Abstract
Background and Objectives
While hearing loss is a known risk factor for dementia, the impact of incident hearing loss on subsequent dementia risk remains underexplored. This study examined the association between newly reported hearing loss and dementia risk in U.S. adults, focusing on critical intervention periods for dementia prevention.
Research Design and Methods
Participants from the Health and Retirement Study who reported no hearing loss or hearing aid use in 2010 or 2012 were included. Incident hearing loss and dementia were assessed via self-report and proxy report. Pooled logistic regression models with inverse probability weighting estimated the cumulative incidence of dementia at 2, 4, 6, and 8 years after baseline. Risk ratios (RR) with 95% confidence intervals were calculated from 200 bootstrap samples. Subgroup analyses were conducted by age, sex, and cardiovascular disease (CVD) status.
Results
Among 13,599 participants, 1125 (8.3%) reported incident hearing loss. Dementia incidence was higher among those with hearing loss (6.6%) compared to those without (4.9%). Starting at 4 years, incident hearing loss was associated with a higher dementia risk, persisting at 8 years (RR = 1.34; 95% CI: 1.05, 1.59). This association was significant among individuals aged 50-64 years and those with CVD.
Discussion and Implications
Incident hearing loss is associated with a heightened dementia risk, particularly in midlife and among individuals with CVD. Future research should investigate the effectiveness of timely interventions aimed at preventing dementia in individuals with hearing loss.
Introduction
Hearing loss affects 20% of adults in the U.S. 1 and poses an important public health burden. Its prevalence increases with age and reaches 65.3% among adults aged ≥71 years. 2 People with hearing loss may experience subsequent social isolation and depression,3,4 which can impact lifestyle (eg, reduced physical activity, 5 poorer adherence to medical treatments 6 ). These factors are associated with dementia, which is a major public health challenge. Epidemiological research has also established that hearing loss is a risk factor for dementia,7-9 accounting for 7% of all dementia cases, which is greater than other risk factors. 9
The associations between hearing loss and dementia suggest that hearing loss may be a potential target for preventing dementia. The groundbreaking ACHIEVE trial recently demonstrated promising results in reducing cognitive decline through hearing interventions among older adults at high risk of dementia. 10 Also, existing literature has shown that risk or prevalence of dementia risk factors are higher among individuals with hearing loss, such as obesity,11,12 cardiovascular disease,13,14 and physical inactivity, 15 so that programs may be launched to reduce these risk factors for further prevention of dementia. Unlike other risk factors such as hypertension and diabetes, hearing loss can often be self-identified, allowing individuals to seek timely treatment.
Most previous studies that have reported associations between hearing loss and dementia have lacked information about the duration of hearing loss,16-21 thus preventing identification of critical intervention periods. A previous study in Taiwan, using health insurance data, reported that newly diagnosed hearing loss was associated with a higher risk of dementia over 13 years. 22 However, this association has not been evaluated in more diverse populations, and some key covariates (eg, education) were not included in the analysis. We sought to investigate the associations of new-onset hearing loss and subsequent risk of dementia in a diverse population in the U.S. In addition, as hearing loss is identified as a midlife risk factor for dementia, 9 hearing loss at different stages of life may be differently associated with risk of dementia. Similarly, it is unknown whether associations between hearing loss and incident dementia may differ between men and women or by other factors, such as cardiovascular disease. Addressing these gaps is crucial for helping individuals with incident hearing loss, particularly those with specific comorbidities, by identifying periods of increased dementia risk and enabling timely intervention.
Using the data from the Health and Retirement Study (HRS), a cohort of representative sample of late midlife (50-64 years) and late life adults (≥65 years) in the U.S., we aimed to examine the associations of incident hearing loss (from any cause) with subsequent risk of dementia. We also examined the potentially different associations in different subgroups (ie, age, sex, cardiovascular risk).
