Abstract
To develop a novel hygroscopic PP film available for functional application, superfine down powder (SDP) was blended with polypropylene (PP) to produce blend pellets, and the extruded pellets were hot-pressed into blend films. Scanning electron microscopy (SEM) photos showed the interface between SDP and PP matrix. X-ray diffraction (XRD) results showed that the crystallinity of PP/SDP blend films decreased with increasing SDP content. Compared to pure PP film, PP/SDP blend films showed higher moisture regain, improved thermal stability and more residues at 600°C according to thermo-gravimetric analysis (TGA). Differential scanning calorimetry (DSC) curves showed that the addition of SDP accelerated the crystallization process of PP matrix. The water absorption of PP/SDP blend films with 50 wt% SDP achieved an increase of 11.9%. It was worth noting that the water absorption and mechanical properties of PP/SDP blend films depended on SDP content.
INTRODUCTION
In conventional filled polymer composites, nature feather fibers are blended with synthetic polymers, thereby producing degradable and reinforced composites and it is also a good way to take advantage of nature protein materials [1–5]. An alternative method to reuse protein fibers is the preparation of keratin solution, which was extracted from these protein fibers using complex chemical methods and was then blended with synthetic polymers to produce protein biomaterials. Although keratin fibers can be applied in polymer composites under high temperature, the dimension of keratin fibers restricts their applications. Unfortunately, keratin solution was prone to decompose in the process of extruding and hot-pressing under high temperature inevitably. Therefore, it is necessary to find a new way to promote the usages of protein fibers for functional applications under high temperature.
In this study, down fiber was ground into superfine down powder (SDP), which not only sustains the excellent properties of protein materials, but also has stable structure against high temperature. So SDP can be blended with thermoplastics resin such as polypropylene to produce functional composites; various shapes of polypropylene (PP)/SDP composites can also be produced without the limit of down fiber dimensions. The preparation and characterizations of powder (wool powder, silk powder, and down powder) promote a new application of nature protein materials [6]. Wool powder was blended with PP to produce composite and its properties were studied [7]. Silk powder [8] was produced as one of the useful physical forms of silk fibroin protein for biomaterial applications [9,10].
As well-known, down fibers are used as fillings in clothes and most of them are often processed into a low quality protein supplement for animal feed after usage. This is a huge waste of energy resources. Down powder/viscose blend films were prepared and the dyeing properties were characterized [11]. PP is an engineering synthetic polymer, which has good mechanical properties and low specific gravity. So PP always was selected to act as matrix and has been blend with CaCO3 [12], glass fiber [13,14], EPDM [15], clay [16], and SiO2 [17,18] to produce composites. It was expected that the addition of SDP could improve the water absorption of PP matrix with a little or no loss of mechanical properties, and the PP/SDP composites could be of low density, degradability, good hydrophilicity, good design, and manufacture flexibility, which will be used in hydrophilic fiber, breathable plastic, agricultural sheet, and even biomedical fields [19].
In present work, down powder was blended with PP to produce the pellets, which was then hot-pressed into films. SEM photographs were used to characterize the compatibility between SDP and PP matrix. The morphologies, microstructure, and crystallinity of composites were also characterized.
EXPERIMENTAL
Materials
Isotactic Polypropylene (iPP) with MFR = 45 g/10 min was obtained from Panjin Co.Ltd., China. Down fibers peeled from duck were purchased from Maolong-wuzhong Down Co.Ltd., Shaoxing.
Preparation of Samples
Down fiber was cut into short pieces on a rotary blade and ground into SDP.
PP pellets with different SDP contents were stirred and then transferred into a twin-screw extruder (SHJ-18, China) for adequately mixing. After dried at 105°C for 2 h, the mixed pellets were pressed into blend films between two pieces of PET films sprayed by silicon to improve the unmolding. The hot pressing was carried out at 5 MPa and 190°C for 5 min, on the plate vulcanization machine (XLB-D350 × 350, China). The blend films with 10 wt%, 20 wt%, 30 wt%, and 50 wt% SDP contents were prepared in this study.
Measurements
PARTICLE SIZE DISTRIBUTION OF SDP
Laser particle analyzer (Chengdu Jingxin powder Analyzer Instrument Co., Ltd. JL-1166, China) was used to measure the average particle size and distribution of the SDP, after the powder was fully dispersed in water.
SCANNING ELECTRON MICROSCOPY (SEM) OF SDP AND BLEND FILMS
Morphologies of down fiber, SDP and cross-sectional morphologies of blend films were examined on a SEM (JSM-5610LV), at 25 kV acceleration voltage after gold coating.
WIDE-ANGLE X-RAY DIFFRACTION OF BLEND FILMS
Wide-angle X-ray diffraction (WXRD) measurements were performed on a D/MAX-βA instrument by use of Cu-ka radiation (wavelength = 0.154 nm) to investigate the structure of samples. Generator intensity was 35 kV; generator current was 30 Ma. The blend films were scanned within the range of 2θ = 5–50° in step of 0.02°.
THERMO-GRAVIMETRIC ANALYSIS OF BLEND FILMS
Blend films were scanned by use of a TG 209 F1 (NETZSCH) from 25°C to 600°C at a heating rate of 20°C/min, in flowing nitrogen atmosphere (10 mL/min).
