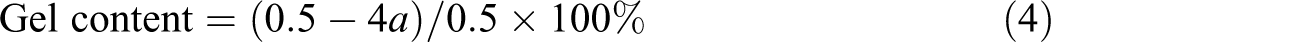Abstract
Monomer casting (MC) nylon-6 was a polymeric modified by grafting 4-amion-2,2,6,6-tetramentylniperidine (TEMP) as reactive-hindered amine onto its chain so as to improve the compatibility and stability of the stabilizer in the polymer matrix. The structure of the reactive stabilizer was confirmed by Fourier transform infrared and proton nuclear magnetic resonance spectroscopic analyses. The effect of TEMP on the thermal stability of MC nylon-6 was studied. It was found that with the increase in TEMP dosage, the thermal weight loss of the samples first decreased and then increased and the preferred dosage of TEMP was about 0.2 mol%. The residual weight percentage (R) of all the samples decreased with heating time, and it does much more slowly for the grafting sample than that of pure one. The thermal degradation characteristic temperatures of MC nylon-6 were improved by grafting TEMP. The long-term aging of MC nylon-6 indicated that the carboxylic acid concentration of the grafting sample was lower than that of the pure sample, and its end amine group concentration was relatively higher during the whole aging process, presenting more stable chemical structure. Ultraviolet absorption and gel content of MC nylon-6 were also reduced by grafting TEMP. The crystallinity (Xc) of MC nylon-6 increased, and the crystallization growth rate and melt temperature (Tm) decreased with aging time, resulting from the annealing process at high temperature and the small molecular products during aging.
Keywords
Introduction
Polyamide 6 (PA6) has a wide range of applications in industry due to its excellent mechanical properties. As one of the most common engineering plastics and fibers, the stabilization and the aging behavior of PA6 have been concerned by people for a long time. Usually, the carbon–hydrogen bond on the methylene group adjacent to nitrogen is considered to be the weakest bond, 1 with an oxidation reaction proceeding on this carbon when subjected to heat, oxygen, light and water, resulting in a decrease in molecular weight, subsequent deterioration in physical and mechanical properties, increased yellowing and eventually embrittlement of PA.2–8 Besides, the observed degradation products implied that all the methylene may be attacked at random by free radicals. 9
Hindered amines are the derivatives of 2,2,6,6-tetramethylpiperidine and are extremely efficient thermal photostabilizers by absorbing oxygen radicals. 10 For the thermal stabilization system of PA, low-molecular-weight compounds such as 4-amion-2,2,6,6-tetramentylniperidine (TEMP) and 4-hydroxy-2,2,6,6-tetramethylpiperidine are often used as the main stabilizers in the present industry due to their high thermostabilizing efficiency. But they are easy to volatilize during high-temperature melting process resulting in the loss of compounds, reduction in the thermo light-stabilizing efficiency and formation of mold deposit.
Monomer casting (MC) nylon-6 prepared by alkali-catalyzed anionic ring-opening polymerization of caprolactam (CL) is one of the commercially prominent members of polyamides, which takes advantages of high molecular weight, high mechanical strength and excellent self-lubricating performance. 11 –14 In this work, MC nylon-6 was a polymeric modified by grafting TEMP as reactive hindered amine onto its chain so as to improve the compatibility and stability of the stabilizer in the polymer matrix. The thermostabilization effect of the TEMP on MC nylon-6 was studied.
Experimental
Materials
The CL monomer was supplied by China Petrolleum and Chemical Co., Ltd with a commercial grade product. Sodium hydroxide (NaOH), as the main catalyst with analytical purity, was purchased from Kermel Chemical Reagent Co., Ltd (Tianjin, China), and toluene-2, 4-diisocyanate (TDI), as the auxiliary catalyst, was purchased from Kelong Chemical Reagent Factory (Chengdu, China). TEMP was purchased from TCI Development Co., Ltd (Shanghai, China). Other processing aids and chemical agents are all commercial grade products and used without further purification.
Material preparation
The pure and grafting MC nylon-6 was fabricated by means of cast molding technique via the mechanism of anionic ring-opening polymerization. 15,16
Synthesis of pure MC nylon-6
A proper quantity of CL was put into a three-necked flask and was heated to about 130°C. After completely melting, the CL melt was refluxed under vacuum for about 30 min to remove water in it. Then a proper amount of NaOH was added under dramatical stirring. The melt was refluxed under vacuum for another 30 min and a certain quantity of TDI was added. After mixing up well quickly, the melt was cast into a preheated mold at 170°C. The reaction lasted for 20 min. The products of MC nylon were then obtained and demolded. 17,18
Synthesis of grafting MC nylon-6
A proper quantity of TEMP and excess quantity of TDI were mixed quickly by agitating in a beaker at ambient temperature. Lots of heat was generated resulting in the formation of the solid intermediate product.
A proper quantity of CL was put into a three-necked flask and was heated to about 130°C. After completely melting, the CL melt was refluxed under vacuum for about 30 min to remove water in it. Then a proper amount of NaOH was added under dramatical stirring. The melt was refluxed under vacuum for another 30 min, and a certain quantity of intermediate product of TDI and TEMP was added into the three-necked flask. After mixing up well quickly, the melt was cast into a preheated mold at 170°C. The reaction lasted for 20 min. The products of MC nylon-6 were then obtained and demolded. 17,18
The synthesis process of MC nylon-6 was shown in Figure 1.
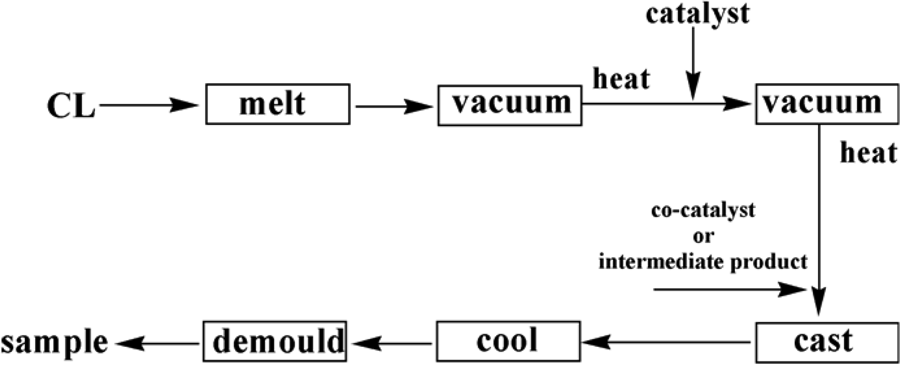
Diagramatic representation of synthesis process of MC nylon-6. MC: monomer casting.
Thermal oxidative aging of MC nylon-6
The samples were cut into pellets and put in an oven at 150°C in the ambient atmosphere to be subjected to thermal oxidative aging. The samples were taken out at regular time intervals and used for physical and chemical characterization. The temperature fluctuation in the oven was ±1°C.
Measurements
FTIR analysis
Fourier transform infrared (FTIR) spectroscopic analysis of TDI, TEMP and the template compound was conducted with Nicolet-560 FTIR spectrometer (Madison, Wisconsin, USA). The scanning rate was 20 scans per second and the resolution was 4 cm−1.
1 H NMR analysis
Proton nuclear magnetic resonance spectroscopy ( 1 H NMR) measurement of template compound was conducted with a Varian Inova-400 NMR spectrometer (Chesterfield, Michigan, USA) at room temperature. The signal frequency was 400 MHz, and the solvent was dimethylsulfoxide-d 6 (DMSO-d 6).
Isothermal weight loss measurement
The isothermal weight loss measurement was carried out in the heating oven at 240°C for 4 h. The thermal weight loss (W%) can be calculated as follows
where W 0 is the original weight of the sample and Wt is the weight of the samples after thermal degradation for the time t.
After the unstable part of MC nylon-6 sample decomposed rapidly, the degradation rate can be obtained by plotting the denary logarithm of the residual weight percentage (R) of the remained sample against heating time (t). The samples underwent thermal degradation in the heating oven at 240°C and then were taken out for the measurement of R per 1 h, respectively.
TGA analysis
Thermogravimetric analysis (TGA) was performed with a TA2950 thermobalance from TA Co. (New Castle, Pennsylvania, USA) with the flow rate of 50 mL min−1 under oxygen atmosphere. The granulated samples of about 10 mg were heated from ambient temperature to approximately 500°C at a rate of 10°C min−1. All samples were dried in the heating oven at 80°C for 2 h before the measurement.
