Abstract
To further improve and enhance the performances and properties of polyvinyl acetate (PVAc), montmorillonite (MMT) and N-hydroxymethyl acrylamide (NMA) were introduced together to polymerize with vinyl acetate (VAc). The exfoliated nanocomposite of PVAc–NMA–MMT was prepared through different synthesis precursors. Linear macromolecular chains of PVAc–NMA were formed in MMT layers. MMT was exfoliated into layers or sheets of nanoparticles and dispersed randomly in PVAc–NMA matrix. Both PVAc–NMA and MMT had small particles with a diameter of 50–100 nm. They dispersed together randomly. PVAc–NMA–MMT was a pseudo-plastic non-Newtonian fluid and possessed the normal stress effect (or Weissenberg effect from the pole-climbing phenomenon. With the change of different synthesis precursors and the increase of NMA content and MMT content in the synthesis system, the molecular weight of PVAc–NMA–MMT increased. PVAc–NMA–MMT had good dispersion, excellent storage stability and high static tensile at 6.47–6.85 MPa.
Introduction
Polyvinyl acetate (PVAc) is an acceptable environmentally friendly adhesive that is usually prepared from the vinyl acetate (VAc) monomer in the mixture of protective colloid, nonion emulsifier, initiator and water. It is nonpoisonous, non-harmful, easily produced, low cost, easy to apply, economical on resources and so on; therefore, it is widely used in the bonding of many porous materials such as wood processing, furniture packaging, building decoration, texture bonding and print bonding. However, its defects, such as poor cold tolerance, poor mechanical stability, poor thermal stability and poor water resistance, directly affect the integrated value of utilization and processing. 1,2 Now, many studies have been done on these topics, including the preparation methods, copolymerization, co-blending, radiation preparation and so on. 3 –10
In our previous work, a natural nanomineral of montmorillonite (MMT) and organic MMT (OMMT) activated by long alkyl chain quaternary ammonium salts were used to polymerize with VAc and the exfoliated nanocomposite of PVAc–MMT 11,12 and PVAc–OMMT was prepared, 13 –20 and an amide derivative of N-hydroxymethyl acrylamide (NMA) from unsaturated carboxylic acid was used to copolymerize with VAc to form the copolymer of PVAc–NMA. 21,22 MMT, OMMT and NMA all improved and enhanced the performances and properties of PVAc at a certain extent, but was not perfect. For example, the static tensile of PVAc–MMT and PVAc–OMMT was high, but their dispersion as well as storage time was poor; PVAc–NMA had good dispersion and long storage time, but its static tensile was unsatisfactory. Therefore, to develop the strongpoint and avoid the shortcoming, in this study, MMT and NMA were introduced together to modify PVAc. The exfoliated nanocomposite of PVAc–NMA–MMT was prepared through different synthesis precursors. The chemical structure investigated by X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR), the dispersion investigated by the method of liquids dispersed in water and transmission electron microscopy (TEM), the rheology investigated by the apparent viscosity, power–law function equation, Newtonian fluid flow equation and Cross–Williamson model viscous equation and the static tensile investigated by the Hooke’s law and Poisson’s ratio equation were described in detail.
Experimental
Samples
Briefly, 1 g of MMT was immersed into 25 g of VAc for 24 h. They were then mixed with 0.5 g of NMA, 70 g of 10% polyvinyl alcohol (PVA) solution, 0.5 g of alkylphenol polyoxyethylene (10) ether, 3.75 g of 10% ammonium persulfate solution, 6.25 g of sodium lauryl sulfate and 250 g (200 g, 150 g, 100 g and 50 g) of water together under stirring vigorously until it became a homogeneous emulsion. Then the temperature rose to 70°C. While stirring vigorously, 3.75 g of 10% ammonium persulfate solution and 25 g of VAc were gradually added into the homogeneous emulsion in 6 h (5 h, 4 h, 3 h and 2 h) for polymerization. Subsequently, the temperature rose to 85–90°C, the emulsion further polymerized for 0.5–1 h. After polymerization, the temperature dropped to below 50°C and 4 g of ethanol, 3 g of water, 0.3 g of sodium benzoate, 0.18 g of sodium bicarbonate and 6 g of di-n-butyl phthalate were added into the emulsion. Finally, PVAc–NMA–MMT-A (PVAc–NMA–MMT-B, PVAc–NMA–MMT-C, PVAc–NMA–MMT-D and PVAc–NMA–MMT-E) was obtained. Here, A, B, C, D and E were used to, respectively, mark the different synthesis precursors with different polymerization time and different proportion of water. Here, VAc and NMA were the main polymerization monomers, PVA was the protective colloid that had a polymerization degree and a alcoholysis degree of 1700 and 88%, respectively, ammonium persulfate was the initiator, alkylphenol polyoxyethylene (10) ether was the emulsifier, sodium lauryl sulfate was the anionic surfactant, di-n-butyl phthalate was the plasticizer, and the mixture of ethanol, water, sodium benzoate and sodium bicarbonate was the pH regulator. In this study, MMT was bought from Zhejiang Huate Group, China, and all other were analytical reagents from Sigma-Aldrich (St. Louis, MO, USA).
Solid content
The solid content (O, %) is calculated by:
Where m1 (g) is the original weight of liquid samples and m2 (g) is the weight of samples dried for 1 h at 105°C.
XRD
Random powder samples were tested using a DX-2000 XRD under Cu Kα of radiating, 40 kV of tube voltage, 30 mA of tube current, scanning from 0.5° to 15° at a rate of 0.02s−1 and λ = 1.54184 Å of wavelength.