DIFFERENTIAL SCANNING CALORIMETRY OF BLEND FILMS
Differential scanning calorimetry experiments were run on a DSC 204 F1 instrument (NETZSCH) under nitrogen purge. The samples were heated from 20°C to 200°C at a heating rate of 20°C/min. The heat of fusion was calculated with the NETZSCH-Protues-Version-4.8.3 software. The degree of crystallinity was gained from the ratio between the fusion heat values of samples (ΔHf) and 100% crystallinity isotactic polypropylene (Hf = 209 J/g). The formula is shown as follows:
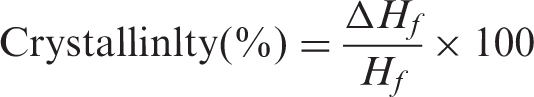
WATER ABSORPTION OF BLEND FILMS
Water absorption behavior was carried out according to the following processes. First, the molded samples (20 × 10 mm2) of blend films were dried in an air-condition oven for 3 h at 60 ± 1°C. Then they were cooled in a dry box for a few minutes until the weight of samples remained the same, and the weight of each sample was determined before water immersion (W1). Secondly, the weighted samples were immersed into water bath at 30 ± 2°C for different time periods before removal from the water bath. Lastly, after these samples were removed from the water bath, the surface of every sample was completely dried with tissue papers and weighed immediately (W2). Water absorption (W3) of each blend film was calculated by the following equation:
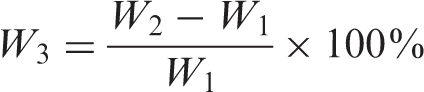
MECHANICAL PROPERTIES OF BLEND FILMS
Stress-strain measurements were performed at 20°C and 65% relative humidity on an Instron 5566 Universal Testing Machine at a gauge length of 30 mm and strain rate of 50 mm/min. The width and thickness of samples were 20 mm and 0.5 mm, respectively. Each sample was tested five times and the results were averaged.
RESULTS AND DISCUSSION
Characterizations of SDP
The particle size of SDP was between 0.2 and 12 µm, as shown in Figure 1. The average size of SDP was around 2.34 µm and 80% SDP was under 4 µm. The most particle size of SDP was around 1µm and 1.5 µm, which kept 22.4% and 22.3%, respectively.
Particle size distribution of SDP.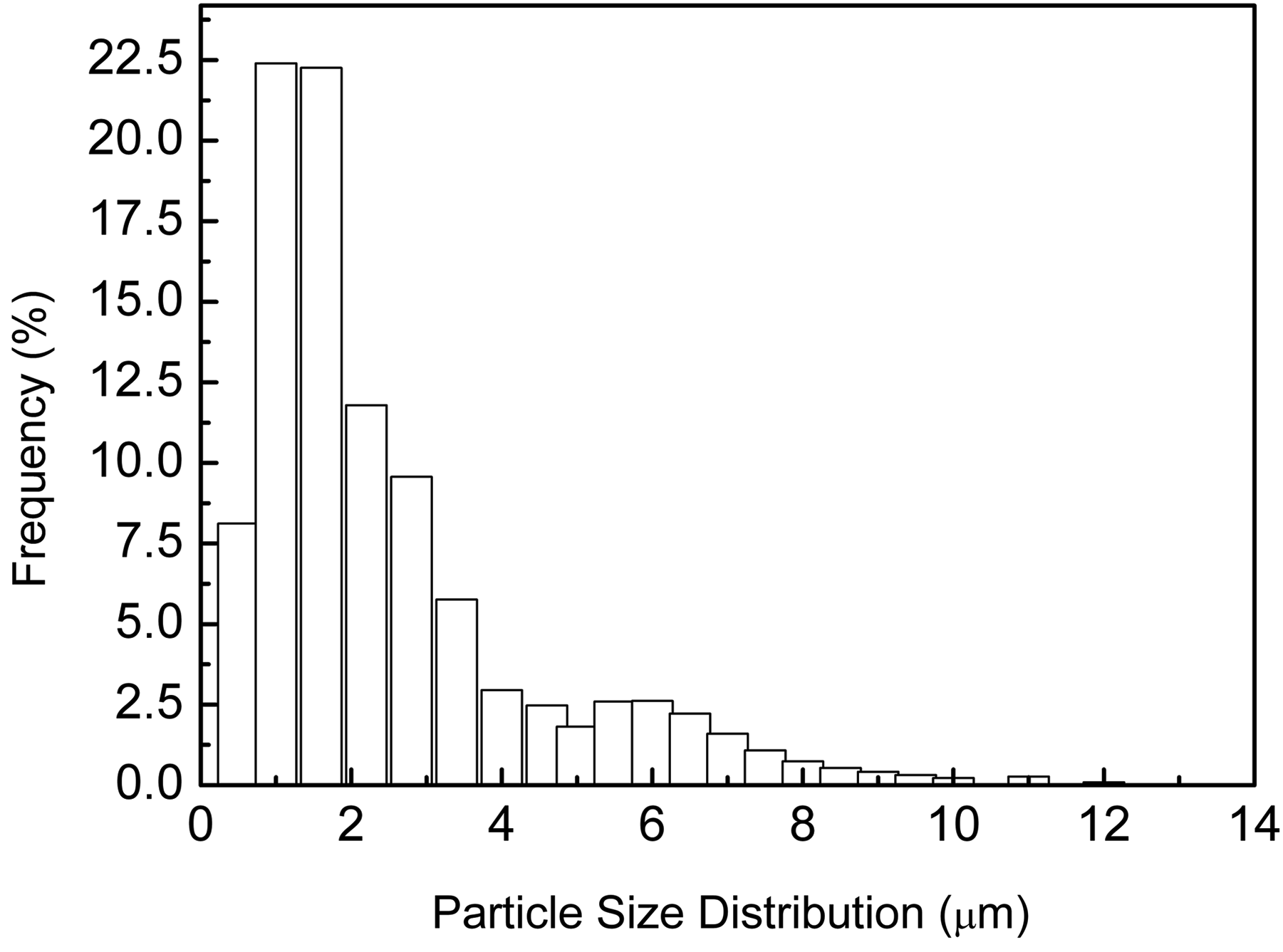
The SEM microphotographs of down fiber and SDP are shown in Figure 2. Figure 2(a) showed a typical untreated down fiber, there were many small branches embedded around the main branch. From the Figure 2(b), it was apparent that most of the SDP were ground into small pieces and some of them were seemed to be needles. Basically, the SDP was solid and showed a roughness surface where many grooves could be found. The powder, which was over 20 µm, was an aggregation of SDP due to strong surface tension.
