Abstract
This work aimed to study the effects of γ radiation on the properties of ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend reinforced with 50 phr of carbon black and cross-linked either with sulfur/tetramethylthiuram disulfide or dicumyl peroxide. Irradiation doses were 100, 200, and 400 kGy. It was observed that doses higher than 200 kGy practically destroy the assessed properties of all obtained elastomeric materials, irrespective of used curing system. However, samples cured with sulfur showed a pronounced decrease in mechanical properties.
Introduction
Polymer blending was recognized in the last few decades as the most promising way to prepare new material with tailored individual properties. Their chemical and physical properties make them suitable engineering materials for chemical industry, electric insulators, and many other applications. 1 Ethylene propylene diene terpolymer (EPDM) is one of the most common polymers for nuclear engineering application and is used, for instance, in nuclear power plants. In this kind of application, polymer sheathing may be exposed to elevated temperatures and γ irradiation, which will accelerate its ageing and consequently degrade its useful application lifetime. 2 Chlorosulfonated polyethylene (CSM) is an important rubber that has been frequently used in many applications such as sheeting cable and geomembrane due to its outstanding resistance to deterioration by heat, oils, ozone, and oxidation. 3
Figures 1 and 2 are showing cross-linking process during sulfur and peroxide vulcanization, describing both the desirable reactions that lead to effective cross-link formation. In sulfur-accelerated vulcanisation (Figure 1), first zinc oxide reacts with accelerator to form zinc–accelerator complex. At high temperatures, zinc–accelerator complex reacts with sulfur to form sulfur-rich zinc accelerator complex that reacts with rubber macromolecules and rubber–sulfur accelerated complex is formed. The cross-linking process takes place in react rubber molecules with rubber–sulfur accelerated complex. Of course, the desired reaction pathway for a polymer radical (P
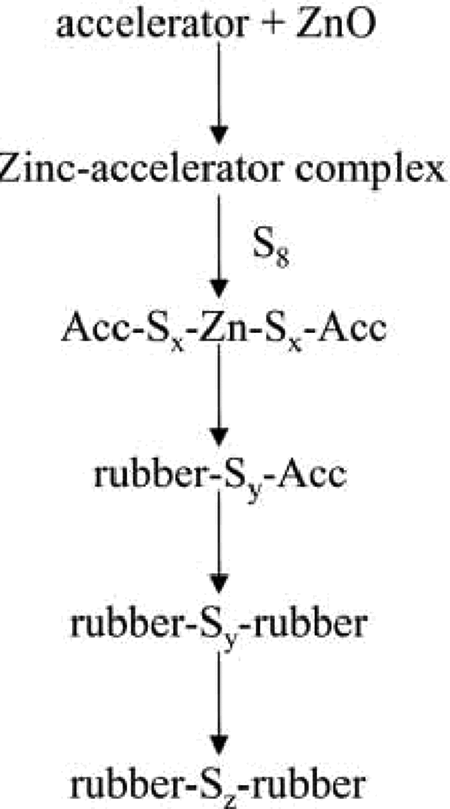
Cross-linking during vulcanization by sulfur.
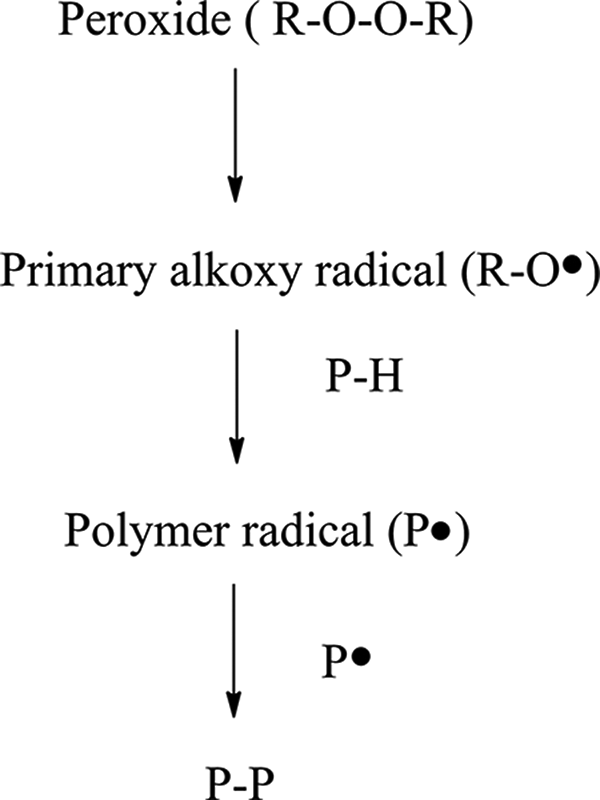
Cross-linking during vulcanization by peroxide.
