Abstract
A flexibly cross-linked shape memory polyurethane (SMPU) was converted to a magnetic polymer by making a composite with a ferromagnetic particle (Fe3O4). The flexibly cross-linked SMPU has already displayed excellent mechanical and shape memory properties; the design of a magnetically responsive material allowed for a unique and novel SMPU. Among the three different composite preparation methods (melt-mixing, solvent blending, and in situ reaction mixing), the melt-mixing method was the best and attained even distribution of magnetic particles and high mechanical and shape memory properties. Differential scanning calorimetric and infrared results showed that the polymer structure was not affected by melt-mixing. In tensile testing, the maximum stress of the composite reached 39 MPa, and the strain at break also increased to 2495%. Shape recovery exhibited maximums as high as 99% and displayed similar values after repetitive shape recovery test cycles. Finally, the magnetic property of SMPU was characterized by a superconducting quantum interference device magnetometer and demonstrated in the magnetic attraction test. The potential applications of the magnetic and flexible SMPU are discussed.
INTRODUCTION
Shape memory polyurethanes (SMPUs) have been extensively studied due to their attractive properties, such as practical and adjustable phase transition temperature ranges, high and reproducible shape recovery, easy and simple processing, and chemical resistance [1–9]. The characteristics of SMPU were explained by a phase-separated structure in which hard and soft domains are formed due to their differences in intermolecular attractions. A cross-linked structure was tested to improve the mechanical and shape memory properties of linear SMPU. An advanced flexible cross-linking method in which SMPU chains were connected by a flexible poly(ethylene glycol) (PEG) displayed high tensile stress and strain capabilities and reliable shape recovery under severe conditions [10–15]. Other than improving the mechanical properties of the polymer, special effects such as electric or magnetic responsiveness, low-temperature shape recovery, temperature or pH sensing, and electric conductivity are actively researched as a mean to develop smart polymer; in such materials, the active functional molecule was chemically coupled to polymer chains to improve the uniform distribution and secure attachment of the functional group. For example, we have reported low-temperature shape recovery polymers that can sustain shape recovery at temperatures as low as −30°C [16]. Electroactive shape memory polymer composites containing carbon nanotubes [17,18], electromagnetic filler [19,20], Ni chain [21,22], or hybrid fibers [23] have been already reported and they demonstrate rapid shape recovery by applying an electric current or electromagnetic field. Magnetic polymer systems are also one of the areas of high research interest. Such materials are attractive for magnetic drug targeting in which a magnetic nanoparticle transports a drug to the area of interest by local application of a magnetic field [24,25]; they are also being investigated for magnetic separations [26] and magnetic resonance imaging [27]. A magnetic and thermoresponsive shape memory polystyrene that responds to the heat generated from magnetic induction has also been reported [28]. The combination of a flex-linked SMPU with magnetite (Fe3O4) particles to form a magnetic SMPU composite is synergistic; the proven mechanical strength and shape recovery of the flex-linked SMPU and the well-known magnetic response of Fe3O4 particles will demonstrate a different type of functional SMPU. In this investigation, a flex-linked SMPU composite that has uniform distribution of magnetic particles and can respond to a magnetic stimulus was studied.
EXPERIMENTAL
Materials
Poly(tetramethylene glycol) (PTMG, Aldrich, Mw = 2000 g/ mole), 4,4′-methylenebis (phenylisocyanate) (MDI, Tokyo Kasei), 1,4-butanediol (BD, Junsei Chemical), PEG (PEG-200, Duksan Chemical, Mw = 200 g/mole), and Fe3O4 (Aldrich) were dried under a high vacuum (0.1 torr) for 12 h before use. Dimethylformamide (DMF, Duksan Chemical, Korea) was distilled over CaH2 before use.
Polymer Synthesis
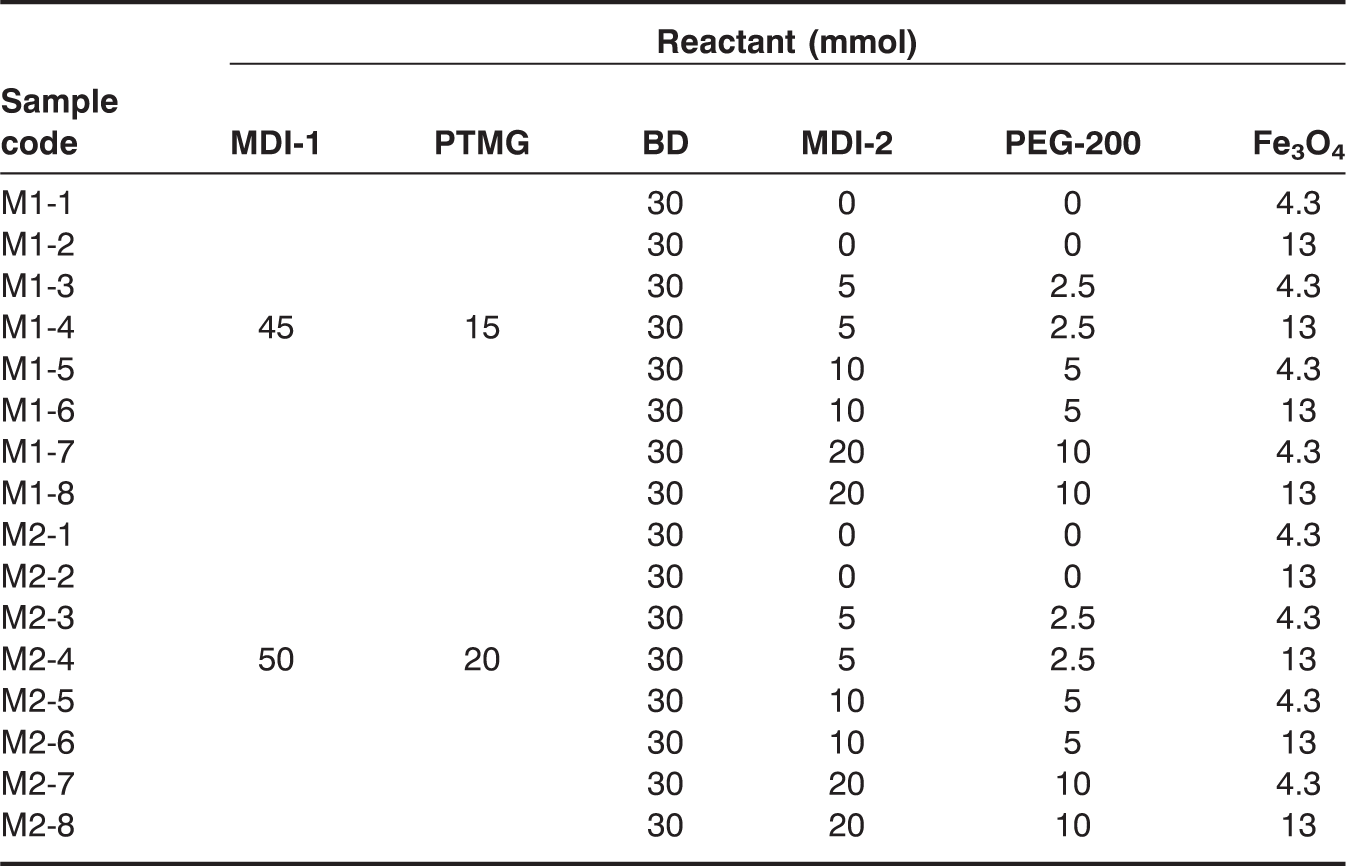
A flex-linked SMPU was prepared by adopting a method described in literature, where the soft segment content was 68 wt% for M1 series and 72 wt% for M2 series, and hard segment content was 32 wt% for M1 series and 28 wt% for M2 series [29]. A mixture of MDI and PTMG in a ratio that is specified in Table 1 was placed in a 500-mL four-necked beaker-type flask equipped with a mechanical stirrer, condenser, nitrogen purge, and oil bath. The mixture was allowed to react for 3 h at 50°C to prepare the prepolymer. BD, functioning as a chain extender, was dissolved in 100 mL of DMF and added to the prepolymer; the reaction continued for 1 h. Subsequently, a second quantity of MDI was added to the reaction mixture mentioned above, and the reaction continued for 1 h. PEG-200 was added to the mixture as a cross-linker, and the mixture was stirred for 2 h under the same conditions. After complete polymerization, the PU product was dried in a 60°C oven for 3 days to remove residual DMF. Schematics of the resulting SMPU structures are shown in Figure 1. The flex-linked SMPU and Fe3O4 composites were prepared by a melt-mixing method with a co-rotating micro twin-screw extruder (Bau Tech BA-10, screw diameter = 19 mm, L/D = 40) at 160°C to 180°C, with a screw torque of 10–11 Nm and a screw speed of 200–250 rpm. The pelletized SMPU/Fe3O4 composite was transformed into 0.4 ± 0.01 mm thick film sheets by melt pressing (Carver 4122) at 185°C and quickly cooling. Specimens for mechanical and shape memory tests were made from the film sheets.
