Abstract
The mass manufacture of tires and the difficulty in storing or disposing them constitute a serious environmental problem. At present, various recycling methods for tires are used, such as grinding, which separates the steel and fibers from the vulcanized rubber, then using the rubber in numerous industrial applications such as pavements, insulators, footwear, etc. The aim of this article focuses on finding a new application for the ground tire rubber (GTR). This would allow including GTR in industrial applications of recycled high-density polythene (HDPE). Tire dust and HDPE thermoplastic have been mixed, and the maximum accepted values of GTR concentration that can be admitted while keeping dielectric, mechanical, and thermal properties within acceptable values, as well as initial polymer microstructure, have been checked. In addition, the suitability of the compounds for antistatic applications is studied. The recycled tire dust which results from the industrial milling processes has been divided by sieve in three different categories according to the size of the particles (<200, 200–500, and >500 µm). This has then been mixed with HDPE in different GTR concentrations (0%, 5%, 10%, 20%, 40%, 50%, and 70%) in order to establish its conduct through electrical, mechanical, thermal, and microstructure tests, which will be held in a temperature range 30–120°C, and with a frequency range 1 × 10−2 and 3 × 106 Hz. The thermal tests determined the enthalpies and fusion temperatures which allow for the observation of changes in crystalline and microstructure of the matrix, and finally, the fracture surfaces of the compound samples have been evaluated using scanning electron microscopy.
Keywords
Introduction
The real problem of used tire accumulation1–3 has led to efforts by the international scientific community to provide solutions designed for their recovery and reusability. 4 Many plastic materials incorporate elastomers to improve their toughness, usually a thermoplastic or heat-stable polymer acts as matrix and the elastomer as disperse phase.5–7 Moreover, as in other two-phase polymer blends,8,9 the interfacial compatibility between the components is important for achieving the desired properties.
One way to increase the compatibility between components is to reduce the degree of ground tire rubber (GTR) crosslinking by means of devulcanization.10–13 Another way to increase interface union is by means of pretreatment with acids, 14 that are produced on the surface of the elastomer, micropores that facilitate the union between phases.15,16 But this method increases the price of the final product, apart from having only a slight influence in improving the properties of the compound. Better results are given using different filler particle sizes, which is the alternative studied in this article. 17 However, as particle size is limited by the technical procedures of pulverization, a simple and economic way has been opted for in order to obtain the classification for the three desired particle sizes (<200, 200–500, and >500 µm), as sieved, although this method has the disadvantage of only making use of a part of the initial amount of GTR. The objective of this study is focused on looking for a new application for GTR mixed with recycled high-density polythene (HDPE), verifying the concentration values admitted by the new compound for which initial values of the polymer’s dielectric and mechanical properties are acceptable,18,19 to enable them to be used in various industrial applications. In order to do this, different HDPE/GTR concentrations have been analyzed (from 0% to 70% GTR), with the three particle sizes indicated. The interface compatibility between components was studied previously using scanning electron microscopy (SEM), and for an improved analysis of the compound, a calorimetric analysis was also undertaken.
Methodology
Materials
HDPE ALCUDIA 4810-B is produced by REPSOL. It is a white compound based on a medium-molecular weight hexane copolymer especially designed for the water bottle manufacturing by extrusion blow molding, and it has a melt flow index of 1 g/10 min at 190°C and a density of 960 kg/m3. The GTR, with a particle size less than 700 µm, was provided by Alfredo Mesalles (Spain). The decomposition temperature range starts at 210°C. By thermogravimetric analysis, it was verified that carbon black content was about 35%. The original GTR was separated by sieving into three categories of particles: <200, 200–500, and >500 µm.
Composite Preparation
The recycled tire powder was dried in an oven at 100°C for 24 h. Six samples of HDPE/GTR compound, varying the composition (5%, 10%, 20%, 40%, 50%, and 70% weight of GTR), were prepared for each particle size. The mixing process was done with a Brabender machine, at 153°C to prevent degradation of the polymer, and a mixing time limited to 4 min. HDPE/GTR laminates (170 × 170 × 2 mm3) were obtained using a hot plate press at 100 kN and 170°C for 10 min. The cooling stage was done with a closed water circuit, which was held in the same press and pressure for 5 min. Samples for testing were properly set up according to the specifications of ASTM-D-638 type V. A sample of pure HDPE was also prepared with the same requirements in order to obtain comparable results.
Scanning Electron Microscopy
SEM was used to analyze the fracture surface of tensile samples. It is possible to analyze the effects of this filling material in the matrix by observing the environment of the reinforcement particles. Some images of the samples were analyzed according to particles size and GTR concentration. A JEOL 5610 microscope was used, and the samples were previously coated with a thin layer of gold in order to prevent the accumulation of static charge in the surface.
Calorimetric Analysis
The dynamic thermal behavior of the samples was analyzed using heat flow differential scanning calorimetry (DSC). The measurements were made with a Mettler TA4000 thermoanalyzer coupled with a DSC 30 apparatus. The sample mass was between 3.0 and 3.5 mg, and it was small enough to avoid problems caused by heat and material transfers. Temperature and enthalpy were calibrated using In and Pb as standards. Samples were heated from 40°C to 200°C at a heating rate of 10 K/min and using synthetic air as purging gas at a flow rate of 40 mL/min.
Mechanical Analysis
Stress–strain tests were carried out with an Instron 3366 – 10 kN universal machine, following the specifications of ASTM-D-638 type V. The test speed was 50 mm/min. The test temperature was 23 ± 2°C, and the relative humidity was of 50 ± 5%. The study of mechanical properties, according to the GTR concentration in the matrix and the particle sizes includes the Young’s modulus, tensile stress, elongation at break, and toughness. Five specimens for testing were used. The mean and standard deviation were calculated for each magnitude, leaving out of account those samples with such a result that was proved to be defective.
Dielectric Analysis
The dielectric analysis was performed only with particles with sizes below 200 µm, as they have provided the best results in previous tests. The parameters and dielectric magnitudes were measured by means of dielectric analysis with BDS40 equipment which has a built-in temperature sensor Novotherm of Novocontrol using a compression mold of 2 cm diameter, and 400 µm of thickness. The measurements were undertaken on a frequency range 1 × 10−2 and 3 × 106 Hz, with a temperature scan between 30°C and 120°C and at a speed of 3°C /min using parallel plate sensors.
Results and Discussion
Scanning Electron Microscopy
Some SEM micrographs
2
of the fracture surface of the tensile test HDPE/GTR tubes
19
are shown in Figure 1. GTR particles do not reach its decomposition temperature range when mixed with the HDPE. So, some dispersed GTR particles can be seen in the HDPE matrix homogeneous medium, as displayed in the micrographs, while the HDPE (thermoplastic polymer) reaches this temperature and the dispersion is proper. The result is a microgranulate mass with a degree of dispersion that depends on the mixing time and temperature, and which does not facilitate cohesion between phases.