SEM microphotographs of down fiber and SDP: (a) down fiber; (b) SDP.
SEM Microphotographs of Blend Films
The SEM microphotographs of cross section for pure PP film and PP/SDP blend films are represented in Figure 3. It was evident that PP/SDP blend films showed more roughness cross section than pure PP film. Interestingly, SDP was dispersed uniformly in PP/SDP blend films with lower power content, as shown in Figure 3(b) and (c). Some aggregated SDP could be found in blend film with 50 wt% SDP, as shown in Figure 3(e). The gaps between SDP and PP matrix could be seen clearly, which meant incompatibility between them. In fact, the interface between SDP and PP matrix could form more tunnels for water transmission.
SEM microphotographs of different PP/SDP films. (a) Pure PP film; PP/SDP blend films: (b) 90/10, (c) 80/20, (d) 70/30, and (e) 50/50.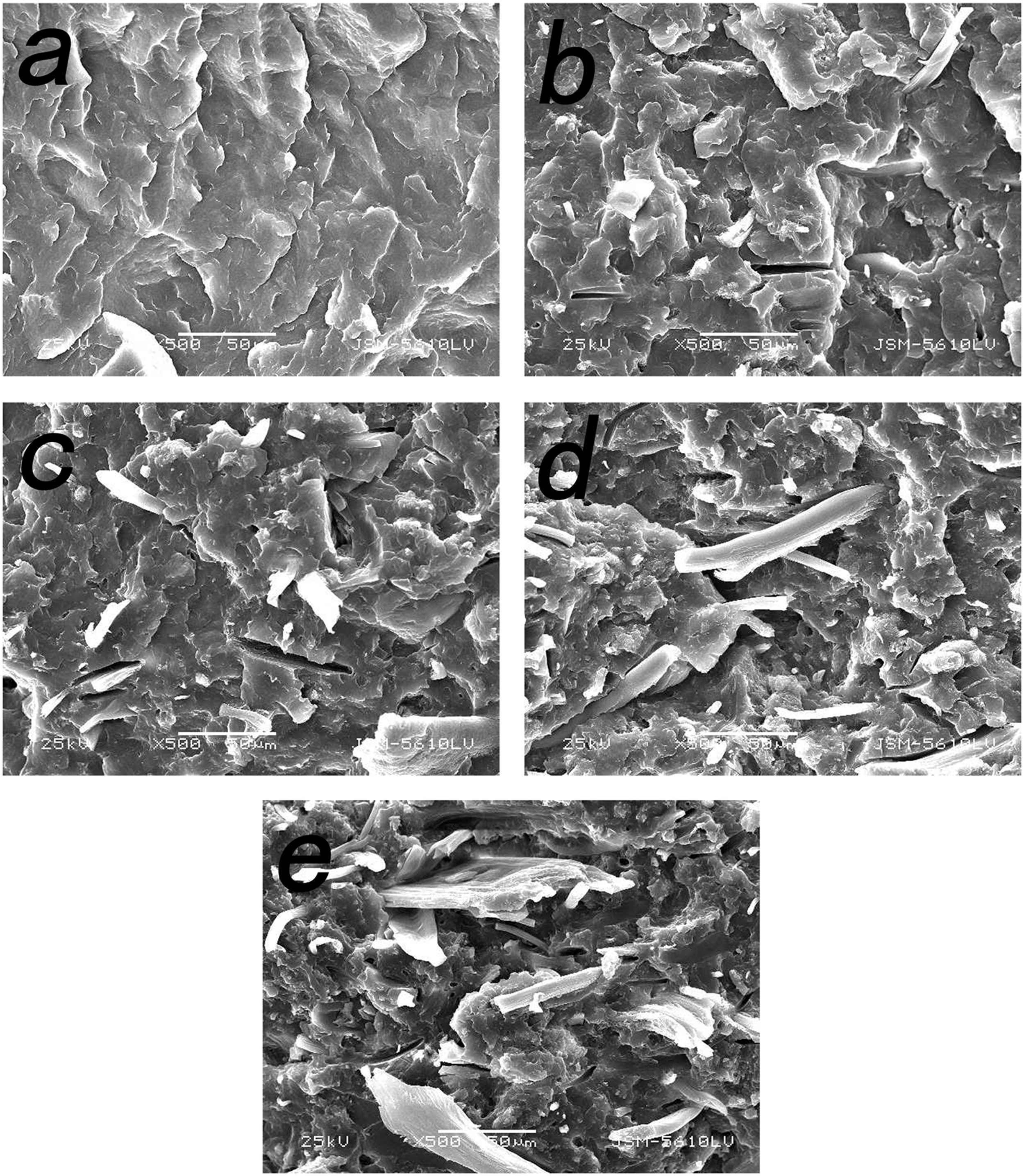
WXRD Analysis of Blend Films
The wide-angle X-ray diffraction patterns of PP/SDP blend films are shown in Figure 4. Isotactic polypropylene exhibits the typical α-monoclinic crystal structure with prominent 2θ peak at about 13.8°, 16.5°, 18.2°, and 20.8° corresponding to the (110), (040), (130), (111) planes, respectively. The intensity of these peaks decreased with increasing SDP content, and the 2θ peak at about 13.8° decreased largely compared to other peaks. It could be attributed to the addition of SDP, which not only had a large amount of amorphous but also destroyed the crystallinity of PP matrix. PP/SDP blend films displayed a new peak at about 16.1° in the WXRD patterns, and the peak was stronger and broader when the SDP content was 20 wt% and then disappeared when the SDP was more than 30 wt%. This might be explained by the forming of β-monoclinic crystals in blend films. When the content of SDP was less than 30 wt%, the SDP could be dispersed uniformly in PP matrix and severed as nucleation agent for forming β-monoclinic crystals, which wasn’t found in WXRD patterns as a result of aggregation of SDP when SDP content was more than 30 wt%.
