Abstract
Background
This study evaluated the diagnostic value and factorial structure of the Subjective Cognitive Decline Questionnaire (SCD-Q) and the Cognitive Failures Questionnaire (CFQ) in the context of early detection of cognitive decline in a memory clinic setting.
Methods
A total of 128 patients were included (AD/MCI: n = 50; non-AD dementia: n = 10; mixed dementia: n = 16; subjective cognitive decline [SCD]: n = 21). Participants completed the modified SCD-Q17 and CFQ and underwent standardized cognitive assessment. Principal component and cluster analyses, regression models, and ROC analyses were used to examine psychometric properties and diagnostic performance.
Results
The SCD-Q17 correlated with objective cognition (CERAD: r = −0.29 to −0.35, P = 0.023-0.005) and differentiated SCD from mixed dementia (AUC = 0.71). The CFQ primarily reflected executive and attentional failures and showed moderate discrimination for non-AD dementia (SCD vs NADD: AUC = 0.67). Cluster analyses identified 2 profiles (impaired vs unimpaired) for both instruments (κ = 0.47). PCA indicated a more redundant structure for the SCD-Q17, whereas the CFQ showed a broader multidimensional structure.
Conclusions
Relative to CERAD performance, both questionnaires distinguished cognitively impaired from unimpaired individuals, but neither provided sufficient precision to classify specific clinical entities.
Keywords
Background
Neurodegenerative diseases such as Alzheimer’s disease (AD) usually develop gradually over several years before a clinical diagnosis is made.1-3 Subjective cognitive decline (SCD), defined as self-perceived cognitive deterioration in the absence of objective impairment, has been proposed as a possible early indicator of increased risk for developing dementia.4,5 Moreover, recent advances in disease-modifying treatments and preventive approaches have intensified efforts to identify cognitive changes at the earliest possible stages.6-8 As such strategies increasingly extend beyond highly specialized centres and target broader populations, brief and easily deployable tools such as self-report questionnaires are gaining importance. 9
Several self-report questionnaires have been developed to assess subjective cognitive complaints and are widely used in both research and clinical settings. Among the most applied questionnaires are the Subjective Cognitive Decline Questionnaire (SCD-Q) and the Cognitive Failures Questionnaire (CFQ).4,10-12 The SCD-Q focuses on perceived changes in memory, executive function, and language over the past 2 years—domains typically affected in the early or preclinical stages of AD.4,10,13,14 In contrast, the CFQ captures a wider range of everyday cognitive lapses, and was originally developed to evaluate memory and attention failures in the general population.4,11,12,15-17
Importantly, neither questionnaire is intended as a diagnostic tool. Rather, subjective cognitive measures are best understood as one component within a broader risk stratification framework. Their primary purpose is not to establish a diagnosis, but to help identify individuals who may be at increased risk and who may benefit from further clinical assessment, particularly when interpreted in combination with clinical, demographic, or biological markers. This perspective highlights their potential value for low-threshold, scalable identification of subtle cognitive concerns that may not be captured by standardized assessments such as the Consortium to Establish a Registry for Alzheimer’s Disease neuropsychological battery (CERAD) or the Clinical Dementia Rating scale (CDR).18,19
The extent to which these questionnaires capture overlapping or distinct subjective cognitive constructs—and their respective diagnostic utility in clinical settings—remains unclear. Previous studies have reported inconsistent associations of self-report questionnaires with objective cognitive performance and clinical outcomes, underscoring the need for a systematic comparison of their psychometric properties.3,20
In this context, this cross-sectional study compared the psychometric structure and diagnostic value of the SCD-Q and CFQ in a memory clinic setting. It also examined their relationship with objective cognitive performance and clinical diagnosis. The aim was to facilitate evidence-based pathways for the early identification and prevention of dementia.
Methods
Study Design and Setting
This observational cross-sectional study was conducted at the Memory Clinic of the Department of Psychiatry and Psychotherapy, LMU Munich, Germany. Participants were consecutively recruited during routine diagnostic work-up between January 2022 and July 2025. The study was conducted in accordance with the Declaration of Helsinki and approved by the local ethics committee (Ethics vote: 22-1117, ClinicalTrials.gov ID: NCT06711952). Written informed consent was obtained from all participants prior to inclusion. Sample size, inclusion and exclusion criteria, and all measures used in the study are described in detail in the following Methods sections.
Study Population and Recruitment
Eligible participants were referred by primary care or specialist providers for cognitive assessment. Inclusion criteria were age >60 years, sufficient German language skills, and the presence of subjective cognitive complaints as the primary reason for referral. Exclusion criteria comprised cognitive impairment due to secondary causes (eg, alcohol abuse, malnutrition, thyroid dysfunction). During the recruitment period, 244 individuals were referred for cognitive assessment and considered for study participation. Of these, 128 participants fulfilled all inclusion criteria and completed the self-report questionnaires prior to the standardized diagnostic assessment. Extreme outliers were evaluated on an analysis-specific basis. Except for one participant excluded due to a multivariate outlier identified using Mahalanobis distance, no individuals were excluded entirely from the dataset. Where applicable, extreme values were handled within individual analyses according to standard statistical assumptions. To minimize bias, participants were recruited consecutively using defined inclusion and exclusion criteria. The clinical diagnostic process was standardized and responses to the questionnaire were blinded. Standardized questionnaires, training and diagnostic criteria were used throughout. The STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines were followed to ensure transparency and reproducibility. 21 A detailed overview of participant flow, inclusion, and diagnostic classification is provided in Supplemental Figure S1.
Diagnostic Procedure and Classification
All participants underwent a standardized clinical diagnostic work-up at the memory clinic. Final diagnoses were established by board-certified specialists in psychiatry or neurology based on a multidisciplinary evaluation and in accordance with current international consensus criteria.5,22 Individuals with dementia were included only if they were in a mild disease stage. All participants were required to have sufficient capacity to provide written informed consent, which was assessed as part of the clinical evaluation.
The diagnostic protocol included a (1) detailed medical history and structured clinical interview, a (2) comprehensive neurological and psychiatric examination, a (3) standardized neuropsychological assessment using the extended CERAD-Plus battery, a (4) routine laboratory testing (eg, metabolic, endocrine, inflammatory parameters) and a (5) structural brain imaging (magnetic resonance imaging, MRI), when available. In a subset of patients, MRI had been obtained externally prior to referral and was integrated into the diagnostic evaluation. Additional investigations (eg, electroencephalography) were considered when clinically indicated.
