Abstract
Objective
This systematic review aims to evaluate recent evidence on interventions for sleep disturbances in dementia, a major clinical concern, with limited pharmacological and non-pharmacological strategies examined in controlled trials.
Methods
We included eight RCTs (N = 666 participants; 2020 – January 2025) in people with dementia and sleep disturbances. Records were identified via MEDLINE, screened using PRISMA-guidelines, and non-randomized or non-intervention studies were excluded. Risk-of-bias was assessed with Cochrane RoB2, and effect sizes (Cohen’s d) were calculated in R.
Results
Z-drugs indicated small-large benefits, with eszopiclone significantly improving sleep efficiency and latency. Orexin receptor antagonists indicated mixed evidence, with low-dose lemborexant (2.5 mg) most favorable, meeting benchmarks for nocturnal awakenings. Circadian modulation light therapy was associated with reduced nocturnal awakenings in small trials, while relaxation-based music therapy indicated little to no effect. Social stimulation with PARO indicated increased sleep time and large positive effects on sleep efficiency, despite missing clinical benchmarks.
Conclusions
Evidence is limited by sparse reporting and few trials per intervention. Z-drug eszopiclone, low-dose ORA lemborexant, and social stimulation PARO show the most favorable profile across outcomes and benchmarks, but pharmacological options require careful risk–benefit consideration. Non-pharmacological approaches appear safer but under-researched. Future trials should standardize outcomes and tailor strategies to patient needs. No external funding. The review was registered in INPLASY: https://doi.org/10.37766/inplasy2025.1.0097.
Keywords
Introduction
Background
Dementia significantly impairs independence and quality of life. 1 Adding to it, sleep disturbances – affecting approximately 19% of dementia patients – exacerbate cognitive impairment and caregiving challenges.2,3 Sleep supports cognitive function and brain health, particularly relevant in patients with dementia. 4 Alzheimer’s disease, the most common dementia subtype, involves pathological accumulations of amyloid-β and tau proteins. 5 The combination of poor sleep, which is associated with increased amyloid, and dementia appears to be a vicious cycle. 6 For this reason, adequate sleep interventions could plausibly influence progression and are thus clinically relevant. Treating sleep problems in dementia addresses multiple disease dimensions.
Numerous pharmacological and non-pharmacological interventions exist for sleep disturbances. In the following passage, the relevant interventions for this review are introduced: The first class of medications discussed is Z-drugs, a class of psychoactive drugs, which are widely used to treat insomnia. Their effects are mediated through the γ-aminobutyric acid-type A receptor, which is linked to sleep regulation. 7 Z-drugs have shorter half-lives than benzodiazepines, but still carry risks of daytime sedation, falls, cognitive deficits, and amnesia. 8
Orexin receptor antagonists (ORAs) are newer sleep medications that target the orexin system, discovered in 1998, inhibiting orexin receptors. 9 Common ORAs include suvorexant and lemborexant. 10 The 2023 European Insomnia Guidelines recommend ORAs for up to 3 months or longer when first-line cognitive-behavioral therapy proves insufficient. 11 Compared to benzodiazepines and z-drugs, ORAs may have a more favorable adverse-effect profile. 12 Recent reviews suggest that ORAs could influence Alzheimer’s pathology, potentially offering protective effects due to interactions with amyloid-β and tau. 13
Light therapy regulates sleep primarily by adjusting circadian rhythms through the suprachiasmatic nucleus, inhibiting melatonin secretion, and modulating alertness through thalamic and cortical activity. 14 Regarding adverse effects, it is considered safe. 15
Next, the precise mechanism of music therapy remains unclear, but involves the limbic system and modulation of endogenous opioids and oxytocin. 16 Music selection depends largely on individual preferences. A 2015 Cochrane Review analyzed the effects of music therapy on insomnia in adults, concluding that moderate-quality evidence supports its effectiveness in improving subjective sleep quality. 17 The review highlighted music therapy’s safety and ease of use. However, most of the included studies had a high risk of bias, and effectiveness was assessed primarily through subjective measures.
Activity therapy uses interactive robotic devices, such as the PARO (a responsive robotic seal; its name comes from the Japanese transliteration of “personal robot”, paasonaru robotto). It aims to reduce behavioral and psychological dementia symptoms through meaningful social interactions, although the definition of “social stimulation” varies widely and the therapy’s efficacy remains limited in evidence.18-20 Recent reviews of social stimulation with PARO in dementia noted moderate reductions in medication use, minor improvements in anxiety and agitation, but negligible effects on sleep duration, and evidence quality was generally low, effectiveness varied by patient interest, and cost-effectiveness was questioned due to significantly higher costs compared to usual care.21-23 For analytical consistency, non-pharmacological interventions were classified by their primary therapeutic mechanism: social stimulation (PARO), relaxation-based (music therapy), and circadian (rhythm) modulation (light therapy).
Current State of Research & Gaps in Knowledge
German guidelines currently lack consensus on sleep treatments in dementia, but mention melperone and pipamperone as options, although evidence is still limited. 24 A 2020 Cochrane review indicated low-certainty evidence for melatonin, ramelteon, and trazodone, with moderate-certainty evidence only for orexin receptor antagonists. 25 The authors found no RCT evidence supporting commonly prescribed drugs, such as antidepressants (eg, doxepin, mirtazapine), antipsychotics (eg, melperone, pipamperone), benzodiazepines, or antihistamines. 26 Recent specific studies remain scarce: the latest pipamperone study 27 dates to 1977, and a 2016 mirtazapine study in Alzheimer’s patients indicated no significant sleep improvement compared to placebo. 28 The Cochrane review found no RCTs on commonly used treatments, but included two melatonin studies in its meta-analysis, both showing no significant improvement and rated as low-certainty evidence. 25 One study found melatonin ineffective in Alzheimer’s, 29 consistent with other trials using different doses and release forms.30,31 A 2014 study reported benefits, but relied on subjective measures like PSQI and sleep diaries. 32
In contrast, a Cochrane review of non-pharmacological interventions (published in 2023, evidence up to January 2022) included 19 RCTs (n = 1335) and concluded with low certainty that physical and social activities, carer-delivered interventions, and multimodal programs may produce small to modest improvements in some sleep outcomes. 33 Meanwhile, the evidence for light therapy, daytime sleep restrictions, slow-stroke back massage, and transcranial electrostimulation was of Low to very low certainty and suggested little to no benefit or remained inconclusive.
