Abstract
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder manifest by motor symptoms including tremor, rigidity, bradykinesia, and postural instability, 1 as well as a variety of non-motor issues including sleep, mood, motivation, and cognition. 2 Both motor and non-motor symptoms can impact quality of life (QoL). 2 The global prevalence of PD is rising rapidly, and in 2021, it was estimated that there were over 11.7 million individuals with PD, representing a substantial morbidity and mortality burden. 3 While there is no cure for PD, medications (eg, dopaminergic replacement therapy [DRT]), surgical interventions, and other therapies can help to relieve some symptoms.4,5 While DRT can improve PD motor symptoms, it can also worsen many non-motor symptoms (NMS). 6 Consequently, there is substantial interest in non-pharmacological options for treating PD. 7
Physical activity (PA) is defined as any movement of the body that uses energy, whereas exercise is a specific type of PA that is planned, structured (characterized by frequency, intensity and time) and performed with the purpose of improving physical fitness. 8 Thus, all exercise is PA while not all forms of PA qualify as exercise. Evidence in the literature has demonstrated that exercise and other forms of PA can ease PD motor symptoms, improve functioning, and perhaps slow the clinical progression of PD.9-12 Maintenance of motor functions (eg, gait, balance, and muscle strength) is critical because it enables the safe performance of daily activities, which in turn, helps individuals to maintain independence. 10 Randomized controlled trials have shown that moderate- to vigorous-intensity PA performed for at least 30 minutes, three times per week, improves the severity of motor symptoms and can slow the progression of symptoms over three to six months,13-15 and a recent proof-of-concept study provided evidence that sustained periods of higher intensity PA have neuromodulatory effects in PD. 16 PA interventions may also have a positive impact on NMS (eg, improvements in depression and anxiety 17 ), but the evidence is limited compared to the effect on motor symptoms.11,18,19 People living with PD have identified a need for advice about accessible and low-cost lifestyle changes, including exercise, to enhance their wellness, sense of agency, daily functioning, and QoL. 20
In 2021, the Parkinson’s Foundation and the American College of Sports Medicine issued updated PA recommendations for individuals with PD. The guidelines recommend 150 minutes of moderate- to vigorous-intensity exercise (ie, 40%-60% heart rate reserve [HRR] and 60%-85% HRR, respectively) each week, albeit only after evaluation by a physical therapist specializing in PD. The updated guidelines specifically recommend that individuals complete aerobic activities three days per week for at least 30 minutes per session of continuous or intermittent movement at moderate- or vigorous-intensity, and complete strength training activities on two or three non-consecutive days per week for at least 30 minutes per session for 10-15 repetitions for major muscle groups. 21 The guidelines also recommend integrating balance and stretching activities at least two to three days per week. 21
PA interventions in PD populations are often trialed under highly controlled, optimal research conditions. Individuals are typically asked to complete structured PA interventions and are continuously monitored (ie, directly supervised in a laboratory or other setting (eg, the SPARX3 Study 22 ), or remotely observed with digital activity trackers, such as pedometers, accelerometers and heart rate monitors (eg, the STEPWISE study 23 )) throughout the trial period, although some behavior change programs have also been trialed (eg, the ParkFit Program 24 ). However, adaptations of PA interventions are common when they are delivered at-scale and may impact intervention effectiveness. 25 There is limited evidence available on the relationship between PA levels and PD outcomes in an unsupervised real-world context. Therefore, additional research is needed to elucidate the potential associations between the frequency and intensity of PA and PD outcomes.
The aim of this study was to assess the relationship between PA frequency and PD outcomes, including motor symptoms, NMS, and QoL in people with PD in a naturalistic environment.
Methods
Data Collection
Data were obtained from the Modifiable Variables in Parkinsonism (MVP) Study. The MVP, initiated in 2013, is a prospective, observational natural history study designed to elucidate lifestyle variables associated with rate of PD progression. The MVP Study was approved using the Bastyr University Institutional Review Board (IRB; #13A-1332) and listed on ClinicalTrials.gov (#NCT02194816). Study participants were recruited via Washington State Parkinson Disease Registry, Michael J Fox Foundation Trial Finder, through social media advertising and in-person through community lectures and outreach around the country. Due to this being an online survey, the IRB issued a Waiver of Documentation of Informed Consent. All study participants included in this analysis reviewed the Participant Information Sheet and provided consent via the online survey. All individuals confirmed that they read and understood the participant information sheet prior to study participation. Study data were collected and managed using Research Electronic Data Capture (REDCap) electronic data capture tools hosted at Bastyr.26,27 REDCap is a secure, web-based software platform designed to support data capture for research studies, providing: (i) an intuitive interface for validated data capture; (ii) audit trails for tracking data manipulation and export procedures; (iii) automated export procedures for seamless data downloads to common statistical packages; and (iv) procedures for data integration and interoperability with external sources.
