Abstract
Poly(methyl methacrylate) (PMMA)/hydroxyapatite (HA) composite has potential application in denture base materials. Surface treatment of HA using zirconate coupling agent (ZCA) has been carried out to improve the interfacial bonding between the PMMA matrix and HA filler. The effects of different concentration of ZCA (i.e., 2–8%) on the mechanical properties of PMMA/HA composites was investigated using tensile and three-point bending flexural tests. The morphological properties of the PMMA/HA composites was characterized using field emission scanning electron microscopy (FESEM). It was found that PMMA/5HA-2% ZCA composites exhibited higher flexural strength compared to that of untreated PMMA/HA composites. This is attributed to the enhancement of interfacial interaction between PMMA and HA, which can be evidenced by the FESEM and EDX technique. The kinetics of simulated body fluid (SBF) absorption of PMMA/HA composites were studied at 37°C for immersion duration of 2 months. The mathematical treatment used in analyzing the data was the Fickian diffusion and utilized Fick's second law of diffusion. It is worth mentioning that the kinetics of SBF absorption of the PMMA/HA composites conformed to Fickian law behavior, whereby the initial SBF absorption follows a linear relationship between the percentage gain at any time
INTRODUCTION
Poly(methyl methacrylate) (PMMA) is widely used in prosthetic dentistry application, for example, denture bases, artificial teeth, and impression trays. The use of heat-polymerized, permanent, acrylic resin denture bases has certain advantages owing to their strong and rigid behavior, in which, they should provide the retention and stability of the final denture. When PMMA-powder and methyl methacrylate (MMA) monomer liquid are polymerized a semi-interpenetrating polymer network structure is formed [1–5]. Denture bases materials are responsible for artificial tooth fixation, stability, and distribution of masticatory forces over a large tissue-bearing area [6]. PMMA continues to be used in denture bases materials, which is attributed to its excellent appearance, ease of processing, ease in repair, accurate fit, stability in the oral environment, and superior esthetics [7]. However, the primary problem is its poor strength characteristics [8]. PMMA has been reinforced with additives (e.g., glass fibers, carbon fibers, barium particles, zirconium particles, silica, hydroxyapatite, etc.) to improve its mechanical, thermal, physical, and rheological properties.
Hydroxyapatite (HA), Ca10(PO4)6(OH)2, was used in various biomedical fields such as dental material, bone substitute, and hard tissue paste. HA can be produced either chemically or from natural resources, such as from corals via hydrothermal transformation, or from bones of humans or (preferably) animals, or from enamel (EHA) or dentine (DHA) of teeth. HA has been extensively used as a substitute material for damaged teeth or bone over the past three decades, and its compatibility with surrounding tissues has been experimentally proven [9–11]. HA-reinforced polymers have many potential clinical applications including use as a bone cement, as bioceramic-reinforced polymers for use as a coating of joint replacement prosthesis, and dental implants. Bio-active ceramics like sintered HA and glass-ceramic hybrids are known to experience good binding characteristics to bones
Denture base acrylic resins are subjected to many various types of intraoral and extraoral stresses. For example, repeated masticatory forces could lead to fatigue phenomena, while high-impact forces may occur as a result of dropping the prosthesis. As a result, stress concentration is generated, and denture base acrylic resin can initiate or propagate existent cracks, thereby influencing the failure rate. To compensate for these problems, the ability of the material to withstand the presence of notches and crack propagation is an important factor affecting denture performance. Thus, there is a need for improvement in the fracture resistance of PMMA denture base materials [15–17]. Approaches to strengthening and/or toughening the acrylic resin prosthesis have included modifying or reinforcing the resin by using filler and fiber, as well as chemical modification to produce graft copolymer high-impact resins. There are also some efforts to be done to increase the impact strength by the incorporation of a rubber-like substances in the acrylic resins [5,15,17]. A rubber phase may be introduced into the acrylic matrix as either co-polymer, rubber particles, or as core-shell particles. The most commercially successful method of reinforcement to date is rubber toughening; however, such materials have compromised flexural properties [18,19].