Objectives
This systematic review aims to synthesize recent interventions and evaluate their impact on sleep disturbances in people with dementia, specifically focusing on changes in sleep parameters, such as total nocturnal sleep time, sleep efficiency, sleep latency, and nocturnal awakenings, compared to prespecified clinical benchmarks for older adults. Earlier evidence is summarized in existing Cochrane reviews.
Research Question
How effective are recent pharmacological and non-pharmacological interventions for sleep disturbances, published since 2020, in improving sleep outcomes relative to the prespecified clinical benchmarks for older adults, and what implications do these findings have for future treatment guidelines?
Materials & Methods
Eligibility Criteria
This review was informed by the eligibility framework of the previously mentioned Cochrane Review of pharmacological interventions (2020). 25 We applied a uniform set of inclusion criteria across all interventions, including randomized, placebo-controlled, and cross-over trials, excluding quasi-randomized ones.
Eligible studies included adults with dementia (any subtype; diagnosed according to DSM/ICD criteria) with sleep problems identified subjectively or objectively. At least 80% of the participants in each study were required to have dementia. Studies focusing on respiratory conditions, such as obstructive sleep apnea syndrome, were excluded.
The sleep outcomes in included studies were measured using objective tools, such as polysomnography or actigraphy.
Importantly, we did not replicate the Cochrane review. We used its criteria as a reference and applied a more inclusive approach of eligibility (more in the Discussion), while focusing only on the sleep related outcomes. We restricted inclusion to studies published from 2020 onwards to provide a recent, integrated synthesis of randomized evidence across pharmacological and non-pharmacological interventions. Evidence before this timeframe has been summarized in the eligibility criteria. Although the 2023 Cochrane Review of non-pharmacological interventions includes trials up to January 2022, we retained the 2020-2021 period to maintain a uniform timeframe across intervention categories and to enable direct, within-review comparisons across intervention classes.
The electronic search was restricted to a single database MEDLINE, selected as the primary biomedical database for this topic. In addition, the review followed PRISMA 2020 guidelines. However, we did not conduct a sensitivity analysis, as we did not plan on performing a meta-analysis.
Search Methods
Studies were identified through a systematic MEDLINE search (2020 – January 2025), using terms covering general therapy keywords (treatment, medication, pharmacotherapy) and a wide range of specific drug names, including antipsychotics, antidepressants, benzodiazepines, stimulants, cannabinoids, anticonvulsants, orexin receptor antagonists, and antihistamines, to capture a broad spectrum of pharmacological options. Non-pharmacological therapies, such as light therapy, phototherapy, neurostimulation, transcranial magnetic stimulation (TMS), cognitive behavioral therapy (CBT), biofeedback, music therapy, and aromatherapy, were also included. The terms were combined with phrases for sleep issues (sleep disturbances, insomnia) and dementia-related conditions (Alzheimer’s, cognitive impairment). To maintain scope, we excluded terms targeting distinct sleep disorders (restless-legs-syndrome, sleep apnea) and caregiver-focused outcomes. We did not include REM-specific terms in the search strategy because the review’s predefined outcome framework prioritized sleep metrics typically derived from actigraphy (eg, sleep efficiency, WASO), consistent with outcome prioritization in the relevant Cochrane review, and actigraphy does not provide REM sleep staging. 34 The primary systematic search was completed in January 2025. Following peer review, a targeted, post-hoc search for REM-sleep-related outcomes was additionally performed in December 2025.
Search terms are documented in the INPLASY registration https://doi.org/10.37766/inplasy2025.1.0097.
Data Collection Process & Study Selection
MEDLINE searches were run independently by two reviewers. The initial search took place from May 1-14, 2024, followed by an update on January 13, 2025, using the same strategy and an expanded list of terms. Duplicates, pre-2020 studies, and non-RCTs were excluded. Remaining studies were assessed using predefined criteria. Outcome data extraction was conducted by one reviewer and subsequently checked by a second reviewer for completeness and accuracy.
Risk of Bias Assessment
Risk of bias was assessed using RoB 2. 35 Data extraction and entry for the risk of bias assessment was conducted by one reviewer. Any uncertainties were discussed with a second reviewer. This single-reviewer risk-of-bias assessment is considered a methodological limitation of this review. Otherwise, the assessment is in accordance with RoB 2 guidance to ensure consistency.
Primary Outcomes
The primary sleep parameters analyzed included total nocturnal sleep time, sleep efficiency, time awake after sleep onset (WASO), number of nocturnal awakenings, and sleep latency.
Summary Measures and Synthesis of Results
For this review, a comparative, analytical approach was used to synthesize the findings of various studies. Effect sizes (Cohen’s d) were calculated using the RStudio meta package for each outcome. Forest plots were used to visualize effect sizes and standard deviations, enabling direct comparisons across studies. This graphical representation serves as a summative representation of the individual study results.
Results
Study Selection
The MEDLINE searches identified a total of 2212 records. After removing non-RCTs, duplicate records, and studies published before 2020, 47 reports were included for eligibility evaluation. Studies were excluded if outcomes were based solely on self-report questionnaires, if full text was unavailable, or if the topic was not relevant to the review question. Upon re-examination of the study design, one study was found to use non-random allocation and was therefore reclassified as non-randomized. It is presented descriptively only and was excluded from RCT synthesis. 36 synthesis. In total, eight studies that met all the criteria were included in the final analysis.
The process of selecting the studies is summarized in Figure 1. PRISMA flowchart 2.0.