3
SEM micrographs of HDPE-GTR for several concentration percentages: (a) HDPE/GTR-10%; (b) HDPE/GTR-20%; (c) HDPE/GTR-40%; and (d) HDPE/GTR-70%.
Figure 1(a), shows the compound with a low GTR concentration (10%) and particles with sizes below 200 µm. It shows that the reinforcement particle is integrated in the matrix giving good interface adhesion. On the surface of the particle, no holes are apparent and the HDPE fragments spread across the entire surface of the particle and stick to it.
Figure 1(d) shows, on the other hand, a compound with high filler concentration (70%) and the same particle size, which results in an increase in the faults and cracks in the matrix, worsening interface adhesion. In this case, the percentage of HDPE is not sufficient to cover the GTR particles, making their union more difficult, with surface cracks and pores of a considerable size appearing. The GTR particles are clean and easy to extract, so the fracture has taken place through the matrix interface. On the other hand, with high GTR concentrations, greater possibilities of particle agglomeration exist, acting this like a large-sized particle.
For intermediate concentrations of GTR in the matrix, different levels of adhesion appear between the components. With 20% GTR (Figure 1(b)), the interfacial cohesion is still acceptable, whereas with percentages of 40% GTR (Figure 1(c)), the particles start to show important discontinuities on their surface with pores and cracks of considerable size that weaken the compound.
Thermal Properties
The calorimetry applied to the compound materials has been used as a tool to detect possible changes in the crystallinity and microstructure of the matrix when adding a second component as reinforcement. 20
Figure 2(a), shows that for small particle sizes (<200 µm), fusion enthalpy tends to increase slightly (5%) when the quantity of GTR is increased until 10%. This phenomenon could be related to the nucleation of the small GTR particles inside the matrix during the preparation of the samples for fusion. The small particles of tire act as nucleation agents increasing the compaction of the structure on its borders. The improvement in crystallization causes an increase of fusion enthalpy which is also related to the improvement in the mechanical characteristics achieved for these particle sizes, such as traction resistance and Young’s modulus. For concentrations of 20–40% GTR, a decrease in fusion enthalpy of 10–15%, respectively, is observed, due to the fact that the effect of particle nucleation is offset by poor interfacial adhesion between components, creating fracture surfaces. With particle sizes over 200 µm, the results show a decrease in fusion enthalpy of 8–14–20% for GTR concentrations of 5–10–20%, respectively. The large particles, on entering the fusion process display a surface with pores and cracks which, if sufficiently large, affect an important part of the HDPE in contact with this surface. This hinders the possibility of forming a compact structure to the compound and facilitating the amorphous state of the particle’s surface in this closed space. For high concentrations (40–50%), the process becomes stable, with no significant difference between particle sizes (decreases of 25–28%, respectively).
(a) Melting enthalpy and (b) onset temperature vs tire content for HDPE-GTR and particle sizes.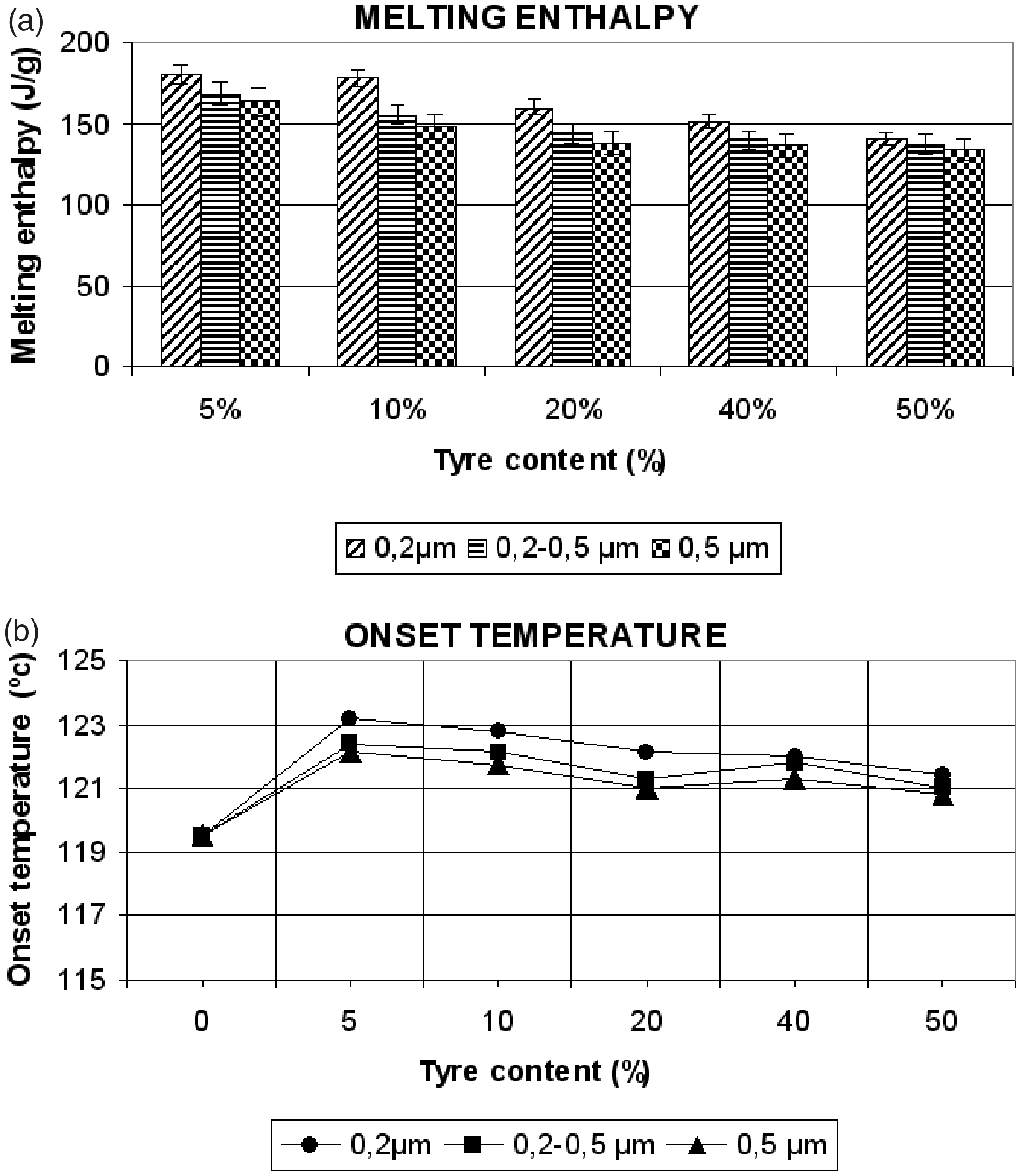
Calorimetric data related to samples’ fusion temperature are shown in Figure 2(b). An increase in fusion temperature could be associated to a more compact crystalline structure. In this case, the results demonstrate that for low GTR concentrations (5–10%) and small particle size (<200 µm), there is a tendency towards an increase in fusion temperatures (6%), which demonstrates its improved adhesion to the matrix. However, the differences in temperature are not significant and it is difficult to establish conclusions on the effect of GTR particles in the HDPE microstructure.