This work aims to study the effects of γ irradiation on the properties of vulcanized EPDM/CSM rubber blend (50:50) filled with 50 phr of carbon black and cured with sulfur or peroxide. The vulcanization characteristics of these materials, such as scorch safety and cure rate, were also analyzed.
Experimental
Materials
Two rubbers were used in this study: (a) EPDM (Vistalon 2504) supplied by Exxon Mobil (France) and (b) CSM rubber (Hypalon 40 S) supplied by Du Pont (Delaware, USA). The compounding recipes of the blends are given in Table 1.
Formulation of ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blends.
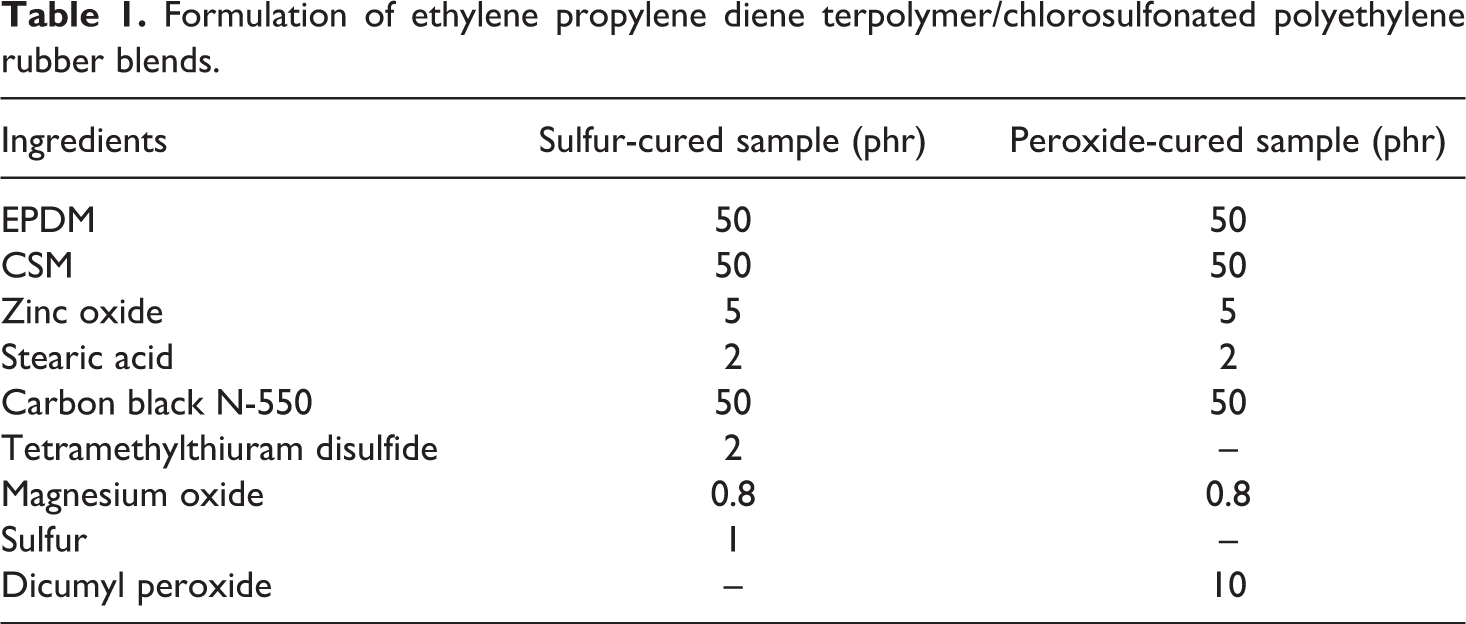
Carbon black (type N550; Degussa, Milan, Italy) with a high structure dibutyl phthalate was added at 121 ml/100 g and an average primary particles size of 40–48 nm was used as filler. Tetramethylthiuram disulfide (Bayer, Leverkusen, Germany), magnesium oxide (Bayer, Leverkusen, Germany), sulfur (Zorka, Šabac, Serbia), and dicumyl peroxide (Akzo Nobel, Flexsys Holandia, The Netherlands) were used as a curing system for blend.