37
summarizing the systematic review process. After identifying a total of 2212 records through MEDLINE, exclusions were made based on publication date, study design, and relevance. Following eligibility assessment, 8 studies were included in the review, with exclusion reasons provided. During reevaluation, one study was reclassified as non-randomized due to non-random allocation and was excluded from the RCT synthesis.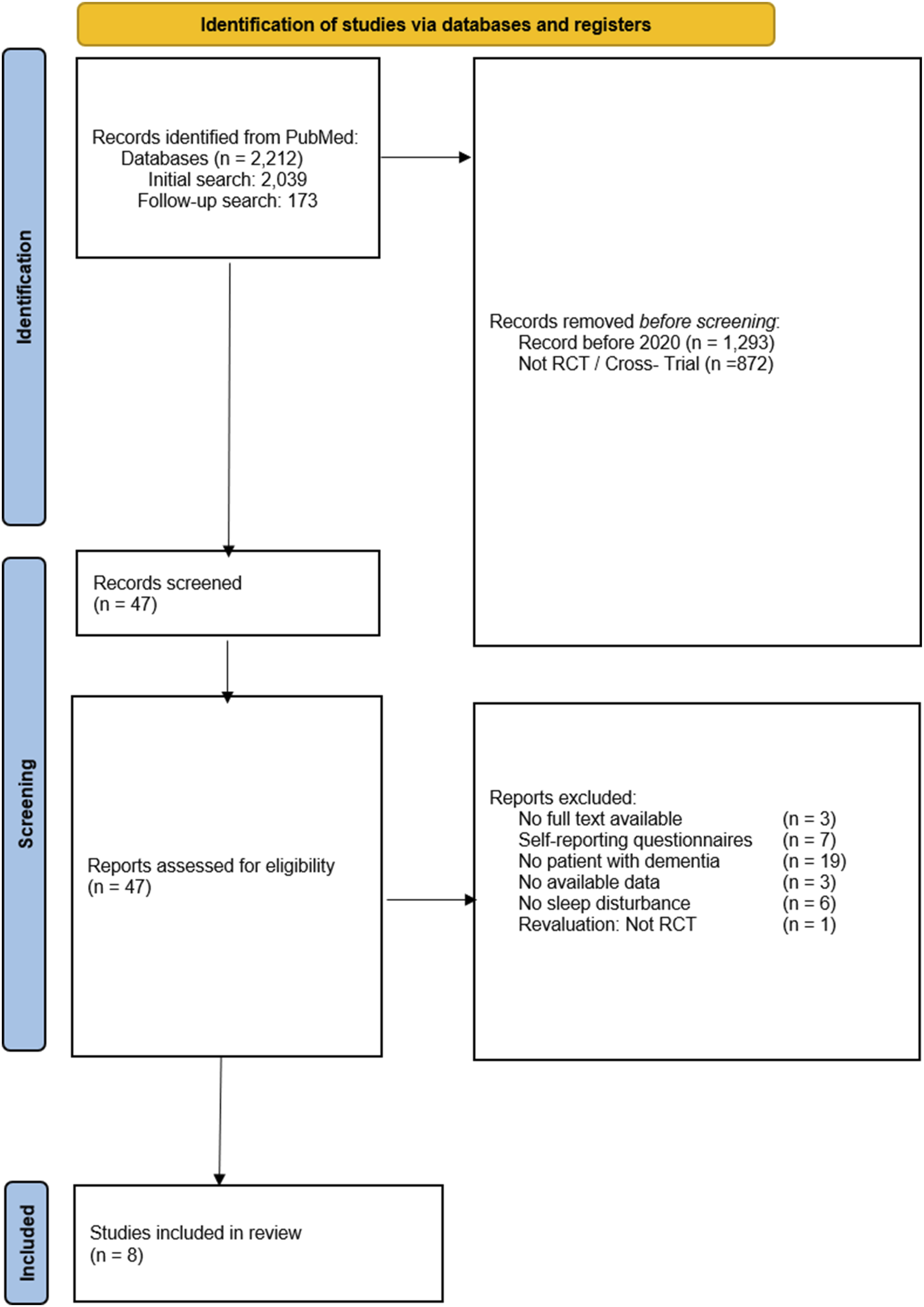
Descriptive Overview and Risk of Bias Within Studies
Summary of Intervention Studies, including Their Settings, Intervention and Control Group Sizes, Descriptions, Durations, and Key Observations.
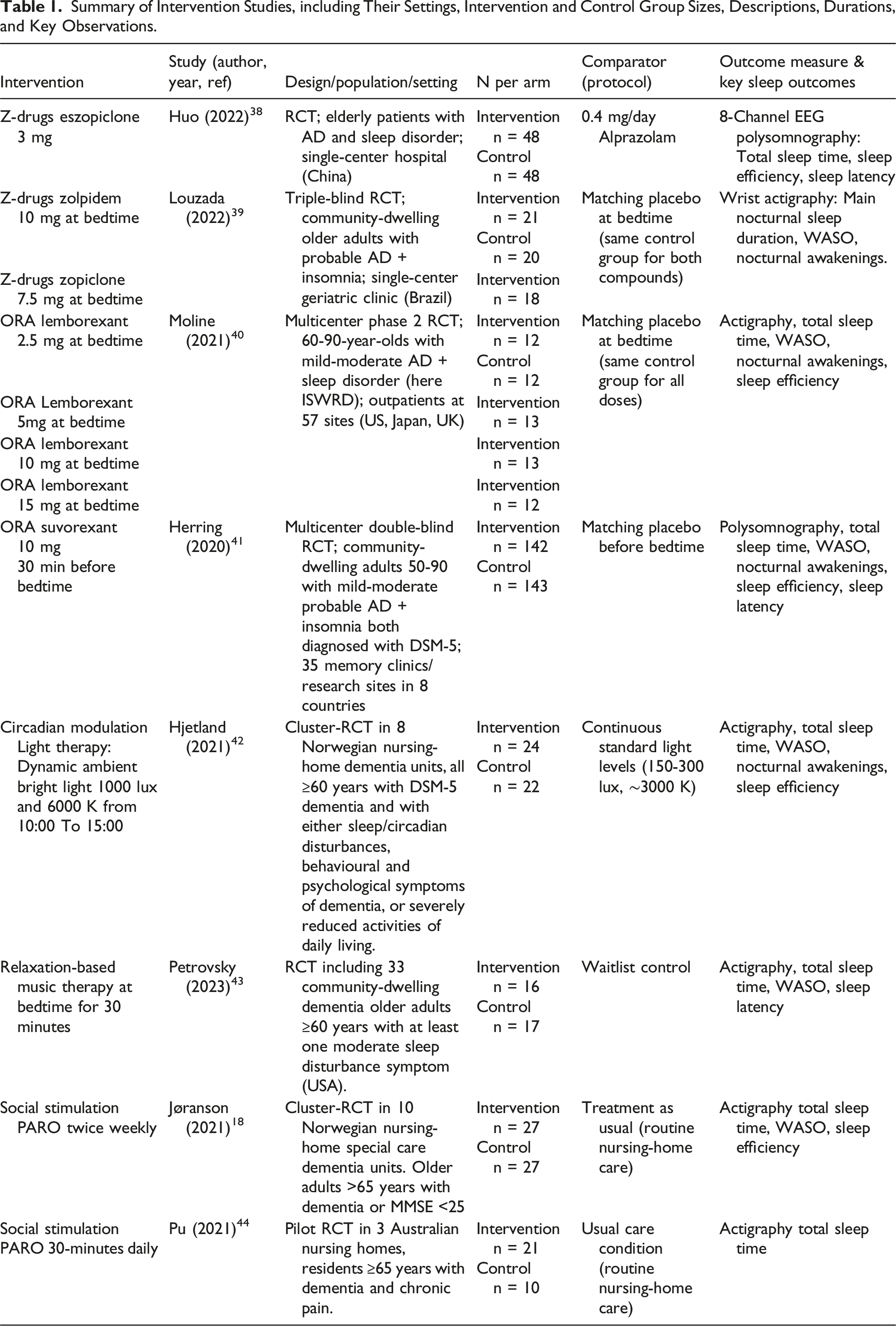
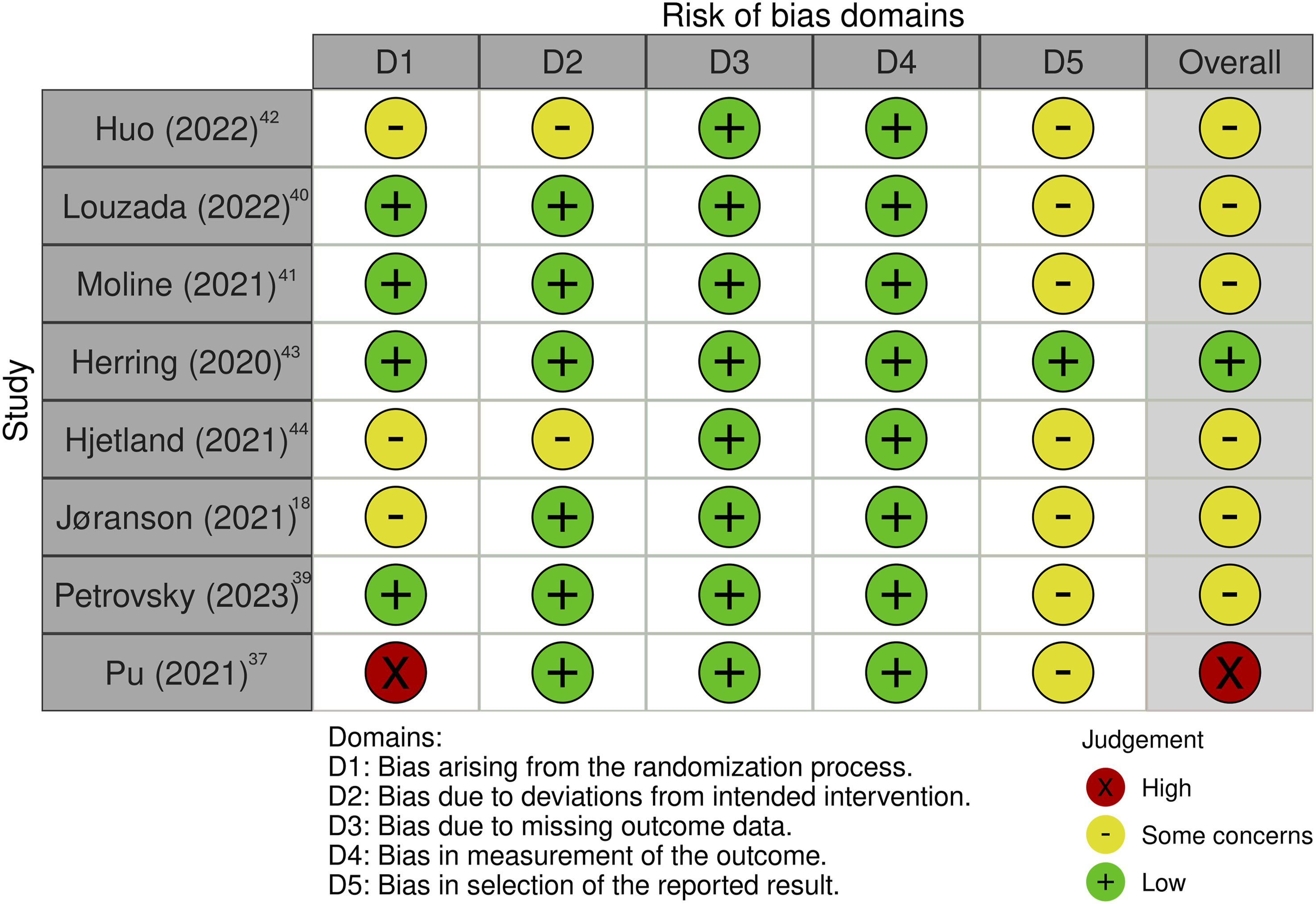
Against this descriptive background, we next summarize risk-of-bias assessments within studies. The risk of bias assessment evaluates studies in five key domains. In this review, most studies suggest a low risk of bias in domains related to deviations from intended interventions (D2), missing outcome data (D3), and outcome measurement (D4). However, concerns were frequently noted in D1 (bias of the randomization process). Similarly, in D5 (selection of the reported results), Pu et al (2021) did not preregister outcomes, raising concerns about selective reporting; most studies were flagged as having “some concerns” in these areas and overall deemed to have a high risk. 44
Results
The comparative analysis below evaluates pharmacological and non-pharmacological interventions against clinical benchmarks. Since not all studies report on every parameter, data availability varies. Certain outcomes are not discussed in detail due to limited data (therefore, no analysis of effect size was possible) or limited interpretability. However, they remain accessible for reference as forest plots in the supplemental material.
