Abstract
Nanosilver/poly(2-acrylamido-2-methylpropanesulfonate sodium) (nano-Ag/PAMPS) composites were synthesized by a microwave synthesis method. The mechanism of catalytic reduction of Ag ion by atomic nitrogen (from PAMPS) was explored. The composites were characterized by the ultraviolet–visible spectroscopy, x-ray diffractometry, transmission electron microscopy, x-ray photoelectron spectroscopy (XPS), Fourier transform infrared spectroscopy, and thermogravimetric analysis (TGA). The results show that Ag nanoparticles (AgNPs) possessed face-centered cubic structure and they were homogeneously dispersed in PAMPS matrix. XPS analysis reveals the interaction between AgNPs and PAMPS matrix. TGA proves that the thermal stability of the pure PAMPS was higher than that of the nano-Ag/PAMPS composites.
Introduction
Research activities on inorganic-polymer nanocomposites are witnessed an exponential growth due to many new characters, such as thermal, electrical, mechanical, optical properties, and so on, which can be widely used in optical materials, electricity materials, biomedical materials, information materials, and other fields. 1 –3
Among the nanocomposites, especially, metal-based materials, such as, silver (Ag)-polymer nanocomposites, are of special interest. 4 –6 This is because nanoscale metal particles such as Ag and gold provide a very exciting research field due to their interesting optical, electronic, magnetic, and catalytic properties. 7 –9 Ag exhibits the highest electrical and thermal conductivities among all the metals and has applications in a variety of areas such as catalysis, conductive inks and pastes, and adhesives for electronic components, photonics, and photography. 10,11
As for the syntheses of metal-based nanocomposites, several methods have been established for the preparation of Ag-polymer nanocomposite, specially, including physical techniques such as thermal evaporation and laser sputtering and chemical techniques such as in situ chemical reduction, 12 sonochemical deposition, 13,14 ultraviolet (UV) radiation reduction, 15 electroless plating 16 and layer-by-layer assembly, 17 and so on. But, there are more or less defects in these synthetic technologies, such as using reductant, complex synthetic steps, difficult post-processing, and so on. Compared with these methods, microwave heating is a completely neoteric method for the preparation of nanosilver (nano-Ag)/polymer composites.
Microwave radiation has the advantage of heating uniformly and making metal nanoparticles (NPs) easily nucleate and manifests the unique superiority in the preparation of nanomaterials. Jiang et al., 18 Pal, 19 and Yao et al. 20 synthesized nano-Ag composites by microwave radiation, but they prepared nano-Ag composites with polymer existing as protective agent, which restricted the selectivity of polymer as matrices of composite materials. Poly(2-acrylamido-2-methylpropanesulfonate sodium) (PAMPS) that is formed by in situ polymerization has eximious dispersion, thermal stability, and a lot of other special performance; it has a greatly protective effect on Ag nanoparticles (AgNPs) as matrix. Xu et al. 21 reported that nanometal composites with PAMPS copolymer as continuous phase were synthesized ultrasonically. The synthesis of AgNPs composites by simpler and easier synthetic routes continues to be a challenging area of research; the microwave reaction can be conducted at room temperature and at ambient pressure. Furthermore, the reaction is free of reducing agents. At present, there is no report on in situ synthesis of nano-Ag/polymer composites by microwave radiation.
In this article, nano-Ag/PAMPS composites were prepared by using the silver nitrate (AgNO3) solution containing PAMPS monomer without a chemical reducing agent, in which Ag ion was reduced to AgNPs and the monomer was polymerized simultaneously under microwave radiation. The composites were characterized by Fourier transform infrared (FTIR), UV–visible (Vis), x-ray diffraction (XRD), transmission electron microscopy (TEM), x-ray photoelectron spectroscopy (XPS), and thermogravimetric analysis (TGA), respectively. The interaction between AgNPs and PAMPS matrix was discussed. Specially, the mechanism of Ag ion reduced to AgNPs catalyzed by PAMPS without metal-reducer under microwave was discussed.