End group analysis
The carboxylic acid
The sample of MC nylon-6 of about 0.3 g was dissolved in 20 mL of phenylcarbinol at 150°C. Then 3 mL of propyl alcohol was added, and the hot solution was titrated with NaOH (about 0.02 mol L−1) using phenolphthalein as the indicator. 19 The content of the carboxylic acid can be calculated with the following formula
where a is the consumption volume of NaOH solution of the sample (in milliliters), b is the consumption volume of NaOH solution for the solvent (in milliliters), C is the molar concentration of NaOH solution (moles per liter) and W is the weight of the sample (grams).
The amine end group
The sample of MC nylon-6 of about 0.3 g was dissolved in 25 mL of mixing solution of phenol-methyl alcohol (volume ratio 1: 1) at 55°C. Then the solution was titrated with HCl (about 0.005 mol L−1) using thymol blue as the indicator. The content of the end amine group can be calculated with the following formula
where A is the consumption volume of HCl solution of the sample (in milliliters), B is the consumption volume of HCl solution for the solvent (in milliliters), C is the molar concentration of HCl solution (moles per liter) and W is the weight of the sample (grams).
UV spectrum analysis
Samples of MC nylon-6 of about 0.2 g were dissolved in 50 mL of a mixed solvent of sulfuric acid/methanol (0.4 mol L−1). The measurement of ultraviolet (UV) absorption of the solution during aging was carried out with an Alpha-1860 UV–visible spectrophotometer (Shanghai Lab-spectrum instruments CO,.Ltd, Shanghai, China).
Gel content
Samples of MC nylon-6 of about 0.5 g were dissolved in 100 mL of formic acid (88 wt%). The solution (25 mL) was filtered through a sand core funnel, and the filtrate was dried in a vacuum oven to a constant weight (a). The gel content can be calculated from the formula
Nonisothermal crystallization analysis
The nonisothermal crystallization was performed with a Netzsch 204 Phoenix DSC (Bavarian, Germany). The temperature scale of (DSC) was calibrated with indium. The granulated samples of about 10 mg were heated quickly from ambient temperature to 250°C under a nitrogen atmosphere. After waiting for 5 min to eliminate the effects of the previous thermal history, the samples were cooled to ambient temperature at a constant rate of 10 K min−1 and then heated to 250°C at the same constant rate.
The quantity of heat absorbed during the melting of the polymer is substantively equivalent to that required to break down the crystal structure. The higher the crystallinity (X c), the higher the melting heat. X c can be calculated using the following formula
where H m is the melting enthalpy and H 0 is the balance melting enthalpy, that is, the melting enthalpy of 100% crystallizing MC nylon-6, which is 190 J g−1.
Results and discussion
Grafting of TEMP onto the molecule of MC nylon-6
A certain quantity of TDI and TEMP was mixed quickly in a liquid state, and the intermediate compound was formed immediately in a solid state through the reaction of amine group of TEMP with isocyanate group of TDI. There are two cases of the reaction and the reaction formula was shown in Figure 2.
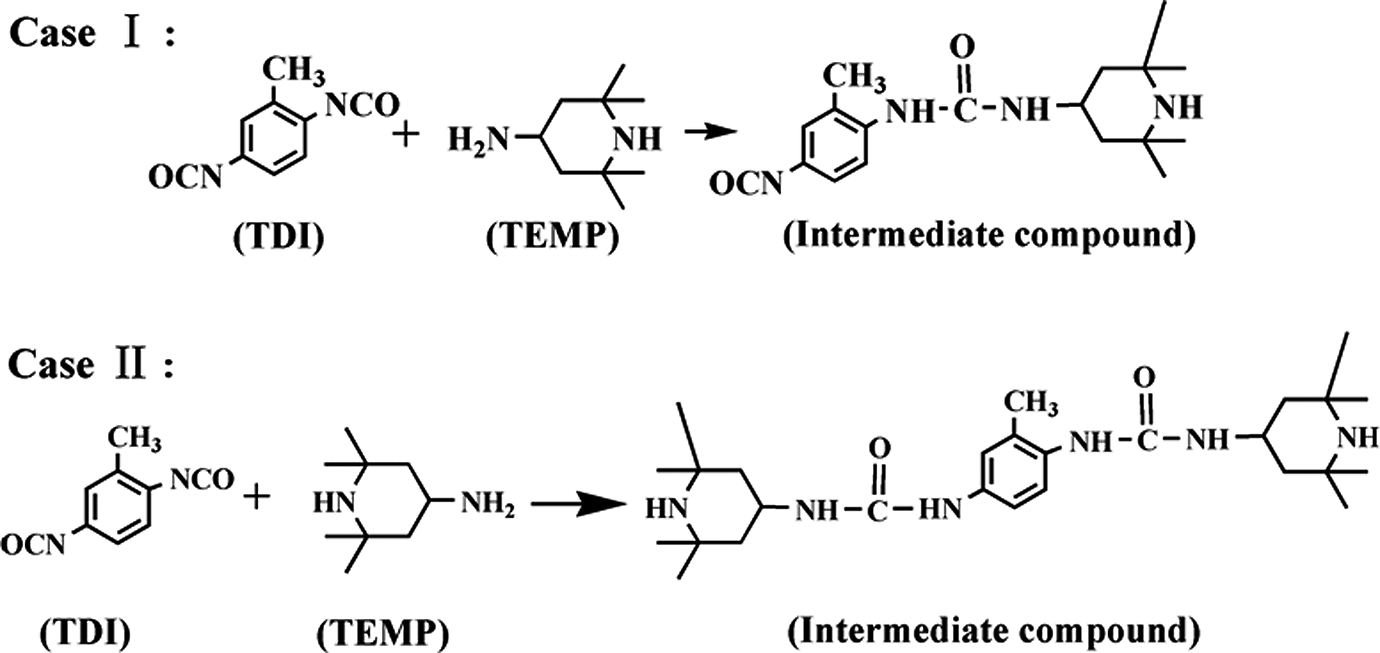
The reaction formula of TDI and TEMP. TDI: toluene-2, 4-diisocyanate; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
Case I may dominate during the reaction of TDI and TEMP due to excess dosage of TDI. The remaining isocyanate (–NCO) group of the intermediate compound can continue to react with CL, which produced the TEMP grafted MC nylon-6. The reaction mechanism was shown in Figure 3.
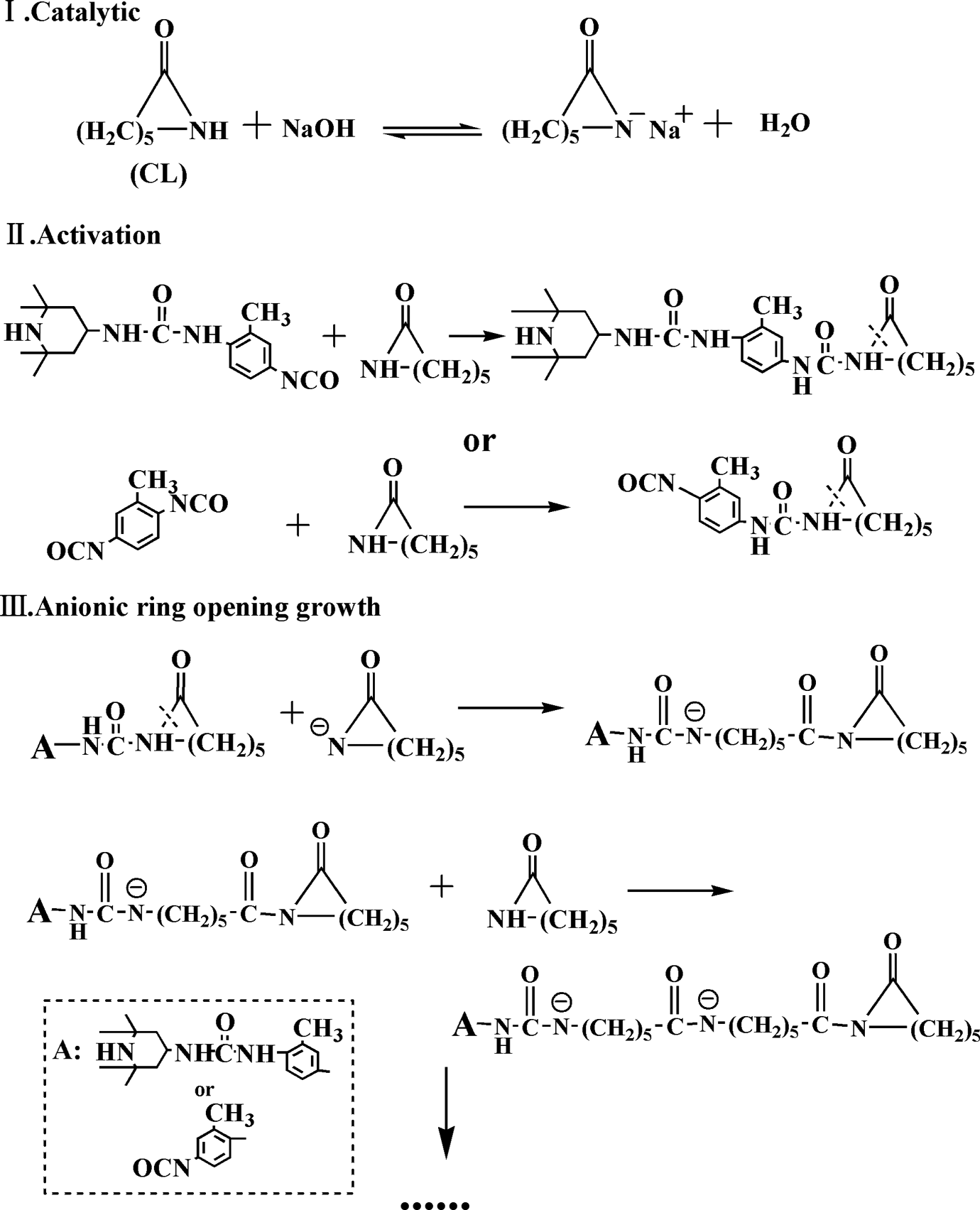
Anionic ring opening polymerization of caprolactam.