Total Nocturnal Sleep Time
Figure 3 illustrates total nocturnal sleep time, the most consistently reported metric. The National Sleep Foundation recommends a minimum of 420 minutes of sleep for older adults.
46
Three out of 12 intervention arms (across eight studies) — relaxation-based music therapy,
43
z-drug zopiclone
39
and 30 minutes of daily social stimulation with PARO
44
— met or exceeded this threshold. All remaining interventions, including the multiple ORA lemborexant doses,
40
fell below the benchmark. Forest plot illustrates the standardized mean differences (SMD) and 95% confidence intervals (CIs) for interventions targeting total nocturnal sleep time. Positive SMD values favor the intervention, while values overlapping zero indicate no significant effect. The effect sizes for the interventions that met the recommendation ranged from around small (SMD = 0.17) to large (SMD = 1.57). The variability in effect sizes is notable across interventions. For example, the study Jøranson et al (2021)
18
(activity therapy PARO) indicate large improvements (SMD >0.8), but these findings were accompanied by wide confidence intervals. Wide confidence intervals, as observed in some studies, suggest high inter-individual variability or small sample size.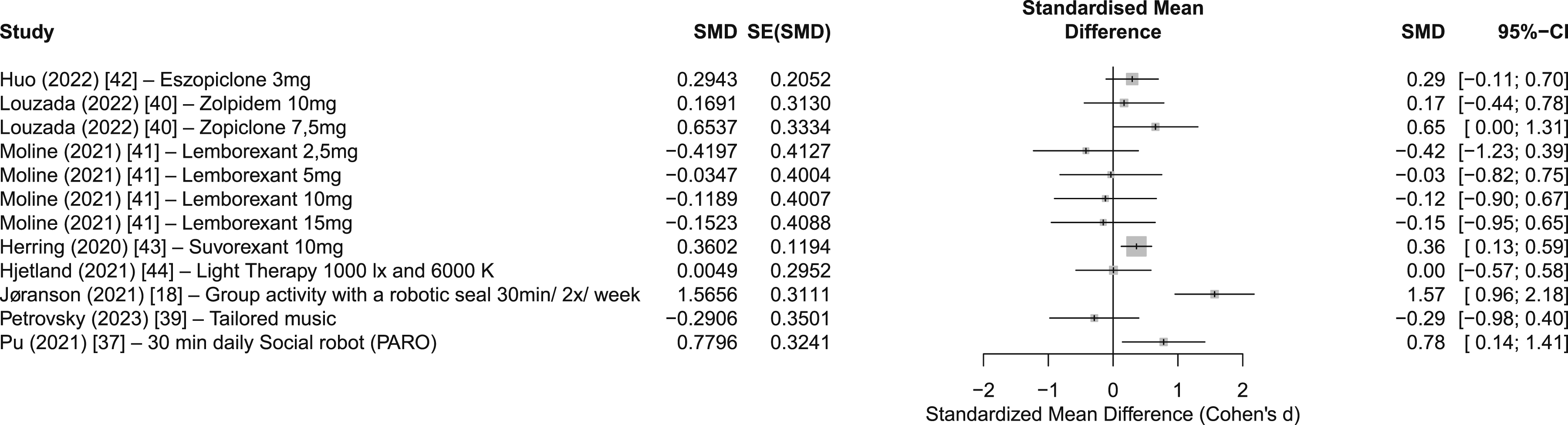
To better understand how individual interventions compare, effect sizes are examined next. The 30-minute daily social stimulation with PARO intervention 44 indicated a moderate effect (SMD = 0.78, 95% CI 0.14 to 1.41, N = 31). Meanwhile, the twice-weekly intervention 18 indicated a large effect (SMD = 1.57, 95% CI 0.96 to 2.18, N = 54), despite not reaching the recommended duration of 420 min (334.90 min reported). Z-drug zopiclone, 39 7.50 mg, met the clinical benchmark with 450 min and indicated a moderate, not-significant effect (CI crossed 0). For total sleep time, small, non-significant effects were observed for Z-drug eszopiclone, 38 ORA lemborexant 40 2.50 mg, and relaxation-based music therapy. 43 In contrast, small, statistically significant effects were observed for ORA suvorexant. 41
In summary, three intervention arms met or exceeded the ≥420-minute benchmark. The twice-weekly social stimulation with PARO schedule 18 indicated the largest significant effect size, but remained clinically subthreshold, while the 30 minutes daily 44 met the benchmark with moderate effect size. ORA suvorexant 41 indicated statistically significant effects, although it did remain clinically subthreshold.
Sleep Efficiency
Beyond sleep duration, sleep efficiency provides a complementary indicator of sleep quality. The National Sleep Foundation defines sleep efficiency as optimal at ≥85% and poor at ≤74%. 46 Six intervention arms exceeded the lower threshold: ORA lemborexant at all doses 40 and social stimulation with PARO administered twice weekly 18 with 79.63%, including Z-drug eszopiclone, which exceeded optimal efficiency at 87.93%. 38 In contrast, two intervention arms fell below clinical benchmark of 74%: ORA suvorexant 41 (72.80%) and circadian modulation light therapy 42 (71.82%) at 1000 lux. Sleep efficiency data were not reported for the remaining four intervention arms.
