Abstract
Three types of nanosilica-based urea–formaldehyde (UF) hybrid composite materials with a formaldehyde-to-urea (F/U) ratio of 0.8 were synthesized (UF with SiO2—Resin 1, UF + SiO2 + thiourea [TU]—Resin 2 and UF + SiO2 + thiosemicarbazide [TSC]—Resin 3). The thermal behavior of obtained materials was studied by nonisothermal thermogravimetric analysis (TGA), differential thermal gravimetry (DTG), differential thermal analysis (DTA) and differential scanning calorimetry (DSC) and supported by data from Fourier transform infrared spectroscopy (FTIR). Nanosilica-based UF hybrid composites have been irradiated (50 kGy) and after that their radiation stability was evaluated on the basis of thermal behavior. The percentage of free formaldehyde in all prepared samples was determined. DTG peaks of unmodified nanosilica-based UF resin (UF with SiO2) are shifted to a high temperature. The minimum percentage values of free formaldehyde (3%) for samples based on UF with SiO2 and UF + SiO2 + TSC after irradiation dose of 50 kGy are detected.
Keywords
Introduction
The study and application of UF resin have a history of more than 100 years. Owing to its low cost, simple synthetic technique and a certain water-resistant bonding strength, UF is one of the main glues used in wood industries so far. 1 As a typical amino resin, UF resin adhesive possesses some advantages, such as fast curing, good performance in the panel, water solubility and lower price. 2 UF resins offer unique potential technical advantages in a variety of applications, in abundance, unmatched competing products. Among those advantages are the above-mentioned low price, the nontoxicity of resin and resin products, and the environmental compatibility of resin-bonded products, such as particleboard. However, they lack in water resistance of the hardened resin owing to the reversibility of the amino ethylene bond and hence the susceptibility to hydrolysis.
The incorporation of nanoparticles into a polymer matrix introduces an enormous number of interfacial surface areas in the system and thus alters the crystallization kinetics of the polymer. It was reported that nanosilica was beneficial in improving the bonding strength of UF adhesive and abate its free formaldehyde emission. Chemical activities of the nanoparticles are excellent. 1 For example, when used as composite fillers, silica materials (quartz, fumed silica, precipitated silica, colloidal silica, etc.) drastically improved the mechanical, thermal and rheological properties of the polymer matrix composites. 3 The surface hydroxyl groups of silica including isolated, vicinal, and geminal silanols are believed to play a key role in most of the aforementioned properties. 4 These new materials, called nanocomposites or organic–inorganic hybrids, afford to combine both the advantages of the organic material as lightweight, flexibility and good moldability, and of inorganic materials such as high strength, heat stable, and chemical resistance. 5,6 Organic–inorganic hybrid materials composed of organic polymers and silica constitute an important class of advanced composites. Organic–inorganic hybrid (nanocomposite) offers a variety of advantageous properties in various applications. These materials have been fabricated in membranes, sheets, and fibers, and they are usually applied in such field as optical, mechanical, electronic, and biomedical appliations. 7,8 Introduction of silica particle into UF adhesive substantially decreases the formaldehyde release into the environment. Silica particles strongly sorbs up to 15 wt% of formaldehyde released in curing of UF resin. 9 –11 The release of formaldehyde is predominantly determined by the mole ratio of formaldehyde to urea in the adhesive system. The other method has been the use of chemical additives called formaldehyde scavengers to reduce formaldehyde emitted from UF resin. 12
High-energy radiation is a well-known technique for modification of polymers. However, little work concerning the effects of γ-irradiation on the thermal properties of modified UF resins has been done. The goal of this work was to determine the radiation stability of the synthesized nanosilica-based urea–formaldehyde (UF) composite materials based on their thermal behavior. The thermal behavior of three types of nanosilica-based UF composite materials (original and irradiated) was investigated using nonisothermal thermogravimetric analysis (TGA), differential thermal gravimetry (DTG), differential thermal analysis (DTA), and differential scanning calorimetry (DSC) supported by the data from Fourier transform infrared spectroscopy (FTIR).
Experimental
Materials
The following are the materials employed in this study: Urea (NH2)2CO (Alkaloid-Skopje, FYR of Macedonia); 35% formaldehyde CH2O (Unis-Goražde, Bosnia and Herzegovina); thiourea (NH2)2CS ([TU] Kemika-Zagreb, Croatia); thiosemicarbazide (TSC) (NH2)2NHCS and nano-SiO2 (Merck, Darmstadt, Germany) with specific surface of 200 ± 25 m2/g.
Synthesis of nanosilica-based UF hybrid composites
Three types of nanosilica-based UF hybrid composites (UF with SiO2—Resin 1, UF + SiO2 + TU—Resin 2, and UF + SiO2 + TSC—Resin 3) with formaldehyde-to-urea (F/U) ratio of 0.8 were synthesized using the same procedure. In the reaction vessel, 60 cm3 of distilled water and 0.1 mol of urea were mixed with magnetic stirrer. Then, 0.015 mol of TU (Resin 2) and TSC (Resin 3) were added. Other components such as 0.121 mol SiO2, 0.12 mol 35% formaldehyde, and 0.6 cm3 of concentrated sulfuric acid were added into the reaction mixture in following order: The pH value lower than 6 and the reaction mixture mixed for 3 h. Then 0.22 mol of sodium hydroxide dissolved in 6 cm3 of distilled water was added to the reaction mixture before stirring. The obtained pH value of resins was higher than 8. The modified UF resin was cured at 110°C for 3 h in a convective drying oven.