Experimental
Preparation of nano-Ag/PAMPS composites
Appropriate amounts of PAMPS (used as obtained with out further purification), AgNO3, and potassium persulfate were dissolved in 50 mL stilled water (used as obtained with out further purification). Subsequently, the mixture was stirred to be homogeneous, while pH of the solution was adjusted to 6.5–7.0 by 5 wt% sodium hydroxide solution and then irradiated by a microwave (Nanjing lingjiang technology Co. Ltd, Nanjing, China) for 15 min. During the reaction procedure, the color of solution was changed from colorless to canary, saffron yellow, even to dark brown with the reduction of Ag ions to Ag atoms and even to Ag clusters. The reaction system was extracted and washed by methanol and dimethyl formamide, and the brown powder was obtained. Finally, the products were subjected to vacuum drying at 100°C for hours. The sample of pure PAMPS polymer was also prepared by means of the similar polymerization approach with the condition of adding no AgNO3.
Characterization of nano-Ag/PAMPS composites
For FTIR spectroscopies, the samples were pressed into pellet together with potassium bromide and were carried on a VECTOR-33 FTIR spectrometer (Bruker Co. Ltd, Billerica, USA), with the wave number ranging from 500 to 4000 cm−1 at a resolution of 2 cm−1. The UV spectrum of the samples were tested by UV-1600 dual-beam UV spectrophotometer (Beijing Rayleigh Instrument Co. Ltd, Beijing, China). XRD patterns of the nanocomposites were determined at a scanning rate of 0.03°s−1 in 2θ ranging from 10° to 85°, using a x-ray diffractometer of XD-3 (Beijing General Instrument Co. Ltd, Beijing, China). The morphology and size of nano-Ag composites were observed by JEM-2010 High-Resolution TEM (Japan Electronics Co. Ltd, Osaka-shi, Japan). X-Ray photoelectron spectroscopy measurement were performed using a ESCALAB 250 spectrometer (Thermo Fisher Scientific Inc, Massachusetts, USA). TGA was conducted on a thermogravimetric analyzer (Thermal Analysis Co., New Castle, USA) at a heating rate of 5°C min−1 under nitrogen protection.
Results and discussion
The FTIR spectra of the nano-Ag/PAMPS composites were shown in Figure s1. Some characteristic peaks of the polymers such as the 3437 cm−1 stretching vibration peak and the 1550 cm−1 bending vibration peak for N–H, as well as the 2927, 1658, 1209, 1043, and 629 cm−1 stretching vibration peak responding to C–H, C=O group, C–N group of sec-amide, S–O, and C–S, respectively, are given. There is no stretching vibration peak for C=C. All those results show that PAMPS as continuous phase of the nanocomposites was successfully prepared with PAMPS by microwave radiation.
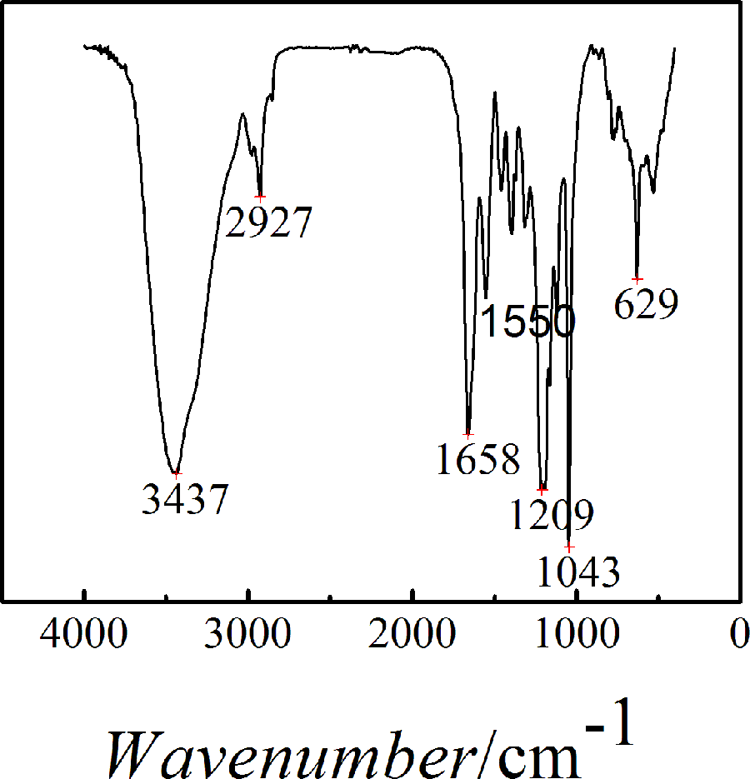
FTIR spectra of nano-Ag/PAMPS composites. FTIR: Fourier transform infrared; Ag: silver; PAMPS: poly(2-acrylamido-2-methylpropanesulfonate sodium).
