Abstract
The influence of ZnO nanoparticles on the dielectric properties, dielectric relaxation behavior, and thermal stability of PC host was investigated at different ZnO nanoparticles concentration. The dielectric study was carried out over a frequency range from 500 Hz up to 1 MHz as a function of ZnO concentration. Results obtained from the best fitting of relative permittivity data with Yan and Rhodes model reveal that the dielectric relaxation is not a single relaxation process and there are two values of relaxation time for each nanocomposite at room temperature and domain frequency range. Furthermore, addition of ZnO nanoparticles to PC host changes the dielectric properties of PC, mainly, increases relative permittivity, dielectric loss, and AC conductivity while decreases the impedance values of PC host. Moreover, effect of temperature on AC conductivity of ZnO-PC nanocomposites at 1 kHz was also considered. Besides, dielectric relaxation behavior of PC was investigated at 165°C (above the glass transition temperature of PC) in the domain frequency range. At this elevated temperature (165°C), dielectric loss data shows a single relaxation peak (α-relaxation) in the domain frequency range and was successfully fitted with Debye equation. Also, it was found that as the content of ZnO nanoparticles increases in the PC host the frequency of the peak maximum (fmax) shifts toward higher frequency value and as a result decreases the value of relaxation time. Additionally, this study shows that the isothermal effect of ZnO nano particles on the α-relaxation of PC host is similar to the effect of temperature. Both of them have a linear dependence with ln(fmax). Furthermore, addition of ZnO nanoparticles to PC host will decrease the thermal stability and glass transition temperature of PC host. Finally, there is a strong evidence from the obtained dielectric and thermal results that addition of ZnO nanoparticles to PC host will highly enhance the chain mobility and also increase the polar character of PC host.
INTRODUCTION
Metal oxide nanoparticles are popular fillers due to their addition to polymers in the expectation of obtaining unique physical properties which cannot be achieved by adding micron-sized particles [1–3].
Transparent polymer such as polycarbonate is well known as a strong polymer, excellent for electrical applications, because of its high dielectric strength and high volume resistivity which decreases only slightly as temperature or humidity is increased. Due to its advantageous properties it can be used in electrical insulators and connectors, medical equipment components, lenses, card guides, aircraft, and missile components. [4]. Furthermore, depending on additives type, aggregation nature, adhesion with polymer host, etc., addition of additives such as metal oxides, salts, ceramic, etc. will modify the physical properties of polycarbonate host. Interesting results were obtained by us in the electrical, optical, and thermal properties of PC/MnCl2 or PC-ceramic composites [4–6]. Besides, there are many studies reporting the effect of additives in the nano-scale (such as CNT and metal oxides) on the physical properties of polycarbonate [7–9]. Particularly, nano-ZnO, as one of the multifunctional inorganic nanoparticles, has drawn increasing attention in recent years due to its prominent physical and chemical properties, such as chemical stability, low dielectric constant, high luminous, effective antibacterial and bactericide along with intensive ultraviolet and infrared absorption. Therefore, nano-ZnO can be potentially used as gas sensors, semiconductors, varistors, piezoelectric devices, antibacterial and bactericide, field-emission displays, and UV shielding materials. Moreover, ZnO nanoparticle, like other nanoparticles, possesses high surface energy, which may result in the agglomeration of particles when ZnO nanoparticles are dispersed in organic solvent and matrices [1–6].
Zinc oxide nanoparticles have been studied as fillers of a series of polymers such as polystyrene, polystyrenebutylacrylate, PMMA, etc. These studies show that addition of small amount of ZnO nanoparticles to the polymer matrix will modify the physical properties of the polymer host such as mechanical, thermal, electrical, and optical properties. The modifications in the physical properties, however, depend on the base polymer, size, distribution and dispersion of the nanoparticles, and on the adhesion at the filler matrix [3,10–15]. Recently, there are also other studies focused on mechanical, thermal and optical properties of ZnO-PC nanocomposites [3,16–17], but to our knowledge, no study has reported the effect of ZnO nanoparticles on the dielectric relaxation behavior of PC and also the correlation between thermal stability and dielectric relaxation behavior.
This work aims to investigate the effect of ZnO nanoparticles on the dielectric properties (such as relative permittivity, dielectric loss, impedance, and AC conductivity), dielectric relaxation behavior, and thermal stability of the PC host at frequency range 500 Hz to 1 MHz and at different temperature values. Correlations between dielectric results, SEM morphology, and thermal analysis (TGA and DSC) are presented and discussed.
EXPERIMENTAL
Films Preparation
MATERIALS AND NANOCOMPOSITE PREPARATION
Zinc Oxide (ZnO)-Polycarbonate (PC) nanocomposites were prepared using the simple casting method at weigh ratios 0, 3, 5, 7, and 10 wt% ZnO nanoparticles. Polycarbonate resin (product by ACROS ORGANICS Company, New jersey (USA), MW 64,000) was dissolved in dichloromethane and stirred at 30°C using the magnetic stirrer for 2 h. ZnO nanopowder (average particle size of 53 nm, Aldrich) was added to polycarbonate solution and re-stirred again for 24 h until ZnO nanoparicles became well dispersed in PC solution and also the composition became highly viscous and homogeneous. This viscous solution was cast in a glass petri dish (dia. of 5 cm). The solvent was evaporated at room temperature for 24 h and placed inside oven at 40°C for 2 h. Samples of different concentrations were kept inside vacuum dissector. The film thickness (∼0.3 mm) was measured using electronic digital micrometer (MOTOTOYO – China) with 0.001 mm resolution.
Scanning Electron Microscopy
The morphology of the prepared nanocomposites were examined using a Jeol scanning electron microscope (J.S.M. 6380) with low and high vacuum. The operation system was controlled by IBM computer. Samples were coated with gold in automatic sputter coater (Polaron SC 500), and then transferred to the electron microscope to be examined. The electrical image shift of
Dielectric Measurements
Dielectric measurements were carried out using an HP 4194 A-impedance analyzer over a frequency range 500 Hz to 1 MHz and temperature range from room temperature up to 165°C. The capacitance ‘C’ and the loss tangent ‘tan δ’ were obtained directly from the bridge from which the AC conductivity (σAC), relative permittivity (ε′), dielectric loss (ε″), and impedance (Z) were calculated. Calibration (short and open) of the impedance analyzer was done before the measurements. Disk shaped specimens were cut from the nanocomposite sheets (0, 3, 5, 7, and 10 wt% ZnO) and placed between the copper plates (10 mm diameter) of a test sample holder placed in a shielded cell designed for this purpose. The two leads of the holder were connected to the terminals of the impedance analyzer [4].
The relative permittivity (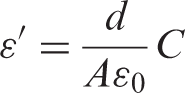
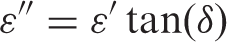
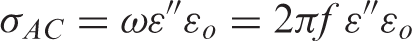
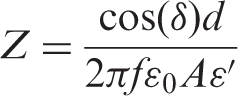
where C is the capacitance of the sample, d is the sample thickness, A is the sample area,
Differential Scanning Calorimetric
The thermal analysis of the samples was studied using Shimadzu DSC 60. The DSC experiments were run at a heating rate of 10°C/min with nitrogen purged at a flow rate of 100 mL/min. Each sample was carefully prepared and measured out to have approximately the same weight (
Thermogravimetric Analysis
A Perkin Elmer Thermogravimetric/Differential Thermal Analysis (TG./DTA 6300 brand) was used to determine the thermal stability of ZnO-PC nanocomposites in the temperature range 50–300°C and scan rate of 10°C/min in air. The weight ratio of ZnO-PC nanocomposites was determined from the TG/DTA scan output. The weight loss ratio was calculated for each ZnO-PC nanocomposite as following: Weight loss ratio (%) = (100% weight ratio) − (weight ratio at 300°C).
RESULTS AND DISCUSSION
Structural Analysis
Figure 1, shows the SEM micrographs for 3 wt% ZnO-PC (Figure 1(a)) and 10% ZnO-PC (Figure 1(b) and (c)). Figure 1(a) shows the formation of nano to micro ZnO agglomerates or clusters which are randomly distributed in the PC matrix. Their size are approximately between 100 and 200 nm. However, such a result was also reported by Carrion et al. [3] when they added 0.5 up to 5 wt% ZnO nanoparticles to PC matrix. Furthermore, these clusters become more closed to each other when ZnO nanoparticles content increased up to 10% (Figure 1(b)) and these clusters will aggregate to form sphere-like clusters in the larger micro scale (Figure 1(c)). However, it is clear that the agglomerates or clusters size increases with ZnO content in the PC host. Additionally, sphere-like morphology indicates a weak adhesion or interfacial bonding between ZnO nanoparticles and PC host [5,18].
