Abstract
The aim of this work was to study the effect of surface-modified calcium carbonate (CaCO3) nanoparticles in the mechanical properties and crystallization behavior of polypropylene (PP) homopolymer. Pimelic acid (Pa) was used as a surface modifier for nano-CaCO3 (nCC). Three compositions of PP/nCC composites were prepared in a corotational twin-screw extruder machine with nCC content of 5, 10, 15, and 20 wt%. The results of scanning electron microscopy showed that the Pa treatment enhanced the interfacial adhesion between the filler and the matrix, indicating the improvement in the compatibility between PP and nCC. The measurements of the mechanical properties showed that the elastic modulus (
Keywords
Introduction
Polymer nanocomposites are a new class of particle-filled polymer composites in which at least one dimension of the dispersed particles is in the nanometer range. Polymer nanocomposites have attracted increasing attention in recent years because of their significant improvement in physical and/or chemical properties over the matrix polymers. 1–3 It is well known that nanoparticles play an important role in their application to polymer nanocomposites. Polymer-based nanocomposites have been extensively studied in research and application because the addition of the nanoparticles provides an increased stiffness, toughness, and dimensional stability. The aggregation of the nanoparticles, however, owing to their high surface energy, strongly decreases the properties of the nanocomposites, which enormously limits their engineering applications. Many studies indicated that the excellent properties of nanocomposites distinctly depend on the particle dispersion which has close relationship with the manufacturing method. 4,5 Many manufacturing methods, such as in situ polymerization, 6 intercalation method, 7 sol–gel method, 8 and melt compounding, 9 have been used to prepare the composites. In all these methods, melt compounding has been regarded as a prevalent means for reasons of economy, practicability, and environment friendliness.
Polypropylene (PP) is one of the most widely used commercial semi-crystalline polymers. The main attraction of PP is its high-performance-to-cost ratio. However, its utilization has been limited because of its poor impact strength (
Experimental
Materials
The grade of PP used in this work was SEETEC homopolymer PP (LG Chem, South Korea). This acts as the matrix. The homopolymer PP has a density of 0.90 g/cm3 and a melt flow rate of 14 g/10 min (2.16 kg at 230°C). We were interested in the effect of surface modification of filler particles on the dispersion and properties of compounds; we studied both unmodified nCC and nCC surface modified by a Pa (analytical reagent and identified as Pa), both kindly supplied by SK Corporation (South Korea). Ethanol (analytical reagent) (Hyo Sung Co. Anyang, South Korea) was commercially available and used as received. CaCO3 nanoparticles were supplied by Shiraishi Kogyo Kaisha Ltd (Japan) and had a density of 2.7 g/cm3 and a diameter of 60–80 nm.
Sample preparation
Surface treatment of CaCO3 nanoparticles
Unmodified nCC was prepared by drying its water suspension in an oven at 100°C to constant weight. Surface treatment of nCC was as follows: 150 g nCC was put into a 500-ml three-necked flask equipped with a mechanical stirrer. Subsequently, a solution of 1.5 g Pa (1% Pa) or 4.5 g Pa (3% Pa) was added to 250 ml ethanol. The mixture was reacted for 1 h at 80°C under vigorous stirring. Then the ethanol was distilled out and the Pa-treated nCC (nCC-Pa) was dried to constant weight at 90°C.
Preparation of PP/nCC-Pa composites
Before melt mixing, PP- and surface-treated CaCO3 nanoparticles were dried in a vacuum oven at 80°C for 6 h and then cooled down to room temperature. The materials were stored in a desiccator prior to processing. nCC, in four different concentrations (5, 10, 15, and 20 wt%), was added to PP to produce binary composites. All the materials were simultaneously added into the extruder after previous mixing. Blending with different filler concentrations was carried out in a twin-screw extruder (Brabender plasticorder, model: PLE-331). The capacity of the mixing chamber being 30 ml. The processing temperature, rotor speed, and blending time were set at 180°C, 60 r/min, and 10 min, respectively. The sample weight of each blending was controlled at 60 g. After 10 min, the mixing chamber of the Brabender apparatus was opened and the resulting mixing was taken out. The resultant mixture was compression-molded in a hot press at 190°C for 5 min between two steel plates under a pressure of 7 MPa. Finally, the pressure was released and the mold removed from the plates. This was followed by cooling to room temperature between two thick-metal blocks kept at room temperature. A template frame was used to ensure a constant film thickness (1 mm). The samples were cut into standard shapes and sizes (according to the ASTM D-638-91 standard) for testing the mechanical properties. The specimens were then sealed in plastic bags as they waited the processing and analysis. There were three composites, PP/nCC, PP/nCC-1Pa, and PP/nCC-3Pa, in this experiment, and the Pa concentrations were 0, 1, and 3 wt%, respectively. The concentration of PP was 100 wt%, when the concentration of nCC was 0%.