Neuropsychological performance was quantified using the CERAD-Plus battery, which assesses multiple cognitive domains including memory, language, visuoconstruction, and executive functioning, and provides age-, sex-, and education-adjusted normative scores.23-25 In the present study, the CERAD total score served as the primary measure of objective cognitive performance.23-25
Diagnostic classification was based on the integrated clinical judgment of the treating specialist, synthesizing neuropsychological results, clinical presentation, laboratory findings, and neuroimaging. Participants were categorized into 4 groups: (1) Subjective Cognitive Decline (SCD), defined as cognitive complaints without objective impairment, (2) Alzheimer-related cognitive impairment (AD spectrum, including MCI due to AD and AD dementia), (3) non-Alzheimer neurodegenerative diseases (NADD; including frontotemporal dementia, Parkinson’s disease, and atypical parkinsonian syndromes); and a (4) mixed aetiology group which comprised individuals with evidence of more than one contributing pathological or clinical factor (eg, neurodegenerative changes in combination with vascular pathology or other relevant comorbidities). These participants were not excluded but analysed as a separate diagnostic category.
Individuals with dementia were included only if they were classified as being in a mild disease stage according to standard clinical criteria applied during routine diagnostic evaluation. Individuals with dementia were included only if they were classified as being in a mild disease stage according to standard clinical criteria applied during routine diagnostic evaluation.
Rationale for Grouping of AD and MCI
For the primary analyses, individuals with MCI attributable to Alzheimer’s disease and those with AD dementia were combined into a single “AD spectrum” group. This grouping was defined a priori and reflects a construct-based approach: both conditions represent different stages of the same Alzheimer-related cognitive phenotype, characterized by overlapping patterns of memory and executive dysfunction. As the present study focuses on relationships between self-report questionnaires and cognitive constructs rather than on diagnostic differentiation per se, this approach allowed meaningful comparison while improving statistical power.
Questionnaires
As part of the initial assessment, participants completed 2 self-report questionnaires prior to the standardized diagnostic evaluation and independently of the final clinical diagnosis. Both instruments were administered in paper-and-pencil format and scored according to their respective manuals.
Cognitive Failures Questionnaire (CFQ)
The 32-item CFQ) assesses everyday lapses in attention, memory, and motor functioning and motor functioning as experienced by the participant during the past 6 months. Items are rated on a 5-point Likert scale ranging from zero (“never”) to 4 (“very often”), with higher scores indicating a greater perceived frequency of cognitive failures (see Supplemental Table S3A for details). 12 A total CFQ score was calculated by summing all item responses, with higher scores indicating a greater perceived frequency of cognitive failures. In addition to the total score, the CFQ captures multiple dimensions of everyday cognitive functioning, including attention, memory, and action control, as described in previous work. 12
Subjective Cognitive Decline Questionnaire (SCD-Q)
The original SCD-Q comprises 24 dichotomous (yes/no) items assessing perceived decline in memory, language, and executive functions relative to the participant’s own self-perceived functioning 2 years earlier and is therefore referred to here as the SCD-Q24. 13 For the present study, we used a modified and shortened version of this questionnaire (SCD-Q17), derived by selecting 17 items and adapting the response format to a 5-point Likert scale to improve sensitivity and response variability. This adapted version was developed by our group and has been described previously. 10 Throughout this manuscript, the term SCD-Q17 refers exclusively to this shortened, modified version, whereas SCD-Q24 denotes the original questionnaire by Rami et al (see Supplemental Table S3B for more information).
The questionnaire begins with 3 introductory questions addressing the presence and personal relevance of subjective cognitive concerns (eg, perceived memory problems, worry, and intention to seek medical advice). Participants who answered “no” to all 3 screening questions were nevertheless included and completed the full questionnaire.12,13 A total SCD-Q17 score was calculated as the sum of all item responses, with higher scores indicating greater subjective cognitive decline. In line with the validation of the SCD-Q17, a cut-off score of 22 points was applied to identify clinically relevant subjective cognitive complaints. 10
Variable Definitions and Data Sources
The primary outcome variables were the total scores for the CFQ and the SCD-Q17, as well as the total CERAD score, which was used as an index of objective cognitive performance. 26 Data were systematically extracted from standardized clinical documentation, including structured interviews, self-report questionnaires, neuropsychological test protocols, and medical records, using a predefined data extraction framework. Participants were recruited consecutively, and all of them completed both self-report questionnaires. No cases were excluded due to missing data on key variables. Outlier detection was performed using Mahalanobis distance, and extreme outliers were excluded. 27
Linguistic Analysis
To explore whether differences in item wording might contribute to the divergent psychometric profiles of the 2 questionnaires, descriptive, non-standardized item-level content review of the SCD-Q17 and CFQ. Instead, we conducted a systematic, item-level content review focusing on structural characteristics of the items, including item length (number of words), syntactic complexity (eg, use of subordinate clauses or temporal comparisons), and semantic focus (eg, concrete everyday behaviours vs abstract or comparative judgments). Word counts and summary characteristics were derived manually and are reported descriptively in Supplemental Tables S4A and S4B. This approach was intended to provide an exploratory comparison of item structure rather than a formal linguistic or readability assessment.
Statistical Methods
Descriptive statistics were computed for all relevant variables. Group comparisons were performed using ANOVA or Kruskal–Wallis tests for continuous variables and chi-squared tests for categorical variables. Correlation (Pearson or Spearman) and linear regression with backward stepwise selection (AIC-based) were used to examine the relationships between scores; multicollinearity was assessed using variance inflation factors (VIF).
ROC analyses assessed the diagnostic utility of both questionnaires in discriminating the SCD reference group from clinical subgroups (NADD, mixed, and AD/MCI), adjusted for age. Principal component analyses (PCA) and hierarchical clustering were applied to explore response patterns and relationships between subjective and objective measures.