Mechanical Properties
Figures 3 and 4 shows the results of the mechanical properties determined by means of the stress–strain test. These properties were analyzed for different concentrations and GTR particle sizes in the HDPE matrix.21,22
(a) Elongation at break (%) and (b) toughness (J) for different HDPE/GTR concentrations and particle sizes. (a) Young’s modulus (MPa) and (b) tensile strength (MPa) for several concentrations HDPE/GTR and particle sizes.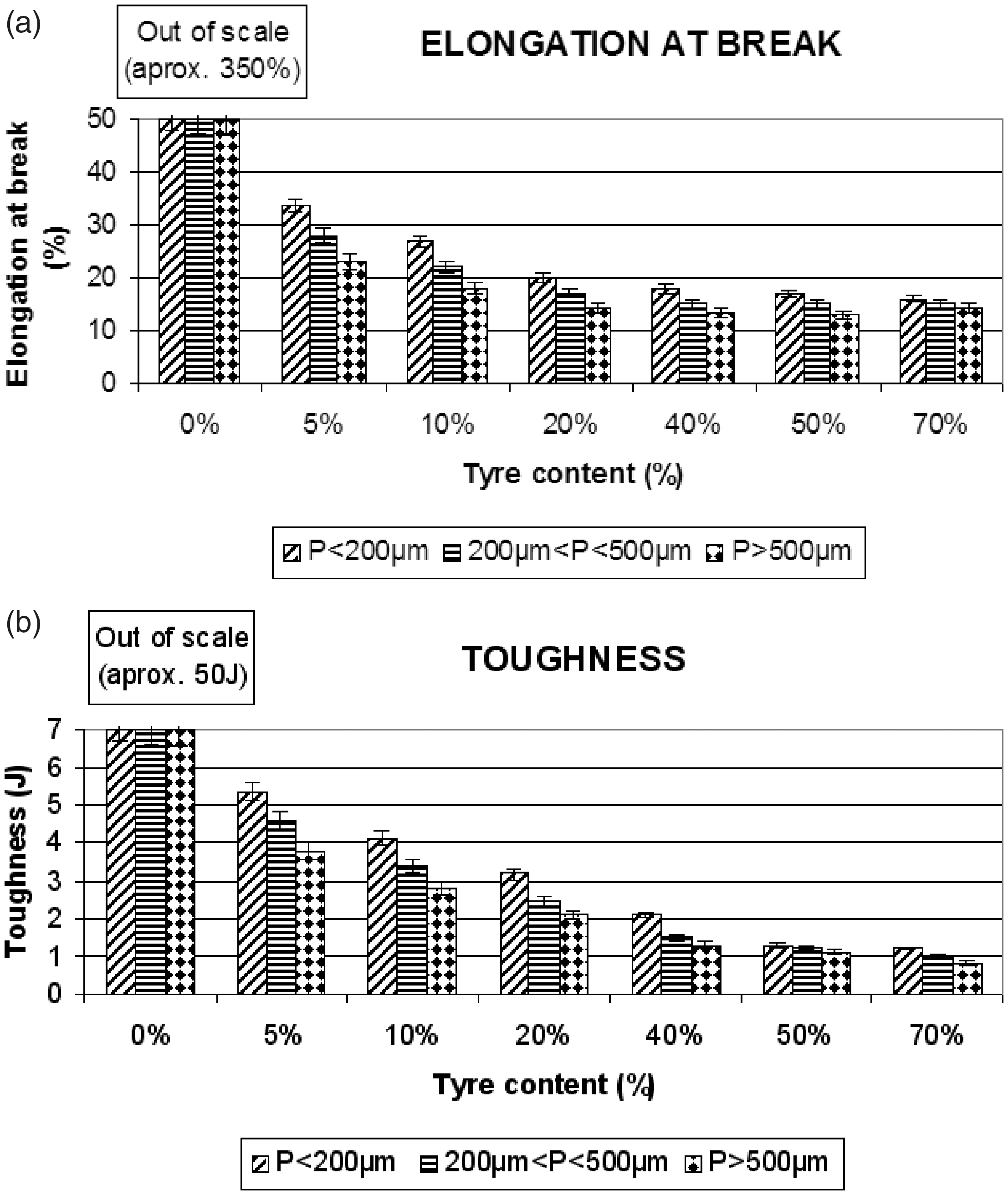
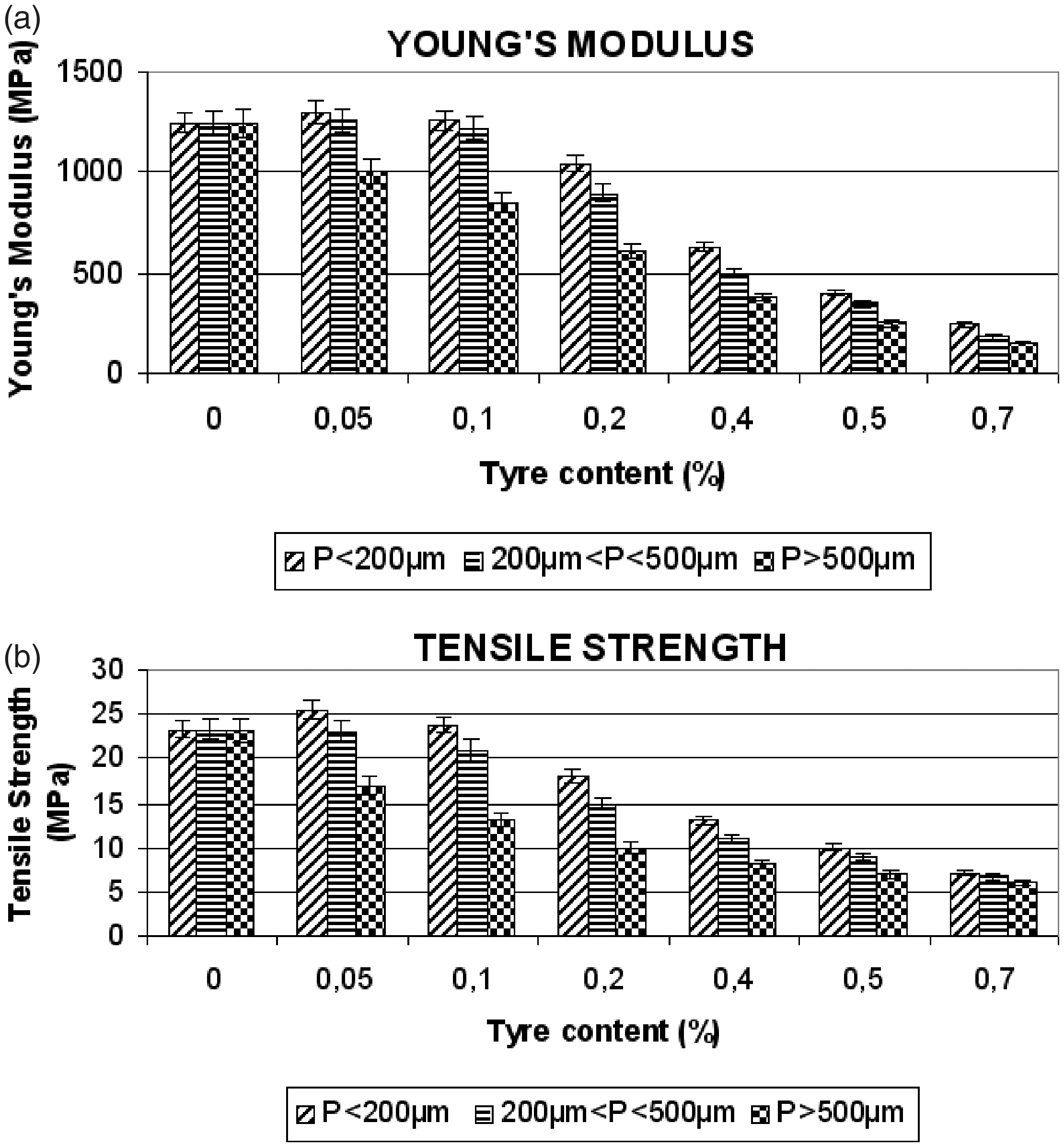
Figure 3(a) shows the compound’s Young’s modulus in which, in contrast to other materials formed by the addition of elastomers, the GTR causes a decrease as much in deformation capacity as in the ductility and plasticity of the HDPE matrix.23,24 The rigidity is seen (for concentrations of up to 10% GTR) to increase with regard to the original HDPE (5–8%) for small-sized particles (<200 µm) and stays practically the same for particles between 200 and 500 µm. This is due to the fact that the composite materials with large particles have a great probability to reduce the interaction in the interphase and the poor adhesion generates that the propagation of some fissure take place very easy. This is obvious in the case of particles over 500 µm, which show a significant decrease in values of mechanical properties in all the cases studied (decreases of 20–35% for concentrations of 5–10%, respectively). When the content of the reused tires increases, interfacial adhesion weakens causing a decrease in rigidity in all cases, no matter the particle size. For concentrations of 40% and 70% GTR, the values are three and five times less than those of the pure HDPE.