At the baseline visit, all individuals with PD are asked for information about their diagnosis. While individuals with any form of parkinsonism, or evidence of prodromal parkinsonism, are eligible to participate in the MVP study, only individuals reporting a diagnosis of “Parkinson’s disease/ Idiopathic Parkinson’s disease (PD)” were included in this analysis. The data presented here are a cross-sectional analysis of baseline data of those individuals.
Outcome Measures
Demographic and Disease Characteristics of the Cohort.
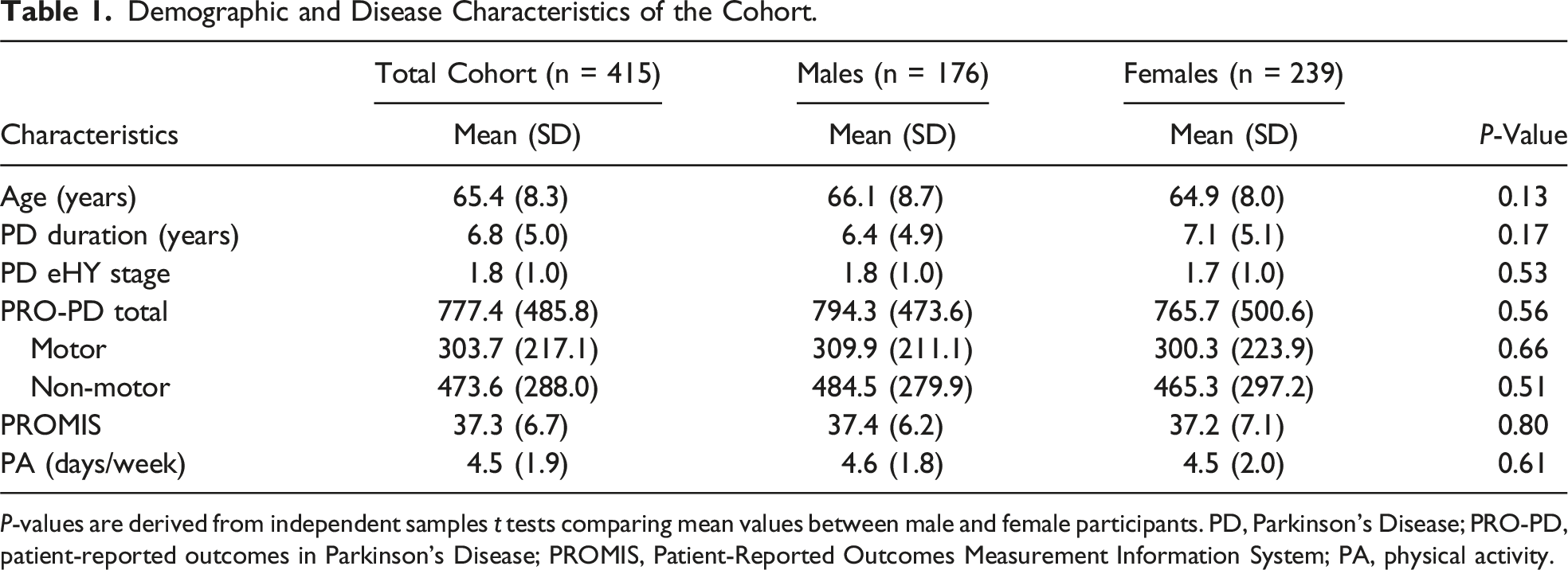
P-values are derived from independent samples t tests comparing mean values between male and female participants. PD, Parkinson’s Disease; PRO-PD, patient-reported outcomes in Parkinson’s Disease; PROMIS, Patient-Reported Outcomes Measurement Information System; PA, physical activity.
