Abstract
Purpose
Agitation is a neuropsychiatric symptom commonly observed in Alzheimer’s dementia, which causes substantial burden for patients and caregivers. In this exploratory analysis, Neuropsychiatric Inventory (NPI) data were pooled from two randomized, double-blind, fixed-dose, placebo-controlled trials of brexpiprazole in agitation associated with dementia due to Alzheimer’s disease. The analysis explored the efficacy of brexpiprazole on agitation and other neuropsychiatric symptoms, including their impact on caregivers.
Major Findings
Brexpiprazole 2 or 3 mg/day (N = 368) showed greater improvement vs placebo (N = 253) from baseline in NPI agitation/aggression domain score (Week 6 onwards), and in the associated occupational disruptiveness/caregiver distress score (Week 4 onwards). Improvements with brexpiprazole vs placebo also occurred on other NPI and occupational disruptiveness/caregiver distress scores, including irritability/lability, aberrant motor behavior, and anxiety.
Conclusions
Over 12 weeks, brexpiprazole was associated with greater improvements vs placebo in agitation/aggression and other neuropsychiatric symptoms, and in associated caregiver disruption/distress. ClinicalTrials.gov identifiers: NCT01862640; NCT03548584.
Plain Language Summary
Alzheimer’s dementia is a common disorder that affects the brain. In addition to problems with thinking and memory, many patients also exhibit agitation. Agitation is a neuropsychiatric symptom (a term for symptoms that affect behaviors and emotions), which are common in Alzheimer’s dementia. There are many types of agitation symptoms, which can be non-aggressive (like wandering or feeling restless) or aggressive (like cursing, shouting, or hitting). Agitation symptoms can cause caregivers to feel anxious and distressed and often lead to patients being moved into nursing homes. A medication called brexpiprazole is approved by the United States FDA to treat agitation symptoms in people with Alzheimer’s dementia. The brexpiprazole clinical trials included an assessment called the Neuropsychiatric Inventory (NPI), which is filled out by doctors based on interviews with caregivers. The NPI measures patients’ neuropsychiatric symptoms (including agitation symptoms) and assesses how caregivers are affected by these symptoms. This article describes an analysis of NPI results from two brexpiprazole clinical trials over 12 weeks. In this analysis, larger improvements were seen with brexpiprazole than with a placebo (dummy drug) in agitation/aggression, as well as in other neuropsychiatric symptoms, including irritability/lability (eg, bad temper or changeable mood), aberrant motor behavior (eg, repetitive habits or fidgeting), disinhibition (eg, acting impulsively or insensitively), and anxiety (eg, feeling nervous or worried). These improvements were accompanied by a reduced impact of the symptoms on caregivers.
Introduction
Alzheimer’s dementia is a highly prevalent disease, which is estimated to affect around 6 million people in the United States and Canada. 1 In addition to cognitive impairment, more than 90% of patients with Alzheimer’s dementia experience neuropsychiatric symptoms, which can include agitation, psychosis, apathy, depression, and sleep disturbances.2-5 Agitation is among the most common neuropsychiatric symptoms, and is present in approximately 50% of patients with Alzheimer’s dementia, in both community settings and nursing homes.6,7 Agitation can manifest as non-aggressive symptoms (eg, repetitive sentences or questions, inappropriate dressing or disrobing, wandering, or restlessness) and aggressive symptoms (eg, cursing or verbal aggression, spitting, hitting, grabbing, or throwing things).8-10
Agitation is associated with a substantial burden for patients and for their caregivers. For patients, agitation symptoms are associated with faster progression of Alzheimer’s dementia, poorer functioning and quality of life, a higher likelihood of being transferred to long-term care, and earlier death.11-13 For caregivers, many experience psychological distress or anxiety, or compromise of their work or daily activities as a result of caring for someone with agitation symptoms in Alzheimer’s dementia.9,14 The caregiver burden and distress associated with agitation and other neuropsychiatric symptoms may result in caregiver burnout.15,16 In the United States, unpaid caregivers of people with agitation symptoms spend an average of 39.1 h per week caring for their loved ones, often in addition to regular employment commitments. 9 As such, caregiver distress is frequently the driving factor for decisions to transfer patients to long-term care settings. 17
The emergence of agitation symptoms in Alzheimer’s dementia is believed to result from dysfunction of the noradrenergic, serotonergic, and dopaminergic neurotransmitter systems, affecting the balance between executive control and emotional drive. 18 Brexpiprazole (2 or 3 mg/day), an atypical antipsychotic with effects on these neurotransmitter systems, 19 is approved in the United States for the treatment of agitation associated with dementia due to Alzheimer’s disease 20 – the first FDA-approved medication for this indication. Brexpiprazole has been approved for similar indications in several other countries. The efficacy and safety of brexpiprazole in this patient population was investigated in a multi-trial Phase 3 clinical program, in which improvements in agitation vs placebo were observed over 12 weeks, and brexpiprazole was generally well tolerated.21-23
While the primary focus of the Phase 3 brexpiprazole trials was agitation (as assessed by the Cohen-Mansfield Agitation Inventory [CMAI] – reported elsewhere),21,22 the trials also utilized the Neuropsychiatric Inventory (NPI) to assess a broad range of neuropsychiatric symptoms and their impact on caregivers. The aim of this exploratory post hoc analysis of pooled NPI data from two Phase 3 trials was to explore the efficacy of brexpiprazole 2 or 3 mg/day on agitation and other neuropsychiatric symptoms, as well as their impact on caregivers, in patients with agitation associated with dementia due to Alzheimer’s disease.
Methods
Trial Designs and Participants
Data were analyzed from two 12-week, Phase 3, randomized, double-blind, placebo-controlled trials of fixed-dose brexpiprazole in participants with agitation associated with dementia due to Alzheimer’s disease, conducted in the United States, Europe, and Russia: Trial 283 (ClinicalTrials.gov identifier: NCT01862640) 21 and Trial 213 (NCT03548584). 22 Another placebo-controlled trial of brexpiprazole (Trial 284; NCT01922258) 21 was also conducted, but is not included in the present analysis because: (1) Trial 284 was not part of the submission to the FDA for the indication of brexpiprazole for agitation associated with dementia due to Alzheimer’s disease 20 ; (2) Trial 284 was flexible-dose rather than fixed-dose (a design differing from the other trials), and (3) Trial 284 included patients who were on lower doses of brexpiprazole (target dose: 1 mg/day). The trials were conducted in accordance with the principles stated in the Declaration of Helsinki, the International Conference on Harmonisation Good Clinical Practice Guideline, and local regulatory requirements, and relevant institutional review boards or independent ethics committees approved the original trial protocols. All participants and/or their legal representatives provided written or electronic informed consent prior to the start of the trials.