Hierarchical cluster analyses were subsequently applied in a data-driven manner to identify latent cognitive complaint profiles based on continuous questionnaire scores in combination with objective cognitive performance (CERAD). No priori cut-off values or diagnostic categories were used to define cluster membership. Cluster solutions were interpreted post hoc by comparing questionnaire scores, CERAD performance, diagnostic groups, and sociodemographic variables. Agreement between cluster solutions derived from CFQ- and SCD-Q17-based models was quantified using cross-tabulation and Cohen’s κ. To account for multiple testing (eg, post hoc comparisons), P-values were adjusted using the Benjamini–Hochberg FDR procedure; q < .05 (two-tailed) was considered statistically significant. Given the naturalistic recruitment, no a priori sample size calculation was performed. Sensitivity analyses were conducted for the primary inferential analyses (correlations, group comparisons, and ROC analyses). Sensitivity analyses showed that with n = 128 and α = 0.05 (two-tailed), correlations of approximately r ≥ 0.25 and moderate ROC performance (AUC ≳ 0.65 vs 0.50) can be detected with 80% power, indicating adequate power for small-to-moderate effects. Formal power calculations are not well-established for exploratory multivariate techniques such as PCA and hierarchical clustering; these analyses were therefore considered exploratory and hypothesis-generating. Given the number of analyses performed, results - particularly from exploratory procedures - should be interpreted cautiously with respect to statistical power and the risk of false-positive findings, despite application of FDR correction.
All analyses were conducted using R (version 2025.05.1 + 513) and IBM SPSS Statistics (version 29). The R packages used were: ggplot2, dplyr, psych, pROC, car, rstatix and others as required. Results were considered statistically significant at P < .05 (two-tailed).
Results
The following section provides a detailed characterization of the sample and diagnostic subgroups. This forms the basis for evaluating the diagnostic value and interpretability of subjective cognitive complaint measures.
Sample Characteristics and Sociodemographics
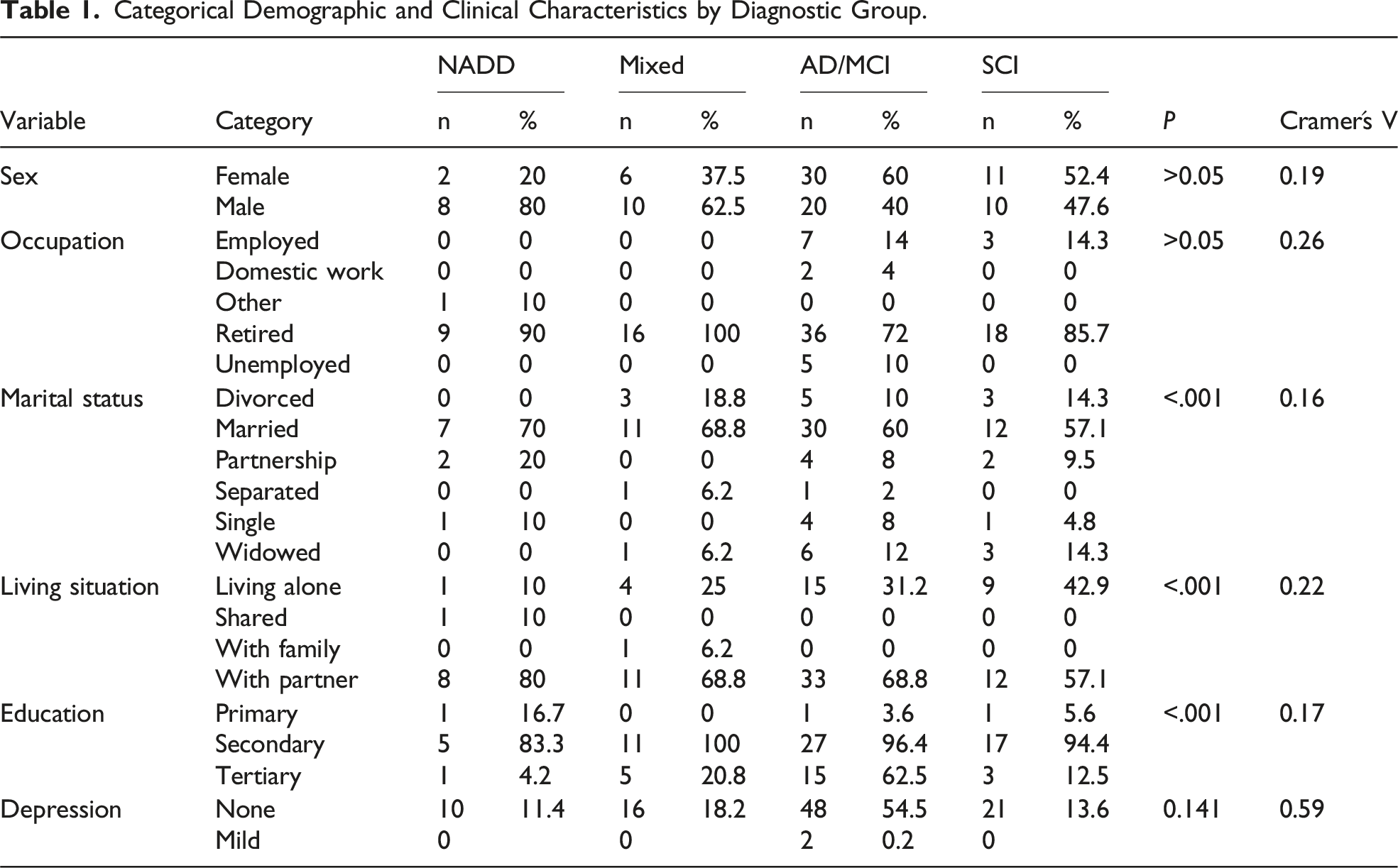
Categorical Demographic and Clinical Characteristics by Diagnostic Group.