Structures of (a) linear SMPU and (b) SMPU with flexible cross-linking. Composition of the magnetic SMPU.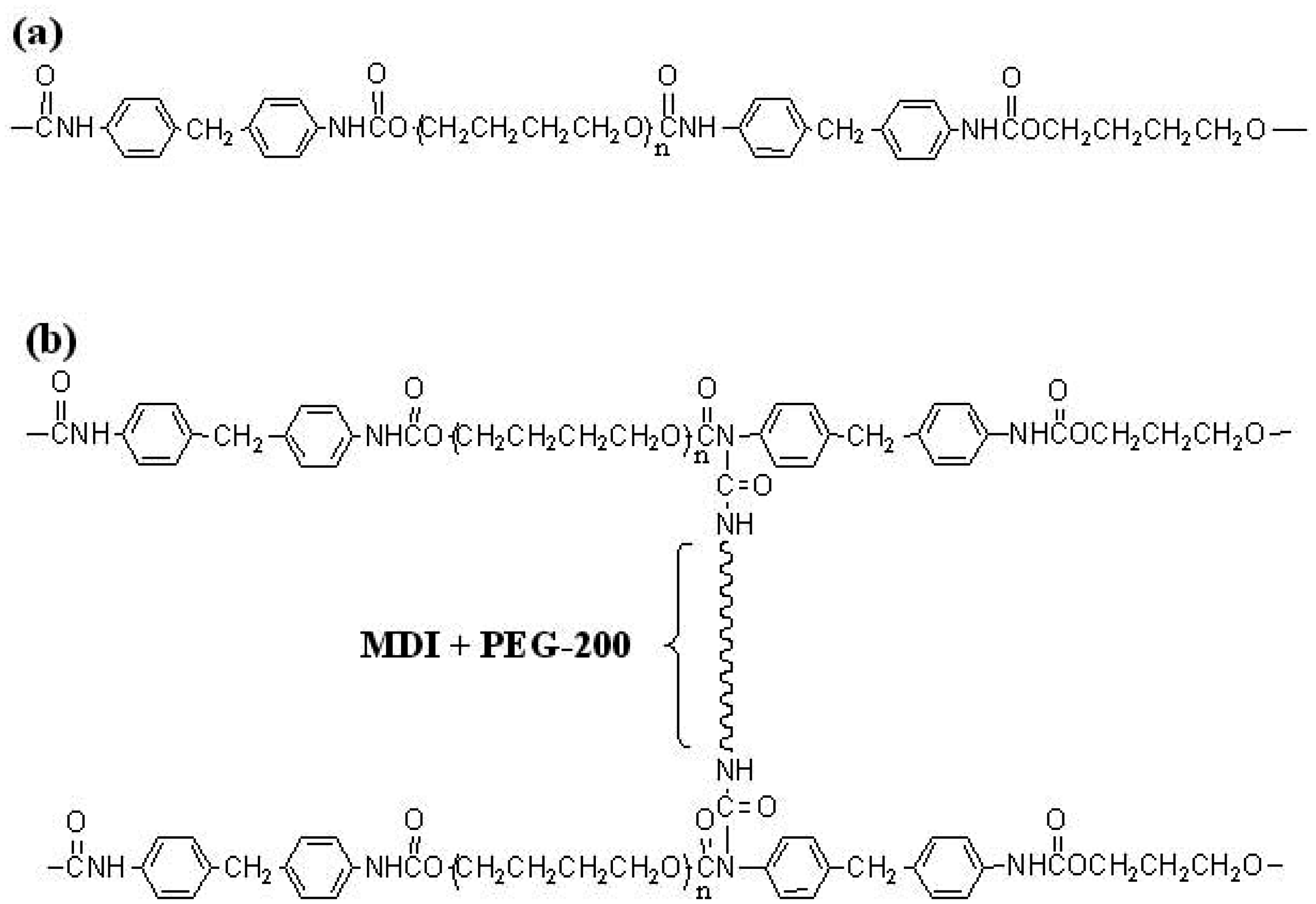
Characterization
A Fourier-transform infrared (IR) spectrophotometer (JASCO 300 E) equipped with attenuated total reflectance capability was used to obtain IR spectra of the SMPU samples. For each sample, 25 scans were taken at resolution of 4 cm−1 and a scan speed of 2 mm/ s. A differential scanning calorimeter (DSC 8000, Perkin Elmer) was used to take calorimetry data for both heating and cooling scans at a rate of 10°C/ min between −50°C and 250°C under N2 flow. After melting at 250°C for 5 min and cooling quickly to −50°C, a 20-mg specimen was warmed to 250°C at 10°C/ min while monitoring the phase transitions. Tensile properties were measured with a Universal Testing Machine (UTM, LR50K, Lloyd Instrument, UK) according to the ASTM D638 standards of a 25-mm gage length, a 10-mm/ min crosshead speed, and a 2.5-kN load cell. The fractured surfaces of composites were imaged with a scanning electron microscope (SEM, JEOL JMS-6360 A).
Shape Memory Test
A UTM equipped with a temperature-controlled chamber was used for cyclic shape memory tests. A specimen with a length L0 was drawn 100% to 2L0 in a temperature-controlled chamber set at 45°C over the course of 2 min and kept at 45°C for 5 min. Afterwards, the specimen was cooled to −25°C in the chamber with liquid nitrogen for 10 min, and the upper grip was released. The shrunken length (L1) was measured after the sample had remained at −25°C for 20 min. Percent shape retention (%) was calculated using Equation (1). The specimen in the chamber was then heated to 45°C for 5 min, and the length (L2) was measured after 10 min at 45°C. Percent shape recovery (%) was calculated using Equation (2). The shape memory test was repeated three times for each specimen.
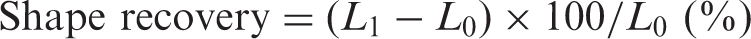
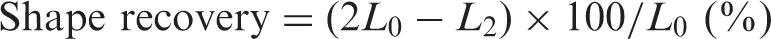
Magnetic Activity Test
A superconducting quantum interference device (SQUID) magnetometer (Quantum Design MPMS5), housed in the Seoul branch of the Korea Basic Science Institute, was used to measure the magnetic field (B-field) of SMPU specimens under the condition of an external magnetizing field strength (H-field) from −2000 to 2000 Oe at room temperature (298 K). Magnetic levitation experiments were carried out with a specimen (5 × 100 mm2) that was fixed at one end and was free to move vertically at the other end. The maximum height of the free end that was raised by a permanent magnet hanging at the top was compared.
RESULTS AND DISCUSSION
Synthesis of Magnetic SMPU
An alternating arrangement of hard (MDI) and soft (PTMG) segments in the SMPU structure was critical for a high shape recovery. The advanced flex-linking of SMPU was different from the conventional cross-linking in that PEG, the flexible cross-linker, was laterally linked between PU chains to increase tensile mechanical stress without decreasing the strain and to attain a reproducible shape recovery [14,15]. The grafting of functional groups to polymer chains is already well studied for electric conductivity, electro-active artificial muscles, antistatic coatings, low-temperature shape recovery, and ion sensing. The lateral cross-linking in the flex-linked SMPU is similar in that grafting of PEG to SMPU chains was made between SMPU chains. The flex-linked SMPU demonstrated superior mechanical and shape memory properties when compared to the linear SMPU and was selected as a polymer matrix for the composite. Magnetic particles selected in this study were Fe3O4, which have been frequently adopted in magnetic polymer research due to their nano-scale particle size, compatibility with SMPU, and ferromagnetic characteristic. Instead of grafting magnetic particles to SMPU chains, magnetic particles were melt-mixed with SMPU in a twin-screw compounder due to the absence of available sites for covalent bonding on the Fe3O4 particle. Other mixing methods were unsuccessfully attempted. Solvent mixing, in which magnetic particles were stirred with an SMPU solution in DMF, had the problem of magnetic particles aggregating around the stirrer. The addition of magnetic particles during SMPU synthesis also had a similar problem, where the magnetic particles attached to stirrer driving shaft. The success of melt-mixing in a compounder originated from the facts that magnetic particles were not attracted to the compounder screw and twin-screw compounding was better in distributing magnetic particles compared to single-screw compounding. Even distribution of magnetic particles in the SMPU matrix could be seen in SEM pictures of the fractured surfaces (Figure 2). The structural differences between linear and flex-linked SMPU can be observed in Figure 1. In Table 1, the amount of Fe3O4 was fixed at either 4.3 or 13 mmol in both the M1 and M2 series while gradually increasing PEG-200 content, and the soft segment content in the M2 series (72 wt%) was larger than that of the M1 series (68 wt%). Therefore, the combination of magnetic responsiveness, very high strength, and high shape recovery was intended to produce a versatile flex-linked SMPU in this investigation.