To understand the shape of AgNPs during reaction, UV-Vis spectrometry of the nanocomposites during different step of reaction were diagrammed in Figure 2. There is an absorption peak in 410 nm for nano-Ag/PAMPS composites, which is analogous with that of Hiramatsu and Osterloh, 22 who reported that the surface plasma resonance absorption peak of nearly spherical shape or spherical AgNPs appeared in the vicinity of 425 nm. The absorption peak of AgNPs coated PAMPS polymer was not shifted with the microwave radiation time for the reaction system, as shown in Figure 2. Therefore, the morphology of AgNPs was not affected by PAMPS polymerization and microwave treatment. It hints that AgNPs are of nearly spherical shape or spherical structure in nanocomposites based on the variance of the width and strength of the absorption peak in the UV-Vis spectra of nanocomposites.
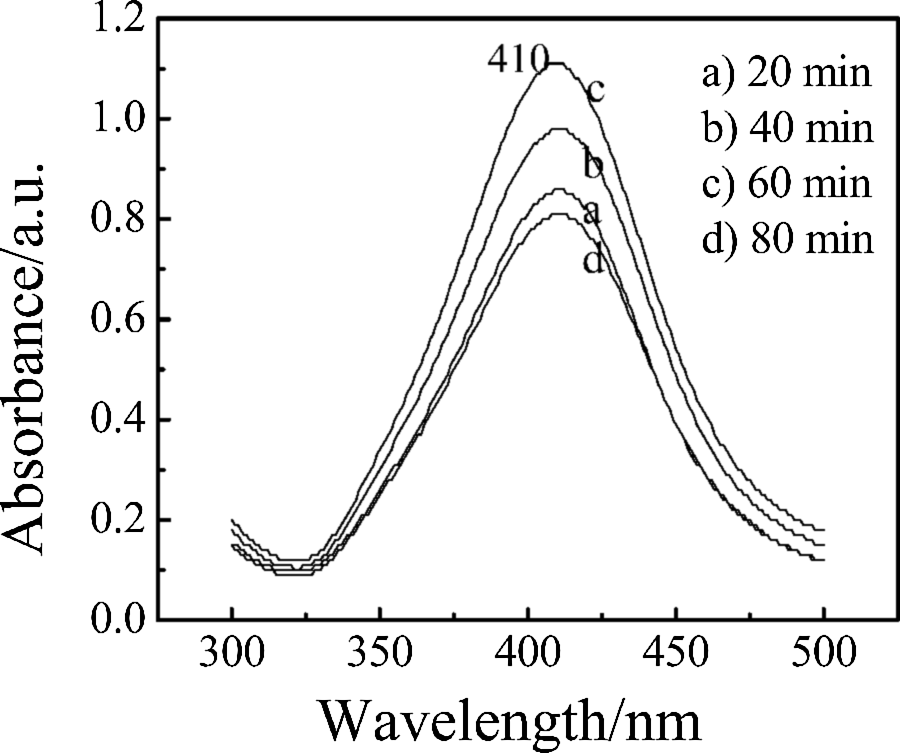
UV-Vis spectra of nano-Ag/PAMPS composites with different microwave time during reaction. UV–Vis: ultraviolet–visible; Ag: silver; PAMPS: poly(2-acrylamido-2-methylpropanesulfonate sodium).
The XRD pattern of nano-Ag/PAMPS composites illuminates that there are four peaks appeared at 2θ = 37.99°, 44.23°, 64.59°, and 77.52°, respectively, which indicates that there are nano-Ag crystal particulates within the nanocomposites. The crystal plane spacing (d) was 0.2367, 0.2046, 0.1442, and 0.1230 nm calculated by 2dsinθ = nλ, corresponding to the (111), (200), (220) and (311) of the crystalline Ag (PDFWIN#87-0597), which suggests that the AgNPs synthesized by microwave radiation have a face-centered structure.