To verify the reaction of TEMP and TDI, a template compound was prepared with 2:1 of molar ratio of TEMP and TDI and purified with acetone.
The structure of the template compound was confirmed by comparison of FTIR spectrum with TDI and TEMP, as shown in Figure 4. For TDI, the band at 2253 cm−1 was attributed to the characteristic absorption peak of isocyanate groups (–NCO), and the wide band at 1520 cm−1 was attributed to skeleton vibration of benzene. For TEMP, the absorbance bimodal peak at 3300 cm−1 was attributed to the stretching vibration of N–H, the band at 2900–3000 cm−1 was attributed to the characteristic absorption peak of piperidine ring. As for the template compound, the above absorbance peak of –CNO disappeared, two new peaks at 1546 cm−1 and 1653 cm−1 can be observed, which were attributed to the characteristic peak of –NHCONH– and its carbonyl group (C=O). The band at 3100–3400 cm−1 was attributed to the stretching vibration of N–H and was strengthened, and the characteristic peaks of piperidine ring and benzene ring still exited. All the results were due to the reaction of TDI and TEMP.
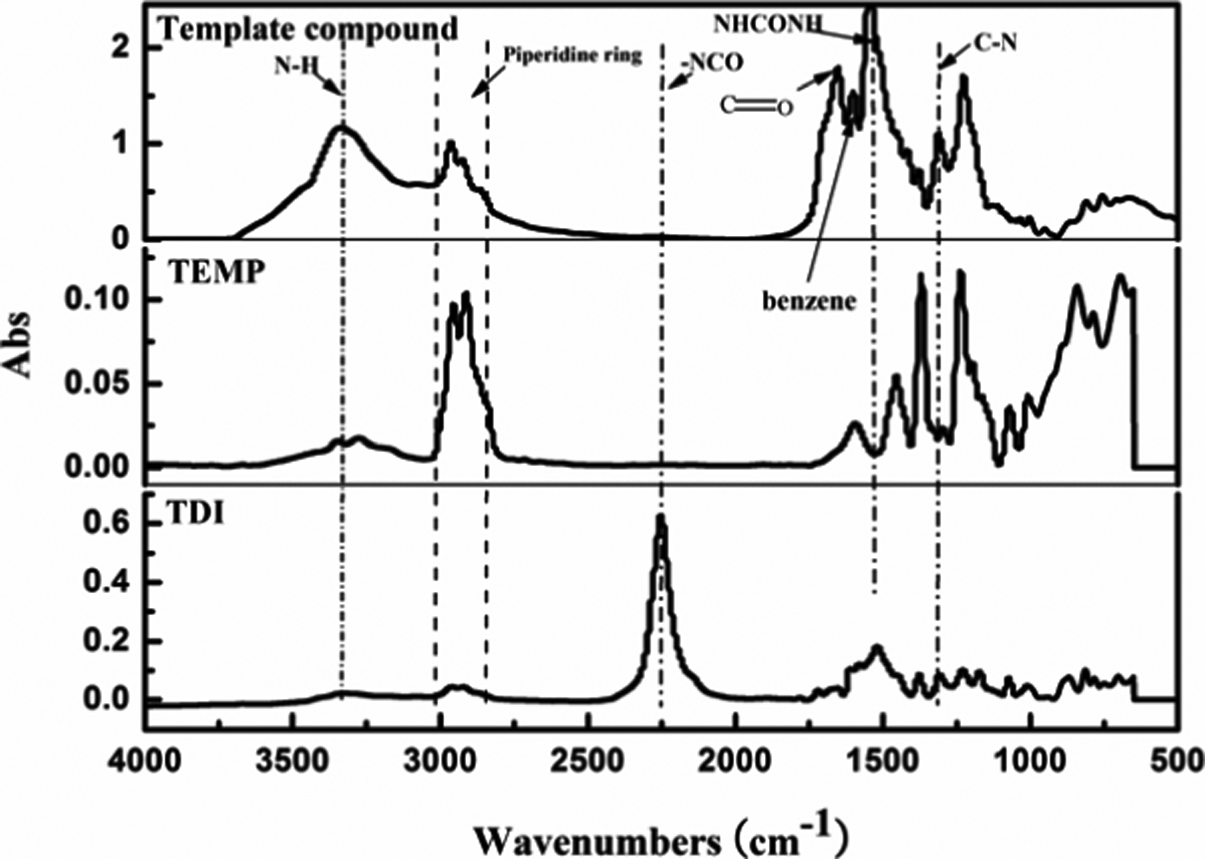
FTIR spectra of TDI, TEMP and the template compound. FTIR: Fourier transform infrared; TDI: toluene-2, 4-diisocyanate; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
Figure 5 shows the 1 H NMR spectra of template compound in DMSO (a) and DMSO-D2O (heavy water) (b) at room temperature. The protons in the methyl group connected to the piperidine ring are evidenced by the chemical shift at 1.02 ppm and 1.15 ppm. The protons in the methylene group on the piperidine ring are exhibited at about 0.90 ppm and 1.73 ppm. The protons in the methane groups on the piperidine ring can be found at 3.85 ppm. The chemical shift at 2.05 ppm is attributed to the methyl group connected to the benzene. The peaks 6, 7 and 8 were attributed to the protons of benzene.
