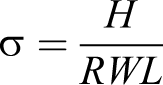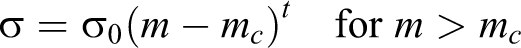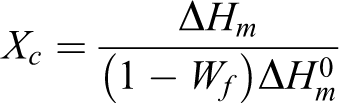Abstract
A nonionic surfactant, Triton X-100, was used to treat multiwalled carbon nanotubes (MWCNTs), and the Triton X-100-treated MWCNTs (TCNTs) were then mixed with polypropylene (PP) to prepare MWCNT/PP composites, followed by characterization using scanning electron microscopic, electrical, mechanical and thermal measurements. It was found that the best usage of Triton X-100 for the treatment of MWCNTs was 80 wt% based on the MWCNTs. The TCNTs could effectively improve the electrical conductivity and mechanical properties of the composites over pristine MWCNTs. The highest electrical conductivity of TCNT/PP composites was 2.1 × 10−2 S/cm at 10% TCNTs. Both MWCNTs and TCNTs could act as nucleating agents to promote the crystallization of PP but the crystallinity of PP decreased with increasing filler’s content. Both fillers caused shifting of the crystallization temperature of PP to higher temperatures. The TCNT/PP composites showed the lower crystallization temperature and lower crystallinity than the MWCNT/PP composites at the same filler’s content. The mechanisms for the results have been discussed.
Keywords
Introduction
Most of polymer materials have very high electrical resistance. In electronic industries, polymer materials are being widely used to produce numerous parts such as circuit boards, IC chip trays, encapsulants for electronic devices, housings of personal computers, frames of monitors, keyboards, mouses, and so on. Many of these applications take the advantages of polymer materials as good insulators, but some applications require polymer materials to be electrically conductive. For example, conductive polymers can be used for electromagnetic shielding, antistatic electricity, printed circuit boards, electrodes of electrochemical devices such as fuel cells, and so on. However, there are only few types of inherently conductive polymers commercially available, but they usually have poor processability because they are not thermoplastic. For example, polyaniline (PANI) is a well-known conducting polymer, but its application is limited as PANI is not thermoplastic and a doping process is necessary to induce electrical conductivity.
A common approach for the development of conductive thermoplastics is achieved through adding conductive fillers into a nonconductive polymer matrix. The commonly used conductive fillers include metal powder, metal fibers, carbon nanotubes (CNTs), carbon black, graphite, and so on.1–7 These conductive filler/polymer composites have attracted much attention recently due to their high potentials in industrial applications.
In all the conductive fillers mentioned above, CNTs exhibit fascinating electronic and mechanical properties because of their unique molecular structures. Fundamental research works on CNTs and their applications have grown rapidly.8–10 However, a number of challenges must be overcome before CNTs can be exploited as conductive fillers for polymers. For example, CNTs are generally entangled together and tend to agglomerate in a polymer matrix. The weak interfacial bonding between CNTs and a polymer matrix also limits full advantage of the superior properties of CNTs.
Several research groups have investigated the electrical properties of multiwalled carbon nanotubes (MWCNTs)/polypropylene (PP) composites, prepared by various methods. For instance, Yu et al. 11 reported the electrical conductivity of PP filled with 3 wt% of m-MWCNT (the MWCNT after modification with the titanate coupling agent) reached more than 10−3 S/m, which was 5 orders higher than that of PP filled with 3 wt% pristine MWCNTs. Pollatos et al. 12 observed the direct current (DC) conductivity to be in the order of 10−6 S cm−1 for the melt mixed MWCNT/sPP (syndiotactic PP) composites at 5 wt% MWCNT loading. Ngabonziza et al. 13 reported the electrical conductivity of 10−3 S cm−1 for the melt mixed MWCNT/PP composites with 10 wt% of MWCNTs. Grossiord et al. 14 observed DC conductivity of 10−2 S cm−1 in the latex-blended MWCNT/iPP (isotactic PP) nanocomposites with 2 wt% of MWCNT as well as the percolation threshold as low as 0.1 wt%. Khare et al. 15 reported the DC conductivity of about 10−5 S cm−1 for the MWCNT/PP composite with 1 wt% of MWCNTs modified by the sodium salt of 6-amino hexanoic acid. The lowest electrical percolation threshold was achieved at 0.1 wt% of the 6-amino hexanoic acid sodium salt-modified MWCNTs.
Recently, surfactants have been used for surface modification of CNTs in order to improve the dispersion of CNTs in a polymer matrix.16 –31 Surfactants have several advantages. Surfactant treatment does not break the structure of CNTs, so it can preserve the electrical and mechanical properties of CNTs. 19 Surfactants may enhance adhesion between the fillers and the polymer,20 –23 thus improving dispersion of the fillers in the polymer or solutions25 –28 and then increasing the mechanical properties of CNT/polymer composites. 24 The most of these researches focused on the effects of surfactants on dispersion of CNTs, and mechanical and thermal properties of CNT/polymer composites, but paid less attention to the electrical properties of composites filled with surfactant-treated CNTs. In the present work, we have used a nonionic surfactant, polyoxyethylene octyl phenyl ether (Triton X-100) to study its effects on surface modification of MWCNTs and further on enhancement of electrical conductivity of MWCNT/PP composites. It is expected that the surface modification of MWCNTs with Triton® X-100 will decrease van der Waals forces among MWCNTs and improve the dispersion of MWCNTs in the matrix, resulting in improvement in electrical conductivity.
Experimental
Materials
Polypropylene (PP) in the form of pellets (characteristic cylindrical diameter 2.5 mm, length 3 mm) was purchased from Sigma-Aldrich, Singapore and its molecular weight was 340,000 according to the supplier. Multiwalled carbon nanotubes (MWCNTs) were purchased from Iljin Nano Tech, Korea, and their diameter and length were about 10–15 nm and 10–20 μm, respectively. Triton X-100 ((CH3)3CCH2C(CH3)2C6H4O(CH2CH2O)9H; molecular weight = 646.86 g mol−1, density at 25°C = 1.07 g mL−1) and absolute ethanol were purchased from Sigma-Aldrich, Singapore.