Techniques
γ-Irradiation
Irradiations were performed in air in the Co-60 radiation sterilization unit with the dose rate of 10 kGy h−1 and the total absorbed dose of 50 kGy.
Free formaldehyde determination
50 cm3 of 1 mol of pure sodium sulfite solution was prepared in a flask. Three drops of thymolphthalein indicator were added. The mixture was carefully neutralized by titration with 1 mol hydrochloric acid until the blue color of the indicator disappeared. A 0.5 g of the resin sample was added to the 25 cm3 of distilled water and 15 cm3 of 0.5 mol/dm3 sodium sulfate solution. A few drops of thymolphthalein indicator were added and the resulting mixture was titrated with 0.5 mol/dm3 hydrochloric acid until complete decolorization. The experiment was carried out once at the end of urea and formaldehyde condensation reaction.
The free formaldehyde CH2O (%) content was calculated from the Eq. (1):
Thermal analysis
The thermal stability was investigated by nonisothermal thermogravimetry (TG, DTA) using a Setaram Setsys Evolution 1750 instrument (France). Samples (6 ± 0.2 mg) were placed in alumina crucibles. An empty alumina crucible was used as a reference. The samples were heated from 30 to 500°C in a 20 cm3/min flow of argon with a heating rate of 10°C/min.
DSC measurements were performed using Setaram 151R (softver SETSOFT 2000 from Setaram, France) instrument in the temperature range from 30 to 450°C (heating rate was 20°C/min). Samples (3 ± 0.2 mg) were placed in alumina crucibles.
FTIR spectroscopy
The FTIR spectroscopy in transmittance mode was used for the characterization of the functional group of the resin. For the solid samples, KBr pellets with 1 mass% of the powdered material were produced. FTIR spectra of samples were recorded at room temperature in the wave number range of 4000–400 cm−1 on a Nicolet spectrophotometer (Model 380, Thermo Nicolet Corporation, Madison, USA), with 4 cm−1 resolution.
Results and discussion
During the manufacture of UF resin, the final reaction products between urea and formaldehyde can range from the simple monomethylolurea to very complicated cross-linked structures. During resin curing, a three-dimensional network structure is built up. The formation of linear condensation products in cure process begins at lower temperature if the resin contains greater amount of reactive methylol groups. 13 Depending on different synthesis conditions and technology, generally we used two-step reaction of urea and formaldehyde, which produces resins with a broad variety of both linear and branched chains. Formaldehyde can be bound with sorbents compatible with UF polymer, in particular, with nanosilica having large specific surface area. Bonding among organic molecules and surface of silica is resulting in the replacement of most of the strongly hydrophilic silanol functionality with a material exhibiting modified, and usually predictable, new or improved properties. 14 The amount of free formaldehyde contained in the UF resin is reduced by its sorption by nano-SiO2 particles. 15 Sorption of formaldehyde on the nanoparticle surface is accompanied by hydrophobic–hydrophilic effect caused by interaction among the surface and formaldehyde oligomers. The stability of the resulting structure is determined by the balance of these forces.
Free formaldehyde determination
The free formaldehyde percentage of the nanosilica-based UF hybrid composites unirradiated and irradiated samples is given in Table 1.
Percentage of free formaldehyde content.
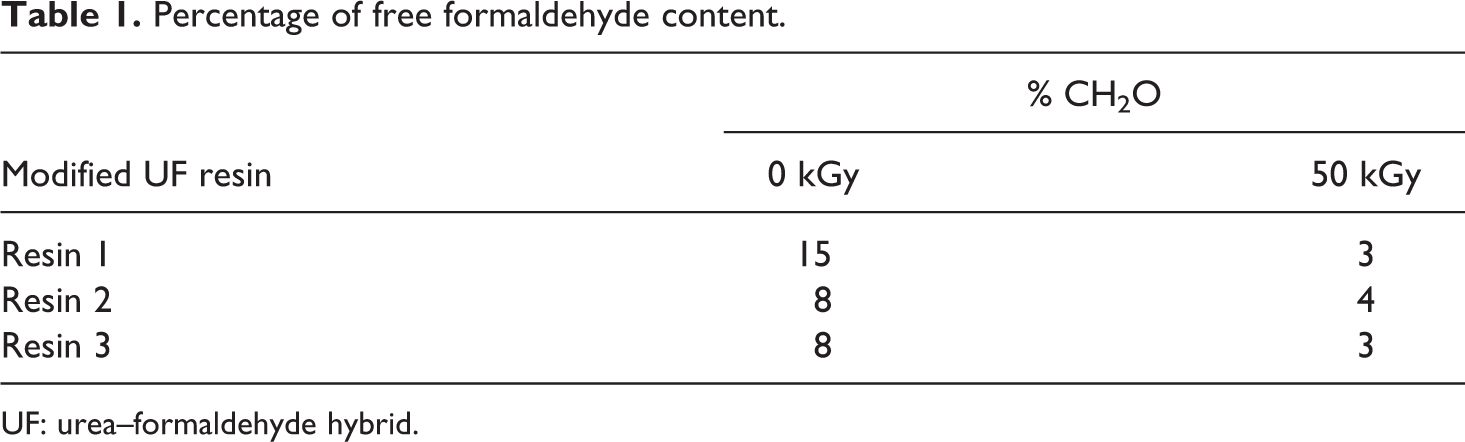
UF: urea–formaldehyde hybrid.
