Abstract
The surface of micro and nanoparticles of calcium carbonate (CaCO3) was modified by 3-mercaptopropyl trimethoxysilane and polymethylhydrogensiloxane. Fourier transform infrared spectra of extracted CaCO3 particles revealed interfacial bonding between coupling agents and CaCO3 particles. Poly(vinyl chloride) (PVC)/surface-modified CaCO3 with different mass ratios of (micro/nano)-CaCO3 particles were melt blended using a Brabender torque rheometer and kept with a constant content of CaCO3. The impact strength, tensile strength, elongation at break, and Young’s modulus of the composites were considerably enhanced at 9/6 mass ratio of (micro/nano)-CaCO3. The mechanical properties were improved relative to the pure PVC due to the interfacial bonding between the filler and the matrix. Scanning electron microscopic images of fractured surfaces of the test specimens showed larger voids with nonsmooth edge around the cavities, as compared with the nano-CaCO3/PVC composite, which is more pronounced by the increase in the micro particles contents.
Introduction
Owing to high mechanical strength, high corrosion resistance, and relatively low cost poly(vinyl chloride) (PVC) is one of the most commonly used plastic materials in the world such as in door and window profiles, electrical insulation, medical tubes, food wraps, outdoor furniture, swimming pool liners, electrical conduit, pressure pipes, and so on. Since PVC has weak impact resistance and poor heat stability, various materials, such as elastomers and inorganic particles, are being used to toughen the polymer. Although rubbery polymers have been widely used for toughening polymers, their incorporation increases cost and decreases strength, heat stability, and modulus. 1 –5
Nanosized calcium carbonate (CaCO3) has become one of the most common fillers used to prepare nanocomposites. 6 –12 This filler has particularly attracted attention in PVC composites because of its ability to eliminate hydrogen chloride gas during thermal degradation of PVC and improve its mechanical properties. 11 However, homogeneous dispersion of CaCO3 nanoparticles in PVC matrix is a challenge in preparing PVC/CaCO3 composites, which is mainly due to the strong tendency of CaCO3 nanoparticles toward agglomeration. 4 It has been reported that surface treatment of fillers through proper coupling agents not only promotes the dispersion of filler into the matrix but also enhances the interfacial bonding of polymer–filler. 13 –15
Mechanical properties of PVC/nano-CaCO3 and PVC/micro-CaCO3 composites have been widely studied. 4 –10,16 –18 In these studies, the CaCO3 particles are modified with fatty acid, titanate coupling agent, and sodium stearate. Tensile strength, yield strength, and elongation at break of both PVC/micro-CaCO3 composites (5–15 phr CaCO3), and PVC/nano-CaCO3 composites (5–20 phr CaCO3) have been improved, as compared with the pure PVC. 9 Another study reports that the addition of 5–25wt% nano- or micro-CaCO3 into the PVC matrix improves impact strength and decreases the tensile strength. 4 The content of nano-CaCO3 particles for maximum impact strength is reported to be about 15 phr for PVC/ nano-CaCO3 composites. 4,16 It is shown that the impact strength of PVC composites at room temperature is increased by 28%, using a mixture of 2.5and 9 phr micro-CaCO3 and nano-CaCO3, respectively. 19
The aim of the present article is to investigate the effects of surface treatment of CaCO3 on mechanical properties (impact strength, tensile strength, elongation at break, and Young’s modulus) of PVC composites filled with various mass ratios of treated micro- and nano-CaCO3 particles. Nano-CaCO3 particles may replace micro-CaCO3 in the composite in a less extent without sacrificing the mechanical properties. Two different methods for the addition of nano-CaCO3 were studied during the melt blending of PVC with treated and untreated nano-CaCO3 to find the effects on particle dispersion and mechanical properties of composites. 20 The addition method of nano-CaCO3 included (1) directly mixing of PVC and nano-CaCO3 and (2) addition of nano-CaCO3 at the onset of PVC fusion. Our results showed that direct mixing of PVC and nano-CaCO3 improved dispersion of nanoparticles and mechanical properties of composites.
Experimental
Materials
Suspension grade PVC with K value 65–67 was obtained from China General pPastic Corporation (CGPC) (Taiwan). Additives such as organic tin stabilizer, processing aid, calcium stearate, and polyethylene wax were supplied by Baerlocher Co., Germany. Two types of untreated CaCO3 (nano and micro) were supplied by Solvay Advanced Functional Minerals Co., Germany. The mean particle size of nano-CaCO3 (Socal 31) and micro-CaCO3 (P3) particles were 62 and 182 nm, respectively. Silane coupling agents consisting of polymethylhydrogen siloxane (PMHS) and 3-mercaptopropyl trimethoxysilane (MPTS) were provided by Merck Co., Germany. Formulation of the PVC used for the preparation of the compounds is given in Table 1.
PVC formulation.

PVC: poly(vinyl chloride); CaCO3: calcium carbonate; PE: polyethylene.
aParts per hundred by weight.
Surface treatment of CaCO3 particles
Nano- and micro-CaCO3 particles were surface treated separately with MPTS and PMHS coupling agents, using a procedure which was reported by Bergstrom et al. 21 Nano-CaCO3 (30 g) has been dried at 100°C for 24 h in vacuum oven. Treating agent MPTS (0.78 g) was added dropwise to dry nano-CaCO3 under vigorous agitations with a home mixer (Mulinex) for 15 minutes. Then, 0.42 g PMHS was added dropwise for 15 minutes and agitation was continued for an additional period of 30 minutes. This blend was placed into the oven at 150°C overnight to ensure complete reaction. Micro-CaCO3 was also modified using a similar procedure with 0.39 g and 0.21 g of MPTS and PMHS, respectively. Consequently, monolayer coverage on nano- and micro-CaCO3 particles was produced. 22,23 Figure 1 shows hydrophobicity and hydrophilicity of treated and untreated CaCO3 particles, respectively. Untreated CaCO3, being hydrophilic, is dispersed in water with agitation, while treated CaCO3 is hydrophobic and forms a foamy layer on the water surface even with vigorous agitation.

Hydrophilicity and hydrophobicity of treated and untreated calcium carbonate.