Surface treatment of MWCNTs with Triton X-100
MWCNTs and Triton X-100 were added into absolute ethanol, at a desired ratio, and then the mixture was subjected to ultrasonication (Cole-Parmer 8900, USA) for 1 h. After ultrasonication, the final product was obtained by drying the mixture in a vacuum oven at 40°C for 2 days. The drying process allowed Triton X-100 molecules to be condensed on the surface of MWCNTs. The weight ratio of Triton X-100 to MWCNTs varied from 0 to 2, in order to find the optimal ratio for the best effect of Triton X-100 on electrical conductivity of a MWCNT/PP composite. The Triton X-100-treated MWCNTs are denoted with TCNTs.
Preparation of composites
The fillers (pristine MWCNTs or TCNTs) and PP were mixed at various desired weight ratios at 200°C by means of a micro-compounder (HAAKE Minilab 2, Germany). The rotation speed was 70 r/min and the mixing time was 10 min. Finally, the PP composite samples were injection-molded using a mini-jet molding machine (HAAKE MiniJet 2, Germany). The injection molding temperature was 210°C; the injection pressure was 600 Pa for 10 s; the mold temperature was 60°C; and the holding pressure was 300 Pa for 5 s. For annealing to improve the electrical conductivity of samples, the samples were kept in an oven at 220°C for 30 min. According to our previous work, the electrical conductivity of injected molded CNT/PP composites could be improved by several orders through a melt annealing process because annealing could allow CNTs to be rearranged in the polymer matrix to construct more networks. The samples were named as ‘xMWCNT/PP’ or ‘xTCNT/PP’ where the prepositional letter ‘x’ stands for the content (in wt%) of MWCNTs or TCNTs in the composite samples. For example, ‘1MWCNT/PP’ means a MWCNT/PP composite containing 1 wt% of pristine MWCNTs, and ‘5TCNT/PP’ represents a TCNT/PP composite containing 5 wt% of TCNTs.
Characterization
The through-plane electrical conductivity of the composite samples was measured according to American Society for Testing and Materials (ASTM) D4496. The samples of a known cross-sectional area and thickness were placed between two copper electrodes and the DC resistance was measured using a Fluke 110 multimeter. The surfaces of the sample in contact with the electrodes were covered with carbon paper in order to reduce discrepancies in conductivity measurement due to microroughness of sample surfaces. It was ensured that the surface area of the electrodes exceeded the cross-sectional area of the samples. The lowest limit of the conductivity measurements was 1 × 10−10 S/cm.
The dispersion state of MWCNTs in the PP matrix was observed using field emission scanning electron microscopy (FESEM, JEOL JSM-7600F, Japan). The mechanical properties of the composites were measured using an Instron Universal Testing Machine according to ASTM D638. The crystallization behavior of PP in its composites was studied by differential scanning calorimetry (DSC) in N2 atmosphere. The sample was first heated to 200°C at 10°C/min and melted completely, and then cooled to 40°C at 10°C/min. In this way, the crystallization curves of the MWCNT/PP composites were obtained.
Results and discussion
Microstructure of composites
The TCNT/PP composites fractured in liquid nitrogen were observed using FESEM. Some of selected FESEM micrographs are shown in Figure 1, where the black area indicates the PP matrix and the white tube-like shapes are the MWCNTs or TCNTs. The FESEM micrographs in Figure 1(a) and (b) were taken from the TCNT/PP samples, while Figure 1(c) and (d) were from the MWCNT/PP samples. From these FESEM images, we observe that TCNTs could be dispersed much more homogeneously than MWCNTs. In the samples of MWCNT/PP composites, very large aggregates of MWCNTs are present. As shown in Figure 1(a), although very few agglomerations are still observed, most of TCNTs are dispersed uniformly. In Figure 1(b), a large network structure across the whole image is not clearly observed, but a small network-like structure could be seen in the lower left corner of the image. Therefore, the 5TCNT/PP composite should have a higher possibility to form a network of TCNTs than the 1TCN/PP composite. But in the 5TCNT/PP composite, the larger number of TCNTs and the shorter interparticular distances among them allowed them much easier to construct networks for the higher electrical conductivity than the 1TCNT/PP composite. The similar phenomenon was also observed in the MWCNT/PP composites.

Field emission scanning electron microscopy (FESEM) micrographs of MWCNT/PP and TCNT/PP composites: (a) 1TCNT/PP, (b) 5TCNT/PP, (c) 1MWCNT/PP, and (d) 5MWCNT/PP. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
In the preparation process of TCNTs, Triton X-100 molecules could be adsorbed onto the surface of MWCNTs by the forces such as π-π stacking between MWCNTs and benzene rings of Triton X-100, van der Waals interaction, or physical adsorption. Triton X-100 adsorbed onto the surface would be able to dissociate bundles or aggregates of MWCNTs into individual ones. The different morphological structures of TCNTs and MWCNTs in the same polymer matrix can be simply known from the FESEM observation. For example, let us compare the FESEM micrographs for the TCNT/PP and MWCNT/PP samples at the same contents (i.e. 1 wt% and 5 wt% fillers; i.e. compare Figure 1(a) with (c), and compare (b) with (d), respectively). In TCNT/PP composites, the TCNTs were dispersed very well and the agglomerates were small. The sizes of the TCNT agglomerates are smaller than 500 nm while some are the individual CNTs. But the MWCNT/PP composite contained the big agglomerates of MWCNTs. The sizes of the MWCNT agglomerates are larger than 1 μm. The results proved that the Triton X-100 could help the dispersion of the MWCNTs, because the Triton X-100 could reduce the surface energy of MWCNTs and decrease the van der Waals force between MWCNTs. The Triton X-100 attached onto the surface of MWCNTs increased the steric hindrance of MWCNTs and acted as a chemical bridging agent of Coulomb interactions. 20 The steric hindrance increased the distance between MWCNTs and decreased the attractive force that made MWCNTs to agglomerate. The chemical bridge of Triton X-100, which was formed between the polymer matrix and MWCNTs, increased the compatibility of MWCNTs with the polymer matrix. All these factors resulted in the better dispersion of MWCNTs in the polymer matrix. A better dispersion of fillers in the polymer matrix will lead to an earlier percolation at a lower volume fraction of fillers.
