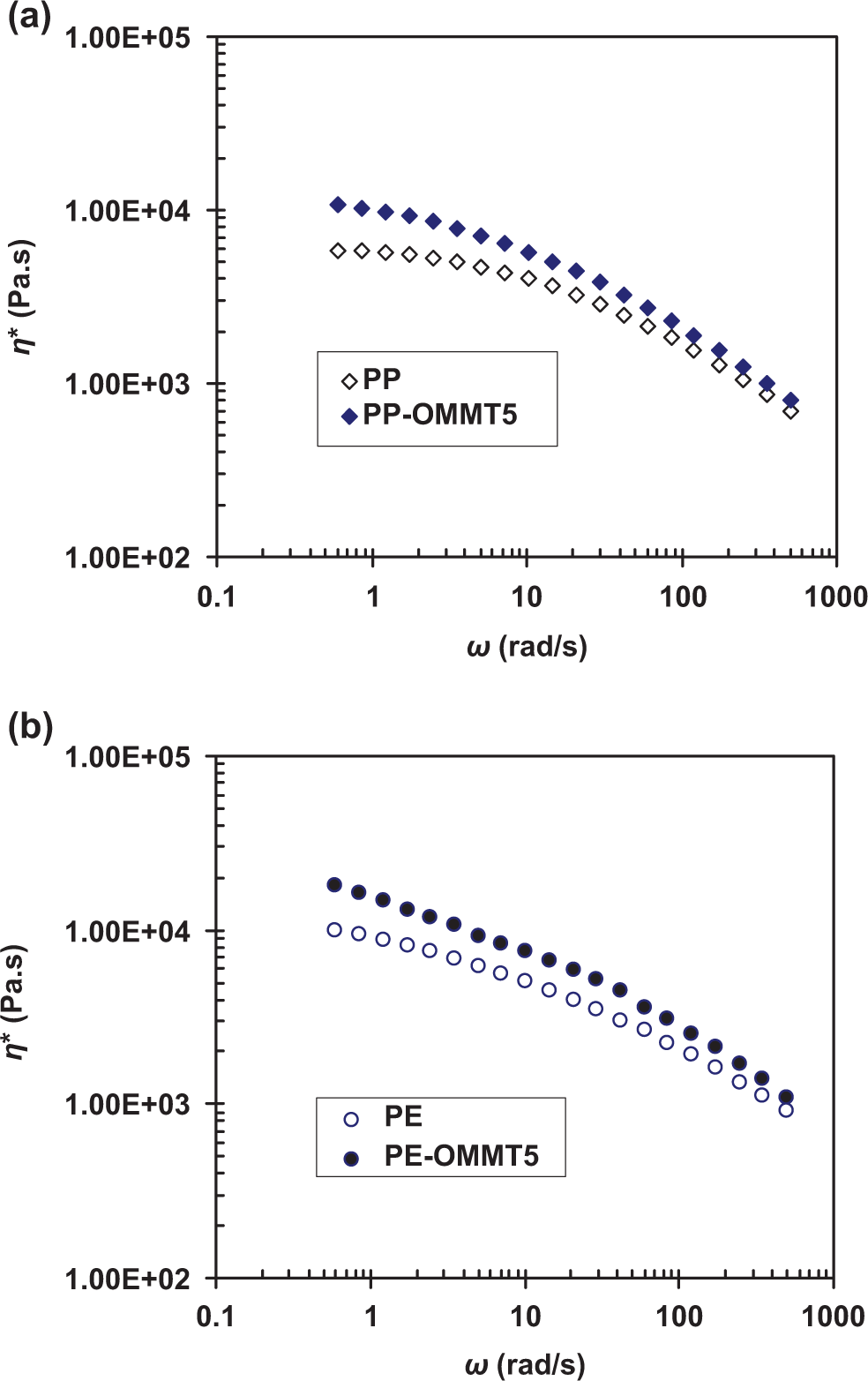
η* vs. ω for PP (a) and PE (b) composite systems measured at 165°C. PP: polypropylene; PE: polyethylene.
The DSC traces of thermal degradation for PP and PE compared to their composites containing CNT and OMMT are shown in Figure 6. The corresponding DSC data (T d and ▵H d) of all samples are also presented in Table 1. The DSC curves of all samples in air show a large degradation exothermic peak involving the thermo-oxidative reaction. Clearly, the addition of CNT or OMMT leads to a remarkable increase in T d of the composites. In addition, all composites have lower ▵H d values than that of its corresponding matrix. By adding the same filler, the ▵H d values of PE composites (PE-CNT5, PE-OMMT5) are slightly higher than those of the corresponding PP composites (PP-CNT5 and PP-OMMT5). The lower ▵H d values were due to lesser decomposition partially caused by the inherent properties of the polymer matrix.
