Abstract
Coir yarns were cured with acrylic acid (AA) and phosphoric acid (PA) using ultraviolet (UV) radiation. Concentration of AA and PA, soaking time and intensity of UV radiation were optimized based on polymer loading (PL). It was found that the formulation containing 4% PA, 30% AA, 64% methanol and 2% photoinitiator showed maximum 35.13% of PL. The grafted yarns were soaked in CuSO4 solution for 30–180 min at different conditions. After withdrawing the yarns, the remaining copper in the solution were measured by atomic absorption spectrophotometer. It was revealed that copper was successfully removed using the grafted coir yarns.
Introduction
Coir fiber is a natural fiber and is obtained from the outer shell or husk of the coconut, the fruit of Cocus nucifera, a tropical plant of the Arecaceae (palmae) family. These fibers are used to make brushes, floor mats, heavy cord, coarse nets, etc. It contains 36–43% cellulose and allied substances comprising 41–45% hemicellulose, 0.15–0.25% lignin and 3–4% pectin. The cell length is about 0.8 mm, spiral angle of 41°–45° having a tensile strength of 131–175 MPa, elongation at break of 15–40% and Young’s modulus of 4–6 GPa, which is hydrophilic glucan polymer consisting of linear chain of 1,4-β bonded anhydroglucose unit that contains alcoholic hydroxyl groups. 1
Many industries, including mining and electroplating, discharge aqueous effluents containing relatively high levels of heavy metals, for example, copper, uranium, cadmium and mercury. Untreated effluents from these manufacturing processes have an adverse impact on the environment. 2 –4 Heavy metals released into the environment have been increasing continuously as a result of industrial activities and technological development, posing a significant threat to the environment and public health because of their toxicity, accumulation in the food chain and persistence in nature. However, it has been proven that large amounts of many heavy metals, such as copper, lead, cadmium or mercury, seriously affect human health. The human body cannot process and dispose of the metals. As a result, they are deposited in various internal organs. Large deposits may cause adverse reactions and serious damage to the body. Those toxic metals can cause the symptoms of memory loss, increased allergic reactions, high blood pressure, depression, mood swings, irritability, poor concentration, aggressive behavior, sleep disabilities, fatigue, speech disorders, cholesterol, triglycerides, vascular occlusion, neuropathy, autoimmune diseases and chronic fatigue.
Traditional methods used for the removal of heavy metals from the environment are in general expensive and potentially risky due to the possibility of the generation of hazardous by-products. Physical and chemical methods have been proposed and applied to remove metal ions from effluents, but, in general, these methods are commercially impractical, either because of high operating costs or the difficulty in treating the solid wastes generated. 5 For example, the use of conventional technologies, such as ion exchange, chemical precipitation, reverse osmosis and evaporative recovery, for this purpose is often inefficient and/or very expensive. Various conventional resins or adsorbents (natural and synthetic) are used. But the fact is that those resins are costly in some cases. If an effluent discharged from a chemical plant contains a large amount of Na+, K+, Ca+, Mg+, Cu2+, and so on and we use a cation exchange resin to remove Cu2+, then along with Cu2+ all those ions will also be removed. As a result, the resin will be exhausted very soon and cannot be used for longer time and will not be economical. It is, therefore, important to develop new methods for metal exclusion and/or recuperation from dilute solutions (1–100 mg/l) and for the reduction of heavy metal ions to very low concentrations. It is well known that cellulosic materials can be obtained and employed as cheap adsorbents and their performance to remove heavy metal ions can be affected upon chemical treatment. In general, chemically modified cellulosic materials exhibit higher adsorption capacities than unmodified forms. Numerous chemicals have been used for the modifications, which include mineral and organic acids, bases, oxidizing agent, organic compounds, and so on. Some of the treated adsorbents show good adsorption capacities for Cd, Cu, Pb, Zn and Ni. Use of an ion selective adsorbent is one of the best solutions of this problem. 6 –14
From the point of view of environmental preservation, fibrous adsorbents for toxic metals such as copper and lead have been extensively developed by radiation-induced graft polymerization. The natural material like coir in many instances is relatively cheap and abundant in supply and has significant potential for modification and ultimately enhancement of their adsorption capabilities. Adsorbent is prepared by direct modification of cellulose with grafting of selected monomers to the cellulose backbone with subsequent functionalization. The heavy metal adsorption capacities for these modified cellulose materials were found to be significant and levels of uptake were comparable, in many instances, to both other naturally occurring adsorbent materials and commercial ion exchange type resins. Many of the modified cellulose adsorbents proved regenerable and reusable over a number of adsorption/desorption cycles, allowing recovery of the adsorbed heavy metal in a more concentrated form. A new type of adsorbent containing phosphoric acid group for the removal of heavy metals was synthesized by radiation-induced graft polymerization of acrylic acid onto coir yarn. The fibrous adsorbents could be synthesized by introducing a functional chelate group, such as phosphate into the coir yarn, wherein AA acts as a carrier of phosphate group. This fibrous adsorbent has high selectivity to heavy ions and higher rate of toxic metal adsorption than that of commercial adsorbent resin in column mode experiment. This high performance of the fibrous adsorbents is attractive for the removal of toxic metals from the streaming water. 15 –24
The aim of this research is to prepare an absorbent for extracting heavy metal ions from coir yarn. The adsorbent was made by treating coir fiber using AA and PA through graft copolymerization technique using ultraviolet (UV) radiation. Here, AA was grafted onto the surface of the coir fiber, which acted as a carrier for phosphate radical (functional group). The adsorbent prepared by this method successfully adsorbed copper ion selectively from waste water. 25 –27
Experimental
Materials
Coir, the natural fiber, was extracted from the husk of coconut (Cocos nucifera) fruit. Coconut was collected from the local market of Bangladesh. The monomer AA, PA and solvent methanol (MeOH) were purchased from Merck (Germany). Photoinitiator Darocur-4043 was procured from Merck, Germany and finally incorporated into the formulation to initiate free radical polymerization reaction.