Lower levels of formaldehyde in Resins 2 and 3 (8%) compared to Resin 1 (15%) can be attributed to the presence of –C=S group (thiocarbonyl group) which acts as a scavenger for liberated formaldehyde. Apparently, free formaldehyde percentage reduced after irradiation, from 15% to 3%, 8% to 3%, and 8% to 4% for Resins 1, 2, and 3, respectively. The percentage of free formaldehyde decreased further, which could be explained as additional resin’s networking.
Thermal analysis
The thermal stability of composites was investigated using nonisothermal TGA, DTG, DTA, and DSC. The thermal behavior of modified nanosilica-based UF resins unirradiated and irradiated samples occurs in four, two, and three main stages for Resins 1, 2, and 3, respectively (Figures 1 –3). The percentage weight loss at different temperatures is summarized in Table 2 for all unirradiated and irradiated resins.
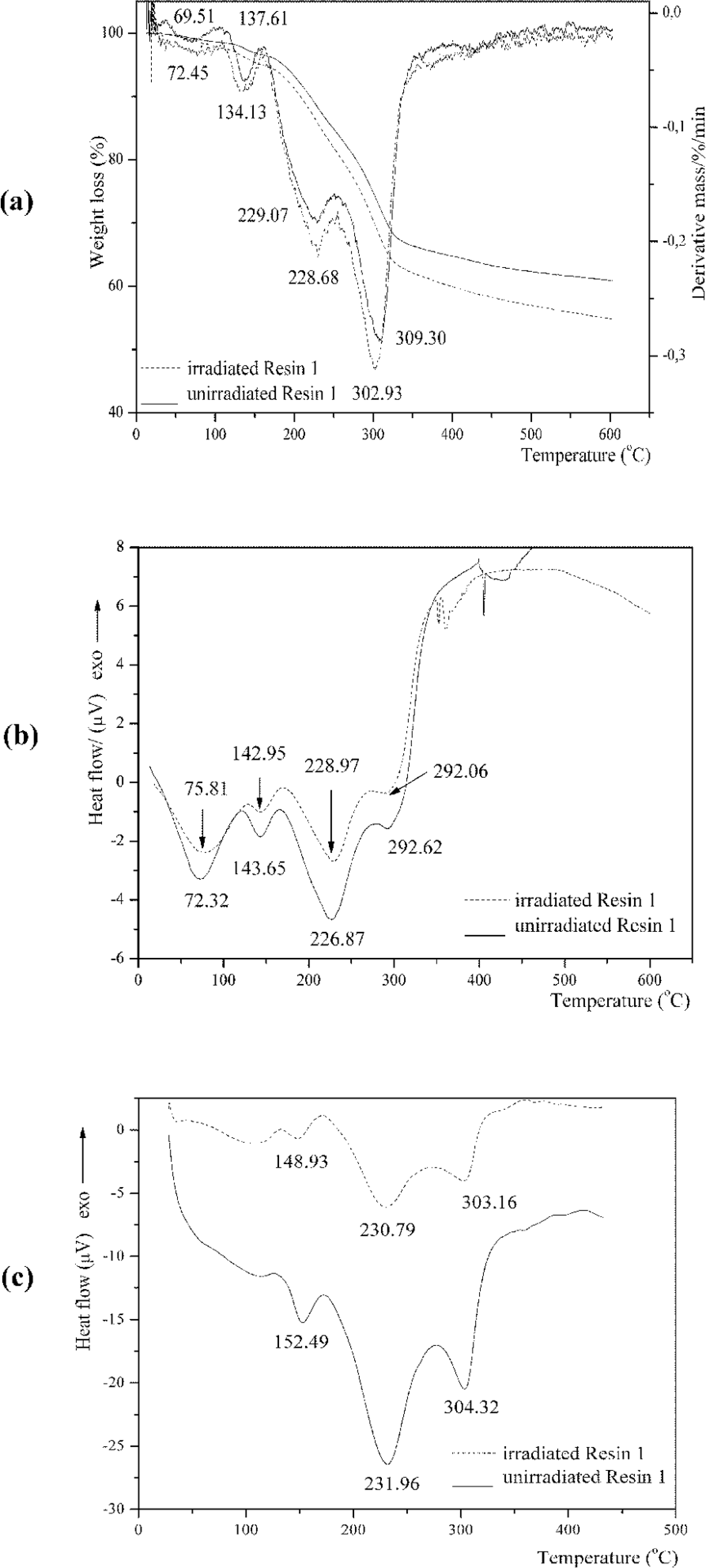
Thermogravimetric analysis/differential thermal gravimetry (TGA/DTG) (a), differential thermal analysis (DTA) (b), and differential scanning calorimetry (DSC) (c) curves of unirradiated and irradiated nanosilica-based UF composite (Resin 1).