The PRO-PD scale is a validated measure for PD symptom severity. 29 Patient-reported outcomes (PRO) are increasingly recognized as appropriate complements to traditional clinical trial measures, and they allow patient perceptions to be considered in the definition of treatment success.30,31 The total PRO-PD score provides the cumulative scale of 32 slider bars that each evaluate a different PD symptom. 32 PRO-PD has been shown to correlate with well-validated endpoints in PD including Unified Parkinson’s Disease Rating Scale, Hoehn/Yahr stage, years since diagnosis, quality of life, and the Non-Motor Symptom Scale in cross-sectional analysis. 32
For this analysis, the total PRO-PD, as well as motor and non-motor subsets, were included as outcomes. The PRO-PD motor subset consisted of the following symptoms: walking, rising from a seated position, tremor, freezing of gait, dressing/eating/grooming, balance, frequent falls, dyskinesia, handwriting, bradykinesia, speech, and stooped posture. The PRO-PD non-motor subset included the variables: constipation, lack of motivation, depression, loss of interest, anxiety, fatigue, daytime sleepiness, temperature dysregulation, orthostatic hypotension, visual disturbances, insomnia, rapid eye movement (REM) sleep behavior disorder, muscle pain, drooling, memory impairment, comprehension disability, hyposmia, sexual dysfunction, urinary dysfunction, and hallucinations.
The 10-question PROMIS Global Scale was chosen as the primary QoL measure for this analysis. The PROMIS Global Scale measure has been validated across diverse chronic conditions and meets the “Recommended” criteria for utilization by the International Parkinson and Movement Disorder Society Task Force on Rating Scale. 33 For PA frequency, participants were asked “On how many of the last seven days did you participate in at least 30 min of physical activity?”
Statistical Analyses
Descriptive statistics were used to characterize demographic and disease characteristics of the cohort and independent samples t-tests were used to assess for group differences between male and female participants. Multiple linear regression models were developed to examine the association between self-reported PA (measured as days/week that the participants completed at least 30 minutes of PA) and patient-reported outcomes (PRO) with total PRO-PD score, PRO-PD motor items, PRO-PD non-motor items or PROMIS scores used as the dependent variable. The linear regression analyses controlled for age, years since diagnosis, and sex. Participant records were excluded from the linear regression analyses if age, years since diagnosis, and/or sex data were missing. Associations of individual symptom scores with PA were assessed using partial correlations, controlling for age, sex, and disease duration. Because enrollment in the MVP Study has historically included a large proportion of female participants, we also examined separate models to assess associations separately in males and females. Only participants reporting a diagnosis of idiopathic PD were included in this analysis. For participants who did not score an element from the 33 individual PRO-PD symptoms, the cohort average for that individual symptom was substituted for any missing values. For other data analyses, such as QoL measures, the most complete dataset was utilized for the relevant variables. Statistical analyses were conducted in SPSS (IBM, Armonk, NY) with alpha level set to 0.05. In this exploratory study, no adjustments were made for multiple comparisons to avoid increasing the risk of type II errors and prevent excluding potential associations that may be explored in future studies.
Results
This study included 415 participants with a self-reported diagnosis of PD; 58% of participants were women. Mean (SD) age and PD disease duration were 65.4 (8.3) and 6.8 (5.0) years, respectively. Participants reported exercising for at least 30 minutes an average of 4.5 (1.9) days per week. Characteristics of the cohort, including a comparison of male and female participants, are detailed in Table 1.