Figure 3 shows the TEM image of nano-Ag/PAMPS composites. It can be seen that the AgNPs within polymer composites were synthesized with uniform size in the range of 10–20 nm. As shown in Figure 3(a), the synthesized samples were homogeneously dispersed in the polymer matrix. The AgNPs were of nearly spherical shape or spherical structure, which is consistent with the results of UV-Vis identification. The face-centered cubic polycrystalline structure was also proved by the electron diffraction analysis in Figure 3(b). According to the basic formula of electron diffraction (d = K/R) and electron microscopy constant K (20.08 mm nm−1), the corresponding crystal plane distances were calculated to be 0.237, 0.205, 0.145, and 0.123 nm, which is consentaneous well with d values of crystal plane distances of (111), (200), (220), and (311) (PDF card no. 87-0597) and consistent with the results of XRD identification. Further, high-resolution electron microscopy (HREM) can provide a deeper insight into the property of the formed particles. Figure 3(c) presents HREM image of the NPs. It can be seen more clearly that the NP is embedded in polymer matrix. Particularly, the lattice fringes reveal a good crystalline order. The spacing of 0.23 nm correspond to the (111) plane. The result further demonstrates that the NPs shown in Figure 3(a) are indeed the nanocrystalline of metallic Ag. It is well known that polymer, as protecting agents, are very effective to inhibit the agglomeration of particles. The in situ occurrence of monomer polymerization and metallic ion reduction should be more beneficial to the formation of well dispersed and narrow size-distributed nanopaticles.

TEM photograph of nano-Ag/PAMPS composites. (a) TEM image of nano-Ag/PAMPS composites, (b) electron diffraction photograph of AgNPs, and (c) HREM image of AgNPs. Ag: silver; PAMPS: poly(2-acrylamido-2-methylpropanesulfonate sodium); TEM: transmission electron microscopy; HREM: high-resolution electron microscopy; AgNP: silver nanoparticle.
To comprehend the interaction between AgNPs as dispersing phase and PAMPS polymer as continuous phase, the nano-Ag/PAMPS composites were examined by XPS. The survey spectrum of nano-Ag/PAMPS was shown in Figure 4(a). The carbon, oxygen, nitrogen, sulfur, and Ag elements were found (See Figure 4(a)), which are consistent with the chemical components of the nano-Ag/PAMPS composites.