FTIR
The solid samples were tested using a NICOLET 380 FTIR.
Apparent viscosity
The liquid samples were tested using a NDJ-1 rotary viscometer. The shear rate (γ) was 0.63s−1, 1.26s−1, 3.14s−1 and 6.28s−1, respectively. The apparent viscosity (η, mPa s) under different shear rate was obtained.
TEM
The liquid samples were observed by a JEM-3100F TEM with magnification 20000×.
Dispersion
Overall, 3–5 drops of liquid samples were added to 20 ml of water in a glass dish (diameter 90 mm). When they became homogeneous, the dispersion was observed comparing with water.
Static tensile
The liquid samples were made into films of 50 mm × 10 mm × 0.5 mm. After drying them to constant weight at room temperature, they were tested by a SANS CMT 5000 computer-controlled electronic universal testing machine under a tensile rate of 10 mm min−1 and a test temperature of 25°C. The test should be finished in 10 min.
Storage time
The liquid samples were stored away from light at room temperature till it appeared as a gel and/or delamination. The cycle was noted as the storage time (S, days).
Results and discussion
Chemical structure
Now, determining a material to be an intercalated or an exfoliated nanocomposite generally depends on its diffraction peak position, shape and intensity of XRD patterns. 23,24 If one or more diffraction peaks corresponding to the large value of d (001) for layered silicate crystal layers appear on XRD patterns, and the diffraction peak position has a certain migration to the small angle compared with that of layered silicate powder, this indicates that the layered silicate is opened, it is called as the intercalated nanocomposite. For the exfoliated nanocomposite, the XRD patterns seem to be linear, no diffraction peak appears within the diffraction angle range of 0.5–5°, this indicates that the ordered structure of clay layers are destroyed and exfoliated completely.
In our previous work, 11 –20 the nanocomposites of PVAc–MMT and PVAc–OMMT were prepared through different synthesis precursors, all of them being exfoliated nanocomposites. In their exfoliation, organics made the first intercalation into MMT, the linear macromolecular chains of PVAc formed in OMMT layers made the second intercalation, so the distance between MMT layers was greatly stretched. The original ordered crystal MMT layers were destroyed completely and fully exfoliated, they dispersed independently in the emulsion with nanometer-thick layers. PVAc–NMA–MMT also was an exfoliated nanocomposite (Figure 1). They had much more significant exfoliation than PVAc–MMT and PVAc–OMMT because the copolymer of PVAc–NMA had longer linear macromolecular chains and higher molecular weight than PVAc, which exfoliated MMT into layers or sheets of nanoparticles more easily, had strong exfoliation on these nanoparticles, and made them to be much smaller. They dispersed in PVAc–NMA matrix randomly such as MMT and OMMT in PVAc.
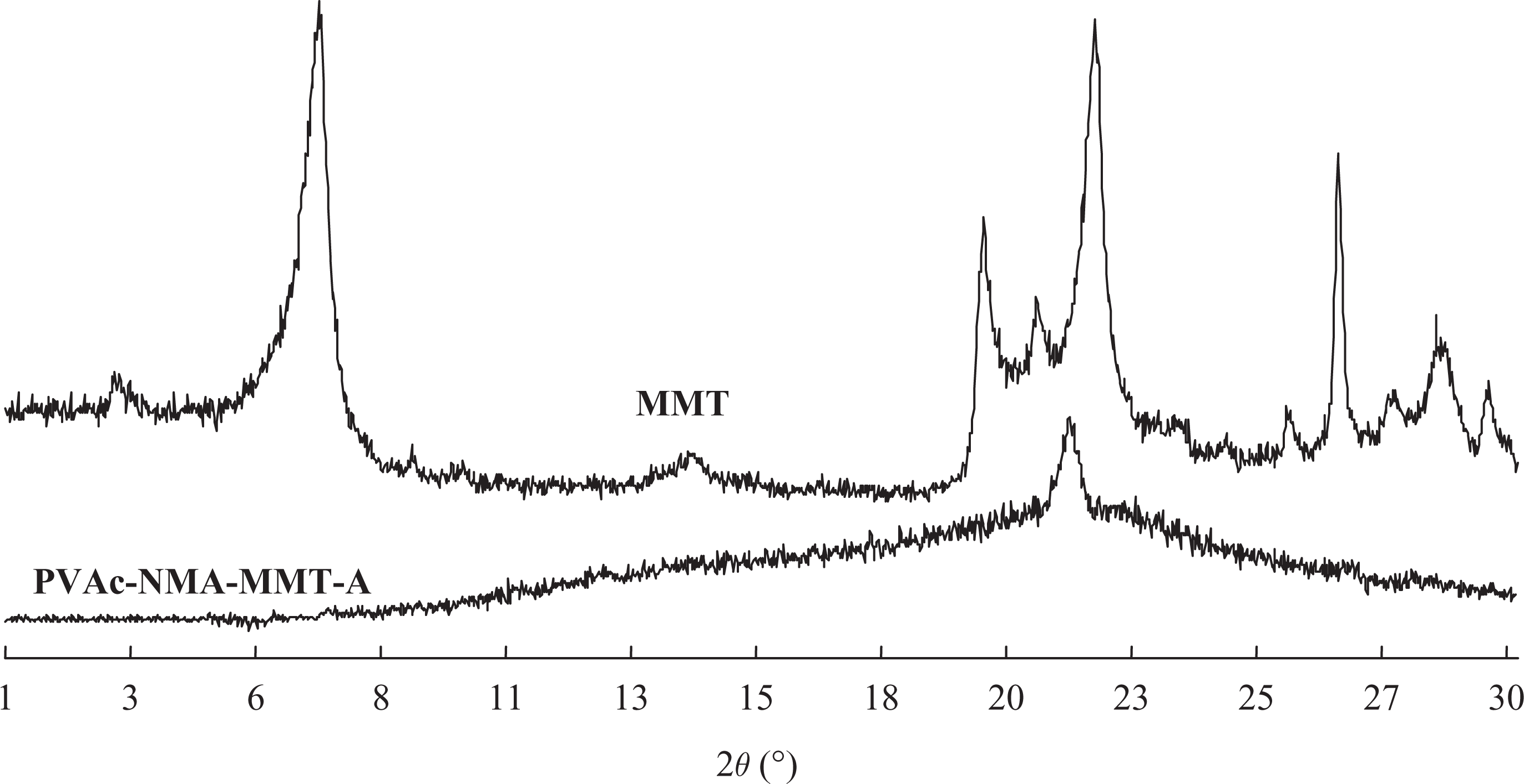
XRD patterns of MMT and PVAc–NMA–MMT. NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate; XRD: X-ray diffraction.