Both z-drug eszopiclone 38 and the social stimulation with PARO intervention 18 were associated with large effect sizes (SMD = 0.94, 95% CI 0.52 to 1.36, N = 96, and SMD = 1.54, 95% CI 0.93 to 2.14, N = 54, respectively). ORA lemborexant, 40 2.50 mg, indicated a moderate, non-significant effect. ORA suvorexant 41 indicated small significant effect size (SMD = 0.35, 95% CI 0.12 to 0.59, N = 285), while ORA lemborexant 40 5 mg was associated with small non-significant effect.
Taken together, these findings suggest that three interventions improved sleep efficiency significantly relative to control groups: ORA suvorexant 41 (small), z-drug eszopiclone 38 (large), and twice-weekly social stimulation with PARO 18 was associated with the largest improvements. Meanwhile, ORA suvorexant 41 did not reach the lower benchmark of 74%, and twice-weekly social stimulation with PARO 18 did not reach the optimal threshold of 85%. In contrast, z-drug eszopiclone 38 exceeded the optimal benchmark.
Wake-After-Sleep Onset & Nocturnal Awakenings
A reduction in nocturnal wakefulness (wake after sleep onset (WASO) and nocturnal awakenings) is another important dimension of sleep quality, and the included studies indicated considerable variability across interventions. For older adults, the recommended duration of wake-after-sleep-onset is ≤ 30 minutes, with longer durations considered uncertain. 46 Only ORA lemborexant 40 remained within the recommended range for WASO across all doses and was consistent with clinically appropriate nocturnal awakenings (≤(2) in the 2.50 mg and 5 mg groups. Higher doses and circadian modulation light therapy at 1000 lux 42 were associated with uncertain clinical benchmarks. Clinical benchmark attainment could not be assessed for three intervention arms for WASO and four intervention arms for nocturnal awakenings due to missing data. Otherwise, where benchmark data were reported, values often remained subthreshold.
ORA Lemborexant 40 2.50 mg and circadian modulation light therapy 42 at 1000 lux indicated large effects in reducing nocturnal awakenings (SMD = −0.85, 95% CI –1.68 to −0.01, N = 62, and SMD = −3.38, 95% CI –4.21 to −2.25, N = 31, respectively). Z-drug Zopiclone 39 and ORA lemborexant 40 5 mg both were consistent with moderate, not significant effects, while the 10 mg and 15 mg doses of ORA lemborexant 40 indicated small, not significant effects. Regarding WASO, social stimulation with PARO 18 administered twice weekly were consistent with a large, significant effect (SMD = −1.41, 95% CI -2.01 to −0.82, N = 54). Moderate but non-significant effects were observed, with estimates trending in both directions (towards the intervention in Z-drug zopiclone 39 and towards placebo in ORA lemborexant 40 5 mg and 15 mg) indicating statistical uncertainty. Meanwhile, Z-drug zolpidem 39 indicated a small, non-significant effect.
Taken together, the findings indicate that ORA lemborexant 40 (particularly 2.5 mg) and circadian modulation light therapy 42 were consistent with the strongest reductions in nocturnal awakenings. However, only ORA lemborexant 40 reached the clinical benchmark of ≤2, despite both indicating large and statistically significant effects. In contrast, no intervention achieved both a significant effect and the ≤30 minutes clinical benchmark for WASO, although the twice weekly social stimulation with PARO 18 was associated with the largest, statistically significant effect.
Sleep Latency
Lastly, sleep latency was assessed, given its clinical relevance as a marker for difficulty initiating sleep. However, unlike other outcomes, only three studies reported this measure, so the findings are interpreted cautiously. Sleep latency ≤30 min is recommended, while ≥60 minutes is insufficient. 46 Relaxation-based music therapy 43 reported latencies of 8.10 minutes (SD = 22.3) and Z-drug eszopiclone, of 21.09 minutes (SD = 6.4), both within the optimal range. However, relaxation-based music therapy 43 indicated only a negligible between-group effect, suggesting limited intervention-specific benefit (SMD = 0.08, 95% CI -0.60 to 0.76, N = 33). At the same time, baseline sleep latency already remained within the clinical threshold. ORA suvorexant 41 remained subthreshold, and the remaining nine intervention arms did not report the relevant data. For sleep latency, Z-drug eszopiclone 38 was associated with moderate effects, with SMD = −0.67, 95% CI -1.08 to −0.26, N = 96, while the other two reported studies indicated negligible effects.41,43
Taking everything together, the evidence suggests that, while the interventions vary in the aspects of sleep they target: Social stimulation PARO,18,44 z-drug eszopiclone, 38 and ORA lemborexant 40 (2.50 mg) appeared most favorable across several outcomes. However, the extent to which these statistical effects are translated into clinically meaningful thresholds varies by outcome and intervention.
Discussion
Data Analysis and Research Gaps
Pharmacological interventions such as Z-drugs, were associated with an increase in total sleep and improvements in efficiency/latency. However, statistical significance and reaching benchmarks varied across compounds and outcomes. Z-drug zopiclone 39 indicated moderate but not significant effects on sleep duration, nocturnal awakenings, and WASO. Z-drug eszopiclone 38 was associated with a large, statistically significant improvement in sleep efficiency and a moderate, statistically significant reduction in sleep latency. By contrast, z-drug zolpidem 39 indicated small, statistically non-significant effects and did not achieve clinical thresholds.
Orexin receptor antagonists were associated with improvements in sleep efficiency and reductions in nocturnal awakenings and nocturnal time awake, relative to clinical recommendations. However, statistical significance and clinical benchmark attainment varied by compound and dose. ORA Suvorexant 41 indicated small effects (some statistically significant), but failed to meet any clinical benchmarks. ORA Lemborexant 40 was associated with the greatest reduction in nocturnal awakenings among the pharmacological interventions (large and statistically significant at 2.50 mg). It also indicated a moderate, but statistically non-significant, gain in efficiency at low doses, with efficacy tapering off at higher doses. For WASO, ORA lemborexant 40 was associated with moderate, statistically non-significant effects favoring placebo, indicating no reliable improvement in this outcome.