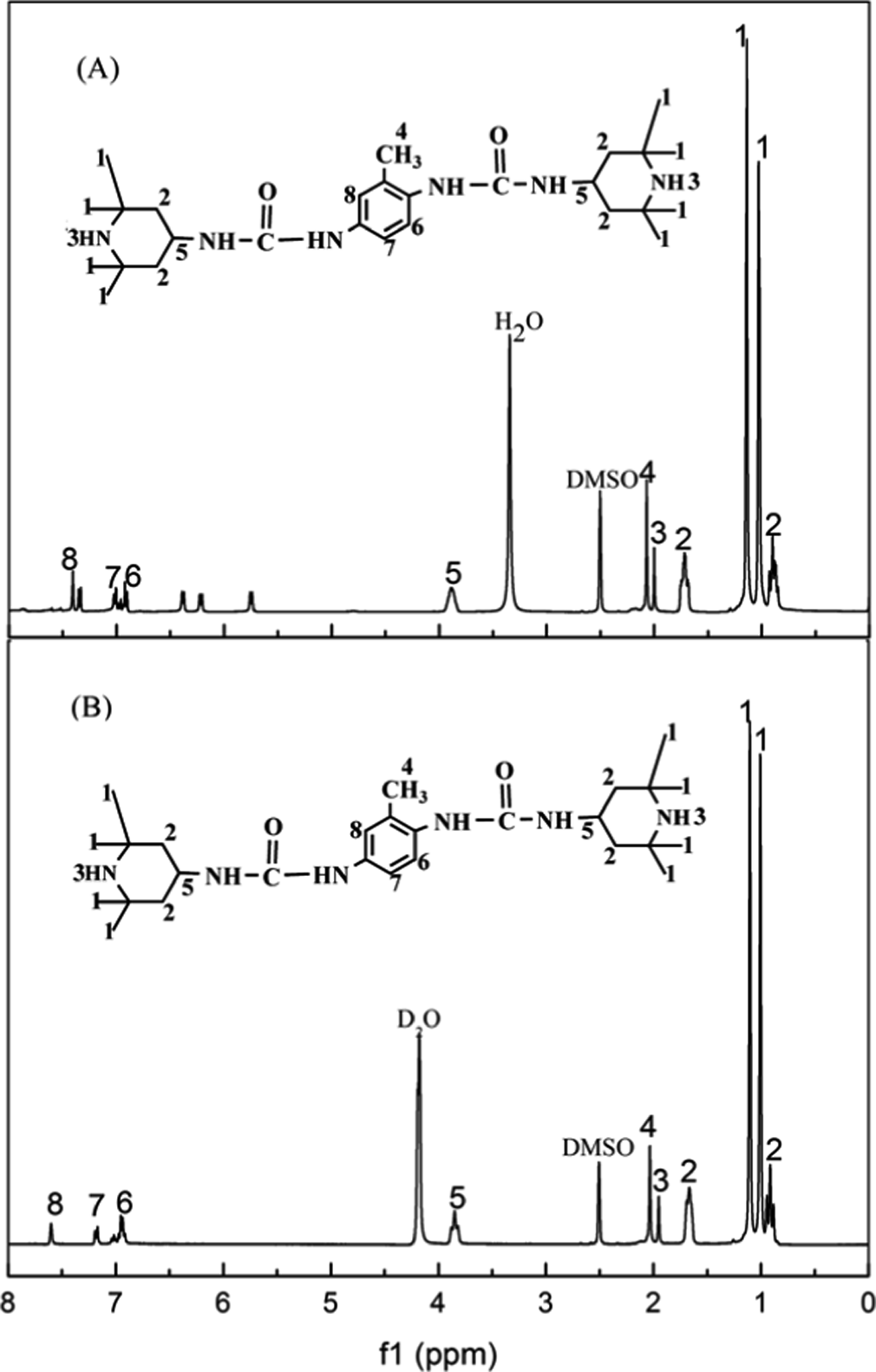
1H NMR spectra of the template compound ((a) in DMSO and (b) in DMSO-D2O (heavy water)). 1H NMR: Proton nuclear magnetic resonance; DMSO: dimethylsulfoxide.
Comparing 1 H NMR spectra of template compound in DMSO (a) and DMSO-D2O (b), it can be seen that the NMR chemical shift at 5.74 ppm, 6.21 ppm, 6.37 ppm and 7.34 ppm in Figure 5(a) disappeared in Figure 5(b), indicating that the active hydroxyl hydrogen on the ureido groups were exchanged by heavy water. The results demonstrated the template compound produced by the reaction of TDI and TEMP contained piperidine ring, benzene and ureido groups.
Effect of TEMP on the isothermal and nonisothermal degradation of MC nylon-6
The effect of TEMP dosage on the isothermal weight loss of MC nylon-6 heated at 240°C for 4 h was shown in Figure 6. With the increase in TEMP dosage, the thermal weight loss of the samples first decreased and then increased, and the preferred dosage of TEMP was about 0.2 mol%. The increase in the thermal weight loss of the sample may be due to the reduction in the molecular weight at high dosage of TEMP.
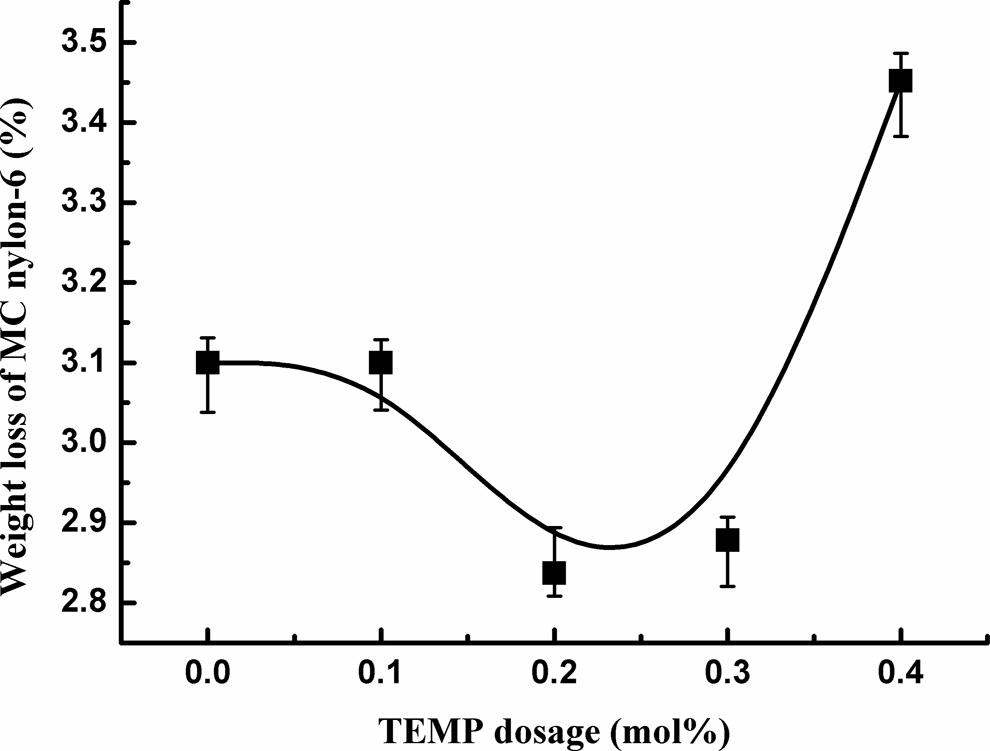
Effect of TEMP dosage on the isothermal weight loss of MC nylon-6. MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
Figure 7 displayed the relationship between denary logarithm of the residual weight percentage (R) and heating time (t). It can be seen that the value of R of all the samples decreased with heating time, and it does much more slowly by addition of 0.2 mol% TEMP than that of pure MC nylon-6, indicating that very small amount of TEMP can show thermostabilization effect on MC nylon-6.
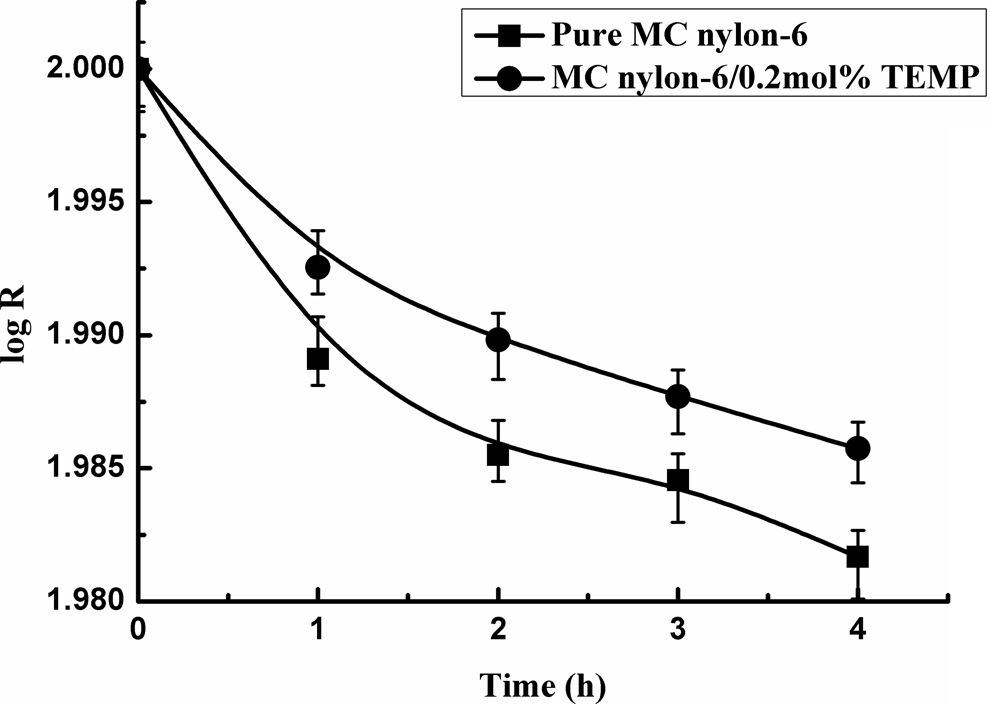
Thermal weight loss rate curves of MC nylon-6. MC: monomer casting.