SEM photographs of (a) SMPU (M1 series control), (b) M1-6, (c) SMPU (M2 series control), and (d) M2-6. Note: The scattered white spots in images (b) and (d) are the Fe3O4 particles.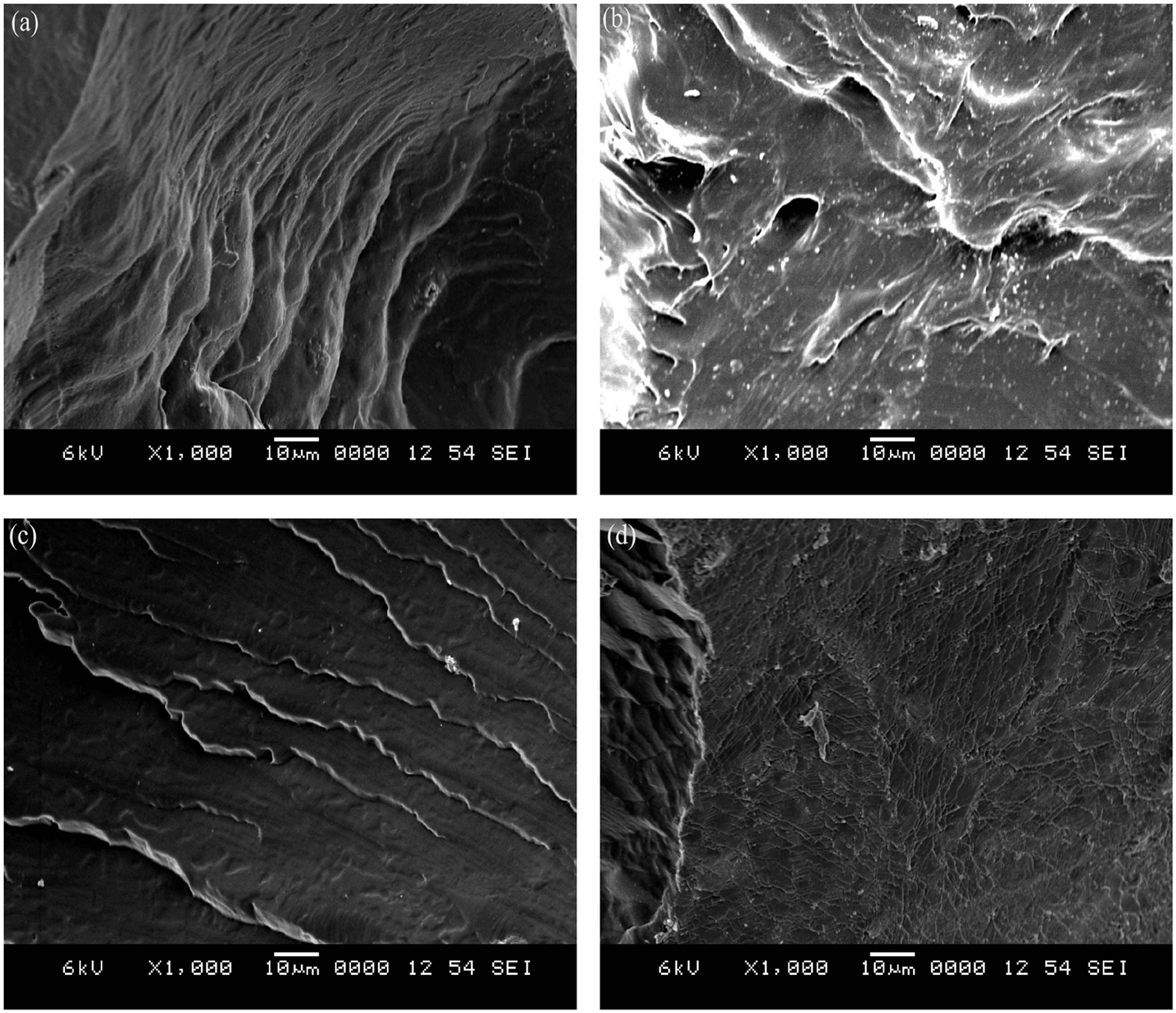
IR and DSC Analysis
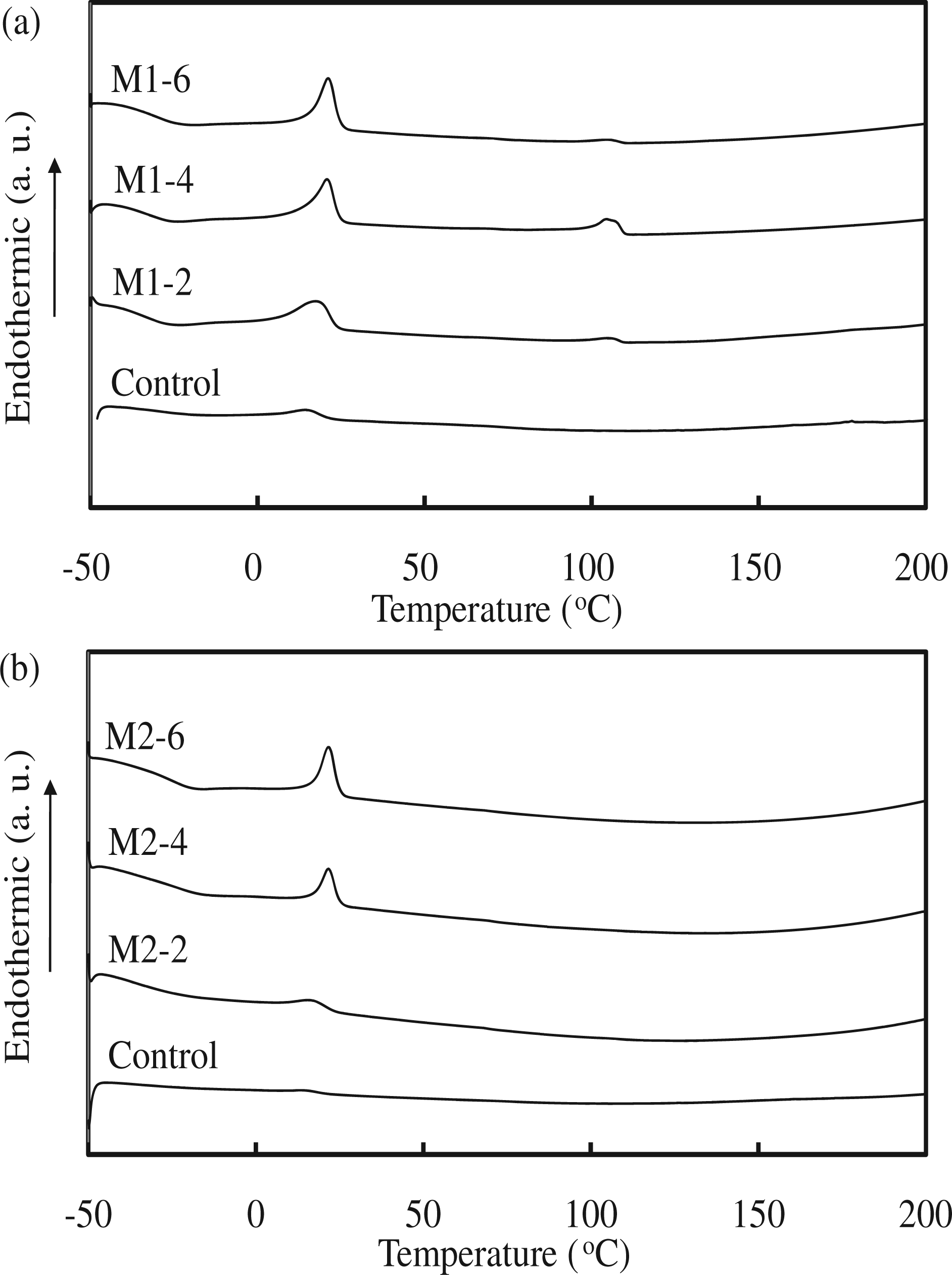
The stretching vibration mode of the hydrogen-bonded urethane carbonyl group appeared in the IR spectrum at 1699–1706 cm−1, which is slightly less than that of the free urethane carbonyl group (1731–1733 cm−1) [30]. The stretching vibration mode of the carbonyl group is also affected by dipole–dipole interactions between SMPU chains. The relative peak areas around both 1700 and 1730 cm−1 did not change with increasing Fe3O4 content for either the M1 series (Figure 3(a)) or the M2 series (Figure 3(b)). Based on the IR results for both the M1 and M2 series, no shift were observed in the bonded carbonyl and free carbonyl as the Fe3O4 or PTMG content increased. Addition of Fe3O4 into the SMPU did not seem to affect the molecular interactions such as hydrogen bonding and dipole–dipole interactions, and hard segment interactions were not disrupted. The glass transition temperature (Tg) of the soft segments of the SMPU were low (under −50°C) and difficult to detect by DSC; instead, the melting temperature (Tm) of this soft segment of the SMPU was obtained. The Tm of the soft segments of both the M1 and M2 series was observed around 20°C. The peak area, corresponding to the enthalpy of the transition, increased with Fe3O4 content; this behavior was presumably due to the interactions between Fe3O4 particles and the SMPU chains, which restricted movement of polymer chains (Figure 4). The thermal analysis results showed that soft segment melting was affected a little by the presence of Fe3O4 particles. Therefore, the soft segment melting temperature was used as the reference point for shape recovery tests.