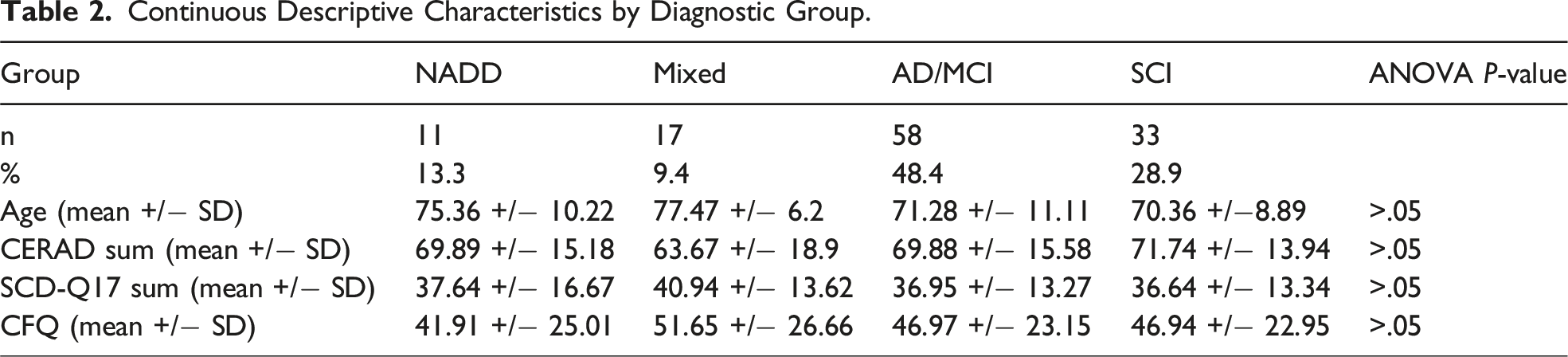
Continuous Descriptive Characteristics by Diagnostic Group.
Self-Reported Cognitive Complaints by Diagnostic Group
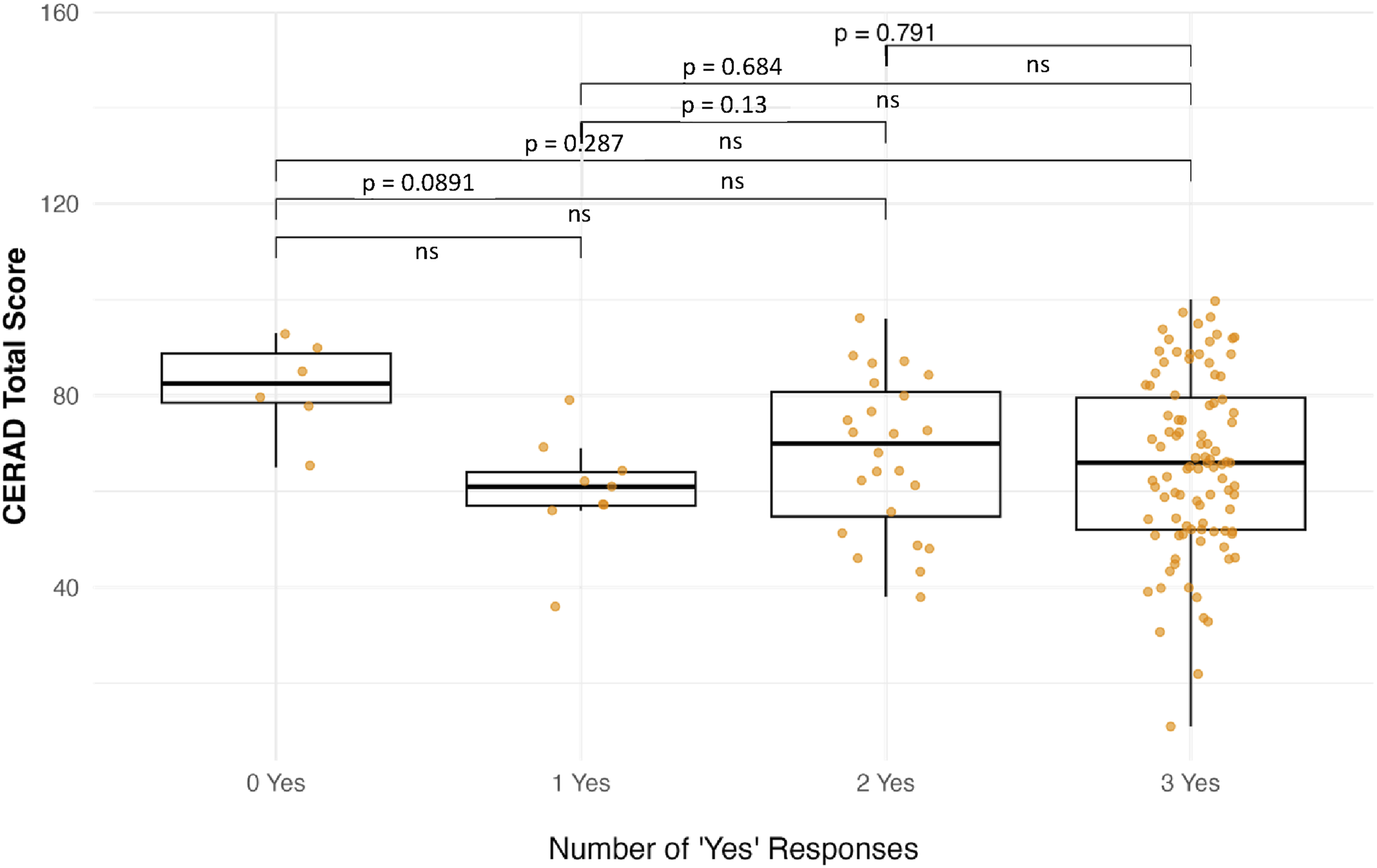
Consistent with this finding, post-hoc pairwise comparisons revealed no significant group differences for either CFQ or SCD-Q17 after correction for multiple testing (all adjusted P-values >.05; see Supplemental Tables S1 and S2). The boxplots in Figure 1 illustrate the total CFQ and SCD-Q17 scores for each diagnostic group. There were no significant differences in CFQ or SCD-Q17 total scores between groups (both ANOVA P > .05), although the mixed group had higher mean CFQ scores on average and AD/MCI patients reported slightly higher SCD-Q17 scores (see Table 2 for details). These findings suggest that subjective cognitive failures, as measured by the CFQ, vary across diagnostic categories, whereas self-reported decline, as assessed by the