TGA is an important method for the study on the thermostability of MC nylon-6. 20,21 Thermal degradation characteristic temperatures such as T onset and T end, respectively, represent the temperature at the intersection point of the tangent line at the fastest thermal weight loss rate with the extension line at the start and end of the degradation. V max and T peak are, respectively, the maximal thermal degradation rate and the temperature at this point.
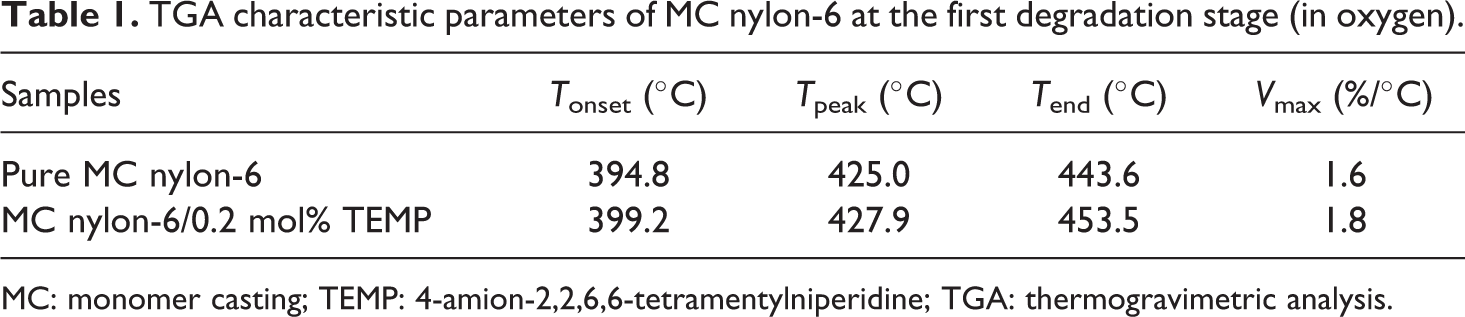
As shown in Figure 8, the TGA curves of the samples presented a two-step inverse ‘S’ shape correlation, which indicated that the degradation process of MC nylon-6 had two stages. For the first stage, compared with pure MC nylon-6, the thermal degradation characteristic temperatures of the grafted sample increased. As listed in Table 1, T onset increased by 5°C and T end increased by 10°C, indicating the stabilization effect of TEMP on MC nylon-6. But for the second stage, when the temperature was above 460°C, the ureido bond between TEMP and TDI was easy to be broken. Therefore, the thermal degradation was accelerated.
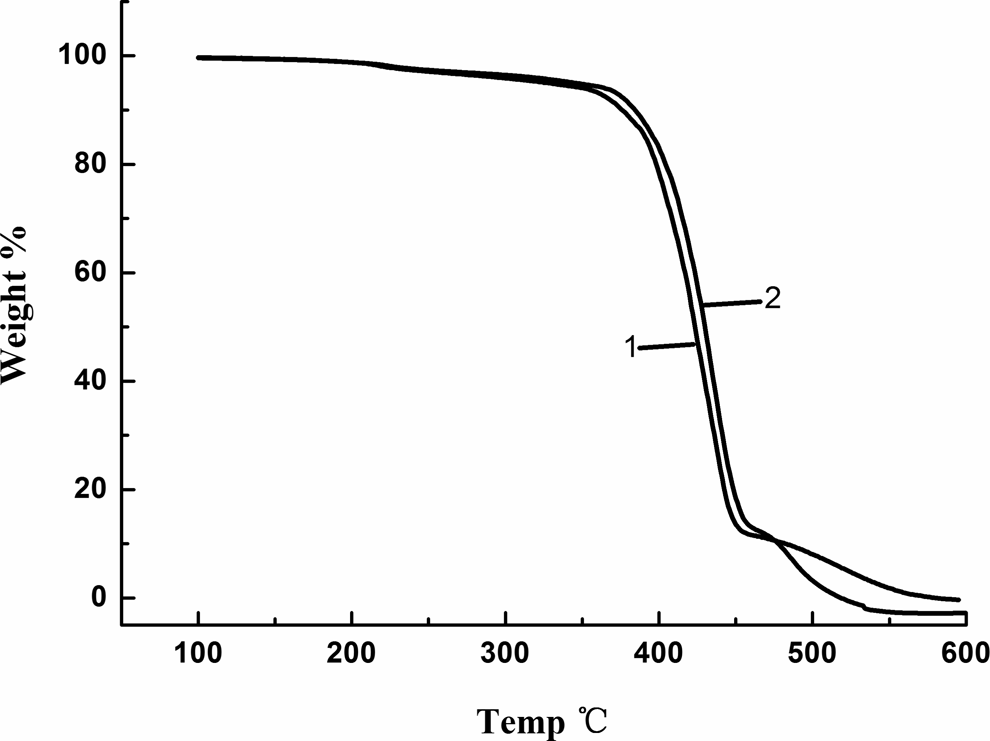
TGA curves of MC nylon-6 ((a) pure MC nylon-6 and (b): MC nylon-6/0.2 mol% TEMP). TGA: thermogravimetric analysis; MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
TGA characteristic parameters of MC nylon-6 at the first degradation stage (in oxygen).
MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine; TGA: thermogravimetric analysis.
Effect of TEMP on the long-term thermal-stability of MC-nylon6
The long-term aging of MC nylon-6 was conducted at 150°C for 9 days. Figure 9 displayed the relationship between denary logarithm of the residual weight percentage (R) and heating time (t) of the samples. It can be seen that the value of R of all samples decreased with heating time, and it does much more slowly by the addition of 0.2 mol% TEMP than that of the pure MC nylon-6.
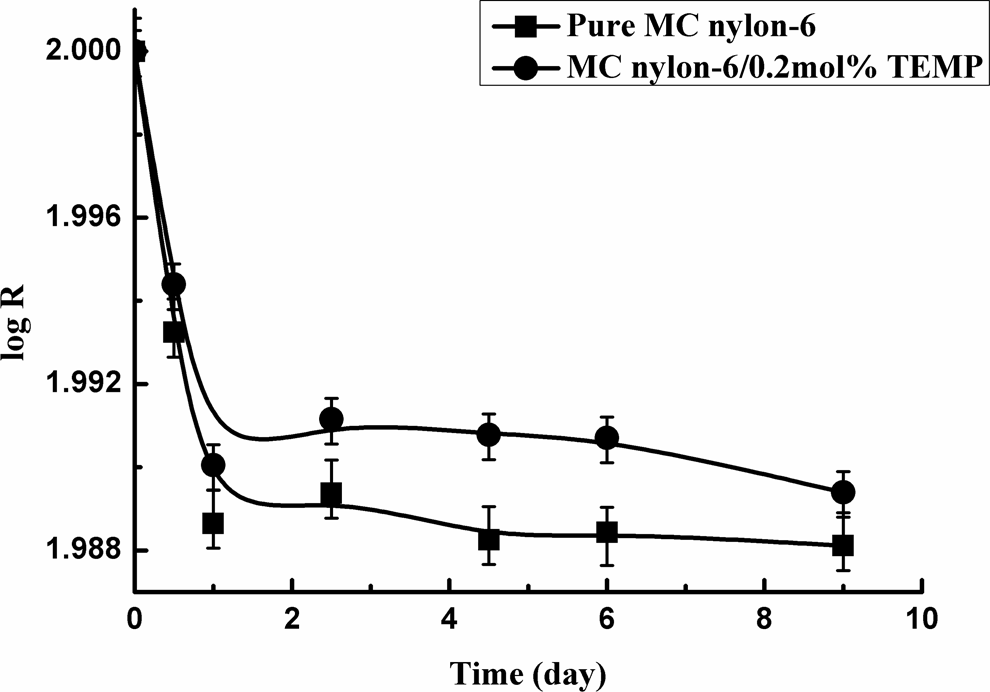
The long-term degradation rate curves of MC nylon-6. MC: monomer casting.