IR spectra of (a) M1 and (b) M2 series. DSC thermograms of (a) M1 and (b) M2 series.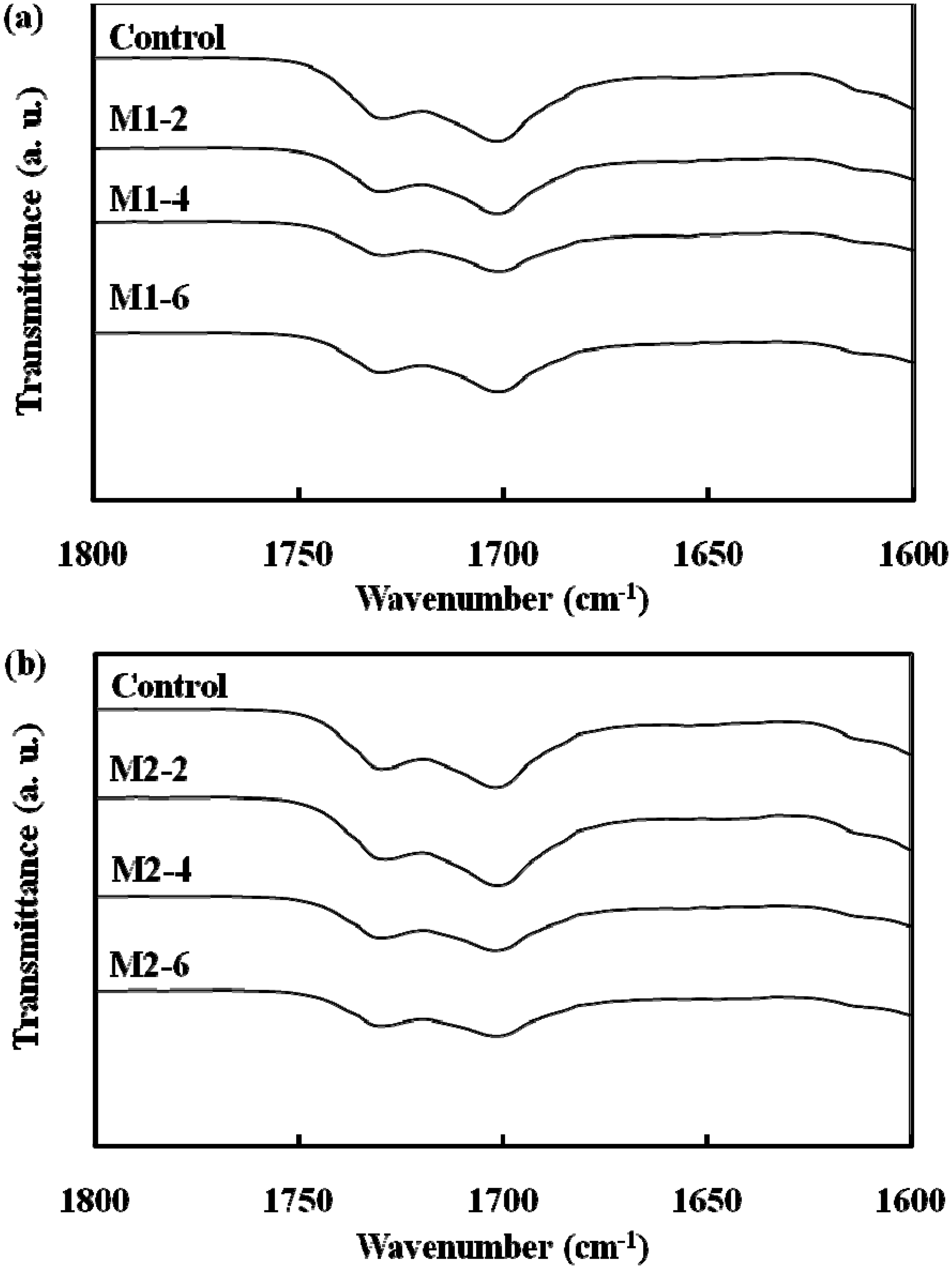
Tensile Properties
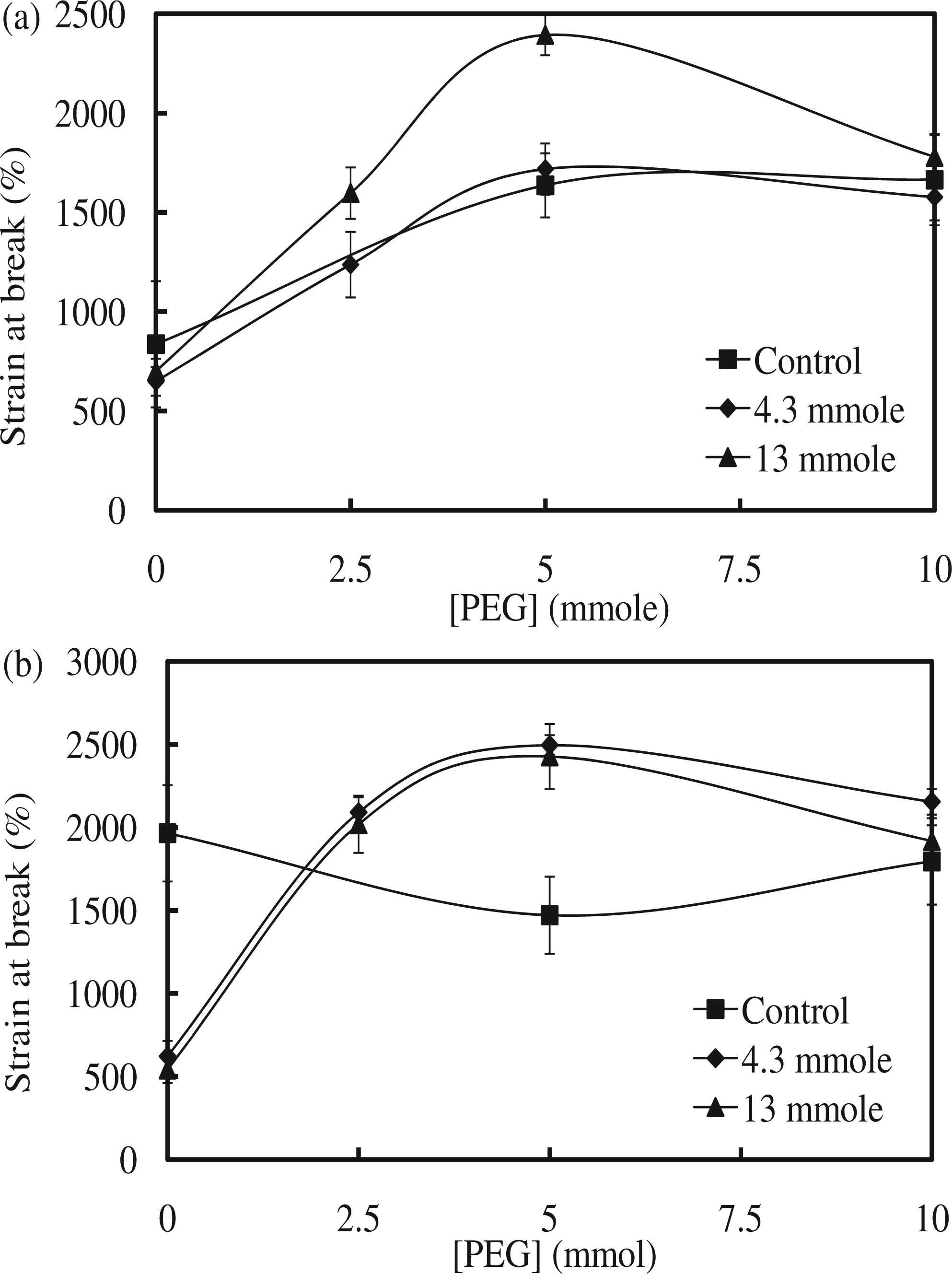
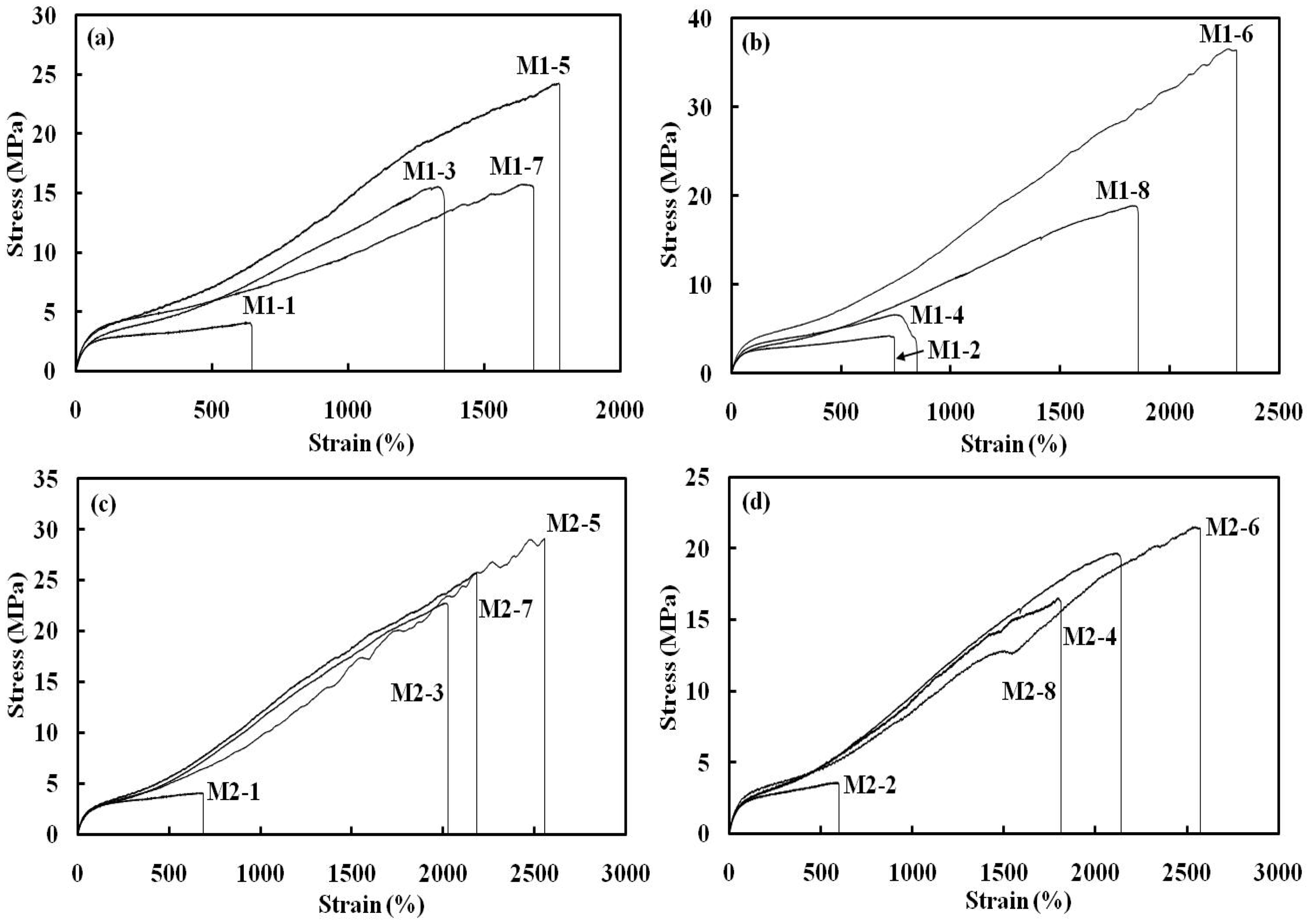
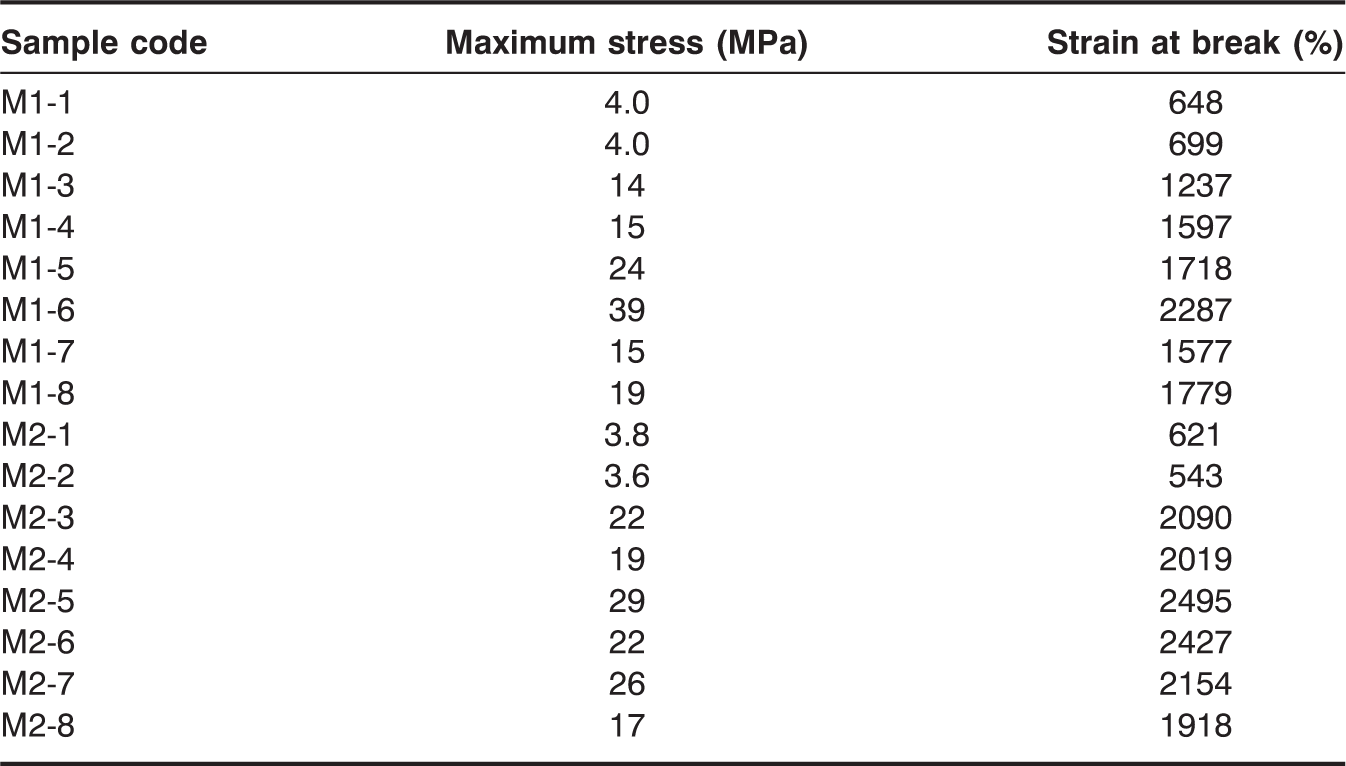
The maximum stress in the M1 series with 4.3 mmol of Fe3O4 increased from 4 MPa for M1-1, to 14 MPa for M1-3, and to 24 MPa for M1-5, before decreasing to 15 MPa for M1-7. The maximum stress in the M1 series with 13 mmol of Fe3O4 also increased from 4 MPa for M1-2, to 15 MPa for M1-4, and to 39 MPa for M1-6; this value slightly decreased to 19 MPa for M1-8. In the M1 series, the highest maximum stress increase was observed at M1-6 (345%) compared to the one without Fe3O4. However, the maximum stress decreased with the addition of magnetic particles when 10 mmol of PEG-200 was used as the cross-linker, compared to the control without Fe3O4. The addition of Fe3O4 did not affect the maximum stress in the case of the linear SMPU, in which PEG-200 was not used. Therefore, proper addition of Fe3O4 increased the maximum stress until a certain point, when further addition of Fe3O4 decreased this value. The maximum stress in the M2 series with 4.3 mmol of Fe3O4 showed an increase, going from 3.8 MPa for M2-1, to 22 MPa for M2-3, to 28 MPa for M2-5, and to 26 MPa for M2-7. The maximum stress in the M2 series with 13 mmol of Fe3O4 also showed an increase, going from 3.6 MPa for M2-2, to 19 MPa for M2-4, to 22 MPa for M2-6, and to 17 MPa for M2-8 (Figure 5). In the M2 series, the highest maximum stress increase was observed at M2-5 (170%) compared to the one without Fe3O4. In the M2 series, a similar trend to that of the M1 series was observed: the maximum stress increased with the addition of Fe3O4 at 5 mmol of PEG-200, but decreased at 10 mmol of PEG-200, compared to the control. Therefore, both M1 and M2 series showed a similar behavior if Fe3O4 particles were included. The effect of the addition of Fe3O4 on the improvement of the maximum stress of both M1 and M2 series was obvious at 5 mmol of PEG-200, but inconsequential at 10 mmol of PEG-200. At 5 mmol of PEG-200, SMPU was less cross-linked, and Fe3O4 particles could have interacted with the polymer chains, resulting in higher maximum stress. When the SMPU was highly