The two analyzed trials were similar in design, as described previously.21,22 In brief, participants were aged 55-90 years; had a diagnosis of probable Alzheimer’s disease by the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA) criteria 24 ; were living in a care facility or community-based setting (transfer between care settings was not permitted during the trials); had a Mini-Mental State Examination score of 5-22 at screening and baseline 25 ; had an NPI or NPI Nursing Home version (NPI-NH) agitation/aggression domain score ≥4 at screening and baseline26-28; and had a caregiver with sufficient contact to describe the participant’s symptoms and behaviors (minimum level of contact: 2 h per day, for 4 days per week). In Trial 213, participants were also required to meet the International Psychogeriatric Association (IPA) definition of agitation, 8 and to meet a criterion based on CMAI Factor 1 (Aggressive agitation),29,30 at screening and baseline. In both trials, key exclusion criteria included dementia or memory impairment due to reasons other than Alzheimer’s dementia, and diagnosis of an Axis I disorder (per Diagnostic and Statistical Manual of Mental Disorders, 4th edition, text revision [Trial 283] or 5th edition [Trial 213] criteria)31,32 including schizophrenia or other psychotic disorder, or bipolar disorder. Current major depressive disorder (MDD) was permitted if asymptomatic (ie, history of MDD, but not a current major depressive episode; Trial 213 only) or if the participant was on a stable dose of antidepressant. Participants with other specified clinically significant comorbidities were also excluded.
Following a period of screening and washout of prohibited medications (up to 42 days), participants were randomized to 12 weeks of double-blind treatment. In Trial 283, participants were randomized 1:1:1 to brexpiprazole 1 mg/day, brexpiprazole 2 mg/day, or placebo, titrated over 4 weeks. In Trial 213, participants were randomized 2:1 to brexpiprazole 2 or 3 mg/day (analyzed as a single arm) or placebo, titrated over 2-4 weeks depending on the dose.
The primary endpoint of each trial was the drug–placebo difference in change from baseline to Week 12 in CMAI Total score, on which greater reductions in agitation frequency were observed with brexpiprazole 2 mg/day and brexpiprazole 3 mg/day vs placebo (results described previously).21,22 The NPI (or the NPI-NH for institutionalized participants) was included as an exploratory (Trial 213) and/or “other” (Trial 283) outcome measure; the NPI is described below.
Assessment of Neuropsychiatric Symptoms and Impact on Caregivers
Overview of the NPI. 28

NPI, Neuropsychiatric Inventory.
Separately, the NPI evaluates the impact of neuropsychiatric symptoms on caregivers, for each of the 12 domains. For institutionalized patients (assessed using the NPI-NH), the caregiver element assesses the impact on professional caregivers and is described as ‘occupational disruptiveness’. For non-institutionalized patients, the caregiver element is described as ‘caregiver distress’. If a symptom is present, occupational disruptiveness/caregiver distress is scored for the corresponding domain (0 [not at all]; 1 [minimally]; 2 [mildly]; 3 [moderately]; 4 [severely]; 5 [very severely or extremely]). If a symptom is absent, occupational disruptiveness/caregiver distress is not evaluated. The NPI occupational disruptiveness/caregiver distress total score is the sum of the 12 caregiver-specific domain scores, with a possible score range of 0 (best) to 60 (worst).
Data Analysis
In this post hoc analysis, data were pooled from Trial 283 and Trial 213 for patients who received fixed-dose brexpiprazole 2 or 3 mg/day (ie, the US FDA-approved therapeutic doses),
20
or placebo. This analysis focused on NPI outcomes from Trials 283 and 213: (1) NPI total score – reflecting the composite of neuropsychiatric symptoms that are present (2) NPI agitation/aggression domain score – reflecting the key neuropsychiatric symptom of interest in Trials 283 and 213 (3) other NPI domain scores – reflecting other neuropsychiatric symptoms (4) NPI occupational disruptiveness/caregiver distress total score – reflecting the overall impact of neuropsychiatric symptoms on caregivers (5) NPI occupational disruptiveness/caregiver distress score in the agitation/aggression domain – reflecting, specifically, the impact of agitation/aggression symptoms on caregivers (6) NPI occupational disruptiveness/caregiver distress scores in other domains – reflecting the impact of other neuropsychiatric symptoms on caregivers.
Efficacy was analyzed in randomized participants who received at least one dose of trial medication and who had a baseline and at least one post-baseline evaluation for the outcome in question. Baseline was defined as the randomization visit. Caregiver outcomes were analyzed for caregivers of participants in the efficacy sample. Least squares (LS) mean change from baseline for brexpiprazole vs placebo was analyzed for each NPI outcome using a mixed model for repeated measures (MMRM) approach on observed cases data, with fixed effects of trial protocol, treatment, and visit week, an interaction term of treatment by visit week, and the interaction term of baseline score by visit week. The analysis was jointly modeled using data from Weeks 0, 2, 4, 6, 8, 10, and 12 for Trial 283, and Weeks 0, 6, and 12 for Trial 213. Due to imbalanced data across time, the default unstructured covariance matrix in the MMRM occasionally failed to converge. As a result, alternative structured covariance matrices with greater model restriction were sequentially explored (in alignment with the pre-defined order used in the primary efficacy analyses of Trial 283 and Trial 213). As an exploratory, hypothesis-generating analysis, all P-values were tested at a nominal 0.05 level, with no adjustment for multiplicity. Finally, Pearson correlations were calculated between changes from baseline in NPI agitation/aggression domain score and the corresponding occupational disruptiveness/caregiver distress score.
Analyses were performed using SAS version 9.4 (SAS Institute Inc; Cary, NC).
Results
Participants
In the trials, 621 participants were randomized to brexpiprazole 2 or 3 mg (N = 368) or placebo (N = 253). Trial completion rates were 320/368 (87.0%) for brexpiprazole 2 or 3 mg and 225/253 (88.9%) for placebo. Full disposition details have been published for a pooled Phase 3 trial sample. 23 In the current post hoc analysis, data from 610 participants were analyzed (brexpiprazole 2 or 3 mg, n = 363; placebo, n = 247).
