Abstract
Objective
Psychosis is a significant neuropsychiatric symptom in Alzheimer’s Disease (AD) and Mild Cognitive Impairment (MCI). While female APOE4 homozygotes are at the highest risk of psychosis, mechanisms underlying this association remain unclear. We investigated whether epigenetic age acceleration (EAA) is associated with psychosis in individuals with AD and MCI, and whether this association varies by sex and APOE4 status.
Methods
Participants with clinical MCI or AD were drawn from the Alzheimer’s Disease Neuroimaging Initiative database. EAA was calculated from DNA methylation data using the PhenoAge epigenetic clock, with the Horvath and Hannum clocks included for comparison. Psychosis status was defined as experiencing hallucinations or delusions at any time, based on the Neuropsychiatric Inventory Questionnaire. Logistic regression models were used to assess associations between EAA and psychosis, including three-way interactions with sex and APOE4 carrier status.
Results
Among 418 total participants (54 with psychosis, 84 female APOE4 carriers), EAA was not associated with psychosis for any of the 3 clocks utilized. However, a significant three-way interaction between sex, APOE4 carrier status, and PhenoAge EAA in predicting psychosis was observed (P = 0.013). Elevated PhenoAge EAA was associated with higher odds of psychosis in female APOE4 carriers (OR = 1.76 per 5-year, P = .025), whereas no associations were observed for other subgroups or clocks.
Conclusions
PhenoAge EAA may serve as a novel biomarker of psychosis among female APOE4 carriers. These findings highlight the potential of second-generation epigenetic clocks for early risk stratification and biological investigations into psychosis in AD and MCI.
Introduction
Psychosis is a common and clinically significant neuropsychiatric symptom (NPS) in individuals with Alzheimer’s disease (AD) and mild cognitive impairment (MCI). Psychotic symptoms, including delusions and hallucinations, occur in approximately 40-60% of individuals with AD, with an annual incidence of 10% among those with AD or MCI.1,2 A recent meta-analysis estimated the prevalence of hallucinations and delusions in MCI as 1.78% and 3.84%, respectively. 3 Psychosis in AD or MCI typically indicates a more severe disease phenotype, including: faster cognitive decline, further dementia progression, greater cortical synaptic impairments leading to disrupted neuronal communication, and several comorbidities such as depression and increased mortality. 1 Additionally, psychotic symptoms are associated with greater functional impairment, 4 represent a significant stressor to dementia caregivers, 5 and lead to earlier patient institutionalization. 6 Despite its prevalence and impact, psychosis in AD and MCI remains poorly understood, and reliable biomarkers to identify those at risk are lacking.
Among known genetic and demographic risk factors, female carriers of APOE4 are at the highest risk of developing AD. 7 Previous research from our group has also shown that female APOE4 homozygotes, both in neuropathological and clinical cohorts, are significantly more likely to develop AD-related psychosis than female non-carriers.8-10 Importantly, this increased risk was not found in males, suggesting a possible sex and genotype interaction that may exacerbate NPS vulnerability. Several hypotheses have been proposed to explain this elevated psychosis risk, including interactions between APOE4 and estrogen pathways, sex-specific aging processes such as menopause, increased vulnerability during periods of low estrogen, mechanisms related to Lewy body formation, or heightened immune activation.8,9,11 However, despite the importance of this interaction and its replication over several lines of research, the underlying biological mechanism remains unknown. 12
Given the limited understanding of the mechanisms linking sex, APOE4 status, and psychosis in AD and MCI, there is growing interest in exploring molecular biomarkers that may provide additional insight. One such candidate is epigenetic age acceleration (EAA), a biomarker derived from DNA methylation patterns that captures deviations from expected biological aging. 13 EAA is thought to serve as a proxy for cumulative biological stress and is influenced by a number of endogenous and exogenous factors. These include sex, immune cell composition, lifestyle behaviours (such as alcohol use and smoking), body mass index (BMI), socioeconomic status, education level, and the presence of chronic conditions like CVD, diabetes, cancer, or HIV. 13 To measure EAA, several epigenetic clocks with differing designs have been developed, which predict age or age-related phenotypes by combining the methylation values of tens to hundreds of CpGs. The Horvath clock is a pan-tissue model trained to predict chronological age based on DNA methylation levels at 353 CpG sites. 14 The Hannum clock is a single-tissue predictor trained on whole blood methylation profiles to estimate chronological age using 71 CpG sites. 15 In contrast, the PhenoAge clock is a second-generation model that was originally developed to better account for differences in risk and physiological status among individuals of the same chronological age. 16 While the Horvath and Hannum clocks produce epigenetic age estimates that correlate well with someone’s chronological age, the DNAm PhenoAge clock uses 513 CpGs to predict someone’s phenotypic age, a composite measure that incorporates chronological age and 9 clinical biomarkers: albumin, creatinine, serum glucose, C-reactive protein, lymphocyte percent, mean cell volume, red cell distribution width, alkaline phosphatase, and white blood cell count. 16 Therefore, the determination of epigenetic age for all 3 clocks is calculated based on DNA methylation data from different CpG sites, but was initially trained on different variables.