Methods
Study Population
The HRS is a longitudinal study that surveys a representative sample of adults ≥50 years in the U.S. Participants of HRS were enrolled in the cohort from 1992, and completed questionnaires every 2 years. 23 Participants in HRS were refreshed with new birth cohorts every 6 years. We created 2 subcohorts which started from Wave 10 (2010, which included the following birth cohorts: The Study of Assets and Health Dynamics Among the Oldest Old [AHEAD]: born 1923 or earlier, starting 1992; Children of Depression [CODA]: born 1924-1930, starting 1998; HRS: born 1931-1941, starting 1992; War Babies [WB]: born 1942-1947, starting 1998; Early Baby Boomer [EBB]: born 1948-1953, starting 2004) and Wave 11 (2012, which included the birth cohort of Mid Baby Boomer [MBB]: born 1954-1959, starting 2010), respectively, and each subcohort was followed up until Wave 14 (2018). The information of self-report dementia became available from Wave 10 (2010). We excluded individuals who had dementia at baseline, and who had self-report of hearing loss or use of hearing aid at the previous wave, and those with hearing aid use at baseline. We additionally excluded individuals with missing information on covariates at baseline. All participants have provided informed written consents, and participants were de-identified in the HRS datasets.
Measurement of Hearing Loss
Information of hearing loss was assessed through self-report questionnaires with the question “Is your hearing excellent, very good, good, fair, or poor (using a hearing aid as usual)?”. Participants who responded “fair” or “poor” were considered suffering hearing loss. In addition, use of hearing aid was also asked in the questionnaire with the question “Do you ever wear a hearing aid?”.
Assessment of Dementia
Dementia at each wave from 2010 was obtained via self-report or proxy (eg, family member or caregiver) reporting. Participants were asked about their diagnosis status at each wave with the question “Had you ever diagnosed them with dementia, senility, or any other serious memory impairment?”, which has been a method used in prior research that has demonstrated moderate agreement with studies employing in-home cognitive assessments for dementia.24,25
Death