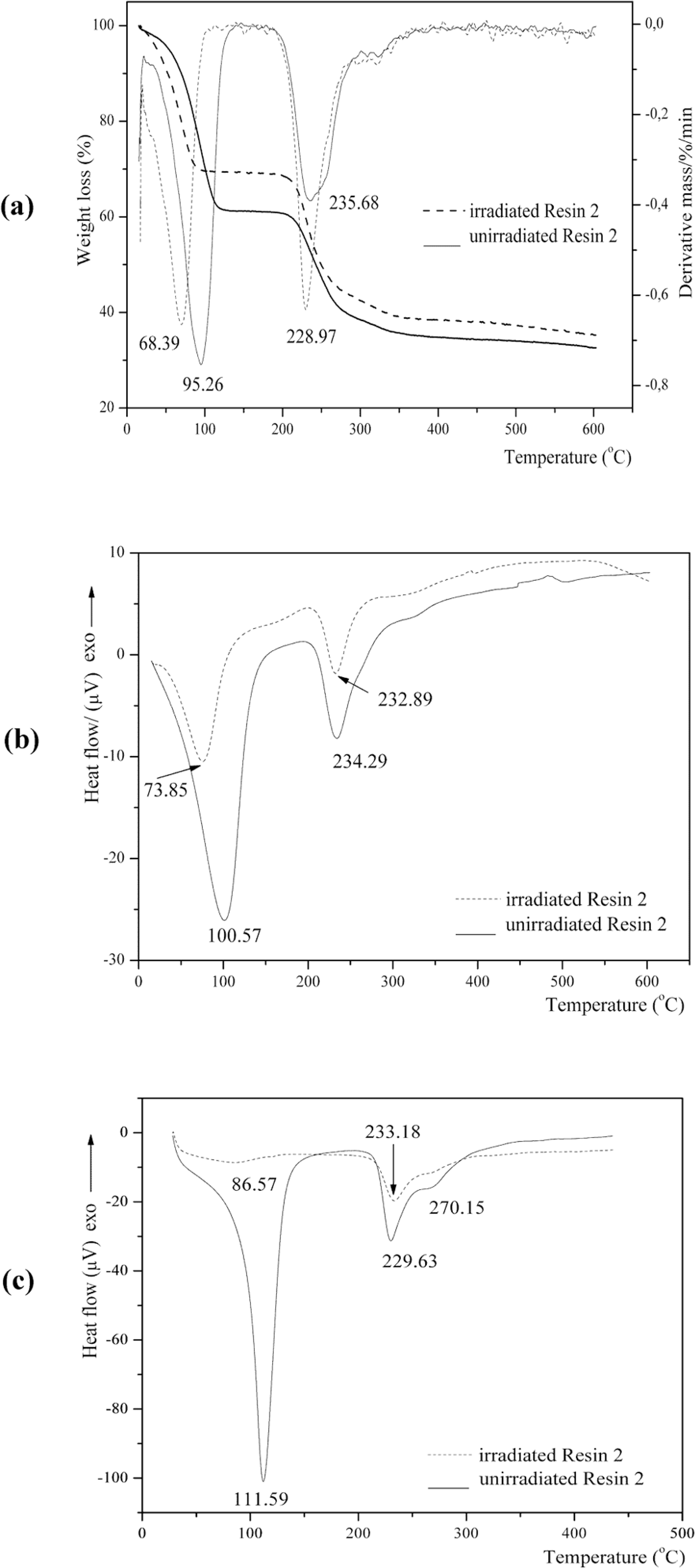
Thermogravimetric analysis/differential thermal gravimetry (TGA/DTG) (a), differential thermal analysis (DTA) (b), and differential scanning calorimetry (DSC) (c) curves of unirradiated and irradiated nanosilica-based UF composite with thiourea ([TU] Resin 2).
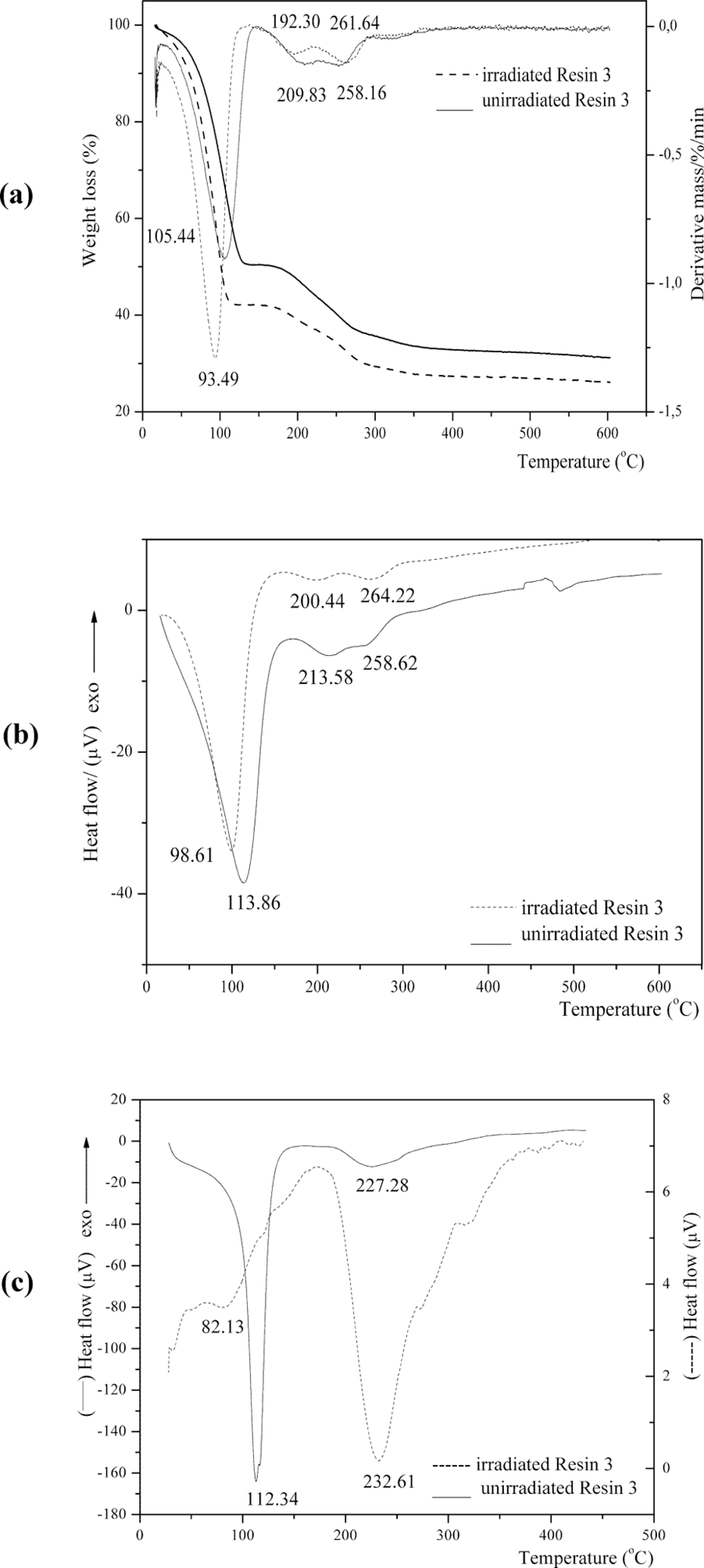
Thermogravimetric analysis/differential thermal gravimetry (TGA/DTG) (a), differential thermal