The mechanical properties of PMMA/HA composites could be limited by the incompatibility between the PMMA and HA. Thus, modification of PMMA/HA composites is required in order to achieve a high performance denture base materials with better mechanical properties. Polymeric compatibilizer and coupling agent can play a role to improve the interaction and adhesion between the organic PMMA matrix and inorganic HA particles [20,21]. Our previous works on the surface treatment of HA using silane coupling agent has demonstrated promising results in mechanical and thermal properties, as well as reduced water absorption and simulated body fluid (SBF) absorption of PMMA composites [22,23]. Water absorption of PMMA/HA is an important study for dentistry science. In oral environment, dental restorative materials are exposed to saliva which contain water. Sometimes, the dental materials are exposed to exogenous substances such as acids, base, salts, alcohols, and oxygen, during drinking or eating. SBF test is a method that is well recognized to characterize the
In this article, we investigate the surface treatment of HA by zirconate coupling agent (ZCA), and its effects on the mechanical and morphological properties of PMMA/HA composites. The influence of the ZCA treated HA on the kinetics of SBF absorption for the PMMA/HA denture base composites have been discussed thoroughly.
MATERIALS AND EXPERIMENTAL
Preparation of PMMA/5HA Composites
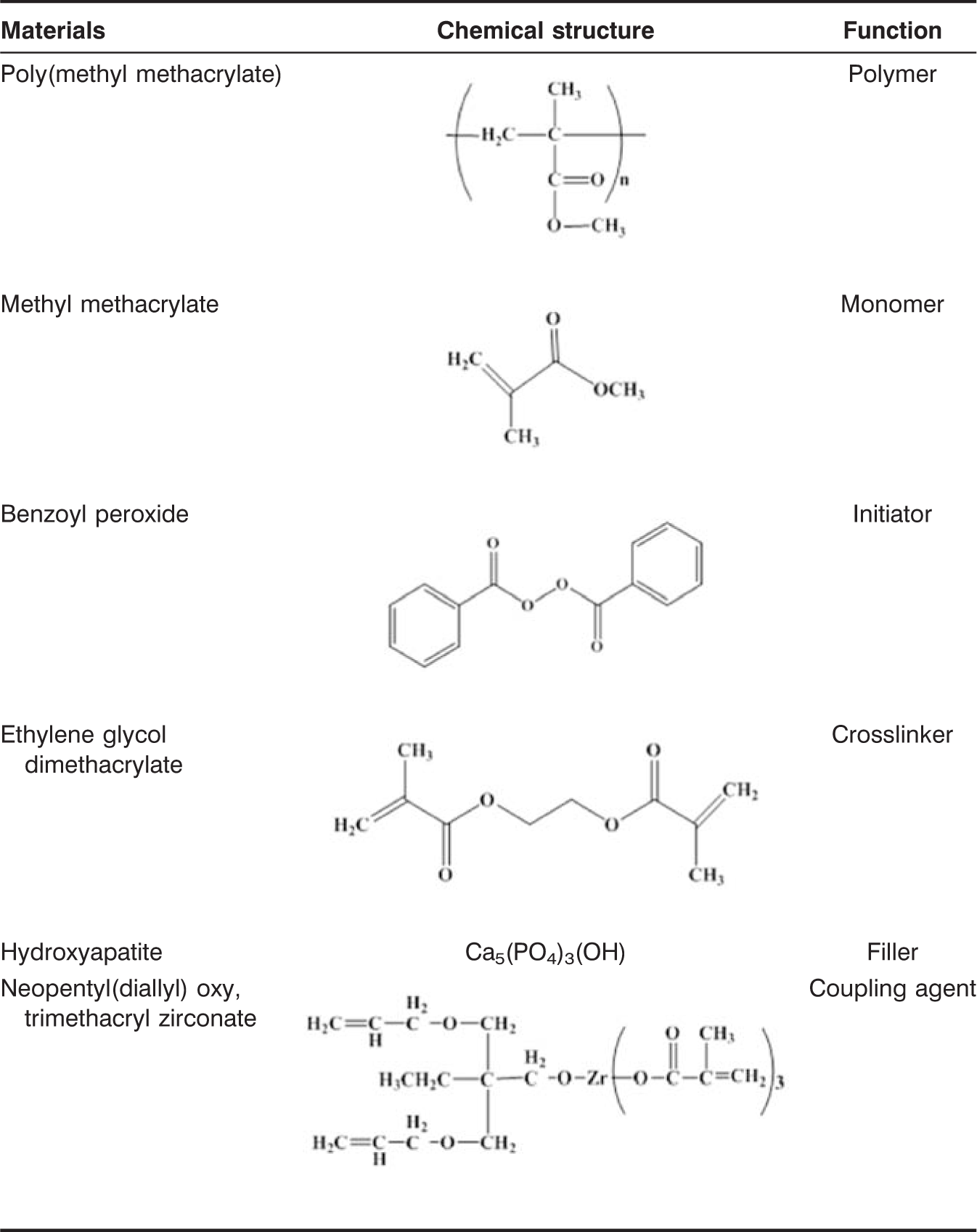
Chemical structure and function of materials.