More Frequent PA is Associated with Improved PRO and QoL in PD
PRO-PD total motor and non-motor scores each decreased (indicating less severe symptoms) with increasing PA frequency (Figure 1A). Similarly, PROMIS scores increased (indicating greater QoL) with more frequent PA (Figure 1B). For each outcome, there was a significant dose-response between activity levels and patient-reported scores (ANOVA P < 0.001, Figure 1).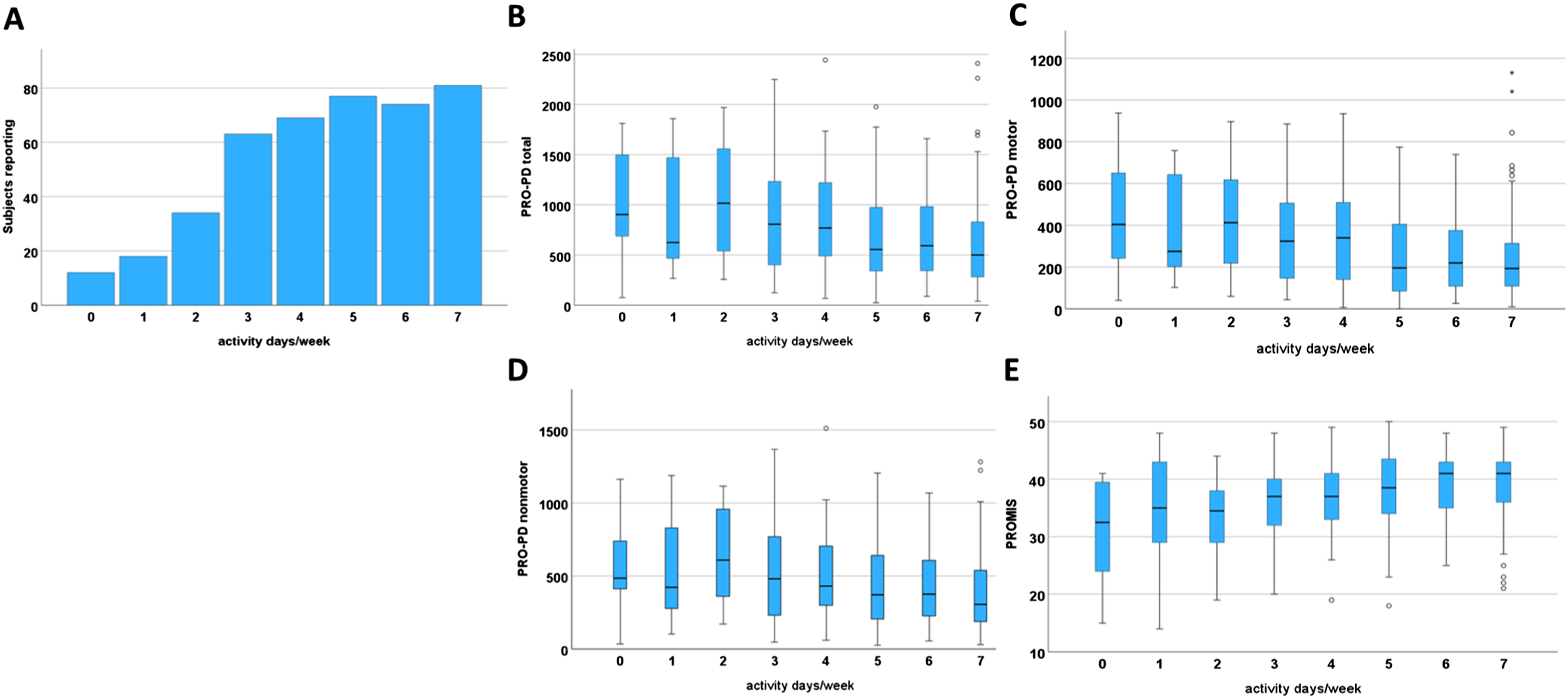
Association of Individual Motor and Non-motor PD Symptoms with PA Frequency
Most, but not all, motor and non-motor domains were inversely associated with the frequency of PA (Figure 2). Using partial correlations controlling for age, sex, and disease duration, PA showed the strongest