analysis (DTA) (b), and differential scanning calorimetry (DSC) (c) curves of unirradiated and irradiated nanosilica-based UF composite with thiosemicarbazide ([TSC] Resin 3).
DTG, DTA, and DSC data of peak values, ▵
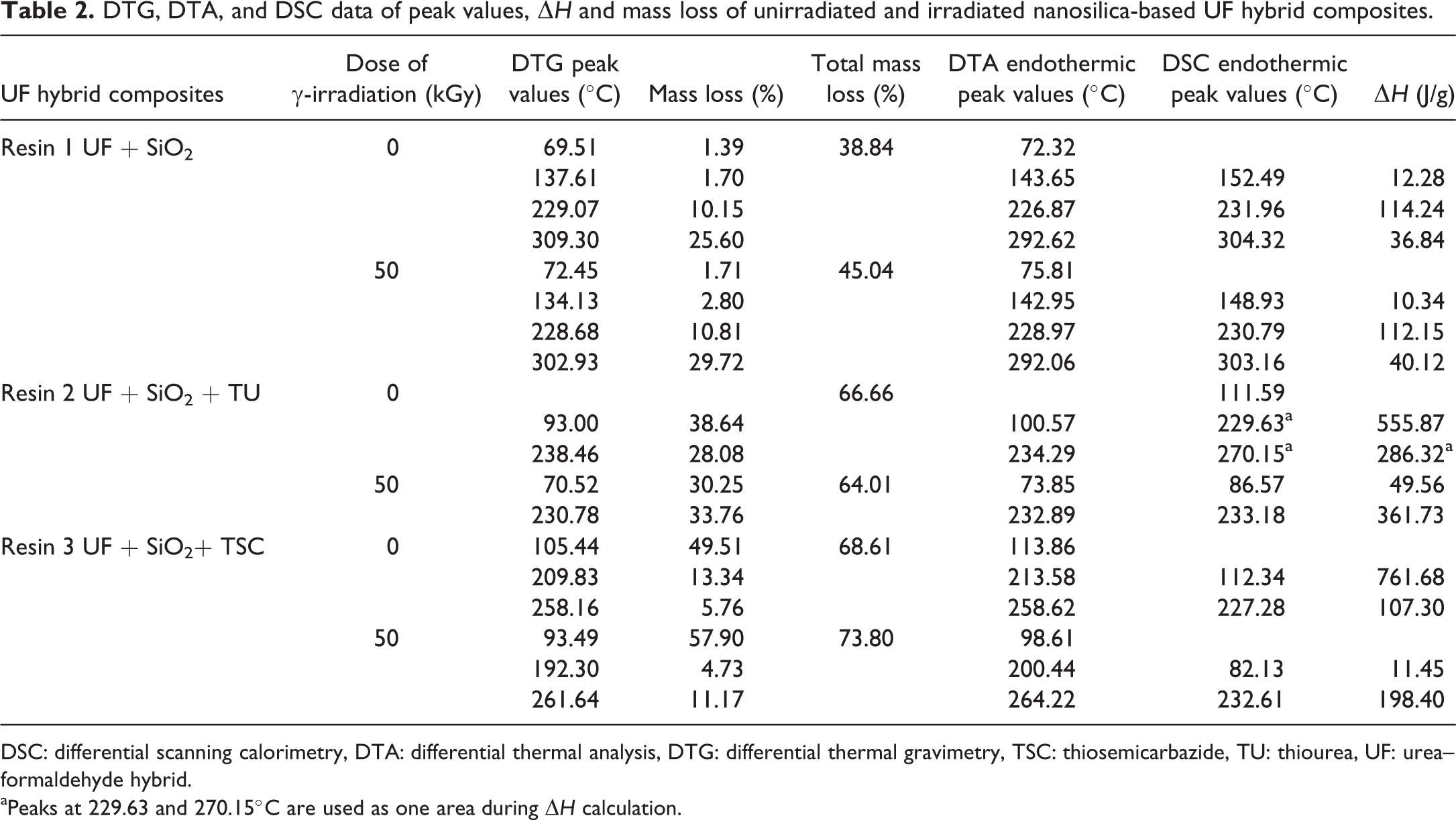
DSC: differential scanning calorimetry, DTA: differential thermal analysis, DTG: differential thermal gravimetry, TSC: thiosemicarbazide, TU: thiourea, UF: urea–formaldehyde hybrid.