Death and the year of death were obtained from household members and were matched with the National Death Index.
Covariates
In addition to incident self-perceived hearing loss, and dementia, several covariates that are associated with these conditions were included in the analysis, such as age, sex, race/ethnicity (non-Hispanic White, non-Hispanic Black, non-Hispanic other, Hispanic), education (below high school, GED, high school graduate, some college, college and above), smoking (never/former/current), body mass index (BMI), household income, prevalent hypertension, diabetes, heart disease, stroke, and cancer, which were obtained or calculated from questionnaires.
Statistical Analysis
Two subcohorts were merged for overall analysis. For descriptive analysis,
Results
Baseline Characteristics
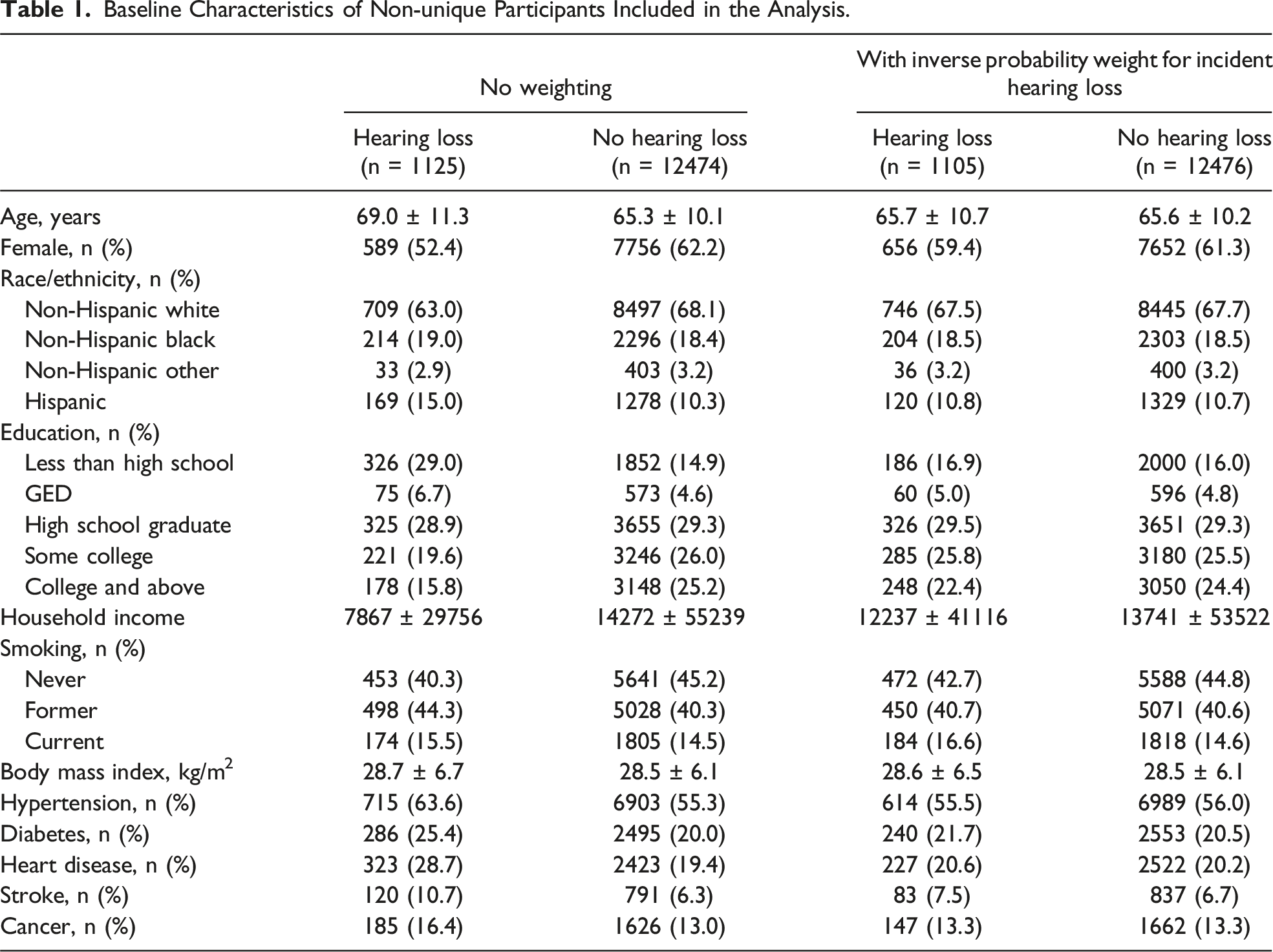
Baseline Characteristics of Non-unique Participants Included in the Analysis.
Incident Hearing Loss and Subsequent Risk of Dementia
Over 8 years of follow-up (median: 6 years), dementia occurred among 6.6% of people with incident hearing loss and 4.9% without incident hearing loss. No significant difference in risk of dementia was found between