To investigate the chemical bond formation between the coupling agent and the surface of modified CaCO3, treated particles were solvent (tetrahydrofuran; THF) extracted using a Soxhlet apparatus. For this purpose, 0.5 g of modified CaCO3 was placed in the extraction thimble of the apparatus and distilled with THF for 12 h under nitrogen atmosphere and then dried in vacuum oven for 24 h at room temperature (25°C).
Sample preparation
Different mass ratios of surface-treated (micro/nano)-CaCO3 (12/3, 9/6, 6/9, and 3/12 (g/g)) were tumbled and mixed in a glass container for 1 h with hand to prepare a mixture for each ratio. Table 2 shows different coded samples prepared by tumble mixing of required ratios of treated nano and micro particles with PVC formulation ingredients (Table 1) for 5 min. The mixed compounds were processed using the Brabender plastograph EC, which was equipped with an electrically heated mixing head (W 50 EHT mixer) having 50 cc volume. The processing temperature, rotor speed, and blending time were set at 170°C, 60r min−1, and 5 min, respectively. The weight of the sample charged into the Brabender plastograph was kept constant at 60 g. The compounded lumps from Brabender have been compression molded using the test molds at 180°C under 100 bar pressure for 5 min to obtain tensile and impact specimens.
Compositions of PVC/(micro/nano)-CaCO3.
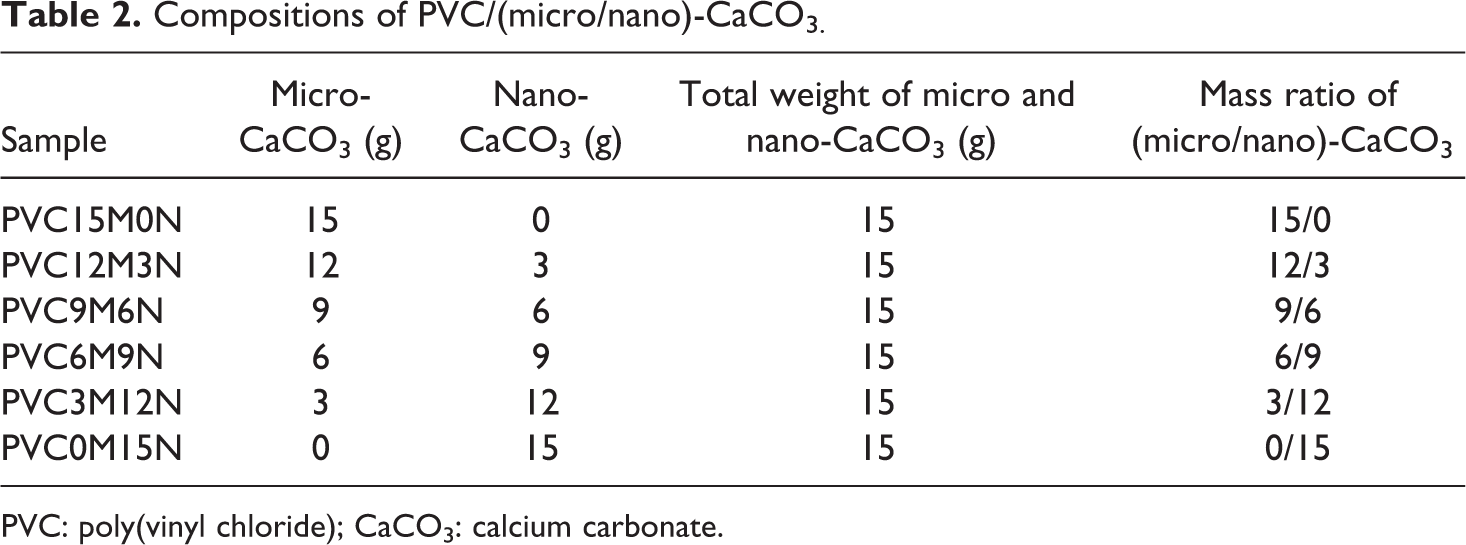
PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
Parameter of B.
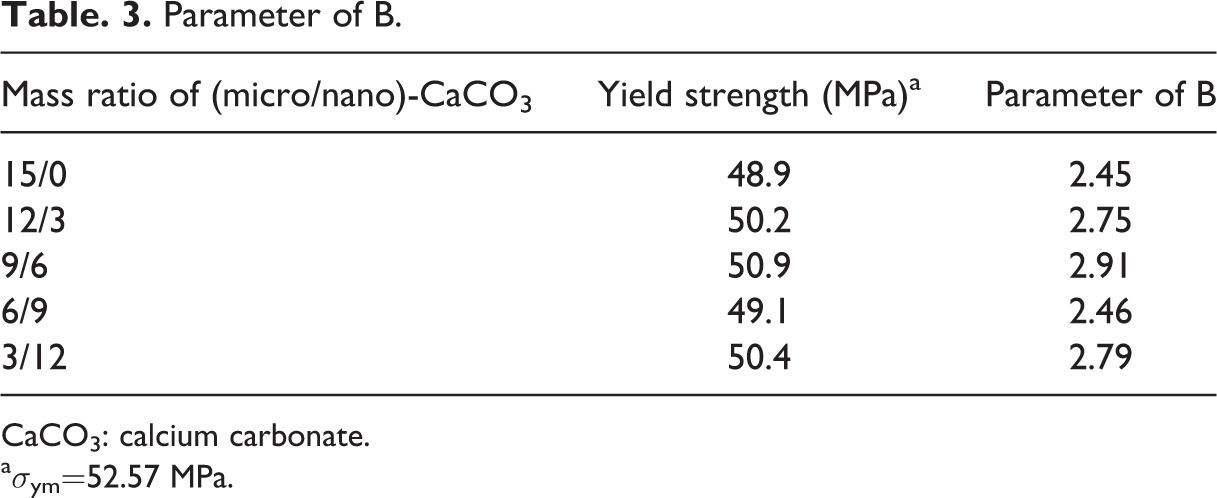
CaCO3: calcium carbonate.
a σ ym=52.57 MPa.