Mixing procedure
Mixing of EPDM with CSM rubber mixtures was carried out on a laboratory open two-roll mill (400 mm diameter and 600 mm working length). The gear friction of the mill is 1:1.4. The hollow rolls were cooled by flushing water in order to regulate the temperature not exceeding 60°C during different stages of mixing, according to ASTM D-15-627. The rubber mixes obtained were sheeted and left for a period of at least 6 h before testing. The addition of ingredients during mixing was carried following the same order and conditions of mixing.
The rubber mixes were compression molded at 160 ± 2°C, using an electrically heated hydraulic press at their optimum cure time derived from rheological measurements. The applied hydraulic force during vulcanization attained 6.7 MPa. Dumbbell shaped TS was punched from the compression molded sheet along the mill grain direction using C-type dumbbell specimens, according to ASTM D-412.
Cure characteristics
The cure characteristics of rubber compounds were carried out using an oscillating disc rheometer (model 4308, Zwick, Germany).
The measured parameters are: M l-minimum torque, M h-maximum torque, t s2-time to 2 units of torque increase above minimum, and M c90-torque at 90% of full torque development.
The cure rate index (CRI) is
These measurements were accomplished according to ASTM D-2084.
Mechanical testing
The tensile properties were determined using the tensile testing machine type Zwick 1445, according to ASTM D-412. The average value of the mechanical properties was calculated using at least three samples. A cross head speed of 50 mm/min was used and the tests were performed at 25°C.
Hardness measurements
Samples of at least 0.12 mm in thickness with flat surface were cut for hardness test. The measurement was carried out according to ASTM D 2240 using Shore A durometer of model 306L type. The unit of hardness is expressed in kilogram per square millimeter.
Gamma irradiation
Radiations of elastomeric materials have been performed in air in the Cobalt-60 radiation sterilization unit with the dose rate of 10 kGy/h and total absorbed dose of 100, 200, and 400 kGy. The radiation dose of 400 kGy can be considered as the relatively big dose, which exceeds the doses for degradation of radiation degradable polymers many times and is also above the typical doses used in practice for radiation modification of polymer-based products.
Swelling measurements
The swelling degree was determined on the basis of equilibrium solvent–swelling measurements in toluene. The samples were submerged in the solvent and after the swelling equilibrium was reached, which means that no change in the weight of the swollen sample was observed, the mass of solvent was determined according to the ASTM D 471. The results were expressed as the mass of solvent absorbed per gram of blend. The percentage of absorption was calculated using the following equation
where m t is the mass of sample at time t and m o is the dry weight of the sample.
Results and discussion
Cure characteristics
The cure characteristics are the torque evolution, the scorch time t s2, optimum cure time t c90, and the CRI of EPDM/CSM rubber blend, which are shown in Table 2.
The cure characteristics and mechanical properties of ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend vulcanized by either sulfur or peroxide at 160°C.