The amido-bond is considered to be the weakest bond in the molecule of polyamide. The rupture of the amido-bond and the adjacent C–C bond resulted in the formation of the carboxylic acid, carbonyl and the end amine group. The decline in the concentration of the end amine group of MC nylon-6 perhaps resulted from the reaction between end amine groups and carboxylic acids or producing the compounds such as tertiary amine or producing the compounds escaped from the matrix by aging degradation. During the thermal oxidative aging of MC nylon-6, the higher the concentration of the carboxylic acid, the more quickly the oxidation aging of the sample, and the further degradation of the polymers maybe promoted. However, the high concentration of amine group in the sample was favorable to its thermal oxidative stability. 22
The variation in the chemical structure (the carboxylic acid and the amine end group) of MC nylon-6 with aging time at 150°C was shown in Figure 10 and 11. It can be seen that with increasing aging time, the carboxylic acid concentration rose and the end amine group concentration dropped monotonically for MC nylon-6. But for the grafting MC nylon-6, they changed much more slowly. Moreover, the carboxylic acid concentration of the grafting MC nylon-6 was lower than that of the pure sample, and its end amine group concentration was relatively higher during the whole aging process. In addition, the ratio of carboxylic acid to end amine group of the grafting MC nylon-6 was always remarkably lower than that of the pure sample, and the difference in the ratio between the two samples became greater with aging time, indicating that the stabilizer of TEMP inhibited the oxidation reaction of MC nylon-6 effectively.
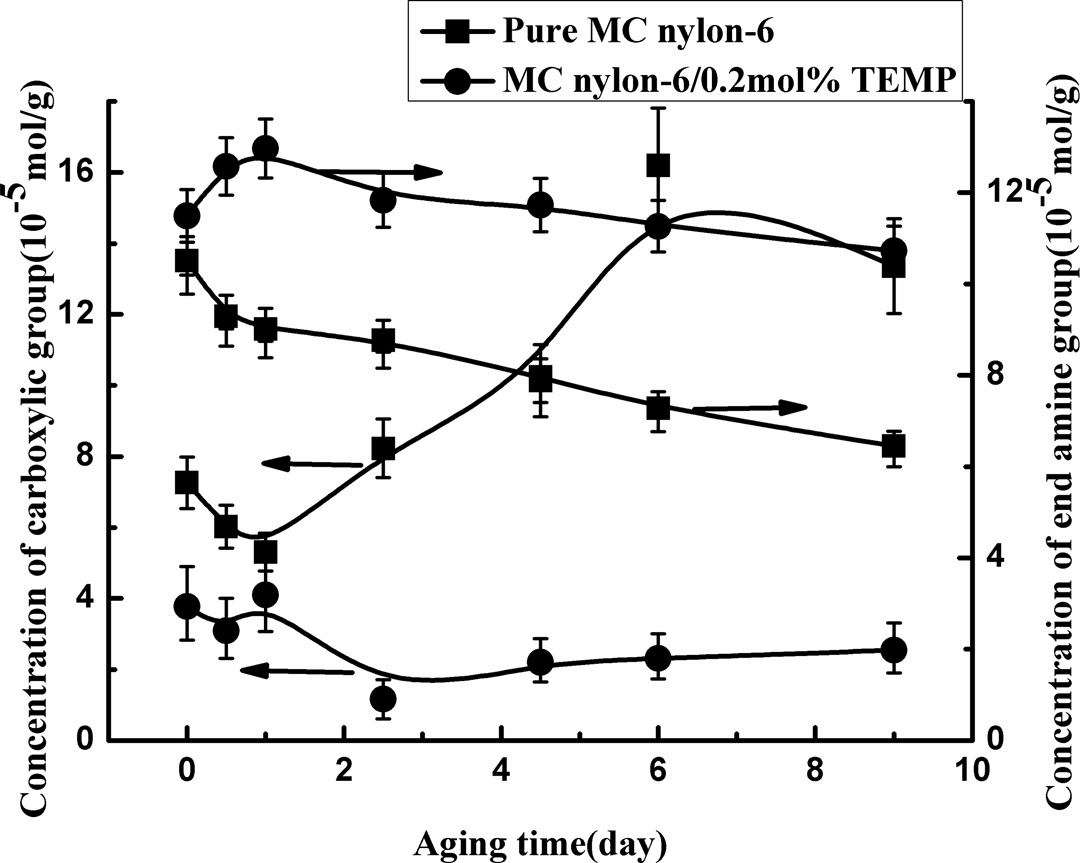
Concentration of carboxylic acid and end amine group of MC nylon-6 as a function of aging time at 150°C. MC: monomer casting.
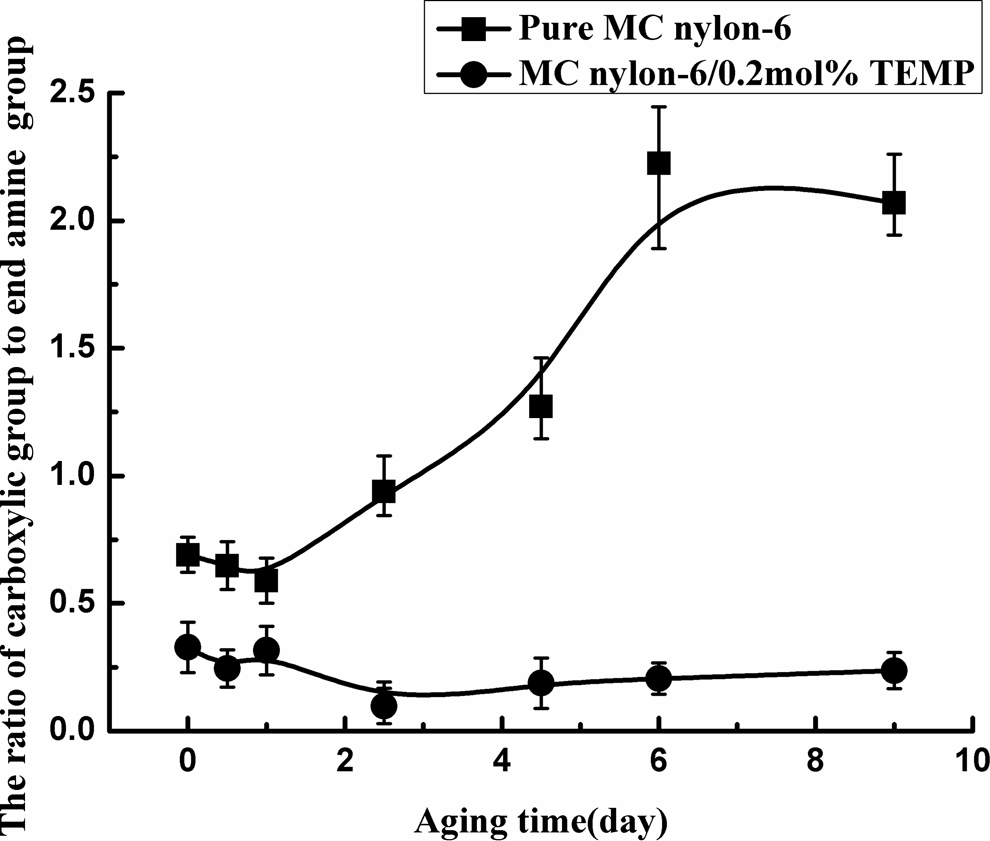
Ratio of carboxylic acid to end amine group of MC nylon-6 as a function of aging time at 150°C. MC: monomer casting.
UV spectra of MC nylon-6 aging at 150°C are shown in Figure 12. UV absorption of MC nylon-6 at the bands of 220 nm and 250–290 nm increased obviously after aging, while absorption intensity of pure MC nylon-6 was significantly higher than that of the grafting MC nylon-6.