Electrical conductivity of composites
The MWCNT/PP and TCNT/PP composites were molded into the square-shaped samples using a mini-jet injection molding machine. Then the resistance of each sample was measured. The electrical conductivity was calculated using the following equation:
where σ is the electrical conductivity in S/cm, L is the sample’s length in cm, W is the sample’s width in cm, H is the thickness in cm, and R is the measured resistance in Ω.
Unfilled PP is an excellent insulating material and has electrical conductivity of 10−17 S/cm. 32 To make a PP composite electrically conductive, electrically conductive fillers must be added and an electrically conductive network must be formed. The electrical conductivities of all the composites prepared in this work are shown in Figures 2 and 3 as a function of filler’s content in wt%. Depending on the type of fillers, the composites behaved differently.
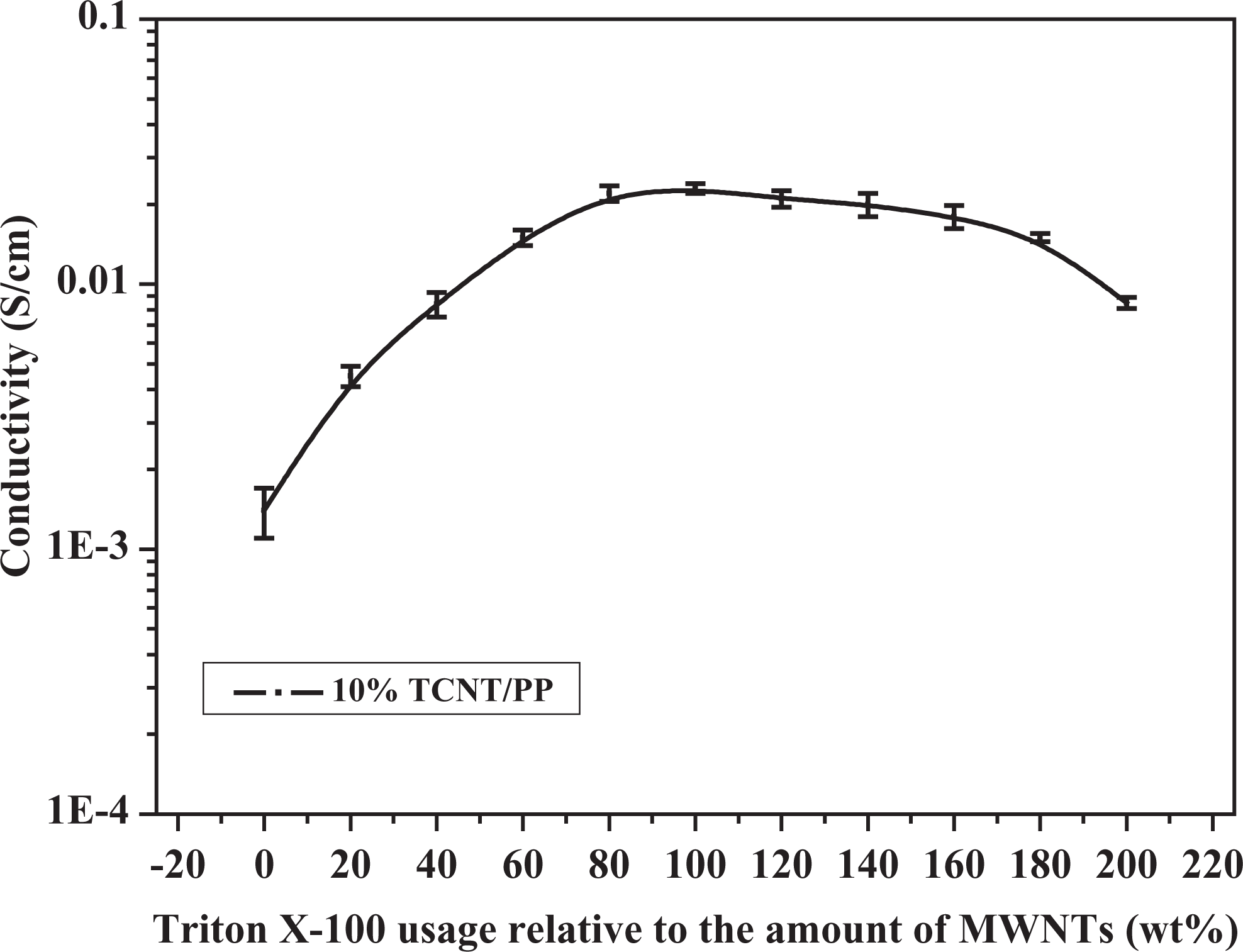
Electrical conductivity of TCNT/PP composites as a function of Triton X-100 usage relative to the amount of MWCNTs. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
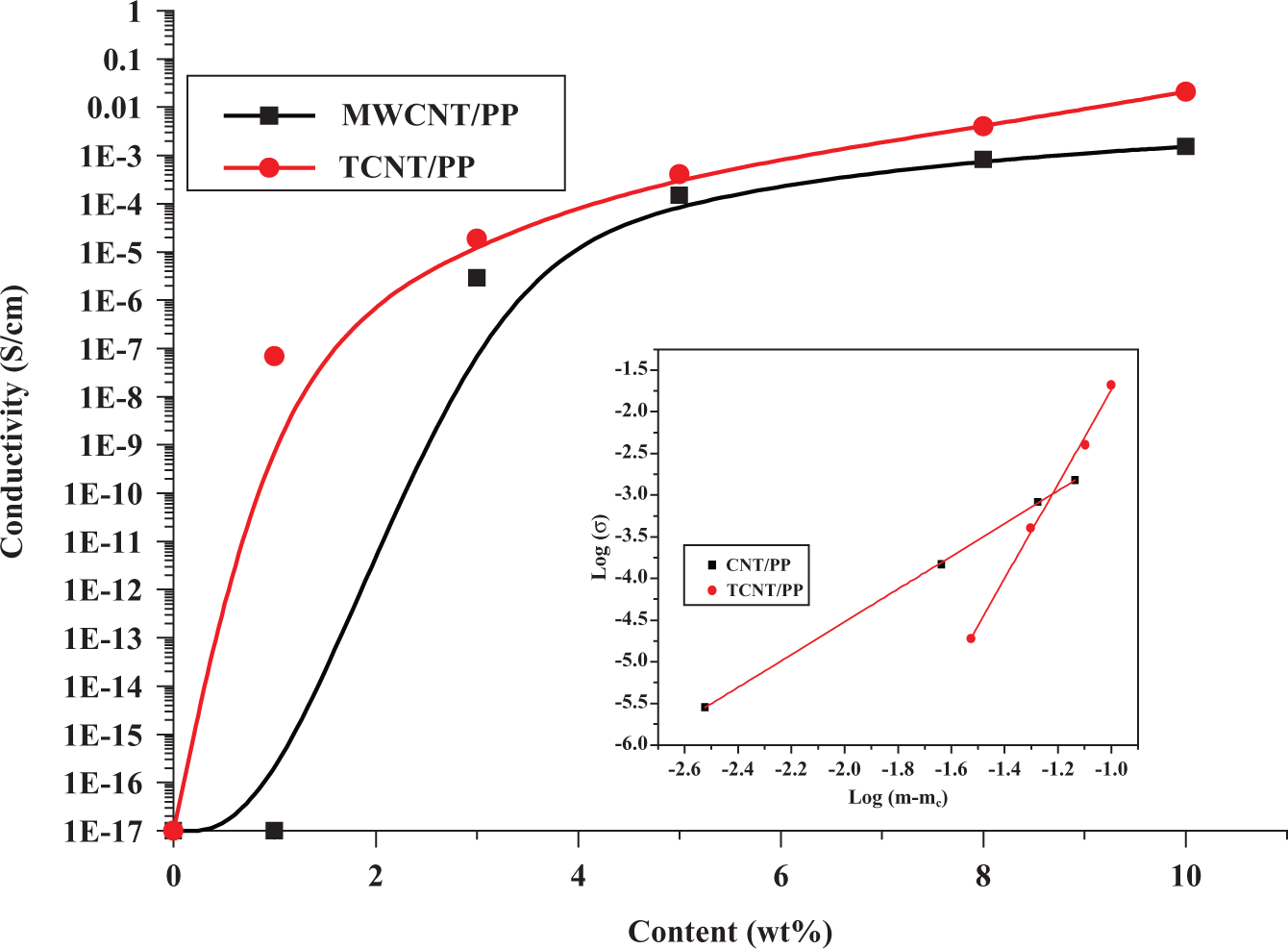
Electrical conductivity of MWCNT/PP and TCNT/PP composites. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
Effect of the usage of Triton X-100 on electrical conductivity of composites