The interaction between PVAc and MMT or OMMT was physical adsorptions or secondary bonds. PVAc and MMT or OMMT were connected or absorbed together to form PVAc–MMT and PVAc–OMMT. 11 –20 The interaction among PVAc, NMA and MMT might be more complicated. Our original purpose was to make MMT to provide a nanoscale reaction surrounding for the main polymerization monomers of VAc and NMA, they reacted and polymerized with each other between MMT layers, and the copolymer of PVAc–NMA exfoliated MMT into nanoparticles. This was verified by XRD (Figure 1) and FTIR (Figure 2). As shown in Figure 2, the absorption peak of PVAc–NMA–MMT near 3000 cm–1 was weaker, even disappeared, that near 1600 cm–1 was a platform from PVAc’s originally slightly sharp absorption peak, and that near 1100 cm–1 was two sharp absorption peaks from PVAc’s originally wide absorption peaks. These differences showed that PVAc–NMA was indeed produced and also PVAc and NMA–NMA. 21,22 The grafted polymer of PVAc–PVA might also be produced according to El-Aasser et al. work. 25 –27 In that article, PVA was mainly used as the protective colloid. The polymerization was mainly occurred between the monomers of VAc and NMA. Even if PVAc–PVA was produced in the polymerization, it would not affect the polymers that we expected, at least it would not bring poor effect on their performances and properties. In other words, as we designed, the copolymer of PVAc–NMA was synthesized between MMT layers. It then had exfoliation on MMT. As PVAc–MMT and PVAc–OMMT, the interaction between PVAc–NMA and MMT also was physical adsorption or secondary bond. PVAc–NMA and MMT were connected or absorbed together to form exfoliated nanocomposites. Moreover, to be precisely because of these physical adsorptions or these secondary bonds, MMT could still keep its good stability in the relatively harsh emulsion intercalation process and guarantee its structural continuity and stability.
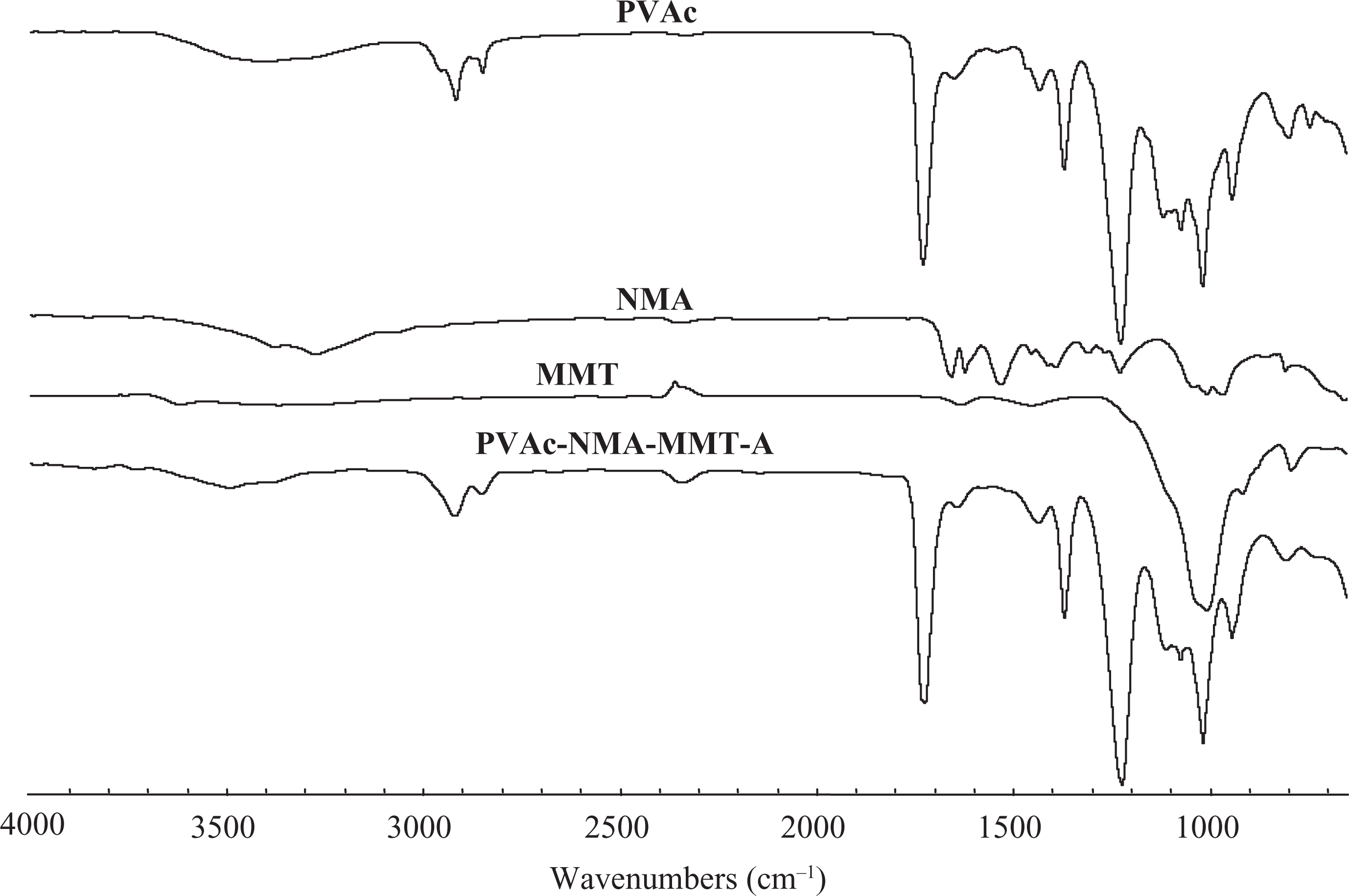
FTIR spectra of PVAc, NMA, MMT and PVAc–NMA–MMT. FTIR: Fourier transform infrared spectroscopy; NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
Dispersion