Methods
The coir yarns were cut into small pieces (150–200 mm in length) and then washed with acetone to remove the foreign particles. After washing, they were dried to remove acetone. A number of solutions were prepared with different concentrations of AA (15–40 wt%) in methanol and 2 wt% PI (Darocur-4043). The samples were soaked in each of the solutions for different soaking time, varying from 15 to 30 min. After withdrawing from the solutions, they were passed under UV curing lamp for different doses (passing 10–40 times). The samples were then kept for 24 h to complete the polymerization reaction. The cured samples were washed in water for 20 h to remove the unreacted monomer and the homopolymers from the sample surface. Then the samples were dried at 105°C until constant weights were achieved. From the final weight, we calculated the percentage of polymer loading (PL). The percentage of AA, soaking time and radiation intensity were optimized for the maximum PL. A number of formulations were again prepared using optimized concentration of AA (30%) and 2–10% (w/w) PA (90%) in methanol. Then the solutions were kept for 2 h. After 2 h, 2% PI was again added in each of the solution. The coir yarns were again soaked in the sample for optimized soaking time of 30 min and were passed through UV radiation for 30 times (optimized). The concentration of PA was again optimized for the maximum grafting. Tensile strength of both treated and untreated virgin coir was measured with a tensile strength testing machine Hounsfield (Model no: H50KS, Serial no: H50KS-0404, made in UK). A number of solutions of CuSO4 were prepared with distilled water and the concentration of Cu2+ solutions was 10 ppm. The treated coir yarn was then used as an adsorbent. The Cu solution 100 ml were taken in 5 separate beakers (1 g AA + PA); among them, 4 were soaked for 30 min, 1 h, 2 h and 3 h, respectively. In the rest of the two beakers, 1 g of pure coir and 1 g of AA grafted with coir yarns were soaked for 3 h. After a certain period, those threads were withdrawn from the solutions and then analyzed by an atomic absorption spectrophotometer to determine the remaining Cu concentration of the solution. After adsorbing, Cu2+ (AA + PA)-grafted coir yarns were soaked in 0.03 M HCl for regeneration. They were soaked in 100 ml acid solution for 1 and 2 h, respectively. For regeneration, Cu2+ containing yarns were also soaked in water for 2 h. After withdrawing the yarns, the solutions were again analyzed by AAS to find out the Cu concentration of the solution.
Results and discussion
Optimization of monomer concentration, soaking time and the number of times UV radiation passed based on PL on coir yarns
PL values of the coir yarns against radiation intensities (the number of times UV radiation is passed) as a function of monomer concentration for various soaking times are represented in Table 1. It was reported that PL values increased with the increase of AA concentrations up to 30% and further increase of AA concentration, PL values was decreased. Again, the PL values increased with an increase in UV intensities, attained the maximum values after the UV radiation is passed for the 30th time and then decreased as the radiation doses increased. From this investigation, it is clear that UV radiation passed for 30 times showed the best performance. At AA concentrations of 15, 20, 30 and 40%, the height of the PL values were found to be 12.3, 14.2, 18.3 and 15.4% at 20 min soaking time, respectively. So, a significant improvement in PL values was found for 30% AA concentration at a passage of UV radiation for the 30th time.
PL (%) against radiation intensities as a function of AA (%).

PL: polymer loading; AA: acrylic acid; UV: ultraviolet.