Wide-angle X-ray diffraction intensity of pure PP film and PP/SDP blend films.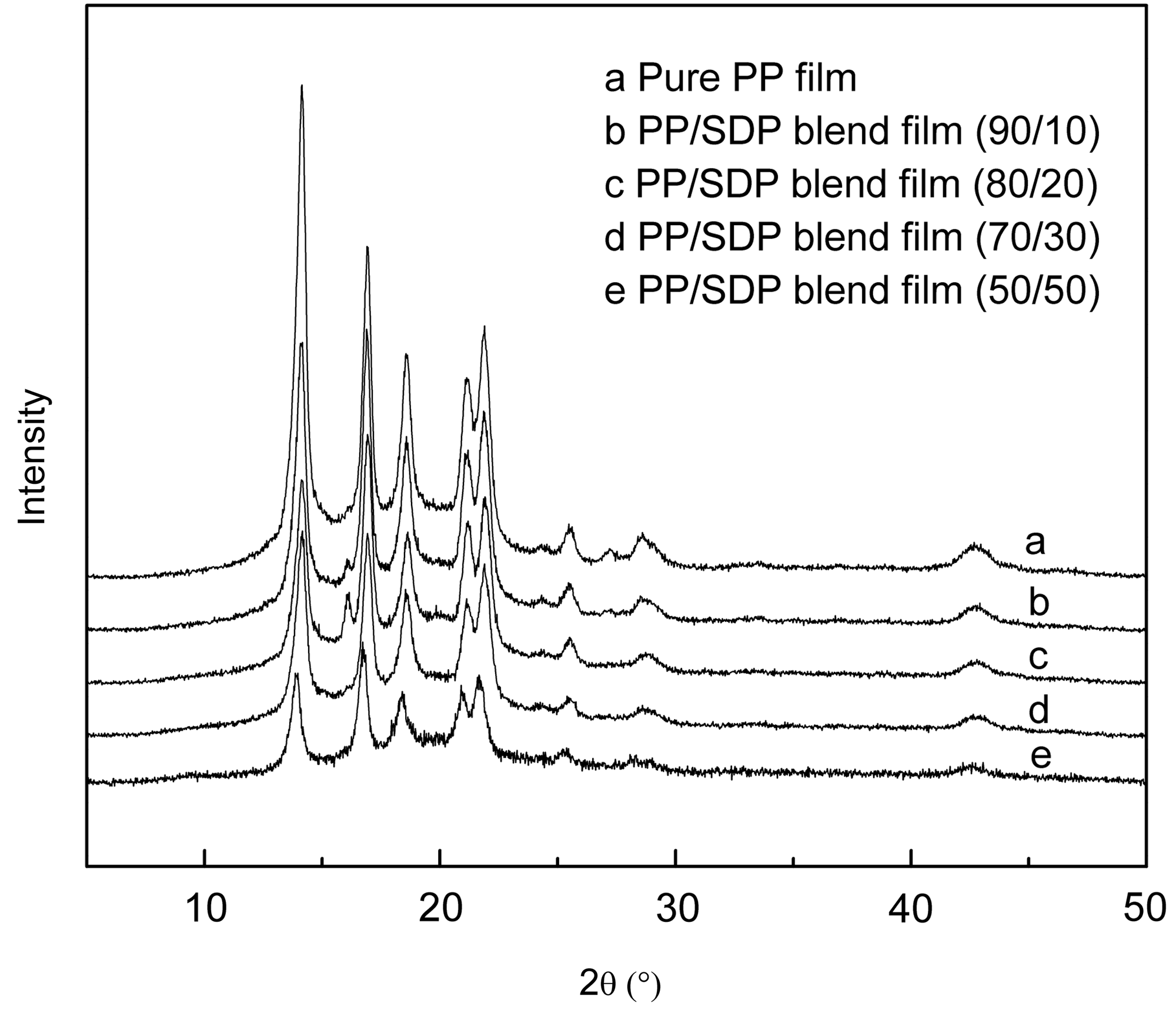
TG Analysis of Blend Films
The TG curves of PP/SDP blend films with different contents of SDP are shown in Figure 5. There were two zones of weight loss observed in the TG curve of SDP. The initial weight loss under about 120 °C corresponded to the evaporation of moisture. The second step started from 220°C to 440°C, which was ascribed to thermal degradation of SDP. An evident weight loss in temperature range of 440–490°C in pure PP curve was associated with the carbonization degradation of PP.