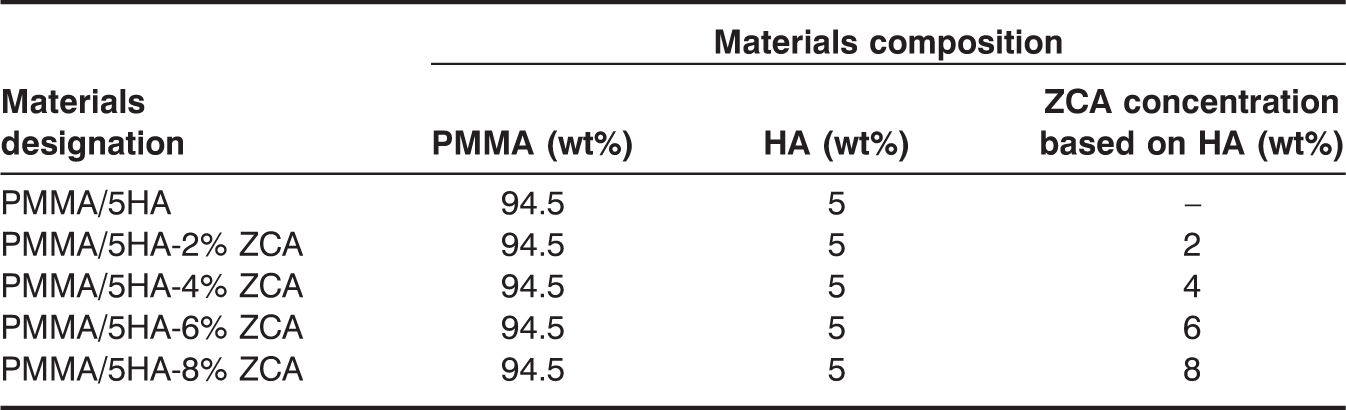
Materials designation and compositions.
Surface Treatment on HA
ZCA (Neopentyl(diallyl)oxy trimethacryl zirconate) was supplied by Kenrich Petrochemical, Inc (USA). The boiling point and flash point of this ZCA are about 104°C and 60°C, respectively. The HA was surface treated using ZCA with different concentration (i.e., 2, 4, 6, and 8 wt% based on HA). The ZCA was added into toluene and was stirred for approximately 30 min in a vessel followed by addition of HA. Later, the HA filler was stirred together with solvent for approximately 1 h. HA filler was then filtered and washed to drive off excess ZCA and solvent. After this, HA filler was dried in oven at 80°C for 24 h.
Characterization of PMMA/5HA Composites
MECHANICAL TESTS
Tensile and three-point bending tests were performed according to ASTM D638 and ASTM D790-03 respectively, using an Instron 3366 machine (USA). The specimen geometry is 100 mm × 13 mm × 3 mm (length × width × thickness). For tensile tests, the gauge length and crosshead speed was set at 50 mm and 5 mm/min respectively. Tensile modulus, tensile strength, and elongation at break were evaluated from the stress-strain data. For the flexural tests, the support span length was set at 50 mm. The testing speed was set at 2 mm/min. At least five samples for each formulation were examined.
MORPHOLOGICAL CHARACTERIZATION
The morphological properties of the fractured surface of the PMMA composites were inspected in a field-emission scanning electron microscope (FESEM, Zeiss Supra 35VP). The sample surfaces were gold coated to avoid electrostatic charging during examination. Energy dispersive X-ray microanalysis system (EDX, EDAX Falcon System) was used to analyze the occurrence of elements in the specimens that were sputtered with gold.