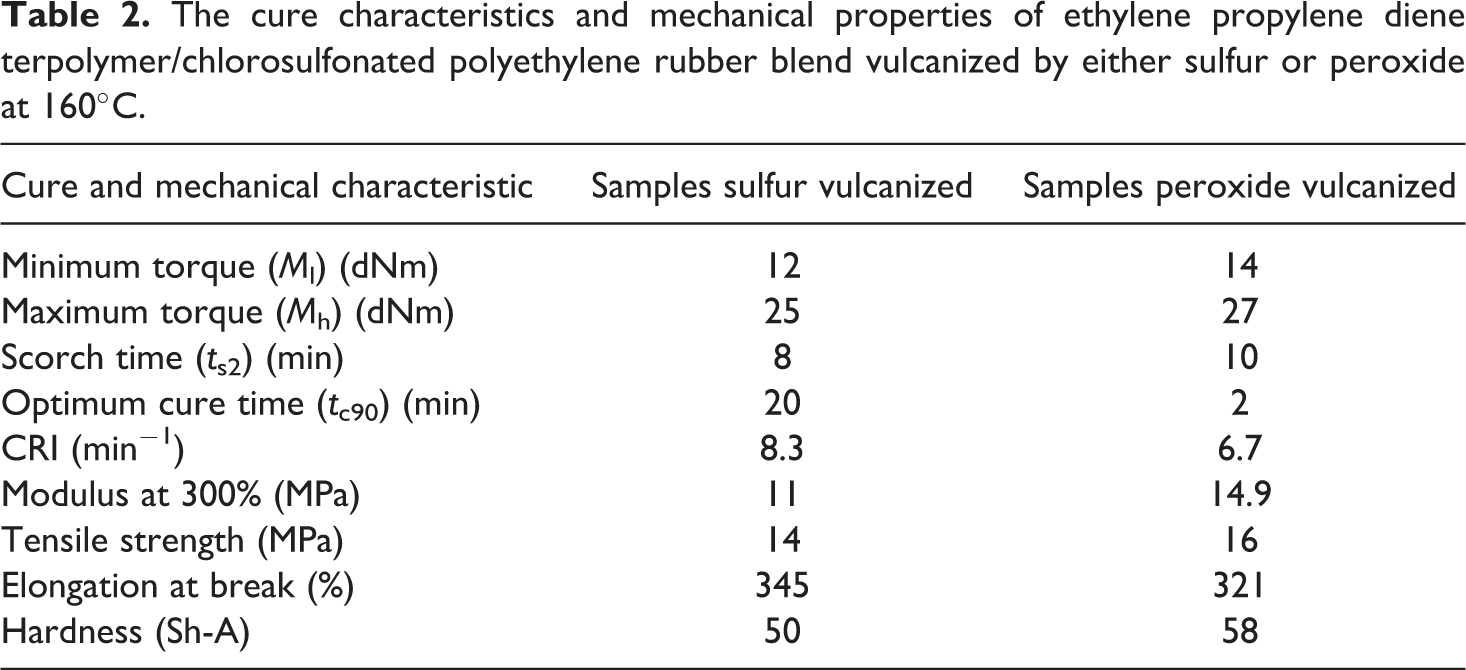
The measurement of vulcanization process was carried out indirectly by determining the torque produced at varying temperatures in MOD rheometer. The higher the torque obtained, the higher the number of cross-links created. Maximum torque used as a measure of cross-link density. The higher torque is obtained in “peroxide-cured” EPDM/CSM rubber blend. Depending on the choice of vulcanization chemical, the start of vulcanization, that is, scorch time (t s2), of a rubber compounds can be rapid or slow. The peroxide-cured rubber blend has slower t s2 than sulfur cured. The higher the cure temperature, the poorer are the mechanical properties of the vulcanizates. More activated precursors for cross-linking 8 –11 are formed as a result of the activation of double bond. For “sulfur-cured” rubber blend, activated precursors accelerated the vulcanization between EPDM and CSM chains and induced faster intermolecular cross-linking between these network precursors.
Mechanical properties
Gamma radiation is a powerful tool for rubber cross-linking; however, exposure to higher dosage degrades take place of prepared composite elastomeric materials. The extent of cross-linking and degradation of polymer network depends on its structural characteristics and the presence of initiators/sensitizers. 12 Vulcanization characteristics of rubber compounds along with mechanical properties of obtained elastomeric materials are presented in Table 2. It can be seen that the values for TS, 17.8 MPa; hardness (Shore-A), 58 Sh-A; and modulus at 300% elongation (M300), 14.9 MPa, for the peroxide-cured rubber blend were higher (14–27%), but elongation at break (321%) is lower when compared with sulfur-cured formulation rubber blend, before irradiation.