SCD-Q17, is more evenly distributed (Figure 1). In their current wording, the three introductory questions were not significantly associated with the CERAD total score and therefore did not meaningfully differentiate objective cognitive performance in this sample (Figure 2). Self-reported cognitive complaints across diagnostic groups (A) CFQ total score by group. (B) SCD-Q total score by group. Association between SCD-Q17 introductory questions and CERAD total score.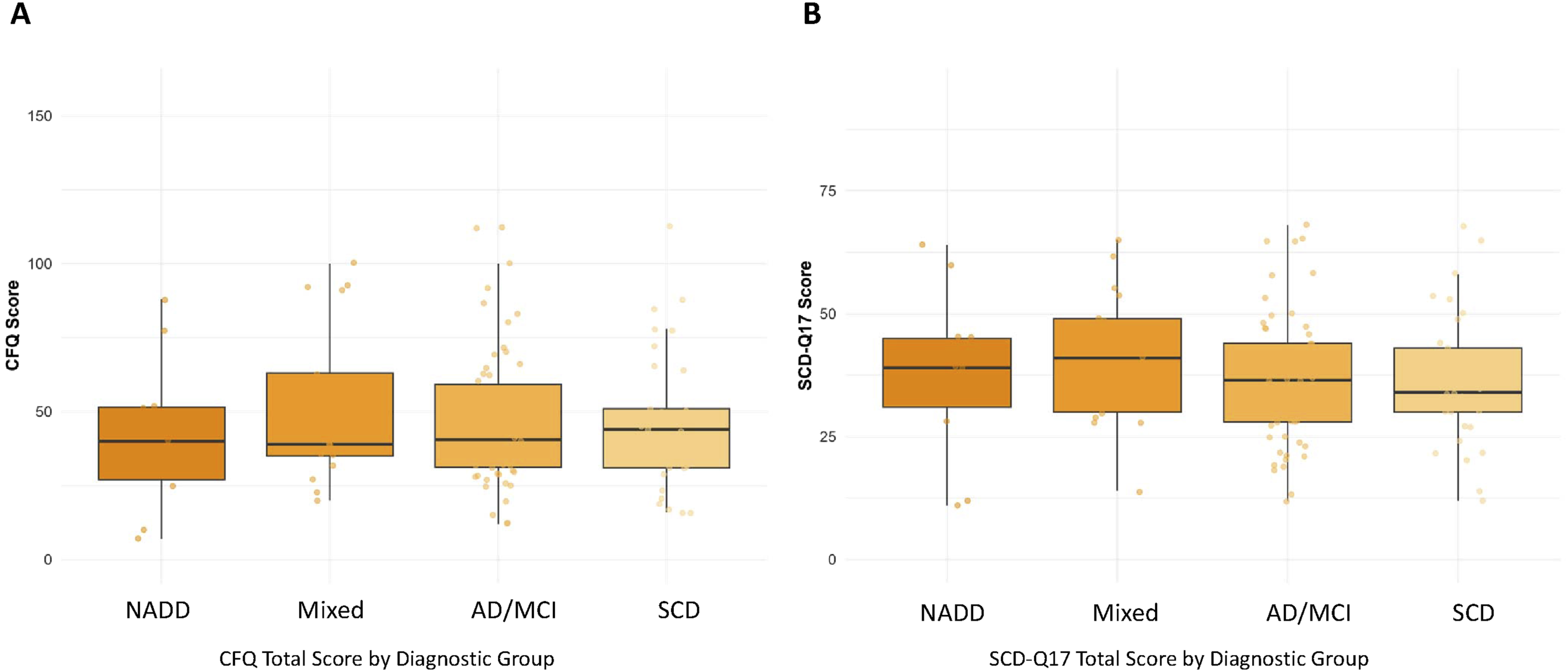
Questionnaire Structure and Factor Analyses
Functional and Factorial Item Mapping of CFQ and SCD-Q17 Items.

Cluster Analysis and Cognitive Profiles
Distribution of Diagnostic Groups Across SCD-Q17- and CFQ-Based Cluster Solutions.
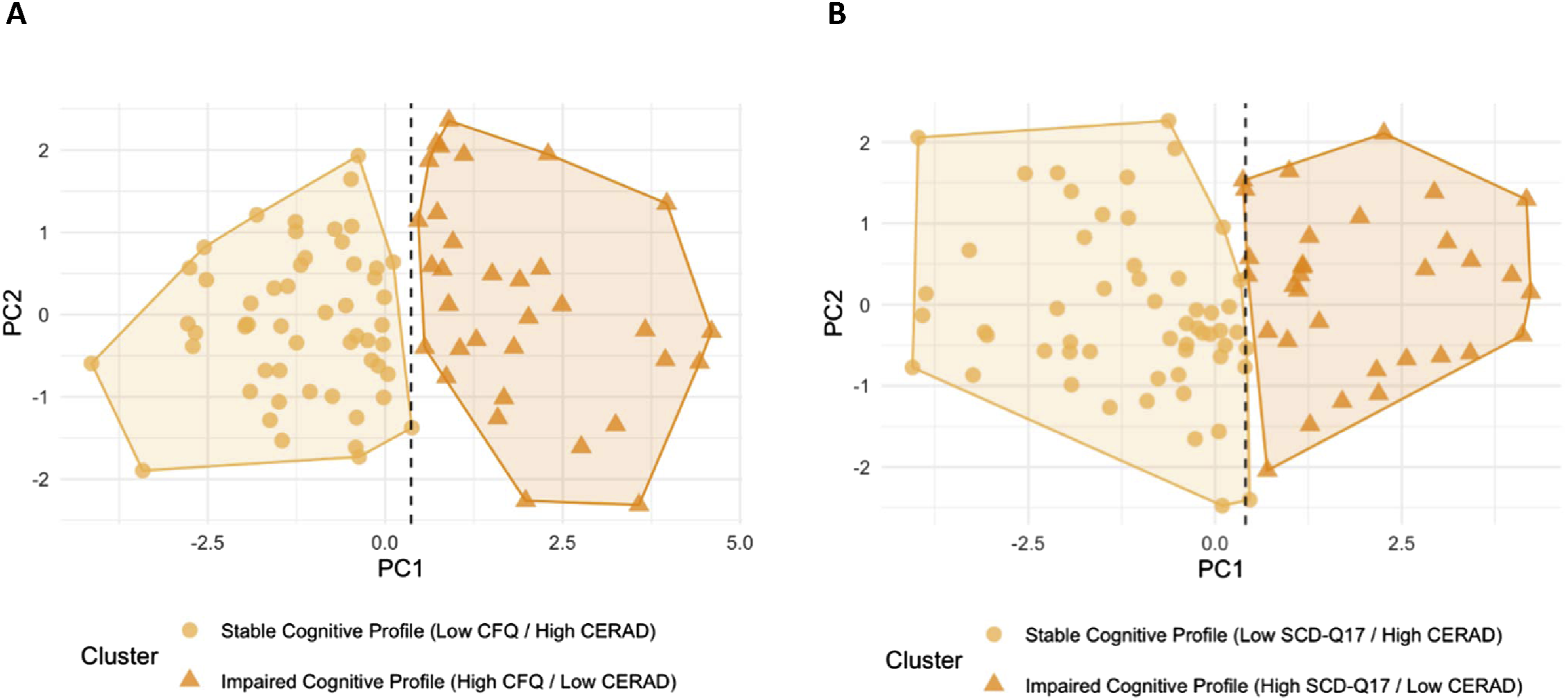
Mapping subjective complaints to objective cognition via CFQ and SCD-Q17 cutoffs (A) Cluster based on CFQ and CERAD scores. (B) Cluster based on SCD-Q and CERAD scores.