Mechanical properties measurements
Mechanical properties of virgin PP and PP/nCC nanocomposites were studied in both tensile and impact tests. Standard specimens were sampled from the compression molded sheet and then conditioned at a temperature of 25 ± 2°C and the relative humidity of 50 ± 5% for 24 h. Tensile testing was performed on a screw-driven universal testing machine (Instron 4466) equipped with a 10 kN electronic load cell and mechanical grips. The tests were conducted at a crosshead speed of 30 mm/min, and data were acquired by a computer. All tests were carried out according to the ASTM D638-91 standard, and five replicates were tested for each sample to get an average value. Izod impact tests of notched samples were carried out according to ASTM D256-93a standard, the instruments was Ceast pendulum impact tester (Model 6545/000). The dimension of the specimens (length × width × thickness) was 63.50 × 13 × 3.20 mm3.
Morphological observation
Scanning electron microscopy (SEM; JEOL, Japan JSM-6360LV) was employed to study the fracture surfaces of all tested samples. The SEM samples were held in liquid nitrogen for 30 min and then broken into two pieces. The fractured surfaces of the specimen were coated with a thin layer (10–20 nm) of gold palladium.
DSC measurements
The melting and crystalline behaviors of virgin PP and samples were measured with a Perkin Elmer DSC-7 (Perkin Elmer, Inc, Wellesley, Massachusetts, USA). First, virgin PP and samples were heated from 25 to 250°C for 10 min to eliminate all of the thermal history in the materials. Then, the samples were cooled to 25°C at a cooling rate (
Results and discussion
Mechanical properties and dispersion of nanoparticles in PP matrix
CaCO3 nanoparticles were incorporated to PP homopolymer in order to achieve better
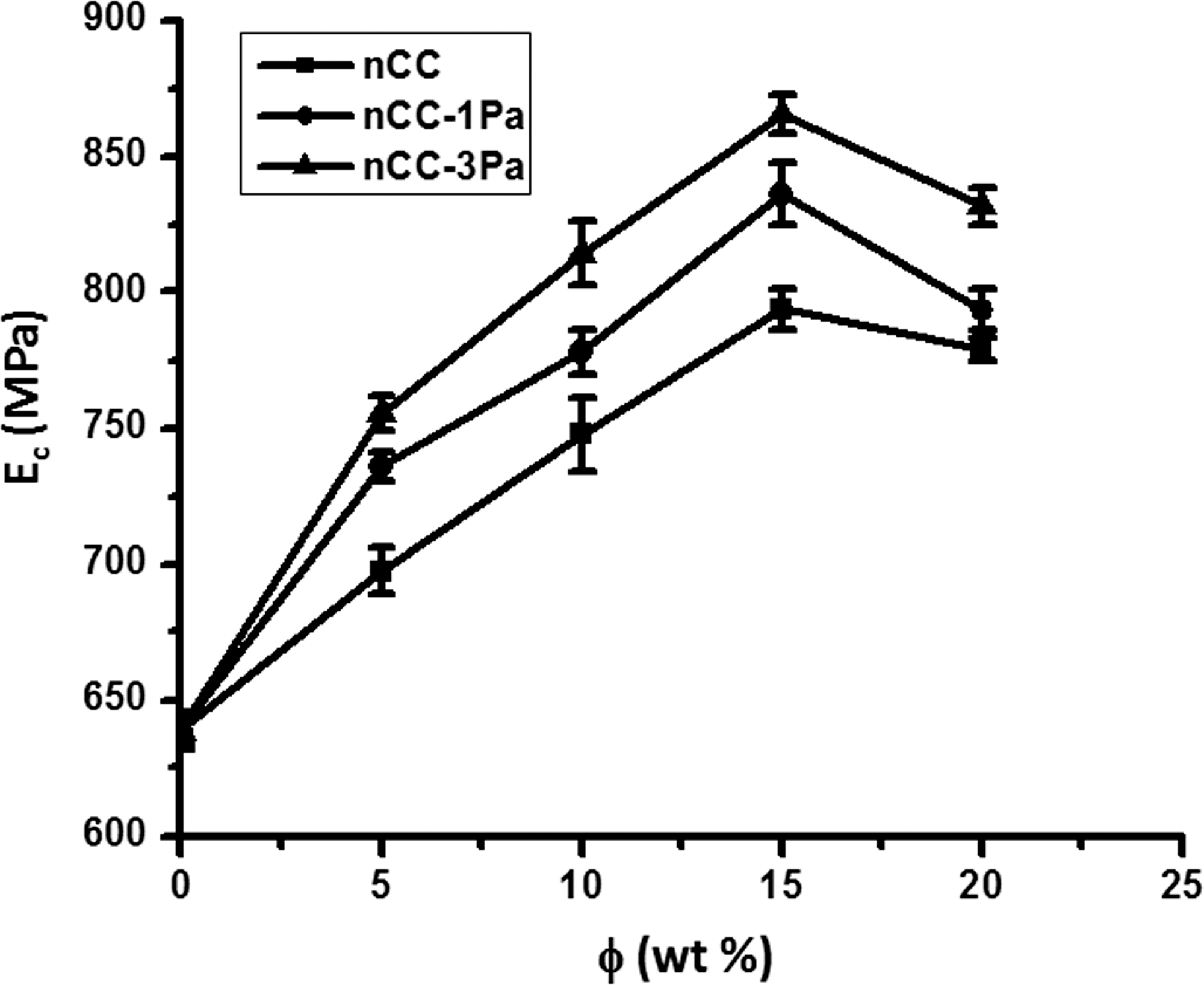
Effects of the filler content (φ, wt%) on the elastic modulus (
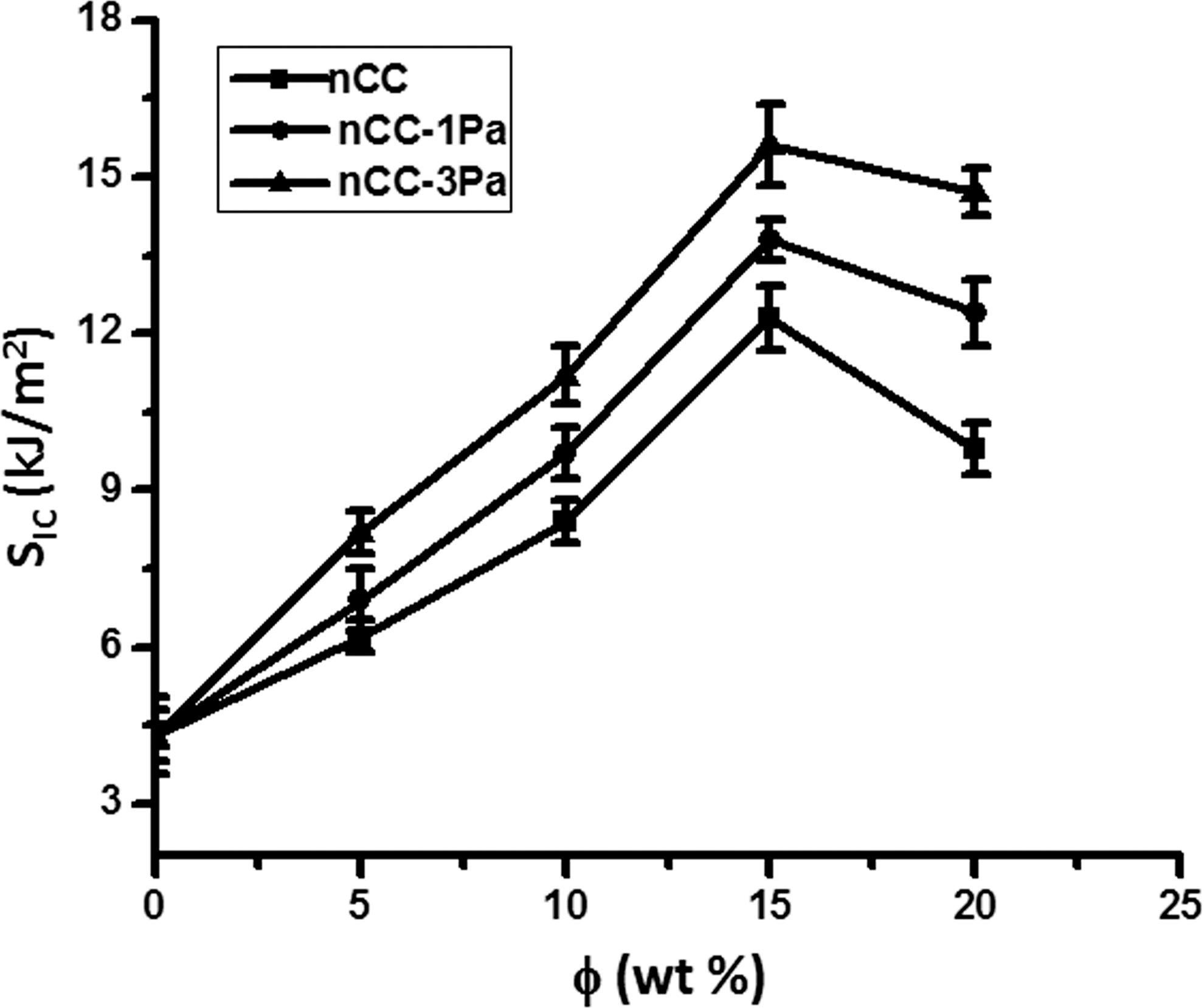