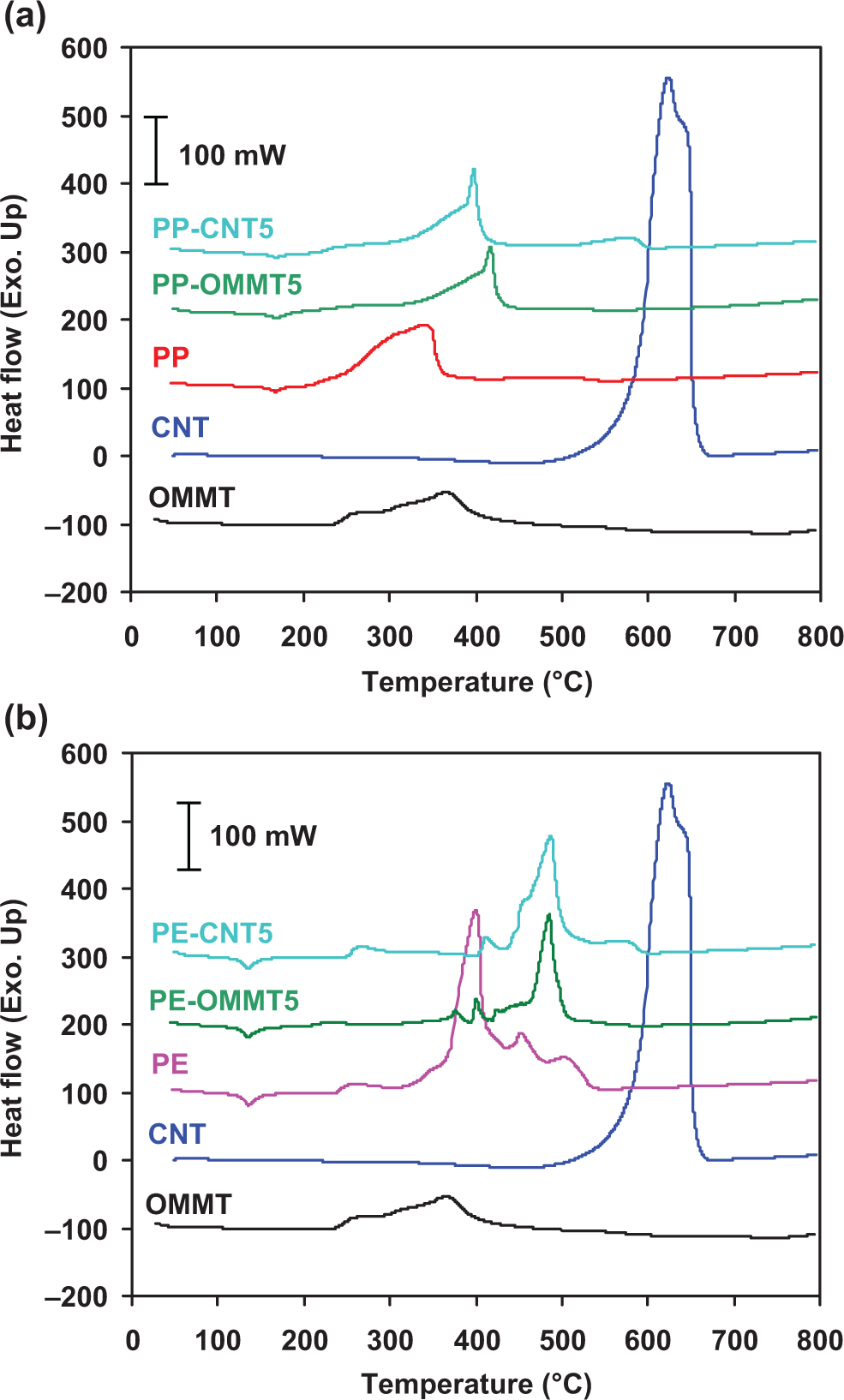
Simultaneous DSC curves of thermo-oxidative decomposition for PP (a) and PE (b) composite systems at a heating rate of 10°C/min in air. DSC: differential scanning calorimetry; PP: polypropylene; PE: polyethylene.
Kinetic analysis
In this work, to obtain the reliable kinetic information on thermal degradation, the isoconversional method, namely, Flynn-Wall-Ozawa (F-W-O) method
32,33
that allows for evaluating kinetic parameters without choosing the reaction model, was applied. F-W-O is a relatively simple method of determining activation energy directly from mass loss versus temperature data obtained at several heating rates. This method is shown by the following equation: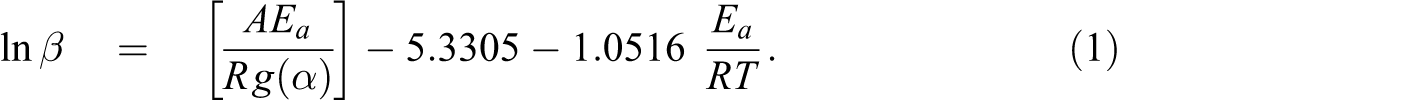
where β is a heating rate in °C/min, Ea is an activation energy in kJ/mol, A is a pre-exponential factor, R is an universal gas constant in J/Kmol, T is an absolute temperature in K, and g(α) is an integral reaction type of kinetic function. In this study, the activation energy calculated by the isoconventional methods is called apparent activation energy because it is the sum value of activation energies of the chemical reactions and physical processes in thermal degradation.