aPeaks at 229.63 and 270.15°C are used as one area during ▵
Common to all resins was that the further heating up to 200°C causes no changes in the reaction heat values. At temperatures over 200°C, the resins began to decompose, showing a series of endothermic effects in similar temperature regions.
The thermal degradation up to 600°C of Resin 1 (Figure 1a) occurred in 4 successive steps. In each step, partial volatilization took place while the polymer undergoes chemical modification to give progressively more stable structures. 16 Above 200°C radicals formed by chain scission induced the formation of cyclic structures in the polymer which undergoes extensive fragmentation above 300°C.
The first-step degradation occurred at a temperature of 49.79–88.26°C; 66.72–112.45°C, and 71.66–125.61°C for Resins 1, 2, and 3, respectively. The mass loss was 1.39% for Resin 1, indicating water evaporation; and 38.64% and 49.51% for Resins 2 and 3 indicating water, formaldehyde, carbon monoxide and dioxide, methane, ammonia, monomethylamine, trimethylamine, methanedithione (CS2), iminomethanethione (HNCS), hydrazine (N2H4), and sulfur dioxide evaporation, which evolved as gaseous products. 16 –18 Due to evaporation of water from the system, the resin molecules became less mobile and the polymeric constituents of the surface area of filler slowed down the contact between reactive sites of the resin.
The second-step degradation started at 122°C and ended at 145.05, 213.73–281.57, and 179.59–268.42°C for Resins 1, 2, and 3, respectively. Third-step degradation started at 181.56–238.35, 282.96 and ended at 600°C for Resins 1, 2, and 3, respectively. The percentage of mass loss at second-step degradation was 1.70, 28.0, and 13.34% for Resins 1, 2, and 3, respectively, indicating polymer degradation, and for third-step degradation it was 10.15 and 5.76% for Resins 1 and 3, respectively.
The rate of the thermal decomposition reaction of all resins, whether unirradiated or irradiated, showed more than one maximum rate with increasing temperature. This behavior indicated that thermal decomposition of these polymers passed through multiple stages, depending on the state of decomposition and not on the components. 19
As it can be seen, in the Figures 1 and 3(a) and (b) the third and fourth degradation regions were overlapped for Resin 1, and second and third degradation regions for Resin 3, which led to 2 simultaneously occurring process degradation with a total weight loss of 35.75% and 19.10%, respectively.
The large endothermic peak of water evaporation with minimum at the 72.32, 100.57, 112.34°C for Resins 1, 2, and 3, respectively, derived from the initial resin water and also from condensation reaction. The endothermic peak with minimum at 226.87, 234.29, and 213.58°C can be attributed to the degradation of methylene ether bridges into methylene bridges and branching and cross-linking reactions in the resins network. 20 Degradation of cured resin begins the liberation of formaldehyde from dimethylene ether groups. This kind of destruction can be regarded as post-curing of resin, as released formaldehyde participates in further reaction, finally giving more stable methylene group. The endothermic peak with a minimum at 292.6 and 258.62°C for Resins 1 and 3, respectively, presented the decomposition of the most stable units in the UF resin-methylenediurea (Figures 1 –3b). These figures (Figures 1 –3b) show that the minimum of the endothermic peak attributed to water evaporation came to lower temperatures for Resin 1 than Resins 2 and 3. Noticeable degradation of cured resins was at 226.87, 234.29, and 200.44°C for Resins 1, 2 and 3, respectively.
Figures 1
–3(c) show DSC curves before and after γ-irradiations and obtained ▵
The DTG peaks shifted to a high temperature, indicating the increase in thermal stability of Resin 1.
High-energy radiation of γ-ray is a well-known tool for the modification of polymers. In polymer irradiation, two phenomena occur at the same time: cross-linking and chain scission. The generation of a cross-linked network between the polymer chains may enhance thermal and chemical resistance as well as stress cracking and dimensional stability. On the other hand, degradation resulting from ionizing irradiation may adversely affect the engineering properties of the polymer. The balance of cross-linking and scission reactions in polyolefin chains, exposed to high-energy radiation processes that produce free radicals, may result in good properties and new applications. These processes have the advantages of being clean and continuous with very good controllability. 22,23
TGA investigations indicate the decrease in the thermal stability of γ-irradiated resins, probably due to chain scission reactions. For all resins, cross-linking reactions are less compared to chain scission reactions, which result in decreased thermal stability.