(a) SEM micrographs for 3% ZnO and (b–c) 10% ZnO.
Dielectric Properties and Dielectric Relaxation Behavior
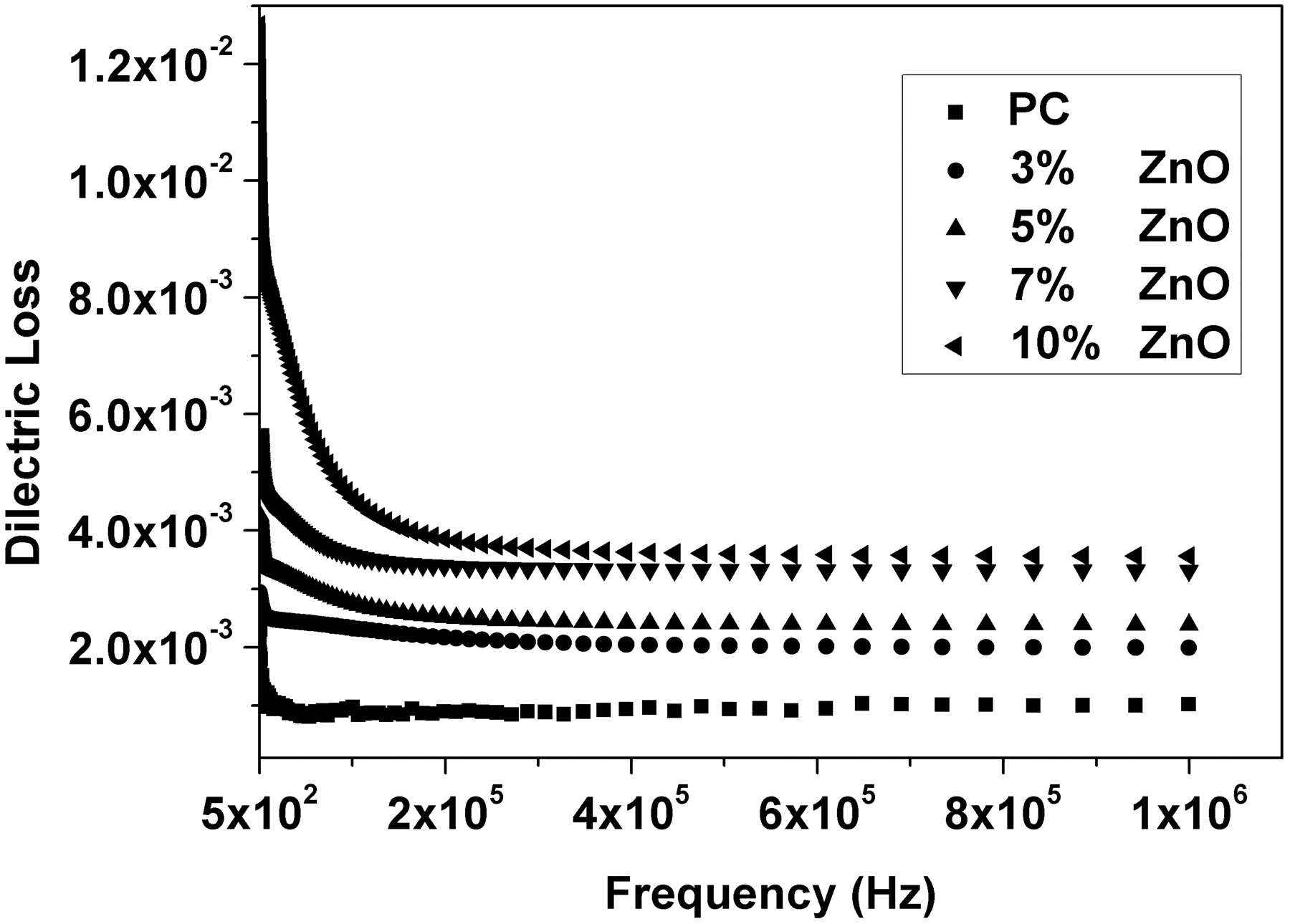
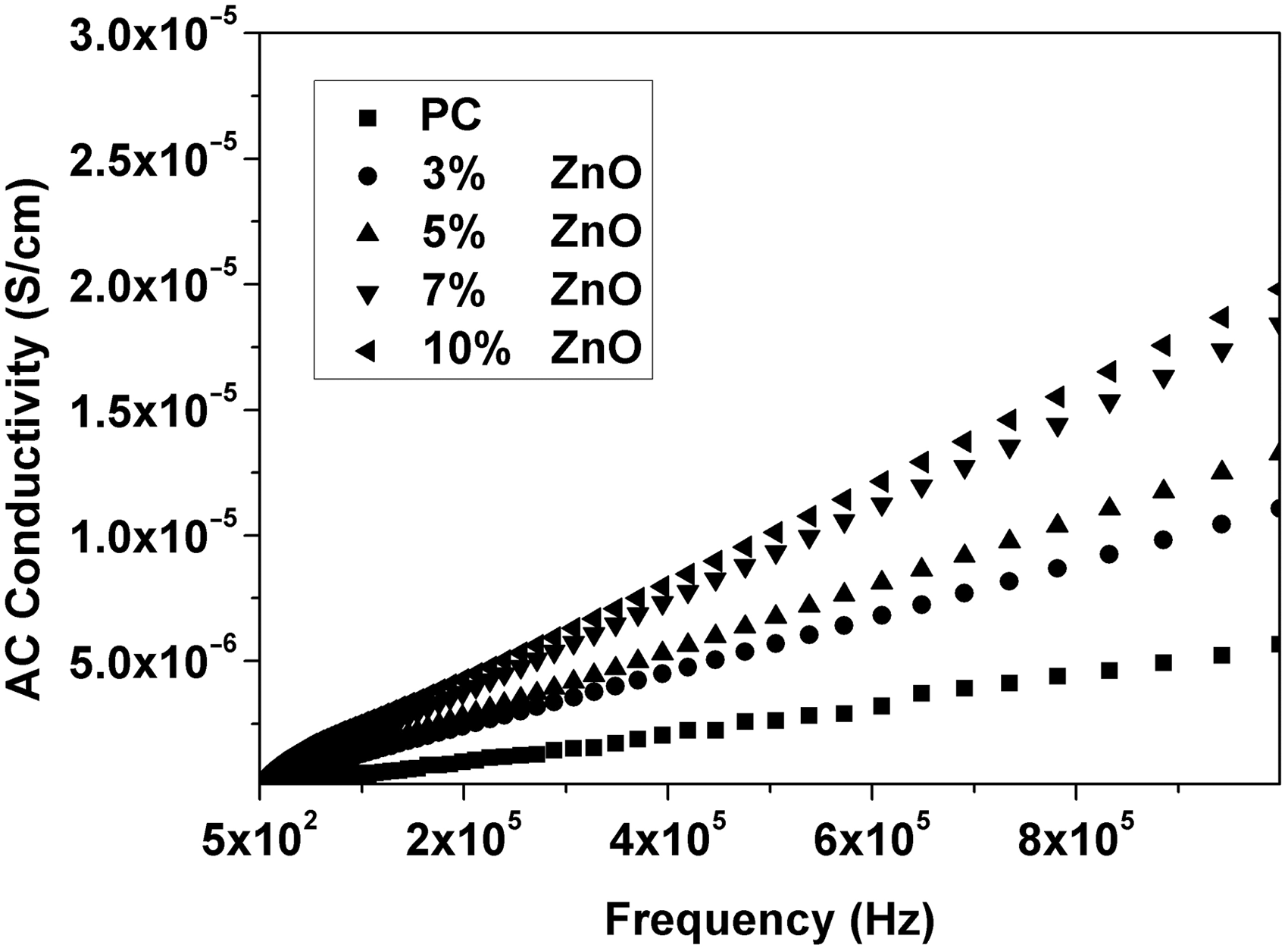
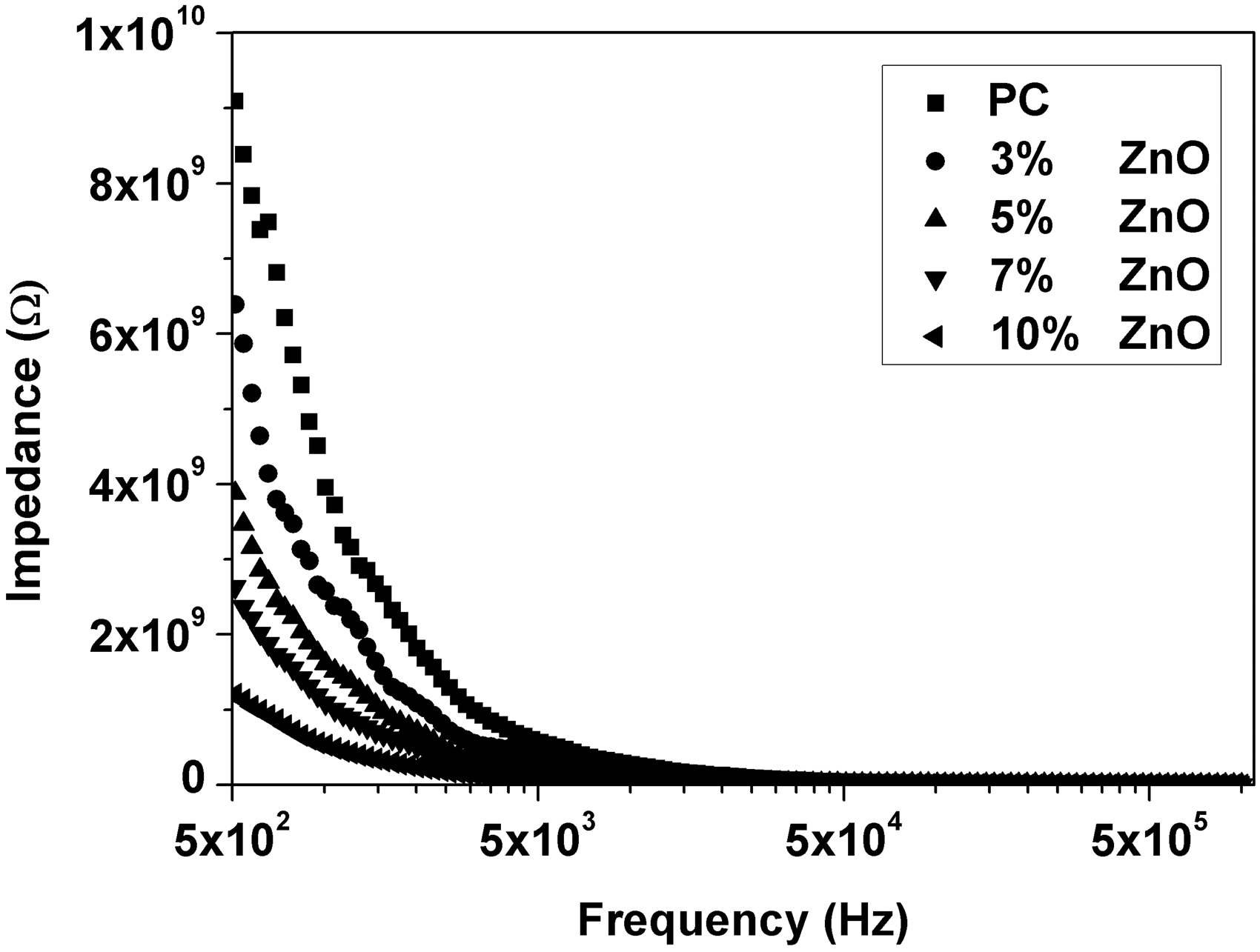
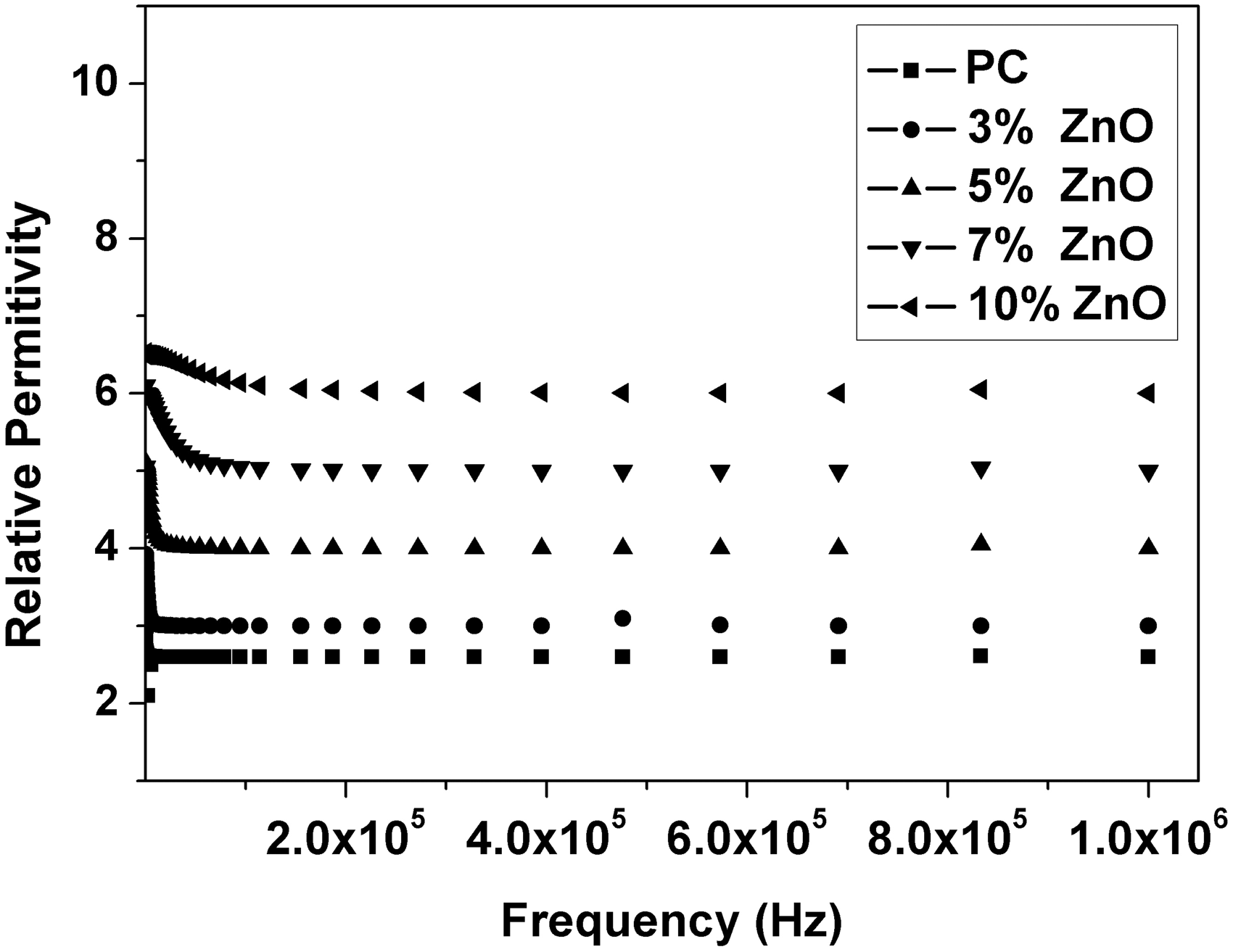
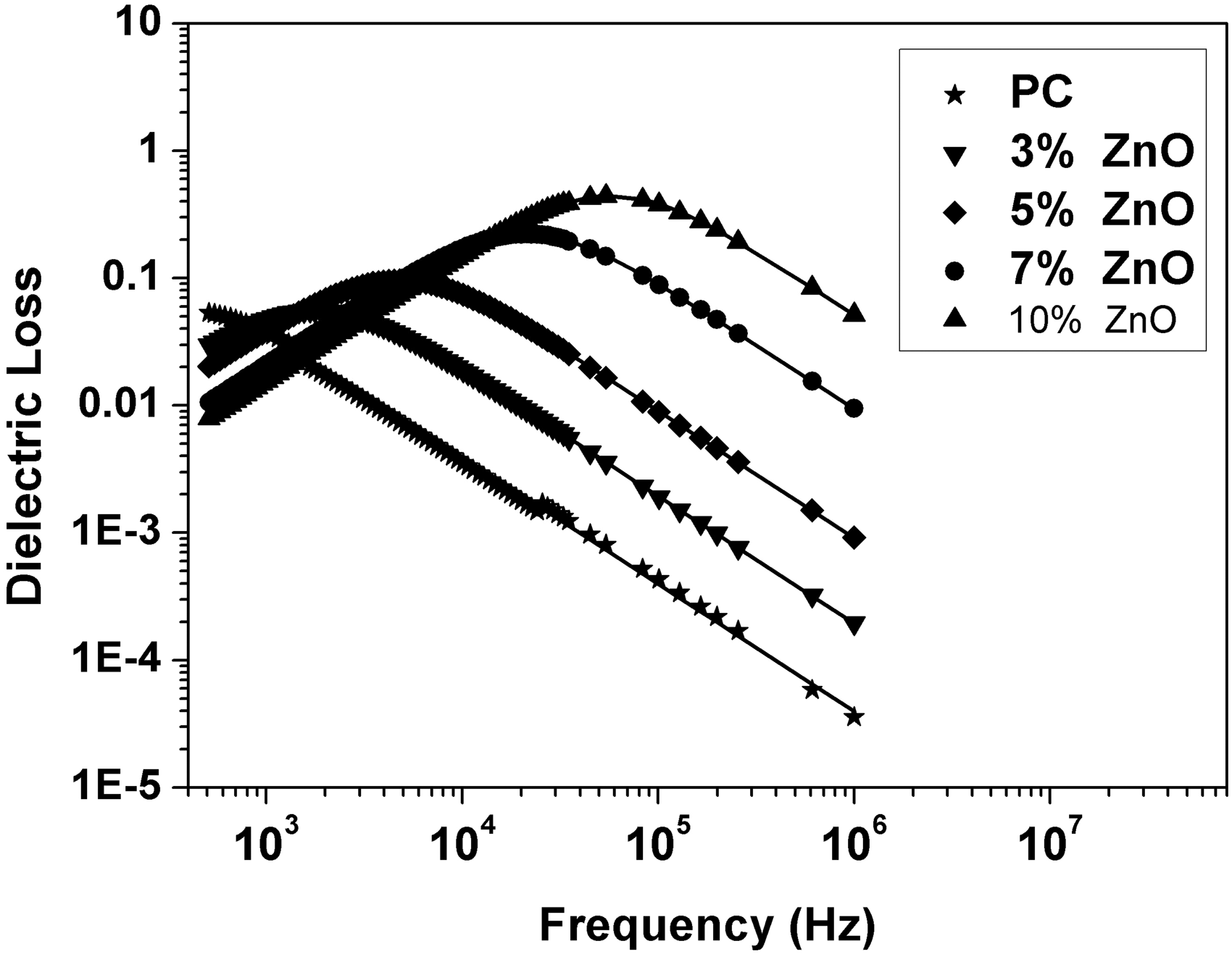
Relative permittivity (ε′), dielectric loss (ε″), AC conductivity (σAC), and impedance (Z) were calculated from the measured capacitance and loss tangent data at room temperature in the frequency range 500 Hz to 1 MHz for 0, 3, 5, 7, and 10 wt% ZnO-PC nanocomposites. The obtained results are presented graphically in Figures 2–5. It is clear that relative permittivity, dielectric loss, and AC conductivity are incased by increasing the concentration of ZnO nanoparticles in the PC host while impedance is decreased. On