Measurements
FTIR spectroscopy
The Fourier transform infrared spectrophotometer (FTIR) analysis was performed on a spectrophotometer (Bruker-Tensor 27, Billerica, MA, USA) at room temperature. FTIR was used to characterize the functional groups of the treated and untreated CaCO3, extracted treated CaCO3, and coupling agents. The spectra were recorded in the range of wave numbers between 600 and 3300 cm−1.
Scanning electron microscopy
The morphology of the fractured surface in tensile testing and fractured surface in notched Izod impact test was studied using scanning electron microscope (SEM-X60, Tokyo, Japan). The fractured specimen surfaces were coated with a thin layer of gold prior to SEM examination. The coating of fractured surfaces was performed by putting the specimen in a high vacuum evaporator and vaporizing the metal held in a heating tungsten basket.
Mechanical properties
Tensile properties were measured using a tensile testing machine (Hounsfield Co., England) according to ISO 527 standard method. The test specimens were tested at a strain rate of 10 mm min−1 at room temperature (20°C). Notched Izod impact test was performed using an impact tester (Zwick/Roell, Germany) according to ISO 180, with impact energy of 2.75 J. Each datum of the tensile and impact tests was the average of six repeated test values. Relative mechanical properties (ultimate tensile strength, Young’s modulus, and elongation at break) were calculated using the ratio of each mechanical property of the composite sample to the mechanical property of pure PVC.
Results and discussion
FTIR analysis
Figure 2(a) to (e) shows FTIR spectra of pure CaCO3 particles, treated CaCO3 particles, extracted CaCO3 particles, MPTS, and PMHS, respectively. In Figure 2(a), the IR absorption at 1411.8, 875.6, and 757.9 cm−1 is assigned to Ca–O stretching and bending vibration, respectively. 24,25 The FTIR of coupling agents corresponding to MPTS and PMHS are presented in Figure 2(d) and (e), respectively. In Figure 2(d), the peaks at 2941.3, 1257.5 and 802.3 cm−1 were assigned to Si–CH3, and 2565.2 cm−1 was attributed to S–H stretching vibration. 26,27 The treated CaCO3 particles in Figure 2(b)shows the shifting of the characteristic peaks of CaCO3 from 1411.8, 875.6, and 757.9 cm−1 to 1407.9, 877.7, and 750.2 cm−1, respectively and the shoulder peak at 1448.4 cm−1 is disappeared. This is due to the interaction between coupling agents and the surface of CaCO3. Also, the appearance of the bands at 1041.5 and 1126.3 cm−1 is indicative of the formation of Si–O–CaCO3 bond on CaCO3 particles. 28,29 This is due to the formation of chemical bonds between the surface of CaCO3 and the coupling agent, MPTS. The physically absorbed coupling agent on the surface of CaCO3 had been removed by extraction, as its FTIR absorption spectra are shown in Figure 2(c). It is observed that the FTIR spectra of unextracted and extracted CaCO3 particles are the same. This indicates the formation of the chemical bond between coupling agents and the surface of CaCO3 particles.
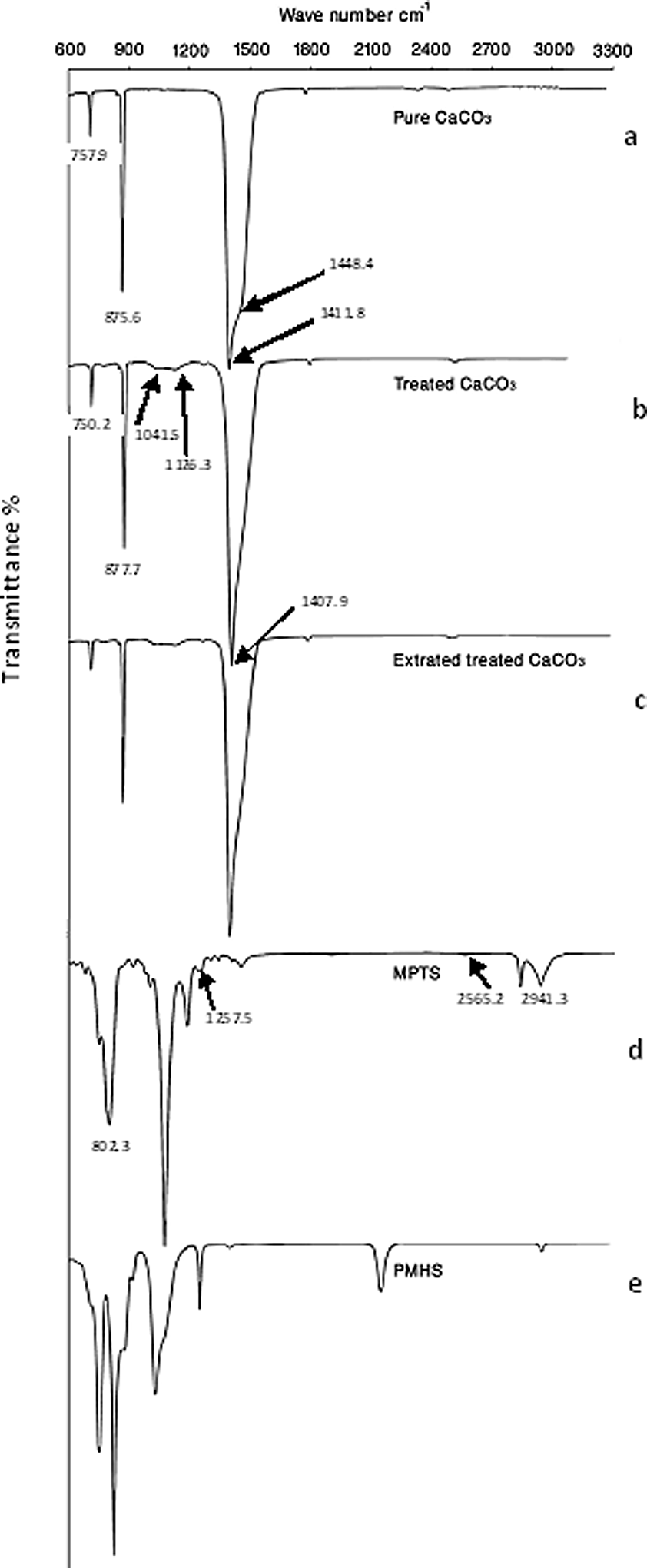
FTIR spectra (a) Pure CaCO3, (b) treated CaCO3, (c) extracted treated CaCO3, (d) MPTS, and (e) PMHS. FTIR: fourier transform infrared; CaCO3: calcium carbonate; MPTS: 3-mercaptopropyl trimethoxysilane; PMHS: polymethylhydrogen siloxane.