people with and without incident hearing loss after 2 years (RR = 1.55, 95% CI: 0.99, 2.12). Starting from 4 years after baseline, incident hearing loss was associated with a higher risk of dementia (RR = 1.50, 95% CI: 1.08, 1.88). Over a total of 8 years, the cumulative risk of dementia remained significantly higher among individuals with incident hearing loss (RR = 1.34, 95% CI: 1.05, 1.59) (Figure 1, Table 2). Cumulative incidence of dementia among participants with and without incident hearing loss over 8 years. Cumulative Incidence of Dementia Among Individuals With and Without Incident Hearing Loss.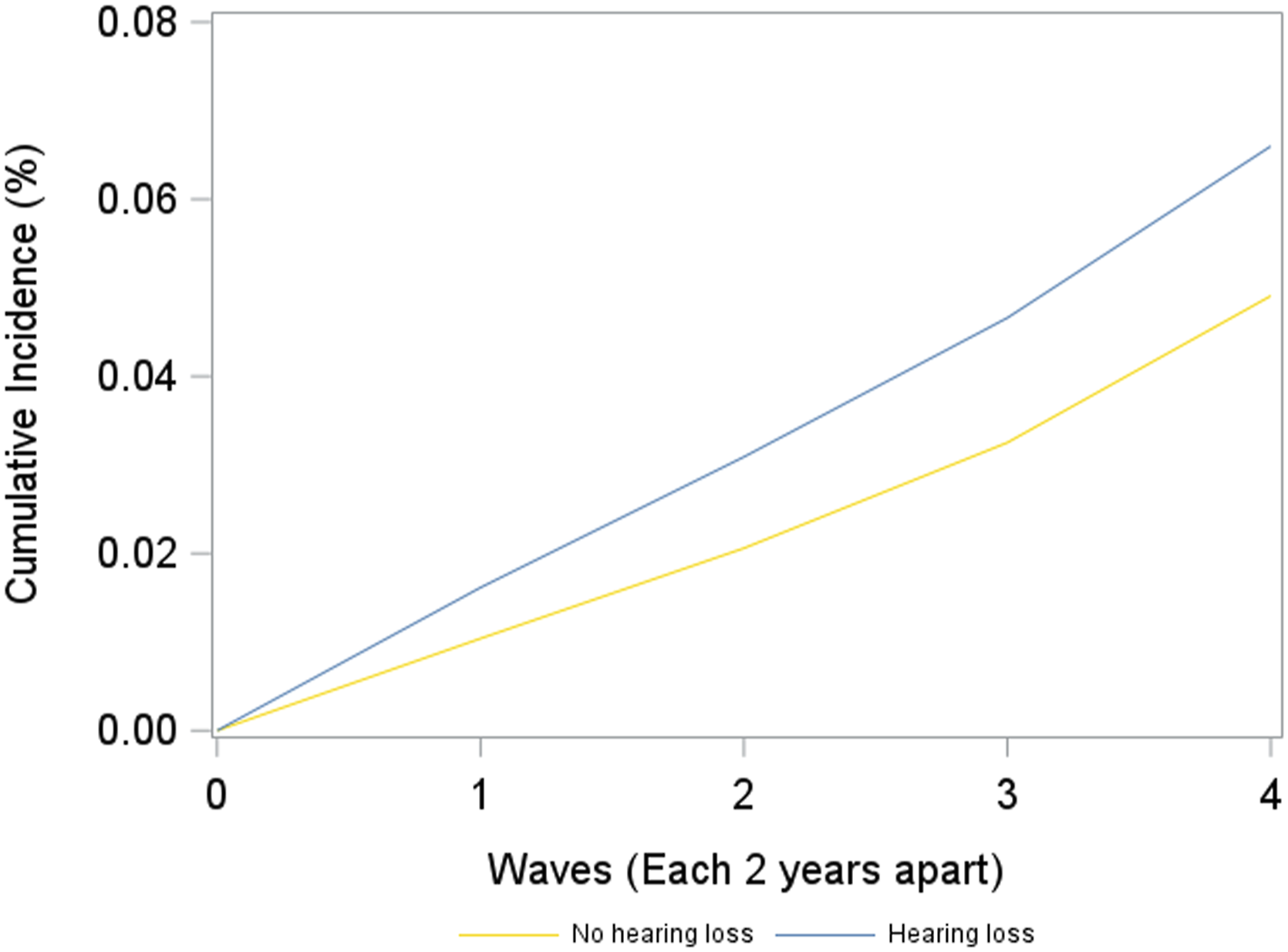

Stratified by age groups (midlife (<65 years) vs late-life adults (≥65 years)), the cumulative incidence of dementia at 8 years after baseline in persons with vs without incident hearing loss was 3.0% vs 1.5% among midlife adults and 8.8% vs 8.0% among late-life adults. Among midlife adults, incident hearing loss was associated with a higher risk of dementia 6 years (RR = 2.53, 95% CI: 1.03, 4.08) and 8 years (RR = 2.60, 95% CI: 1.30, 4.10) after baseline. However, no association was found between