the other hand, the decrease of relative permittivity and dielectric loss with frequency could be attributed to the insufficient time for dipoles to align before the field changes direction [18–19]. The variations of relative permittivity, dielectric loss, AC conductivity, and impedance with ZnO concentration could be attributed to the ions diffusions and orientational polarizations in addition to the contribution of Maxwell-Wagner polarization [6,18–20]. However, it is very clear that ZnO nanoparticles will increase the polar character of the PC host.
Relative permittivity as a function of frequency and ZnO concentration for ZnO-PC nanocomposites. (Experimental data (symbol curves) and theoretical data (dark-solid curves)). Dielectric loss as a function of frequency and ZnO concentration for ZnO-PC nanocomposites. AC conductivity as a function of frequency and ZnO concentration for ZnO-PC nanocomposites. Impedance as a function of frequency and ZnO concentration for ZnO-PC nanocomposites.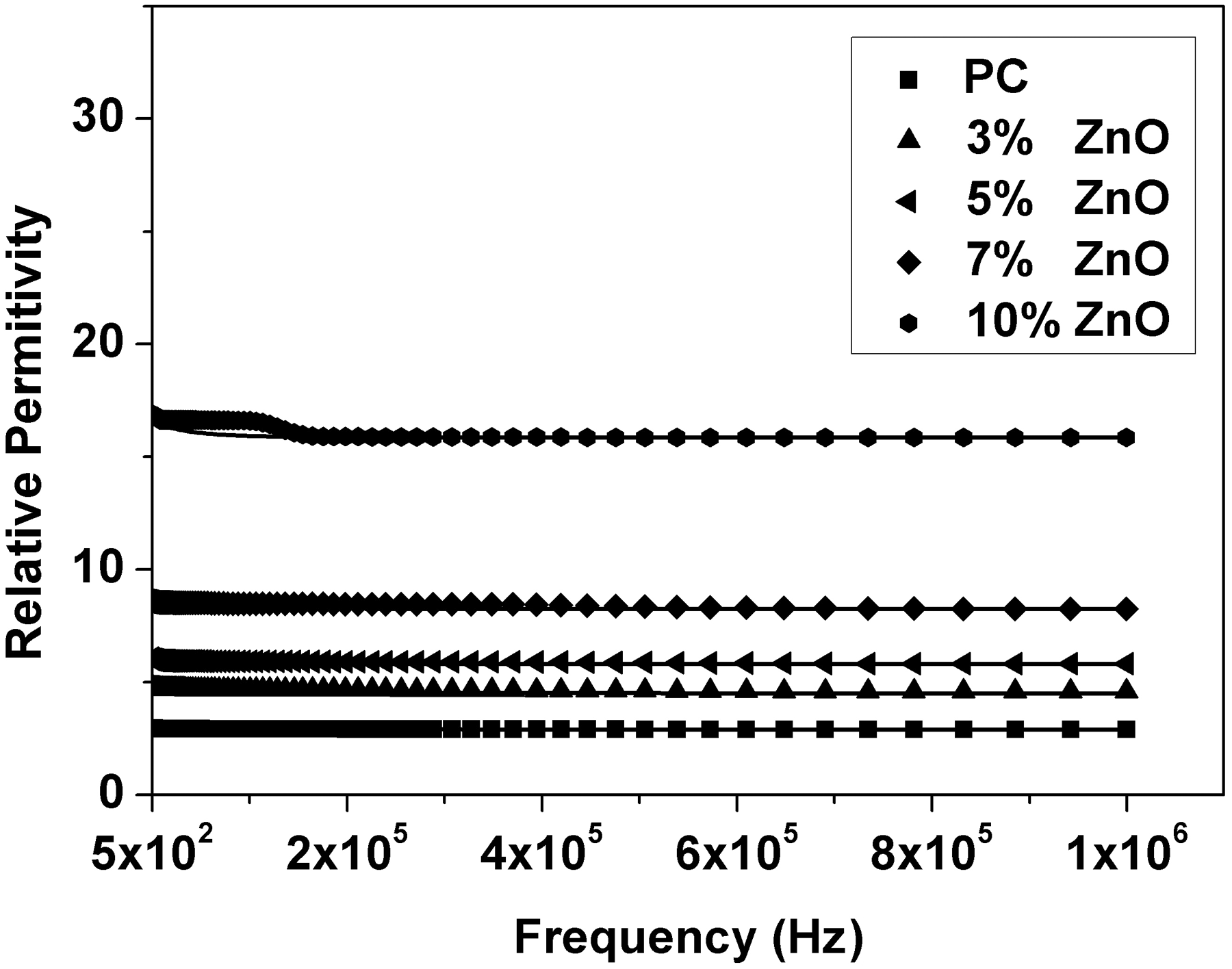
In our previous research [4], Yan and Rhodes model was used to estimate the values of relaxation time for PC/MnCl2 composites at room temperature. Two values of relaxation time were found for PC/MnCl2 composites at room temperature. In the present work, this model was also used to estimate relaxation time values at room temperature for ZnO-PC nanocomposites.
According to the Yan and Rhodes model, relative permittivity (ε′) is given as: [4]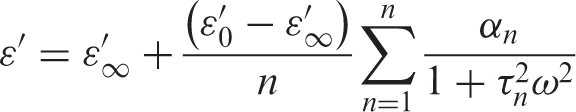
where ∑nn=1 αn = n and
Relaxation time values, glass transition temperature, and the weight loss ratio for ZnO-PC nanocomposites.