At a low monomer concentration, the vinyl monomer promotes the rapid propagation reaction with the help of PI leading to network polymer structure through graft copolymerization reaction of their double bonds. As the AA concentration is increased, the amount of residual concentration is also increased, as a consequence of faster rate of formation of three-dimensional network structures causing restricted mobility. After the attainment of the maximum monomer concentration, the decrease in PL values at higher monomer concentration may be caused by two factors. At higher AA concentration, radical–radical recombination process may be dominant; thus, more homopolymers are created rather than monomer-backbone reaction. The other reason could be that the swelling of the coir backbone with MeOH was insufficient due to the low methanol concentration. As a result, monomer molecules are incapable of penetrating the coir molecules in the presence of low solvent concentration. This may cause a smaller number of reacting sites at the coir backbone and thus continue to reduce the active sites. The PL values decrease with increasing radiation dose because at higher radiation dose coir yarn may degrade.
In the above investigation, the soaking time was fixed at 20 min, but it was found that with the increase in soaking time, a significant impact was found on the PL values. The best PL values could be obtained for 20 min soaking. The highest PL value was reported as 18.3% at the passage of UV radiation for the 30th time and at an AA concentration of 30% for 20 min soaking time. On the other hand, it was reported that the highest PL value (13.2%) was achieved at a 30% concentration of AA solution for 15 min soaking time. From this investigation, it is clear that the coir yarns soaked for 20 min in AA concentration of 30% at the passage of UV radiation for the 30th time yielded the maximum PL values (18.3%)
Optimization of PA concentration
Polymer loading
Figure 1 shows the PL value as a function of PA concentration. The value increases with increase in PA concentration. The highest value of 35.1% is obtained for 4% PA. The PL value decreases with increase in PA by more than 4%, which may be due to the lower rate of polymerization at the higher PA concentration. It may also be possible that the higher concentration of acid may change or degrade the main polymeric chain and hence the PL value decreases. When PA is used, the PL values enhance quite substantially up to 4%. Acid helps to speed up the copolymerization process through the cross linking of individually segmented units of the polymer. Acidification of the solution changes the mobility of the solution at an equilibrium condition of the components during the copolymerization process at the zone of the reactivity associated with the partitioning of the various components present in the solutions. The rate of reactivity coupled with the partitioning, thus, enhanced the overall copolymerization process in the presence of acid.
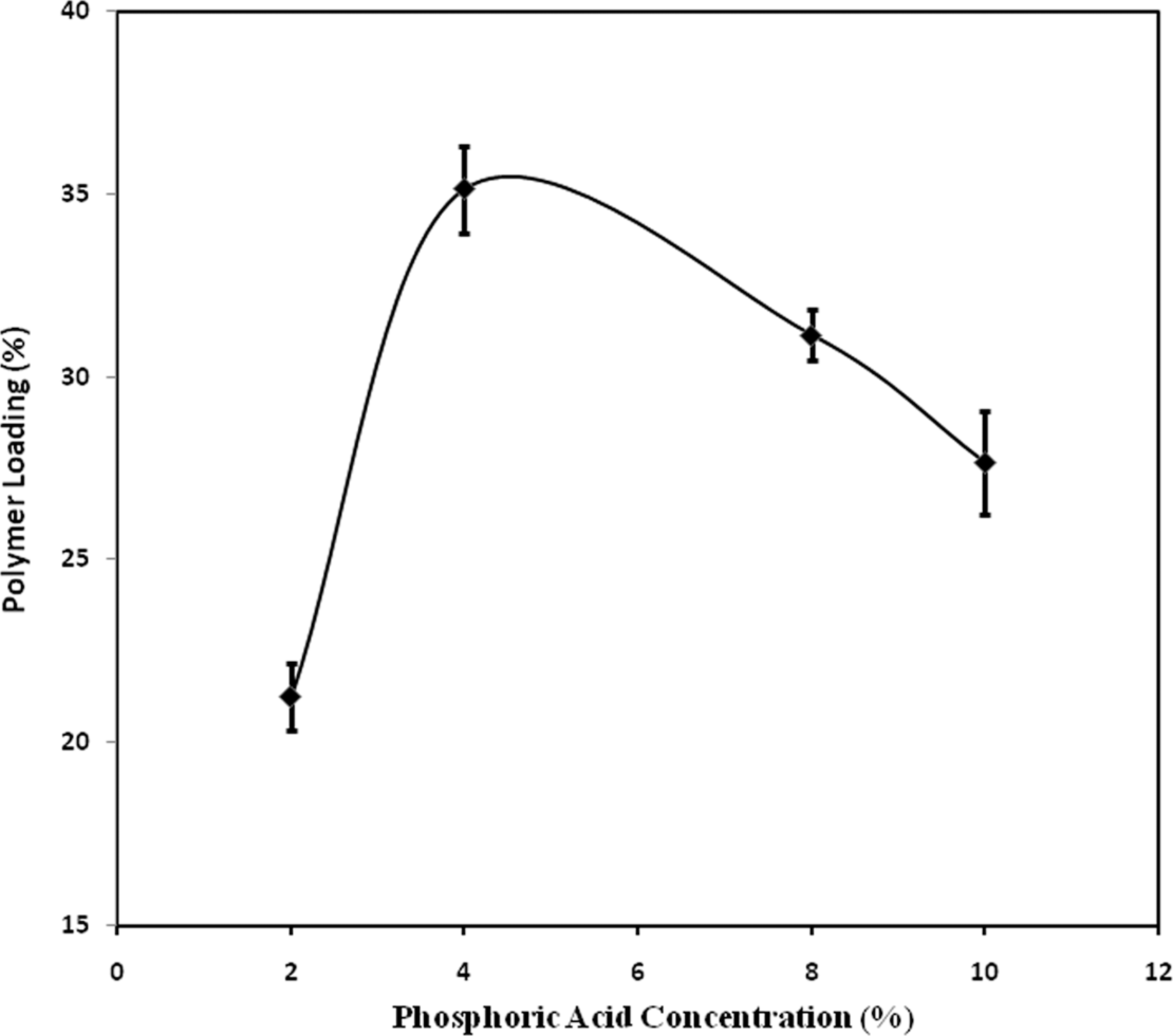
Polymer loading values of coir yarn against percentage concentration of phosphoric acid.