The plots of isoconversional activation energy as a function of conversion extent (α) using F-W-O method for PP and PE and its composites are shown in Figure 7. For PP composite system (Figure 7(a)), the activation energy of PP is nearly constant at about 70 kJ/mol at α = 0.05–0.9. With the addition of CNT, the activation energy of the PP-CNT5 progressively increases from 80 to 180 kJ/mol as α increases from 0.05 to 0.85. For OMMT composite, Ea of PP-OMMT5 moderately increases from ≈60 to ≈95 kJ/mol as α increases from 0.05 to 0.35. At α > 0.25, the activation energy of PP-OMMT5 is nearly constant at ≈90 kJ/mol. For PE composite system (Figure 7(b)), the activation energy of PE shows a slight increase from ≈80 to 125 kJ/mol during initial degradation (α < 0.20). These values agree well with the activation energies found for decomposition of peroxides, 23 which suggests this process to be a rate-limiting step of the thermo-oxidative degradation. The remaining reaction shows unstable value of the activation energy. The activation energy of PE-CNT5 gradually increases and remains constant at ≈225 kJ/mol for α > 0.45. For PE-OMMT5, the activation energy starts from 60 kJ/mol and progressively increases throughout the degradation process. As compared with OMMT loading, the results of the activation energy, according to our finding, show that the addition of CNT into the polyolefins causes better improvement in thermo-oxidative stability. There are a number of research reporting that CNT can enhance the thermal stability of PP, 34 polystyrene, 35 and acrylonitrile-butadiene-styrene (ABS) elastomer. 36
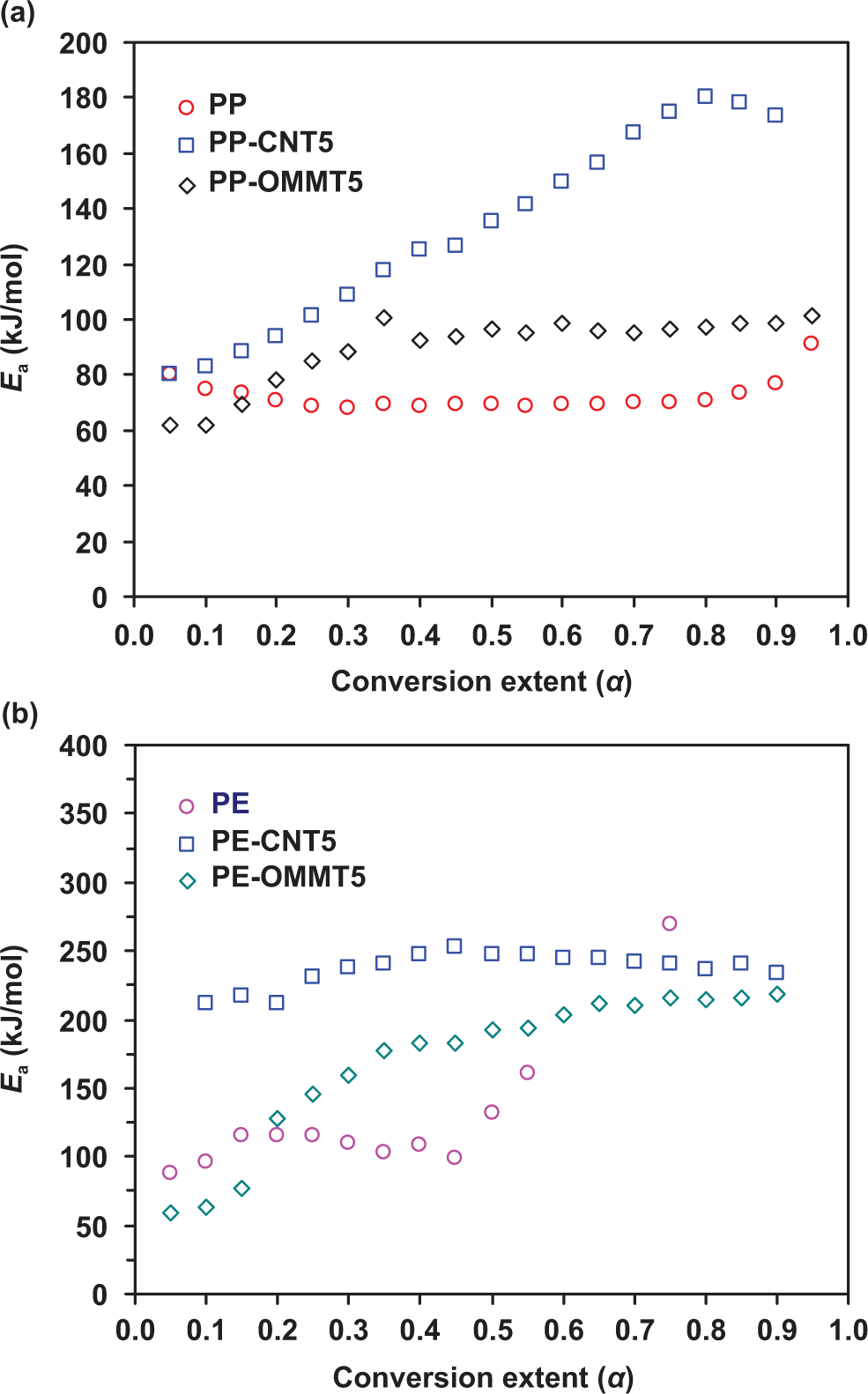
Dependence of activation energy (Ea ) on conversion extent (α) for of thermo-oxidative decomposition of PP (a) and PE (b) composite systems at a heating rate of 10°C/min in air. PP: polypropylene; PE: polyethylene.