Figure 3(b) shows the tensile strength, which displays a behavior similar to Young’s modulus. In this way, for low GTR concentrations (5–10%) and small-sized particles, the value of tensile strength increases slightly (4–8%), whereas after concentrations greater than 10%, the values drop markedly. For 20% GTR, tensile strength diminishes by 25% for particles less than 200 µm, and 35% for particles between 200 and 500 µm. For larger concentrations (40% GTR), the values stabilize regardless of, in this case, particle size, with values some 2.5 times as low as those of pure HDPE. With regard to particle size greater than 500 µm, even for low GTR concentrations (5%), tensile strength diminishes by 25%.
The addition of reused tire particles in all the compounds produces a notable decrease of elongation at break (Figure 4(a)) and of toughness (Figure 4(b)). The tenacity and the elongation at break of the GTR compounds display values much lower than the matrix of pure HDPE (around seven times lower). Considerations on particle size are hardly important in this case, showing differences between particle sizes which do not reach 14%. The decrease in elongation at break has been documented in other compounds25,26 and it is related to the imperfect interfacial adhesion between the components. The reduction in the rubber’s capability to deform influences the decrease in stretching and, consequently, a decrease in hardness.
Electrical Characteristics
The dielectric analysis was performed only with particles with sizes below 200 µm, as they have provided the best results in previous tests.
Conductivity