Using the Horvath clock, researchers have found that individuals who develop psychosis exhibit an EAA of approximately 2.8 years per chronological year, compared to non-converters to psychosis. 17 Additionally, both Horvath and PhenoAge EAA of the dorsolateral prefrontal cortex has been associated with greater amyloid burden, cognitive decline, and memory loss in AD.16,18 Interestingly, a large meta-analysis including over 30 studies from 2022 suggested that overall, current, first-generation epigenetic clocks (eg, Horvath and Hannum) are not clinically useful biomarkers of dementia or cognitive aging. 19 However, more recent research has found that second-generation, blood-based epigenetic clocks (eg, PhenoAge) can predict progression from cognitively normal aging to MCI or AD, as well as poorer longitudinal cognitive outcomes. 20 To date, no research has examined whether EAA is specifically associated with psychosis risk within AD or MCI populations.
Therefore, the present study aimed to examine whether EAA is associated with psychosis in individuals with AD or MCI, and whether this effect is moderated by sex and APOE4 carrier status. Given prior evidence linking female APOE4 homozygosity to elevated psychosis risk, we hypothesized that EAA would be associated with greater odds of psychosis, particularly among female APOE4 carriers. Additionally, we prioritized PhenoAge as our primary model of interest, including the Horvath and Hannum clocks for comparison to assess whether second-generation clocks that incorporate phenotypic aging markers are more sensitive to psychosis in those with AD or MCI.
Methods
Participants and Data Source
The sample population was collected from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database (https://adni.loni.usc.edu), a public-private partnership launched in 2003 by Principal Investigator Michael W. Weiner, MD. The goals of ADNI are to investigate clinical, imaging, genetic, and biomarker changes associated with the progression of normal aging to MCI and AD. Study participants were identified if they had a clinical diagnosis of AD or MCI (n = 1438), and had available genomic DNA methylation data, Mini-Mental State Examination (MMSE) scores, Neuropsychiatric Inventory Questionnaire (NPI-Q) scores, sex, date of birth, smoking status, and APOE genotype. Participants missing any of these data were excluded (n = 1020).
In the ADNI database, patients were divided into 3 cohorts (cognitively unimpaired, MCI, and AD) based on established diagnostic criteria that included clinical assessments such as the Clinical Dementia Rating, MMSE, the Wechsler Logical Memory II paragraph recall test (part of the neuropsychological battery), and clinical judgement. 21 As per the ADNI protocol, diagnoses at each site were confirmed by an experienced neurologist or psychiatrist, and patients were excluded if they had a significant neurologic disorder other than AD (eg, Parkinson’s disease, Hungtinton’s disease, multiple sclerosis, brain tumors, etc.), a lifetime history of schizophrenia, major depression or bipolar disorder within the past year, or alcohol abuse/dependence within the past 2 years. 21 Additionally, individuals with a history of significant head trauma or persistent neurological deficits, as well as those with structural brain abnormalities, were also excluded. APOE genotyping in ADNI-1 was performed using polymerase chain reaction (PCR) amplification with HhaI restriction enzyme digestion, while later iterations used allele-specific PCR, with all data undergoing manual quality control before submission to the ADNI Genetics Core. 22
For the purpose of this study, psychosis status was defined as a binary outcome (psychosis present vs no psychosis), determined by a positive score (>0) for either hallucinations or delusions on the NPI-Q at one or more visits. The NPI-Q, a well established tool for determining psychosis status,8,23 has been validated against the full NPI,24,25 which reported reliable sensitivity (0.826), specificity (0.917), positive predictive value (0.864), and negative predictive value (0.892) in determining psychosis. 26 To enhance statistical power and better reflect the continuum of cognitive impairment across which psychosis can emerge, participants with AD and MCI were analyzed together in all models. 27 Because our primary objective was to identify factors differentiating AD and MCI patients with vs without psychosis, cognitively impaired participants without psychosis were considered our reference group rather than including cognitively healthy controls. This approach isolates within-diagnosis variability in psychosis risk (our main question of interest), rather than replicating known global differences in epigenetic age between AD and healthy aging. 20