Figure 3 shows the effect of γ irradiation dosages on the TS rupture of sulfur and peroxide-cured blends. It can be seen that the TS values for both blends decrease with increasing irradiation dose. The initial stages of degradation of EPDM and CSM rubber generally indicate a random breaking of bonds in the polymer chain at higher dosages. 13 It is also interesting to note that the peroxide-cured blends exhibited higher retention of TS compared to sulfur-cured systems, but lower retention for irradiation doses higher than 200 kGy. This is due to the presence of short and rigid C–C bonds in the peroxide-cured systems, which resist degradation due to γ irradiation.
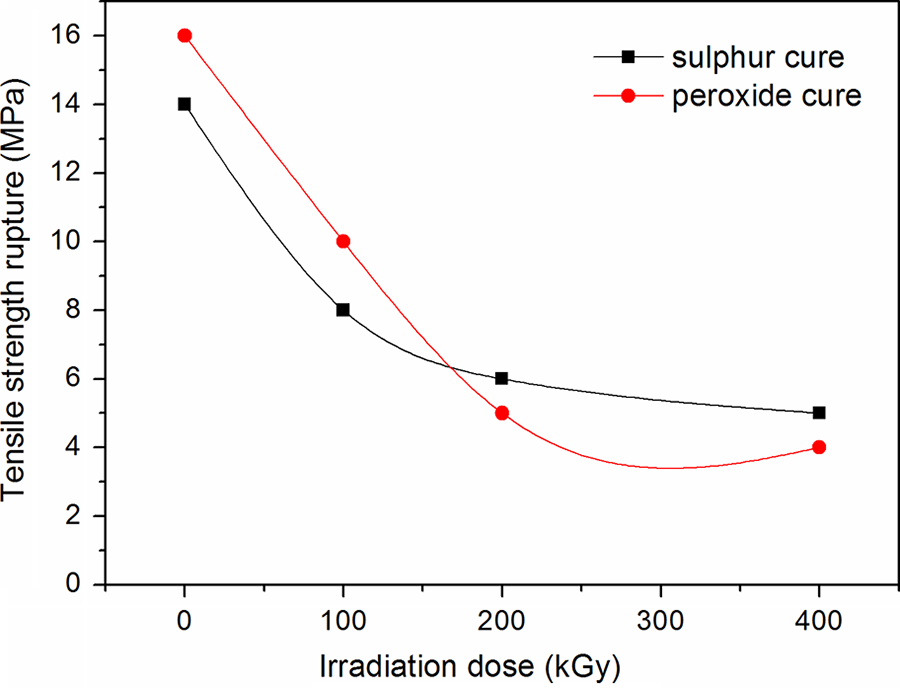
The effect of irradiation dose on tensile strength rupture for ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend cured with sulfur and peroxide.
Effect of irradiation dose on the 100% modulus retention for EPDM/CSM rubber blends for different cross-linking systems is presented in Figure 4. The peroxide-cured blends exhibited higher retention of modulus compared to sulfur-cured systems. This could be due to the higher cross-linking level achieved in the peroxide-cured blends during the exposure to γ irradiation.
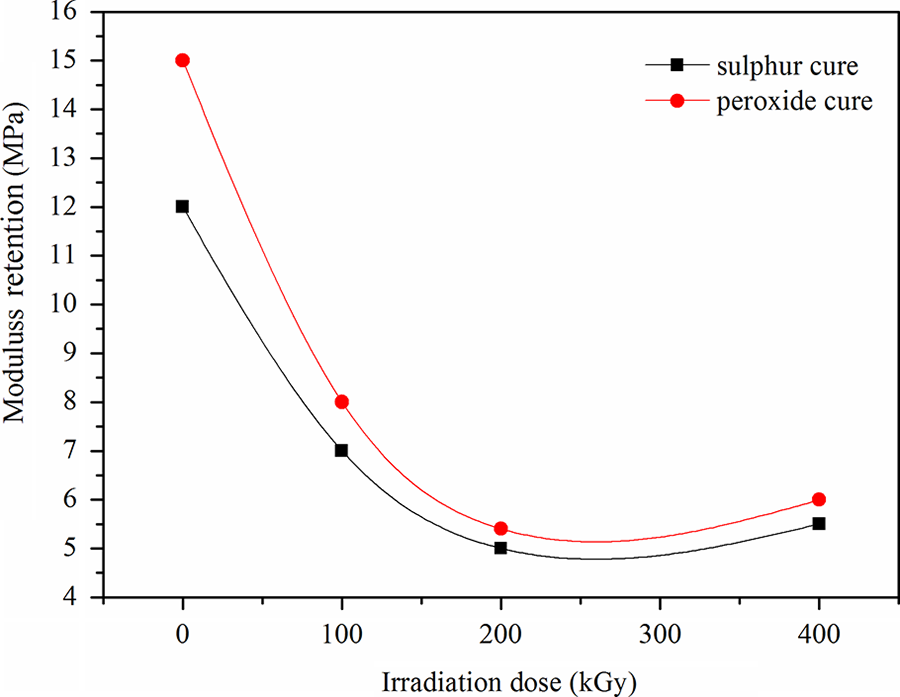
The effect of irradiation dose on modulus retention for ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend cured with sulfur and peroxide.