Figure 5 displays the real conductivity results of the different compounds HDPE/GTR at 30°C and 120°C. The pure HDPE conductivity measurements incorporated an important dispersion due to the low conductivity of such material and have not been included. In the zone corresponding to high frequencies of the log–log plots, the conductivity dependency on the frequency is seen as linear. This fits a sublinear dispersive conductivity model Equation (1) as it is common in polyethylene and similar disordered materials,
27
Conductivity σ for HDPE/GTR at (a) 30°C and (b) 120°C in function with the frequency: ○, HDPE; □, HDPE + 5%GTR; ◊, HDPE + 10%GTR; Δ, HDPE + 20%GTR; ⊲, HDPE + 40%GTR; ▿, HDPE + 50%GTR; and ⊳, HDPE + 70%GTR.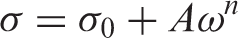
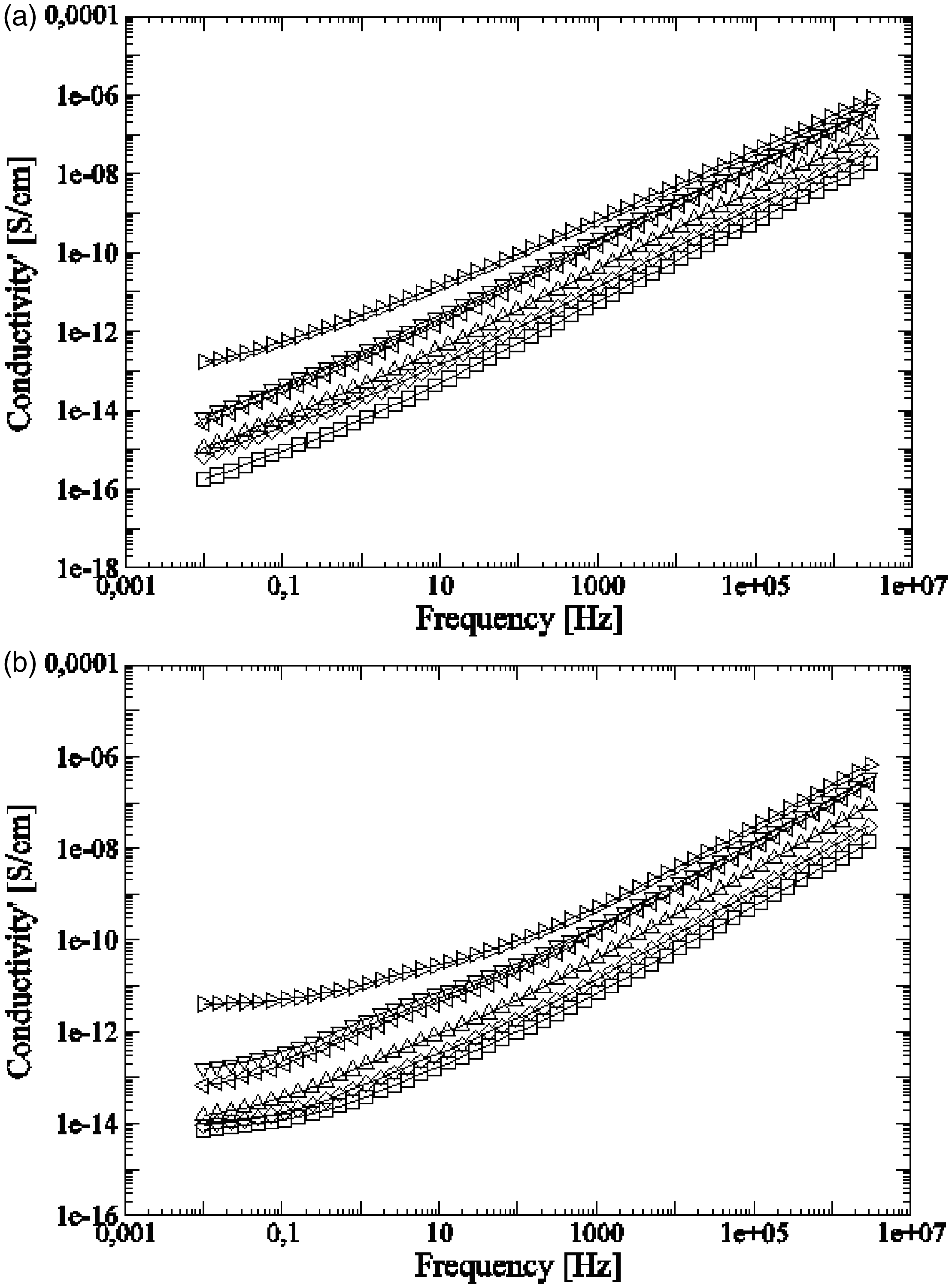
Since HDPE has a very low DC conductivity, for most temperatures and GTR concentrations, the crossover frequency that delimits one or other regime is located below the range of frequencies analyzed. Only at high temperatures or for high GTR concentrations, it is possible to see clearly the change in the slope at the low frequencies region of the spectra. The reason is that by increasing the temperature or the GTR concentration, and therefore the black carbon amount in the composed material, the DC conductivity rises and shifts the crossover frequency to higher values. As it could be expected, there is also a direct relationship between the GTR concentration and the conductivity in both DC and dispersive regimes. The carbon black present within the GTR particles is much more conductive than the insulating polymers and it is usually used to improve the electrical properties of these materials. 28 In this case, the addition of GTR raises the DC conductivity up to values suitable for antistatic applications (10−11 to 10−16 S/cm 29 ). On the other hand, when comparing both graphs (Figure 5(a) and (b)), it is observed how the conductivity at low frequencies is about one or two orders greater in size when measured at 120°C basically due to the increase in DC conduction. For high frequencies, the increase with temperature is much smaller.