In order to find the best usage of Triton X-100 relative to the amount of MWCNTs, the TCNTs with the different usages of Triton X-100 were prepared and used as fillers of PP composites. The results for electrical conductivity of TCNT/PP composites are shown in Figure 2. The conductivity of TCNT/PP composites increased monotonously with increasing Triton X-100 loading but tended to level down after 80 wt%. Thus, Triton X-100 reached the highest effect on improving electrical conductivity of composites at a level of 80 wt% based on MWCNTs, and the further increase in the usage of Triton X-100 decreased the electrical conductivity. The effect of Triton X-100 on improving electrical conductivity is the result from two factors: dispersing and blocking. A schematic description of the proposed effect of Triton X-100 on electrical conductivity of the composites is shown in Figure 4. When Triton X-100 helped the dispersion of MWCNTs, at the same time, it also acted as the blocking parts for conducting electrons because Triton X-100 itself is not electrically conductive. Therefore, the more usage of Triton X-100 means the more blocking effect, thus resulting in the lower electrical conductivity.
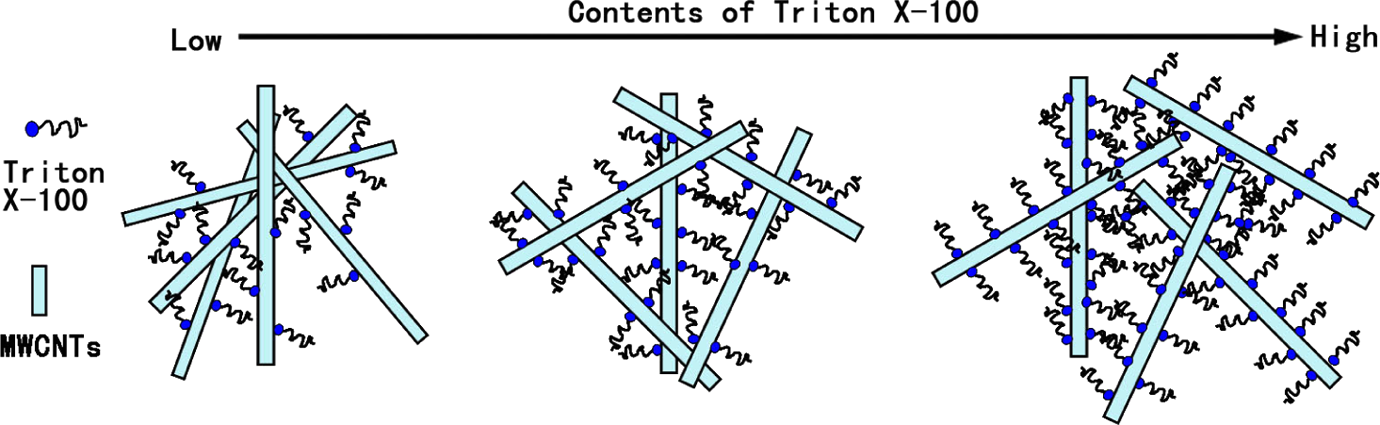
Schematic representation for the effect of different contents of Triton X-100 on the electrical conductivity of composites.