Mechanical properties
Figures 3 to 6 show variations in impact strength, tensile strength, elongation at break and Young’s modulus of the composites containing different mass ratios of (micro/nano)-CaCO3, respectively. Mechanical properties of base poly (vinyl chloride) and relative mechanical properties of PVC nano-composite are reported in tables 4 and 5, respectively. Figure 3 shows the effect of various ratios of (micro/nano)-CaCO3 on the relative impact strength of the samples. By increasing the content of nano-CaCO3, the relative impact strength reaches a maximum (i.e., sample PVC9M6N) and then decreases. In this study, since micro and nano-CaCO3 are used to prepare the composites, different mechanisms are expected to influence the mechanical properties of these composites. The impact performance of the PVC filled with micro-sized CaCO3 particles could be described by crack bowing mechanism. 8 According to this mechanism, the rigid particles resist in the path of a propagating and growth crack. Therefore, the crack tends to bow between the neighboring particles. In the case of introducing nanoparticles into the polymer matrix, the stress concentrator mechanism could be used to explain toughening of polymer. The nanoparticles could act as stress concentration points and promote cavitation at the particles–polymer matrix boundaries during loading. 8,10 Since nano- and micro-CaCO3 particles are present into the PVC matrix, the contribution of both mechanisms (crack bowing and stress concentrator) leads to improving the impact strength of the composites. There may be the synergic effect of the two particles types on impact strength, especially when the mass ratio of (micro/nano)-CaCO3 is 9/6. Figure 4 shows