Epigenetic Age Calculations
Illumina Infinium HumanMethylationEPIC BeadChip data were downloaded and analyzed for all eligible participants. To standardize time point selection and reduce potential confounding from disease progression, the earliest available blood-based methylation sample was used for each participant. Raw intensity data (.IDAT) files were processed using the minfi R package with standard normalization and quality control procedures. 28 Beta values, which represent the proportion of DNA methylation at individual CpG sites (ranging from 0 to 1), were extracted and used to calculate epigenetic age (DNAm age) estimates using the methylclock R package. 29 Three clocks were applied: Horvath (trained on multiple tissues, 353 CpG sites), Hannum (trained on adult blood, 71 CpG sites), and PhenoAge (trained on peripheral blood to predict phenotypic age, 513 CpG sites). We selected these 3 clocks as they are the most widely validated and commonly used measures of DNAm age across clinical research. To our knowledge, no prior studies have examined epigenetic aging in the context of psychosis in AD or MCI, so our goal was to begin with the most established and reproducible clocks to provide a strong foundation for future work.
Chronological age was calculated by subtracting each participant’s date of birth from the date of their methylation blood draw. Since only the month and year of birth were available, the 15th day of each month was used as an approximate birthday. EAA was defined as the linear residual from regressing each measure of epigenetic age on chronological age.
Statistical Analyses
Raw methylation data were processed in R (version 4.5.0), while all subsequent statistical analyses were performed in Python (version 3.10). Group differences in demographic variables between participants with and without psychosis were assessed using independent samples t-tests, Mann-Whitney U tests, and chi-squared tests, as appropriate. Using the Pearson method, we assessed the correlation between epigenetic age and chronological age for all 3 clocks. To account for participant-level variability in timing of the methylation blood draw relative to psychosis onset, descriptive statistics were used to summarize the timing of methylation sample collection relative to psychosis onset.
Logistic regression models were used to assess associations between EAA as a continuous independent variable (predictor) and psychosis status (dichotomized outcome). Separate main-effects models were run for each epigenetic clock. Covariates included sex, APOE4 carrier status (defined as the presence of at least one ε4 allele; includes both APOE4 heterozygotes and homozygotes), MMSE score, and smoking status. Odds ratios (ORs) were reported per 5-year acceleration in epigenetic age to improve interpretability, consistent with prior studies.
To test the hypothesis that female APOE4 carriers have different associations with EAA, a second model was run for each clock testing a three-way interaction term: Sex * APOE4 carrier status * EAA. All lower-order interactions and main effect terms were included (see Supplemental Information), and statistical significance was set at P < 0.05. When a significant interaction was identified, post-hoc analyses were performed to assess the association between EAA and psychosis within each sex and APOE4 genotype subgroup. ORs and 95% confidence intervals (CIs) were calculated by exponentiating the beta coefficient.
Multiple correction comparisons were not applied across epigenetic clocks, as PhenoAge was selected a priori as the primary model of interest based on prior evidence of its increased performance relative to first generation epigenetic clocks. Similarly, subgroup analyses were hypothesis-driven, with a focus on female APOE4 carriers given previous research identifying this subgroup as specifically vulnerable to psychosis in AD.
Results
Baseline Population Demographics and Epigenetic Age Measures by Psychosis Status.