cross-linked and strengthened with a high PEG content like the one with 10 mmol of PEG-200, Fe3O4 particles may have hindered the molecular interaction between the polymer chains, reducing the maximum stress. The maximum stress generally decreased if the content of inorganic particles was increased in the composites. The gradual increase of the maximum stress of control samples, one without Fe3O4, in both the M1 and M2 series as the PEG content was increased was simply due to more flexible cross-linking by PEG. The strain at break in the M1 series with 4.3 mmol of Fe3O4 increased from 648% for M1-1, to 1237% for M1-3 and to 1718% for M1-5, before decreasing to 1577% for M1-7. Strain at break in the M1 series with 13 mmol of Fe3O4 also increased from 700% for M1-2, to 1597% for M1-4, and to 2287% for M1-6; this value decreased to 1780% for M1-8. Unexpectedly, strain did not decrease as the Fe3O4 content increased. The strain of polymer composites generally decreased when the stress was improved due to increased rigidity. However, the flexible cross-linking made improvements of both stress and strain possible, and such characteristics were not hindered by the addition of Fe3O4. The strain at break in the M2 series with 4.3 mmol of Fe3O4 also showed a similar trend as the PEG content increased: 621% in M2-1, 2090% in M2-3, 2495% in M2-5, and 2154% in M2-7 (Figure 6). The strain at break in the M2 series with 13 mmol of Fe3O4 increased from 543% in M2-2, to 2019% in M2-4, and to 2427% in M2-6; this value decreased to 1918% in M2-8. The strain at break of the SMPU of both M1 and M2 series was found to increase with the addition of Fe3O4; the synergistic effect between the SMPU and Fe3O4 particles originated from the flexible cross-linking. Results of tensile properties are summarized in Table 2. The combined improvement in stress and strain after the addition of Fe3O4 can also be observed in the stress–strain curves (Figure 7(a) and (b) for the M1 series; Figure 7(c) and (d) for M2 series), in which values for both maximum stress and strain at break were increased relative to samples without Fe3O4. In general, the simultaneous improvement of both the maximum stress and strain at break after the addition of Fe3O4 was very intriguing and helpful in designing a strengthened polymer composite.