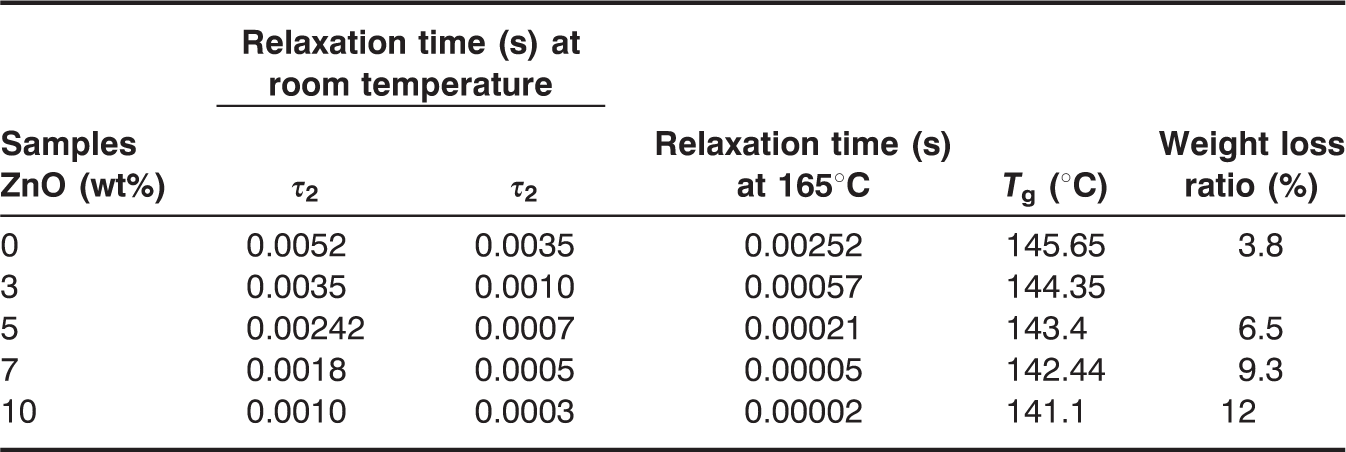
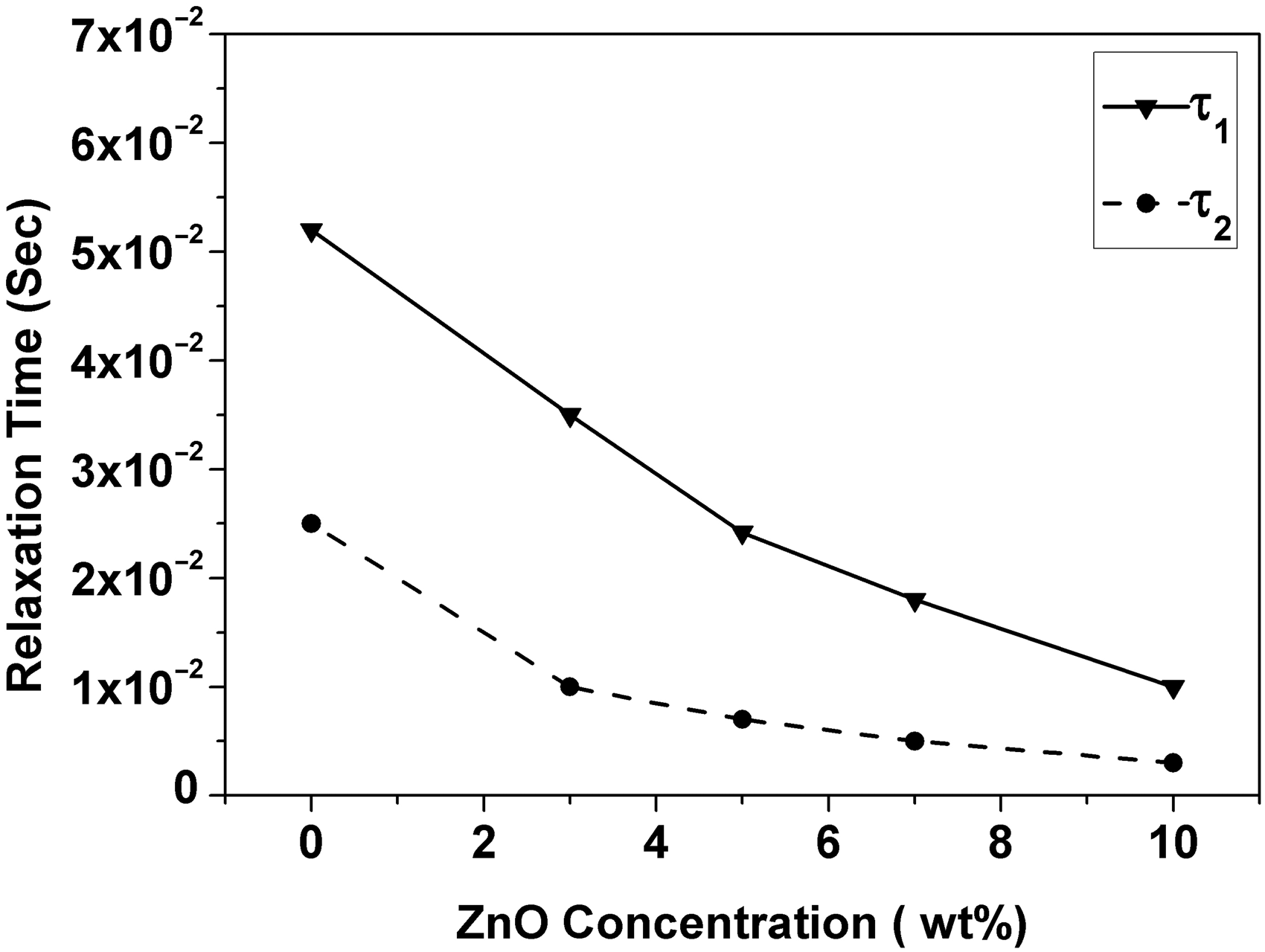
Relaxation time values vs ZnO concentration for ZnO-PC nanocomposites.
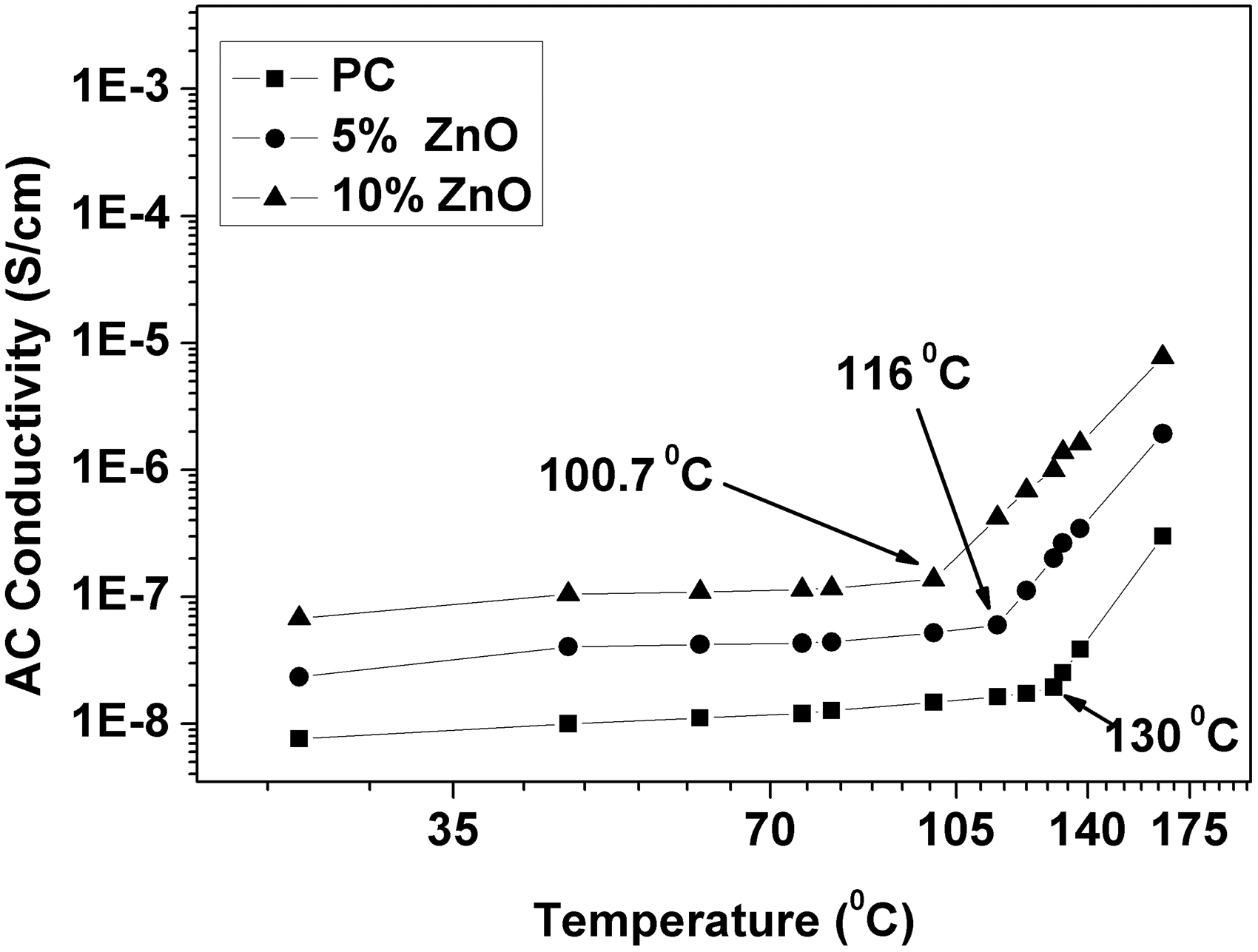
Figure 7 shows the dependence of AC conductivity with temperature at 1 kHz. It is very clear that AC conductivity is starting to jump to the higher values at 130°C (near the glass transition temperature of PC) which is already expected due to the increase in the chain mobility at this stage. Furthermore, addition of 10% ZnO nanoparticles to PC host will shift this jump toward lower temperature values (100.7°C). Generally, these variations could be attributed to the existence of two competitive mechanisms which might be responsible for these changes [18]: segmental mobility of polymer chains and thermal expansion of PC host and ZnO disrupting the chains of contacting nanoclusters. AC conductivity vs temperature at 1 kHz for ZnO-PC nanocomposites.