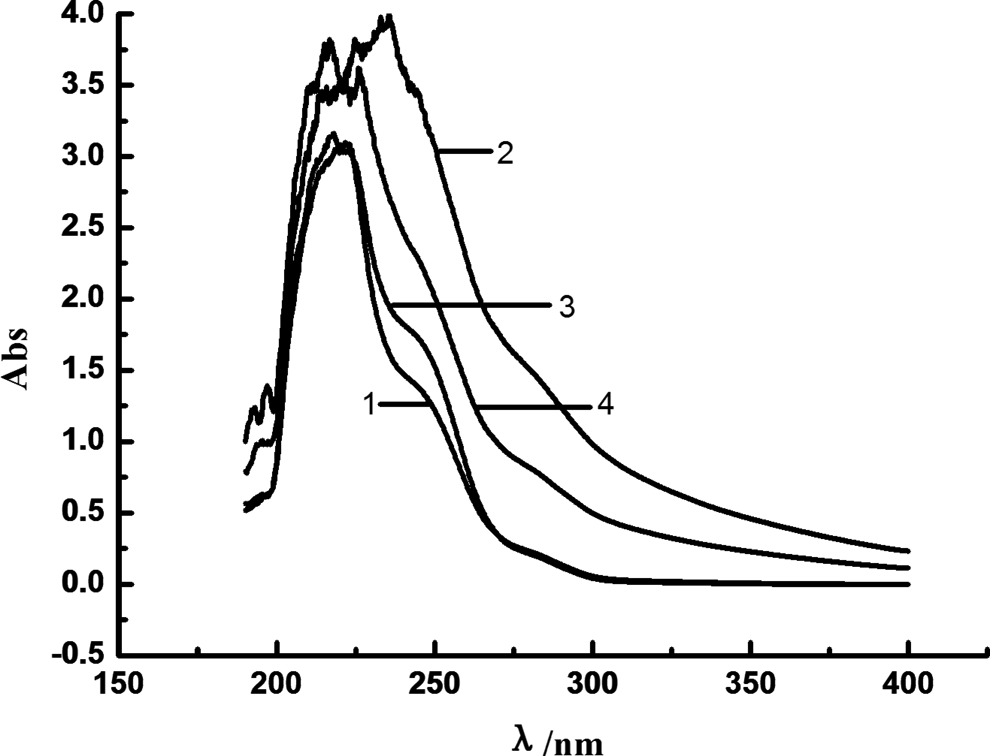
Ultraviolet spectrum of MC nylon-6 as a function of aging time at 150°C ((a) pure MC nylon-6/0 day; (b) pure MC nylon-6/9 days; (c) MC nylon-6/0.2 mol% TEMP/0 day; (d) MC nylon-6/0.2 mol% TEMP/9 days). MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
A series of degradation reactions were initiated under the thermaloxidation aging, as shown in Figure 13. Before aging, a strong UV absorption peak at about 220 nm of MC nylon-6 should be attributed to the electronic π → π* transitions of the carbonyl amide groups. After aging, degradation products such as unsaturated aldehydes, ketones, carboxylic acids and other compounds were generated, and π → π* or n → π* electron transition of carbonyl groups in these compounds may result in increasing UV absorption at about 220 nm. The UV absorption at 250–290 nm was enhanced due to the formation of isolated carbonyl groups during aging.
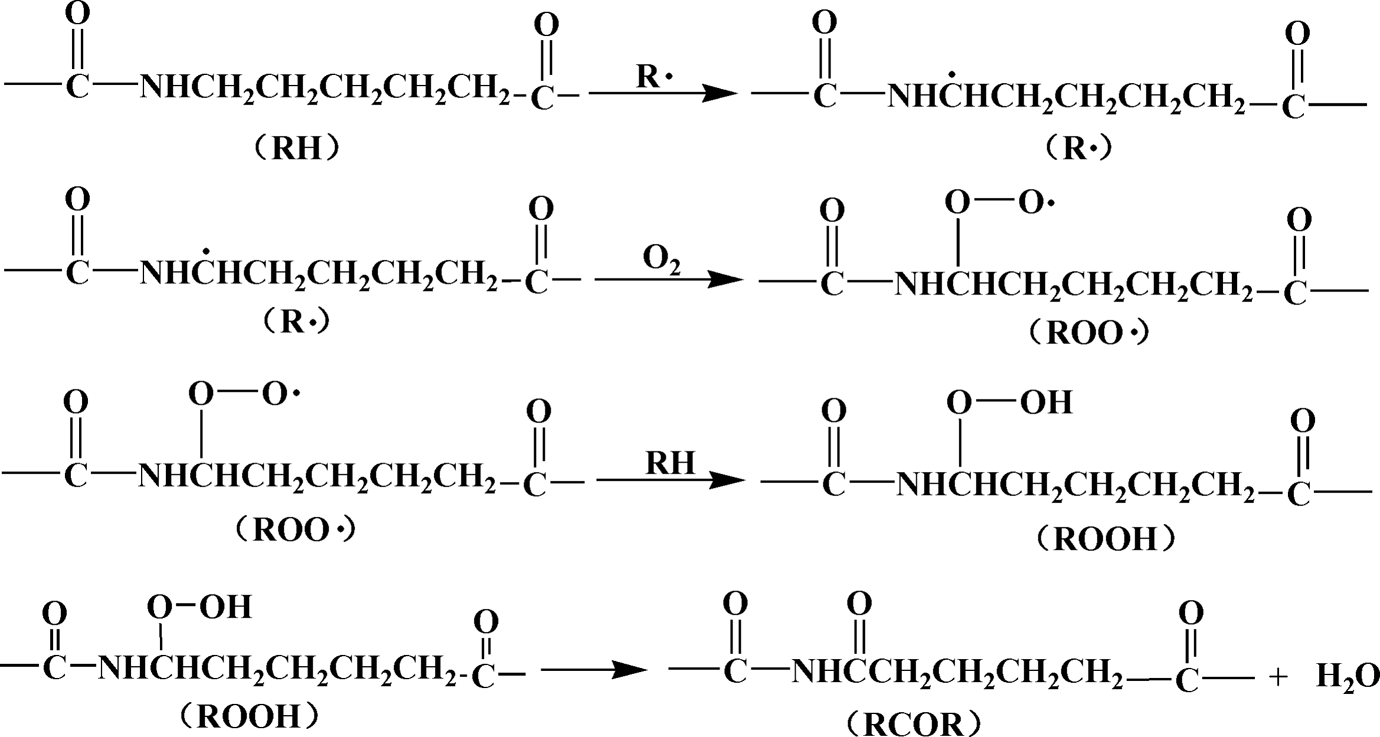
The degradation reactions of MC nylon-6 under the thermal-oxidation aging. MC: monomer casting.
During thermo-oxidative aging of polymer materials, molecular degradation and cross-linking usually occurred simultaneously. The gel content of MC nylon-6 as a function of aging time was measured, as shown in Figure 14. It can be seen that the gel content of the sample decreased at the initial stage of aging when molecular degradation dominated under thermal-oxidation, and then increased quickly when molecular cross-linking dominated. The gel content of the sample of the grafting MC nylon-6 was always lower than that of the pure sample, indicating the stabilizing effect of TEMP on MC nylon-6.
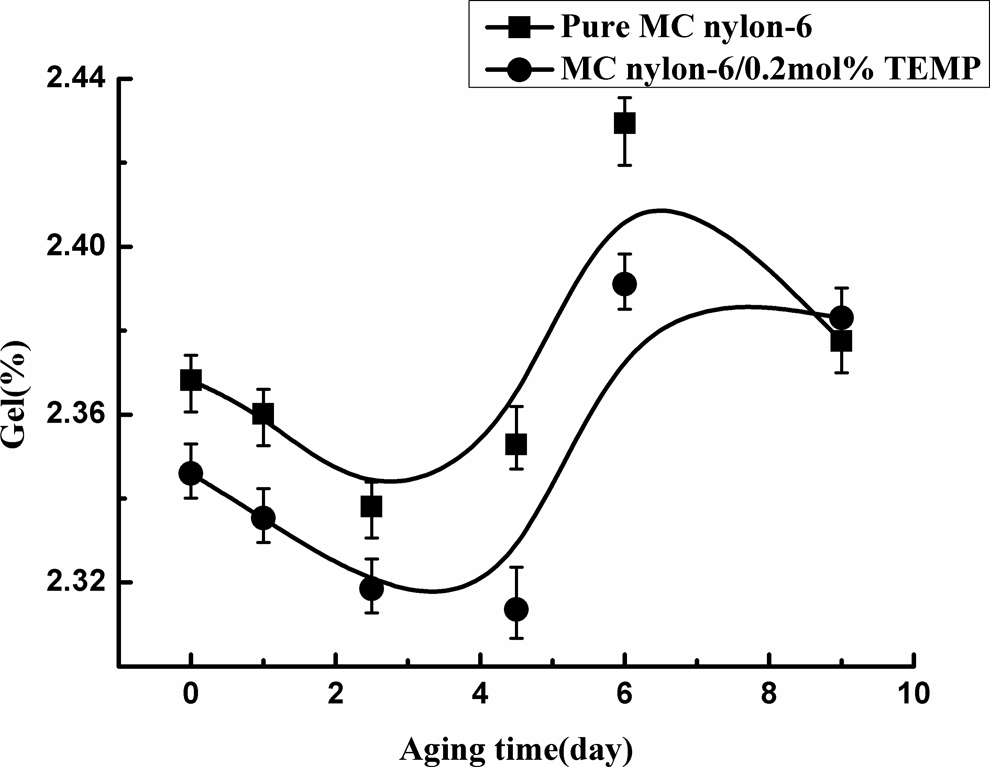
Gel content of MC nylon-6 as a function of aging time at 150°C. MC: monomer casting.