Effects of the filler content on the impact strength (
Figure 3 illustrates the correlation between the tensile yield stress (σyc) and the content of nCC of the three nanocomposites. It can be seen that when φ < 5%, the σyc of all three composites increases slightly with increasing φ. For φ > 5%, σyc decreases gently, but PP/nCC-3Pa and PP/nCC-1Pa were higher than that of PP/nCC. The maximum appears at φ = 5%. It means that the σyc of PP can be enhanced to some extent, when PP is filled with suitable content of nCC. The decrease in the σyc of the obtained composite at higher filler content (φ > 5%) was attributed to the agglomeration of the nCC particle’s weak interfacial adhesion that existed between the PP matrix and the nanoparticles, and hence the load-bearing capacity of cross-sectional area of composites decreased, and only a small amount of stress could be transferred from the matrix to inorganic particles; hence, the tensile stress showed decrement in the magnitude; in this case, agglomerated particles easily debonded from the matrix and could not bear any fraction of external load ultimately decreasing the tensile stress. These results are consistent with the results of the research study carried out by by Li et al. 24 In contrast, outstanding effectiveness of nCC-Pa increasing the Tensile strength (TS) of PP composites was demonstrated. As the concentration of the filler increased in the composites filled with nCC-Pa, the TS was first remarkably increased and then decreased. When the concentration of the filler was 5 wt%, the maximum TS found was 36.1 MPa for nCC-1Pa and 36.4 MPa for nCC-3Pa, which was 2 and 3% higher than that of the unmodified composite, respectively. Altogether, the mechanical properties of the three composites were as follows: PP/nCC-3Pa > PP/nCC-1Pa > PP/nCC, a composite with good mechanical properties could be achieved when the filler loading was 15 wt%.