Dispersion or suspension is very important to emulsions because good dispersion often means long storage time, which has a great practical value on application. From the visual, all PVAc–NMA–MMT emulsions were similar to that of PVAc–MMT-A, PVAc–MMT-B, PVAc–MMT-C, PVAc–OMMT-A, PVAc–OMMT-B, PVAc–OMMT-C and all PVAc–NMA emulsions, 11 –22 they were viscous, milk-white, homogeneous, fine emulsions, and all had no coarse particles, no foreign bodies and no delamination. PVAc–MMT-D, PVAc–MMT-E, PVAc–OMMT-D and PVAc–MMT-E had delamination, separation and precipitate, however, PVAc–NMA–MMT-D and PVAc–NMA–MMT-E did not demonstrate these phenomena showing their dispersion was good. Moreover, when some were dropped in water, their dispersion was similar (Figure 3). Compared with water, no coarse particle was found in the dispersion of all PVAc–NMA–MMT emulsions, their dispersion was good.

Dispersion of PVAc–NMA–MMT in water. NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
To further study the dispersion, PVAc–NMA–MMT-A was chosen and observed by TEM (Figure 4). Under observation, the particles were very small. Their diameter was 50–100 nm. This differed from PVAc–MMT, PVAc–OMMT and PVAc–NMA. In PVAc–MMT and PVAc–OMMT, PVAc particles were larger, their diameter was 250–500 nm, and MMT particles and OMMT particles were smaller, their diameter was 50–100 nm and 25–100 nm, respectively. 11 –20 In PVAc–NMA, PVAc–NMA particles were also larger, their diameter was about 250 nm, and other smaller particles were the condensed polymer of NMA–NMA, their diameter was about 50–100 nm. 21,22 These differences showed that PVAc–NMA had much more significant exfoliation on MMT, and MMT also greatly affected PVAc–NMA. PVAc–NMA particles were much smaller similar with that of MMT particles, so it was difficult to distinguish them clearly. These particles all were round or similar round, and dispersed together randomly, as platelets in flat form, but not uniformly dispersed as described by Chien et al. 28,29 In addition, no interesting structure, such as ‘Core-Shell’ structure, ‘Strawberry’ structure, ‘Sandwich’ structure, ‘Swallow’ structure, ‘Tactoids’ structure and so on was found in PVAc–NMA–MMT that once appeared in PVAc–MMT and PVAc–OMMT. 11 –20 In fact, these interesting structures might exist but were very hard to be found out clearly. The dispersion was too random, irregular, disorderly and even messy. Maybe it is because of the formation of smaller PVAc–NMA particles and their random dispersion with MMT particles, PVAc–NMA–MMT had a good dispersion for application or storage.