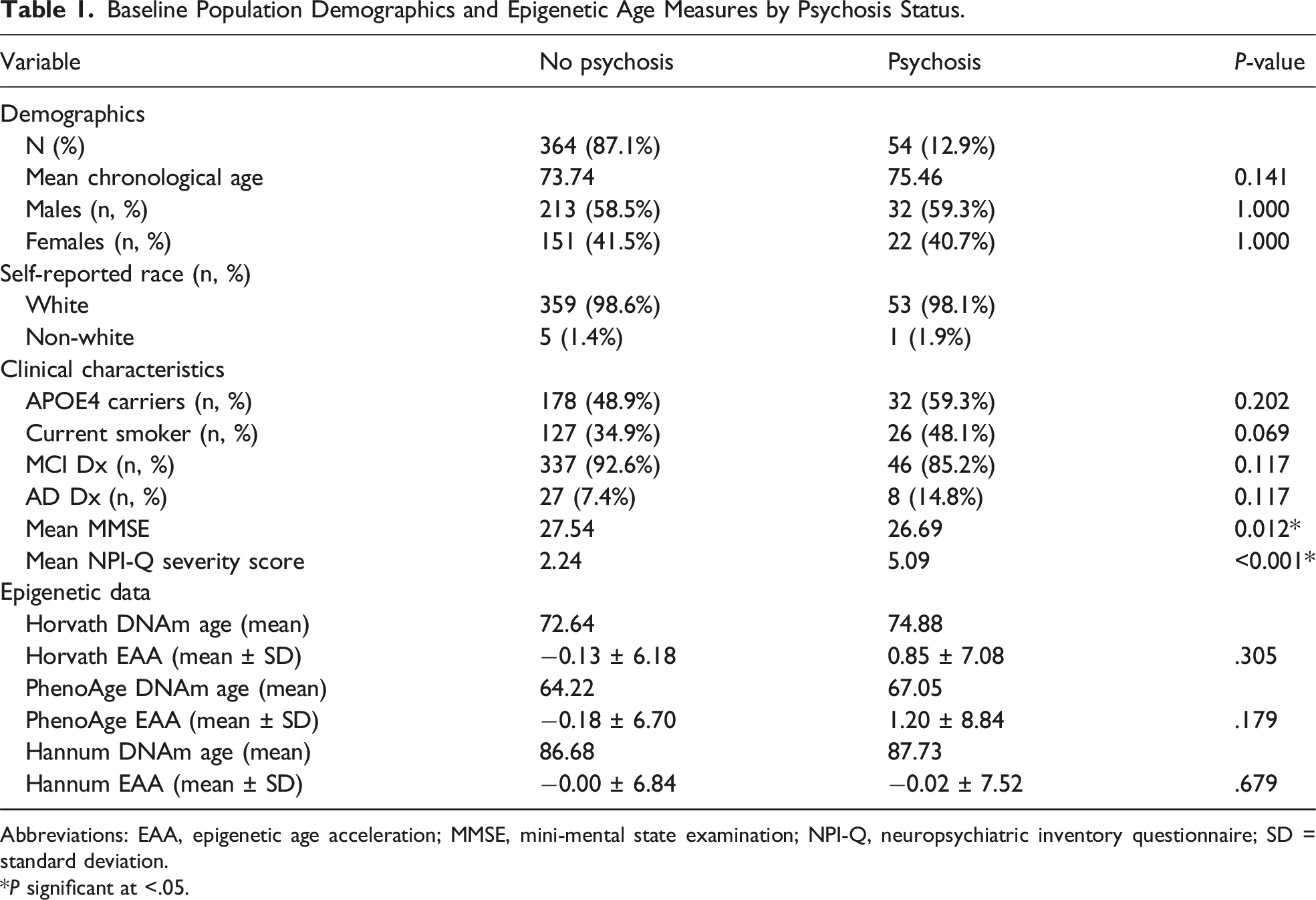
Abbreviations: EAA, epigenetic age acceleration; MMSE, mini-mental state examination; NPI-Q, neuropsychiatric inventory questionnaire; SD = standard deviation.
*P significant at <.05.
Using the Pearson method, epigenetic age across all 3 clocks was significantly and moderately correlated with chronological age: Horvath (r (416) = .68, t = 19.02), PhenoAge (r (416) = .69, t = 19.68), and Hannum (r (416) = .58, t = 14.60); all P < .001.
Mean EAA did not differ significantly between the psychosis and non-psychosis group for any clocks, as determined by independent samples t-tests or non-parametric alternatives when the assumption of normality was not met (Table 1). The largest observed difference was in the PhenoAge clock, where individuals with psychosis had a mean EAA of +1.20 years (SD: 8.84) compared to −0.18 years (SD: 6.70) in the non-psychosis group, though this difference was non-significant (t (416), P = .179).
Associations Between EAA and Psychosis: Main Effects and Interaction Effects Models Across Three Clocks.