However, to realize effect of ZnO nanoparticles in the dielectric relaxation behavior of PC host, the dielectric parameters of ZnO-PC nanocomposites were measured at 165°C (above the glass transition temperature of PC) Figures 8–10 show AC conductivity, relative permittivity and dielectric loss of ZnO-PC nanocomposites at 165°C respectively Generally, increasing of temperature up to 165°C and also increasing ZnO nanoparticles content will increase AC conductivity, relative permittivity, and dielectric loss. These variations, however, could be attributed to the same reasons mentioned above.
AC conductivity vs v frequency at 165°C for ZnO-PC nanocomposites. Relative permittivity vs frequency at 165°C for ZnO-PC nanocomposites. Dielectric loss vs frequency at 165°C for ZnO-PC nanocomposites. Solid curves represent Debye fittings.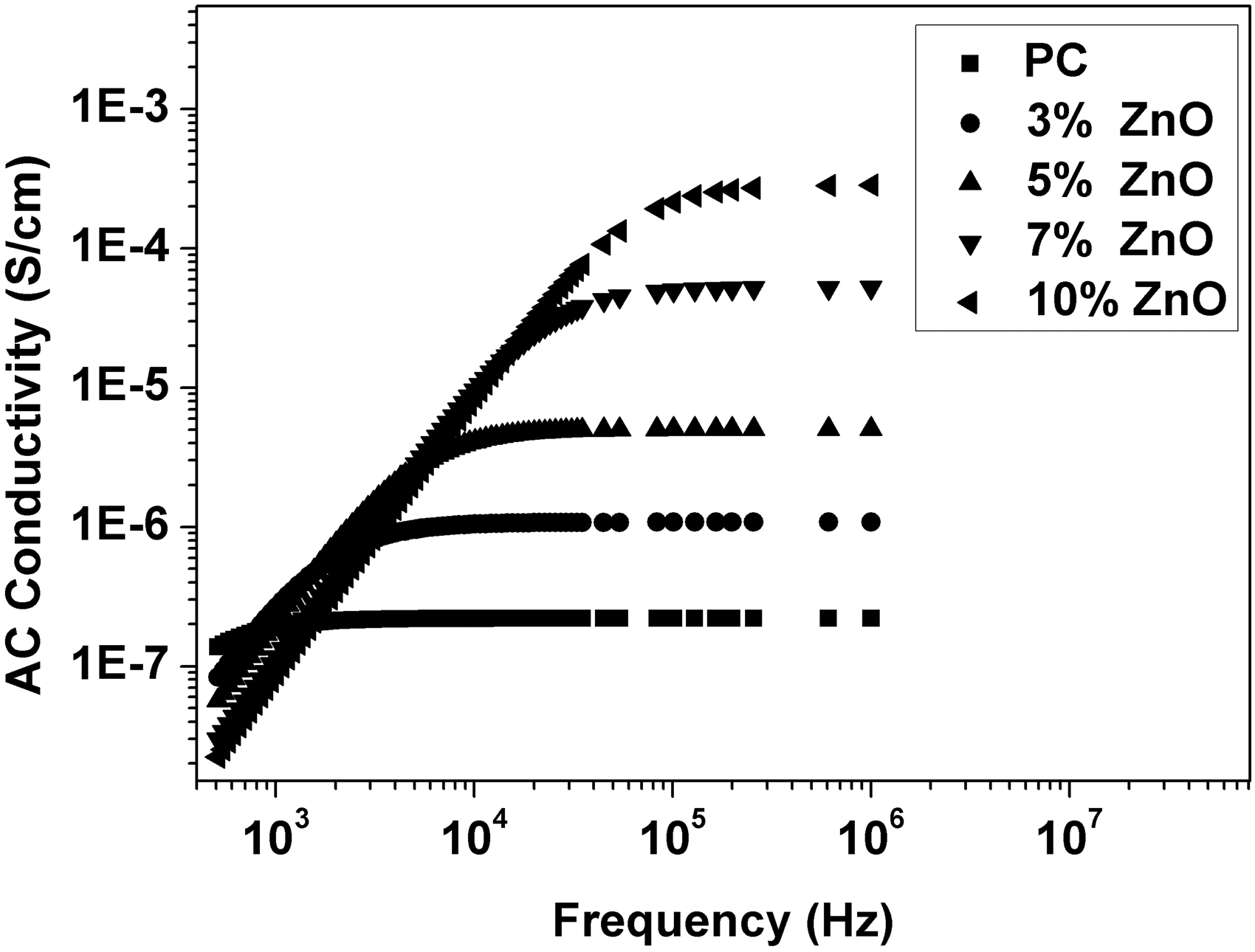
Campbell et al. [21] found that an isothermally single relaxation peak (α-relaxation) appears in the dielectric loss-frequency curve for PC at around 160°C. They reported that as the temperature is increased, the frequency of the peak maximum (fmax) is also increased, due to the polymer chains attaining greater mobility. Also they found that the relation between the ln(fmax) and temperature (T) is a linear relation. However, this relaxation can be also seen in the dielectric loss versus frequency curves of ZnO-PC nanocomposites at 165°C as shown in Figure 10. Additionally dielectric loss experimental data was fitted using Debye equation [22] to estimate the values of relaxation time. Debye equation is given as: [21,22]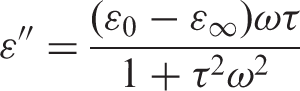
The obtained results from these fittings are presented graphically with those experimental data in Figure 10 (solid lines) while the obtained relaxation time values from the best fitting are listed in Table 1. From Figure 10, it is very clear that all ZnO-PC nanocomposites show single relaxation (α-relaxation) at 165°C in the domain frequency range. Furthermore, addition of ZnO nanoparticles to PC host will isothermally shift the frequency of the peak maximum toward higher frequency values and reduce the relaxation time value of PC host. Besides, the relation between ln(fmax) and ZnO concentration is linear relation as shown in Figure 11. It is very clear that the isothermal effect of ZnO nano particles on the α-relaxation of PC is similar to the temperature effect [21]. Both of them have a linear dependency with ln(fmax). This strongly indicates that ZnO nanoparticles highly enhances chain mobility of the polymer host.