Tensile strength
The tensile strength of sample increases with increase in PA of up to 4% and the highest value is 72.1 MPa, which is shown in Figure 2. Decrease in the tensile strength value may be due to the fact that the rate of polymerization decreases at high acid concentration. The other reason may be that the concentrated PA solution may change or degrade the polymeric backbone. H+ ion breaks the long chain of the coir yarn thus rendering the reduced tensile strength of the yarn.
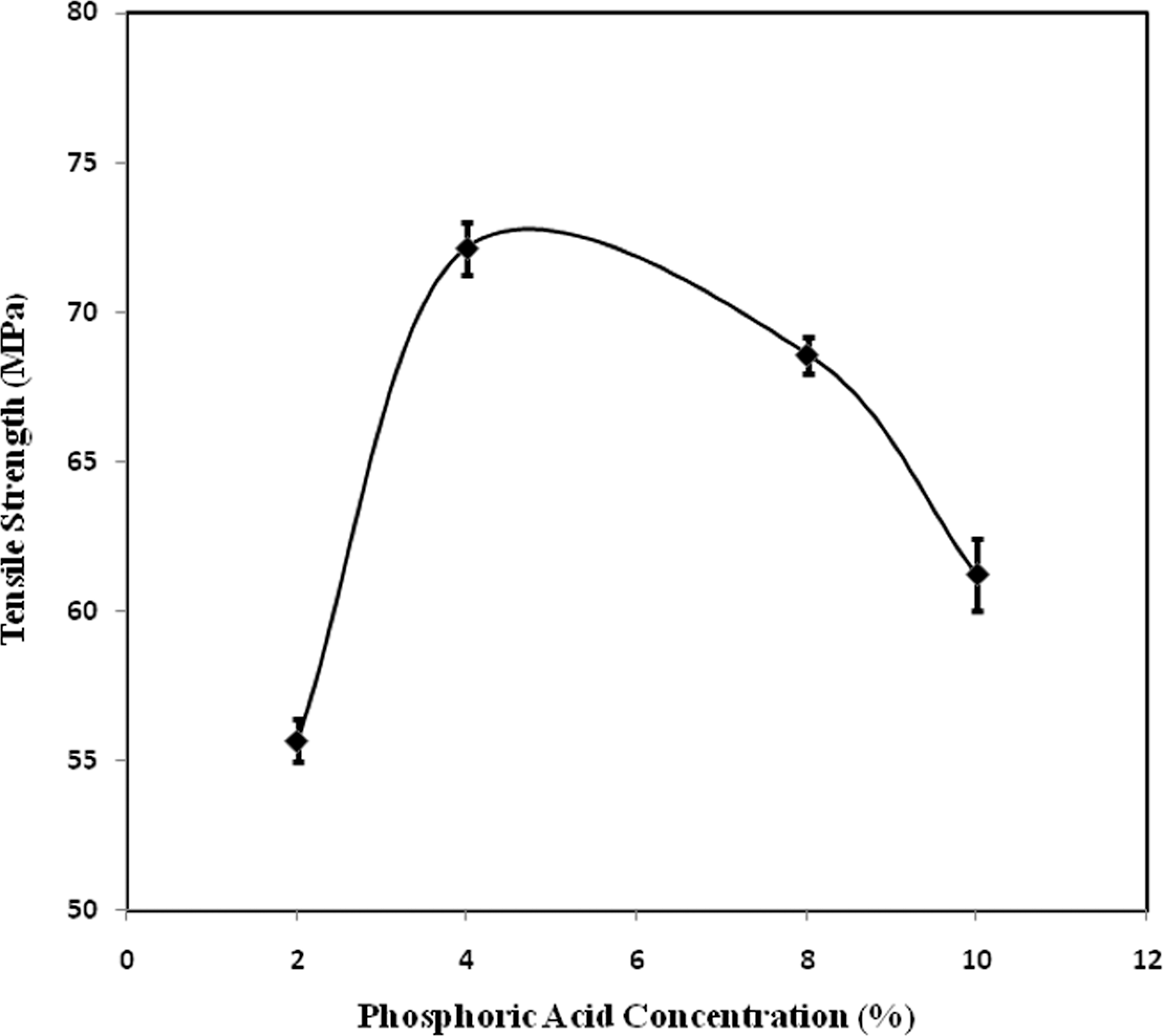
Tensile strength values of AA + PA-grafted coir yarn as a function of percentage concentration of PA. AA: acrylic acid; PA: phosphoric acid.