SIMULATED BODY FLUID ABSORPTION
The specimens (in flexural bar geometry) were dried in a vacuum oven at 70°C until a constant weight was attained. The specimens were placed in a container of SBF. The containers were then put in a water bath at temperature of 37°C. The SBF was supplied by B-Braun Medical Industries (Penang, Malaysia). The weight change of the specimens was measured as a function of time after removing the SBF on their surfaces. The percentage change at time 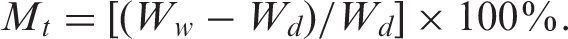
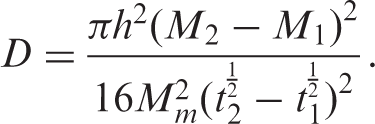
RESULTS AND DISCUSSION
Mechanical Properties of PMMA/5HA Composites
Effect of different concentration of zirconate coupling agent on the flexural properties of PMMA/5HA composites.

Effect of different concentration of zirconate coupling agent on the tensile properties of PMMA/5HA composites.
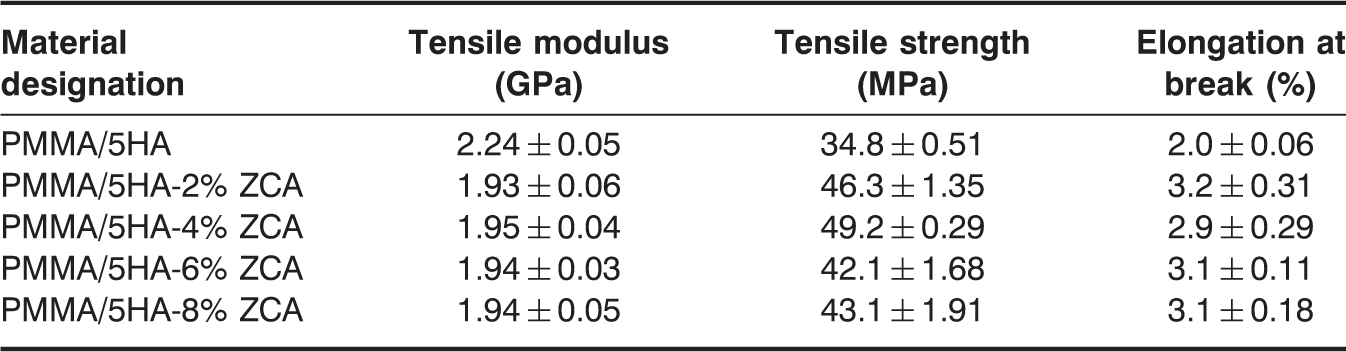
Morphological Properties
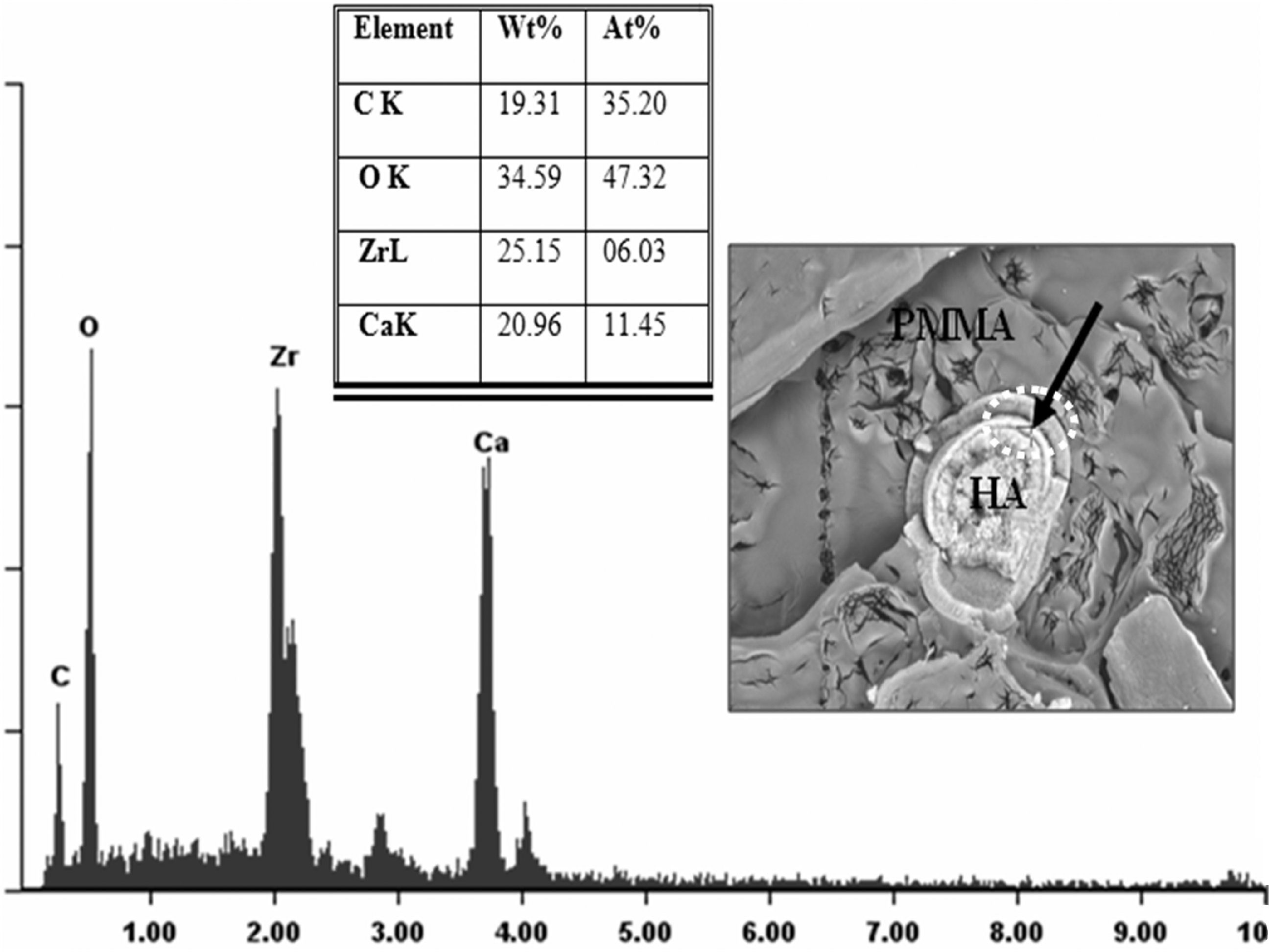
The FESEM micrographs of PMMA/5HA and PMMA/ZCA treated 5HA composites are shown in Figure 1. The smooth surface exhibited by PMMA indicates a typical fractographic feature of brittle fracture behavior. In general, filler can act as stress concentrators in polymer matrix and lead to initiation of failure deformation. In Figure 1(a), it was found that the fracture proceeds along the interface of PMMA and HA. There is noticeable gap between the PMMA and HA particle, which indicates the weak filler-matrix interface. It is interesting to note that, the ZCA treated HA was surrounded by ‘mono- and multi-layer track-like’ interfacial structure (cf. Figure 1(b) and (c)). It is believed that the ZCA can be absorbed onto the surface of HA and form the multilayer through hydrogen bonding. This indicates that the ZCA are able to provide interfacial bonding between HA filler and PMMA matrix. Nonetheless, Figure 1(c) shows the HA particles were pulled off from PMMA matrix. This is maybe due to the excessive concentration of ZCA which induce the plasticizing effects, rather than interfacial contact enhancement. Figure 2 shows the EDX spectra from PMMA/ZCA treated-5HA composite. Recall that the HA used for this study contains O, Na, P, and Ca. Note that the elements of O, Zr, Ca can be detected in the ‘multilayer track-like’ interfacial structure. This may prove that the interface multilayer is associated to ZCA, which contains the Zr element. These morphological observation evidenced the improved adhesion between PMMA and HA in the presence of ZCA. Figure 3 shows the possible interaction between HA and ZCA. It is hypothesized that the interaction between PMMA and HA can be controlled by the concentration of ZCA. One may find that plasticization predominated as the overloading of ZCA.