Maximum stress of (a) M1 and (b) M2 series. Strain at break of (a) M1 and (b) M2 series. Stress–strain curves of (a) M1 series with 4.3 mmol of Fe3O4, (b) M1 series with 13 mmol of Fe3O4, (c) M2 series with 4.3 mmol of Fe3O4, and (d) M2 series with 13 mmol of Fe3O4. Tensile mechanical properties.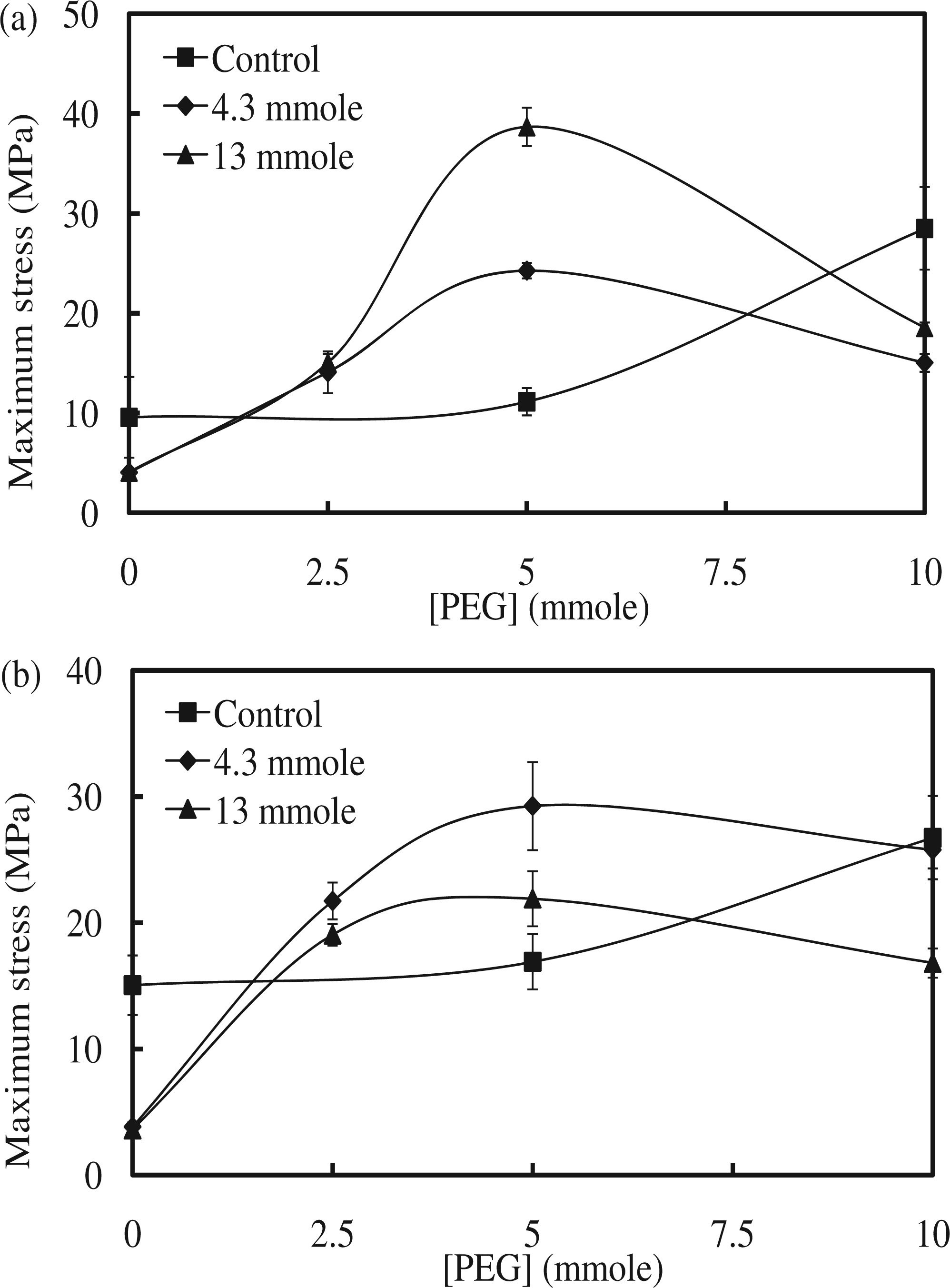
Shape Memory Effect
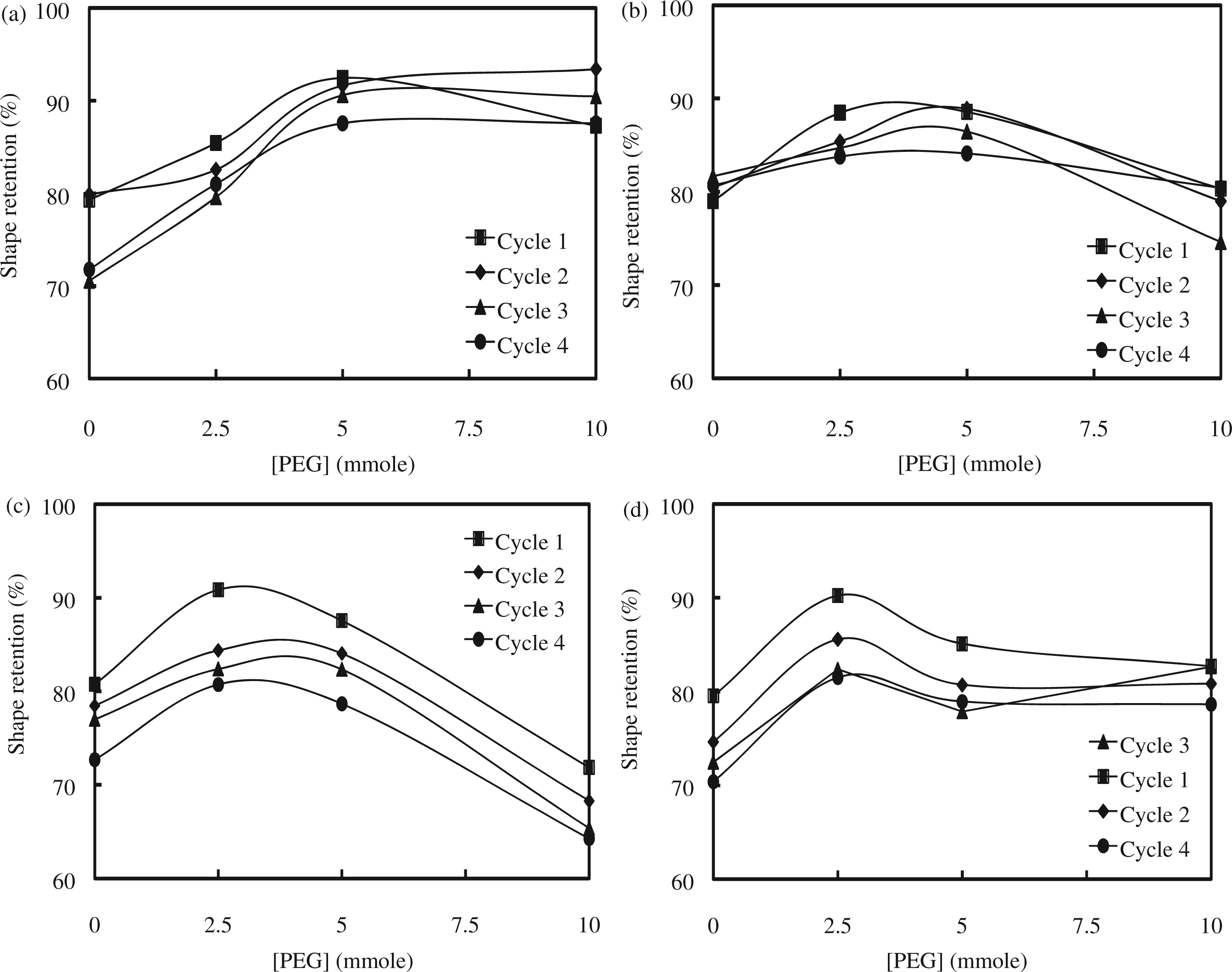
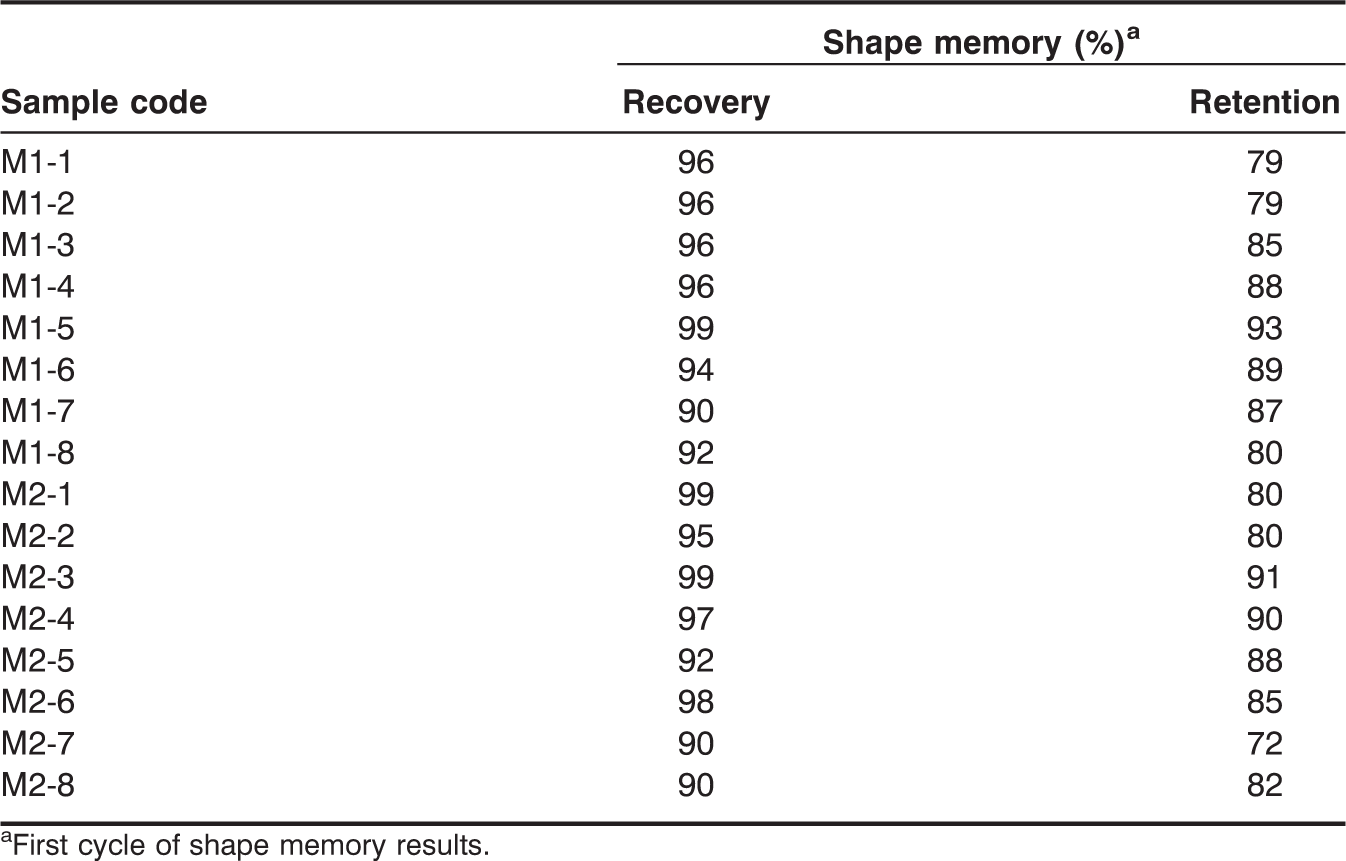
Shape recovery and retention of the SMPU were investigated by a cyclic shape memory test at Tm ± 20°C (Figures 8 and 9). The Tm of the soft segment was selected for shape memory tests instead of the Tg which is too low for temperature control using our apparatus and is too low for practical applications [11,12]. Shape recovery of the M1 series with 4.3 mmol of Fe3O4 was 96%, 96%, 99%, and 90% for M1-1, M1-3, M1-5, and M1-7, respectively, in the first test cycle and remained above 90% after four cyclic tests. Shape recovery of the M1 series with 13 mmol of Fe3O4 was 96% for M1-2, 96% for M1-4, 93% for M1-6, and 92% for M1-8; these values gradually decreased after repeated test cycles. Shape recovery of the M2 series with 4.3 mmol of Fe3O4 was 99% for M2-1, 99% for M2-3, 92% for M2-5, and 90% for M2-7 in the first test cycle; these values remained around 90% after four cyclic tests. Shape recovery of the M2 series with 13 mmol of Fe3O4 was 95%, 97%, 97%, and 91% for M2-2, M2-4, M2-6, and M2-8, respectively, and decreased as the cyclic tests continued. Shape recovery of both the M1 and M2 series was above 90% and slightly decreased as PEG content increased. Shape recovery of the SMPU was reproducible, as judged from the four cyclic tests. Finally, the addition of Fe3O4 did not significantly alter the results of shape recovery. The shape retention experiments were conducted at a very low temperature (−20°C) and tested the tendency of an SMPU to retain its shape against a shape recovering force. Shape retention, in general, increased for all of the SMPUs with increasing PEG content up to 10 mmol of PEG. The addition of Fe3O4 did not affect the shape retention. The M2 series was expected to have a better shape retention than M1 series because the M2 series contained a greater amount of soft segments (based on comparing the PTMG content of each series). However, the difference