inverse association with walking, arising from a chair, balance, and dressing among motor symptoms (Table 2). Among NMS, problems with motivation, excessive sleepiness, and sexual dysfunction demonstrated the strongest inverse association with PA frequency. Conversely, cognitive complaints including memory and comprehension scores as well as smell loss were not significantly associated with PA. Correlation Between Individual Motor and Non-motor Parkinson’s Disease Symptoms and Physical Activity Levels. Data are partial correlations (controlling for age, sex, and disease duration) between individual symptoms and days/week of reported physical activity and associated P-values (significant P-values are in bold).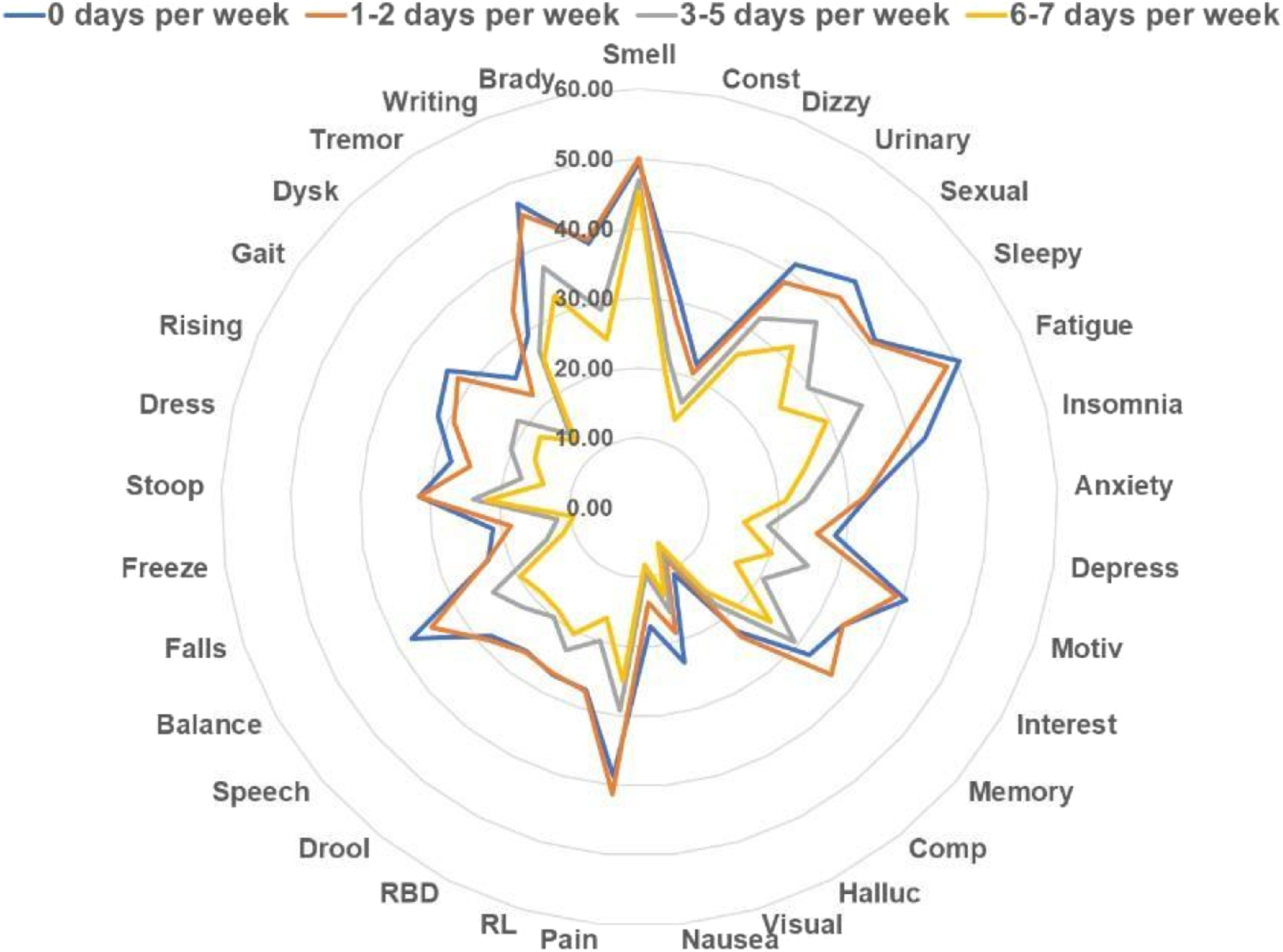