The nonisothermal DSC curves of the pure and grafting samples of MC nylon-6 under different aging time with a heating and cooling rate of 10 K min−1 were shown in Figure 15, from which the onset crystallization temperature (T onset), the crystallization peak temperature (T c), the melting temperature (T m), the melting enthalpy (ΔH m), the crystallinity (Xc) and the crystalline half-peak width (ΔW) can be obtained, as listed in Table 2.
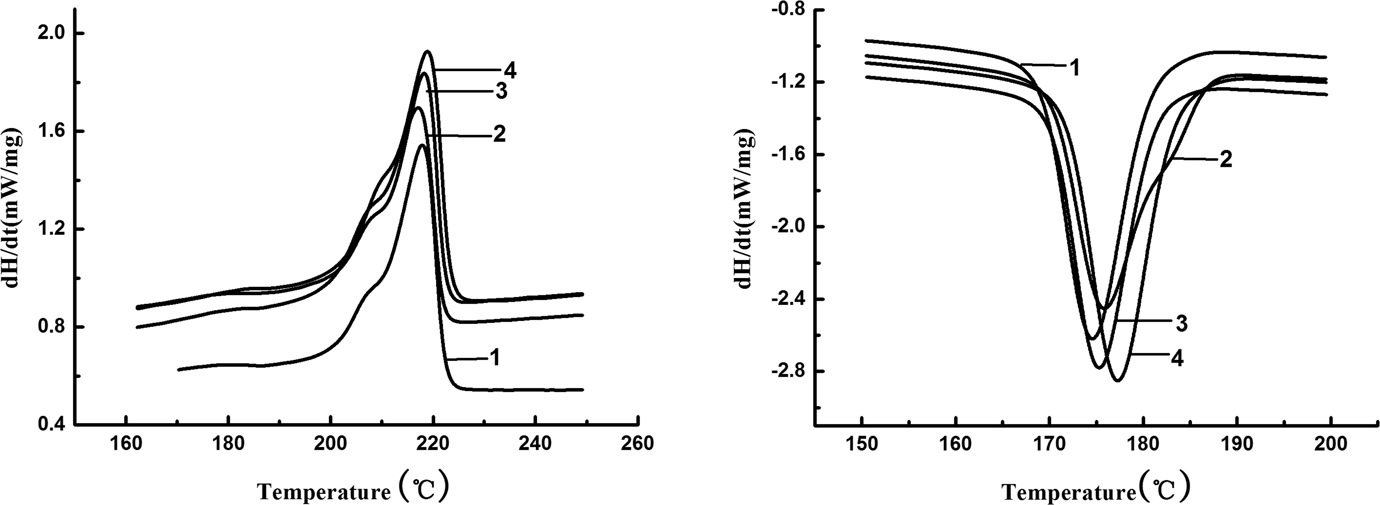
DSC curves of MC nylon-6 at different aging time with heating and cooling rate of 10 K min−1 ((a) Pure MC nylon-6/0 day; (b) pure MC nylon-6/9 days; (c) MC nylon 6/0.2 mol% TEMP/0 day; (d) MC nylon 6/0.2 mol% TEMP/9 days). DSC: differential scanning calorimetry; MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
Crystallization parameters of MC nylon-6 at different thermal aging time.a
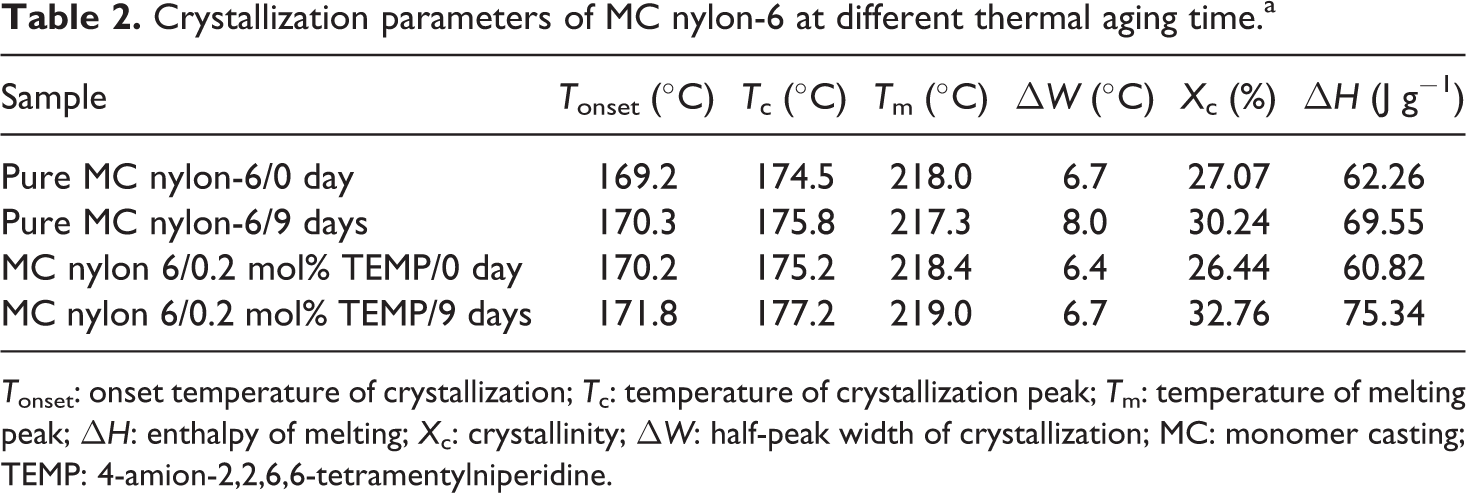
T onset: onset temperature of crystallization; T c: temperature of crystallization peak; T m: temperature of melting peak; ΔH: enthalpy of melting; X c: crystallinity; ΔW: half-peak width of crystallization; MC: monomer casting; TEMP: 4-amion-2,2,6,6-tetramentylniperidine.
It can be seen that for both pure and grafting samples of MC nylon-6, the crystalline fraction showed an increase in crystallinity for the aged sample, indicating the fact that post crystallization occurred as a result of the aging process, which can yield mobile chains in the amorphous region suitable to join the polymeric crystallites. However, the small molecules and polymers with low molecular weight produced by degradation may also contribute to a rise in the crystallinity. On the other hand, the thermal oxidative aging would primarily occur on the weak bond of the molecular chain in the amorphous region of the polymer, and the molecular degradation in this region resulted in the increase in the concentration of the crystalline region of MC nylon-6. Meanwhile, some of low-molecular-weight products have nucleation effect on MC nylon-6, making it to crystallize at comparatively high temperature. The low regularity of the molecular chains of the sample with branch chains of oxides and peroxides produced by oxidation would lead to a decrease in the crystalline growth rate, presenting an increase in ΔW.
Conclusion
MC nylon-6 was a polymeric modified by grafting TEMP as reactive-hindered amine onto the chain of MC nylon-6 so as to improve the compatibility and stability of the stabilizer in the polymer matrix. The structure of the reactive stabilizer was confirmed by FTIR and 1 H NMR analysis. The effect of TEMP on the thermal stability of MC nylon-6 was studied. The result showed that with the increase in TEMP dosage, the thermal weight loss of the samples first decreased and then increased, and the preferred dosage of TEMP was about 0.2 mol%. The residual weight percentage (R) of all samples decreased with heating time, and it does much more slowly for the grafting sample than that of pure MC nylon-6. The thermal degradation characteristic temperatures of the grafting sample were also higher. The long-term aging of MC nylon-6 indicated that the carboxylic acid concentration of the grafting MC nylon-6 was lower than that of the pure sample, and its end amine group concentration was relatively higher during the whole aging process, presenting more stable chemical structure. UV absorption of MC nylon-6 at the bands of 220 nm and 250–290 nm increased obviously after aging, while absorption intensity of pure MC nylon-6 was significantly higher than that of the grafting sample. The gel content of the grafting MC nylon-6 was also lower than that of the pure sample. X c of MC nylon-6 increased, the crystallization growth rate and T m decreased with aging time. The anneal process at high temperature and the small molecular products may be responsible for it.
Footnotes
Funding
This work was supported by the Key Natural Science Fund of China (51133005).