Ln(fmax) vs ZnO concentration at 165°C for ZnO-PC nanocomposites.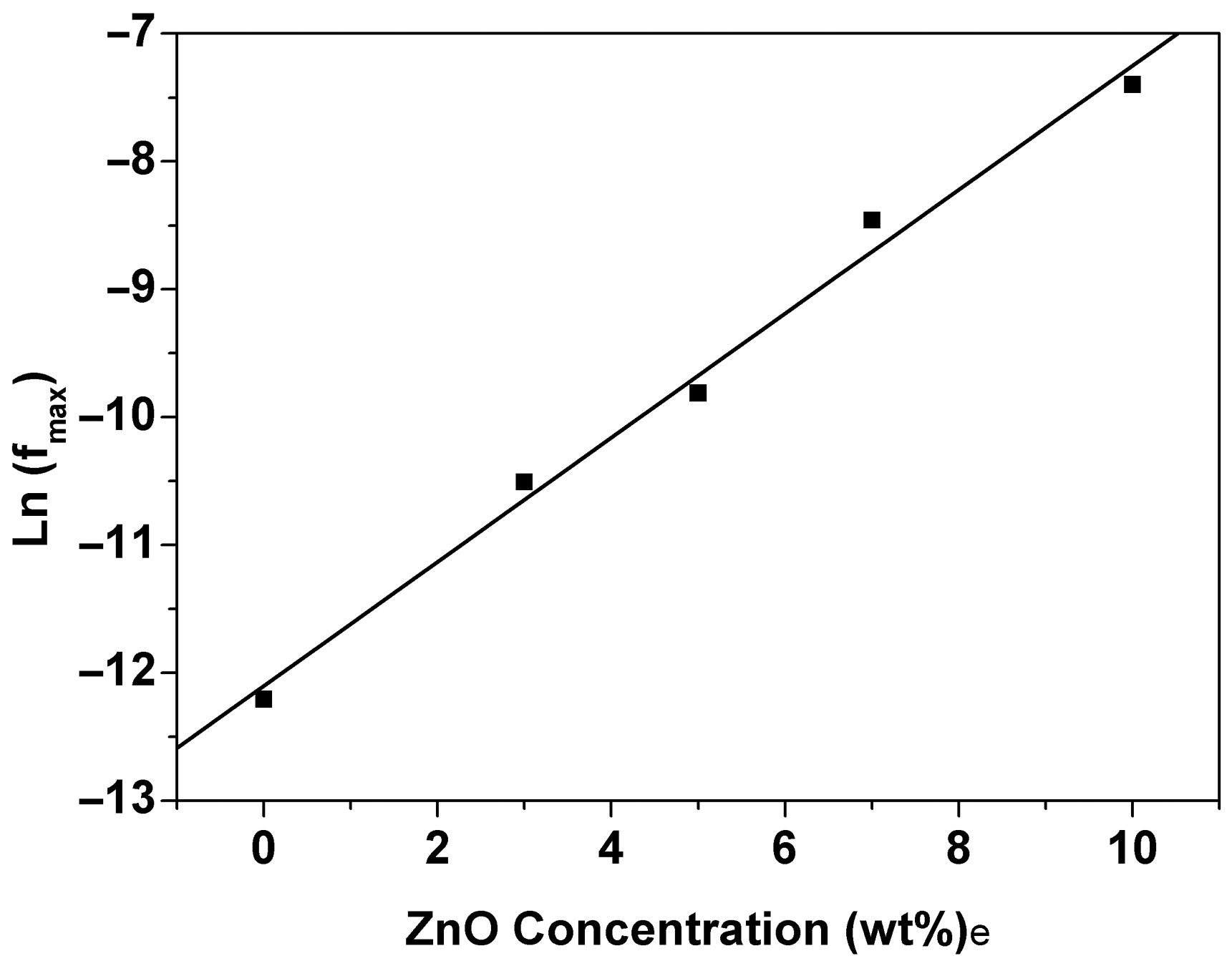
Thermal Analysis
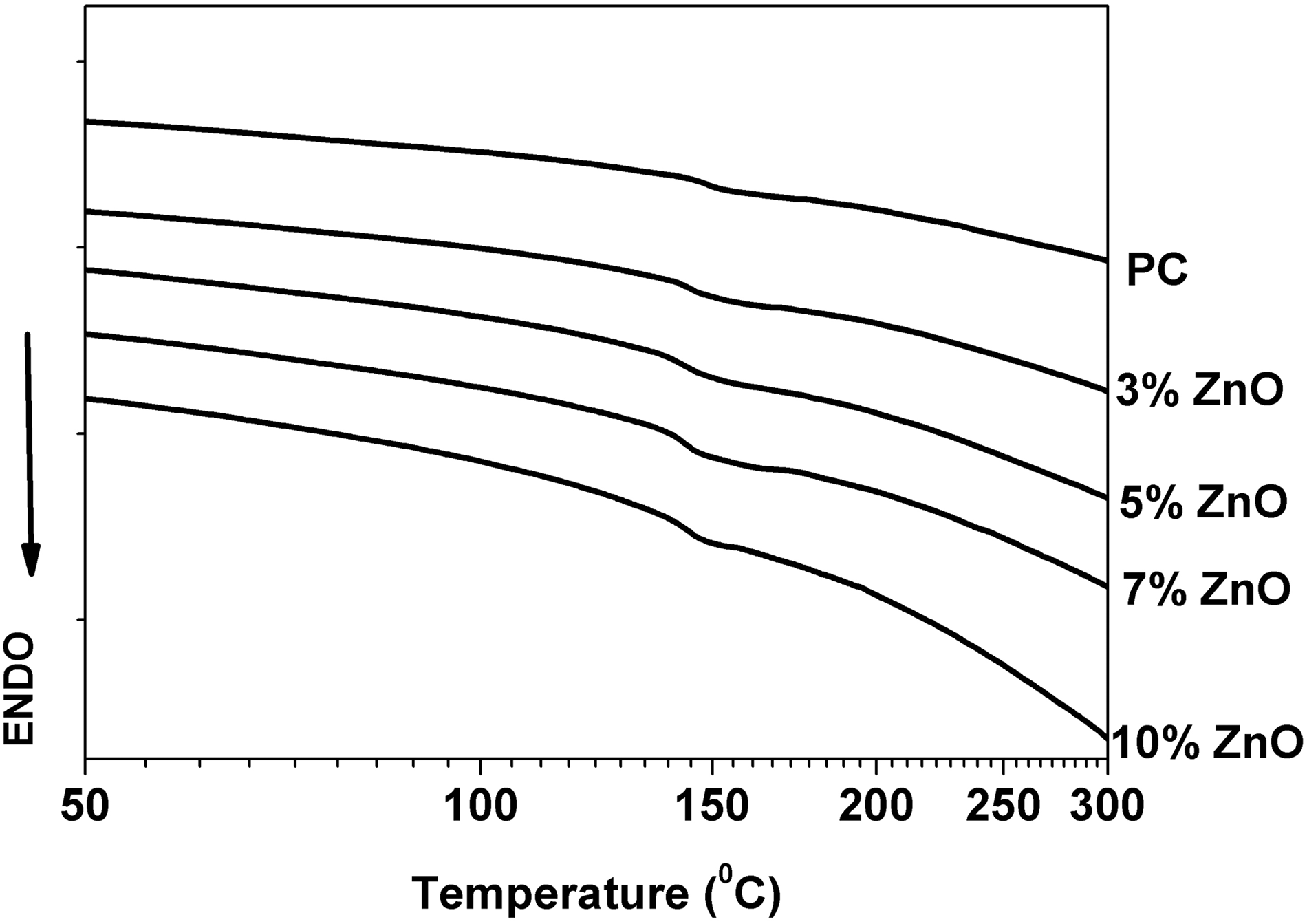
Glass transition temperate (Tg) values were determined for all ZnO-PC nanocompsites from the DSC curve as shown in Figure 12 and the obtained Tg values are listed in Table 1. Figure 13 shows the DSC curves of ZnO-PC nanocomposites. DSC results reveal that addition of ZnO nanoparticles to the PC host will decrease the Tg value from 145.65°C (for PC) up to 141.1°C (for10% ZnO-PC).