Participant baseline demographic and clinical characteristics have been previously published for the individual trials21,22 and for a pooled Phase 3 trial sample, 23 and were generally similar between participants receiving brexpiprazole 2 or 3 mg and placebo. In the present pooled sample, 50.0% and 56.7% of participants in the brexpiprazole group and placebo group (respectively) were living in a care facility (and were assessed using the NPI-NH); 50.0% and 43.3% were living in community-based settings (and were assessed using the NPI).
NPI Scores at Baseline.
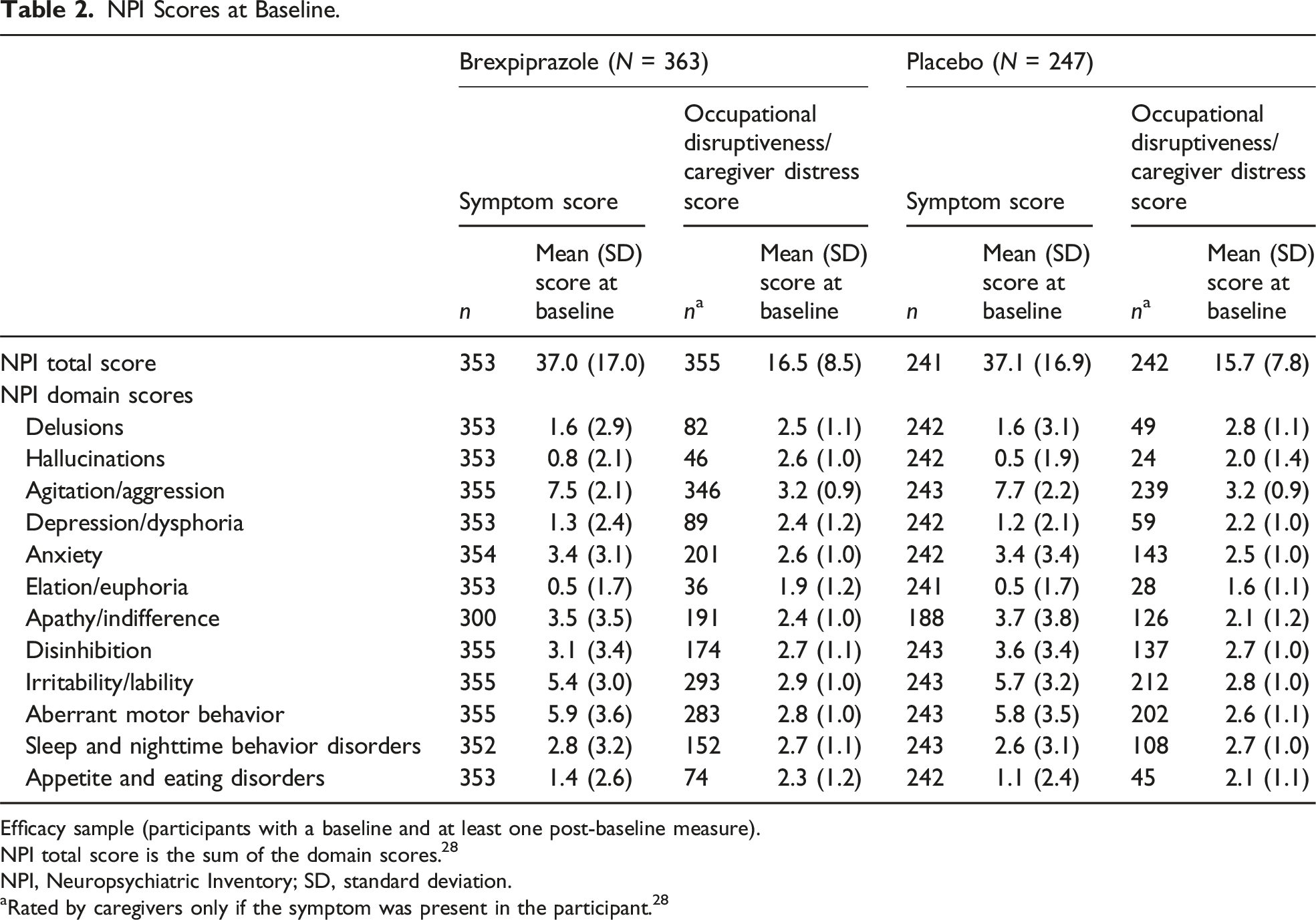
Efficacy sample (participants with a baseline and at least one post-baseline measure).
NPI total score is the sum of the domain scores. 28
NPI, Neuropsychiatric Inventory; SD, standard deviation.