Abbreviations: EAA, Epigenetic Age Acceleration, MMSE, Mini-Mental State Examination. All regression coefficients were evaluated using Wald z-tests (df = 416). The main effect of DNAm age acceleration is derived from a main effects-only model. The 3-way interaction term is derived from the full interaction model, with all main effects and lower order interactions included. The reference group are male APOE4 non-carriers. Odds ratios reported per 5 year increases in EAA.
*P significant at <.05.
Subgroup Analysis of Psychosis Odds per 5-Year Increase in PhenoAge EAA.
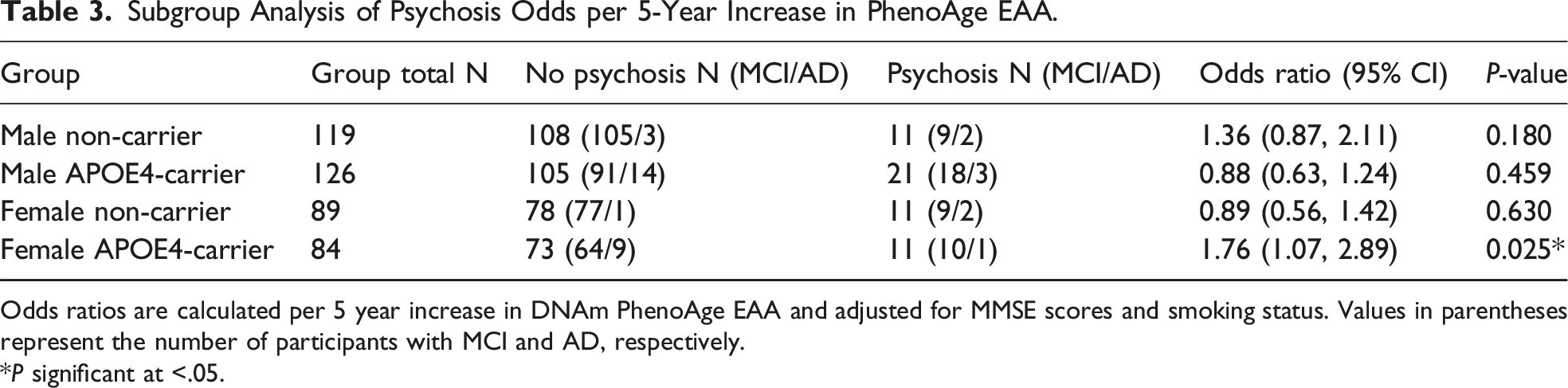
Odds ratios are calculated per 5 year increase in DNAm PhenoAge EAA and adjusted for MMSE scores and smoking status. Values in parentheses represent the number of participants with MCI and AD, respectively.
*P significant at <.05.
Notably, higher cognitive status, as measured by the MMSE, was a consistent protective factor for psychosis development across all clocks in both main and interaction models (OR Range: 0.85-0.86, all P < .05). Smoking status was not significantly associated with psychosis across any models, though it trended towards significance in many of them (OR Range: 1.64-1.74, P-value range: 0.062 - 0.099). Full model outputs, including covariates, main effects, and all lower-order interaction terms, are available in the Supplemental Information.
Discussion
Our preliminary investigation suggests a potential association between PhenoAge EAA and psychosis among female APOE4 carriers with MCI and AD. Importantly, this association was not found across male subgroups or female non-APOE4 carriers. Among female APOE4 carriers, our findings suggest that epigenetic age measured from peripheral blood may contribute to psychosis risk independent of chronological age or cognitive status, although causality cannot be inferred. Additionally, this significant three-way interaction suggests that the relationship between biological aging and psychosis is influenced by both sex and genetic risk, aligning with prior work identifying that female APOE4 carriers are a biologically vulnerable subgroup for AD-related psychosis, whereas sex and APOE4 independently do not independently contribute to psychosis risk.8-10 In contrast, no significant associations were observed for the Horvath or Hannum clocks, highlighting the potential specificity of second-generation epigenetic clocks in capturing AD-specific aging processes. Additionally, higher MMSE scores (covariate) were consistently associated with lower odds of psychosis across all models, aligning with previous research suggesting that patients with more advanced cognitive decline are at increased risk for psychosis. 1
The specificity of our findings to the PhenoAge clock, with no significant findings for the Horvath and Hannum clocks, likely reflects differences in what each clock captures. Out of the 513 CpGs incorporated into the DNAm PhenoAge model, only 41 CpGs overlapped with the Horvath clock, 6 CpGs with the Hannum clock, and 5 CpGs among all 3 clocks.16,20 Moreover, while the Horvath and Hannum clocks estimate chronological age based on general age-related methylation patterns, PhenoAge incorporates CpGs linked to clinical markers of physiological decline, better reflecting functional aging across tissues and organs.30,31 Moreover, the PhenoAge EAA model is particularly relevant for understanding psychosis risk because its biomarkers directly reflect biological processes, such as inflammation, metabolic dysfunction, and oxidative stress, that are known to contribute to neurodegeneration and psychosis vulnerability. Indeed, our findings align with recent research suggesting that PhenoAge outperforms earlier DNAm clocks in predicting disease-related morbidity, healthspan, physical function, and specific indicators of cognitive impairment and AD.16,20 Future research should continue to examine whether second-generation epigenetic clocks like PhenoAge consistently provide greater sensitivity to disease-related morbidity in AD and MCI.