Non-pharmacological approaches indicated mixed effectiveness. Circadian modulation light therapy, 1000 lux, 42 was associated with a large, significant effect on nocturnal awakenings, but uncertain clinical relevance. Evidence is limited to a single, small trial (total N = 46), which restricts generalizability; effects on other outcomes remain unsupported.
Relaxation-based music therapy 43 was associated with sleep latency values within the recommended threshold. However, baseline sleep latency also remained within the recommended threshold. Similar improvements in control groups point to possible placebo or methodological effects. This is supported by trivial effect size on latency and a negative small effect on sleep duration.
Social stimulation with PARO18,44 effects were associated with the most consistent benefit among non-pharmacological interventions. For total sleep time, daily social stimulation with PARO 44 met the ≥420-minute benchmark, whereas twice-weekly social stimulation with PARO 18 indicated large, statistically significant effects but remained below the benchmark. Only the twice-weekly study 18 reported sleep efficiency, indicating statistically significant improvements but not reaching the ≥85% optimal threshold.
Data gaps — especially in sleep latency (reported in only three studies) and nocturnal awakenings (four studies) — limit conclusions regarding broader efficacy. Inconsistent outcome reporting underscores the need for standardized measurement and comprehensive evaluation of key sleep parameters. It is commonly argued that non-pharmacological interventions should be fully explored before initiating pharmacological treatment. 47 However, particularly in outpatient care settings, prescribing benzodiazepines and other pharmacological agents remains common, while non-pharmacological interventions are often underutilized. 47 This pattern may reflect economic constraints and shortage of health care personnel, which can limit the feasibility of non-pharmacological interventions.
In summary, evidence remains fragmented across outcomes and interventions, limiting conclusions. This underscores the need for better standardization and more robust trials, particularly for non-pharmacological approaches.
Broader Discussion
In addition to efficacy, side effects and economic feasibility must be considered. The risk–benefit evaluation of Z-drugs, particularly in older adults, remains under debate. Z-drugs were introduced as improved alternatives to benzodiazepines, offering similar efficacy, but fewer side effects. Clinicians favored them due to better daytime functioning, increased total sleep time, and perceived safety regarding tolerance and dependency risks. 48
However, recent studies indicate that both z-drugs and ORAs were associated with increased risk of falls and fractures, comparable to benzodiazepines — especially in older patients with dementia.49,50 Evidence also points to rising dependence and misuse of z-drugs, suggesting prior risk assessments may have underestimated the problem. 51
REM sleep may be clinically relevant in neurodegeneration, in particular, isolated REM sleep behavior disorder is a strong prodromal marker for dementia with Lewy bodies. 52 Benzodiazepines alter sleep architecture and are generally associated with reduced REM sleep. 53 Although benzodiazepines are recognized to increase the risk of cognitive impairment in older adults, observational evidence regarding long-term dementia risk is mixed.54,55 By contrast, dual orexin receptor antagonists (DORAs), such as suvorexant and lemborexant, can influence REM parameters (eg, increased REM sleep time) in sleep-architecture studies. 56
To assess whether omission of REM-related terms could have led to missed dementia RCTs of DORAs, we conducted a post-hoc, targeted, supplemental search in December 2025 (covering 2020-2025) MEDLINE check outside the prespecified main search strategy, combining dementia/DORA terms with “REM”. While this check returned 41 records, title/abstract screening confirmed that no additional eligible RCTs were missed by the original January 2025 cutoff.
Non-pharmacological interventions offer a promising alternative, with potentially fewer side effects, though current evidence remains limited. Pharmacological treatments may improve some sleep outcomes, but involve risks such as dependency and sedation — especially problematic for older patients with dementia. In contrast, safer, non-pharmacological approaches often require structured environments and are thus more feasible in institutional settings.
Given these considerations, prioritizing non-pharmacological methods first, when feasible, is often recommended as an initial approach due to their minimal side-effect profiles. Even if these interventions do not fully resolve sleep disturbances, they might reduce symptoms sufficiently to reduce the overall need for medication, thus taking a more holistic view of patient care. Further research is needed to inform clinical guidance. Advanced dementia limits patient participation in many non-pharmacological interventions; however, circadian modulation light therapy may remain relatively feasible even in later stages, underscoring the need for interventions adaptable to varying levels of dementia severity. Research involving dementia patients presents ethical challenges, particularly regarding informed consent in advanced stages, as well as frequent compliance issues due to behavioral symptoms.57,58 Such factors may lead to selection bias, limiting evidence mostly to milder cases. Additionally, clinicians must carefully evaluate if sleep-related interventions align with patient preferences and best interests, as patients themselves may not desire further medication or interventions, complicating treatment decisions and ethical considerations in clinical practice.
Taken together, treatment decisions should balance modest and uncertain efficacy against safety and feasibility constraints, with REM-related questions and dementia-stage limitations highlighting priorities for future research.
Clinical Implications
Pharmacological options may be considered for patients with severe symptoms, or when non-pharmacological approaches are not feasible, while weighing risks. Clinicians may benefit from considering non-pharmacological strategies before pharmacotherapy, particularly in nursing home settings, where adherence can often be supported. Treatment strategies may benefit from being tailored to the patient’s clinical profile and goals. For severe insomnia requiring rapid improvements in sleep efficiency, sleep latency, and nocturnal awakenings, Z-drugs or ORAs may be considered based on individual risk–benefit assessment. For patients able to adhere to a structured regimen, social stimulation with PARO and circadian modulation light therapy may be considered where feasible and supported by setting/resources. However, non-pharmacological therapies can be difficult to implement in routine care. Combined or personalized strategies that account for dementia severity and comorbidities may improve outcomes. Future guidelines may benefit from incorporating structured assessment tools to tailor treatments to individual needs. Policy initiatives may support funding for non-pharmacological therapies in institutional settings, where adherence is generally higher. In addition, integrating cost–benefit analyses into clinical trials would further support clinicians and policy decision-making.