aRated by caregivers only if the symptom was present in the participant. 28
Changes in Neuropsychiatric Symptoms
Over 12 weeks, greater improvement in the composite score of neuropsychiatric symptoms (which includes the key neuropsychiatric symptom of agitation/aggression) was observed with brexpiprazole (LS mean [standard error (SE)] change in NPI total score: −17.6 [0.7]) vs placebo (−14.3 [0.7]) (Figure 1). The LS mean difference between treatment groups at Week 12 was −3.31 (95% confidence interval [CI]: −5.09, −1.52; P < .001). Improvements vs placebo (P < .01) were observed at Weeks 6, 10 and 12 (Figure 1). Change from baseline in NPI total score. **P < .01, ***P < .001 vs placebo; MMRM, efficacy sample. LS, least squares; MMRM, mixed model for repeated measures; NPI, Neuropsychiatric Inventory; SE, standard error.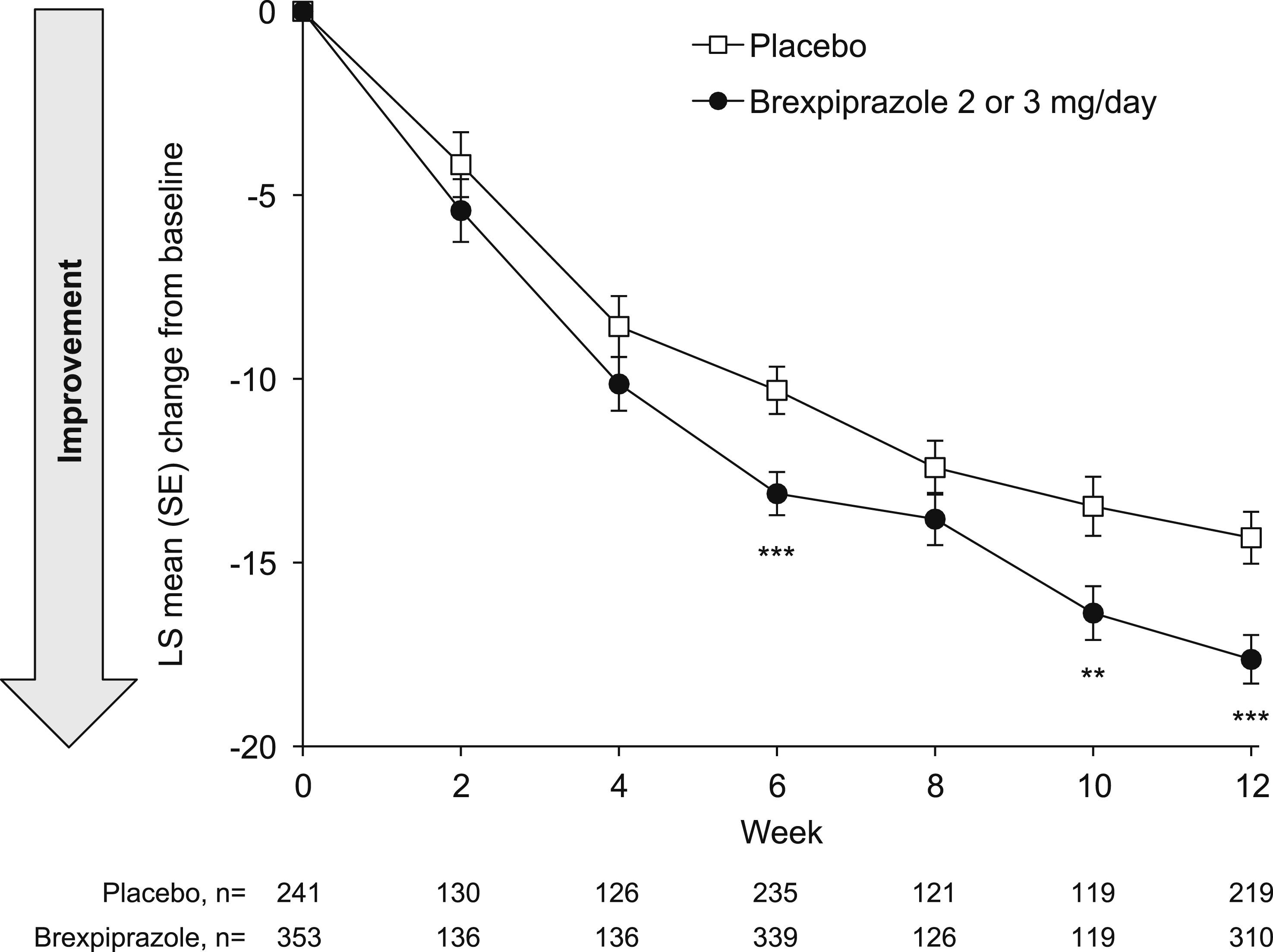
On the key neuropsychiatric symptom of agitation/aggression, brexpiprazole showed greater improvement vs placebo from baseline to Week 12 (LS mean [SE] change in NPI agitation/aggression domain score: brexpiprazole, −4.0 [0.2]; placebo, −3.4 [0.2]) (Figure 2). Improvements vs placebo occurred at Week 6 (P < .01) and were maintained until Week 12 (LS mean difference at Week 12: −0.60; 95% CI: −1.04, −0.16; P < .01) (Figure 2). Change from baseline in NPI agitation/aggression domain score. **P < .01, ***P < .001 vs placebo; MMRM, efficacy sample. LS, least squares; MMRM, mixed model for repeated measures; NPI, Neuropsychiatric Inventory; SE, standard error.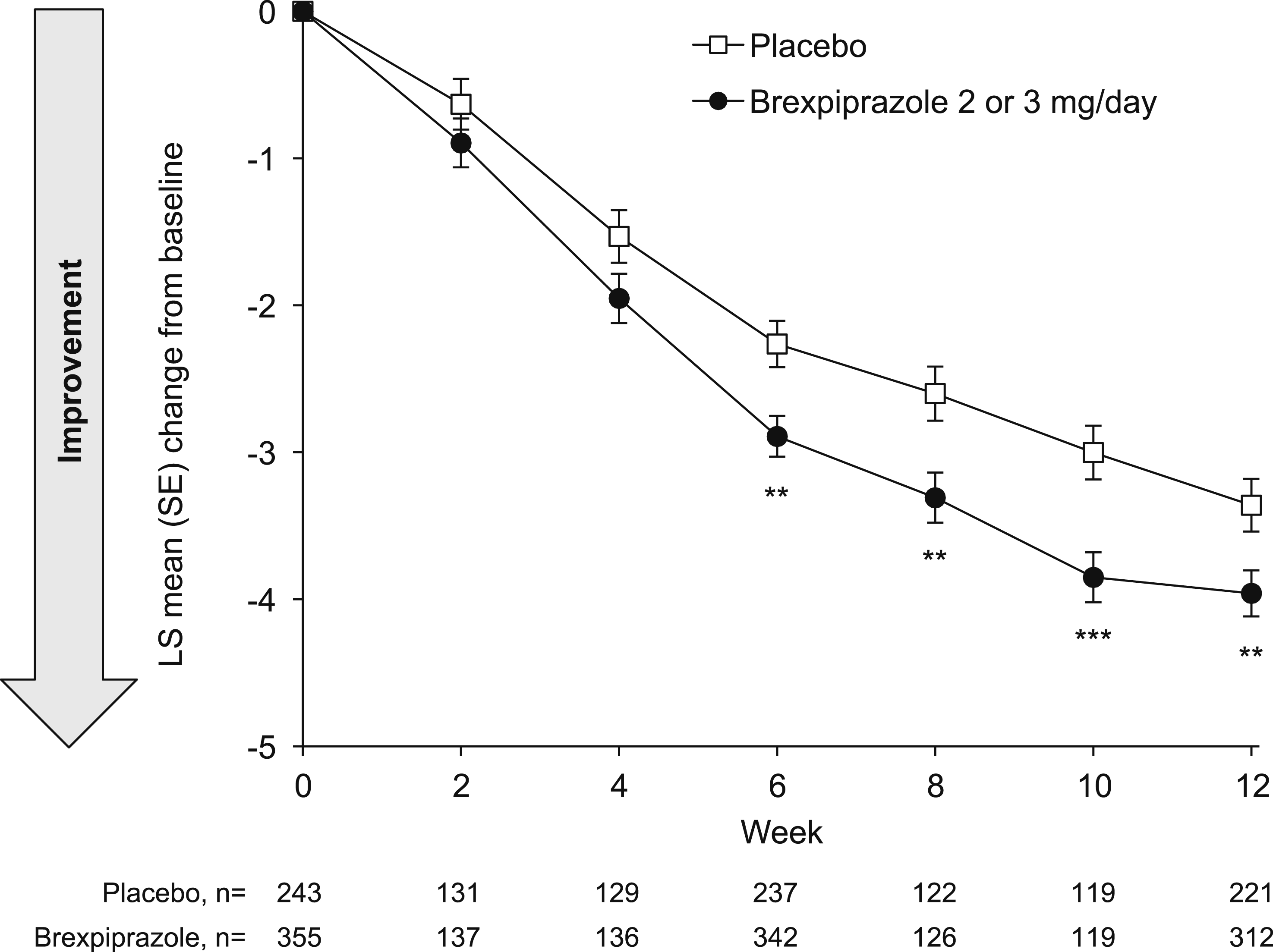