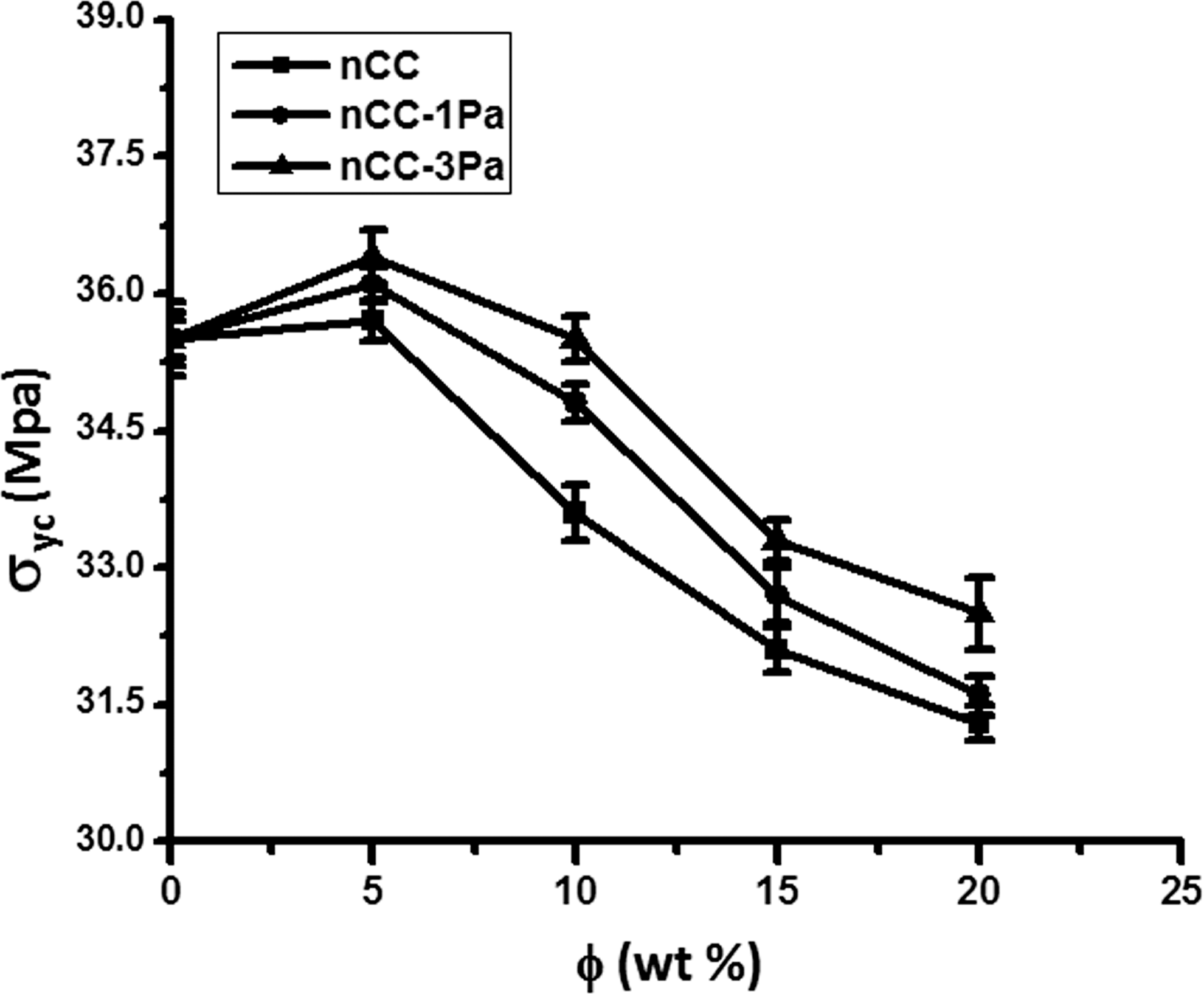
Effects of the filler content on the tensile yield stress (σyc) of PP/nCC, PP/nCC-1Pa, and PP/nCC-3Pa composites. PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
Figure 4(a) to (c) presents SEM micrographs of the impact-fracture surface of PP/nCC, PP/nCC-1Pa, and PP/nCC-3Pa nanocomposites, respectively, with 10 wt% filler. The particles of nCC in Figure 4(a) were dispersed in the matrix of PP irregularly, and some big aggregates were exposed on the fracture surface, with a particle size greater than 3 μm. By observing these aggregations, we found that they were formed of many primary particles. The PP/nCC sample had a large particle size of 3–4 μm. From an analysis of the mechanical properties, we determined that this composite exhibited impact energy only slightly higher than that of virgin PP and much lower than that of the PP/nCC-3Pa composites. Figure 4(b) and (c) presents photographs of the impact fracture surface of PP/nCC-1Pa and PP/nCC-3Pa composites. In PP/nCC-1Pa, there were no aggregates and the average particle size was smaller than that of nCC. When the amount of the Pa covering the nCC particles increased, the dispersion of the nCC-3Pa particles in the PP matrix became more uniform, as Figure 4(c) demonstrates. Most nCC-3Pa particles were under 80 nm, and the combination of nCC-3Pa particles with the PP matrix was very close; this was the main reason for the high toughness.