The elevated risk for AD-related psychosis among female APOE4 carriers has already been well established, yet the biological mechanisms remain unclear. 8 Based on our findings, we hypothesize that elevated PhenoAge EAA may reflect a metabolically dysregulated and pro-inflammatory state that could contribute to psychosis vulnerability in already at-risk subgroups. Of the 9 clinical biomarkers that make up the PhenoAge model, red cell distribution width (RDW), serum glucose, and C-reactive protein are the most heavily weighted. 16 RDW, which captures variability in erythrocyte size and volume, has been linked to elevated levels of oxidative stress and neuroinflammatory markers like IL-6.32,33 Similarly, hyperglycemia exacerbates neuroinflammation, oxidative damage, amyloid beta accumulation, and vascular dysfunction, particularly in already inflammatory conditions.34,35 C-reactive protein, an acute pro-inflammatory marker, can penetrate the blood-brain barrier at high doses, inducing reactive gliosis and impairing CNS function. 36 Together, these biomarkers may point to a systemic inflammatory state that can contribute to brain dysfunction.
This phenotype is mirrored at the molecular level, where individuals with accelerated DNAm PhenoAge show upregulation of pro-inflammatory signalling pathways, including NF-κB, the JAK-STAT cascade, and toll-like receptor (TLR) signaling. 16 NF-κB, the master regulator of inflammation, is upregulated by microglia and astrocytes to promote sustained cytokine production and neuroinflammation. 37 These combined systemic and molecular disruptions may promote processes such as microglial activation, dopaminergic dysregulation, and synaptic dysfunction – mechanisms that have been implicated in psychosis in MCI, AD, and other psychiatric conditions.38,39 Female APOE4 carriers may be especially vulnerable due to APOE4 driven increases in cytokine expression, and the loss of estrogen’s anti-inflammatory effects post-menopause.40,41 Indeed, research from Wang et al suggest that APOE4 exerts stronger neuroinflammatory effects in aging females than in males. 42 Additionally, there is evidence that women experience accelerated epigenetic aging during and after menopause, potentially compounding inflammation-related vulnerability. 43 While neuroinflammation represents a plausible mechanistic pathway, future research should further investigate the CpGs and molecular features captured by PhenoAge to clarify its role in AD-related psychosis.
Our findings have potential clinical implications. Currently, there are few reliable biomarkers and no targeted interventions for psychosis in those with cognitive impairment, largely due to a lack of a clear mechanistic understanding of its development. While some tau mechanisms have been implicated for psychosis in later life, 27 traditional AD biomarkers like amyloid plaques are not as strongly associated with psychosis, and are often expensive (eg, PET scan) or invasive (eg, lumbar puncture) to measure.20,44 Peripheral blood PhenoAge EAA may represent a feasible and accessible candidate biomarker for psychosis in MCI and AD, pending replication in larger, independent cohorts. Our findings also offer preliminary insight into potential biological pathways associated with psychosis, such as neuroinflammation and metabolic dysfunction, which could serve as targets for future investigation of therapeutic interventions. This is especially important given that traditional antipsychotics show limited efficacy in MCI and dementia-related psychosis, and are generally avoided due to significant risk of adverse events among older populations.45,46
Given that PhenoAge captures several modifiable biological processes, it is also worth investigating whether DNAm age itself can be reversed. For example, a recent RCT demonstrated that targeted diet and lifestyle interventions (including supplemental nutrition, an exercise and stress management program, and a sleep prescription of more than 7 hours a night) could reverse epigenetic age, as measured by the Horvath clock. 47 Similarly, a phase one human clinical trial by Fahy et al found that a combination therapy of human growth hormone, DHEA, and metformin significantly reduced epigenetic age across all 4 clocks tested: Horvath, Hannum, PhenoAge, and GrimAge. 48 These findings suggest that epigenetic aging is modifiable, raising the possibility that interventions targeting PhenoAge could mitigate psychosis risk among female APOE4 carriers. Future trials should expand upon and investigate other targeted lifestyle interventions (eg, smoking cessation) to investigate whether PhenoAge EAA can be reversed and reduce psychosis risk in female APOE4 carriers. Additionally, longitudinal cohort studies could track changes in epigenetic age and lifestyle factors over time in high-risk MCI or AD populations who may develop psychosis.