Considering neuropsychiatric symptoms (domain scores) other than agitation/aggression, the change from baseline at Week 12 generally favored brexpiprazole, with greater improvements vs placebo (P < .05) on irritability/lability, aberrant motor behavior, disinhibition, anxiety, and elation/euphoria (Figure S1). No individual neuropsychiatric symptoms worsened relative to baseline with brexpiprazole or placebo treatment.
Changes in the Impact of Neuropsychiatric Symptoms on Caregivers
From Week 4 onwards, the overall impact of symptoms on caregivers showed greater reduction with brexpiprazole compared with placebo (LS mean [SE] change from baseline to Week 12 in NPI occupational disruptiveness/caregiver distress total score: brexpiprazole, −7.0 [0.3]; placebo, −5.4 [0.4]) (Figure 3). The LS mean difference between treatment groups at Week 12 was −1.62 (95% CI: −2.55, −0.70; P < .001). Change in NPI occupational disruptiveness/caregiver distress total score. *P < .05, ***P < .001 vs placebo; MMRM, efficacy sample. LS, least squares; MMRM, mixed model for repeated measures; NPI, Neuropsychiatric Inventory; SE, standard error.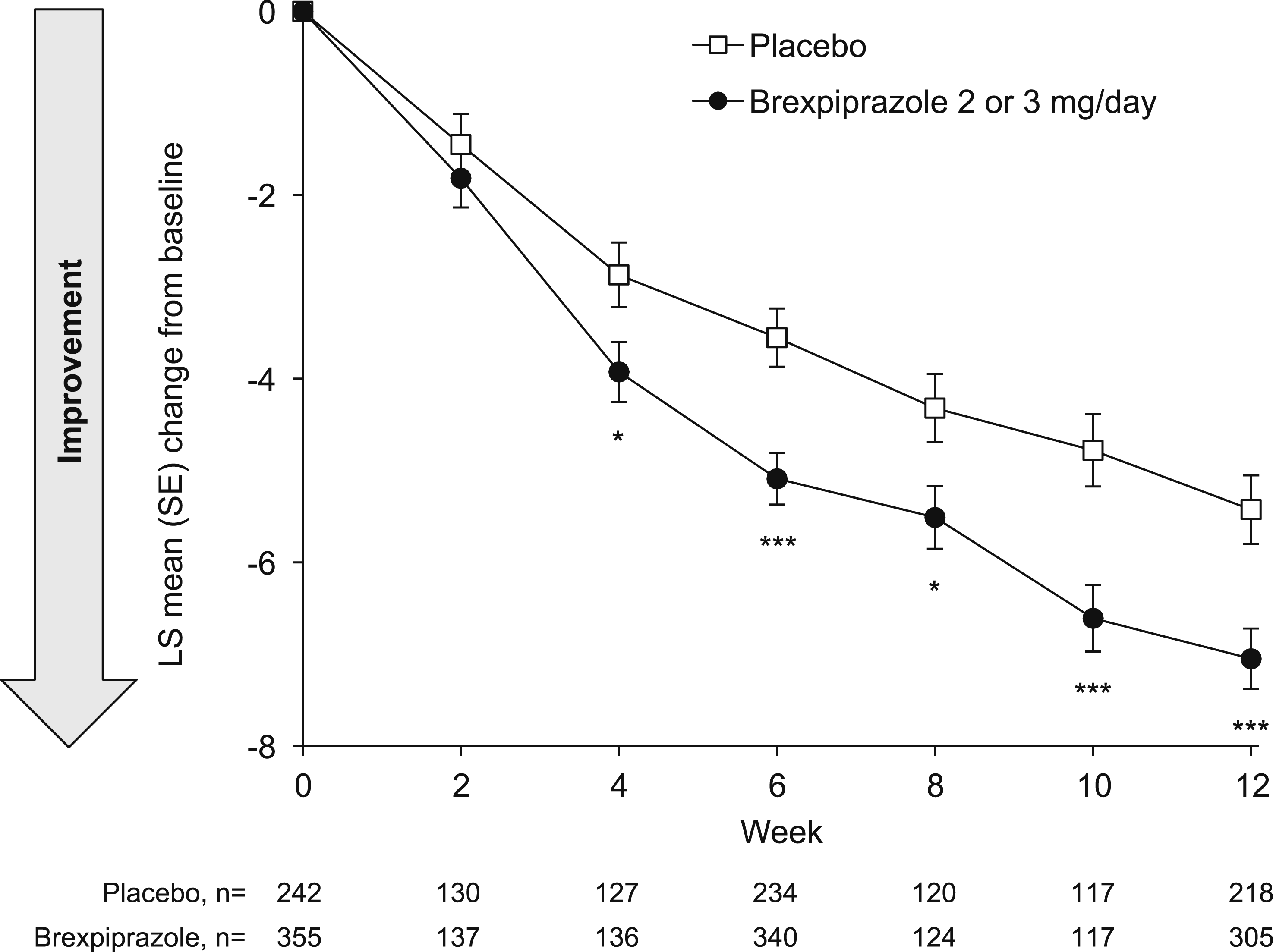