Scanning electron micrographs of composites (10 wt% filler): (a) PP/nCC, (b) PP/nCC-1Pa, and (c) PP/nCC-3Pa. PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
Melting and crystallization behavior of PP and its nanocomposites
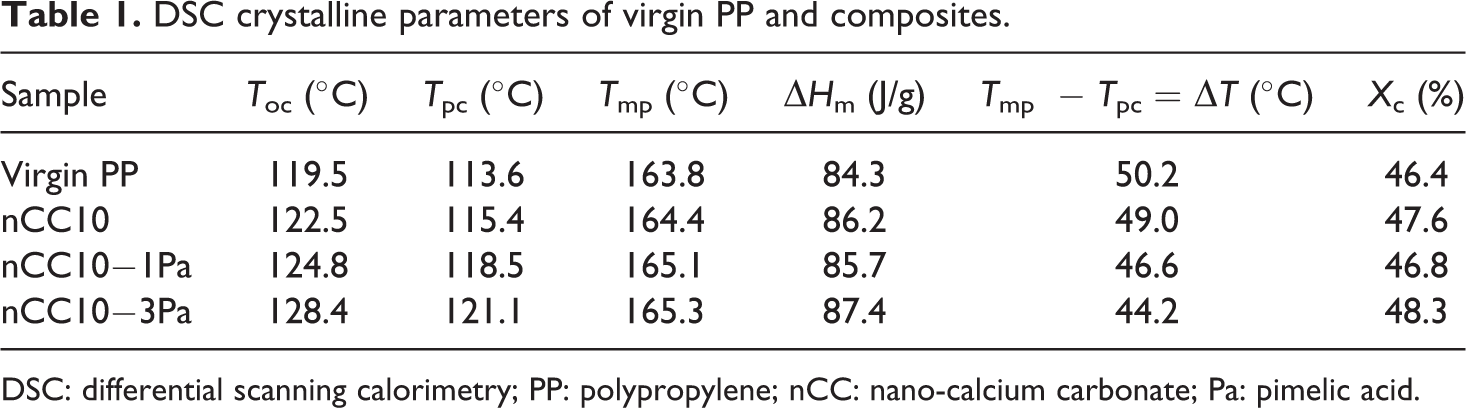
Figure 5 shows the nonisothermal crystallization curves of PP and PP/10 wt% nCC (nCC10), PP/nCC10-1Pa (nCC10-1Pa), and PP/nCC10-3Pa (nCC10-3Pa) composites at the cooling rate of 10°C/min. From these curves, it can be seen that the addition of nCC10, nCC10-1Pa, and nCC10-3Pa fillers significantly change the crystallization behavior of PP. It is found that the crystallization peak of the specimens shifts to a high temperature, and the movement of nCC10-3Pa was the most obvious. Table 1 represents the differential scanning calorimetry (DSC) data for nonisothermal crystallization and melting.