Overall, the observed effects were moderate to large in magnitude. Among female APOE4 carriers, a 5-year increase in PhenoAge EAA was associated with approximately 1.7-fold higher odds of psychosis, and a 3-fold increase relative to other subgroups. However, while these effect sizes are notable within the context of biomarker studies in Alzheimer’s disease, they should be interpreted as preliminary and clinically suggestive rather than definitive, given wider confidence intervals and relatively smaller subgroups.
Limitations and Conclusion
While our results are of interest, this study had several expected limitations, each of which points to clear directions for future research. First, the timing of psychosis onset relative to methylation blood draw varied across our sample. To minimize this variation, we used the earliest available EAA measurement for each participant and determined the time interval between methylation blood draw and symptom onset. Notably, the median interval indicated that for most participants, EAA preceded the onset of psychosis (5.98 months prior). However, this was not the case for all participants, meaning we cannot establish strict temporal or causal relationships. Interestingly, Iftimovici et al followed an ultra–high-risk group for schizophrenia over time and found that converters to psychosis initially exhibited epigenetic age deceleration, followed by rapid acceleration during the transition period. 17 Future research should adopt prospective designs with repeated methylation sampling to better clarify the timing and directionality of EAA changes in relation to psychosis development in AD and MCI.
Second, the decision to combine MCI and AD participants increased our study’s statistical power but introduced some clinical heterogeneity, especially with a relatively smaller proportion of AD participants in our sample. Considering psychosis is much more prevalent in AD than MCI, it is possible that associations between EAA and psychosis may differ based on diagnosis type and severity of disease. 49 This limitation was also present in our subgroup analyses, where lower sample sizes across sex and genotype groups with psychosis limited statistical power and prevented separate analyses of APOE4 homozygotes vs heterozygotes. Moreover, given that psychosis is a secondary phenotype in AD and MCI, its occurrence may vary within these populations, complicating the detection of clear associations. Future research should therefore explore these differences with larger sample sizes and specific diagnostic cohorts (eg, AD-only cohort).
Third, our AD subsample was diagnosed based on clinical data and not neuropathologically confirmed, and psychosis status was determined via NPI-Q scores and not formal criteria. 50 Consensus from the International Psychogeriatric Association defines psychosis as the presence of hallucinations (visual or auditory) or delusions that persist for at least one month, are severe enough to impair daily functioning, and are not better explained by other medical conditions like schizophrenia or delirium. 51 Better verification of AD and psychosis status would therefore improve the validity of the study’s methodology. Another limitation is the relatively smaller number of individuals with psychosis compared to the non-psychosis group, which may exaggerate the effect of variables like sex, APOE4 carrier status, and MMSE in this cohort. Furthermore, the ADNI cohort is quite homogeneous and lacks diversity, thus limiting the generalizability of our findings. Finally, although the observed associations were specific to PhenoAge, the biological mechanisms involved in this link remain correlational and require further investigation through experimental and longitudinal studies.
In conclusion, our study identified a significant three-way interaction between PhenoAge EAA, sex, and APOE4 carrier status. Specifically, EAA was associated with significantly higher odds of psychosis in female APOE4 carriers, with the strength of this association being roughly 3 times greater relative to male non-carriers. Supporting this, subgroup analyses indicated that female APOE4 carriers had a significant association between EAA and psychosis (OR = 1.76 per 5-year increase, P = 0.025), whereas no significant associations were observed in any other sex or genotype combinations. Our study provides preliminary evidence that second-generation epigenetic clocks may capture aspects of neuropsychiatric vulnerability beyond traditional AD biomarkers. Moreover, they offer valuable insight into the moderating physiological factors that may be associated with psychosis risk in AD and MCI, paving the way for future mechanistic and intervention-focused research.