To compare the performance of CNT and OMMT as a thermal-stabilizing modifier of the polyolefin-based composites, the relative activation energies (Ea of the composite divided by that of its corresponding matrix) of all composite systems are shown in Figure 8. It is seen that the relative Ea of all samples are mostly unstable with α. At α < 0.20, PP-OMMT5 and PE-OMMT5 show the relative Ea lower than unity. At α ≥ 0.20, the relative Ea of most samples is higher than unity, indicating improved thermo-oxidative stability of the composites over that of its corresponding polymer matrix. Especially for PE-CNT5, the relative Ea value is found to be 2.0–2.5 times that of PE in the α range from 0.0 to 0.50. It is noticed that at high α (α > 0.60), the thermo-oxidative stability of OMMT-containing composites is better than that of CNT composites.
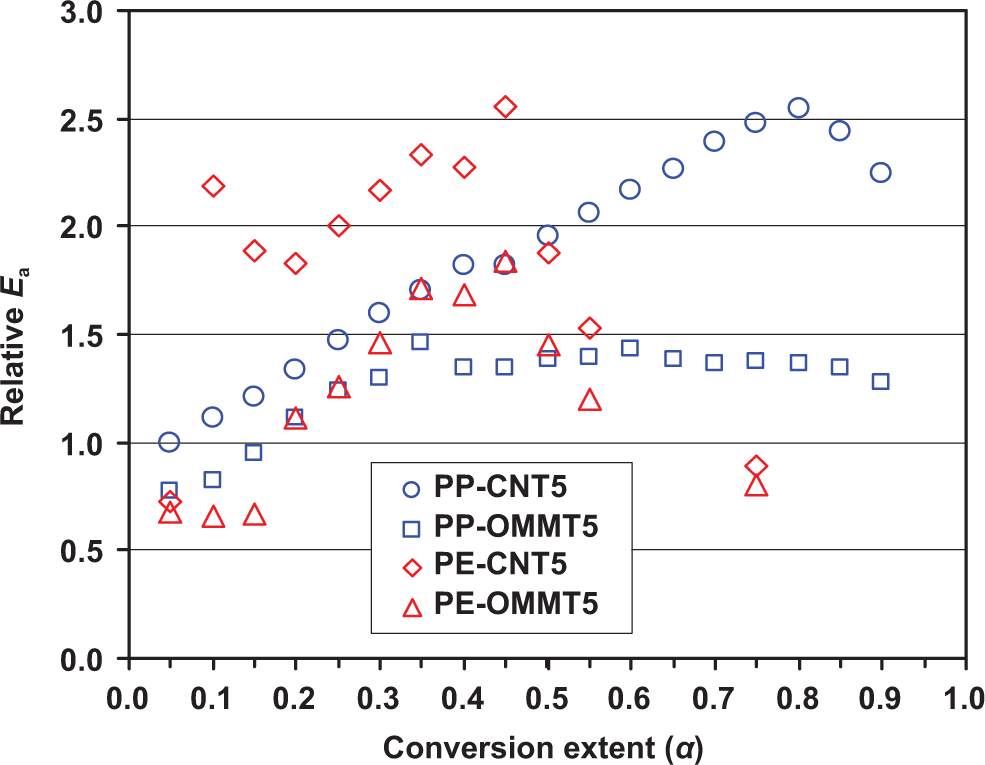
Relative activation energy (Ea (composite/Ea (matrix)) for the composite as a function of conversion extent (α).