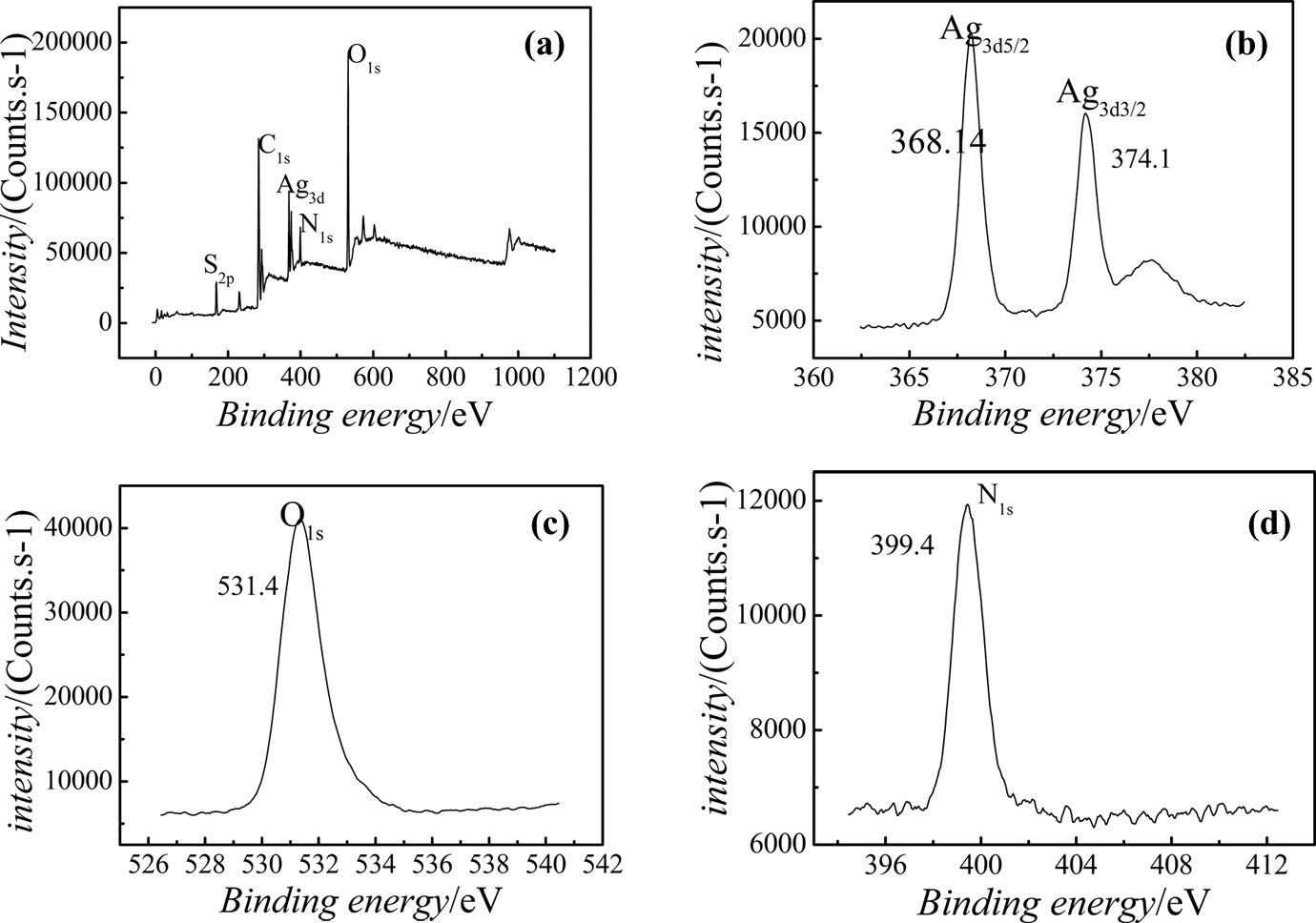
XPS spectra of nano-Ag/PAMPS composites. (a) Survey XPS of nano-Ag/PAMPS powder, (b) XPS of Ag3d, (c) XPS of O1s, and (d) XPS of N1s. XPS: x-ray photoelectron spectroscopy; Ag: silver; PAMPS: poly(2-acrylamido-2-methylpropanesulfonate sodium).
In Figure 4(b), the energy spectrum peaks of Ag3d3/2 electrons are positioned at 374.1 eV, which is higher than the standard energy spectrum peaks of Ag3d3/2 electrons that are positioned at 373.0 eV. The energy spectrum peaks of Ag3d3/2 electrons within nanocomposites are moved to the high binding energy, which indicate that the chemical environment of AgNPs was changed already; it was possibly due to the coordination interaction between certain atoms of polymer and AgNPs. The binding energy within the electronic shell was increased because of decreasing of valence electron density and the shielding effects of domestic electronic shell.
A better subdivision of the spectrum for O1s was observed in the XPS spectrum for the composites (see Figure 4(c)). The O1s binding energy (531.4 eV) of C=O was lower than the standard O1s binding energy (532.1 eV), that is, the O1s binding energy is moved to the lower region, which indicates that carbonyl oxygen atom accepting electronics cloud from other atoms. A better subdivision of the spectrum for N1s was observed in the XPS spectrum (see Figure 4(d)). The N1s binding energy (399.4 eV) was lower than the standard N1s binding energy (399.88 eV), that is, the N1s binding energy shift to lower region, which indicates that nitrogen atom accepted electronics cloud from other atoms, too.
Based on above discussion, it is suggested that the electron density of the nitrogen atoms and carbonyl oxygen atoms of PAMPS polymer was decreased, due to the transfer of electrons from Ag particles with the increase in binding energy. Therefore, the electron binding energy of the nitrogen atoms and oxygen atom shifts to a low binding energy level, as reported in the literature. 23 The specific interaction between nitrogen atoms and carbonyl oxygen atoms belonged to PAMPS polymer, and AgNPs within the nanocomposites could be confirmed by the shift of the N1s and carbonyl O1s peaks to lower binding energy.