Bigger Picture – Placement Among the Literature
This review was informed by the 2020 Cochrane Review but was not designed as a direct update. 25 In the Cochrane Review, ORAs were the top-performing intervention and the only ones rated with moderate-certainty evidence. In our synthesis, ORAs were generally associated with small-to-large improvements in sleep efficiency and nocturnal awakenings, with inconsistent statistical significance. However, effects varied by compound, dose and were not consistently supported across outcomes, making their overall efficacy difficult to assess. Moreover, evidence of inconsistent dose–response patterns (particularly with Lemborexant) raises concerns about potential adverse effects on sleep that warrant further studies. This indicates the complexity of the research in this field. Research specifically addressing medications like melperone, pipamperone, doxepin, valproate, quetiapine, mirtazapine, valerian, and silexan in dementia populations remains limited, despite continued clinical use. This evidence gap limits reliable treatments for dementia-related sleep disturbances.59-62
As regards the corresponding Cochrane review 33 of non-pharmacological interventions, only one intervention class overlapped with our review: Circadian modulation light therapy. In the Cochrane review, light-based interventions were the most frequently studied approach (six trials), but certainty of evidence was very low, and effects were inconsistent, likely due to heterogeneity in protocols. Overall, Cochrane concluded that evidence for light therapy remains inconclusive. In our synthesis, circadian modulation light therapy indicated a large, significant reduction in nocturnal awakenings but did not meet clinical benchmarks. For other outcomes, evidence of benefit was not supported. Given the small sample size (N = 35), which limits generalizability, our findings are broadly consistent with Cochrane.
Differences between our included trials and those in Cochrane likely reflect differences in scope, eligibility operationalization, and trial classification, as there was no overlap in included non-pharmacological trials between both. This review included three trials that were excluded in Cochrane because baseline sleep disturbance was not obligatory, reflecting differences in operationalization of sleep-related eligibility criteria. Cochrane required sleep disturbance as an explicit inclusion feature. Our approach prioritized symptom-based evidence of baseline sleep impairment from reported measures, even when a formal diagnosis labeling of sleep disturbance was not specified. Further differences relate to non-overlapping search timeframes. In addition, one study was classified as non-randomized in Cochrane; following closer appraisal of the allocation procedure, we reclassified it as non-randomized and excluded it from the RCT synthesis.
Limitations
The exclusive use of a single database, restricting inclusion to studies published from 2020 onwards, and the restriction to English-language publications may contribute to an incomplete representation of the available evidence. This may have resulted in incomplete retrieval of eligible studies indexed elsewhere. Although study selection was conducted independently by two reviewers, limiting database coverage remains a methodological constraint and may affect the completeness of the evidence base. The risk-of-bias assessment was completed by one reviewer and discussed with a second reviewer; as needed, residual subjectivity cannot be excluded. Moreover, the absence of differentiation between dementia subtypes and failure to distinguish between care settings (institutional vs community-dwelling individuals) or stages of disease severity may introduce further imprecision and limit the generalizability of the findings. Conclusions are limited by small numbers of trials and heterogeneity.
Conclusion
This review provides a recent synthesis of dementia-related sleep treatments, an area with complex challenges and major impacts on patients and caregivers. By examining recent studies on a range of interventions, from pharmacological approaches such as z-drugs and orexin receptor antagonists to non-pharmacological strategies such as circadian modulation light therapy, and social stimulation with PARO, this work seeks to contextualize recent RCT evidence alongside Cochrane reviews by offering complementary evidence under a broader operationalization of sleep-related eligibility criteria. These preliminary insights highlight the potential of interventions to address dementia-related sleep disturbances by improving some outcomes in a small number of trials.
While statistical significance did not always translate into clinically meaningful improvements, social stimulation with PARO18,44 and z-drug eszopiclone 38 were associated with the most favorable profiles across outcomes, when considering both effect sizes and clinical benchmarks. Larger trials with standardized outcome reporting are needed to clarify the role of these interventions in clinical practice.
The value of this review lies in its attempt to shed light on the nuanced efficacy of these interventions, acknowledging the vast scope of future research necessary to refine and expand our understanding. The findings underscore the continued need for rigorous methodological research to evaluate intervention efficacy based on clinically recommended thresholds for key sleep parameters, providing clearer guidance to tailor treatment to the diverse needs of patients with dementia.
Supplemental Material
Supplemental material - Treatment Options for Sleep Disturbances in Dementia A Systematic Review
Supplemental material for Treatment Options for Sleep Disturbances in Dementia A Systematic Review by Duc Viet Pham, Arnim Quante in Journal of Geriatric Psychiatry and Neurology
Supplemental Material
Supplemental material - Treatment Options for Sleep Disturbances in Dementia A Systematic Review
Supplemental material for Treatment Options for Sleep Disturbances in Dementia A Systematic Review by Duc Viet Pham, Arnim Quante in Journal of Geriatric Psychiatry and Neurology
Supplemental Material
Supplemental material - Treatment Options for Sleep Disturbances in Dementia A Systematic Review
Supplemental material for Treatment Options for Sleep Disturbances in Dementia A Systematic Review by Duc Viet Pham, Arnim Quante in Journal of Geriatric Psychiatry and Neurology
Supplemental Material
Supplemental material - Treatment Options for Sleep Disturbances in Dementia A Systematic Review
Supplemental material for Treatment Options for Sleep Disturbances in Dementia A Systematic Review by Duc Viet Pham, Arnim Quante in Journal of Geriatric Psychiatry and Neurology
Footnotes
Acknowledgements
We would like to thank Stephen Schüürhuis, Charité Institute of Biometry and Clinical Epidemiology, for their consultation with the statistical analyzes in this study. I, Duc Viet Pham, would like to thank Prof. Dr Med. Surjo R. Soekadar and Fernanda Cristina de Carvalho Santos for the valuable knowledge and insights they shared, which have greatly enriched my understanding of research practices and methodologies.
Author contributions
Duc Viet Pham - Conducted the literature search, performed data extraction and statistical analysis, drafted the manuscript. Email:
Declaration of Conflicting Interests
The authors report that they do not have conflicts with any product or concept mentioned in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data used in this study were derived from publicly available sources and are accessible to the public. The corresponding author can provide specific datasets and detailed instructions for accessing them on request.
Supplemental Material
Supplemental material is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