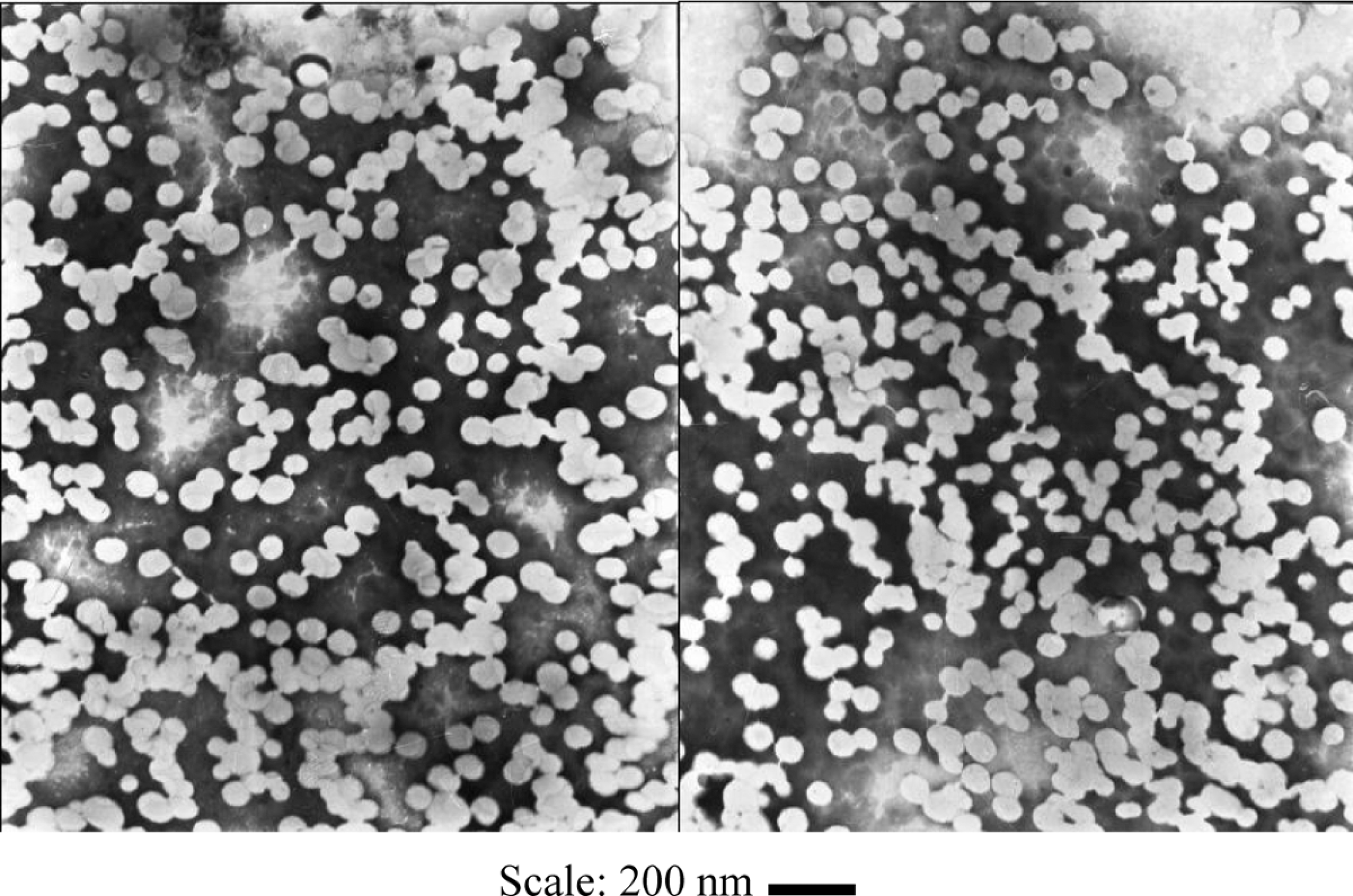
TEM images of PVAc–NMA–MMT. NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate; TEM: transmission electron microscopy.
Rheology
The apparent viscosity of PVAc–NMA–MMT (Table 1) varied with the different synthesis precursors. From precursor A to E, the apparent viscosity increased. The different synthesis precursors made the great differences on the apparent viscosity. As described above, PVAc–NMA–MMT was synthesized in different weight of water, respectively, was 250 g, 200 g, 150 g, 100 g and 50 g, and with different polymerization time, respectively, was 6 h, 5 h, 4 h, 3 h and 2 h. These would affect the microstructure of final products significantly, so the apparent viscosity was different.
Apparent viscosity.
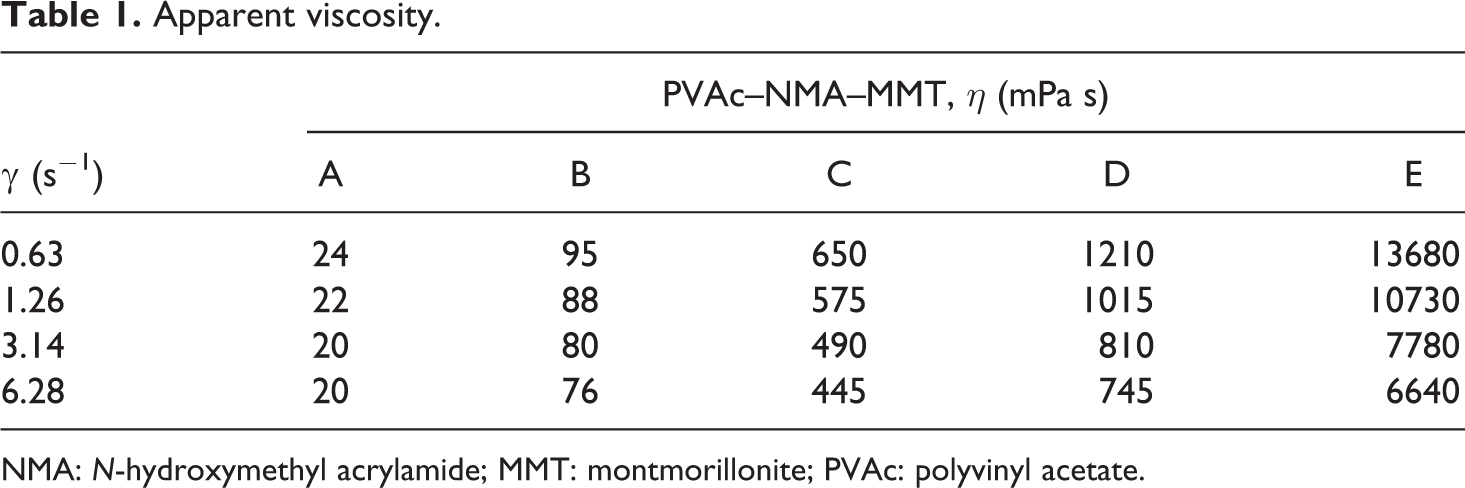
NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
Compared with PVAc, the apparent viscosity of PVAc–MMT, PVAc–OMMT and PVAc–NMA was higher 11 –22 and that of PVAc–NMA–MMT was much higher than all of them. NMA and MMT both were integrated to contribute to this. On one hand, NMA was added into the polymerization, NMA–NMA and PVAc–NMA were produced, and the intermolecular and intramolecular hydrogen bond-driven self-assembly 30 –34 might appear in PVAc–NMA. On the other hand, (i) there is an immobilized hydrodynamic layer for polymer on the surface of MMT layer; its thickness and the gyration radius of polymer are in the same magnitude order. According to the scaling theory, if the relaxation time of restricted molecular chain is much longer than its bulk molecular chain, and its thickness roughly equals to the gyration radius of macromolecular chain, the viscosity of restricted polymer or melt can increase by several times. Moreover, (ii) MMT plays a role of physical cross-linking points in the composite, so some similar cross-linking structure will also exist there. 35 The friction between anisotropic MMT layers in the composite will also make the viscosity increase. 36 When MMT content reaches a certain amount, this role will get very obvious that the viscosity depends on the MMT content greatly.