TG curves of pure PP film, SDP and PP/SDP blend films.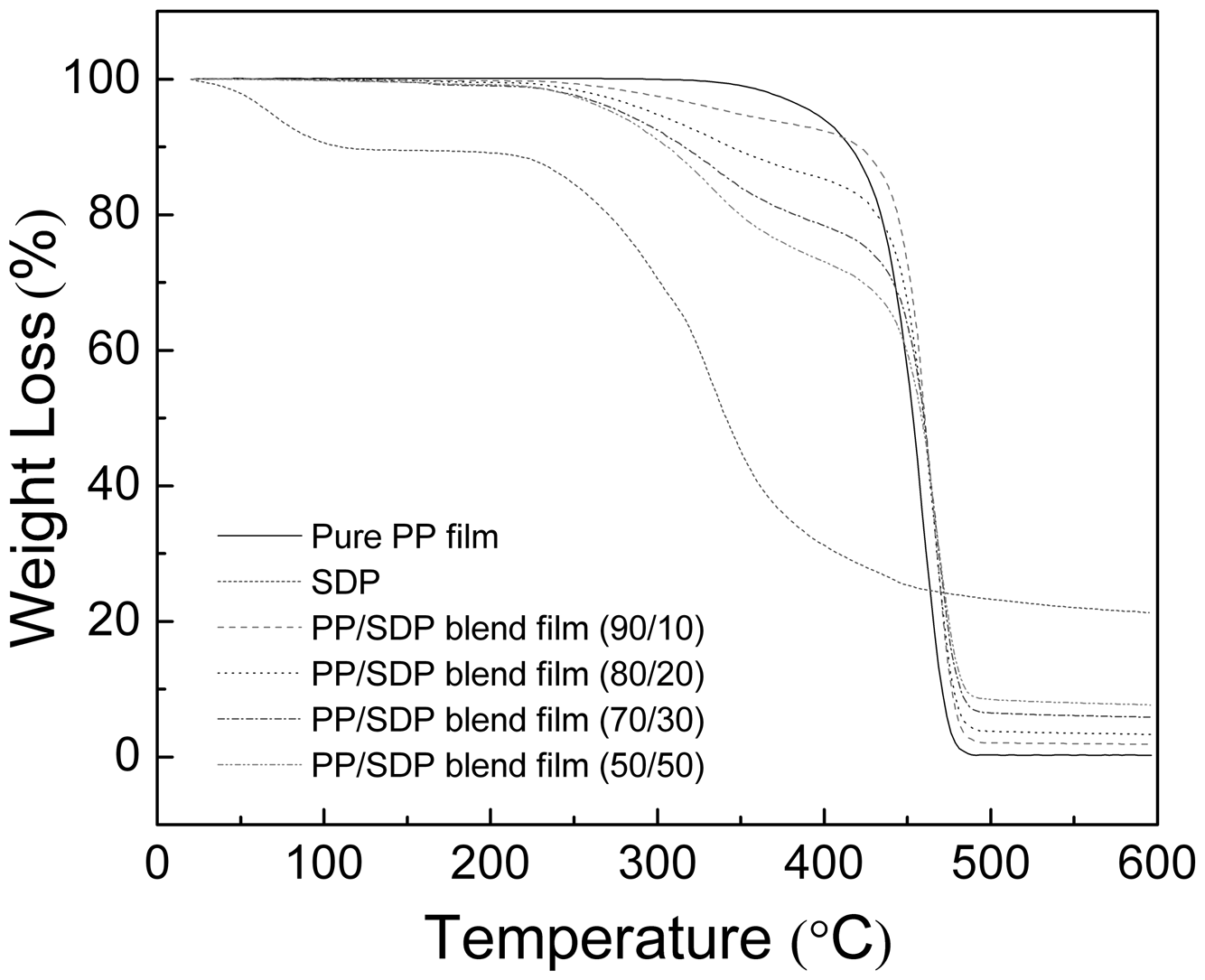
The weight loss of SDP in different blend films could be used to quantify SDP content in blend films. The TG curve of SDP showed about 68.1% weight loss in the range of 180–450°C, and then showed 68.3% weight loss at 600°C, which indicated that SDP retained many residues around 600°C. However, the TG curve of pure PP film showed 99.746% weight loss at 600°C. After the addition of SDP, the PP/SDP blend films left more residues than pure PP film around 600°C and the residues of samples increased with the increase in SDP content, which are summarized in Table 1. Based on the DTG curves of blend films shown in Figure 6, the peaks of PP/SDP blend films shifted higher temperatures compared to pure PP film, which meant that the addition of SDP increased the thermal stability of PP/SDP blend films.
DTG curves of pure PP film, SDP, and PP/SDP blend films. The weight retained of different PP/SDP blend films at 600°C.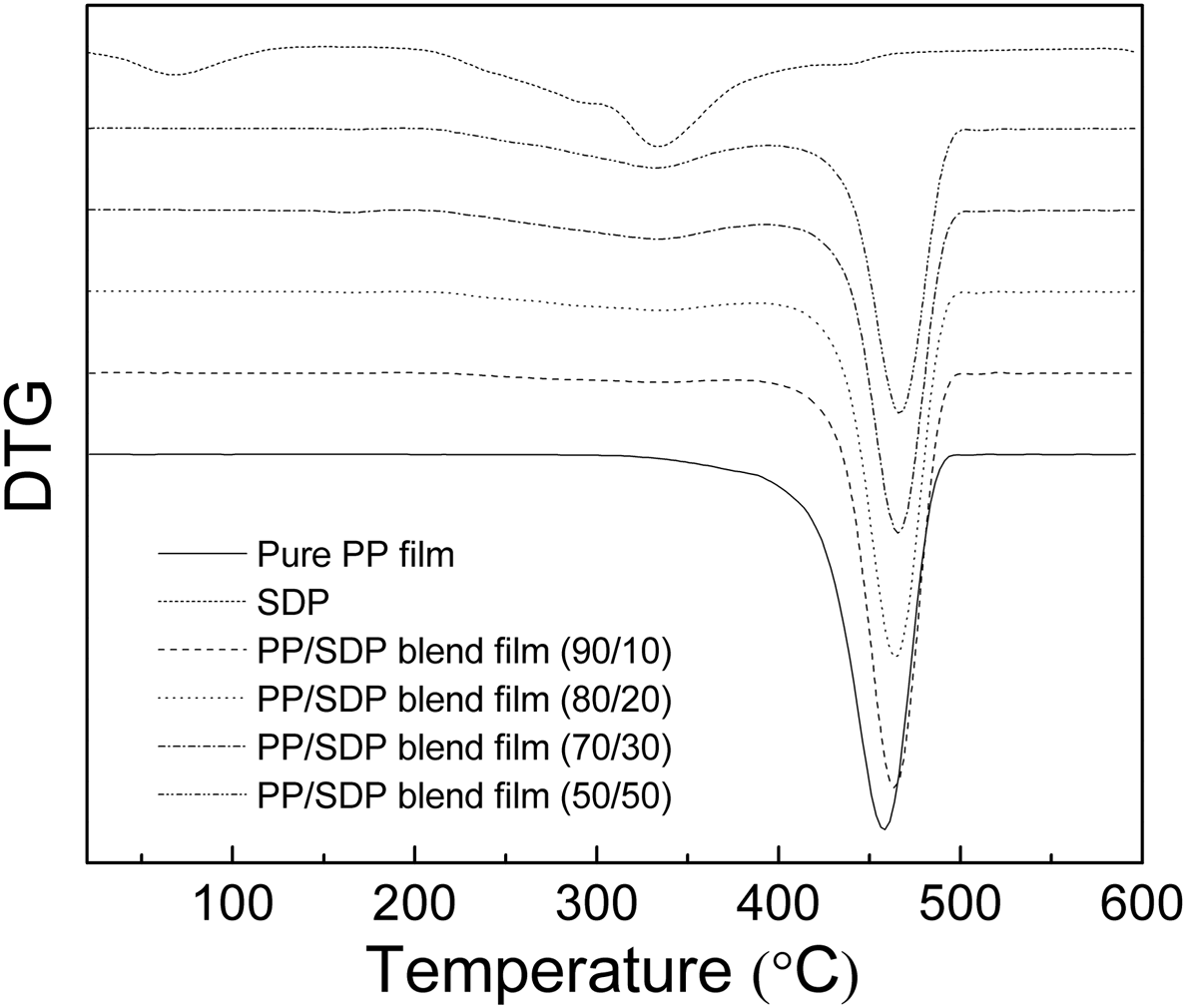

DSC Analysis of Blend Films
The DSC heating and cooling thermograms of pure PP film and PP/SDP blend films are shown in Figure 7, and the values of relevant thermodynamic parameters are summarized in Table 2. In the 1st heating run, the PP/SDP blend films showed no evident new melting peak. So there was no new molecular structure taking place in the