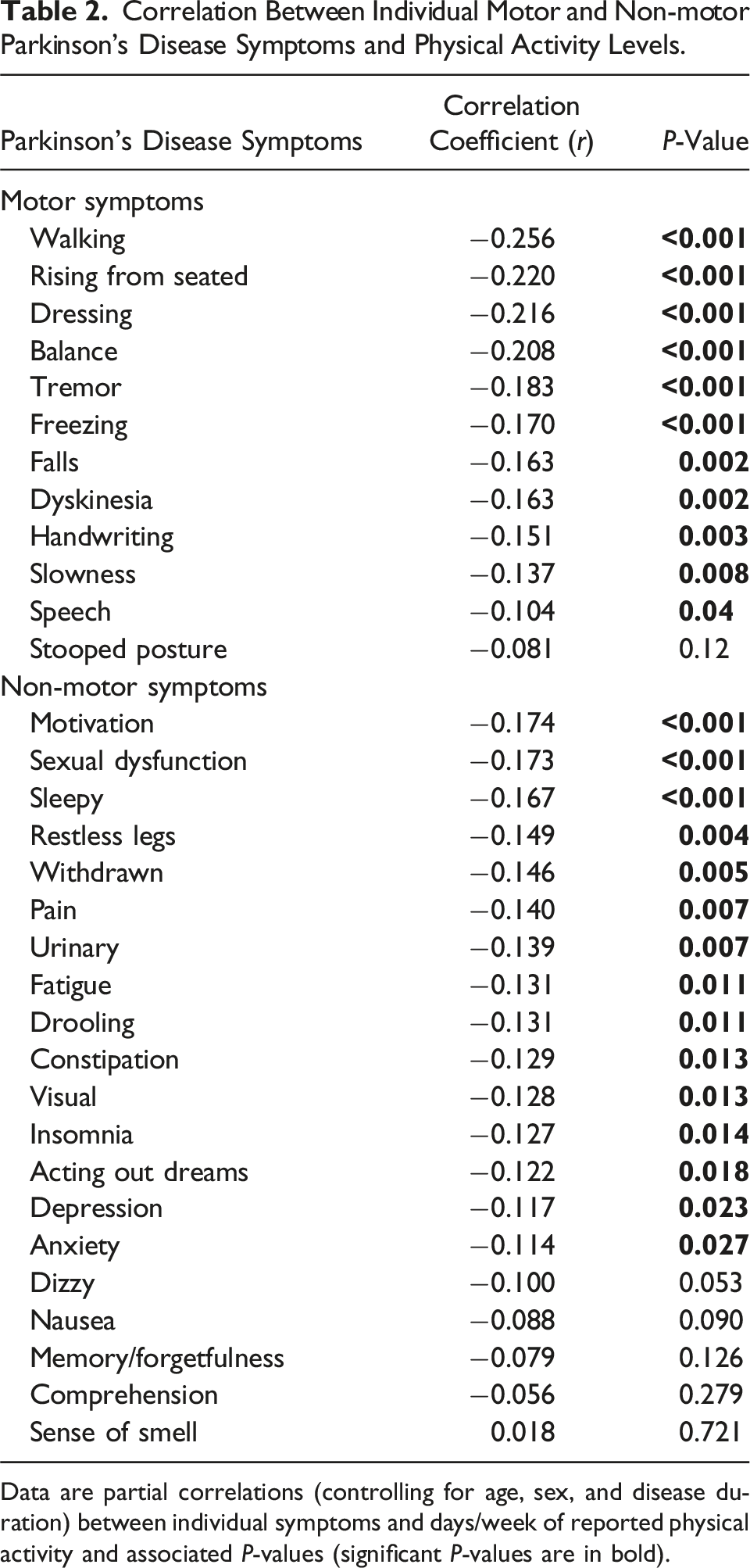
Sex-specific Association Between PA, PRO, and QoL in PD
Sex-specific Differences in the Associations Between Physical Activity and Patient Reported Outcomes in Parkinson’s Disease.
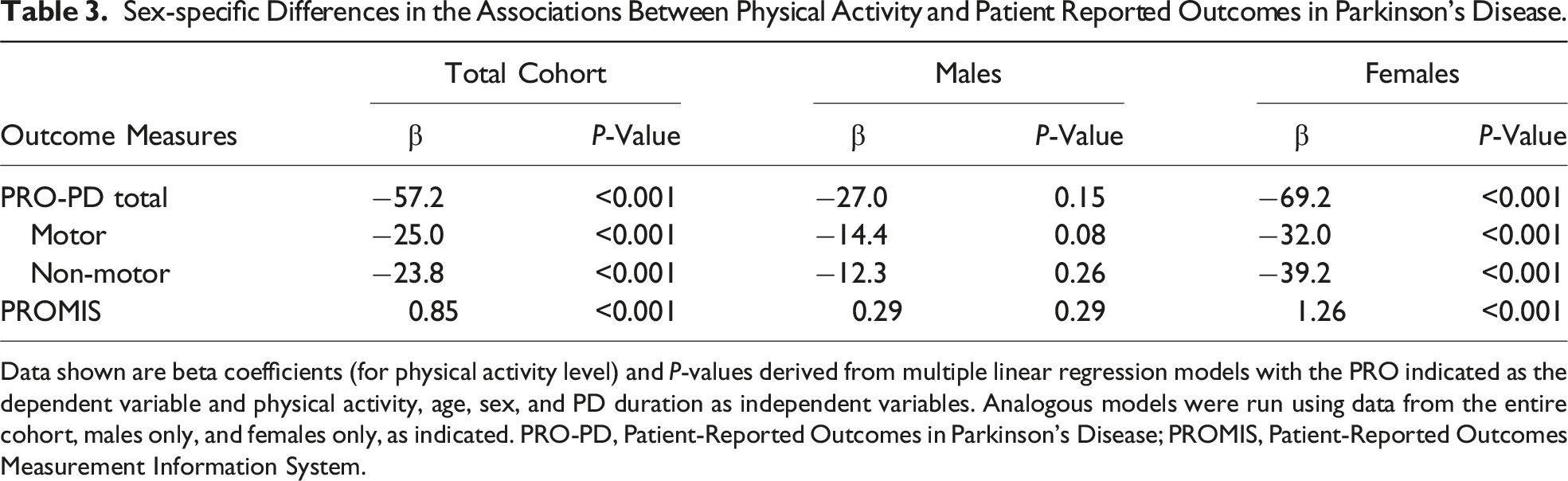
Data shown are beta coefficients (for physical activity level) and P-values derived from multiple linear regression models with the PRO indicated as the dependent variable and physical activity, age, sex, and PD duration as independent variables. Analogous models were run using data from the entire cohort, males only, and females only, as indicated. PRO-PD, Patient-Reported Outcomes in Parkinson’s Disease; PROMIS, Patient-Reported Outcomes Measurement Information System.