Cu uptake
Figure 3 shows that the metal uptake increases significantly up to 2 h, after which the Cu uptake is almost same. This phenomenon occurs due to the fact that after 2 h, the threads become almost saturated with Cu. There is no more vacant site in those threads to take more Cu from the solution. It is possible to remove more Cu within 2 h from the same solution if the amount of thread is increased so that the total vacant sites will also increase. It was found that the Cu uptake was 1.56, 2.45 and 3.89 mg/g of thread for 30 min, 1 h and 2 h, respectively.
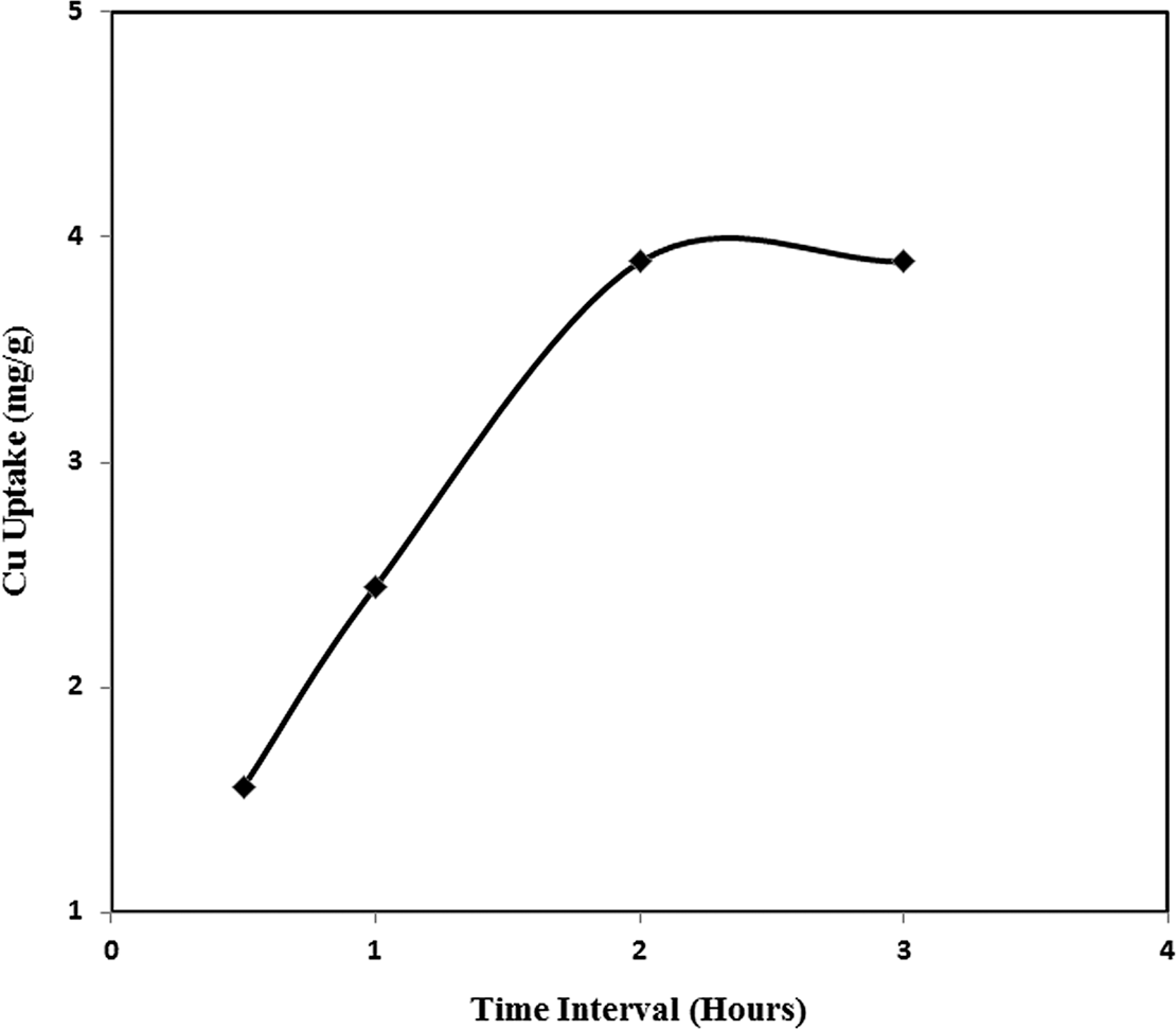
Cu uptake of AA + PA incorporated coir yarn from 10 ppm solution of Cu for different time intervals. AA: acrylic acid; PA: phosphoric acid.