FTIR spectroscopy
FTIR spectra and characteristic bands of original and irradiated samples are shown in Figure 4 and Table 3. Due to the complexity of structure in the polymer, the absorption frequencies are broad for resin spectra. Broadening may also be observed due to the presence of by-products in the resin, such as water and excess formaldehyde, which allows hydrogen bonding with the reactive functional groups like CH2OH, NH2, and NH present in resin samples. 24 However, spectra of cured resins showed sharper characteristic absorption peaks in this region. In the UF Resin 1, a medium absorption peak is observed at 3437 and 3342 cm−1, which is the characteristic absorption of the NH-stretching mode for NH2 group. Similar absorption bands for Resins 2 and 3 were recorded in the same region. A medium absorption band in all resin spectra appears in the range of 2969–2958 cm−1, which is ascribed to the symmetrical C–H stretching mode of CH2 of ether, CH2OH, and N–CH2. A very strong absorption band is observed around ∼1660 cm−1 in all spectra, which may be assigned to the C=O stretching vibration (amide-I) in CONH2 group. The strong absorption bands around 1550 cm−1 can be assigned to NH bending mode in 2°-amine (amide-II). The cross-linking between two methylol groups provide ether linkages (–CH2–O–CH2) to which –NH is attached to both sides. A weak absorption band around ∼1440 cm−1 can be assigned to CH mode in –CH2O, N–CH2–N and NH–CS–NH thiocarbonyl group. The weak absorption bands around 1390–1351 cm−1 for all polymer samples may be ascribed to C–H bending mode in –CH2/–CH2OH/N–CH2–N. The medium absorption band in the region of 1150–1130 cm−1 may be assigned to asymmetric stretching vibrations of –N–CH2–N–, ν(C–O–C) of ether linkage and ν(Si–O–C) and νass(Si–O–Si) of siloxane or silicone. 1,7 The band at ∼780–880 cm−1 may be assigned to asymmetric stretching vibrations of C–O in CH2OH, N–H wagging in 1° and 2° amines and ν(Si–O) and 473 cm−1, respectively. Stretching vibrations of C–S and bending vibrations of CH appeared at 617–620 cm−1. δs(C=S) and νas(C=S) vibrations appear at 781.13 and 1455.32 cm−1, respectively. 25
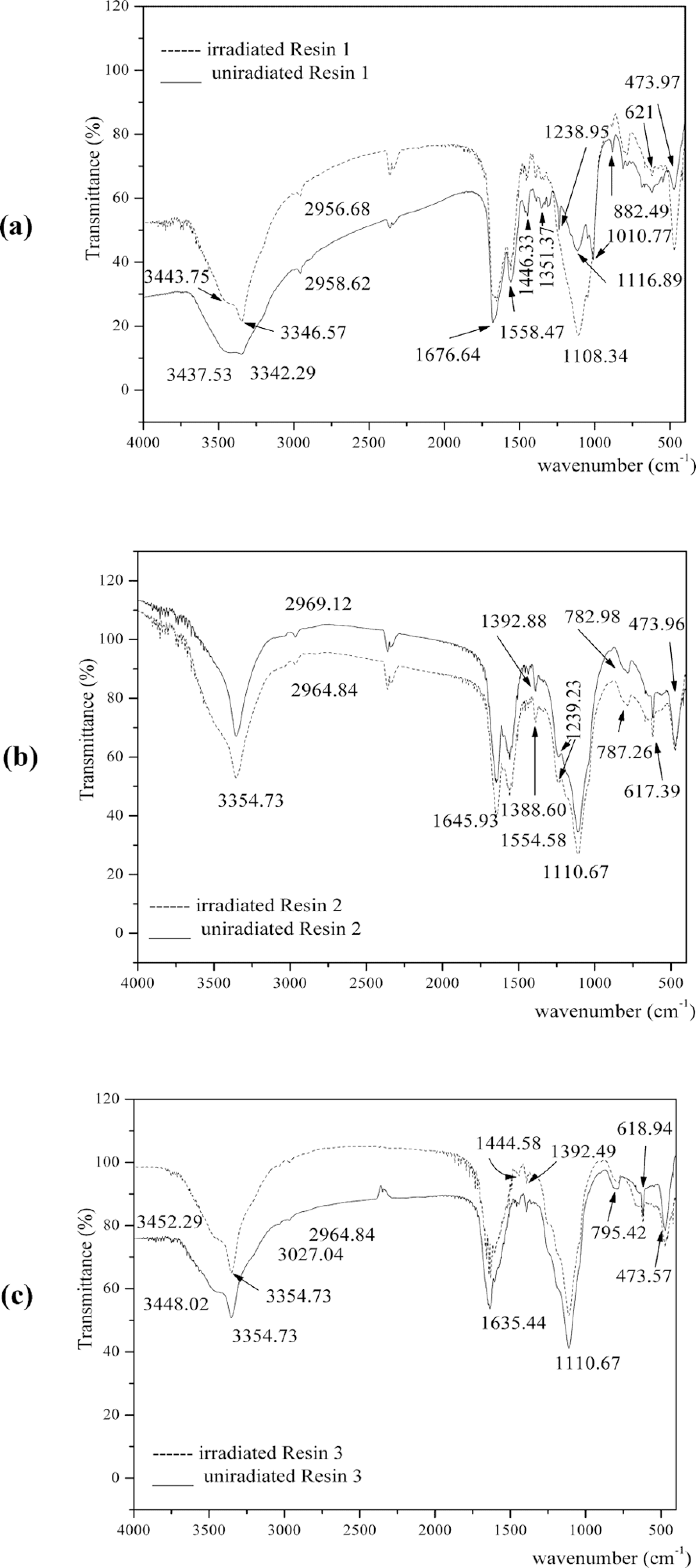
Fourier transform infrared spectroscopy (FTIR) spectra of unirradiated and irradiated nanosilica-based UF composites: (a) Resin 1, (b) Resin 2, and (c) Resin 3.