in shape retention between the M1 and M2 series was minimal. Cyclic shape retention tests also demonstrated that the shape retention of both the M1 and M2 series remained mostly constant after four cyclic tests; for example, the initial shape retention values of M1-6 (88%) and M2-6 (85%) were slightly reduced to 84% and 79%, respectively, after four cycles. Shape memory test results are summarized in Table 3. Considering shape recovery, shape retention, and tensile mechanical test results, both the M1 and M2 series performed excellently when compared to the previous SMPUs; M1-5 and M2-3, in particular, exhibited outstanding shape recovery for an SMPU. The addition of Fe3O4 to the SMPU did not interfere with the molecular interactions necessary for their excellent performance in shape memory and mechanical properties.
Cyclic shape recovery tests of (a) M1 series with 4.3 mmol of Fe3O4, (b) M1 series with 13 mmol of Fe3O4, (c) M2 series with 4.3 mmol of Fe3O4, and (d) M2 series with 13 mmol of Fe3O4. Cyclic shape retention tests of (a) M1 series with 4.3 mmol of Fe3O4, (b) M1 series with 13 mmol of Fe3O4, (c) M2 series with 4.3 mmol of Fe3O4, and (d) M2 series with 13 mmol of Fe3O4. Shape memory properties. First cycle of shape memory results.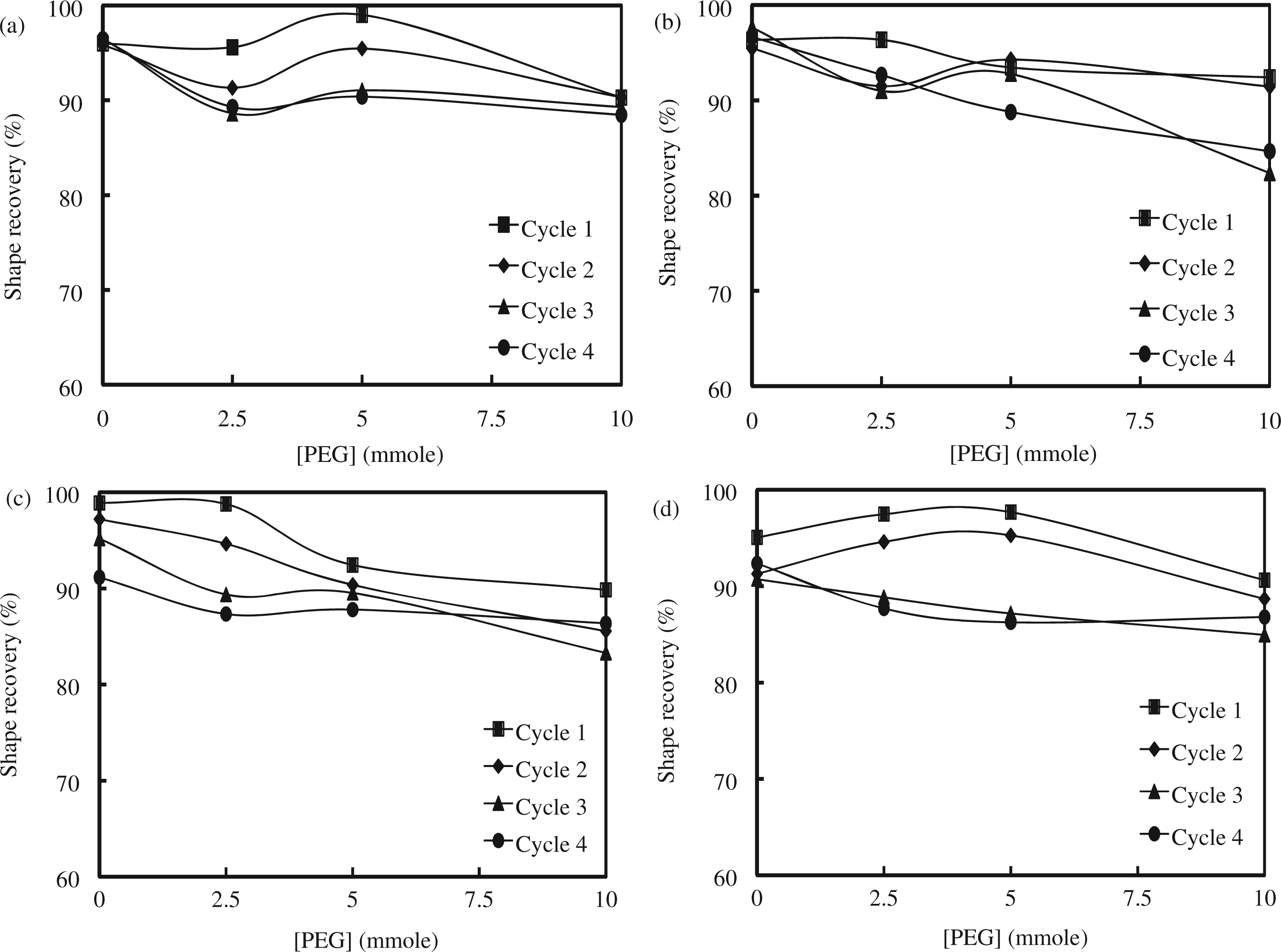
Magnetic Property
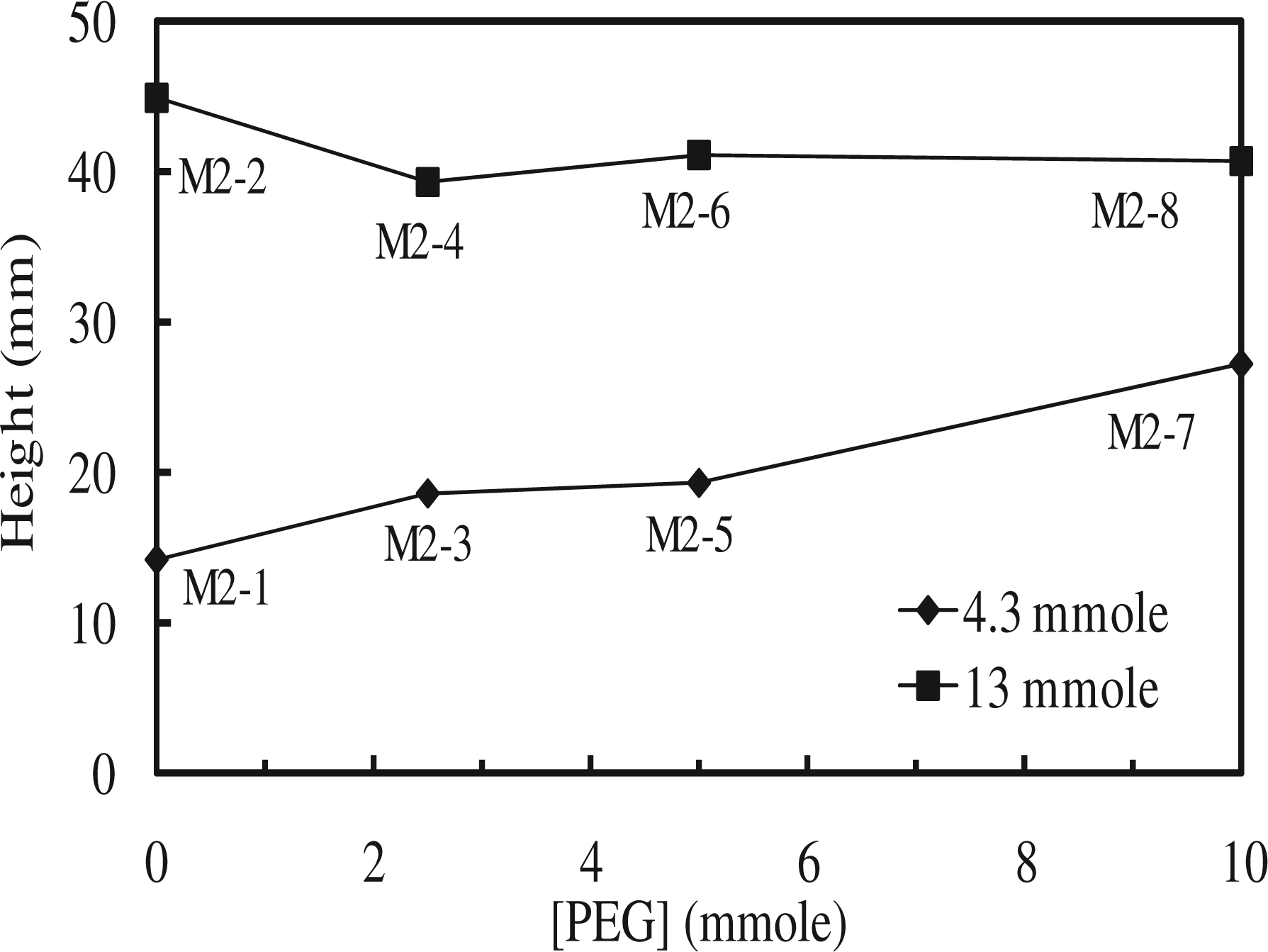
The magnetization of SMPU specimens was tested by a SQUID magnetometer that measured the magnetized field (H-field) of a material by an external magnetic field (B-field). As seen in Figure 10, Fe3O4 alone did show a significant response to the external magnetic field, and the control, the SMPU without Fe3O4, did not respond to the external magnetic field. The M1 and M2 series showed an intermediate magnetic response depending on the amount of Fe3O4 included in the specimen. The M2 series showed a slightly larger magnetic response than the M1 series at the same Fe3O4 content because the M2 series had a higher content of soft segments and better flexibility. Therefore, magnetization of the SMPU depended on the Fe3O4 content and the direction of the magnetic field. In line with the magnetization test, the maximum height that the SMPU could be raised by a permanent magnet was compared for M2 series, as shown in Figure 11 and plotted in Figure 12. For simplicity, only