FESEM micrograph of (a) PMMA/5HA, (b) PMMA/5HA-2% ZCA, (c) PMMA/5HA-4% ZCA. EDX spectra of PMMA/5HA treated with ZCA (Note: The EDX spectra was taken from the arrow pointed area). Possible interaction between HA and zirconate coupling agent.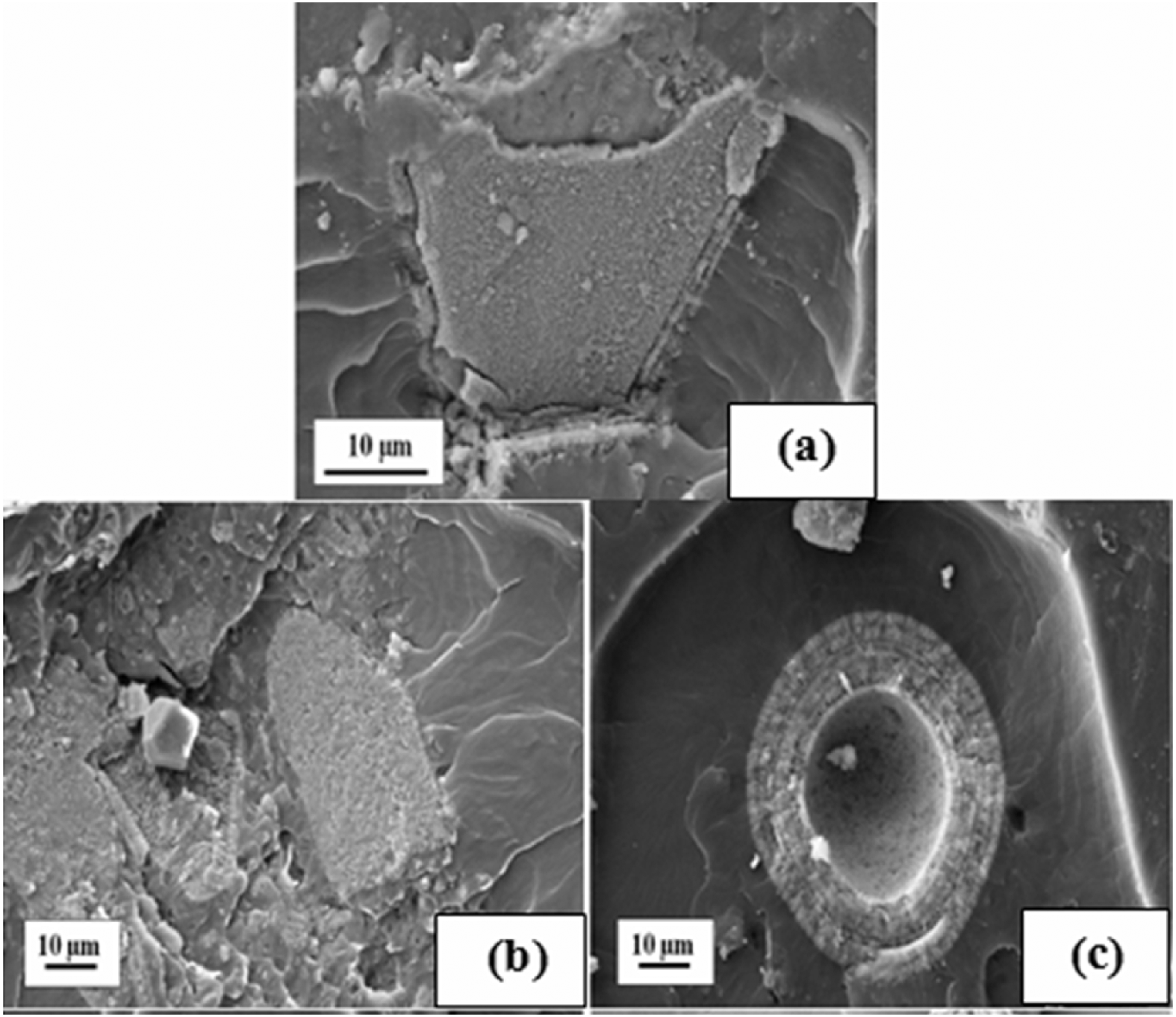
Kinetics of SBF Absorption
Figure 4 shows the effect of ZCA concentration on the SBF uptake of PMMA/5HA composites upon exposure to SBF immersion for 2 months. All specimens showed an SBF uptake process involving