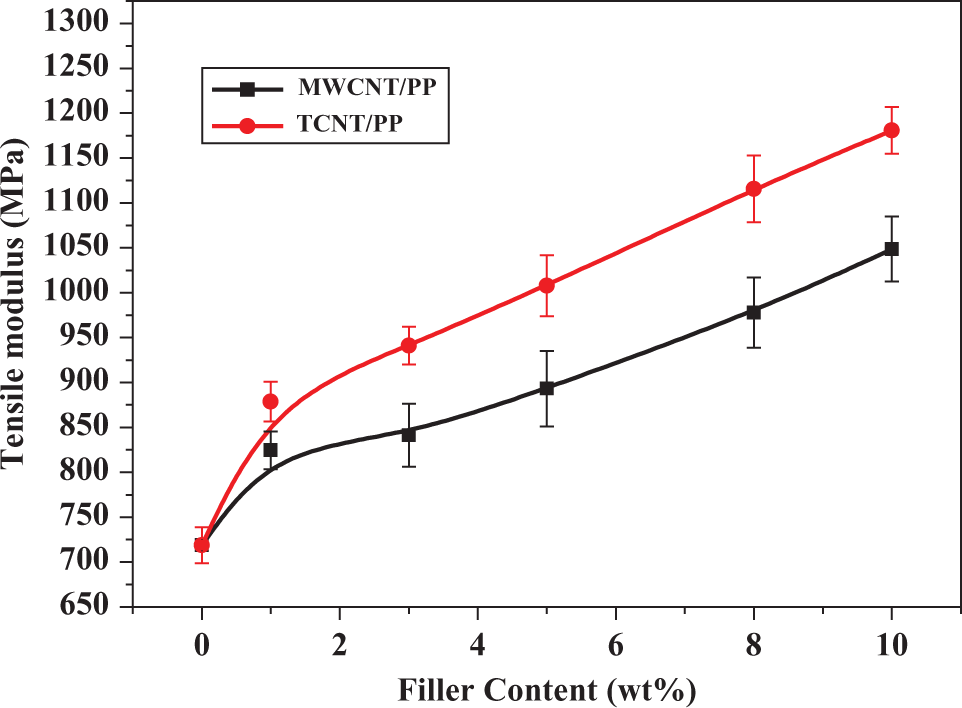
Tensile modulus of MWCNT/PP and TCNT/PP composites. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
Effect of TCNTs on improving electrical conductivity of composites
The electrical conductivity of a composite can be best described by a scaling law based on the percolation theory:
where σ is the electrical conductivity of the composite, m is the filler’s content, mc is the percolation threshold, and the exponent t is related to sample dimensionality.33–38 The electrical conductivity can be significantly increased by several orders of magnitude when the percolation threshold mc is achieved due to the formation of infinite conductive clusters by filler particles.
As we expected, Triton X-100 could help improve the electrical conductivity of MWCNT/PP composites due to the improved dispersion and distribution of MWCNTs in the PP matrix. The approach also preserved the delocalized π-electron network on the nanotube sidewall and ensured minimal perturbation of the defect-sensitive electrical properties. According to the result of the usage amount of Triton X-100, we choose the TCNTs in which the percentage of Triton X-100 was at a level of 80 wt% based on MWCNTs. In other words, the percentage of Triton X-100 in the TCNTs was 44.4 wt%. In Figure 3, the composites containing TCNTs exhibited the more pronounced enhancement in electrical conductivity than their counterparts containing pristine MWCNTs. The curve fitting using equation (2) gives the percolation threshold mc = 0.2 wt% and mc = 2.7 wt% for TCNT/PP and MWCNT/PP composites, respectively. Thus, the results proved that the TCNTs can significantly decrease the percolation threshold.
The evolution of electrical conductivity for these two types of CNT/PP composites showed the different patterns. The conductivity of MWCNT/PP composites increased monotonously with increasing filler loading but tended to level off after 8 wt% loading. The electrical conductivity of TCNT/PP composites also increased monotonously with increasing filler loading but tended to continue increasing after 8 wt% loading. At the same content of 10 wt%, the TCNT/PP composite reached the highest conductivity of 2.1 × 10−2 S/cm, while the MWCNT/PP composite only reached a conductivity of 1.5 × 10−3 S/cm, which is almost one order lower than the former, showing the advantage of TCNTs as an effective conducting filler to improve the electrical conductivity of a polymer composite.
The electrical conductivities of MWCNT/PP and TCNT/PP composites are mainly determined by the state of fillers: dispersion and distribution. As proved by FESEM, for the pristine MWCNTs, they tended to form big agglomerates and the dispersion of pristine MWCNTs in the polymer matrix was not good (see Figure 1(c) and (d)). On the other hand, TCNTs can be dispersed better in the polymer matrix (Figure 1(a) and (b)) for the reasons discussed above. The better dispersion means that the TCNTs can construct more networks to allow electrons to pass through, leading to a higher electrical conductivity.
Mechanical properties of composites
Figure 5 shows that the tensile modulus of composites increased with increasing the content of TCNTs or MWCNTs. Although these two kinds of composites showed the similar trends in tensile modulus, they exhibited the different trends in tensile strength. The addition of either MWCNTs or TCNTs to PP could improve the tensile modulus, but when the filler’s content was higher than 8 wt%, the tensile strength of MWCNT/PP composites decreased. For example, the addition of 1 wt% MWCNTs to PP increased the modulus by 20%. At 10 wt%, the modulus still increased by about 45%, but the tensile strength decreased by about 10% (Figure 6). These results are consistent with what Koval'chuk et al. observed. 39 The agglomerates of MWCNTs present in the composites may act as defect sites affecting tensile deformation of a polymer. Thus, the effect of MWCNTs on tensile strength of PP is determined by two factors: reinforcement by MWCNTs and defects by MWCNTs. At low contents of MWCNTs (e.g. 1 wt%), the reinforcement is the major factor for increasing tensile strength of a polymer, while at high contents of MWCNTs (e.g. 10 wt% and above) the defects by MWCNTs become important, which decreases tensile strength.