incident hearing loss and subsequent risk of dementia among late-life adults (Figures 2(A) and (B), Table 3). Cumulative incidence of dementia for individuals with and without incident hearing loss among participants who were (A) 50-64 years, (B) ≥65 years, (C) men, (D) women, (E) with cardiovascular disease at baseline, (F) without cardiovascular disease at baseline. Cumulative Incidence of Dementia for Individuals With and Without Incident Hearing Loss, Stratified by Age, Sex, and Cardiovascular Disease at Baseline.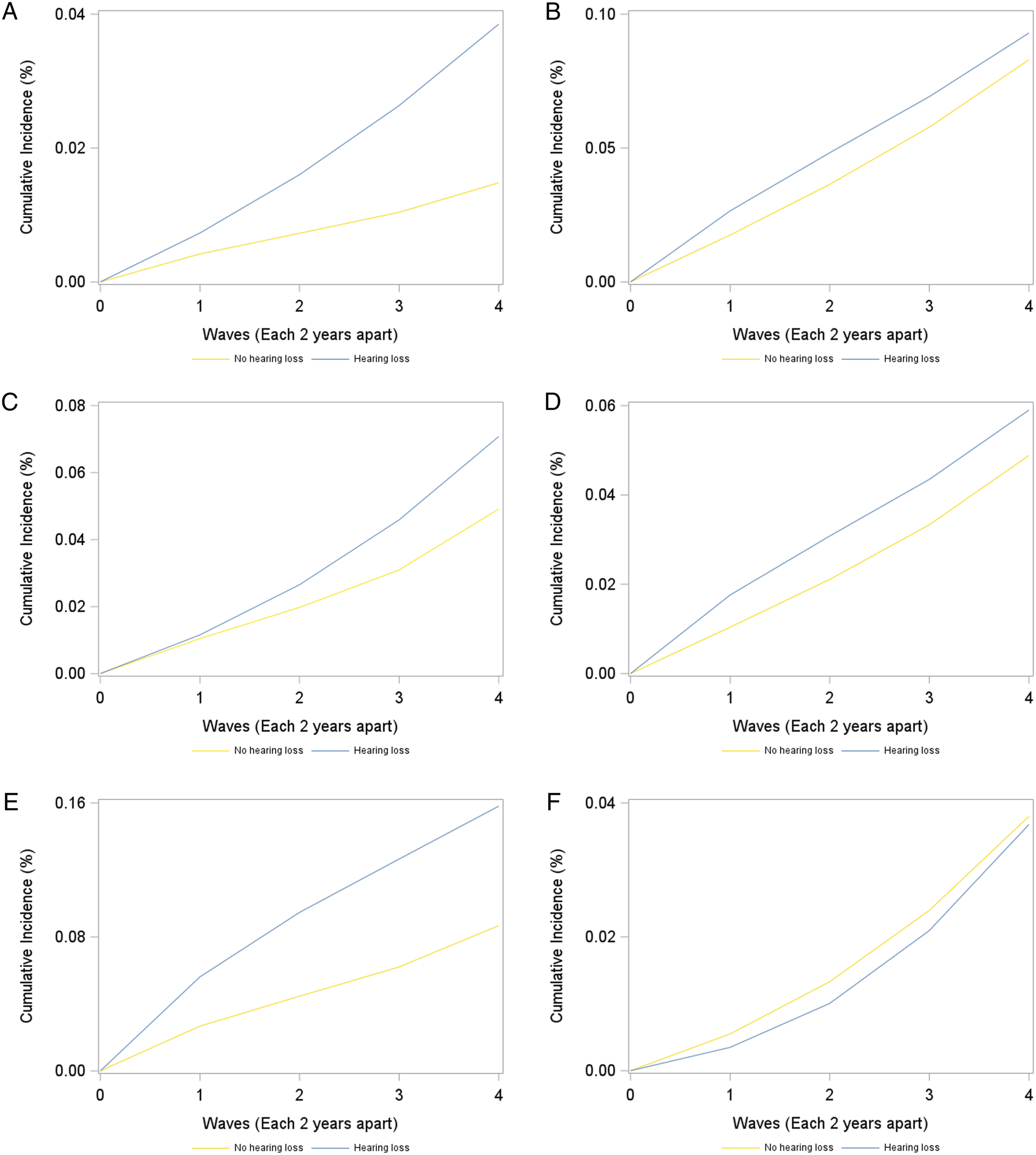
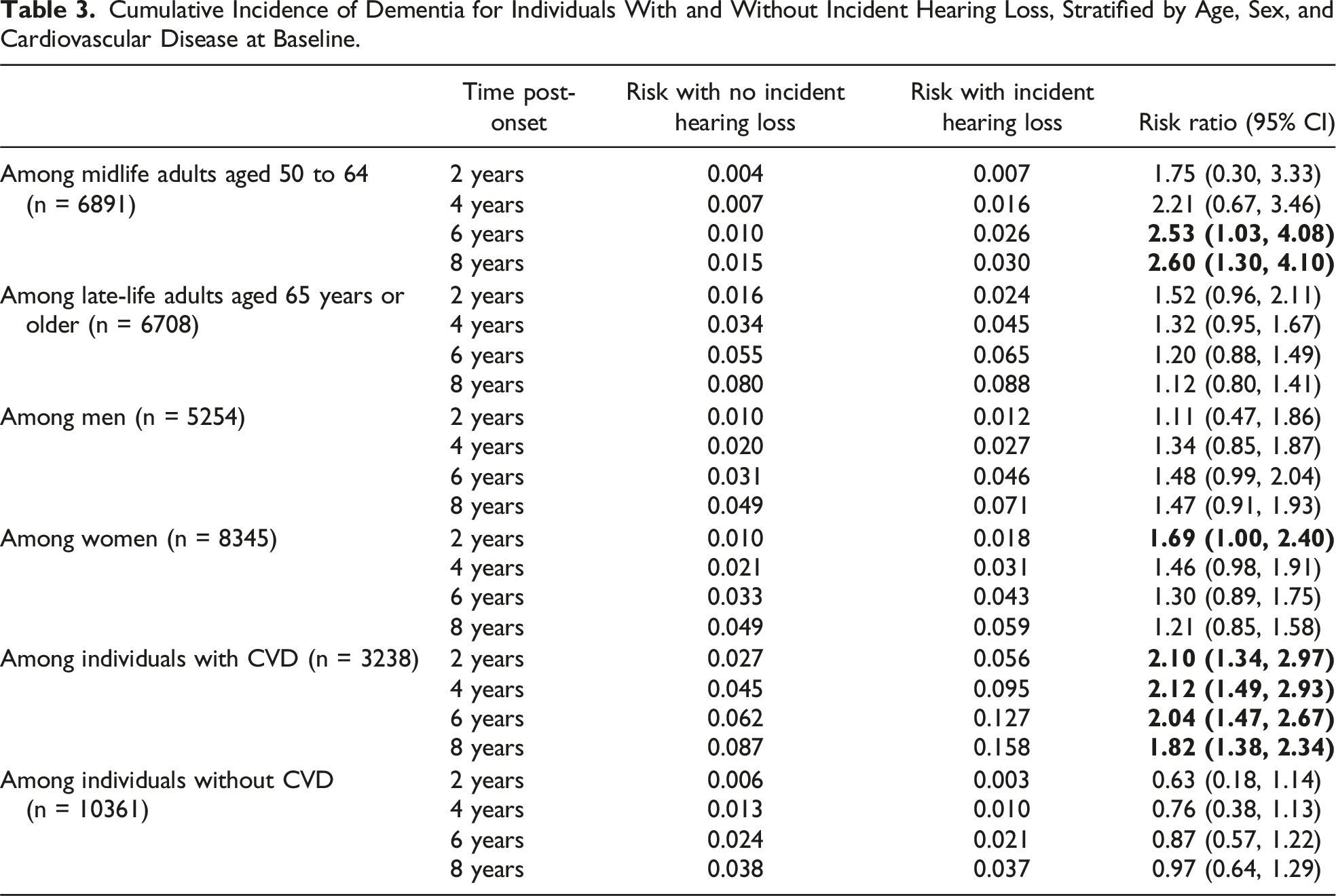
Stratified by sex, the cumulative incidence of dementia 8 years after baseline in persons with vs without incident hearing loss was 7.1% vs 4.9% in men and 5.9% vs 4.9% in women. No significant association was found between incident hearing loss and subsequent risk of dementia among men. For women, incident hearing loss was associated with a higher risk of dementia 2 years after baseline (RR = 1.69, 95% CI: 1.00, 2.40), but the associations were no longer statistically significant beyond 2 years of follow-up (Figures 2(C) and (D), Table 3).