Regarding specific symptoms, the impact of agitation/aggression symptoms (the key neuropsychiatric symptom of interest) on caregivers showed greater reduction from baseline to Week 12 with brexpiprazole vs placebo (LS mean [SE] change in NPI occupational disruptiveness/caregiver distress score in the agitation/aggression domain: brexpiprazole, −1.1 [0.1]; placebo, −0.9 [0.1]) (Figure 4). The LS mean difference between treatment groups at Week 12 was −0.26 (95% CI: −0.42, −0.11; P < .01). Beyond agitation/aggression, greater reductions (P < .01) in impact on caregivers (NPI occupational disruptiveness/caregiver distress domain scores) with brexpiprazole vs placebo were observed for other neuropsychiatric symptoms: irritability/lability, aberrant motor behavior, depression/dysphoria, and anxiety (Figure S2). All NPI occupational disruptiveness/caregiver distress domain scores showed numerical improvement relative to baseline. Change in NPI occupational disruptiveness/caregiver distress score in the agitation/aggression domain. **P < .01, ***P < .001 vs placebo; MMRM, efficacy sample. LS, least squares; MMRM, mixed model for repeated measures; NPI, Neuropsychiatric Inventory; SE, standard error.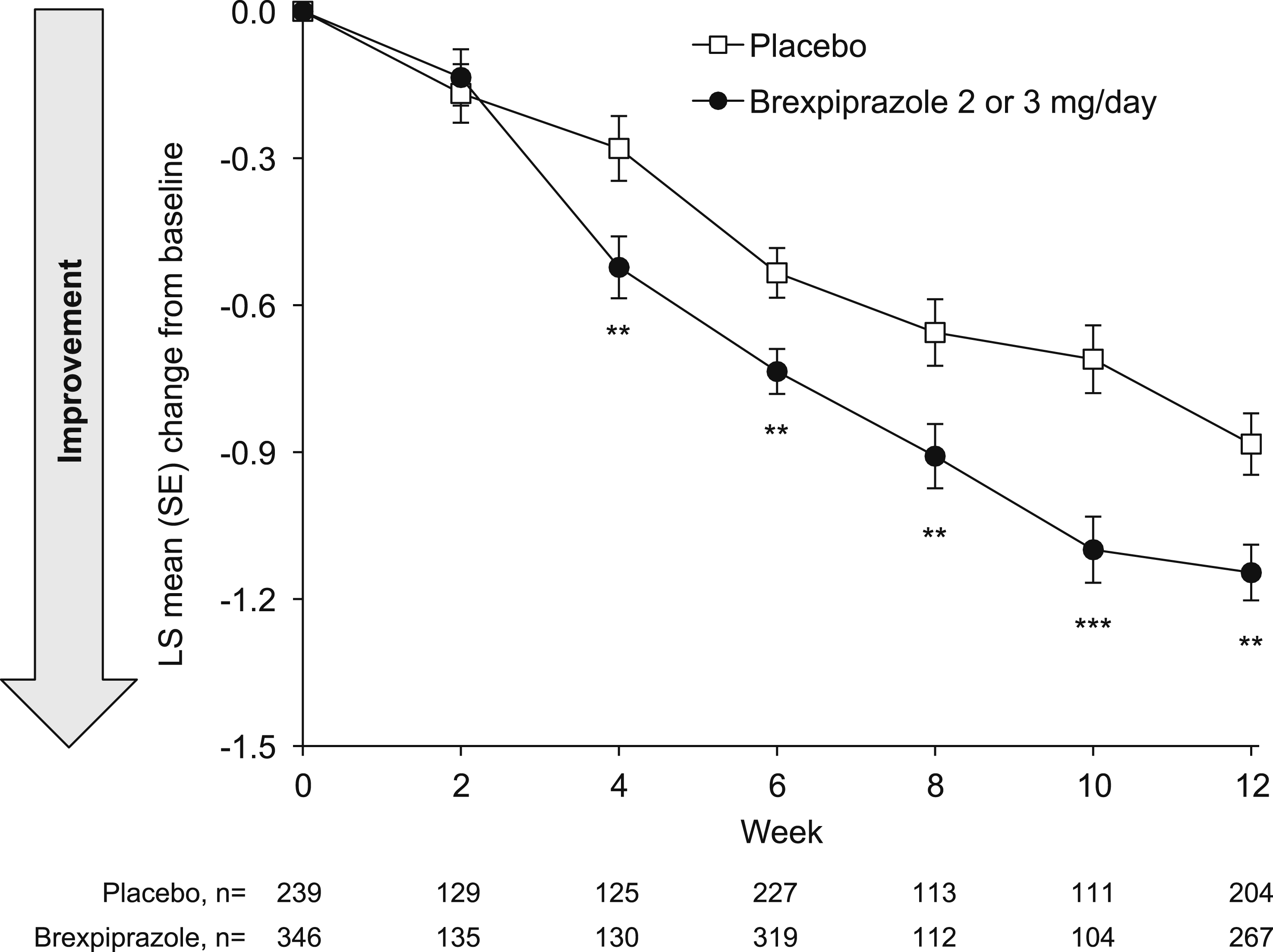
At Week 12, there was moderate correlation (Pearson’s r, 0.58) between changes from baseline in NPI agitation/aggression domain score and corresponding occupational disruptiveness/caregiver distress score.