the variation of elongation at break of the composites versus the mass ratio of (micro/nano)-CaCO3. The elongation at break shows again a maximum at 9/6 mass ratio of (micro/nano)-particles. It is seen that the elongation at break for all the samples is significantly higher than that of PVC without filler, showing similar variations to impact strength. This improvement in elongation at break is related to the formation of micropores and cavities to absorb the applied stress during longitudinal extension. 9,30 In this study, in spite of the lack of any impact modifier in the composites, elongation at break is improved for all the samples. On the contrary, the other authors used some impact modifiers such as methacrylate–butadiene–styrene (MBS = 10 phr) in their composites. 9,30 A gradual decrease in elongation at break is observed, when the particles with mass ratios more than 9/6 is used in the PVC matrix.. The tendency of the nanoparticles to aggregate in the polymer matrix may cause such a reduction in elongation. 30 Figure 5 shows variation of relative ultimate tensile strength (UTS) of the composites versus mass ratio of (micro/nano)-CaCO3 particles. An increase in the UTS is obtained with a decrease in this ratio up to 9/6. It is interesting to note that UTS reaches a maximum at such ratio, followed by a gradual decrease with more reduction in the ratio. It is worth mentioning that all the measured tensile strengths in this study are higher than that of the pure PVC. This improved tensile strength is attributed to the enhanced interfacial interaction between the filler particles and the PVC matrix. However, other researchers reported that the tensile strength of both surface treated PVC/nano-CaCO3 and