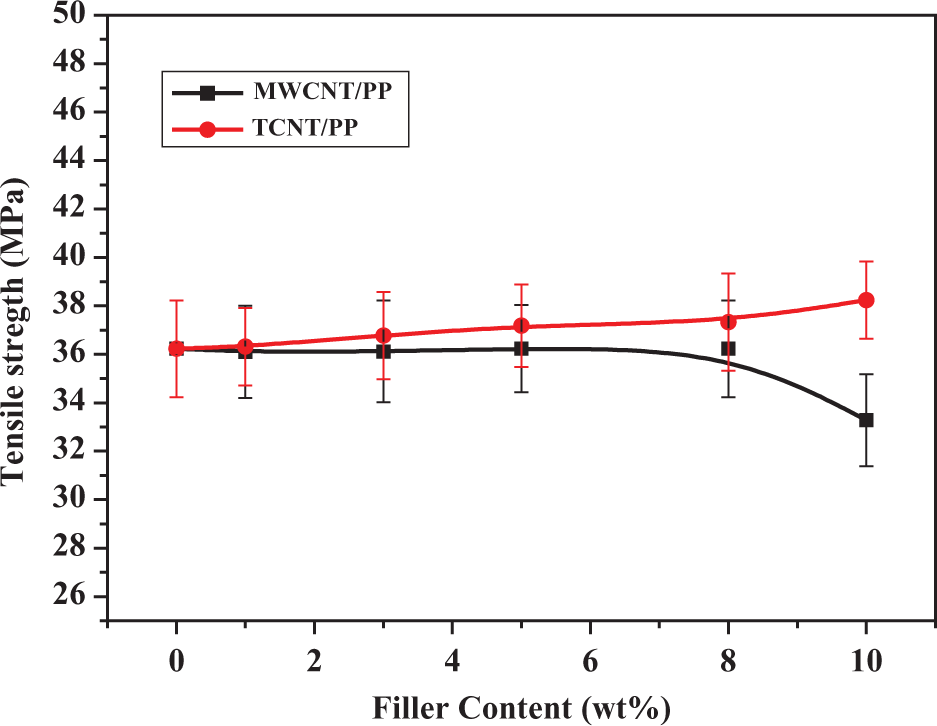
Tensile strength of MWCNT/PP and TCNT/PP composites. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
Compared with the MWCNT/PP composites, the TCNT/PP composites exhibited much higher modulus and consistently higher tensile strength. For instance, the addition of 1 wt% TCNTs to PP increased the modulus by 22%. At 10 wt% TCNTs, the modulus increased by almost 64% and the tensile strength was increased by about 6% (see Figures 5 and 6, respectively). Since both MWCNTs and TCNTs could act as defect sites affecting tensile deformation of a polymer, the reason why they affected the tensile strength differently can be considered from the fact that Triton X-100 improved the dispersion of MWCNTs in the matrix as well as the interfacial interactions between MWCNTs and the polymer matrix, as confirmed by the FESEM image (Figure 1). Moreover, considering the difference in agglomerates’ size between MWCNTs and TCNTs, it is obvious that the smaller agglomerates of TCNTs have less negative effect on tensile strength of a matrix polymer.
Crystallization of PP in composites
The crystallization properties of the PP composites were studied using DSC. Since both types of fillers (MWCNTs and TCNTs) do not show any thermal transitions in the experimental temperature range (40–200°C) for DSC, the crystallinity of PP, Xc , could be calculated according to the following equation:
where Wf
is the weight fraction of fillers, ▵Hm
is the measured melting enthalpy, and
Figure 7 shows the nonisothermal crystallization curves for the MWCNT/PP and TCNT/PP composites on cooling from 145°C at 10°C/min. For each composition, there is only one single exothermic peak appearing between 145 and 90°C, which was identified as the crystallization peak, and the peak temperature of the exothermic peak was defined as its crystallization temperature, Tc . The crystallization temperature of unfilled PP is 111.6°C. From Figure 7, it is interesting to note that the addition of both fillers could shift the crystallization temperature of PP to higher extent. The higher crystallization temperatures of PP in the composites compared to that of unfilled PP mean that crystallization started earlier in the composites than in the unfilled PP. This is because the fillers could act as nucleating agents to induce crystallization of the polymer melt in the cooling process. The effect of fillers on Tc is found to be different in such an order: MWCNTs > TCNTs. For example, 5 wt% MWCNTs could shift Tc of PP from 111.6 to 129.4°C, while 5 wt% TCNTs could change the Tc to 125.0°C. Thus, Tc of the 5MWCNT/PP was 4.4°C higher than that of the 5TCNT/PP. This result suggests that MWCNTs were a more effective nucleating agent for PP than TCNTs. The difference in affecting Tc between MWCNTs and TCNTs is considered to be due to the stronger interfacial interaction between TCNTs and PP in the TCNT/PP composites than that between MWCNTs and PP in the MWCNT/PP composites. The stronger interaction at the TCNT-PP interface would tend to ‘attract’ polymer chains from crystallization. As a result, the PP in the presence of TCNTs could not crystallize as early as it in the presence of MWCNTs. Some research works in the literature also reported that MWCNTs can act as a nucleating agent in many kinds of polymer matrixes to accelerate crystallization.41–46 Furthermore, no significant change in the melting point of PP was detected in the composites studied.