To investigate the thermostability of the nanocoomposites, the TGA curves of the pure PAMPS and nano-Ag/PAMPS composites were measured (See Figure 5). It can be seen that the decomposition temperature of PAMPS in the nanocomposites was 297°C, which was lower than that of pure PAMPS (357°C). This is consistent with the result of Lee et al., 24 who showed that the thermal stability of the polymer was reduced due to the presence of AgNPs in their composites. It is possibly that AgNPs are homogeneously dispersed in polymer matrix to rapidly transmit heating effect and to promote decomposition of PAMPS polymer.
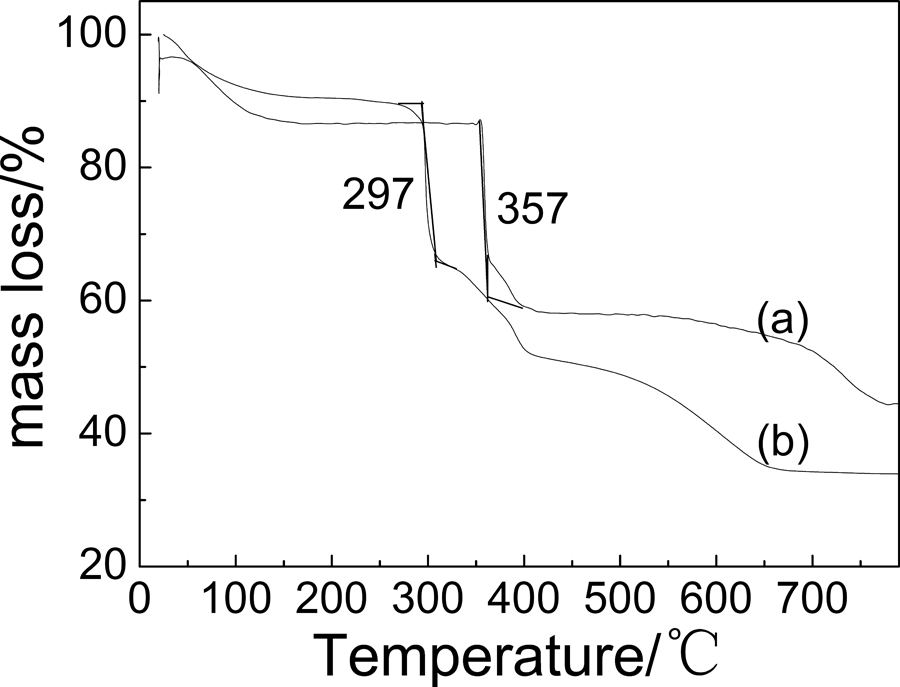
TGA curves of (a) PAMPS and (b) nano-Ag/PAMPS composites. Ag: silver; PAMPS: poly(2-acrylamido-2-methylpropanesulfonate sodium); TGA: thermogravimetric analysis.