Irradiation affects on the elongation at break retention property for the two vulcanization systems, as shown in Figure 5. Sulfur-cured samples showed higher elongation at break retention compared to peroxide-cured samples for all irradiation doses. A significant reduction in elongation leading can be observed for peroxide-cured samples because of polymeric chain scission, with sudden reduction at lower doses and almost leveling off for doses above 200 kGy. Changes in the vulcanized network structure are responsible for the change in the vulcanized’s properties, due to the curing-system formulation. 13 Peroxide-cured samples experienced a non-significant reduction in properties at doses up to 200 kGy; above this dose, sudden properties’ reduction have occurred, proportional to doses, probably due to a higher selectivity in bonds rupture verified for the cross-linker. For peroxide-cured samples, there was a reduction in the properties proportional to the applied dose; for doses higher than 200 kGy, a sudden reduction in properties probably imparted by very large polymeric chain degradation was observed.
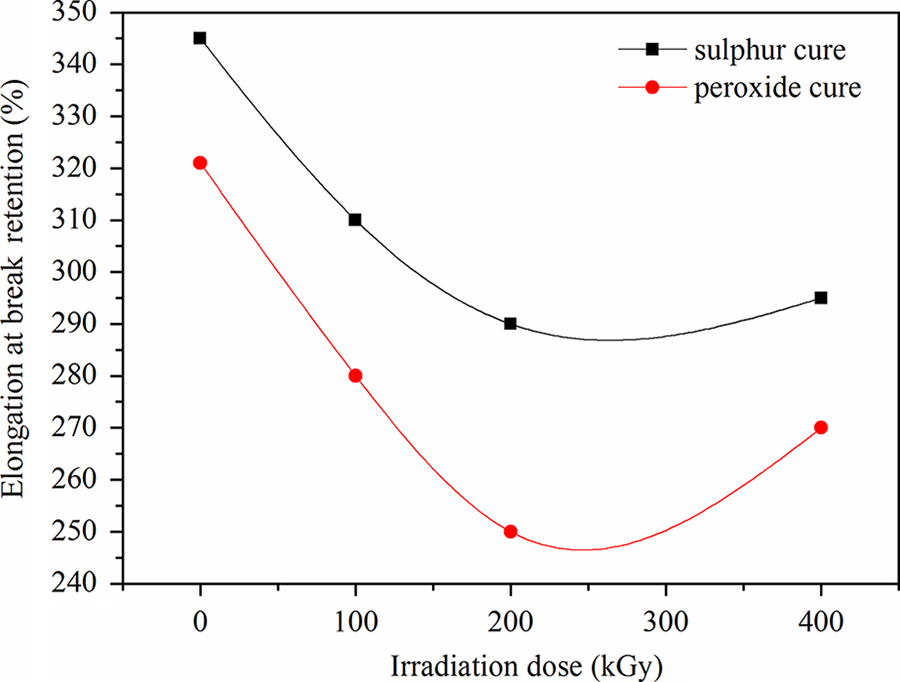
The effect of irradiation dose on elongation at break retention for ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend cured with sulfur and peroxide.