PVC/micro-CaCO3 composites was significantly lower than that of the pure PVC. 4,31 Figure 6 shows an increase in Young’s modulus of PVC/(micro/nano)-CaCO3 composites with decreasing mass ratio up to 9/6. Further decrease in the mass ratio decreases the Young’s modulus. It is reported that the effect of surface treatment of particulate filler on Young’s modulus in the literature is conflicting. 13 Interestingly, it can also be seen that Young’s modulus of PVC composites is higher than that of PVC without filler. This is due to the presence of the rigid filler into the PVC matrix that restricts the mobility and deformability of the matrix. 32
Mechanical properties of poly(vinyl chloride) without filler.
Relative mechanical properties of PVC/(0 g micro/15 g nano)-CaCO3.

PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
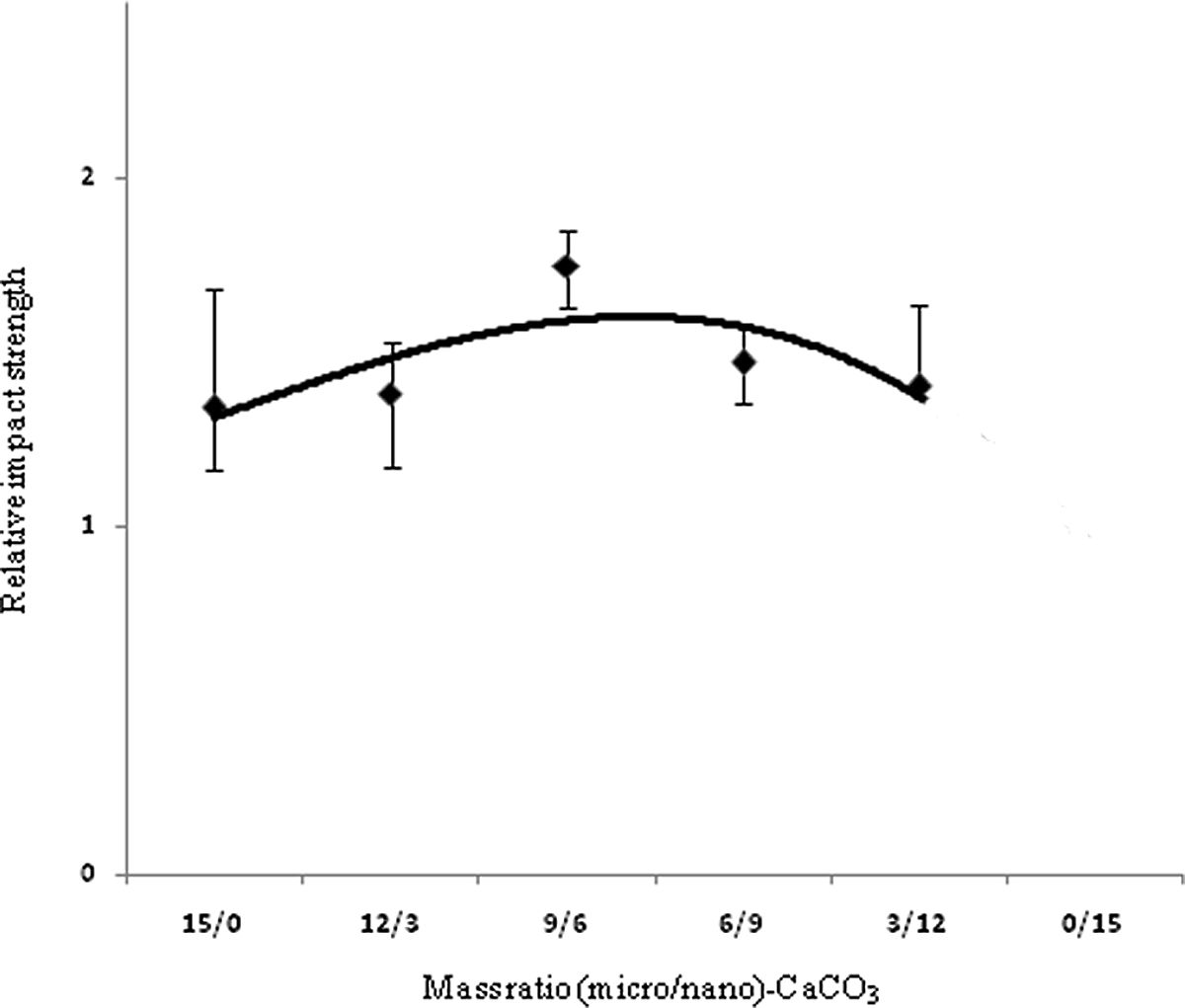
Relative impact strength of PVC composites versus mass ratio of (micro/nano)-CaCO3 particles. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
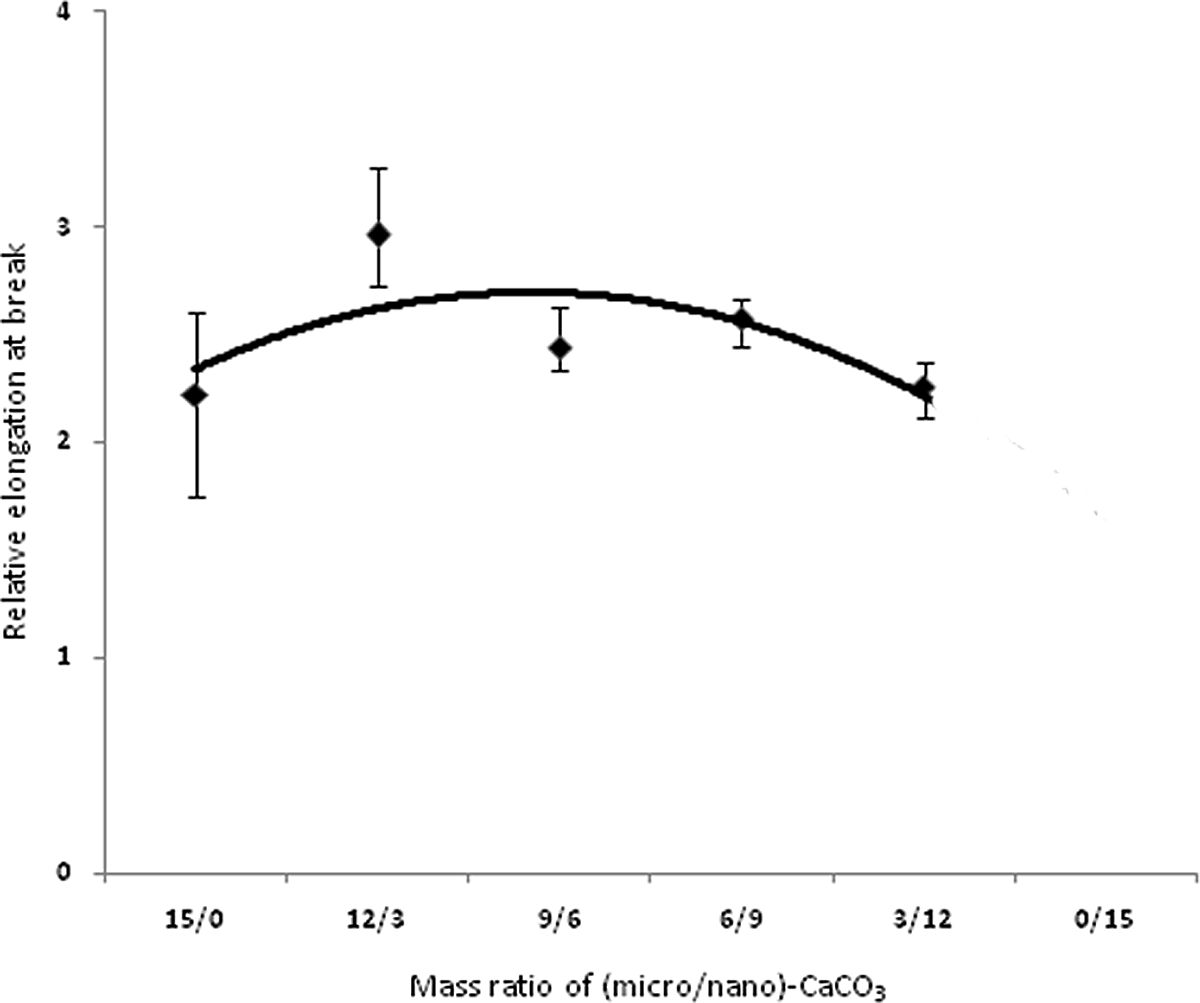
Relative elongation at break of PVC compositesversus mass ratio of (micro/nano)-CaCO3 particles. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
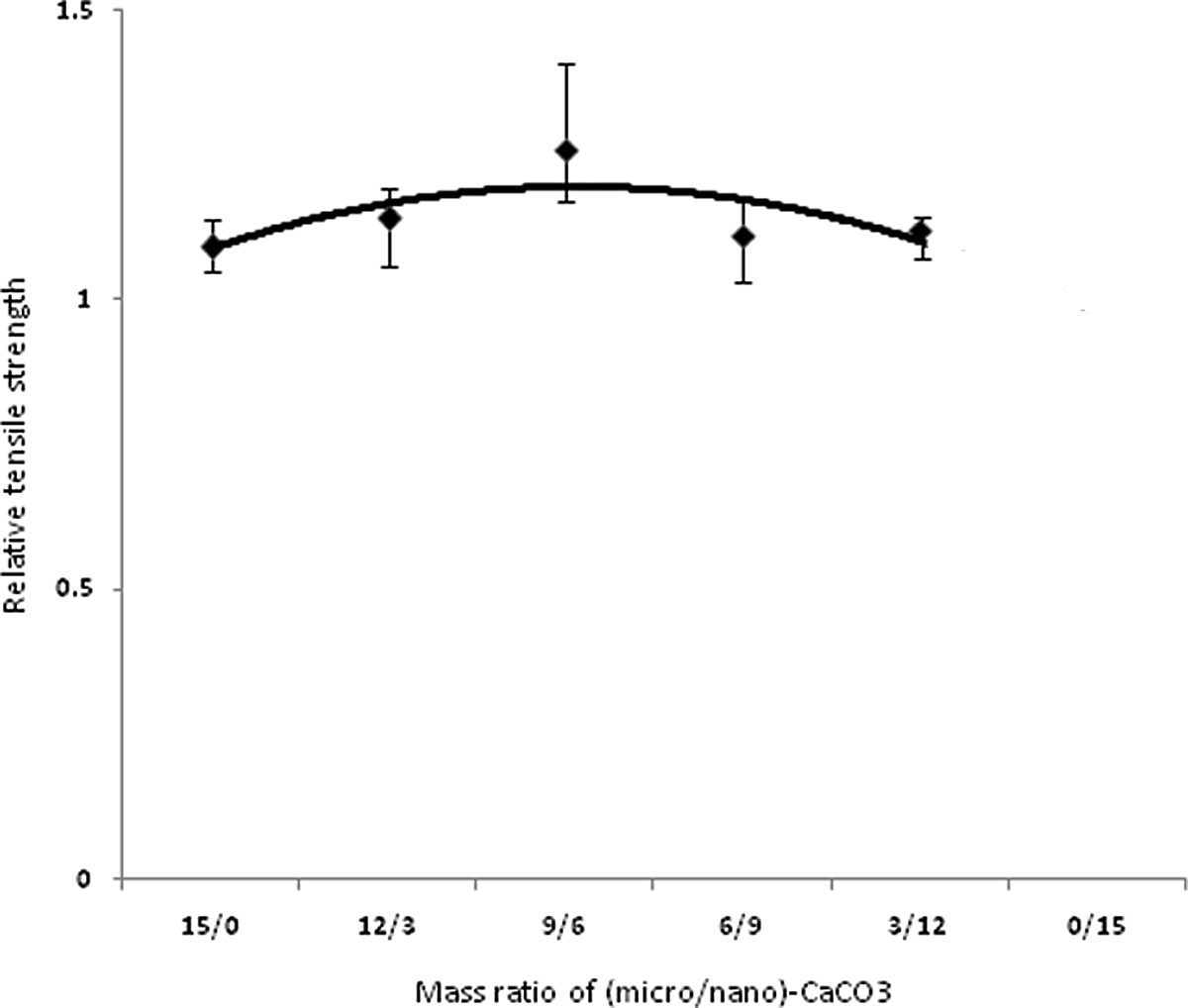
Relative tensile strength of PVC composites versus mass ratio of (micro/nano)-CaCO3 particles. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
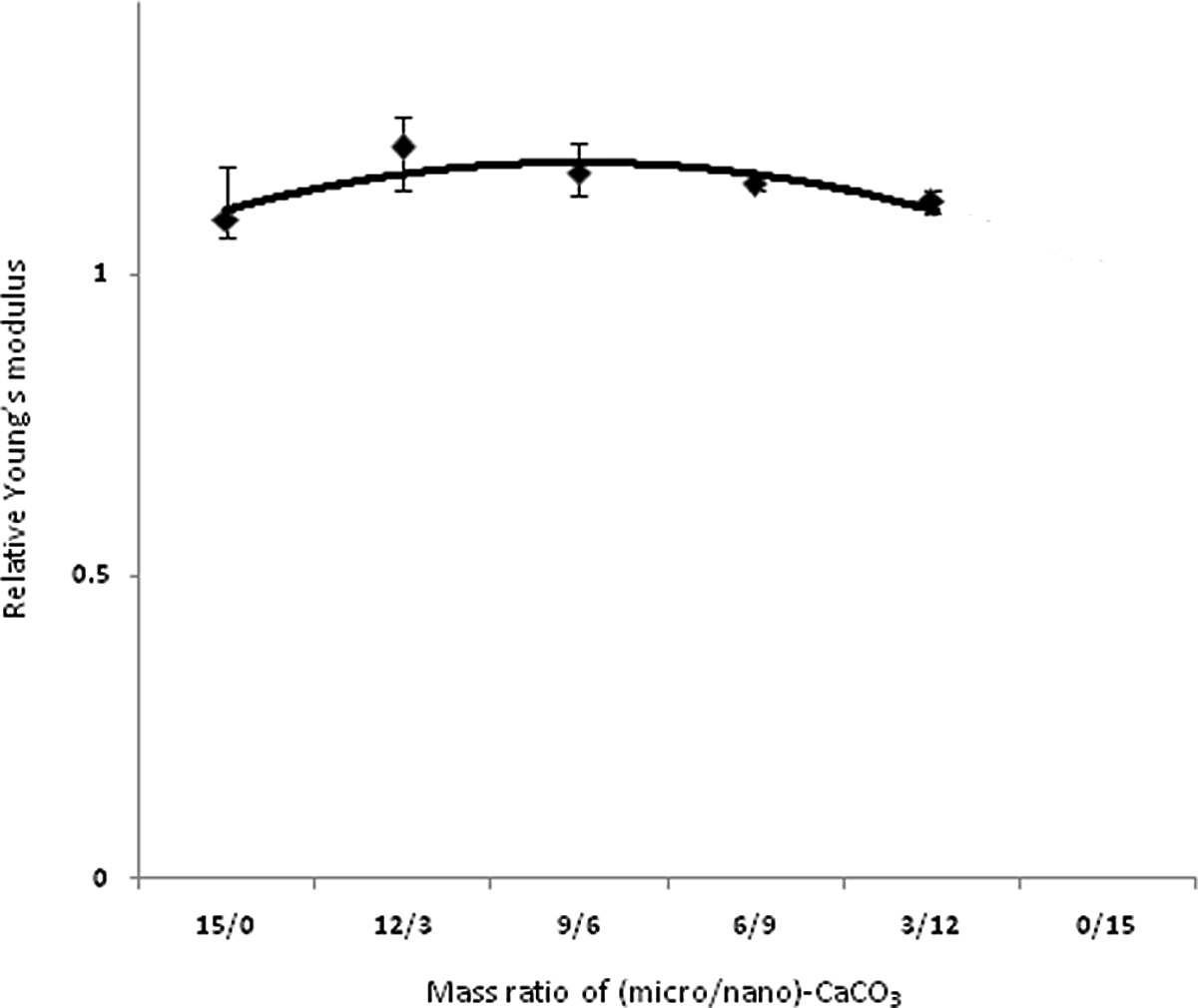
Relative Young’s modulus of PVC composites versus mass ratio of (micro/nano)-CaCO3 particles. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
Turcsanyi et al. proposed a model (equation (1))to describe the effect of interfacial interaction on the yield strength of particulate-filled composites.