Discussion
In this analysis of patients with agitation associated with dementia due to Alzheimer’s disease, the composite of neuropsychiatric symptoms improved with brexpiprazole 2 or 3 mg/day vs placebo, as captured by the NPI total score. Of particular relevance to this patient population, improvements in agitation/aggression, along with other neuropsychiatric symptoms (including irritability/lability, aberrant motor behavior, disinhibition, and anxiety), were observed with brexpiprazole vs placebo at Week 12. These results are consistent with a previous analysis of NPI outcomes in a separate 10-week, placebo-controlled trial of brexpiprazole 1 or 2 mg/day in Japan. 33 In the Japanese trial, greater improvements were observed with brexpiprazole vs placebo in agitation/aggression (1 mg/day and 2 mg/day), and in certain other neuropsychiatric symptoms (2 mg/day). 33 The current results are also consistent with analyses of CMAI data from the Phase 3 brexpiprazole trials, in which a meta-analysis showed that brexpiprazole was efficacious on agitation, 34 and a pooled subgroup analysis showed that brexpiprazole reduced agitation regardless of care setting, severity of cognitive dysfunction, presence of co-occurring behavioral symptoms, and concomitant use of dementia medications or antidepressants. 35
This analysis indicates that the effects of brexpiprazole may extend across agitation behaviors to include other neuropsychiatric symptoms that can occur in patients with Alzheimer’s dementia. Beyond agitation/aggression, the neuropsychiatric symptoms with the greatest improvements with brexpiprazole were irritability/lability, aberrant motor behavior, disinhibition, and anxiety. Other analyses suggest that these specific neuropsychiatric symptoms are closely aligned with agitation/aggression.36,37 The literature regarding this relationship between neuropsychiatric symptoms is sparse, but overlapping features may exist. Further research on the relationships between certain neuropsychiatric symptoms is warranted.
Baseline scores were similar between treatment arms, but varied between individual neuropsychiatric symptoms, with typically higher baseline scores (indicating greater symptom presence) for agitation/aggression, irritability/lability, and aberrant motor behavior. As noted, baseline agitation/aggression scores were influenced by trial inclusion criteria (enrolled patients had an NPI agitation/aggression domain score ≥4).21,22 Since irritability/lability and aberrant motor behavior are closely aligned with agitation/aggression,36,37 the higher baseline scores in these domains could be expected. Baseline scores were low for some other neuropsychiatric symptoms (eg, hallucinations; elation/euphoria; appetite and eating changes), which limited the potential scope for improvement in these symptoms. Overall, the heterogeneity of neuropsychiatric symptoms at baseline was reflective of clinical practice. 38
Improvements in neuropsychiatric symptoms were associated with reductions in the overall impact of neuropsychiatric symptoms on caregivers, as well as the specific impact of agitation/aggression, irritability/lability, aberrant motor behavior, depression/dysphoria, and anxiety. For agitation/aggression specifically, caregivers reported moderate impact (score of 3) at baseline, which reduced to mild impact (score of 2) at Week 12, on average. These results are consistent with the effects of brexpiprazole 2 mg/day on caregiver impact in the trial of brexpiprazole in Japan. 33 In the current analysis, change in agitation/aggression domain score correlated with change in agitation/aggression occupational disruptiveness/caregiver distress score, highlighting the relationship between symptomatic improvement and benefit to caregivers. In general, symptoms of agitation, irritability, and anxiety are among those with the greatest associated caregiver burden. 39 The presence of agitation symptoms can influence caregiver decisions to transfer their patients to long-term care. 9 Therefore, improvement in agitation symptoms, and associated reductions in impact on caregivers, may allow patients to remain living at home for longer. As patients in long-term care settings tend to incur higher total annual health care costs than patients living in private homes, 40 enabling patients to live at home for longer may ease the economic burden associated with agitation symptoms in patients with Alzheimer’s dementia.
In these analyses, greater improvements (P < .05) in neuropsychiatric symptoms with brexpiprazole vs placebo were observed as early as Week 6, and greater improvement in impact on caregivers was observed as early as Week 4. Caregivers may spend a large proportion of their week caring, 9 and may have been caring for many years. Reduction in burden 1 month after starting treatment may be a valuable benefit for caregivers. Further work is needed to determine if bridging treatments could help to reduce agitation during the first month of brexpiprazole treatment, as well as to determine the optimal duration of brexpiprazole treatment. 41 Two extension trials indicated that agitation continued to improve over up to 24 weeks on brexpiprazole, with consistent safety.42,43
Clinical meaningfulness is difficult to quantify for the NPI because (1) different domains reflect different symptoms, which may not be equally burdensome, and (2) the nature of frequency × severity scoring means that scores are asymmetric (domain scores of 5, 7, and 11 cannot be achieved). 44 According to caregivers, any reduction in the frequency or severity of agitation behaviors is meaningful. 45 A meaningful within-patient change threshold has been established for the CMAI, and a previous analysis indicated that a greater proportion of participants on brexpiprazole had a meaningful response than those on placebo. 46 Regarding the NPI as an assessment tool, the concordant findings between effects on symptoms, and accompanying changes in impact on caregivers, support convergent validity. This aligns with a previous analysis that found a statistically significant relationship between neuropsychiatric symptom scores and caregiver burden. 47 In clinical practice, alternative scales with faster administration may be preferred, such as the NPI-Q or the Agitation in Alzheimer’s Screener for Caregivers.44,48,49 The NPI-C-IPA, in which items from the NPI Clinician version are mapped onto the IPA agitation criteria, may be a valuable addition to future trials. 50 Future work may also consider use of emerging real-time technologies to track the neuropsychiatric symptoms of dementia, such as objective quantification via sensors51-53 and ecologically informed momentary assessments.54,55
The safety and tolerability of brexpiprazole in this patient population have previously been published. 23 Safety analyses indicate that brexpiprazole was generally well tolerated, with no specific safety concerns. 23 Across the three 12-week brexpiprazole trials, death was reported for 6 participants on brexpiprazole (0.9%; intracranial hemorrhage, acute purulent meningoencephalitis, pneumonia, airway obstruction, end-stage Alzheimer’s dementia, and heart failure) and 1 participant on placebo (0.3%; respiratory insufficiency due to pneumonia). 23 No deaths were attributed to brexpiprazole treatment. 23 Safety was not specifically assessed in the current analysis; however, no worsening of any neuropsychiatric symptom was observed over 12 weeks, which supports the tolerability of brexpiprazole in this patient population.
A key strength of this analysis is the evaluation not only of changes in patients’ symptoms, but also the associated changes in impact on caregivers. Secondly, the use of a caregiver-based assessment reflects the nature of information-gathering in clinical practice, which may facilitate earlier symptom identification, diagnosis and treatment. 56 Lastly, based on the authors’ experience, the baseline neuropsychiatric symptoms in the analyzed sample were broadly representative of patients in clinical practice.