DSC thermogram ofr polycarbonate (PC). DSC curves of ZnO-PC nanocomposites.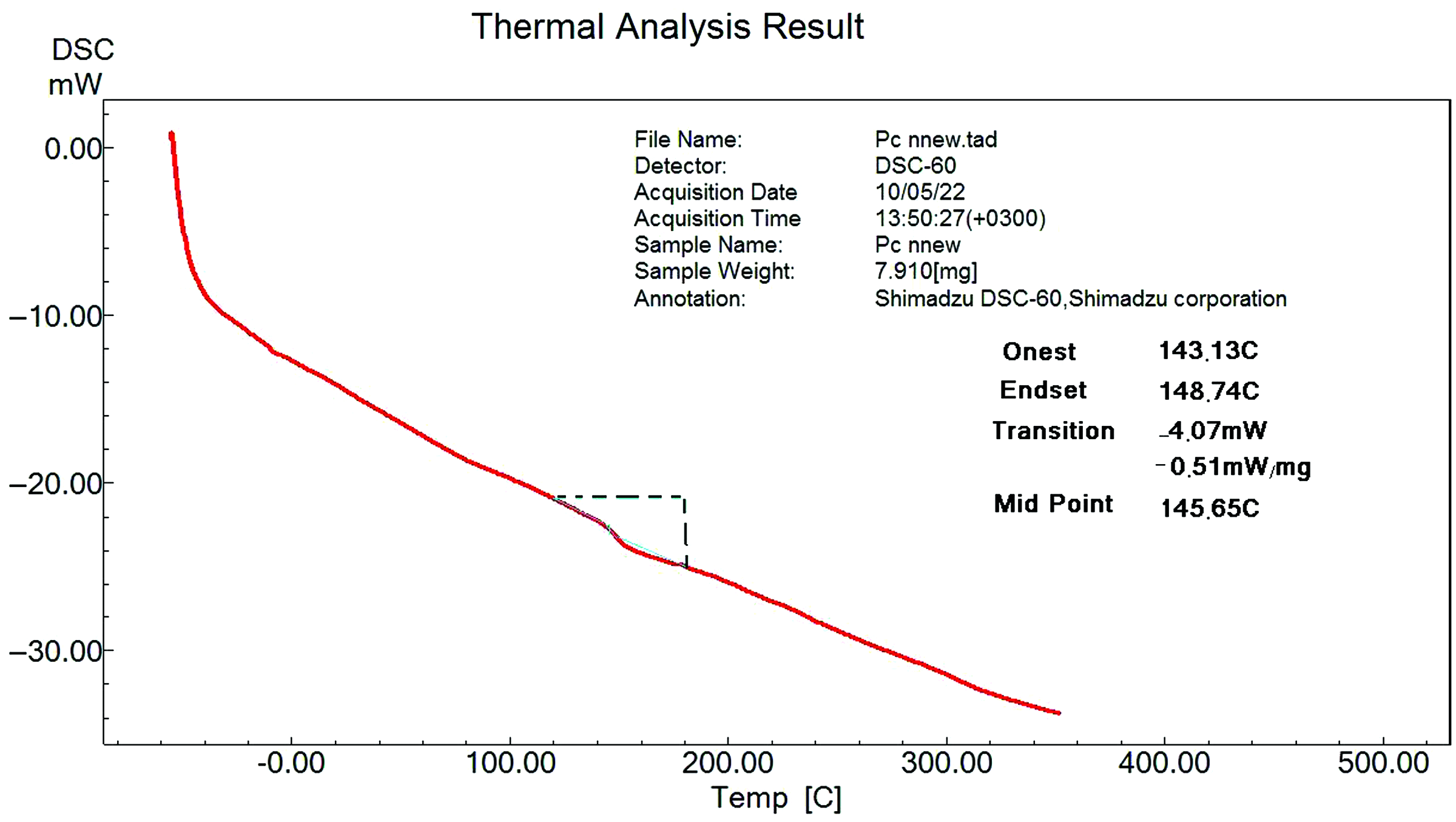
The results obtained from TGA show that the weight loss ratio increases with increasing of ZnO nanoparticles content (see Figure 14 and Table 1). Besides, Carrion et al. [3] also found that addition of ZnO nanoparticles up to 5 wt% to the PC host will decrease Tg value of PC. Also, they indicated that the thermal stability of polymer/ZnO nanocomposites depend on the base polymer, the nanoparticle morphology, and the processing conditions. However, the above results indicate that addition of ZnO nanoparticles to PC host will enhance the chain mobility and decrease the thermal stability of PC host. This confusion, however, is in a good agreement with those results obtained from dielectric relaxation.
TGA curves of ZnO-PC nanocomposites.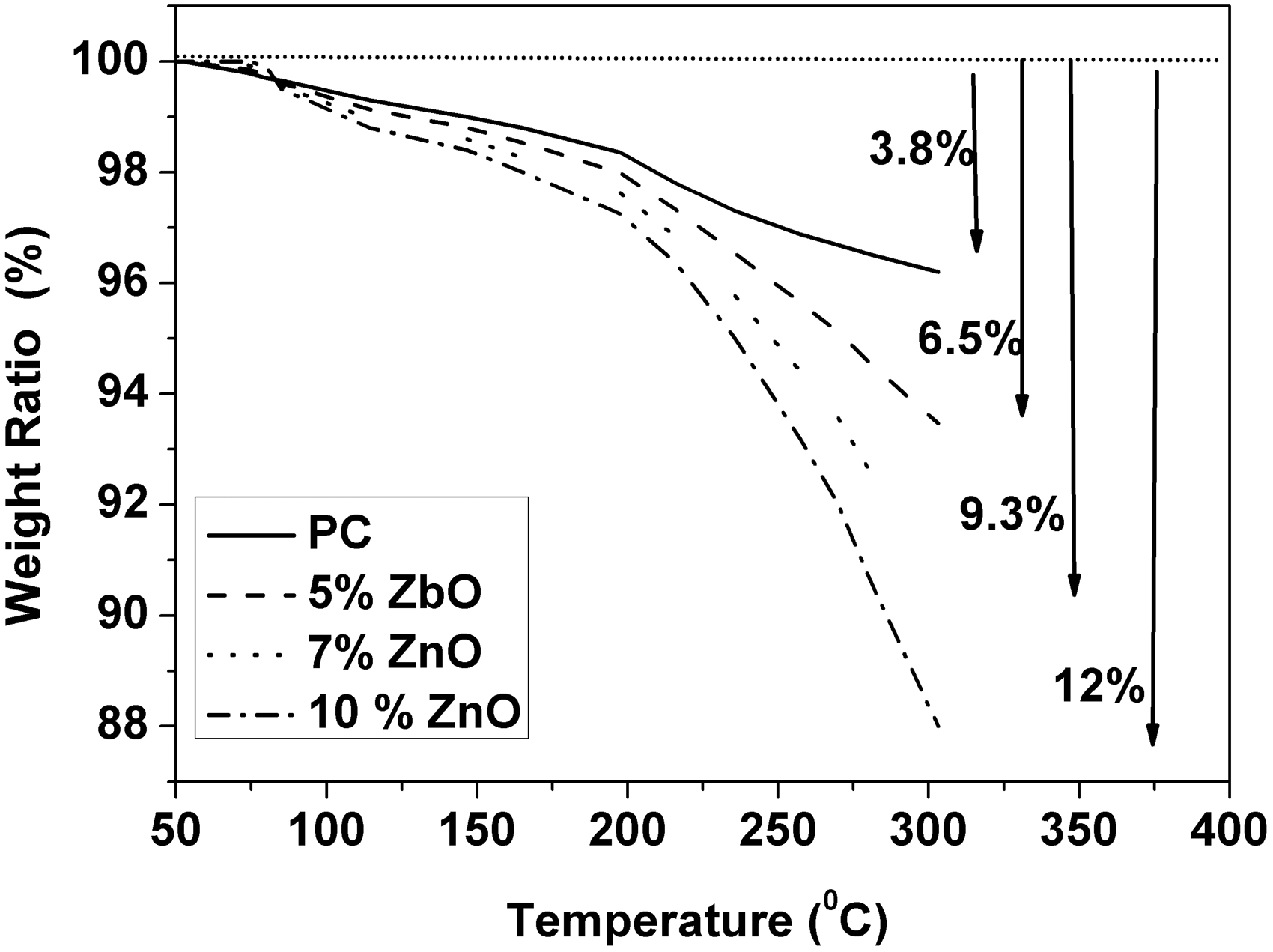
CONCLUSIONS
ZnO-PC nanocomposite films were prepared at different ZnO weight ratios 0, 3, 5, 7, and 10 wt% to investigate effect of ZnO nanoparticles on the dielectric relaxation behavior and thermal stability of PC host. Relative permittivity, dielectric loss, and AC conductivity increases with the increasing of ZnO content while impedance decreases with ZnO content in the frequency range 500 Hz to 1 MHz at room temperature. Also increasing the temperature up to 165°C will increase relative permittivity, dielectric loss, and AC conductivity at the domain frequency. Results obtained from the fittings of relative permittivity with Yan and Rhodes model reveal that the dielectric relaxation is not a single relaxation process and there are values of relaxation time for each nanocomposite at room temperature and domain frequency range. Additionally, dielectric loss data shows a single relaxation peak (α-relaxation) at 165°C in the domain frequency range. Furthermore, as the content of ZnO nanoparticles is increased in the PC host, the frequency of the peak maximum (fmax) shifts toward higher frequency value and as a result decreases the value of relaxation time. Also, this study shows that the isothermal effect of ZnO nano particles on the α-relaxation of PC is similar to the effect of temperature. Both of them have a linear dependency with ln(fmax). Addition of Zno nanoparticles to PC host decreases the thermal stability of PC host. Finally, there is a strong evidence from the obtained results such as dielectric relaxation, dielectric parameters, and thermal stability that ZnO nanoparticles highly enhance the chain mobility of PC host and increase the polar character of PC host.
Footnotes
ACKNOWLEDGMENTS
We would like to thank Mr. E.F. Barqawi (Physics department, College of Science, KFU, KSA) for his technical help during all the stages of this work. Thanks for Dr Bassam Sewellh and all the member staff of the Chemistry Department at the University of Jordan for providing PC resin. Many thanks to Mr Ayman Gannam (Physics Department, College of Science, King Fahd University, KSA) for his technical help.