extruding and hot-pressing process. With the increase in SDP content, the melting temperature of PP/SDP blend films decreased and then increased when SDP content was 50 wt%. This suggested that the addition of SDP destroyed the crystallinity of PP matrix. Interestingly, the aggregation of SDP lowered this damage. Moreover, the transition enthalpy values and the melting points (Tm) for PP/SDP blend films decreased with increasing SDP content, as shown in Table 2. These results could be attributed to the destruction of crystallinity of PP matrix after addition of SDP.
DSC curves of pure PP film and PP/SDP blend films: (a) 1st heating run; (b) 1st cooling run. DSC temperatures and heats of transition for different PP/SDP blend films.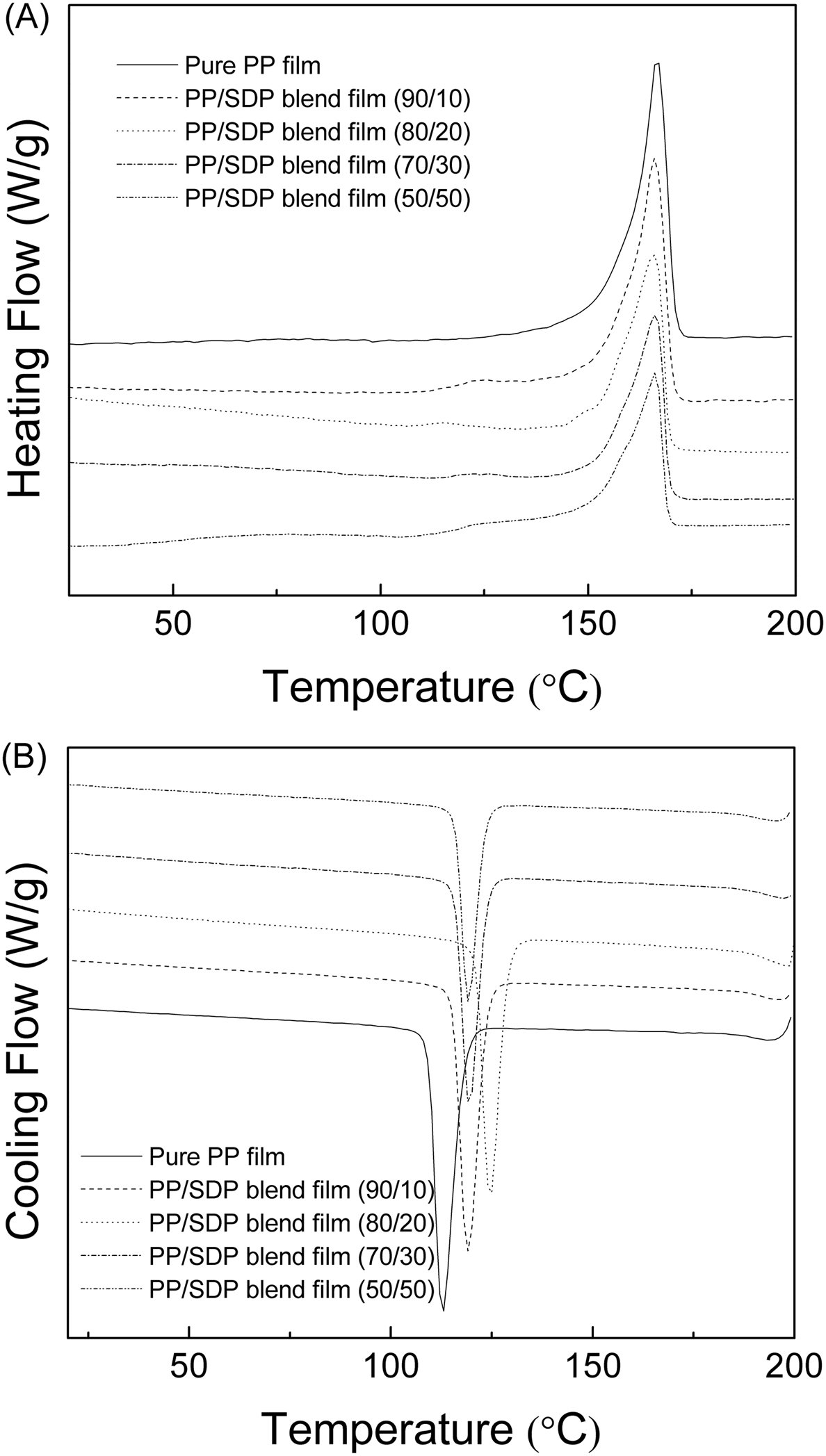
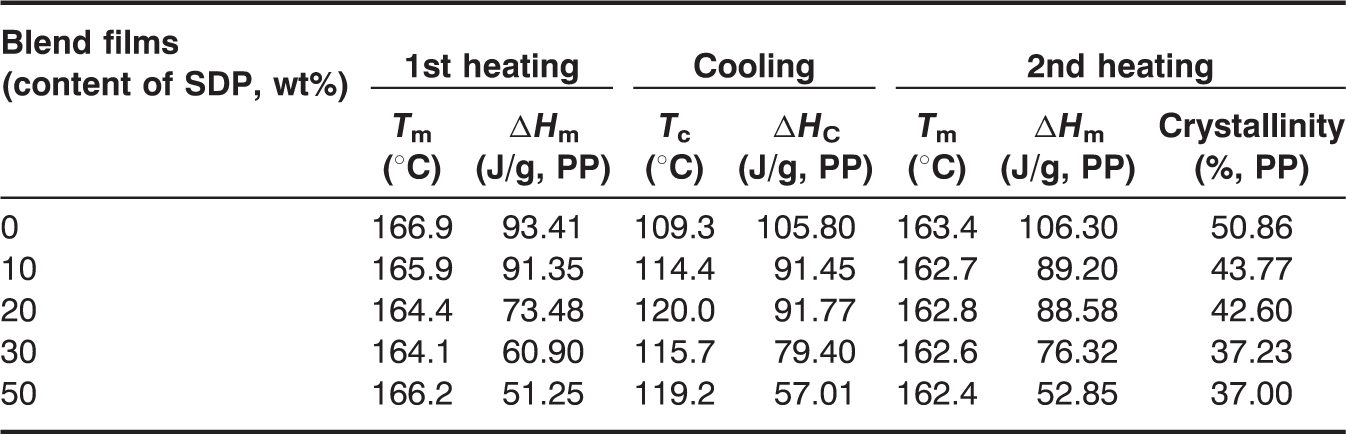
In the cooling run, with increasing SDP content the temperature of crystallization (Tc) of blend films increased and then decreased when SDP content was more than 30 wt%. This may be attributed to the decrease in nucleating ability of aggregated SDP when SDP content was more than 30 wt%.
Water Absorption Characteristics
PP is a hydrophobic synthetic polymer. However, SDP has a high moisture regain because of the presence of amide, carboxyl groups and amorphous regions. So it was expected to improve the water absorption of pure PP film with the addition of SDP. The percentage weight gain of blend films immersed into water is shown in Figure 8.