33,34
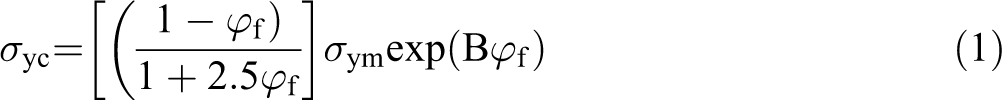
where σ yc, σ ym, and φ f are yield strength of the composite, yield strength of the matrix, and the volume fraction of the filler, respectively. B is the interfacial interaction parameter between the particles and the polymer. A higher value of B corresponds to stronger interfacial interaction between particle and polymer. 33,34 In the present work, the parameter B was calculated using equation (1) and the data is shown in Table 3. Parameter B acquires the greatest value for the composites prepared at 9/6 mass ratio of micro/nano-CaCO3 particles, confirming that the highest mechanical properties are expected from this composition.
CaCO3 particles could act as stress concentrator and nucleation sites, which could promote cavitation at the particles–PVC matrix boundaries during loading. These cavities lead to plastic deformation and an increase in the size of the crack-tip process zone; thus, the work required for crack propagation results in the improvement of impact strength. 8,10 It is believed that maximum cavities created at 9/6 ratio provide the maximum energy dissipation resulting in the highest impact strength.
Fracture surface observations using SEM analysis
Fracture surface micrographs of PVC/(micro/nano)-CaCO3 composites and pure PVC are shown in Figure 7(a) to (g). It can be seen that the addition of (micro/nano)-CaCO3 particles alters the fracture behavior relative to that of PVC without filler significantly. Figure 7(a), showing the image of pure PVC, illustrates a relatively smooth texture containing a lot of cavities that lead to a brittle failure. 24 The effect of various ratios of (micro/nano)-CaCO3 particles on fracture surface of the samples is shown in Figure 7(b) to (g). It can be clearly seen that the (micro/nano)-CaCO3 particles are debonded from the PVC matrix and voids are formed. The formation of voids around the particles, being a result of their stress concentration effect, causes plastic deformation. Figure 7(b) and (c) belong to the PVC0M15N and PVC3M12N samples, respectively. Comparison of Figure 7(b) with Figure 7(c) reveals that the presence of micro-CaCO3 particles resulted in larger voids and agglomeration relative to the nanoparticles in the samples (as shown by the arrows in Figure 7(b) and (c)). As more micro-CaCO3 is introduced into the composites, the fractured surfaces become rougher (Figure 7(e) to (g)). It can also be seen that the edge of the cavities becomes rough and non-smooth with increasing the content of micro particles in the composites (as shown by the arrow in Figure 7(b) and (g). This may also be due to the effect of size and number of the particles. These particles themselves behave like stress concentrator points. As the content of micro- CaCO3 is increased in the composites, the size of particles becomes larger and the number of particles decreases in the composites. Therefore, the number of the stress concentration points decreases and the value of concentrated stress at these points increases. As a result, debonding around a big particle may happen easily, relative to small particle and increasing energy dissipation. Subsequently, plastic deformation is developed for craze propagation. However, in the case of the increase in nano-CaCO3 particles, the number of particles is relatively higher and the applied stress is distributed between larger numbers of particles. Thus, the value of stress concentration on each particle is relatively lower, making a smaller driving force for initiation and propagation of crack. Sahebian et al. concluded that the nanoparticles tend to decrease their surface contact with matrix through agglomeration. These agglomerated particles decrease the stress concentrator points and so increase the value of stress concentration. 35

Scanning electron microscopic images of the impact strength fracture surface specimens of pure PVC. (a) PVC with various mass ratios (micro/nano)-CaCO3, (b) PVC0M15N0/15, (c) PVC3M12N, (d) PVC6M9N, (e) PVC9M6N, (f) PVC12M3N, and (g) PVC15M0N. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
Figure 8(b) to (g) shows SEM images of fractured surfaces (from tensile tests) of the composites containing various mass ratios of (micro/nano)-CaCO3. The micrograph of PVC sample without filler shows a fibrous feature in Figure 8(a). Cavities, fibrous arrangement, and fibrous shape are clearly seen in Figure 8(b) to (g). These fibrous structures at break and cavities could partially act as absorber of tensile force. 29 The fracture surface of PVC9M6N sample contains longer and thinner fibrils as compared to the other samples (Figure 8(e)). Longer fibrils are due to the strong bond and interfacial interaction between PVC matrix and treated CaCO3 particles. Figure 8(f) and (g) show the break surface of composites for PVC12M3N and PVC15M0N samples, respectively. Shorter fibrils can be observed, as compared with Figure 8(e), confirming the lower tensile strength. The large number of agglomerates can be seen in the break surface of the composite for sample of PVC15M0N in Figure 8(g), resulting in lower mechanical properties.