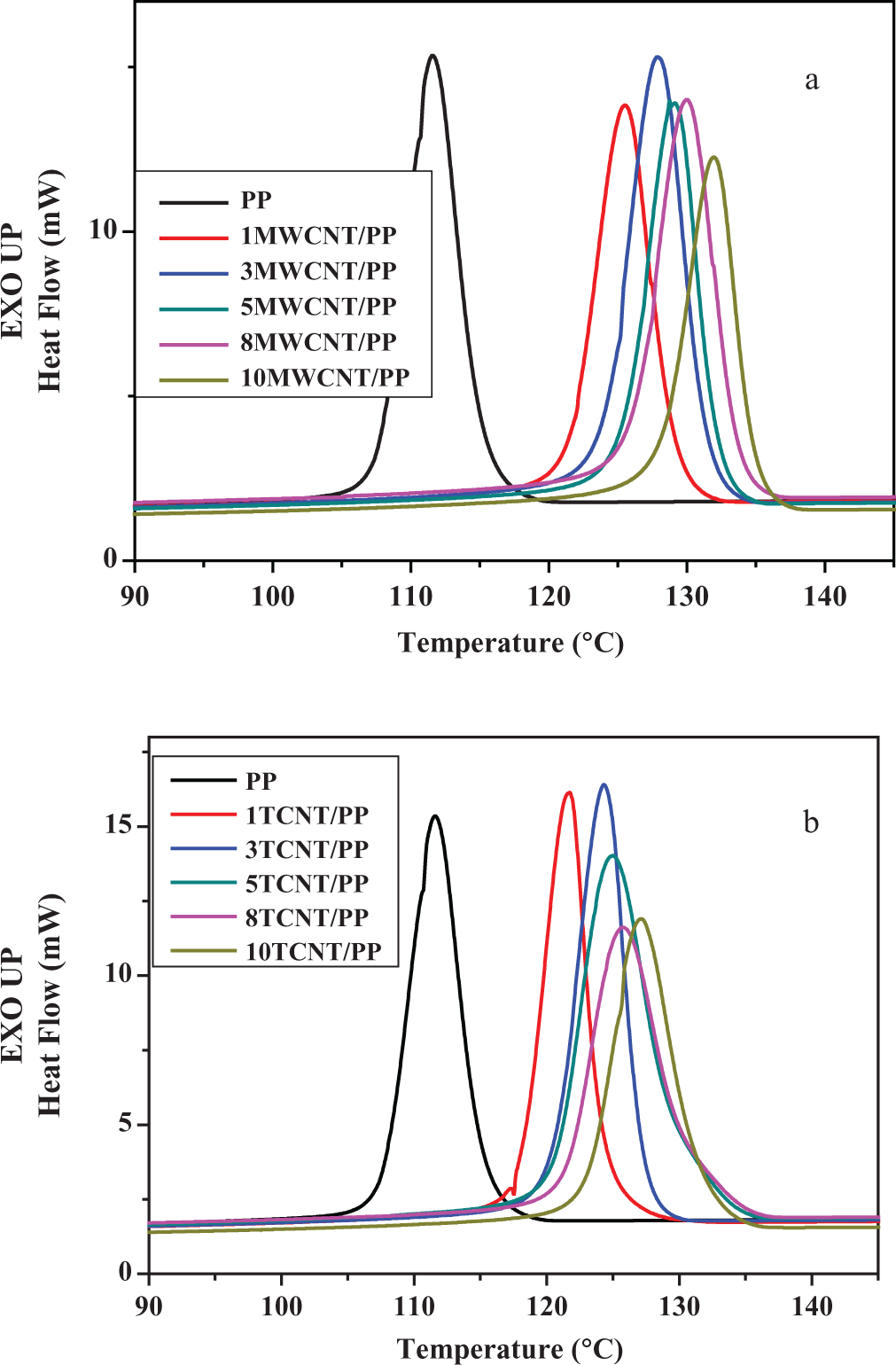
Nonisothermal crystallization curves for (a) MWCNT/PP composites and (b) TCNT/PP composites at a cooling rate of 10°C. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
The values of crystallinity obtained using equation (3) are shown in Table 1 and Figure 8. From Figure 8, we can see that the crystallinity of PP decreased evidently with increase in the content of fillers and both types of CNT/PP composites were lower in crystallinity than unfilled PP. The reason for the lower crystallinity of CNT/PP composites than unfilled PP can be explained as follows. The filler particles could provide nucleating sites for crystallization of PP, but at the same time the mobility of PP chains is restricted by solid filler particles. 47 In addition, solid filler particles may also act as barriers affecting crystal growth. As a result, although crystallization of PP in a composite could occur earlier at a higher temperature, the amount of PP crystals formed was limited due to the presence of filler particles. The crystallinity of TCNT/PP composites had the similar trend as the MWCNT/PP composites but was much lower than the latter. This can be explained by the stronger interaction at the TCNT-PP interface which would attract some polymer chains in the vicinity of TCNTs from crystallization. As a result, the PP in the presence of TCNTs could not crystallize as much as it in the presence of MWCNTs.