a rapid Fickian process which is linear to The effects of zirconate coupling agent concentration on the SBF uptake of PMMA/HA composites.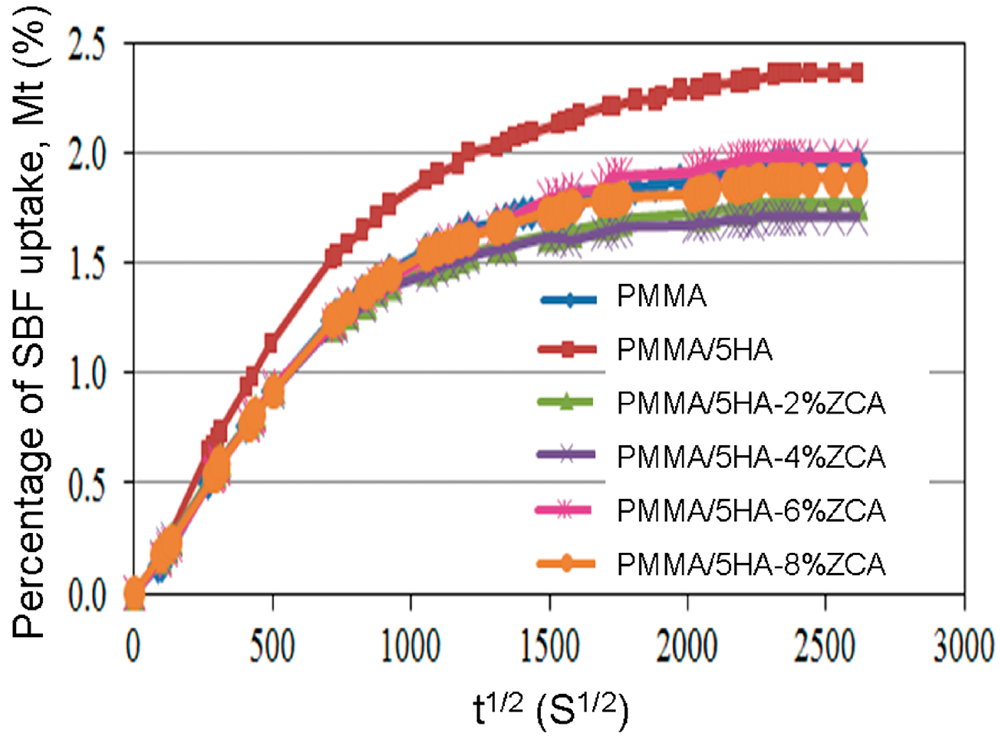
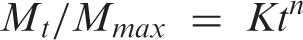
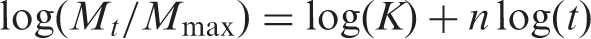
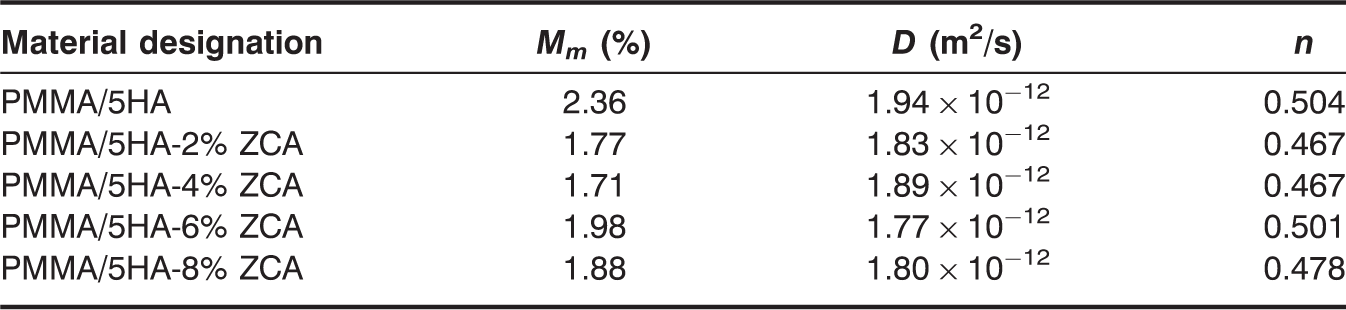
Maximum SBF absoprtion (Mm), diffusion coefficient (D) and diffusional exponent (n) of PMMA/5HA composites and zirconate coupling agent treated PMMA/5HA composites after subjected to SBF tests.