Supplemental Material
Supplemental material - Peripheral Blood Epigenetic age Acceleration is Associated With Psychosis in Female APOE4 Carriers With Mild Cognitive Impairment and Alzheimer’s Disease: A Pilot Study
Supplemental material for Peripheral Blood Epigenetic age Acceleration is Associated With Psychosis in Female APOE4 Carriers With Mild Cognitive Impairment and Alzheimer’s Disease: A Pilot Study by Dylan Kwan, BHSc, Neda Rashidi-Ranjbar, MD, PhD, Joseph E. Marinas, MD, Andrew Namasivayam, MD, Nathan Churchill, PhD, Luis R. Fornazzari, MD, Zahinoor Ismail, MD, Vincenzo DeLuca, MD, PhD, Tom A. Schweizer, PhD, David G. Munoz, MD, Corinne E. Fischer, MD, and For the Alzheimer’s Disease Neuroimaging Initiative in Journal of Geriatric Psychiatry and Neurology.
Footnotes
Acknowledgements
ADNI funding: Data collection and sharing for the Alzheimer’s Disease Neuroimaging Initiative (ADNI) is funded by the National Institute on Aging (National Institutes of Health Grant U19AG024904). The grantee organization is the Northern California Institute for Research and Education. In the past, ADNI has also received funding from the National Institute of Biomedical Imaging and Bioengineering, the Canadian Institutes of Health Research, and private sector contributions through the Foundation for the National Institutes of Health (FNIH) including generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc; Biogen; Bristol Myers Squibb Company; CereSpir, Inc; Cogstate; Eisai Inc; Elan Pharmaceuticals, Inc; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc; Fujirebio; GE Healthcare; IXICO Ltd; Janssen Alzheimer Immunotherapy Research & Development, LLC; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co, Inc; Meso Scale Diagnostics, LLC; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics.
Author Contributions
Dylan Kwan: Writing – original draft, Methodology, Investigation, Conceptualization, Data Curation, Formal Analysis. Neda Rashidi-Ranjbar: Writing – review & editing, Conceptualization, Methodology, Formal Analysis. Joseph E. Marinas: Writing – review & editing, Conceptualization. Andrew Namasivayam: Writing – review & editing, Conceptualization. Nathan Churchill: Writing – review & editing, Conceptualization, Methodology. Luis R. Fornazzari: Writing – review & editing, Conceptualization, Methodology. Zahinoor Ismail: Writing – review & editing, Conceptualization, Methodology. Vincenzo DeLuca: Writing – review & editing, Conceptualization, Methodology. Tom A. Schweizer: Writing – review & editing, Conceptualization, Methodology. David G. Munoz: Writing – review & editing, Conceptualization, Methodology. Corinne E. Fischer: Supervision, Methodology, Conceptualization, Investigation, Writing – review & editing.
Declaration of Conflicting Interests
Corinne E. Fischer reports grant support from Cortexyme, Brain Canada, Vielight Inc, Hoffman La Roche, Novo Nordisk, NIH/NIA, CIHR/CCNA, the Hilary and Galen Weston Foundation, Mito2i, and the Temerty-Tanz Centre for unrelated work. Corinne E. Fischer has received honoraria for lectures from the Icahn School of Medicine and the Care of the Elderly Course. Corinne E. Fischer served as Chair of the ISTAART NPS PIA (2018–2020) and the IPA Psychosis Working Group. Zahinoor Ismail reports receiving funding grants from NIA, CIHR, CCNA, Brain Canada, ADDF, Weston Foundation, Gordie Howe CARES. Zahinoor Ismail reports receiving consulting fees from Otsuka/Lundbeck, Roche, Novo Nordisk, Eisai, Eli Lilly. Zahinoor Ismail reports participation on a data safety monitoring board for the OCEANS study Johns Hopkins and BioXcel BXCL501. All other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data collection and sharing for the Alzheimer’s Disease Neuroimaging Initiative (ADNI) is funded by the National Institute on Aging (National Institutes of Health Grant U19AG024904). The authors wish to acknowledge the St. Michaels Hospital Foundation Heather and Eric Donnelly endowment for their support.
Supplemental Material
Supplemental material is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