To obtain more kinetic parameters (n, lnA), the Kissinger method
37
is widely used to calculate the kinetic parameters at a fixed conversion. This method involves the temperature (T
max) at the maximum mass-loss rate (dα/dt)max. The apparent activation energy can be determined using this method without precise knowledge of the reaction mechanism. This method is based on the following equation: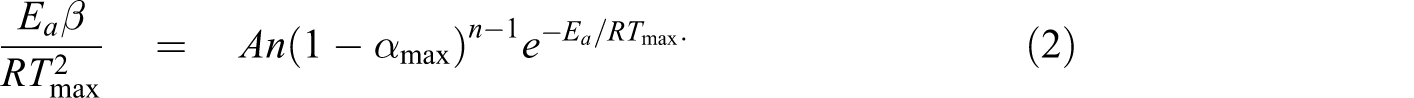
Kissinger assumes that the product 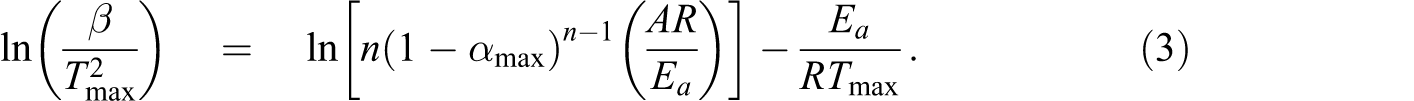
The slope of 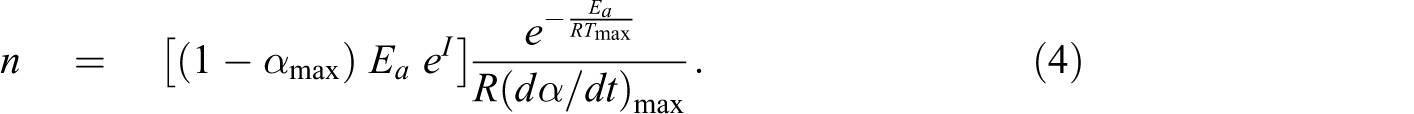
Then, the A value can be calculated by the substituting n into the intercept equation:
In the present work, Kissinger method determining the kinetic parameters at a fix conversion was employed in order to relatively compare the whole kinetic information (Ea , lnA, and n) among the neat polymer matrices and its composites containing different fillers. Table 2 shows the kinetic parameters corresponding to the first major degradation step for all samples in air. It is seen that the kinetic parameters of the neat matrices and the polymer composites are different. For PP composite system, the Ea values are in the following order: PP-CNT5>PP-OMMT5>PP. Note that the Ea of PP is much lower than that of PP-CNT5. For PE composite system, the Ea value of CNT-containing composite is much higher than those of PE-OMMT5 and PE, respectively. The results of Ea for all samples calculated using Kissinger method show a similar trend as that calculated using F-W-O method. The decomposition order (n) of the composite is slightly higher than that of its polymer matrix, suggesting that the decomposition mechanism of the composite becomes more complex compared with the neat matrix. No significant difference in reaction orders was observed between the composites with different fillers.
Nonisothermal kinetic parameters for the first major degradation step in air of the neat polyolefins and its composites calculated using Kissinger Method.a
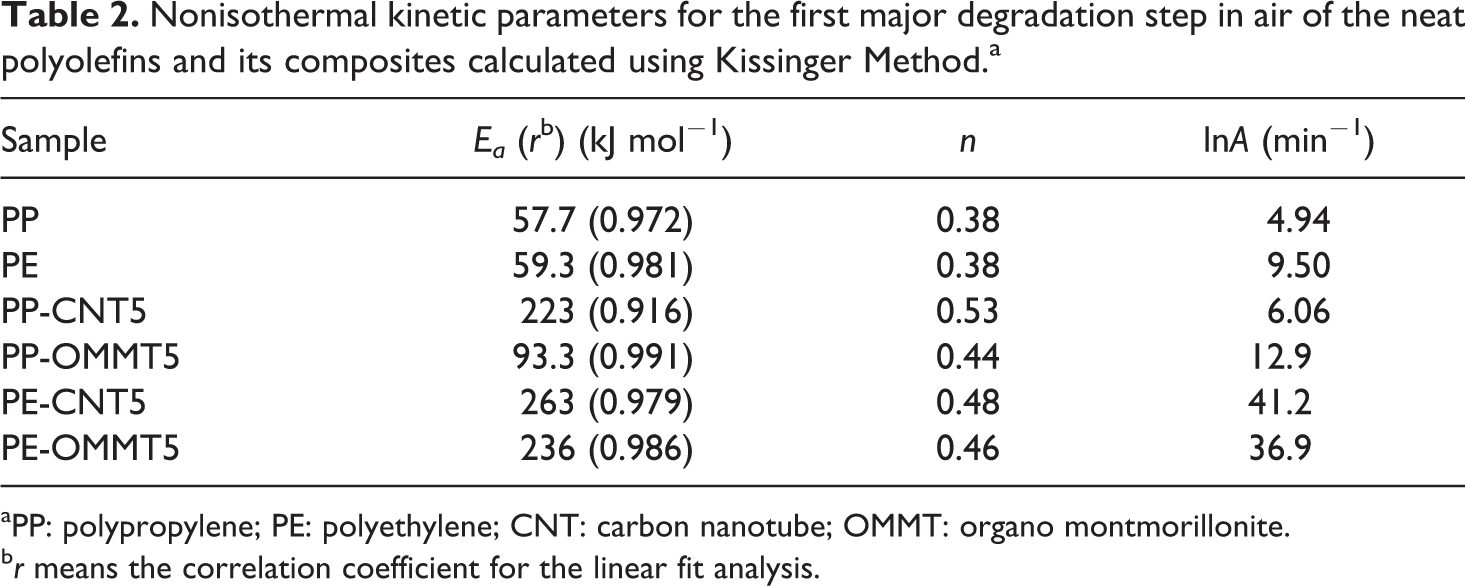
aPP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
b r means the correlation coefficient for the linear fit analysis.