Moreover, the results also show that η decreased with the increase of the shear rate. This phenomenon indicated that PVAc–NMA–MMT was a pseudo-plastic non-Newtonian fluid. Its characteristic is that the apparent viscosity decreases with the increase of shear rate. The orientation of slender molecular chains for a polymer in the flow direction causes the decrease of viscosity, so it is also often called the shear thinning fluid. This rapid shear thinning behavior of PVAc–NMA–MMT was much greater due to the close contact between polymer and silicate layers that changed the relaxation dynamics of polymer chains, 37 –43 the intermolecular and intramolecular hydrogen bond-driven self-assembly 30 –34 and the role of plasticization from small molecular intercalation agents. This would facilitate materials processing and application.
Based on the apparent viscosity, the rheology of PVAc–NMA–MMT was further studied by the power–law function equation, Newtonian fluid flow equation and Cross–Williamson model viscous equation. The fluid consistency (Ï, mPa s), flow index (i), zero shear viscosity (η0, mPa s), limit viscosity (η∞, mPa s), characteristic time (ζ, s), average molecular weight (M w) and number average molecular weight (M n) were obtained (Table 2). These i of PVAc–NMA–MMT all were less than 1, so it was a pseudo-plastic non-Newtonian fluid that coincided with the results of shear thinning fluids obtained from the apparent viscosity. Ï, η0, M w and M n increased from precursor A to E and those of PVAc–NMA–MMT all were much higher. Their trends were similar to the apparent viscosity. η∞ was 0 mPa s at all the time. ζ is a characteristic constant for materials, always expressed by time, mainly by s. Its variation trend of PVAc–NMA–MMT increased from precursor A to C, then decreased to precursor D, and increased to precursor E again, not similar to PVAc, PVAc–MMT, PVAc–OMMT and PVAc–NMA, 11 –22 because they were different polymers or nanocomposites.
Rheological results.
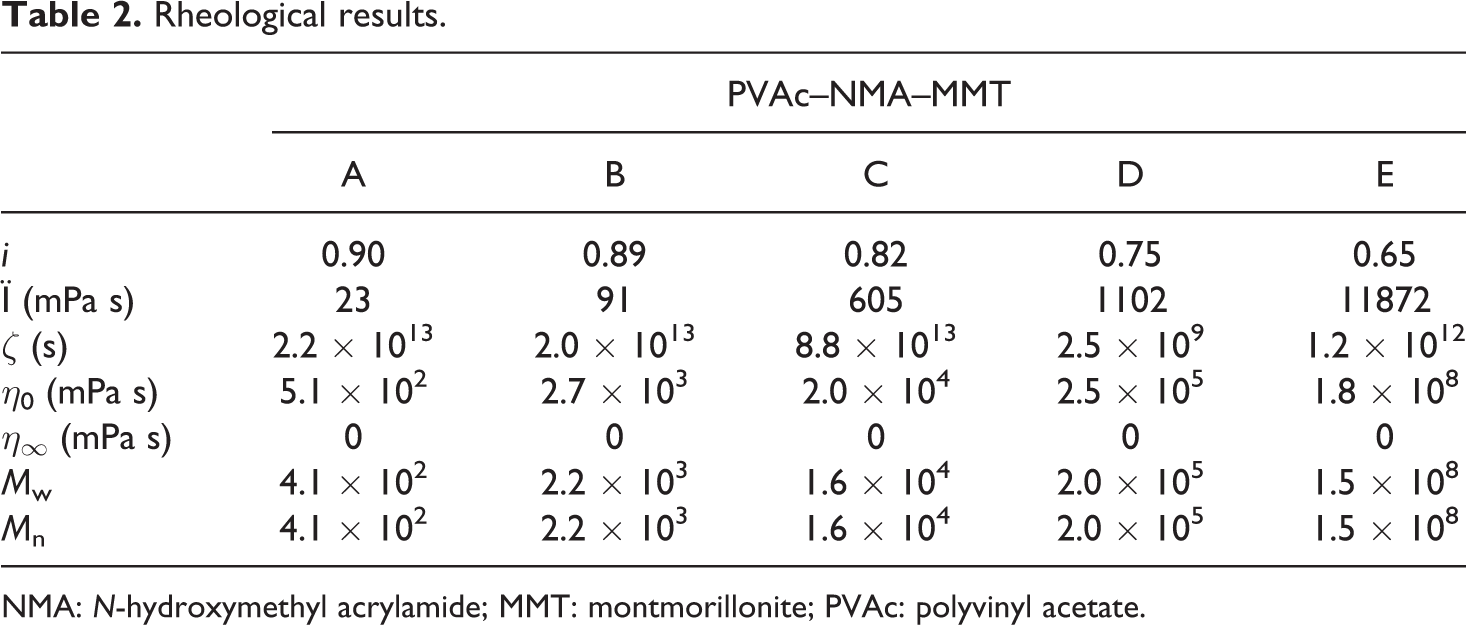
NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
From precursor A to E, the molecular weight increased, and PVAc–NMA-MMT was much higher than PVAc, PVAc–MMT, PVAc–OMMT and PVAc–NMA. 11 –22 In the synthesis, MMT had various effects on the emulsion polymerization. On one hand, MMT absorbed emulsifier molecules, then reduced the latex particles in the system, promoted the decomposition rate of initiator molecules, and thereby decreased the molecular weight of polymer. On the other hand, in the emulsion