Discussion
Overall, participants with higher levels of PA had less severe total, motor, and non-motor PD symptoms, as well as higher QoL. Although a variety of PA interventions have been shown to improve PD motor outcomes in the highly controlled settings of clinical trials, less is known about the relationship between activity levels in community settings and important patient-centered outcomes.34,35 People living with PD have identified a need for additional information about accessible, low cost lifestyle interventions to improve their QoL. 20 Providing individuals with guidance for PA from the day of diagnosis has been identified as a strategy to improve self-agency and instill hope for individuals with PD. 36 The results of this study suggest that PA interventions for individuals with PD can successfully be translated to community settings, given that improvements in PD outcomes were observed in an unsupervised real-world context. However, this cohort reported higher levels of PA (4.5 days/week on average) than prior reports in both early and mixed stage PD populations.37-40 Given their willingness to participate in the MVP study, it is possible that this cohort is also more motivated to participate in PA than the general PD population.
The findings from this study also suggest that improvements in patient-reported outcomes from PA are dose-dependent (Table 3). Specifically, the results indicate completing PA five to seven days per week provides maximum benefits. This has implications for clinical recommendations and suggests that health care providers should (i) emphasize that the recommendations represent the minimum amount of PA that individuals with PD should complete, and (ii) recommend that individuals with PD try to incorporate additional PA above the minimum each week to maximize benefits, provided that their individual health status allows for the safe completion of activities.
The use of PRO in this study provided valuable information about the association between PA levels and specific PD symptoms. PRO are underutilized in clinical trials and provide important information not often captured by traditional assessments.41-43 Motor symptoms were broadly associated with activity levels (Figure 2, Table 2), reminiscent of improved overall motor function in numerous trials of aerobic, resistance, and balance PA interventions in PD.44-47 However, there is limited evidence in the literature examining the potential link between PA and NMS.11,18,19 This study demonstrated strong inverse associations between problems with motivation, excessive sleepiness, sexual dysfunction, and PA frequency (Table 2). The observed relationship between sleep-related symptoms and PA activity was consistent with results from a recent interventional PA trial where sleep was the primary outcome. 48 However, cognitive symptoms (memory, comprehension) were not significantly associated with PA in this study (Table 2), in contrast to clinical trial data suggesting that PA improves cognition in PD.49,50 While this inconsistency may relate to the use of subjective rather than objective cognitive measures or intensity of PA, it highlights the need to target non-motor outcomes in PA trials using validated scales.
Higher self-reported QoL, measured by the PROMIS Global Scale, was strongly associated with increased PA frequency (Figure 1, Table 3). Previous interventional trials of PA in PD have generally failed to demonstrate this relationship,51,52 in part, because QoL has been inconsistently included as an outcome. However, a systematic review and meta-analysis indicated that PA interventions, especially aerobic exercise, can significantly improve QoL in individuals with PD, although certain domains of QoL (ie, mobility, activities of daily life, and social support) improved more than others and there was high heterogeneity among included studies. 53
In sex-specific multivariate models, the association between PA and PRO was only statically significant in women. Studies in the general population have reported that women are somewhat less active than men, implying that there could be a differential benefit to increasing PA levels. 54 Furthermore, women may preferentially engage in different types or intensity of PA and derive cardiovascular benefits of PA at lower intensity than men.55,56 However, the sex differences observed in this study must be reproduced in independent cohorts, and the potential mechanisms and implications warrant further investigation. Bespoke guidelines for wellness strategies that account for age, gender, and other sociodemographic factors are needed, as large gaps have been identified in the management of these populations in the PD context. 57
This study has limitations that should be acknowledged and considered in the interpretation of the findings. Relying on participant self-report of a diagnosis, instead of a neurological evaluation performed by a trained clinician, is a limitation of this study. Individuals with PD can misunderstand the diagnosis, cognitive impairment can interfere with ability to understand or recall the diagnosis, and individuals with PD may be misdiagnosed early in the disease, either due to lack of specialist involvement or the overlapping presentations of different forms of parkinsonism early in the disease. This diagnostic certainty is further complicated as the defining characteristics of PD are still evolving. 58 Data regarding daily dopamine use for participants was not available.