Permittivity and dielectric modulus
Figure 6 shows for different compounds of HDPE/GTR, the values of real permittivity ( (a) Real and (b) imaginary dielectric permittivities of HDPE/GTR at 30°C in relation to frequency: ○, HDPE; □, HDPE + 5%GTR; ◊, HDPE + 10%GTR; Δ, HDPE + 20%GTR; ⊲, HDPE + 40%GTR; ▿, HDPE + 50%GTR; and ⊳, HDPE + 70%GTR.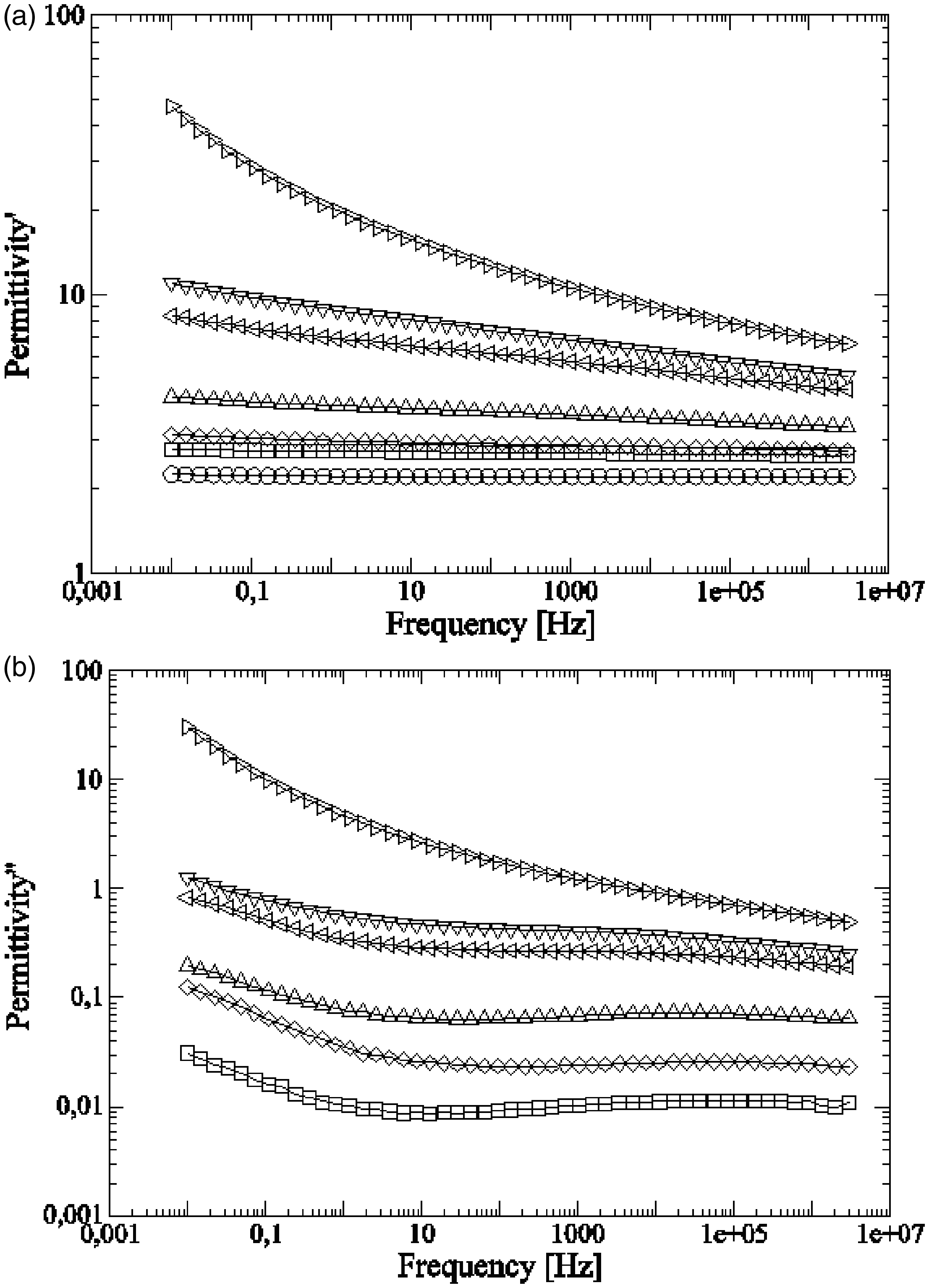
Both real permittivity and dielectric loss factor increase as GTR content rises. The real permittivity decreases as frequency increases, being this tendency less significant for low GTR concentrations. In the case of HDPE with no GTR,
The permittivity and dielectric loss for a frequency of 50 Hz and several temperatures between 30°C and 120°C are shown in Figure 7. GTR higher contents are directly related with higher (a) Real dielectric permittivity and (b) dielectric loss of HDPE/GTR, at 50 Hz in relation to temperature: ○, HDPE; □, HDPE + 5%GTR; ◊, HDPE + 10%GTR; Δ, HDPE + 20%GTR; ⊲, HDPE + 40%GTR; ▿, HDPE + 50%GTR; and ⊳, HDPE + 70%GTR.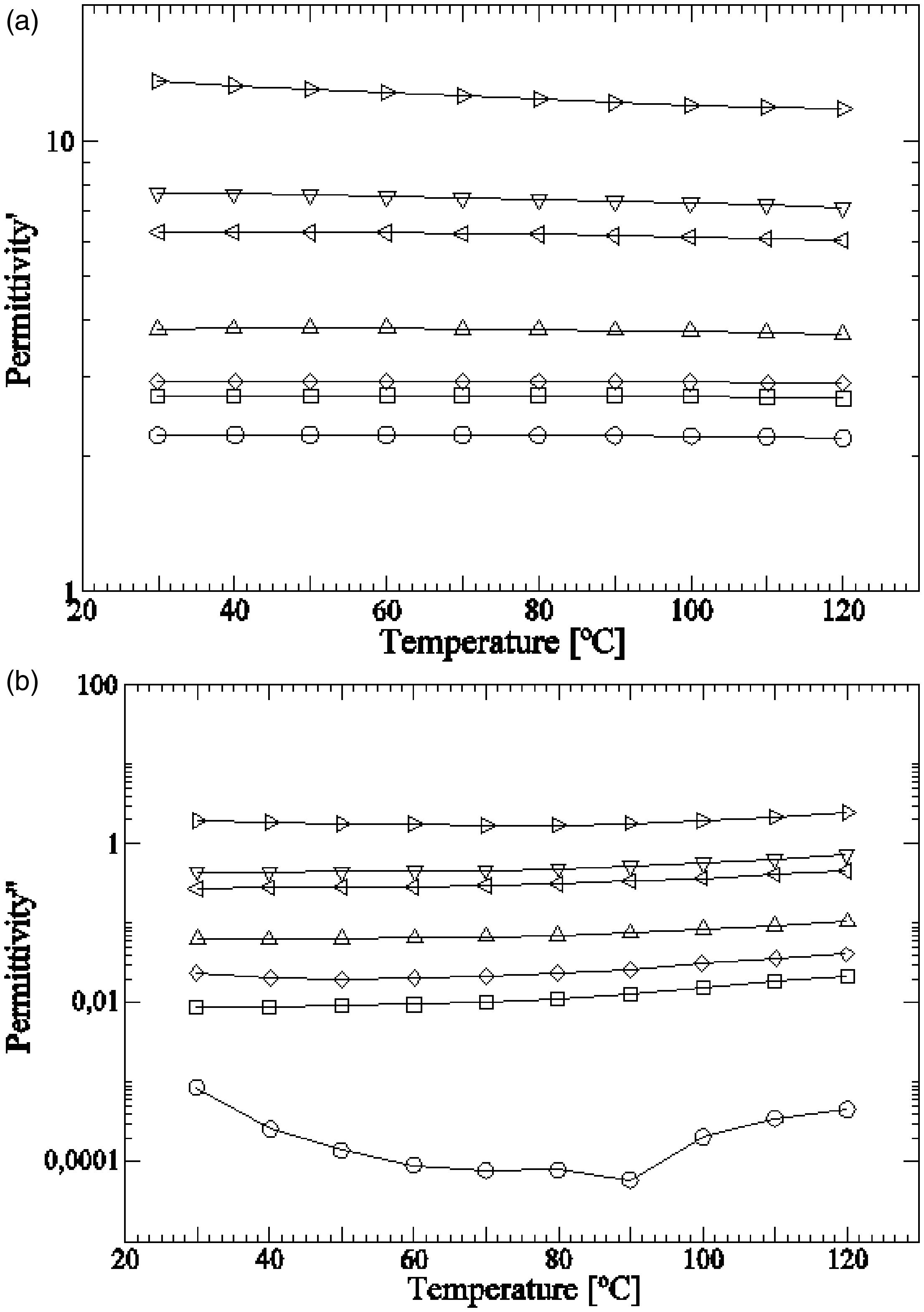
Generally, the permittivity seems to decrease slightly when temperature rises. Nevertheless, dielectric loss factor results show a differentiated behavior of HDPE from the HDPE/GTR compounds. The data obtained from the HDPE sample show that
Relaxational interfacial phenomena in heterogeneous materials usually are located at very low frequencies, and so they are not visible in low-temperature measurements for the frequency range studied. As they are thermally activated processes, they would appear in this frequency range for higher temperatures. However, at such frequencies and temperatures, there are several phenomena that typically darken the interfacial relaxations (electrode polarization and conduction phenomena). In order to overcome these problems, it can be convenient to use the formalism of the dielectric modulus:35,36