Figure 6 shows the effect of γ irradiation dosages on the retention of hardness of sulfur and peroxide-cured blends. It can be seen that the retention hardness values for both compositions increase with increasing irradiation dose reaching its maximum value at 200 kGy and then decrease for doses higher than that. The peroxide-cured EPDM/CSM rubber blend has higher retention hardness than sulfur-cured EPDM/CSM rubber blend. Hardness is proportional to the entangled bonds formed during the vulcanizing process and consequently directly affected by the amount of sulfur or peroxide present in vulcanizing systems. The higher is the amount of sulfur and peroxide, the higher is the hardness and the degree of cross-linking. Peroxide-cured blend showed slightly higher hardness values, pointing to a higher degree of cross-linking of these mixtures. These samples showed equal losses in properties for all doses, as shown in Figure 6, indicating that irradiation did not affect the compound’s hardness, probably because the EPDM units are practically non-reactive toward sulfur under the condition of vulcanization and only the units are involved. Peroxide-cured samples showed an increase in hardness related to the dose applied, as a function of a decrease in molecular weight, because cross-linking would be expected only through the C–C bond. For sulfur and peroxide-cured samples, the hardness remain almost unchanged for almost all dose values, whereas for doses higher than 200 kGy, samples showed a sudden reduction in hardness values indicating polymer degradation, because of the dose-dependent nature of chains scission. 14 In fact, the vulcanized samples with “sulfur” and “peroxide” showed that mechanical properties decreases proportionally with the increase in radiation. This decrease was caused by a loss of molecular weight in the irradiated polymers, because irradiated EPDM/CSM rubber blends loses molecular weight event doses, which are not that high—this loss can be proved particularly through viscosity measurements. 15 The vulcanized sample with sulfur showed initially a sharp decreasing at low doses, probably because the presence of polysulfides (C–S–Sn–C), where n < 41 16 have lower resist to irradiation. For the samples cured with peroxide, the molecular weight loss occurred proportionally to the dose, showing that the loss in molecular mass follows the increase in the dose and more severe property losses were observed at doses above 200 kGy. 17 –21 When the radiation dose increased to 400 kGy, the variation in properties becomes greater.
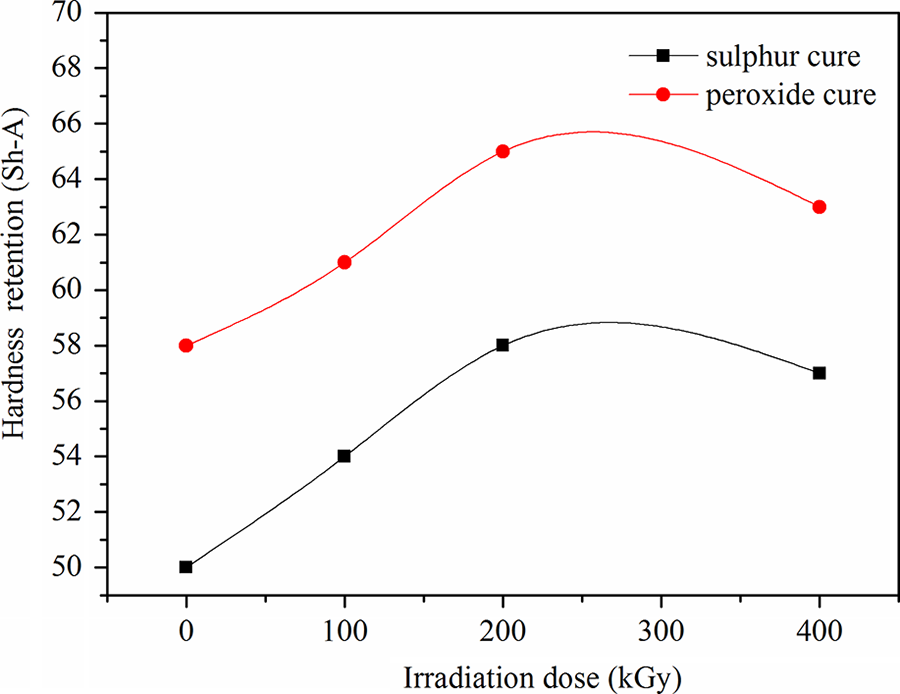
The effect of irradiation dose on hardness retention for ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend cured with sulfur and peroxide.