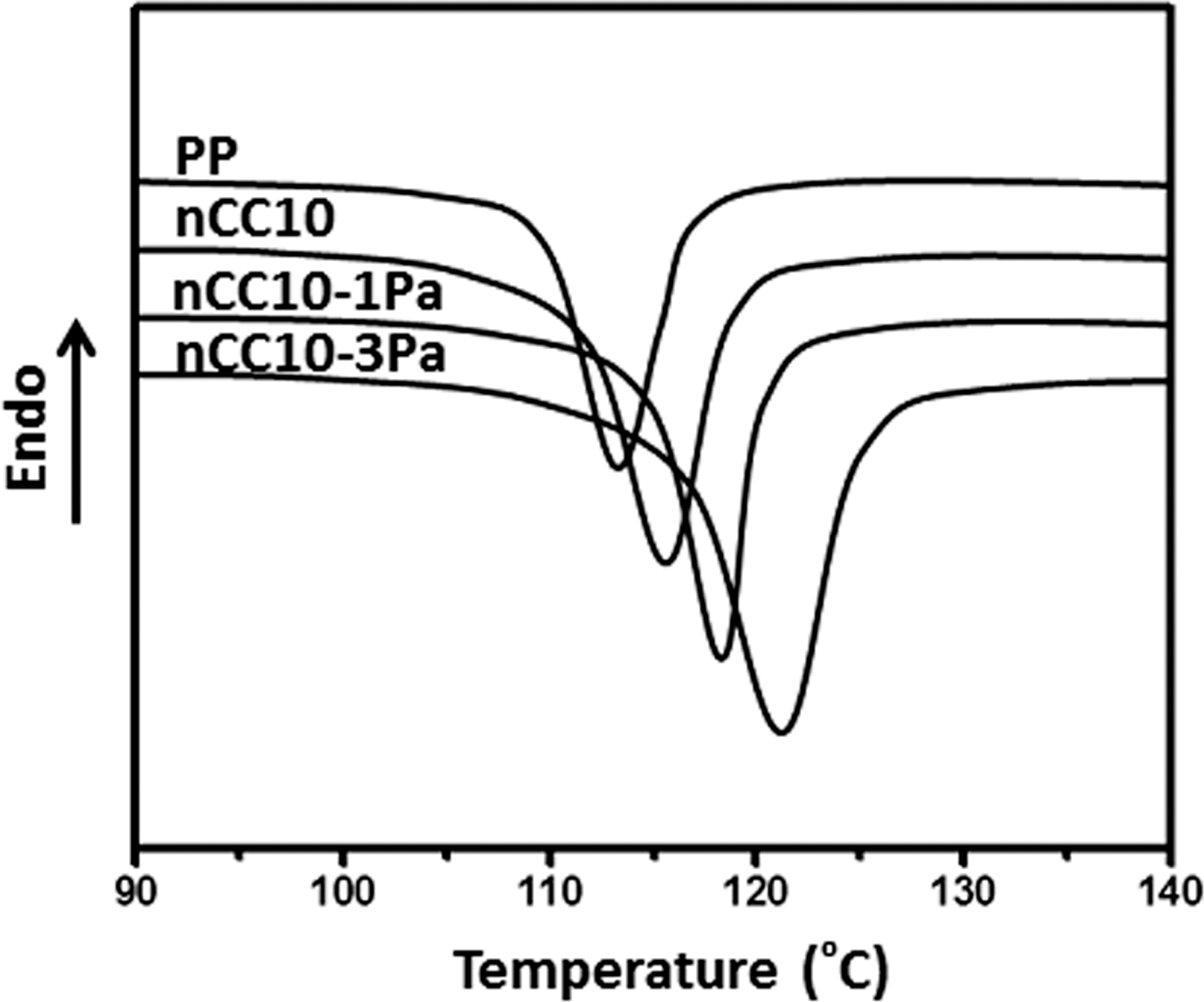
DSC crystallization curves of virgin PP, PP/10 wt% nCC (nCC10), nCC10-1Pa, and nCC10-3Pa composites. DSC: differential scanning calorimetry; PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
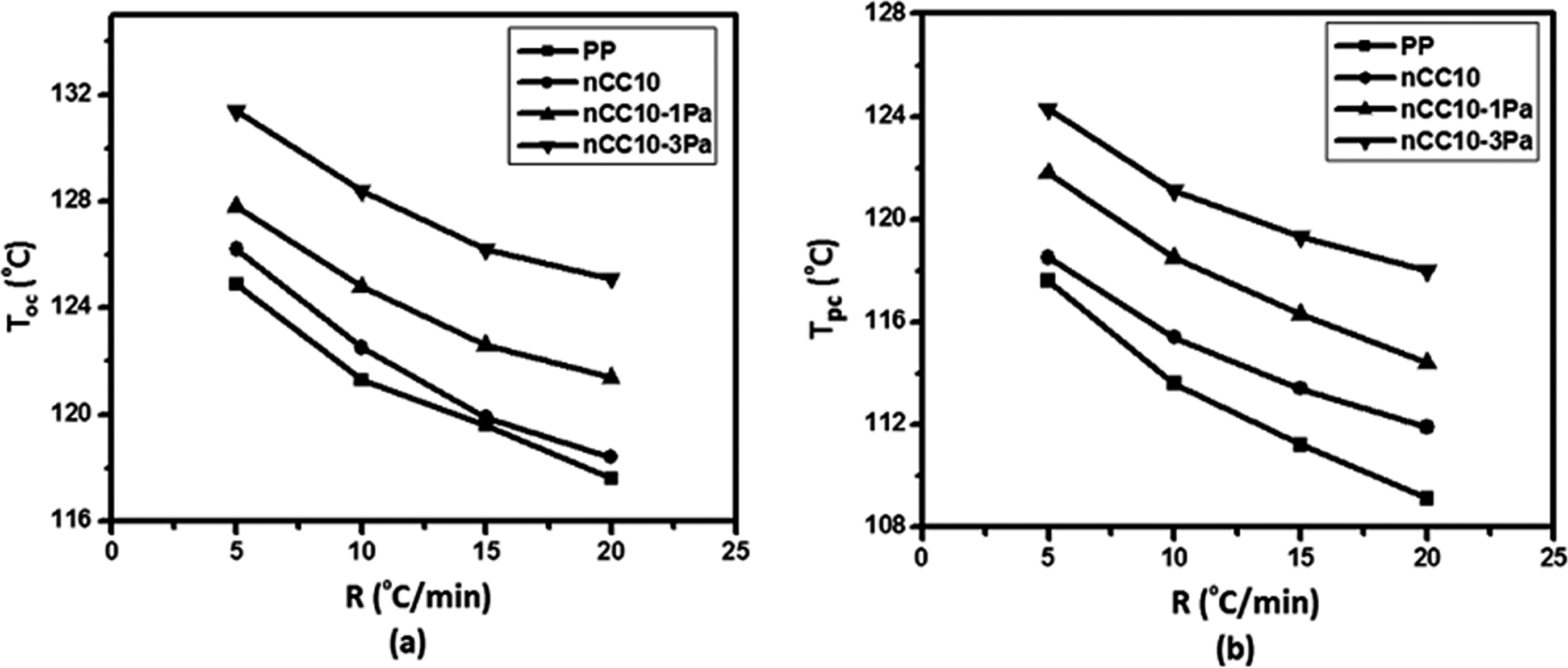
(a) The onset crystallization temperatures (
DSC crystalline parameters of virgin PP and composites.
DSC: differential scanning calorimetry; PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
After the process of crystallization, the samples were reheated. The DSC heating curves of PP and nanocomposites are displayed in Figure 7. It indicates that there are nearly no changes in the shape of curves for virgin PP and nanocomposites. Both virgin PP and nanocomposites seem to have the same value of melting temperature ( DSC melt curves of virgin PP, nCC10, nCC10-1Pa, and nCC10-3Pa composites. DSC: differential scanning calorimetry; PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.