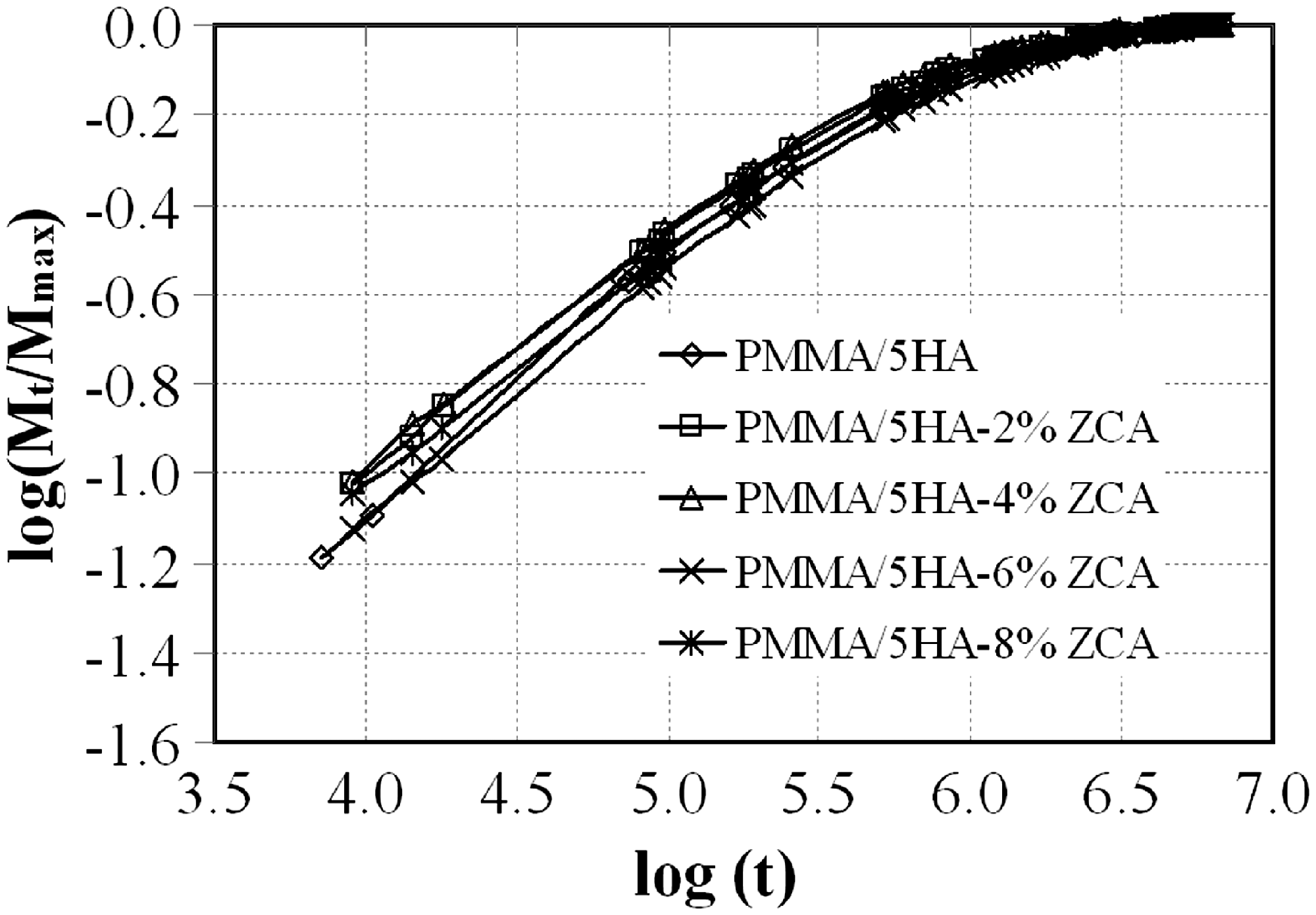
Plot log(Mt/Mmax) vs log(t) for PMMA/HA composites.
CONCLUSIONS
Based on this work devoted to study the effect of ZCA on the microstructure, mechanical properties and SBF absorption behavior of PMMA/HA composites, the following conclusions can be drawn:
Surface treatment of HA using ZCA has resulted improvement in flexural modulus and strength of PMMA/5HA composites, attributed to the better interfacial interaction. The interfacial contact between PMMA and HA can be evidenced using SEM and EDX technique. The SBF absorption of PMMA/HA composites followed the kinetics of Fickian diffusion process. Moreover, the maximum SBF absorption and diffusion coefficient of PMMA/5HA was reduced by the ZCA treatment.
Footnotes
Figure 4 appears in color online ![]()
ACKNOWLEDGEMENT
This work was supported by the Science Fund [MOSTI, Malaysia, grant number 6013318]; Universiti Sains Malaysia [Incentive Grant, grant number 8021013]; Universiti Sains Malaysia [Research University Postgraduate Research Grant Scheme, grant number 8031015]; and USM Postgraduate Fellowship.