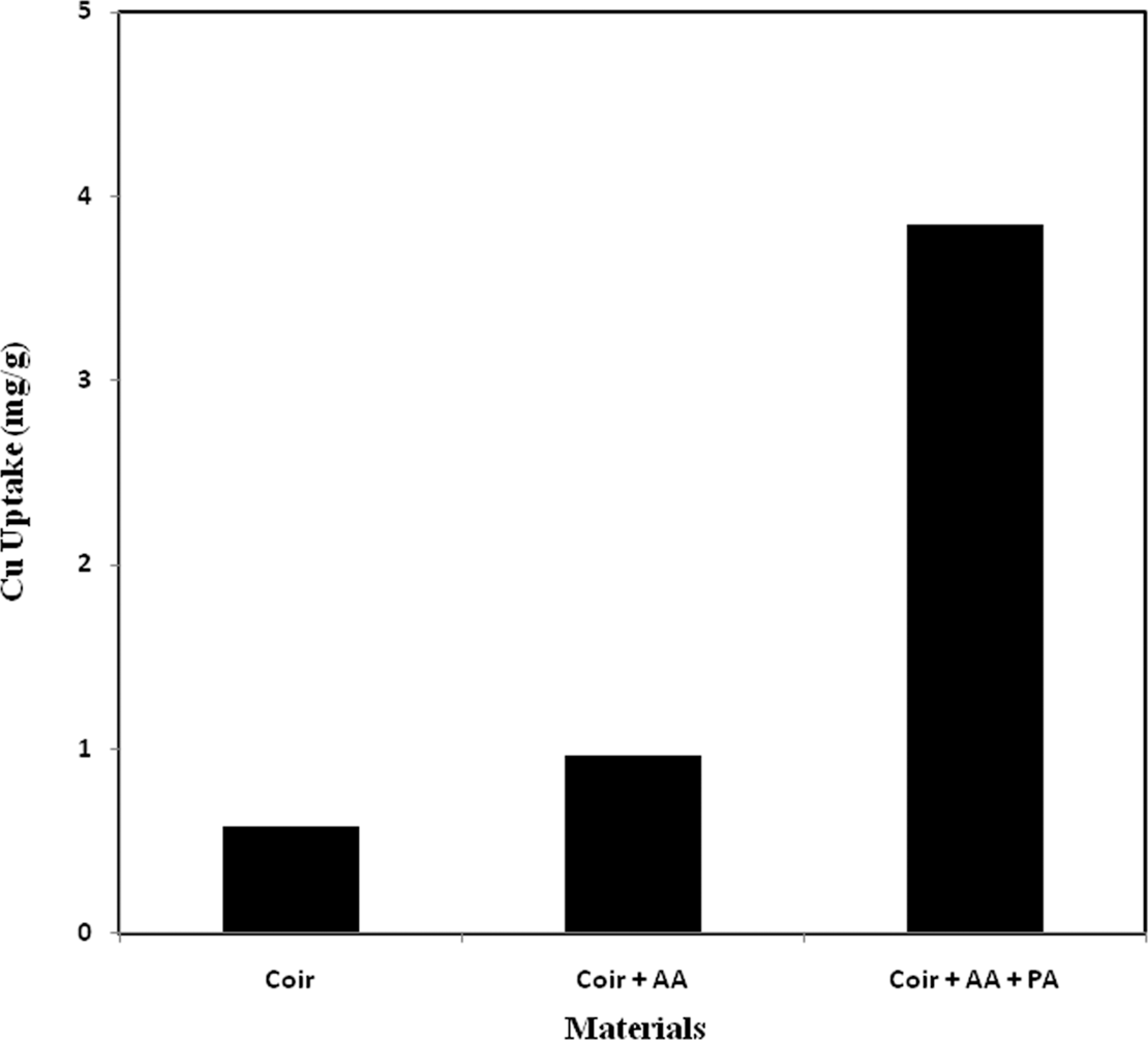
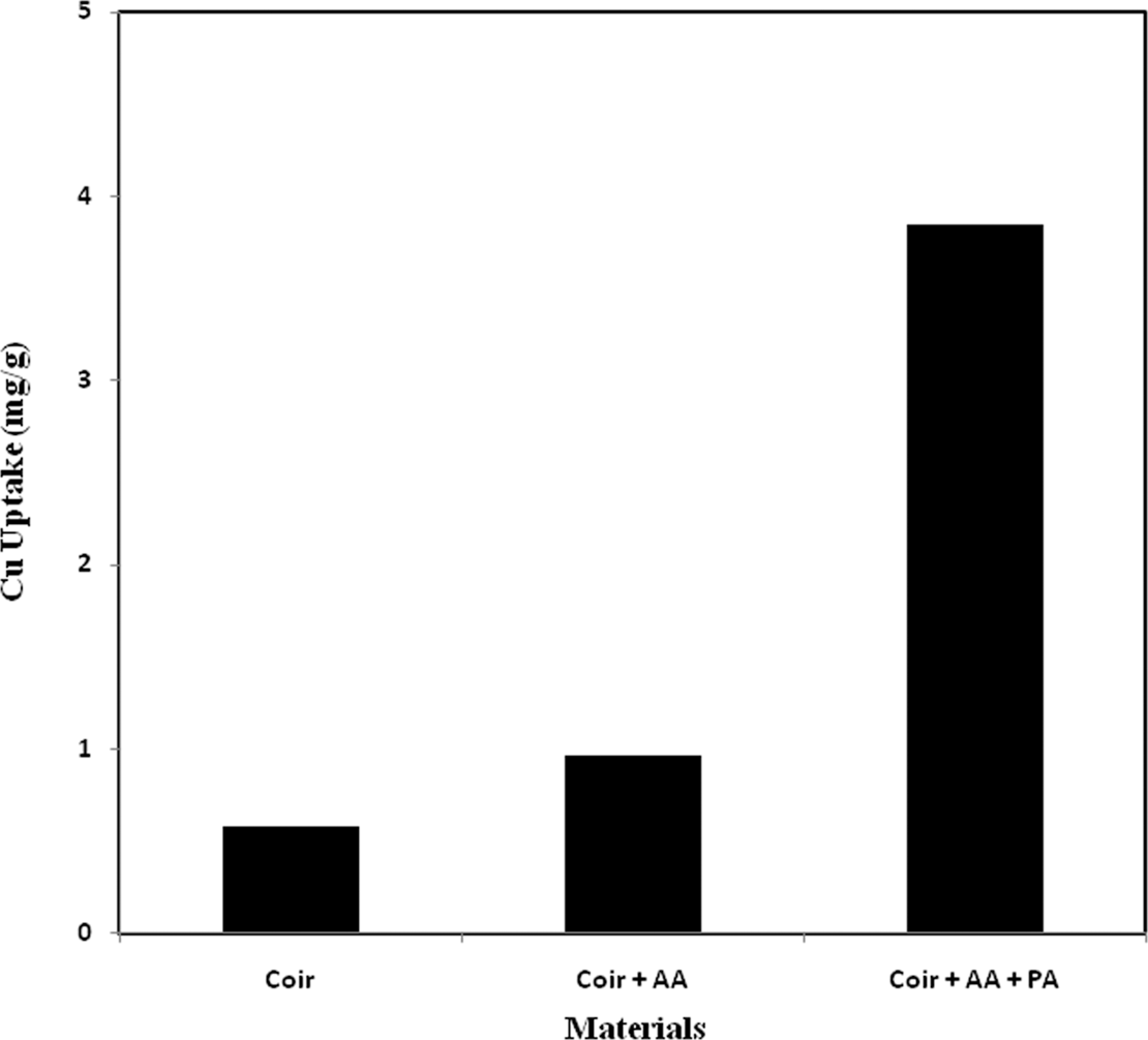
Comparative study of Cu uptake
The comparative study of Cu uptake between pure, AA-grafted and AA + PA-incorporated coir is presented in Figure 4 as a function of Cu uptake. Figure 4 shows that the pure and the AA-grafted threads removed a very small amount of Cu because some molecules of Cu which are just in touch with the surface of those threads can be removed. Cu uptake was found to be 0.58, 0.96 and 3.85 mg/g of thread for pure, AA-grafted, and AA + PA- incorporated coir yarn, respectively.