Figure 8 shows the DSC nonisothermal crystallization curves of nCC-3Pa with different filler contents. These curves imply that the crystallization temperature increased with increasing concentration of nCC-3Pa. DSC crystalline parameters of nCC10-3Pa with different filler contents are listed in Table 2. The data demonstrate that the concentration of the nCC10-3Pa fillers had some influence on PP crystallites; the crystallization temperature increased by 5.5, 7.5, 10.1, and 13.3°C when the filler concentration was 5, 10, 15, and 20 wt%, respectively. Additionally, the degree of crystallinity of PP was increased by the addition of the nCC-3Pa filler. The minimum
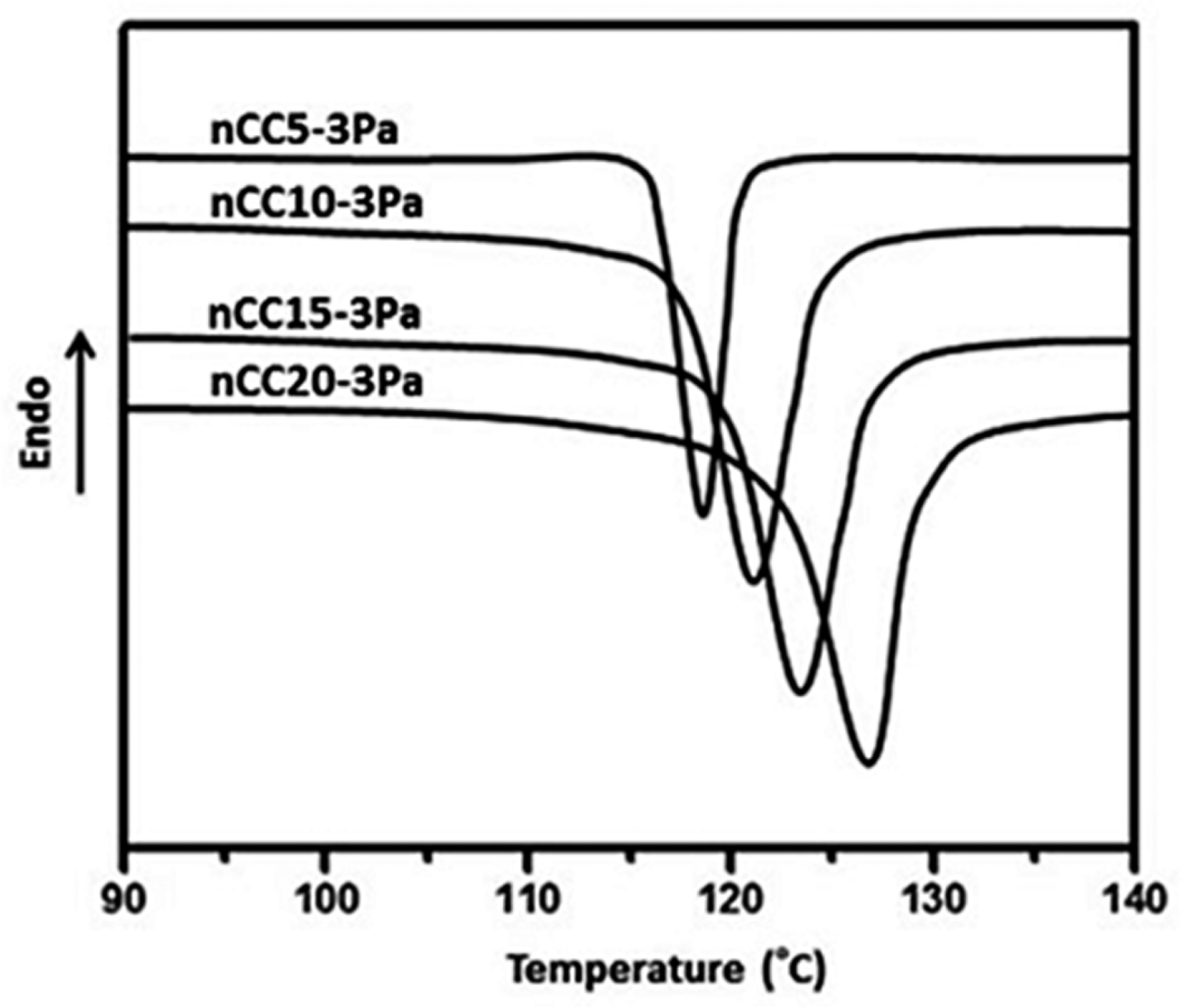
DSC crystalline curves of PP/nCC-3Pa with different filler contents: (1) 5, (2) 10, (3) 15, and (4) 20 wt%. DSC: differential scanning calorimetry; PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
DSC crystalline parameters of PP/nCC-3Pa (nCC-3Pa) with different filler contents.
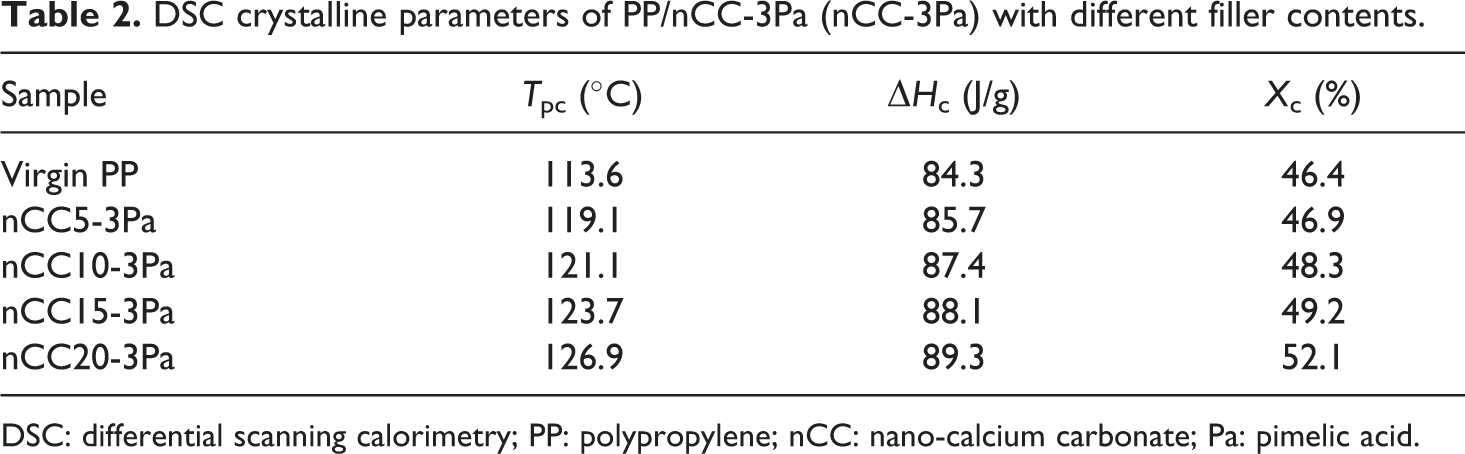
DSC: differential scanning calorimetry; PP: polypropylene; nCC: nano-calcium carbonate; Pa: pimelic acid.
Conclusions
From the above discussion, we can gain the following conclusions:
In this work, PP/nCC, PP/nCC-1Pa, and PP/nCC-3Pa nanocomposites were prepared by melt blending. The
When φ < 15%, the
When φ < 5%, the σyc increases with addition of φ and then decreases gradually. It appears that the maximum σyc occurs at φ = 5% in the range from 0 to 20%.
Unlike untreated nCC, SEM micrographs show a uniform size distribution for nCC-Pa, which dispersed well in PP. After surface modification by the Pa, the adhesion between the filler particles and PP matrix was enhanced and the aggregation could be avoided.
DSC measurements show that, in comparison with virgin PP and PP/nCC, the addition of the nCC-Pa fillers led to a higher crystallization temperature, and nucleation and crystallization improved at the same time.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