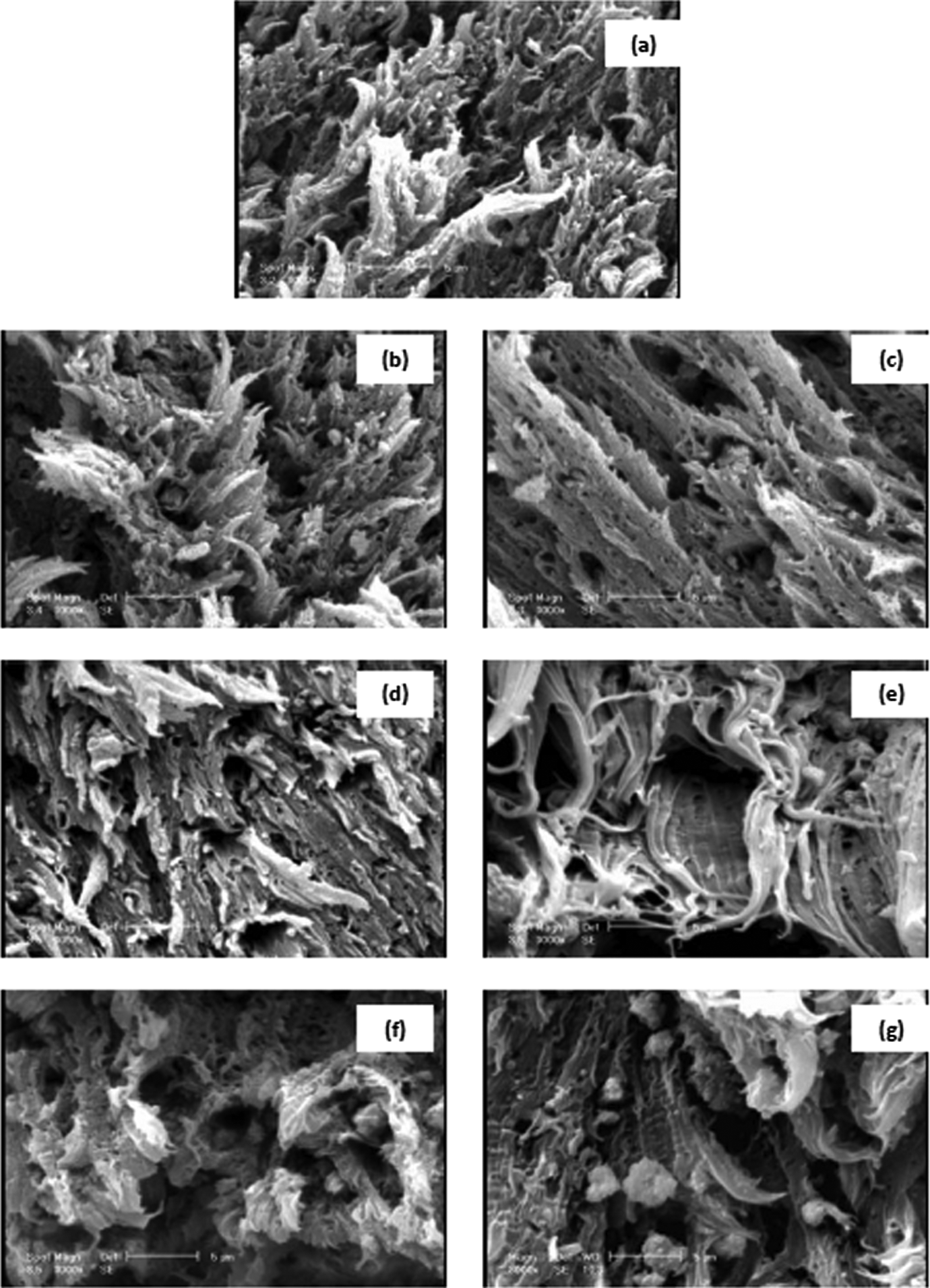
Scanning electron microscopic images of the tensile strength fracture surface specimens of pure PVC. (a) PVC with various mass ratios (micro/nano)-CaCO3, (b) PVC0M15N, (c) PVC3M12N, (d) PVC6M9N, (e) PVC9M6N, (f) PVC12M3N, and (g) PVC15M0N. PVC: poly(vinyl chloride); CaCO3: calcium carbonate.
The differences between SEM micrographs of impact strength tester fractures and tensile strength fractured specimens are due to the type of testing. The uniaxial tensile test at low strain rate causes the polymer chains and segments to be oriented and extended in the form of fibrils. In the impact test, very complex stress in a short time acts on the specimen and the orientation of the polymer chains and segments does not occur.
Conclusions
Micro- and nano-CaCO3 particles were surface treated with MPTS and PMHS. FTIR spectra of treated CaCO3 particles revealed interfacial bonding between coupling agent and the surface of particles. The characteristic peaks of CaCO3 including 1411.8, 875.6, and 757.9 cm−1 are shifted to 1407.9, 877.7, and 750.2 cm−1, respectively. PVC composites filled with different mass ratios of (micro/nano)-CaCO3 particles were prepared via a melt blending method with Brabender torque rheometer. The maximum impact strength, elongation at break, tensile strength, and Young’s modulus were obtained at 9/6 mass ratio of (micro/nano)-CaCO3 in the PVC matrix. These results show the considerable reinforcement of the PVC with the addition of various ratios of (micro/nano)-CaCO3.
The fracture surfaces of impact strength testing revealed that with increasing micro-CaCO3 particles, the surface was shifted from smooth to rough and larger cavities were formed at the fractured surface. The fracture surfaces of tensile strength revealed that fibrils that appeared on the fracture surfaces and fibrils of composites for the sample containing 9/6 mass ratio particles (PVC9M6N) were longer and thinner than those of the other mass ratios of (micro/nano)-CaCO3. The replacement of a portion of the micro-CaCO3 particles with nanosized particles significantly improves mechanical properties. We have shown that the applications of the prepared composites in this work are preferable and favorable when compared with the pure PVC. It is concluded that interfacial interactions between PVC and (micro/nano)-CaCO3 are improved, resulting in better mechanical properties, as compared with the pure PVC.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