ROC Analysis and Diagnostic Differentiation
Receiver operating characteristic (ROC) analyses were conducted to evaluate how well the CFQ and SCD-Q17 distinguish persons with SCD from the other clinical subgroups. All ROC analyses were adjusted for age by incorporating it as a covariate in the underlying classification models prior to ROC curve estimation. Diagnostic accuracy was quantified using the area under the curve (AUC), sensitivity, specificity, and the Youden index. Sensitivity represents the proportion of true positives correctly identified, specificity the proportion of true negatives correctly identified, and the Youden index (J = sensitivity + specificity − 1) identifies the optimal cut-off by maximizing their combined value. AUC values of 0.5 indicate chance-level discrimination, values around 0.7 reflect acceptable performance, and values ≥0.8 indicate good discrimination. Higher sensitivity indicates better detection of affected individuals, whereas higher specificity reflects better exclusion of non-affected individuals; higher Youden values indicate a more optimal balance between both.
Diagnostic Performance of the CFQ and SCD-Q17 Questionnaires (ROC Analyses).
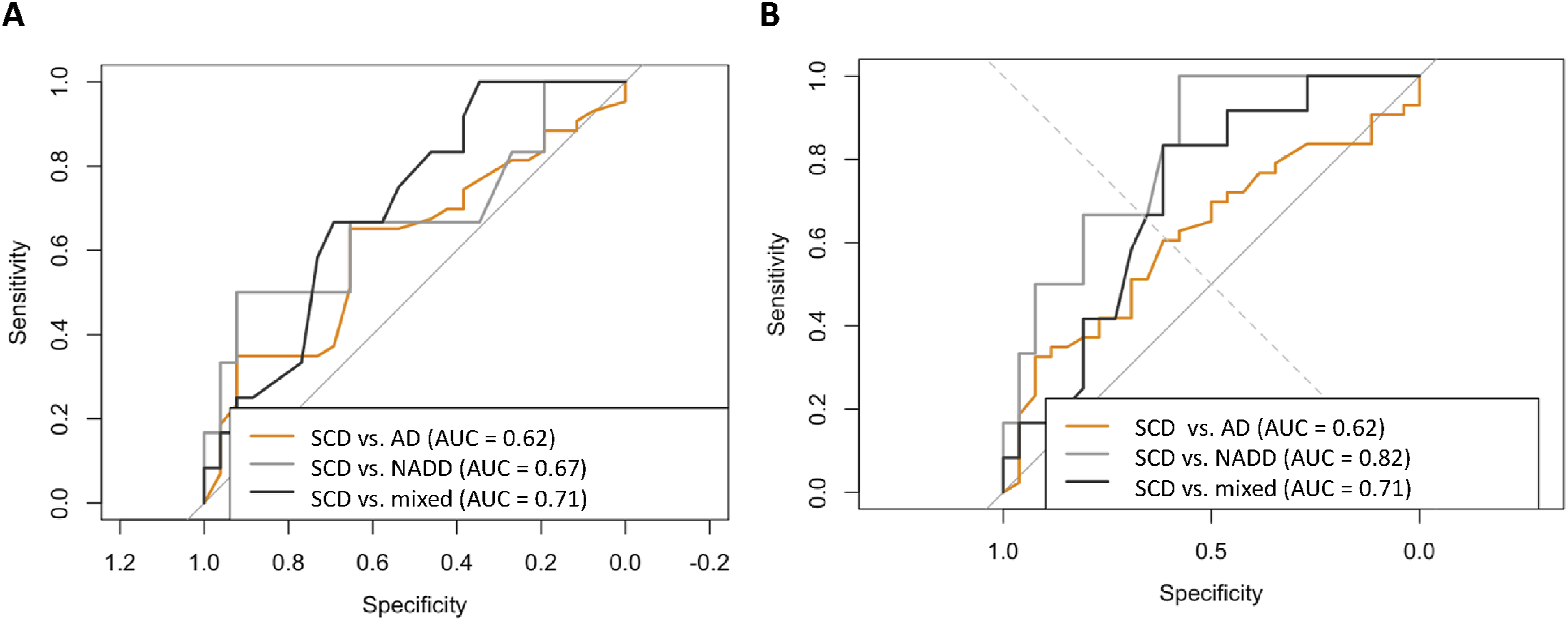
Diagnostic discrimination based on subjective complaint scores (A) CFQ-differentiation of diagnostic groups. (B) SCD-Q-differentiation of diagnostic groups.