Three-dimensional diagrams of the imaginary component of the electric module (M″) with relation to temperature and frequency for HDPE: (a) HDPE, (b) HDPE/GTR-5%, (c) HDPE/GTR-20%, (d) HDPE/GTR-40%, (e) HDPE/GTR-50%, and (f) HDPE/GTR-70%.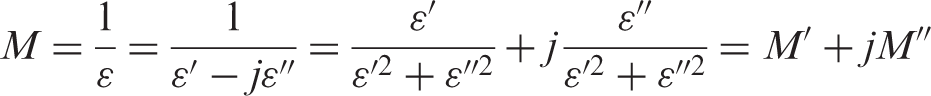
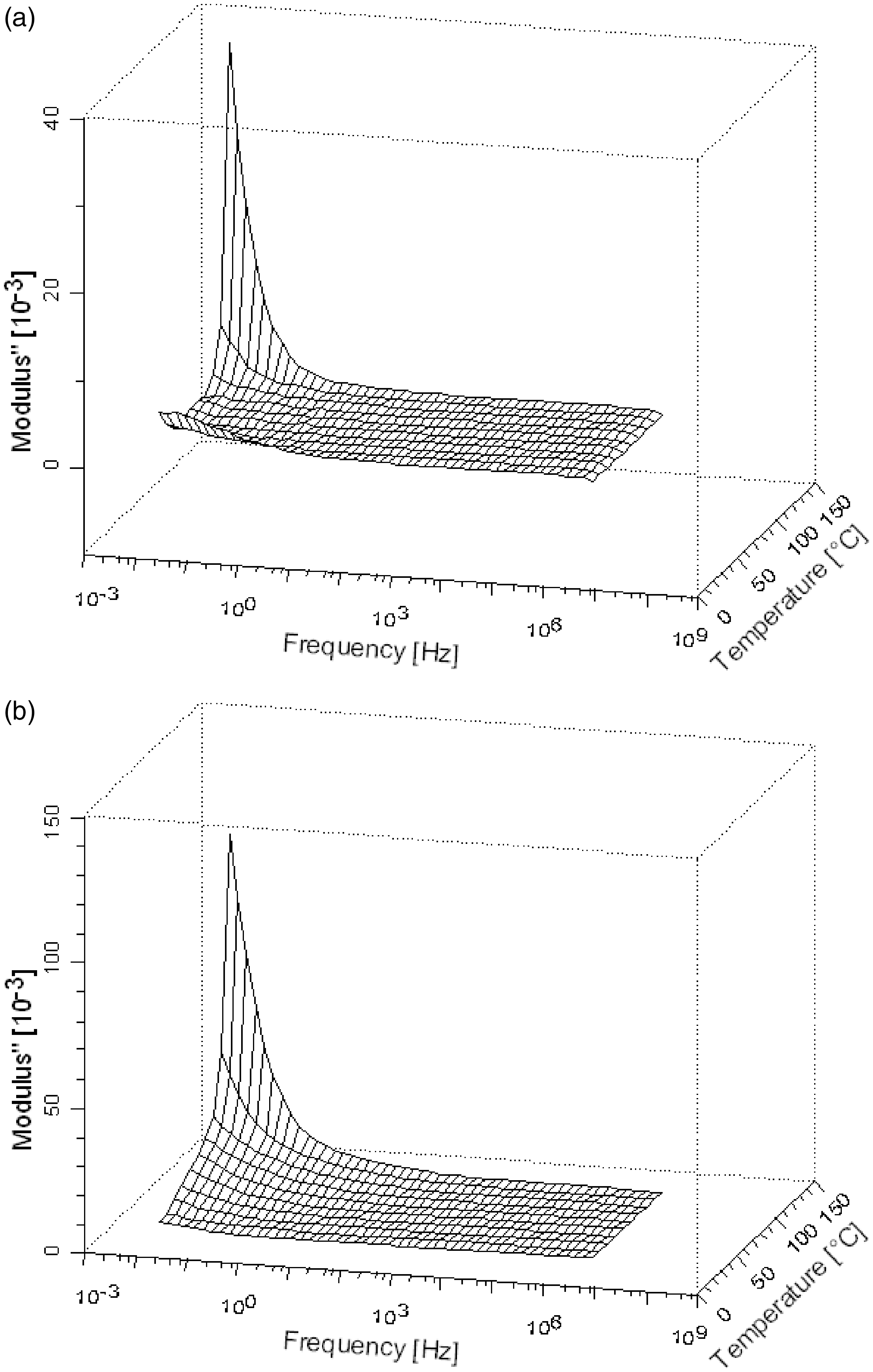
On the other hand, for low GTR concentrations, the α′ peak is placed at very low frequencies, an it does not appear in diagrams. However, it seems that for GTR concentrations of 5%, the relaxation is greater than of pure HDPE. For higher GTR concentrations, the α′ relaxation decreases, shifts to higher frequencies, and finally, it almost vanishes for 70%. Such evolution is analogous to that of crystallization, which, as it has been observed by DSC, is higher for 5% of GTR and drops if the concentration increases. The fact that α′ relaxation should be related to amorphous–crystalline interfaces is consistent with this apparent direct dependency.
In Figure 9, the maximum relaxation frequency, MWS in relation to the reciprocal of the temperature is represented. The points fit well to a straight line. This behavior obeys the Arrhenius expression for thermally activated relaxations,
Arrhenius plot of log f in relation to 1/T for the MWS relaxation: ○, HDPE + 20%GTR; □, HDPE + 40%GTR; ◊, HDPE + 50%GTR; and Δ, HDPE + 70%GTR.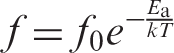
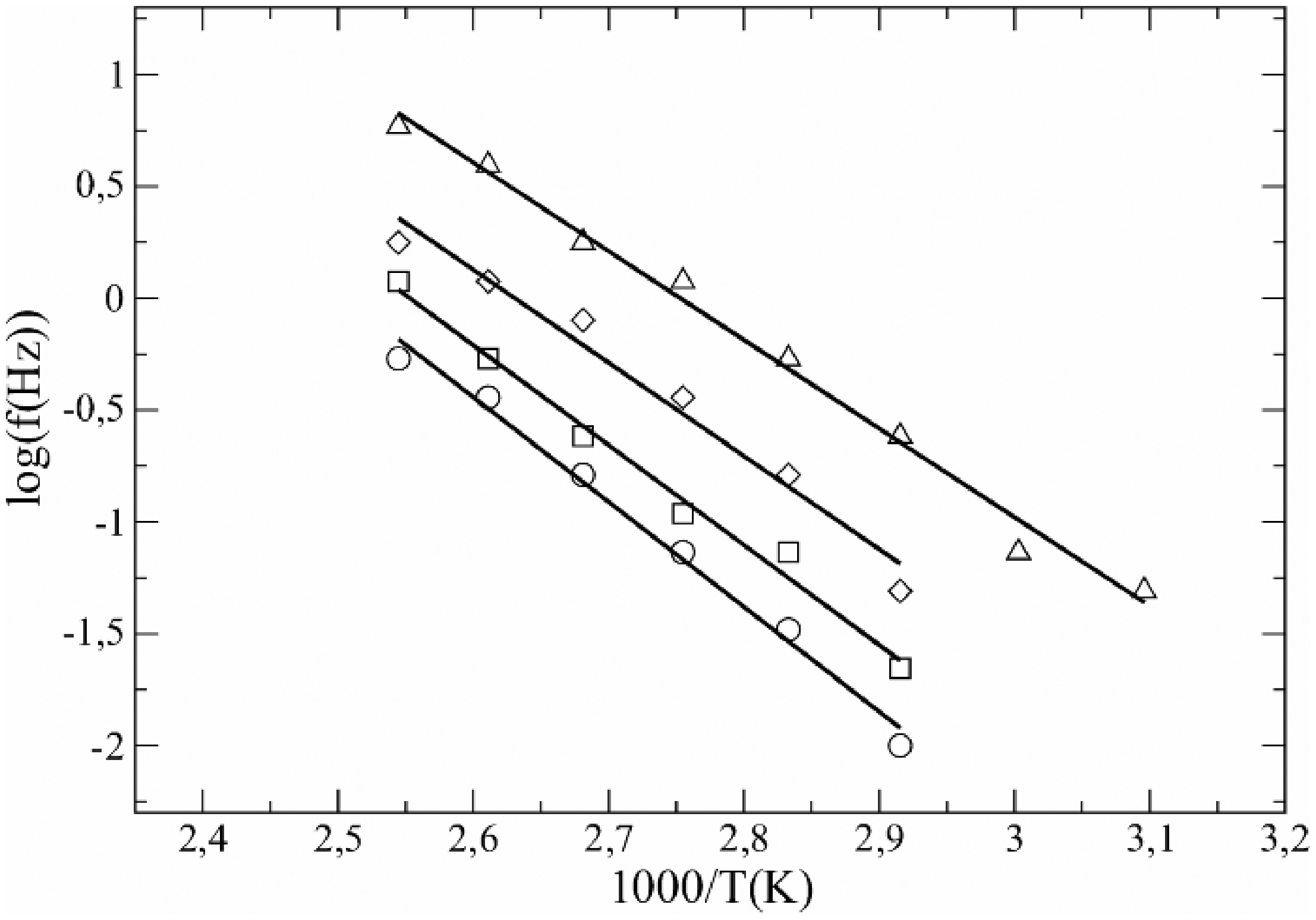
Activation energy and characteristic time of MWS relaxation for any GTR concentration.