Percentage weight gains of pure PP film and PP/SDP blend films.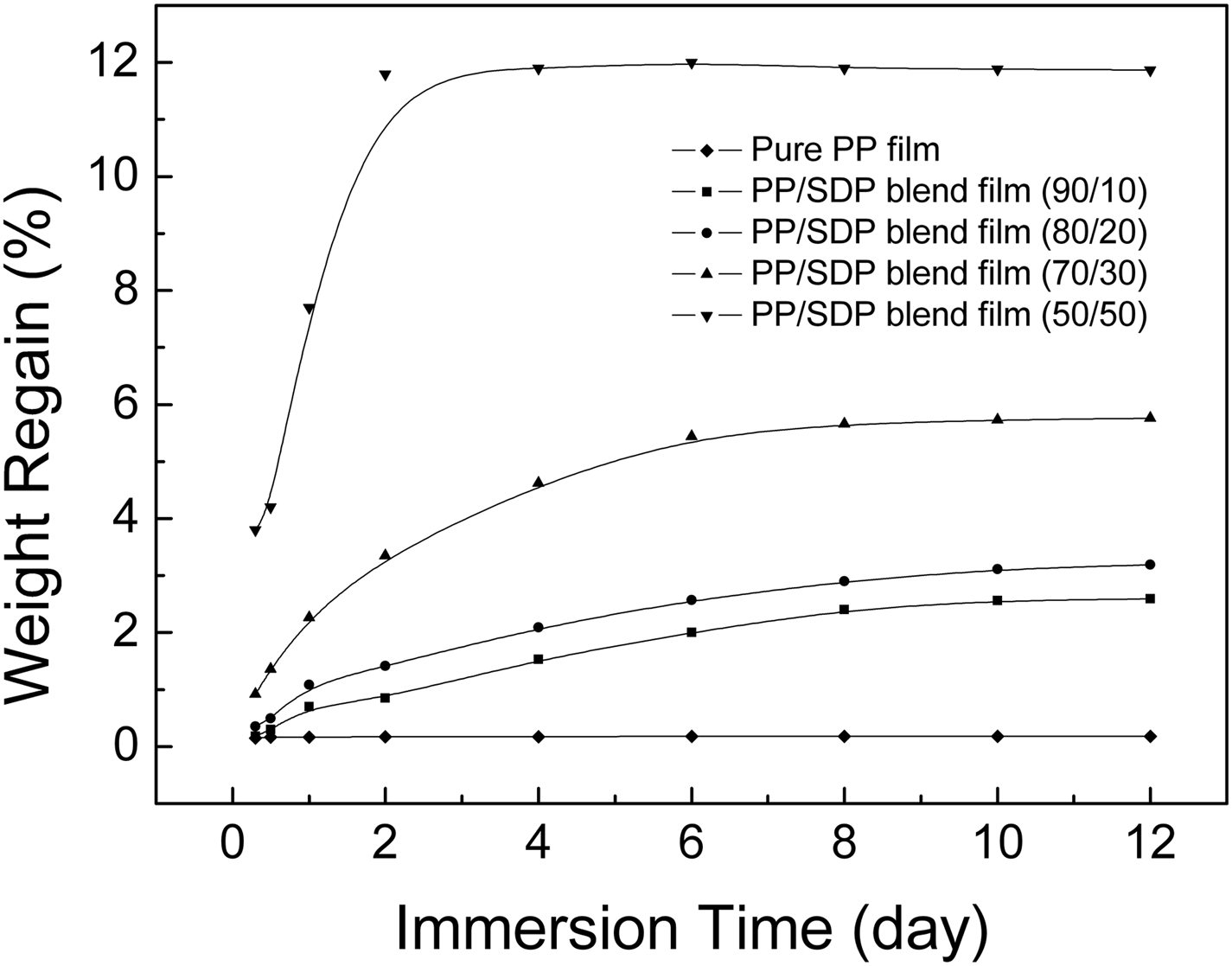
As shown in Figure 8, PP/SDP blend films exhibited reasonably good water absorption compared to pure PP film. In the 1st day, the water absorption of blend films containing 10 and 20 wt% SDP increased quickly, and then went up slowly. The water absorption of blend film with 30 wt% SDP enhanced markedly in the first 6 days and then increased slightly after that. The water absorption of PP/SDP blend film with 50 wt% SDP increased sharply in the first 2 days and remained constant until the water absorption was about 12 wt% in the 4th day. The water absorption of PP/SDP blend films were found entirely dependent on SDP content. Moreover, the water absorption increased largely when the content of SDP was above 30 wt%. This indicated that the continuous SDP formed more tunnels by which water molecules could pass through, and the gaps between SDP and PP matrix could also form more tunnels.
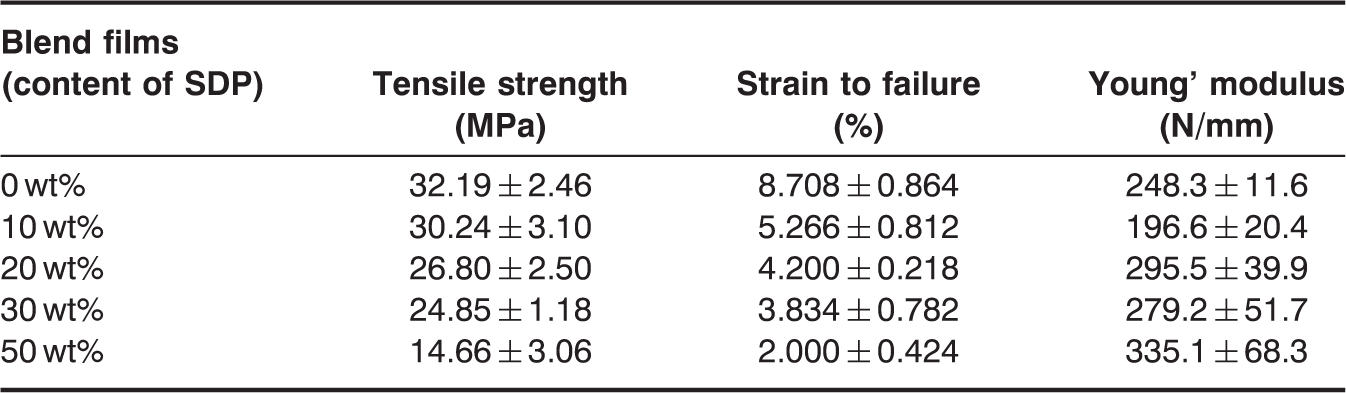
Mechanical Properties of Blend Films
Tensile strength, elongation at break, and Young’s modulus for PP/SDP blend films with different contents of SDP.
CONCLUSION