The intensity of all bands in FTIR spectra of modified UF resins is increased and shifted to lower wave number according to lower thermal stability after γ- irradiation.
Important IR-characteristic bands observed for unirradiated and irradiated nanosilica-based UF hybrid composites.
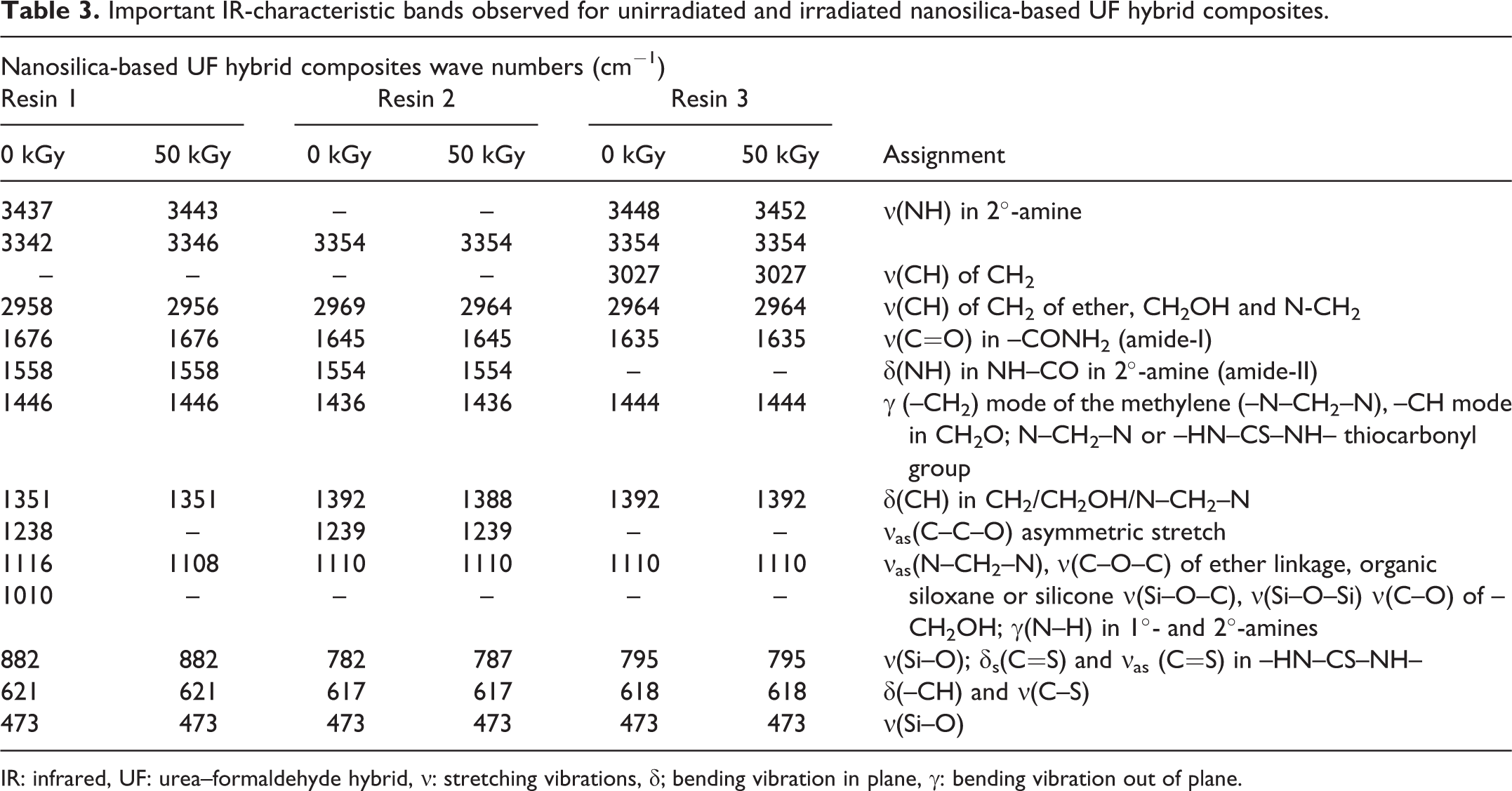
IR: infrared, UF: urea–formaldehyde hybrid, ν: stretching vibrations, δ; bending vibration in plane, γ: bending vibration out of plane.
Conclusion
The percentage of free formaldehyde for nanosilica-based UF hybrid composite modified with TU and TSC is less than that of unmodified UF resin. The percentage of formaldehyde is significantly reduced after γ-irradiation. TG/DTG and DTA curves show that the modified UF resin has good thermal stability. TGA investigations indicate decreased thermal stability of γ-irradiated resins. DSC investigations indicate increase in the values of ▵H after γ-irradiation of all resins. The intensity of all bands in FTIR spectra of nanosilica-based UF hybrid composites is increased and shifted to lower wave number exposed according to lower thermal stability after γ-irradiation.
Footnotes
Funding
Financial support for this study was granted by the Ministry of Science and Technological Development of the Republic of Serbia (Projects Numbers 45022 and 45020).