Another limitation of the study is that “physical activity”, which includes daily tasks like walking, household chores, gardening, sports, and workout routines, and “exercise,” which is specifically intended to enhance physical health, are not defined in the study. This can create conceptual confusion, diluting the clarity of the findings, and introduces validity concerns, as participants may only be reporting exercise and thus underestimating overall PA. Despite these limitations, this simplified messaging can aid in pragmatic public messaging in supporting a “Just Do Something” approach. Subsequent analyses should include a validated measure for PA, which was not available for this analysis.
Due to the cross-sectional nature of the data used in this study, it is not clear whether a lower level of symptom severity allows for a higher level of PA or if a higher level of PA leads to improved symptom severity and function in individuals with PD. It is also possible that both effects occur and contribute to a positive feedback loop. For example, walking symptoms were better in participants with higher PA levels, and it seems equiprobable that participants with better gait (or other motor symptoms) might be more active, or that PA could improve gait symptoms (Table 2). Additionally, PRO are subjective by nature and PA levels were self-reported in this study. Therefore, these data were subject to potential recall bias. However, self-reported PA levels have been shown to have a high level of agreement with objectively measured step counts in at least one PD cohort. 38 Additionally, each symptom in PRO-PD is assessed through a single question allowing for stratification by symptoms. Future studies using detailed, validated questionnaires targeted at domains of interest may yield additional information.
This study assessed PA frequency, but information about the intensity or type of PA was not available. Evidence indicates that PA intensity may be an important factor that contributes to the efficacy in improving PD outcomes,13,14,59 although an observational study suggested that a larger volume of PA, regardless of the intensity, is associated with slower disease progression. 60 A systematic review and meta-analysis also demonstrated that most different types of exercise have a beneficial effect on both the severity of PD motor symptoms and QoL. 9 The questionnaires used in concert with the PRO-PD scale have been modified to collect information on PA type and intensity for future studies.
Conclusion
This study demonstrated that higher PA levels are associated with better patient-reported motor symptoms, NMS, and QoL in PD. Future research should focus on the potential sex differences in the association between activity levels and PD outcomes, as this could offer insights into the relationship between PA and PD symptoms. The remotely acquired PROs, PROMIS and PRO-PD, were sensitive to changes in PA frequency and provided a unique description of patient-perceived symptoms associated with lack of PA in a real-world setting. Based on self-reported PA frequency data, 6-7 days per week of PA may be associated with improved PROs in PD.
Footnotes
Acknowledgments
We thank Bastyr University Research Institute for resources and for housing the MVP study. This team was brought together by the RAND REACH Center, which is supported by the National Center for Complementary & Integrative Health of the National Institutes of Health under Award Number U24AT012549. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Contributions
James F. Morley - Study design, data analysis, first draft of manuscript. Indu Subramanian – Study design, data interpretation, manuscript edits. Joshua Farahnik – Data management, manuscript edits. Leah Grout - Data interpretation, manuscript edits. Cristal Salcido - Data interpretation, manuscript edits. Josi Kurtzer – Data interpretation, manuscript edits. Laurie K. Mischley – study concept, data collection, manuscript edits.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Potential conflict: author LKM owns the copyright of the PRO-PD scale, which was used as an outcome measure in this study; however, LKM was not directly involved in the data management or analysis. Furthermore, LKM has made the PRO-PD scale freely available.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no financial support for authorship and/or publication of this article. The MVP Study providing these data was initiated with the support of the NIH K01-ATT004404, and has been funded by community donations made by Bill and Sondra Fondren, Mark Claude, Sondra Wolfe Elias, Debbie Krasnow, Mike S., and other anonymous donors.