Mechanism of Ag ion reduction with nitrogen-containing compound by microwave
To explain the forming mechanism of nano-Ag with no chemical reducing agent by microwave method, the catalytic reduction of Ag ion by N atom within PAMPS matrix is discussed. The color did not change, and the AgNPs was not found if only the reaction system was irradiated by microwave. However, when PAMPS was added in the AgNO3 solution, the color changed after microwave radiation to show the formation of the AgNPs from the reduction of Ag ion. So the mechanism of Ag ion reduced to nano-Ag under the effect of PAMPS may be given as follows:
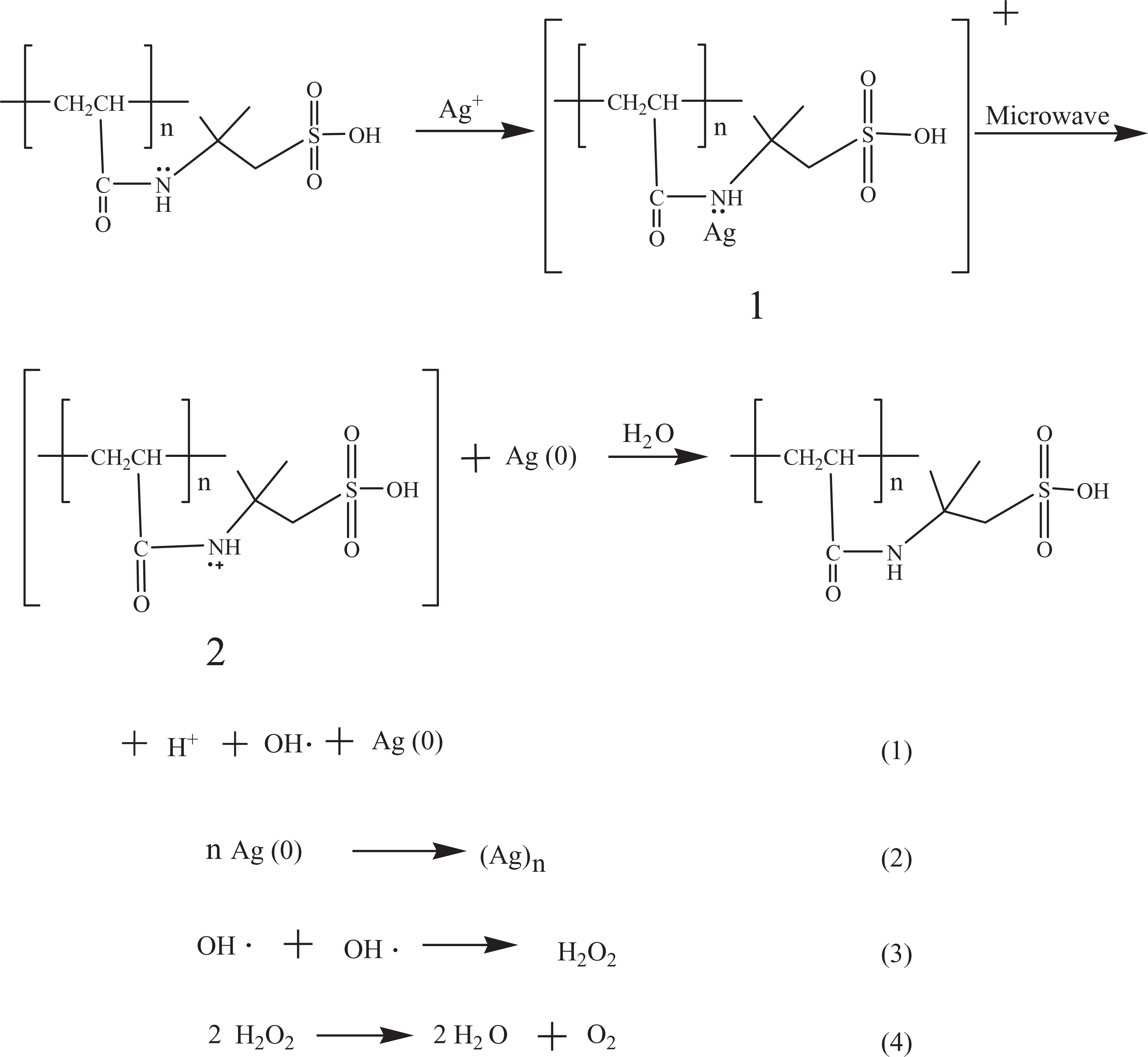
The pairs of electron within the nitrogen atom attached to sec-amine of PAMPS molecular can coordinate with Ag+ to form the structure
Conclusion
The nano-Ag/PAMPS composites were prepared in situ by an in situ microwave synthesis method. The UV-Vis spectrum revealed that the morphology of AgNPs was not affected by microwave treatment. TEM displayed that AgNPs were homogeneously dispersed in polymer matrix. XPS indicated that there was chemical action between AgNPs and nitrogen atoms and carbonyl oxygen atoms from PAMPS polymer. TGA proved that the thermal stability of the nano-Ag/PAMPS composites was lower than that of the pure PAMPS. The mechanism of catalytic reduction of Ag ion by N atom from PAMPS has also been proposed.
Footnotes
Funding
This work was financially supported by the Natural Science Foundation of China (Grant No. 51173002), the Natural Science Foundation of Anhui Province, China (Grant No. 090414183), and the doctor project of Anhui University of Science and Technology.