Swelling properties
CSM rubber has excellent resistance to most chemicals and this explains why percentage of swelling of sulfur and peroxide-cured EPDM/CSM rubber blend matrix is low. Figure 7 illustrates the variation in swelling degree retention as a function of irradiation dose for all prepared rubber blends composites.
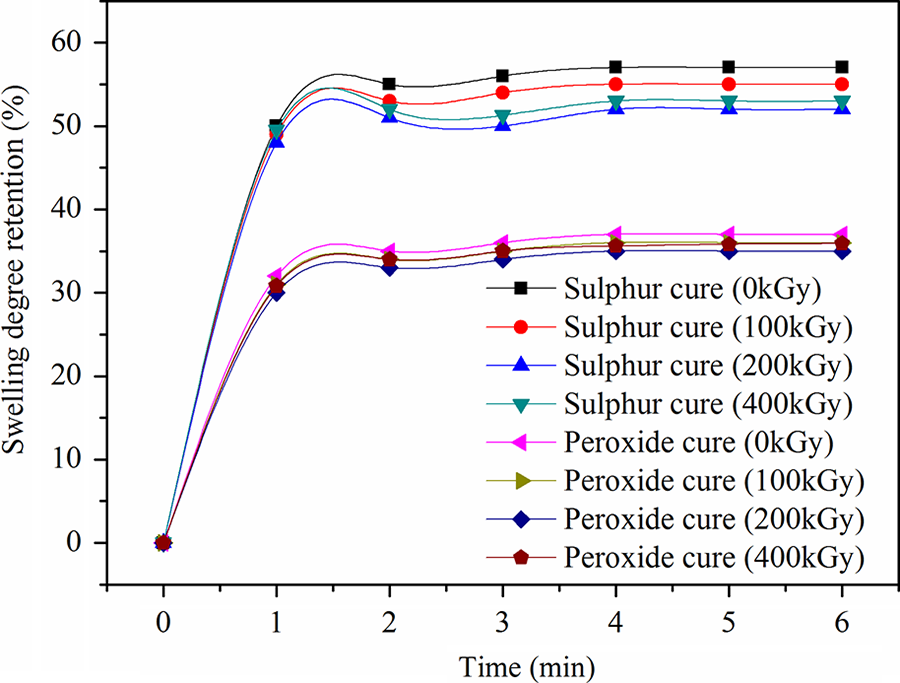
Swelling degree retention for ethylene propylene diene terpolymer/chlorosulfonated polyethylene rubber blend cured with sulfur and peroxide.
The swelling degree values for unirradiated as well as corresponding irradiated ones are also given in the same figure for sake of comparison. It can be seen that these unirradiated composite have attained comparatively high swelling degree values. On the other hand, the swelling degree values decreased with irradiation dose at 200 kGy and then they increased over any further increase in dose until 400 kGy. These data indicate clearly that the irradiated composites possess better resistance to be swelled by toluene until reach 200 kGy by means of cross-linking that occurs at low doses then degradation may then predominate with respect to sulfur and peroxide-cured EPDM/CSM rubber blends composite.
Conclusion
The elastomeric materials cross-linked by different systems have quite different sensitivities to irradiation. The values for TS, hardness (Sh-A), and modulus at 300% elongation (M300), for the peroxide-cured rubber blend were higher (from 14 to 26%), but elongation at break value is lower when compared with sulfur-cured rubber blend, before irradiation. The highest degradation occurred in sulfur vulcanized materials, being observed that for low doses the cross-linking was preserved, where as for high doses, just degradation occurred. Peroxide-cured samples were more stable at low doses, as their properties were maintained constant, whereas at higher doses, they showed severe degradation. At doses higher than 200 kGy, there was a major loss observed in the assessed properties for all rubber blend, irrespective of the vulcanization system used, except for the sulfur-cured samples. Irradiated composites possess better resistance to swelling in toluene.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was granted by the Ministry of Science and Technological Development of the Republic of Serbia (Projects Numbers 45022, 45020, and 172056).