Isothermal decomposition behavior
Both nonisothermal and isothermal TGs are complementary and necessary to obtain a full concept description of the decomposition process. To elucidate the thermo-oxidative degradation behavior, the isothermal TG and DTG curves of PP, PE, PP-CNT5, and PP-OMMT5, PE-OMMT5 and PE-CNT5 at 320°C in air are presented in Figure 9, and the isothermal decomposition data are presented in Table 3. W 20 min and W 30 min represent the amount of char left after heating for 20 and 30 min, respectively. t max represents the time at maximum weight-loss rate, (dα/dt)max for the first stage of decomposition. For PP composite systems, the neat PP and its composites start to first degrade after heating for 10 min. It is seen that PP exhibits a rapid decomposition during the first 20 min of isothermal heating whereas PP-CNT5 and PP-OMMT5 show a relatively slower decomposition as seen from Figure 9(Ia). However, at the initial decomposition region, PP isothermally degrades with highest rate, whereas the decomposition rates of PP-CNT5 and PP-OMMT5 are nearly the same (Figure 9(Ib)). Note that, by isothermal heating for 20 min, the highest amount of char left is observed for OMMT-containing composite, followed by PP-CNT5 and PP, respectively. This result agrees well with that obtained from the nonisothermal investigation. Moreover, the t max of PP and its composites are mostly comparable at ≈15 min.
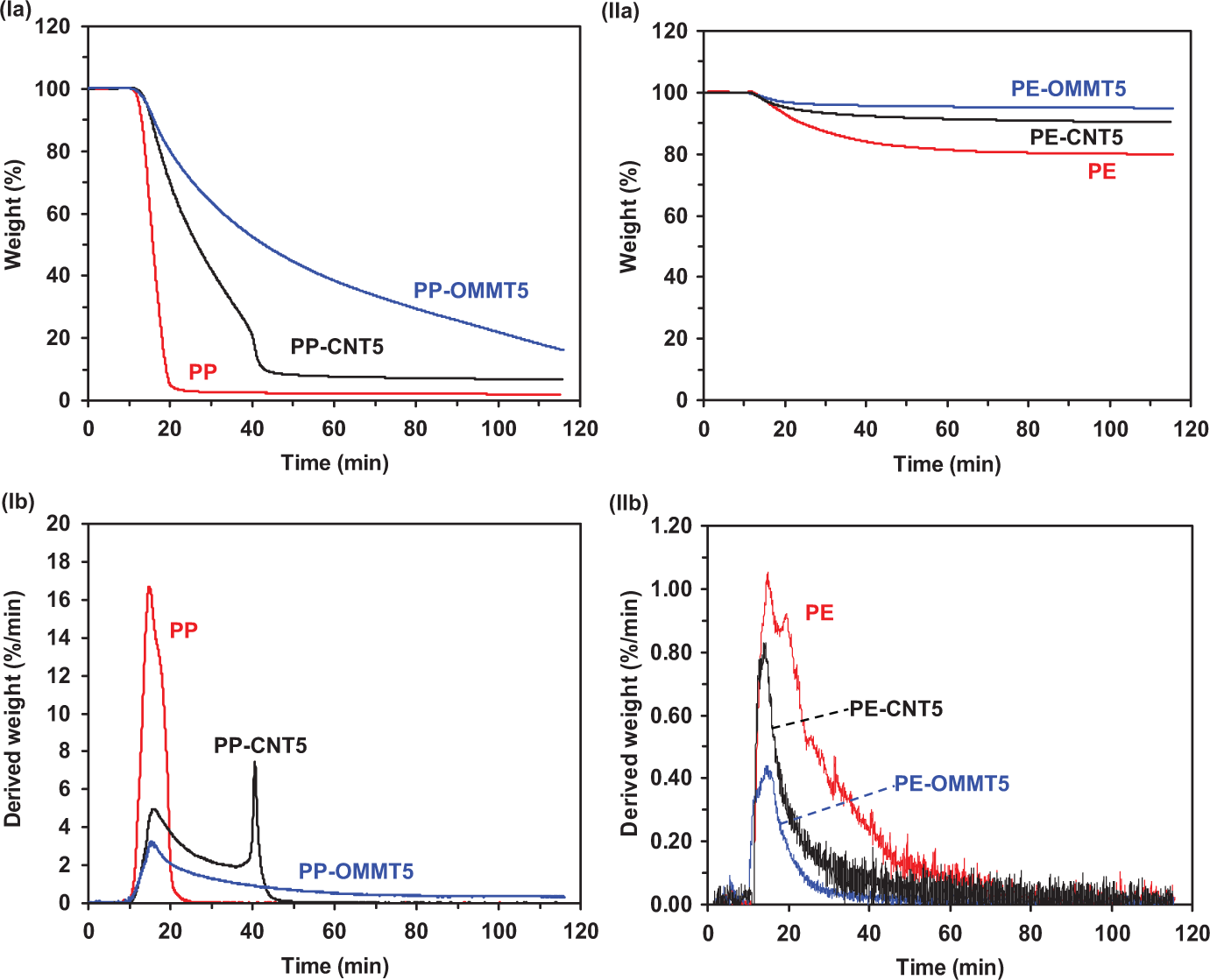
Isothermal TG (row a) and DTG (row b) curves of the composites based PP (column I) and PE (column II) thermoplastics under heating at 320°C in air. TG: thermogravimetric; DTG: derivative TG; PP: polypropylene; PE: polyethylene.
Thermo-oxidative decomposition data of the neat polyolefin and its composites under isothermal heating at 320°C in air.a
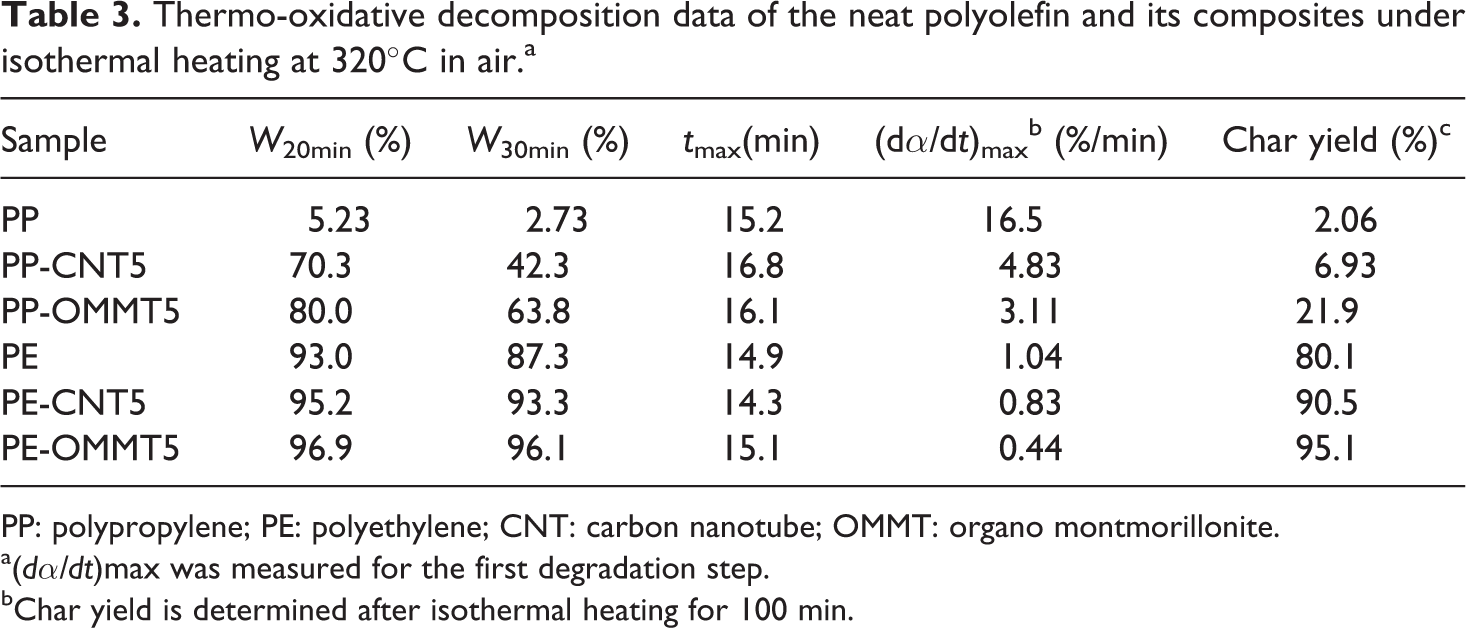
PP: polypropylene; PE: polyethylene; CNT: carbon nanotube; OMMT: organo montmorillonite.
a(dα/dt)max was measured for the first degradation step.
bChar yield is determined after isothermal heating for 100 min.