This analysis is limited by its post hoc nature, meaning that results should be considered exploratory and hypothesis-generating. There were no adjustments for multiple testing, which increases the risk for Type I errors (false positives) and means that results should be confirmed in prospectively collected data with prespecified analyses that correct for multiple comparisons. 57 NPI outcomes were exploratory or “other” endpoints in Trials 283 and 213, and were therefore not powered for comparisons between brexpiprazole and placebo. The NPI was assessed less frequently in Trial 213 than in Trial 283 in order to reduce the burden of questionnaires on patients and caregivers. However, treatment differences at Weeks 6 and 12 were generally similar between the individual trials (data not shown), indicating that the overall results were not driven by one trial. Data for occupational disruptiveness (in nursing homes) and caregiver distress (at home) were combined to measure the social/environmental effects of patients’ symptoms, and future studies could analyze these aspects separately in each setting. The 2 mg and 3 mg doses were pooled to increase sample size and thereby increase confidence in the results, and future work may wish to consider the effects of individual doses. While trial designs were similar, Trial 213 was enriched for aggressive agitation but Trial 283 was not.21,22 Inclusion of institutionalized and non-institutionalized patients enabled a broader, more representative sample, but involvement of different settings may have resulted in heterogeneity in symptom reporting and the nature of the impact on caregivers. Trial participants were required to have agitation symptoms at baseline (NPI agitation/aggression domain score ≥4), whereas some other neuropsychiatric symptoms were present in only a small number of patients, limiting the comparability of treatment effects of brexpiprazole across symptoms. Finally, symptoms were attributed to dementia of the Alzheimer’s type, but no confirmatory biomarkers were collected in the trials.
Conclusions
In this exploratory post hoc analysis, the key neuropsychiatric symptom of agitation/aggression improved over 12 weeks with brexpiprazole 2 or 3 mg/day vs placebo in patients with agitation associated with dementia due to Alzheimer’s disease. Improvements in agitation/aggression symptoms were accompanied by reductions in associated caregiver disruption and distress. Additionally, the composite score of neuropsychiatric symptoms improved in patients treated with brexpiprazole, and there were concurrent reductions in other neuropsychiatric symptoms, including irritability, aberrant motor behavior, disinhibition, and anxiety. Similar to agitation/aggression, improvement in other neuropsychiatric symptoms was generally accompanied by associated reduction in caregiver disruption and distress. Prospective studies are needed to confirm these results.
Supplemental Material
Supplemental Material - Efficacy of Brexpiprazole on Neuropsychiatric Symptoms and Impact on Caregivers: Pooled Neuropsychiatric Inventory (NPI) Analysis in Patients With Agitation Associated With Dementia due to Alzheimer’s Disease
Supplemental Material for Efficacy of Brexpiprazole on Neuropsychiatric Symptoms and Impact on Caregivers: Pooled Neuropsychiatric Inventory (NPI) Analysis in Patients With Agitation Associated With Dementia due to Alzheimer’s Disease by George T Grossberg, Marwan N Sabbagh, Sanjeda R Chumki, David Wang, Pedro Such, Zhen Zhang, Anton M Palma, and Jeffrey L Cummings in Journal of Geriatric Psychiatry and Neurology
Footnotes
Acknowledgements
Writing support was provided by Zoe Aliwell, BSc, and colleagues of Cambridge (a division of Prime, Knutsford, UK), funded by Otsuka Pharmaceutical Development & Commercialization Inc. and H. Lundbeck A/S. Parts of this work were presented as a poster at the Society for Post-Acute and Long-Term Care medicine (PALTC) annual conference; March 9-11, 2024; San Antonio, TX and the American Association for Geriatric Psychiatry (AAGP) annual meeting; March 15-18, 2024; Atlanta, GA. Jeffrey L Cummings is supported by NIGMS grant P20GM109025; NIA R35AG71476; NIA R25AG083721-01; NINDS RO1NS139383; Alzheimer’s Disease Drug Discovery Foundation (ADDF); Ted and Maria Quirk Endowment; and Joy Chambers-Grundy Endowment.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: George T Grossberg has received consulting fees from Acadia, Abbott, Axsome, Biogen, Bristol Myers Squibb, BioXcel, Eisai, Karuna, Lundbeck, MapLight Therapeutics, Otsuka, and Takeda. He is on Safety Monitoring Committees for Anavex, Quince, Merck, Newron, and Oligomerix. Marwan N Sabbagh has received consulting fees from Abbvie, Alzheon, Anavex, Cognito Therapeutics, Eisai, GSK, Lilly, NeuroTherapia, Novo Nordisk, Signant Health, and Synaptogenix. He is on the board of Directors for CervoMed (previously known as EIP Pharma). Sanjeda R Chumki, Malaak Brubaker, Zhen Zhang, and Anton M Palma are full-time employees of Otsuka Pharmaceutical Development & Commercialization Inc. David Wang is a full-time employee of Lundbeck LLC. Pedro Such is a full-time employee of H. Lundbeck A/S. Jeffrey L Cummings has provided consultation to Acadia, Acumen, ALZpath, Annovis, Aprinoia, Artery, Axsome, Biogen, Biohaven, BioXcel, Bristol-Myers Squibb, Eisai, Fosun, GAP Foundation, Green Valley, Janssen, Julius Clinical, Karuna, Kinoxis, Lighthouse, Lilly, Lundbeck, LSP/eqt, Mangrove Therapeutics, Merck, MoCA Cognition, New Amsterdam, Novo Nordisk, onocC4, Optoceutics, Otsuka, Oxford Brain Diagnostics, Praxis, Prothena, ReMYND, Roche, Scottish Brain Sciences, Signant Health, Simcere, Sinaptica, T-Neuro, TrueBinding, and Vaxxinity pharmaceutical, assessment, and investment companies. He owns the copyright of the Neuropsychiatric Inventory.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Otsuka Pharmaceutical Development & Commercialization Inc (Princeton, NJ, USA) and H. Lundbeck A/S (Valby, Denmark). The sponsors were involved in the design of the research, the analysis and interpretation of data, and the writing and reviewing of this article.
Ethical Considerations
This analysis used pooled data from published clinical trials. The trials were approved by relevant institutional review boards or independent ethics committees and were conducted in accordance with the principles stated in the Declaration of Helsinki, the International Conference on Harmonisation Good Clinical Practice Guideline, and local regulatory requirements.
Consent to Participate
This analysis used pooled data from published clinical trials. All participants and/or their legal representatives provided written or electronic informed consent prior to the start of the trials.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