In this study, SDP was blended with PP to produce higher hygroscopic films. It was confirmed that the addition of SDP could increase the water absorption of PP matrix largely, and PP/SDP blend films had potential applications in different fields and industries. SEM microphotographs showed that SDP dispersed uniformly in PP matrix when the content of SDP was less than 30 wt%. A new diffraction peak at about 16.1° appeared in PP/SDP blend films when SDP content was less than 30 wt% due to the forming of β-monoclinic crystals. However, the peak disappeared when SDP content was more than 30 wt% due to the aggregation of SDP. TG curves showed that the residues of PP/SDP blend films at 600°C increased with increasing SDP content, and DTG curves showed that PP/SDP blend films had improved thermal stability. The WXRD and DSC curves showed that the crystallinity of blend films decreased due to the addition of SDP, which has a large of amorphousness and destroyed the crystallinity of PP matrix. The temperature of crystallization (Tc) of PP/SDP blend films increased due to the nucleating ability of SDP. The water absorption of blend films increased due to the tunnels formed by continuous SDP and the gaps between SDP and PP matrix especially when SDP was more than 30 wt%. The average values of tensile strength and strain to failure decreased with the increase in SDP content due to poor interfacial adherence between SDP and PP matrix. In addition, the Young’ modulus of PP/SDP blend films increased because SDP was stiffer than PP matrix.