For PE composite system (Figure 9(IIa) and (IIb)), the isothermal TG and DTG curves show much difference from those of PP composite system. That is, a relatively slower decomposition is observed for PE composite systems. The decomposition of PE becomes relatively more rapid, whereas a slow decomposition is observed for PE-CNT5 and PE-OMMT5. This indicates that the addition of OMMT and CNT into PE significantly renders the retardation of the thermo-oxidative reaction. Moreover, the thermo-oxidative stability of PE is better than PP. Note that, as seen from Figure 9(IIa), PE, PE-OMMT5 and PE-CNT5 isothermally degrade with leaving high amount of char (> 80 wt%). From Figure 9(IIb), the (dα/dt)max of the PE and PE composites are much lower than those of PP and its corresponding composites. Moreover, the unstable DTG curves revealed for PE composite system indicates more complex decomposition mechanism compared with the PP composite system. However, the kinetic study of isothermal decomposition is also complementary and necessary to obtain a concept description of decomposition behavior as well as the nonisothermal analysis. Since the results obtained from kinetic analysis of isothermal decomposition investigated in this work are in agreement with those obtained from nonisothermal TG data, the kinetic results of isothermal decomposition are not shown here.
However, in case of PE matrix, the presence of CNT and the OMMT causes similar effects on the thermo-oxidative stability but for PP matrix, there are some differences. This is observable both for isothermal and for nonisothermal heating. The difference partly arises from some different decomposition mechanisms of PP and PE. Like PE, the degradation of PP occurs by random scission followed by radical transfer process. Unlike PE, however, PP degradation does not involve chain branching or cross-linking and the reaction products are different from those of PE degradation. Therefore, although the same type of fillers was added, thermal stability may be different as a result of the different environments of the chemical species.
Morphology of the polymers after isothermal heating
Morphological observation on the surfaces of the samples after isothermal heating at 320°C for 20 min is shown in Figure 10. For PP composite system (column I), the neat PP in the original form of sheet sample is degraded by leaving some residues. Interestingly, PP-CNT5 exhibits thermally stable material as seen from that the sheet specimens. In the case of PP-OMMT5, a large distortion of the specimen is observed, indicating the less thermo-oxidative stability compared with PP-CNT5. For PE composite system, the morphological results show the same trend as that of PP composite system. The addition of CNT into PE polymer exhibits the remarkable improvement in thermo-oxidative stability as seen from a slight change in the specimen geometry of PE-CNT5. However, as compared with PP composite system, the PE composite system seems to show better thermo-oxidation resistance under isothermal heating. The reasons of the improved thermo-oxidative stability of the polyolefins on addition of CNT are mainly governed by two distinct physical processes. 38 First, the structure network layer acts as a shield and re-emits much of the incident radiations back into the gas phase, decreasing the polymer degradation rate. Second, the presence of CNT increases the thermal conductivity of the polymer. As a result, the time to protect the heat of the polymer is prolonged. In the case of OMMT, the improved thermal stability can be ascribed by the formation of a barrier against heat and volatiles by migration of the clay nanolayer toward the material surface, followed by char formation, together with increased melt viscosity for exfoliated nanocomposites. 31,38

Morphology of the neat polyolefin and its composites after isothermal heating at 320°C for 20 min in air.
Conclusions
The direct comparison of heat-stabilizing performance of OMMT and CNT as a minor blend component for polyolefin-based composites was investigated using nonisothermal and isothermal TGs in air. Before blending, OMMT and CNT show the lowest and highest thermo-oxidative stability, respectively. Although T onset of OMMT shows the lowest value compared to those of the polyolefins, the thermo-oxidative stability of OMMT-containing composites is much improved when compared with that of its corresponding polymer matrix. From DSC profiles of nonisothermal degradation, the exothermic processes for all neat materials and composites were observed in air. The thermo-oxidative stability is strongly affected by the inherent properties of polymer matrix, types of fillers, and heating modes. The activation energy calculated by F-W-O and Kissinger methods suggests that the incorporation of OMMT and CNT into PP or PE significantly improves the thermo-oxidative stability of the composites. From nonisothermal investigation, as compared with OMMT, CNT acts as the better heat-stabilizing material for the polyolefin-based composites. Under isothermal heating at 320°C in air, the neat polyolefin and its composites mostly start to degrade after isothermal heating for 15 min. The morphological evidences after isothermal heating suggest the heat-stabilizing performance of CNT as an appropriate modifier for the polyolefin-based thermoplastic composites over that of OMMT.
Footnotes
Acknowledgments
We also would like to acknowledge PERCH-CIC for partial financial support. Moreover, the authors thank Dr. Taweechai Amornsakchai, Department of Chemistry, Faculty of Science, Mahidol University for the gift of clay and CNT samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to express our profound gratitude and sincere appreciation to Faculty of Science, Mahasarakham University, for financial support (fiscal year 2013).