the M2 series was selected for comparison because it had a higher soft segment content and better flexibility for the levitation test. The M2 series with 13 mmol of Fe3O4 (M2-2, M2-4 M2-6, and M2-8) had levitation values around 40 cm, while the M2 series with 4.3 mmol of Fe3O4 (M2-1, M2-3, M2-5, and M2-7) exhibited levitation of approximately 15 cm. The levitation height in the M2 series was affected more by the Fe3O4 content, as opposed to the PEG content. The SMPU with flexible cross-linking did not lose its pliability after cross-linking and was able to attain similar heights compared to the linear one. The magnetic response of an SMPU with flexible cross-linking was demonstrated in this investigation and will be useful in fields such as fibers, foams, elastomers, and insulating materials with magnetic property.
Magnetization of PUs measured by SQUID magnetometer. Magnetic attraction of M2 series: (a) control, (b) M2-1, (c) M2-2, (d) M2-3, (e) M2-4, (f) M2-5, (g) M2-6, (h) M2-7, and (i) M2-8. Note: The inset number designates the raised height (mm). Comparison of the maximum magnetic attraction height of SMPUs with 4.3 mmol of Fe3O4 and 13 mmol of Fe3O4.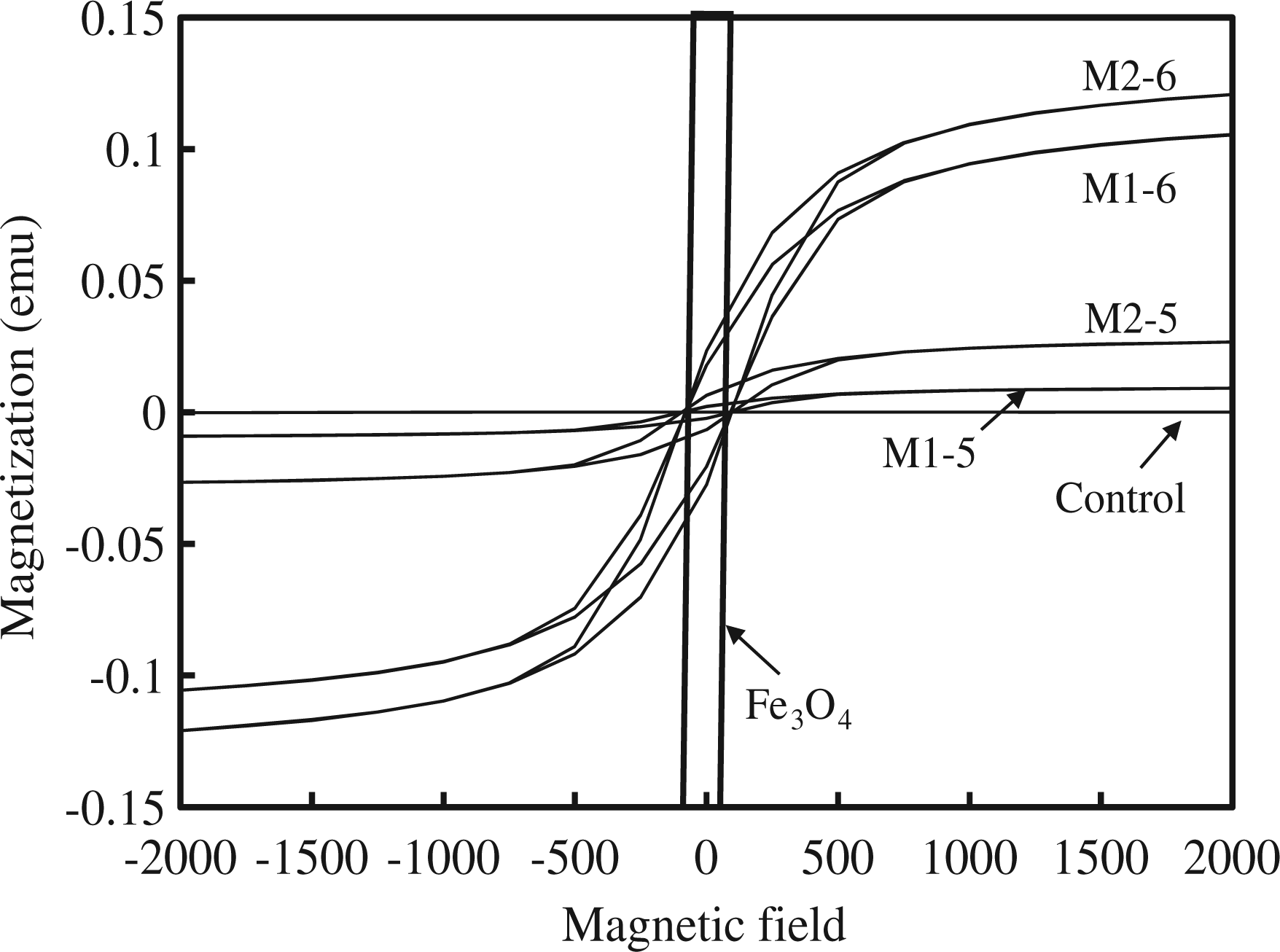

CONCLUSION
The SMPU composite prepared by melt-mixing with Fe3O4 was investigated for magnetic activity. Characterizations of thermal transitions, tensile behavior, and shape memory properties supported the fact that the addition of Fe3O4 did not affect the characteristics of a linear SMPU. Two series, M1 and M2, with different soft segment, PEG, and Fe3O4 content were compared. The shape recovery of both series was greater than 90% for all materials and reproducible through four cyclic tests. Shape retention of the entire series remained greater than 80%. The magnetic activity was demonstrated by vertically raising the SMPU specimens by a permanent magnet; the height was dependent on the amount of Fe3O4 included in the composite. These preliminary findings described a promising method for making magnetic SMPU composites with a uniform distribution of magnetic particles.
Footnotes
Figure 11 appear in color online ![]()
ACKNOWLEDGMENT
This research was supported by Basic Science Research Program through the National Research Foundation funded by the Ministry of Education, Science and Technology (R11-2005-065).