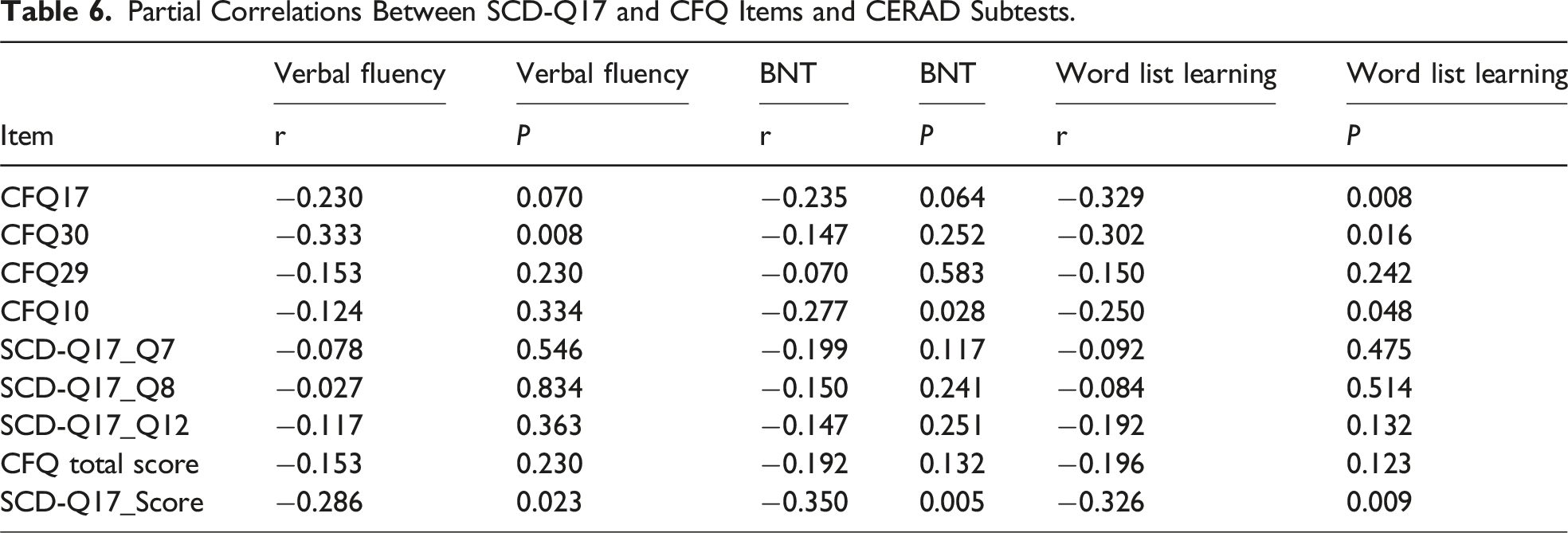
Correlations with CERAD Subdomains
Partial Correlations Between SCD-Q17 and CFQ Items and CERAD Subtests.
Linguistic Characteristics of Questionnaires Items
To provide descriptive context for the differing psychometric profiles of the SCD-Q17 and CFQ, we conducted an exploratory, non-standardized item-level content review focusing on structural characteristics of item wording. Although both questionnaires employ a 5-point Likert response format, their items differ in linguistic structure and semantic focus. Items of the SCD-Q17 frequently involve temporal comparisons and self-referential judgments (eg, perceived decline relative to 2 years earlier) and often use more complex sentence constructions. In contrast, CFQ items tend to describe concrete, everyday cognitive lapses using shorter and more behaviourally anchored formulations. These descriptive differences in item wording and structure are summarized in Supplemental Tables S4A and S4B and are reported here to contextualize the observed differences in factor structure and response patterns between the 2 instruments.
Discussion
This study compared the psychometric structure and diagnostic utility of 2 widely used self-report questionnaires, the CFQ and the SCD-Q17, to clarify their respective roles in the early assessment of cognitive complaints. Although SCD is associated with an increased risk of subsequent cognitive decline, many affected individuals remain cognitively stable over time; accordingly, subjective complaints should be interpreted as indicators of perceived impairment and potential vulnerability and should not be interpreted as definitive markers of prodromal AD.5,28 The present findings are discussed in terms of risk stratification and not diagnostic prediction. This study aimed to refine interpretations, reduce redundancy and outline realistic implications for clinical and research use.
Within this framework, the CFQ and SCD-Q17 assess complementary yet conceptually distinct dimensions of subjective cognition, which has implications for their clinical use. The SCD-Q17 primarily targets perceived memory-related decline, whereas the CFQ captures a broader spectrum of everyday cognitive lapses. Within this framework, our findings show that the CFQ and SCD-Q17 capture partly distinct aspects of subjective cognition and provide complementary information. While both instruments demonstrated only modest diagnostic discrimination, they differentiated broader complaint profiles across the sample, supporting their use for risk stratification and symptom characterization rather than categorical diagnosis. The choice of questionnaire should therefore be guided by the clinical question, with the SCD-Q17 emphasizing memory-focused concerns and the CFQ offering a broader assessment of everyday cognitive functioning.
Implications for Questionnaire Design: Complexity, Redundancy, and Usability
Against this conceptual background, differences in item wording and structure may further shape how subjective complaints are framed, perceived, and reported. At the same time, both questionnaires may benefit from further linguistic refinement to optimize clarity and usability. Rather than advocating expanded clinical algorithms based on the current questionnaires, our results therefore argue for refinement and simplification—reducing redundancy, clarifying domain coverage, and optimizing language to improve accessibility without compromising construct validity. Future research should systematically examine how item formulation affects response behaviour, interpretability, and clinical applicability.1,2,5,29,30
Clinical Context and Practical Considerations
Positioning brief self-report questionnaires at the entry point of a clinical pathway can enhance accessibility and scalability while minimizing reliance on specialized services. In such models, case finding begins with tools such as the SCD-Q and CFQ and, when indicated, proceeds to targeted cognitive assessment and biomarker-based evaluation. 6 At the same time, early identification is only meaningful if it is linked to timely counselling, preventive strategies and psychosocial support. Consistent with the modest diagnostic discrimination observed in the present study, the use of questionnaires as entry points in clinical assessment pathways requires clear justification, particularly for individuals with minimal symptoms or who are clinically asymptomatic. Results should therefore be communicated with caution and framed as indicators of subjective concern and potential vulnerability rather than as diagnostic markers, taking into account ethical and economic consideration.3,7,31
Limitations
Several limitations should be considered when interpreting these findings. First, the study was conducted in a specialized memory clinic setting. Referral-based samples differ substantially from community-based populations, and longitudinal evidence indicates that progression rates from subjective cognitive decline to MCI or dementia are lower outside specialist settings. Findings from memory clinics may therefore overestimate conversion risk, and differences in case mix related to age, education, and socioeconomic factors limit generalizability to broader populations.28,32,33 Second, the sample was restricted to individuals presenting with subjective cognitive complaints, which introduces a degree of conceptual circularity. As the questionnaires were evaluated only within the population they are designed to identify, the present study cannot assess their full discriminative utility across the general population. Consequently, it remains unclear whether these instruments should be applied exclusively to individuals who spontaneously report cognitive concerns or more broadly as part of an initial assessment, including individuals who may not self-identify as cognitively impaired. Additional limitations include the cross-sectional design, the absence of gold-standard biomarker confirmation (eg, cerebrospinal fluid analysis), unequal subgroup sizes, and the lack of informant ratings, which limits conclusions regarding insight or awareness. Although conducted at a single memory clinic, the cohort reflects a typical real-world diagnostic population, including individuals with comorbid affective disorders, thereby enhancing the validity of the findings. Finally, while the sample size was sufficient to detect small-to-moderate effects in primary analyses, statistical power for exploratory multivariate procedures is inherently limited. Accordingly, results from exploratory analyses should be interpreted cautiously and considered hypothesis-generating.