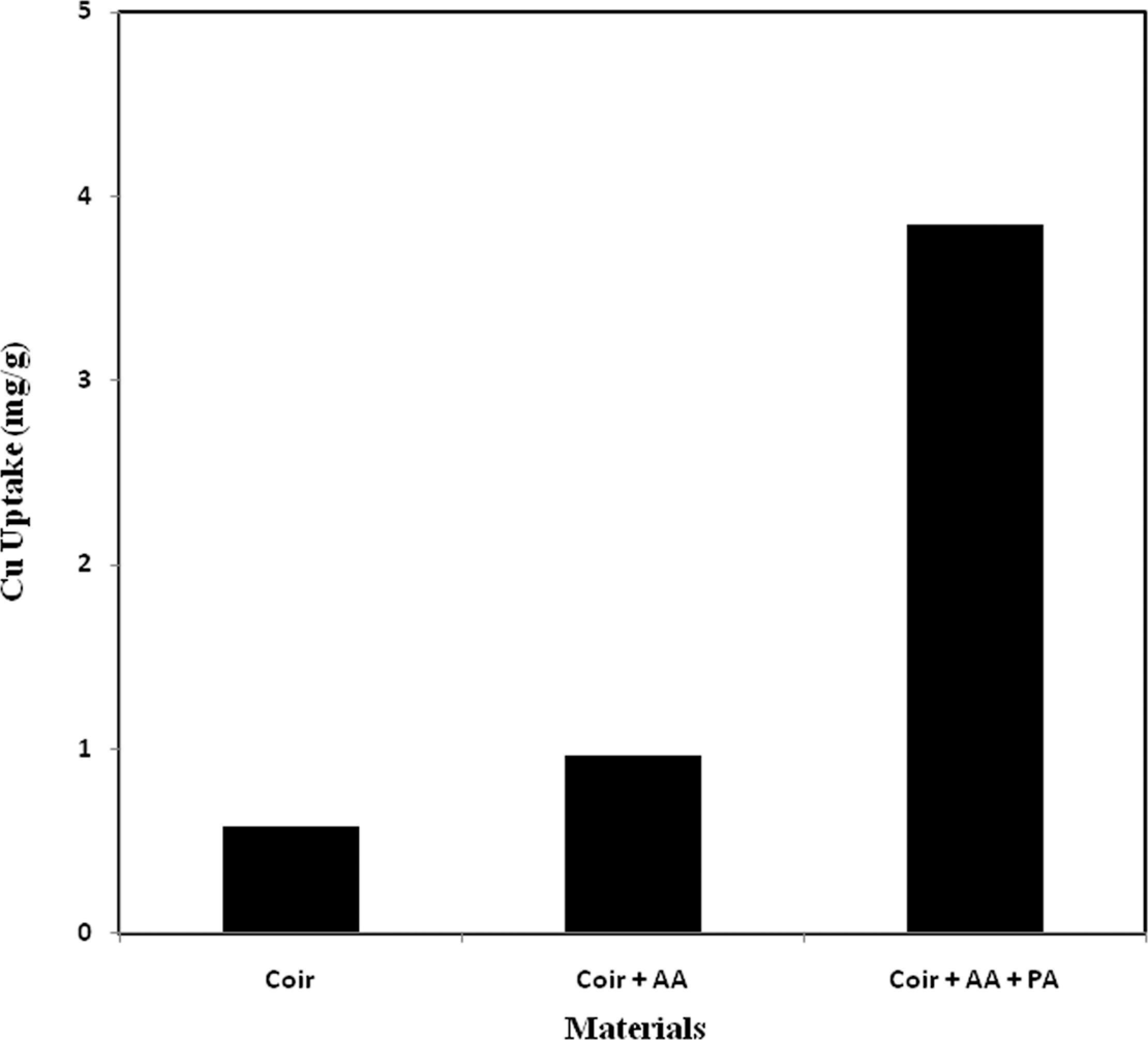
Comparative study of Cu uptake from 10 ppm Cu solution among pure, AA grafted, and AA + phosphoric acid grafted coir yarn. AA: acrylic acid.
Regeneration of adsorbent
After taking metal, the threads have to be regenerated in acid so that it can be used again as an adsorbent. In Figure 5, regeneration of Cu is presented as a function of time interval when threads are soaked in 0.03 M HCl. The recovered concentration of Cu in the solution is increased with increasing time. By regeneration, 0.53 and 0.98 mg of Cu per gram of thread was recovered for 1 and 2 h, respectively.
Recovery of Cu after regenerating coir yarn in 0.03 M HCl solution.
Comparative study of regeneration
Comparative study of regeneration between water and HCl is presented in Figure 6 as a function of recovered Cu concentration. The concentration of Cu found in water is very negligible, which indicates that the adsorption that has occurred was not simple physical adsorption, but chemisorption. If it is physical adsorption, then the Cu can be extracted from the thread by water wash alone. Since it is chemisorption, it requires external energy such as heat, stirring or another ion exchange reaction for the adsorbed Cu to be extracted from the threads. Hence, the threads must be regenerated in an aqueous HCl solution. Cu of 0.12 and 0.98 mg were recovered per gram of thread in water and in acid for 2 h, respectively.
Comparative study of recovery of Cu from coir yarn after regenerating in water and acid for 2 h.
Conclusions
Coir yarns were modified using acrylic and PA via UV treatment. Based on optimized polymer loading (35.1%) of coir yarns, ion selective adsorbent was prepared. The optimized formulation was found to be 4% PA, 30% AA, 64% methanol and 2% PI. Copper was effectively removed from the CuSO4 solution and thus indicated the potentiality of the modified coir yarns as ion-selective adsorbent. It was also proved that the regeneration of Cu was also possible from the grafted coir yarn.