Stratified by cardiovascular disease status, the cumulative incidence of dementia 8 years after baseline in persons with vs without incident hearing loss was 15.8% vs 8.7% among individuals with cardiovascular disease and 3.7% vs 3.8% in those without cardiovascular disease. Among people with existing heart disease or stroke, incident hearing loss was associated with a higher risk of dementia starting 2 years after baseline (RR = 2.10, 95% CI: 1.34, 2.97), which remained elevated 8 years after baseline (RR = 1.82, 95% CI: 1.38, 2.34). No association was found between incident hearing loss and risk of dementia among those without heart disease or stroke (Figures 2(E) and (F), Table 3).
Sensitivity Analysis
In a sensitivity analysis that included individuals with new use of hearing aids at baseline, dementia occurred among 6.7% of people with incident hearing loss and 5.0% without incident hearing loss. No significant difference in risk of dementia was found between people with and without incident hearing loss after 2 years (RR = 1.47, 95% CI: 1.00, 1.94). Starting from 4 years after baseline, incident hearing loss was associated with a higher risk of dementia (RR = 1.48, 95% CI: 1.14, 1.86). Over a total of 8 years, the cumulative risk of dementia remained significantly higher among individuals with incident hearing loss (RR = 1.34, 95% CI: 1.07, 1.62) (Supplemental Figure 1, Supplemental Table 1).
Discussion
In a national sample of late midlife and late-life adults in the U.S. without use of hearing aids, we found that incident hearing loss was associated with a higher risk of subsequent dementia 4 years later. This association was observed only among individuals aged 50-64 (but not ≥65) years and those with prevalent cardiovascular disease. While the association between hearing loss and dementia risk is well established, our results provide a clearer timeframe for timely treatment and intervention to further prevent dementia.
Few studies have examined the association between incident hearing loss and incident dementia. In a population-based study conducted in Taiwan with 16270 participants using the National Health Insurance Research Database, Liu et al demonstrated that newly diagnosed hearing loss was associated with a slightly higher incidence of dementia compared to those without hearing loss after applying propensity score matching (hazard ratio: 1.11; 95% CI: 1.01-1.22). 22 Moreover, the subgroup analysis showed that this association was more prominent among participants aged 45 to 64 years but not significant for participants aged 65 to 74 years or 75 years and older. Our results align with those findings, although not with other studies that reported significant associations between hearing loss and incident dementia among older adults.26,27 A possible explanation is that those studies examined prevalent hearing loss, which may have begun in midlife, rather than incident hearing loss in older adulthood. Adults who enter late life with good hearing ability may have lower risk of dementia despite late onset of hearing loss, consistent with prior evidence that hearing loss specifically in midlife is a risk factor for dementia. 9 Furthermore, the Taiwanese study suggested the association became significant 5 years after hearing loss diagnosis, 22 similar to our findings showing no association before 4 years post-baseline. Our results did not show a significant association between incident hearing loss and dementia risk in sex-stratified analyses, which differs from this previous study that the association was significant among women. 22 However, we observed a similar trend over 8 years, suggesting potential sex-based variations that warrant further investigation. Overall, by using a nationally representative sample from a diverse population, our findings contribute valuable new evidence to the existing literature.