Integrative Summary and Outlook
In summary, the present study demonstrates that the CFQ and SCD-Q capture complementary, but distinct aspects of subjective cognitive complaints. Consistent with the structural analyses, the CFQ reflects a multidimensional profile encompassing executive, attentional failures, whereas the SCD-Q is more narrowly focused on perceived memory-related decline. These differences were further supported by the present findings, which showed modest diagnostic discrimination for both instruments but clear differentiation of broader complaint profiles across the sample. Together, the results indicate that subjective questionnaires are more informative for risk stratification and symptom characterization than for categorical diagnostic classification. From a clinical perspective, these findings underscore that the CFQ and SCD-Q are not interchangeable and should be selected based on the clinical question. The SCD-Q may be most useful when memory-dominant decline is the primary concern, whereas the CFQ provides a broader perspective on everyday cognitive functioning. Importantly, neither questionnaire alone is sufficient for diagnostic decision-making, reinforcing their role as adjunctive tools within a broader clinical assessment. Future research should primarily focus on longitudinal validation to clarify how different subjective complaint profiles relate to cognitive trajectories over time. Additional priorities include evaluating refined questionnaire versions with reduced redundancy, assessing performance in non-specialist and community-based settings and integrating informant-based perspectives where patient insight might be limited. These steps are essential to determine how subjective cognitive measures can be optimally embedded into scalable, ethically sound assessment pathways (Figure 5). Ultimately, optimized self- and proxy-report questionnaires could facilitate the earlier identification of individuals at risk, enable more tailored interventions, and support the development of effective prevention strategies across a broad range of conditions associated with cognitive decline. Proposed clinical pathway for cognitive screening and case-finding in AD and NADD.
Supplemental Material
Supplemental Material - Subjective Cognitive Complaints and Objective Cognitive Performance Across Diagnostic Groups in Memory Clinic Patients: A Cross-Sectional Comparison of the SCD-Q and CFQ Questionnaires
Supplemental Material for Subjective Cognitive Complaints and Objective Cognitive Performance Across Diagnostic Groups in Memory Clinic Patients: A Cross-Sectional Comparison of the SCD-Q and CFQ Questionnaires by Paulina Tegethoff, Marleen Taute, Sophia Rutt, Fortese Neziraj, Anna Hufnagel, Robert Perneczky, Carolin Kurz in Journal of Geriatric Psychiatry and Neurology
Supplemental Material
Supplemental Material - Subjective Cognitive Complaints and Objective Cognitive Performance Across Diagnostic Groups in Memory Clinic Patients: A Cross-Sectional Comparison of the SCD-Q and CFQ Questionnaires
Supplemental Material for Subjective Cognitive Complaints and Objective Cognitive Performance Across Diagnostic Groups in Memory Clinic Patients: A Cross-Sectional Comparison of the SCD-Q and CFQ Questionnaires by Paulina Tegethoff, Marleen Taute, Sophia Rutt, Fortese Neziraj, Anna Hufnagel, Robert Perneczky, Carolin Kurz in Journal of Geriatric Psychiatry and Neurology
Footnotes
Author’s Note
R.P. has received honoraria for advisory boards and speaker engagements from Roche, EISAI, Eli Lilly, Biogen, Janssen-Cilag, AstraZeneca, Schwabe, Grifols, Novo Nordisk, and Tabuk, and is shareholder of Medotrax GmbH and Vistim Ltd. as well as Founding Chairman of the Board of InRAD Foundation.
Acknowledgements
We are deeply grateful to all participants and their families for their valuable time, trust, and willingness to contribute to this study. Their engagement made this research possible.
Author Contributions
Paulina Tegethoff: Conceptualization, Methodology, Formal Analyis, Writing – original draft, Writing – review & editing, Visualization. Marleen Taute: Conceptualization, Methodology, Formal Analyis, Writing – original draft, Writing – review & editing, Visualization. Sophia Rutt: Conceptualization, Methodology, Formal Analyis, Writing – original draft, Writing – review & editing, Visualization. Fortese Neziraj: Writing – review & editing. Anna Hufnagel: Writing – review & editing. Robert Perneczky: Conceptualization, Writing – review & editing, Supervision. Carolin Kurz: Conceptualization, Methodology, Formal Analyis, Writing – original draft, Writing – review & editing, Visualization, Supervision. Paulina Tegethoff and Marleen Taute have equal contribution.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R.P. was supported by the German Center for Neurodegenerative Disorders (Deutsches Zentrum für Neurodegenerative Erkrankungen, DZNE), the Hirnliga e.V. (Manfred-Strohscheer Stiftung), the Davos Alzheimer’s Collaborative, the VERUM Foundation, the Robert-Vogel-Foundation, the National Institute for Health and Care Research (NIHR) Sheffield Biomedical Research Centre (NIHR203321), the University of Cambridge – Ludwig-Maximilians-University Munich Strategic Partnership within the framework of the German Excellence Initiative and Excellence Strategy, the European Commission under the Innovative Health Initiative program (project 101132356), the Deutsche Forschungsgemeinschaft (DFG, 1007 German Research Foundation) under Germany’s Excellence Strategy within the framework of 1008 the Munich Cluster for Systems Neurology (EXC 2145 SyNergy – ID 390857198), the Alzheimer’s Association and Gates Ventures.
Clinical Trial Registration
No material requiring third-party permission has been included, and the work is not associated with any clinical trial registration.
Ethical Considerations
This manuscript reports investigator-initiated original research involving patient data. Informed consent was obtained from all individual participants included in the study. This cross-sectional observational study was conducted at the Memory Clinic, Department of Psychiatry and Psychotherapy, LMU Hospital Munich (Germany) between January 2022 and July 2025. Ethical approval for the study was obtained from the Ethics Committee of the Ludwig-Maximilians-Universität München (vote number: 22-1117, clinical trials NCT06711952).
Consent to Participate
Written informed consent was obtained from all participants prior to inclusion. The study was conducted in accordance with the Declaration of Helsinki and relevant institutional guidelines.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