polymerization, MMT layers dispersed in the polymerization system and made the viscosity increase; they blocked the proliferation of free radicals, even made them lose activity, so the molecular weight of polymer increased. When MMT content was less in the system, the former factor might be the dominant factor, the molecular weight was low. When MMT content was more in the system, the latter factor dominated, so the molecular weight was high. Therefore, as in our synthesis precursors, MMT was 2% of VAc and it was a reasonable addition. From precursor A to E, the water weight in the reaction decreased and was 250 g, 200 g, 150 g, 100 g and 50 g, respectively. In other words, with the decrease of water in the synthesis from precursor A to E, MMT content increased in the synthesis system, so their molecular weight increased. Meanwhile, with the increase of MMT, NMA content also increased in the synthesis system, so much more grafting were carried out between VAc monomers, NMA monomers or between VAc and NMA, and much more macromolecular chains were produced. As same as other polymers, PVAc–NMA–MMT also possessed the normal stress effect that is the pole-climbing phenomenon, also called as the Weissenberg effect.
Static tensile
In the static tensile test, the results obtained only were the maximum break force of PVAc–NMA–MMT films. Many other static tensile data, including the static tensile strength (σ, MPa), break strain (∊, mm mm−1), vertical strain (∊ x , mm mm−1), horizontal strain (∊ y , mm mm−1), break elongation (∊t, %), elastic modulus (E', MPa) and Poisson’s ratio (ι), would be calculated from the maximum break force through Hooke’s law and Poisson’s ratio equation. As shown in Table 3, PVAc–NMA–MMT’s σ was from 6.47 MPa (PVAc–NMA–MMT-E) to 6.85 MPa (PVAc–NMA–MMT-D), much higher than PVAc, PVAc–MMT, PVAc–OMMT and PVAc–NMA whose maximum σ was 3.11 MPa, 3.49 MPa, 6.45 MPa and 4.33 MPa, respectively. 11 –22 NMA and MMT improved σ greatly, and it seemed that the different synthesis precursors had not very significant influence on this, neither others. Its ∊(∊ x ), ∊ y , ∊t, E' and ι were little affected by the different synthesis precursors, but however, the static tensile of PVAc–NMA–MMT was much better, its σ was higher, its ∊t was greater, its E' was larger and its ι was more reasonable.
Static tensile.