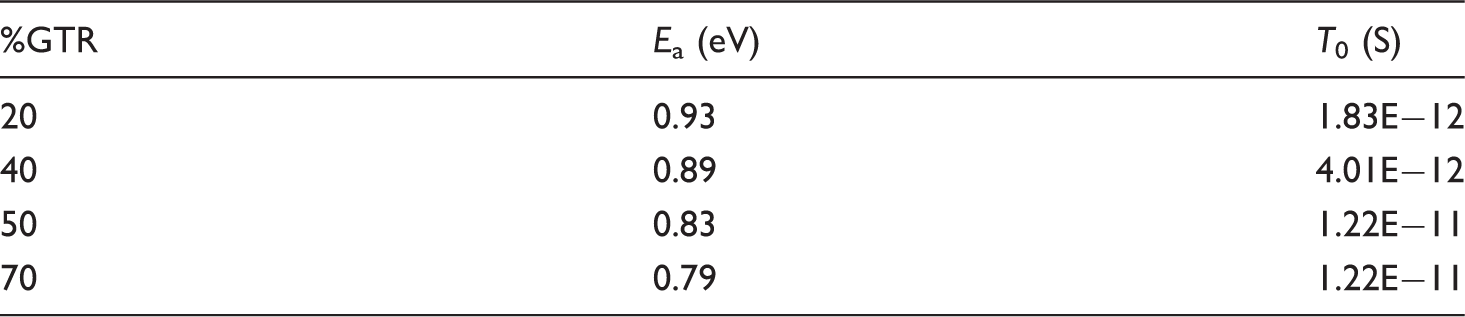
Only for 20% GTR concentration or higher, the MWS peak has been clearly located and, so, a linear fit performed. Table 1 shows that there is a light decrease in
Conclusions
The SEM microphotographs show relative differences in their results depending on particle size of concentration. With reference to particle size, when they are small they adhere better to the matrix due to its high specific ruggedness and the small size of its pores and cracks. On the other hand, large particles cause an increase in faults and cracks in the matrix, worsening interfacial adhesion. The GTR concentration also influences the microstructure of the compound worsening in all the cases its interfacial union, forming agglomerates that cause cracks and pores of considerable size on its surface.
The calorimetric study indicates that the differences in fusion enthalpy are small when the GTR concentration was varied which means that the matrix is only slightly altered by the presence of the filler in terms of its crystalline structure. Nevertheless, this fusion enthalpy tends to increase slightly for compounds with particle sizes inferior to 200 µm and low GTR concentrations (5–10%), due to the effect of the nucleation of the GTR particles in the interior of the HDPE. Generally, particles with sizes greater than 200 µm cause a decrease in fusion enthalpy in all cases, demonstrating greater fragility.
In principle, the stress–strain test should show that on adding the GTR reinforcement in the matrix, the mechanical properties of the compound increase, which would make its incorporation positive. This happens for concentrations of up to 10% and small-sized particles, the reinforcement–matrix union is correct, improving mechanical properties such as tenacity and tensile strength. Nevertheless, in excess of a 10% GTR concentration, and with all particle sizes, the mechanical properties diminish, this decrease being more significant when large-sized particles are in use.
With respect to dielectric analysis, it has been observed that real conductivity as well as permittivity and dielectric loss factor increase with the GTR concentration of the compounds. As it occurs in similar materials, the conductivity presents a sublinear dispersive behavior in all cases. On the other hand, permittivity decreases with frequency in composite samples but remains constant in pure HDPE. The loss factor shows an initial decrease due to conductive processes and a subsequent β relaxation in all samples. The analysis with the temperature shows that the real permittivity slightly decreases while the dielectric loss factor increases with temperature, except for pure HDPE, which presents its own dielectric complex spectrum. The dielectric modulus formalism has enabled us to study the MWS relaxation due to interfacial polarization. By fitting the Arrhenius model, the activation energy and the characteristic time of this thermally activated relaxation for any studied compound are obtained. It has found that the activation energy decreases with GTR concentration whereas the characteristic time increases. Finally, although the insulating features are reduced with GTR addition, the conditions for antistatic applications are improved.
The results reached through the analysis of these compounds, obtained exclusively from recycled polymers, indicate that by considering only the size of the GTR particle as a variable, and without any type of previous pretreatment with acids (which have proven to be ineffective and costly), the concentration limit in order that the mechanical and dielectric properties of the compound maintain acceptable values, is around 10–15% GTR concentration. Other methods should be tested such as previous devulcanization of the GTR, to verify if these percentages could rise to 20–25% GTR, which would make its use viable in diverse fields of industry.
Footnotes
Acknowledgments
The authors thank the Ministry of Science and Technology for the funding received in order to develop the project MAT 2007-64569, and the Department of Nuclear Physics, University Polytechnic of Catalonia, Terrassa Section, for their support in undertaking the dielectric tests and with special thanks to M. Mudarra and J. Belana.