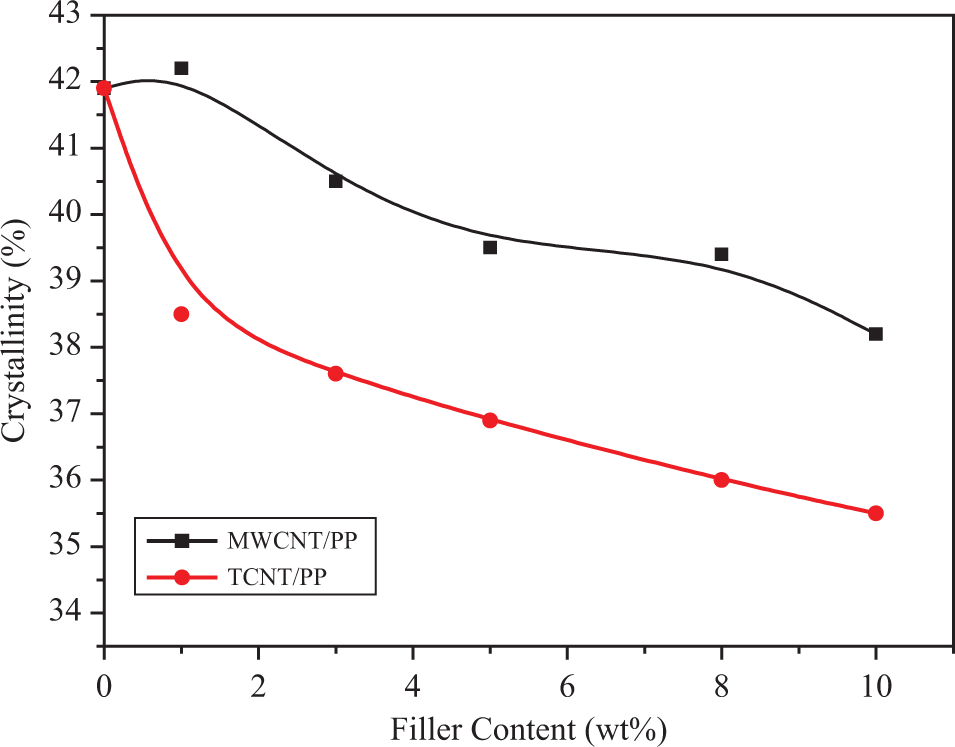
Crystallinity curves of MWCNT/PP and TCNT/PP composites. MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
The DSC data for MWCNT/PP and TCNT/PP compositesa
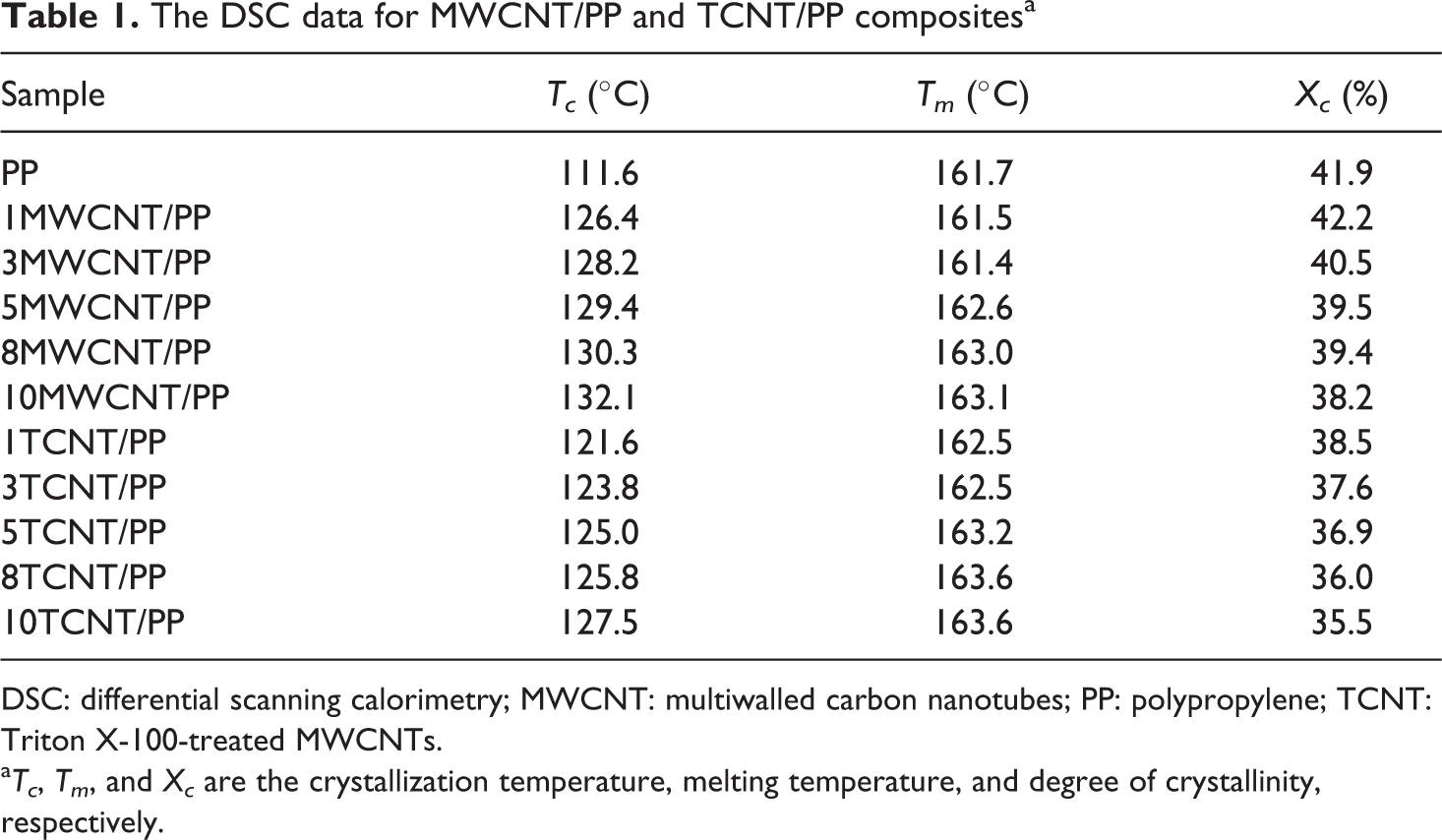
DSC: differential scanning calorimetry; MWCNT: multiwalled carbon nanotubes; PP: polypropylene; TCNT: Triton X-100-treated MWCNTs.
aTc, Tm, and Xc are the crystallization temperature, melting temperature, and degree of crystallinity, respectively.
Conclusion
In summary, we have successfully prepared TCNT/PP composites using Triton X-100 as a dispersing aid. The best usage of Triton X-100 was 80 wt% based on the MWCNTs.
Triton X-100 was able to significantly decrease the percolation threshold for electrical conductivity. The electrical conductivity of composites containing TCNTs was notably higher than those containing pristine MWCNTs, confirming the advantage of the TCNTs as an effective conductive filler.
Both pristine MWCNTs and TCNTs could improve tensile modulus and strength, but the TCNTs had a better effect than pristine MWCNTs due to the fact that Triton X-100 could help improving the dispersion of MWCNTs by reducing the surface energy and van der Waals forces between MWCNTs.
The DSC results confirmed that both pristine MWCNTs and TCNTs acted as nucleating agents in the PP matrix and promoted the nucleation for crystallization of PP. Compared with the MWCNT/PP composites, TCNT/PP composites had lower Tc and Xc because TCNTs enhanced the interaction at the CNT-PP interface.
Footnotes
Funding
This work was supported by the A*STAR SERC Grant (0721010018), Singapore.