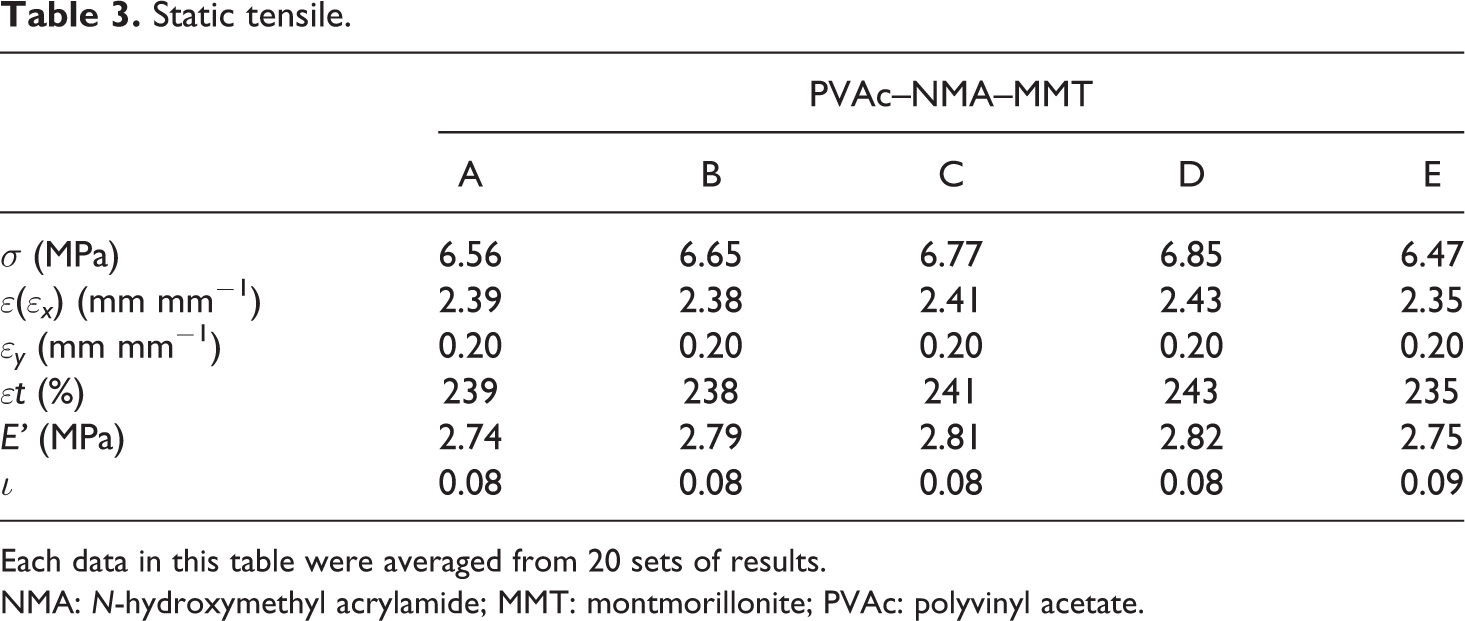
Each data in this table were averaged from 20 sets of results.
NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
PVAc–NMA–MMT as an exfoliated nanocomposite was prepared successfully, and MMT played an important role in strengthening and toughening of the static tensile. First, MMT provided a nanoscale reaction surrounding for the main polymerization monomers of VAc and NMA. It could be considered to be a special reactor, a nanoreactor, a reaction mesoscopic surrounding modulated by nanoscale that was embodied as the reaction medium, the reaction carrier, the reaction interface and so on. 44 In PVAc–NMA–MMT, as a nanoreactor, MMT provided a special nanoscale surrounding and limited the reaction between VAc and NMA only happening in the nanometer range. The randomly dispersed nanoscale layers or sheets of MMT blocked the movement of PVAc–NMA chains effectively and affected the interaction between these chains, so strengthened and toughened the static tensile. Second, the interlayer effect of inner and outer for MMT layers and MMT as physical cross-linking points also influenced the strengthening and toughening. On one hand, the interlayer had special effect. PVAc–NMA chains were linked directly with the inner and outer surface of MMT layers together in the form of physical absorptions. 45 The combined regions all were the square of nanoscale, so their set effect was very significant. On the other hand, in the nanocomposites, the dispersed layered silicates in polymer matrix always worked as the physical cross-linking points. They could be screwed or anchored together with polymer chains that also strengthened and toughened the static tensile. Finally, in the nanocomposite, the better the dispersion of silicate layers in the polymer matrix, the greater the combination probability between them, and the more the physical cross-linking points, so thus the static tensile would be strengthened and toughened more significantly. 46,47 This effect would be much more obvious especially for exfoliated nanocomposites, as the synthesized PVAc–NMA–MMT, it precisely was an exfoliated nanocomposite.
Besides these, PVAc–NMA–MMT contained the cross-linkable monomer of NMA, which had the active functional group of hydroxymethyl and the double bonds conjugated with the carbonyl group. Under heat or in an acidic condition, NMA would play a cross-linking role, made the molecules be further cross-linked, so the thermosetting polymer of PVAc–NMA was generated that could improve the films’ tensile strength and the adhesion to the bonded substrate to a great extent. In PVAc–NMA–MMT, the system itself had the weak acid surrounding, and/or by the help of heat during curing or hardening, this self-cross-linking reaction occurred naturally. The intermolecular and intramolecular hydrogen bond-driven self-assembly in PVAc–NMA–MMT also had helpful contribution to the static tensile.
The solid content of PVAc–NMA–MMT was higher than PVAc, PVAc–MMT, PVAc–OMMT and PVAc–NMA 11 –22 shown in Table 4. From precursor A to E, O gradually increased, especially precursor D and E, their O was much higher, so they would be helpful for the development of high solid content emulsions. S for PVAc–NMA–MMT was good all at more than 180 days, which showed the storage stability was very excellent due to its good dispersion described above.
Solid content and storage time.

NMA: N-hydroxymethyl acrylamide; MMT: montmorillonite; PVAc: polyvinyl acetate.
Conclusions
Compared with PVAc–MMT, PVAc–OMMT and PVAc–NMA, PVAc–NMA–MMT developed their strongpoint and avoided their shortcoming, its dispersion was better, and its static tensile was much higher, up to 6.47–6.85 MPa. MMT was exfoliated into layers or sheets of nanoparticles, dispersed together with PVAc–NMA particles randomly. PVAc–NMA–MMT was a pseudo-plastic non-Newtonian fluid, and with the change of different synthesis precursors and the increase of NMA content and MMT content in the synthesis system, the molecular weight of PVAc–NMA–MMT increased. PVAc–NMA–MMT had good storage stability and high static tensile, its synthesis method was easy and simple, could be used to modify PVAc.
Footnotes
Funding
The authors are grateful for the financial support of the National Nature Science Foundation of China (30930074).