We also found that dementia risk was significantly higher ≥2 years after incident hearing loss among individuals with cardiovascular disease. Cardiovascular disease is independently associated with a higher risk of dementia,28,29 and individuals with CVD often have comorbidities 30 such as diabetes, 31 hypertension, 32 and hyperlipidemia, 33 which all independently contribute to the development of dementia. Both hearing loss and CVD may potentially increase the risk of depression,34,35 which is associated with a higher risk of dementia. 36 Also, some pathological processes common in CVD, such as chronic stress and inflammation, may exacerbate pathology of dementia.37,38 The evidence to date suggests that hearing loss, when combined with cardiovascular disease, may be associated with accelerated dementia progression, highlighting the need for early treatment targeting both conditions. Given the high prevalence of heart disease and stroke among older adults, many of whom also experience hearing loss, public health efforts should focus on promoting timely treatment. Educational programs encouraging the use of hearing aids could be evaluated as a potential strategy to mitigate dementia risk. 39
Our findings have important public health implications. First, newly recognized self-perceived hearing loss serves as a useful indicator of higher dementia risk, particularly within 4 years or sooner among those with cardiovascular disease. These insights provide a concrete timeline for screening and intervention to prevent dementia. Health care providers, including primary care physicians, neurologists, and cardiologists, should work collaboratively to identify and manage risk factors associated with hearing loss and dementia. 40 Additionally, hearing loss may serve as another intervention target to prevent cognitive decline. Prior observational studies have shown that hearing aid use is associated with a lower risk of dementia,21,41 and the ACHIEVE trial demonstrated significant reductions in cognitive decline among individuals at high risk for dementia following hearing intervention. 10 Using target trial emulation, a recent work has shown that initiating hearing aids may reduce the risk of dementia among individuals with hearing loss. 42 While promising results have been shown with hearing interventions, the current prevalence of hearing aids use among people with hearing loss is low (90% of individuals reporting hearing difficulties in the Medicare did not have hearing aids 43 ), which could be attributed to the low coverage of hearing aids in traditional Medicare. 44 Therefore, promoting accommodation of timely treatment for hearing loss in the U.S. health care system could be essential for public health professionals, 45 given the high prevalence of hearing loss in the U.S. and the associations between hearing loss and the risk of dementia.
It is important to note that our study relied on self-perceived hearing loss, which may differ from clinical diagnoses. The World Health Organization defines hearing loss as pure-tone average of hearing thresholds >25 dB at 0.5, 1, 2, and 4 kHz in the better hearing ear among adults, 46 as also in prior epidemiological studies. 8 While self-reported hearing loss can be subjective, it also offers some advantages. Individuals who perceive hearing difficulties may be more proactive in seeking medical care, even if their hearing loss is not clinically significant. Furthermore, self-perceived hearing loss can negatively affect quality of life, leading to social isolation and depression, which are also risk factors for dementia. Future studies should explore how different measures of hearing loss—whether self-reported or clinically diagnosed—affect cognitive outcomes and identify the pathways connecting hearing loss to dementia.
Our study has several limitations. First, dementia diagnoses were based on self-reports, which may introduce misclassification. Additionally, unmeasured confounding factors such as access to an otolaryngologist or a neurologist associated with both hearing loss and dementia could not be fully accounted for, despite our use of inverse probability weighting. Nevertheless, our large sample size, repeated measurements, and longitudinal design allowed us to investigate the association between self-perceived incident hearing loss and subsequent dementia risk over 8 years.
In conclusion, incident hearing loss is associated with an elevated risk of dementia 4 or more years later, particularly among midlife adults and individuals with cardiovascular disease. Timely interventions targeting hearing loss could play an important role in dementia prevention. Future research should explore how different measures of hearing loss affect dementia risk and assess the effectiveness of early interventions.
Supplemental Material
Supplemental Material - Incident Hearing Loss and Subsequent Risk of Dementia: The Health and Retirement Study 2010-2018
Supplemental Material for Incident Hearing Loss and Subsequent Risk of Dementia: The Health and Retirement Study 2010-2018 by Jingkai Wei, Youngran Kim, Yike Li, Donglan Zhang, and Casey Crump in Journal of Geriatric Psychiatry and Neurology
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health and Retirement Study, (RAND HRS Longitudinal File 2020 (V1)) public use dataset. Produced and distributed by the University of Michigan with funding from the National Institute on Aging (grant number NIA U01AG009740). Ann Arbor, MI, (March 2023). RAND HRS Longitudinal File 2020 (V1). Produced by the RAND Center for the Study of Aging, with funding from the National Institute on Aging and the Social Security Administration. Santa Monica, CA (March 2023). The HRS (Health and Retirement Study) is sponsored by the National Institute on Aging (grant number NIA U01AG009740) and is conducted by the University of Michigan.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
