Abstract
Background and Objectives
Non-Hispanic Black populations (NHB) have a significantly higher prevalence of dementia than non-Hispanic Whites in the U.S., and the underlying risk factors may play a role in this racial disparity. We aimed to calculate risk scores for dementia among non-Hispanic White (NHW) and non-Hispanic Black populations aged 50-64 years over a period of 10 years, and to estimate potential differences of scores between NHW and NHB.
Research Design and Methods
The Health and Retirement Study from 2006 to 2016 was used to calculate the Cardiovascular Risk Factors, Aging, and Incidence of Dementia (CAIDE) risk score, a validated score for predicting dementia risk. Weighted average CAIDE score, as well as CAIDE score for modifiable factors hypertension, obese, hypercholesterolemia, physical inactivity), and non-modifiable factors (age, sex, education) were calculated for adults aged 50-64 years with normal cognition for 2006-2008, 2010-2012, 2014-2016. The associations of race with CAIDE score and elevated CAIDE score were examined.
Results
A total of 10,871 participants were included in the analysis. The CAIDE score showed declining trends for NHB from 2006 to 2016, while NHB consistently had a higher total CAIDE score and CAIDE score for modifiable factors from 2006 to 2016, but not for non-modifiable factors.
Discussion and Implications
NHB had a higher level of dementia risk factors than NHW among adults aged 50-64 years in the U.S. from 2006 to 2016, and the difference is attributable to modifiable risk factors, which holds promise for risk reduction of dementia.
Introduction
Dementia is a major public health issue in the U.S., with an estimated 6.5 million American adults aged 65 or above currently living with Alzheimer’s dementia. 1 Although the overall prevalence of dementia has decreased, there are consistent disparities in dementia by race. 2 For example, non-Hispanic Black (NHB) populations have a higher prevalence of dementia compared to non-Hispanic White (NHW) populations. 3 However, despite these observed disparities in different racial groups, the reasons for these differences remain unclear. As the process of cognitive decline may start early in midlife, prevention strategies for dementia may need to be based on established risk factors from midlife.
A variety of modifiable and nonmodifiable risk factors for dementia have been identified. In addition to nonmodifiable risk factors such as age, sex, and education level, cardiovascular risk factors, particularly those in midlife, are considered significant risk factors for dementia.4–6 Dementia in late life (≥65 years) may reflect the severity of midlife risk factors for dementia, and the prevalence of cardiovascular risk factors, such as high blood pressure, 7 obesity, 8 and high cholesterol 9 prior to late life are particularly important, with better prediction of incident dementia. Since NHB populations have a higher prevalence of dementia than NHW over time, it is likely that NHB populations may have a more severe profile of risk factors for dementia in midlife. Existing literature demonstrates that NHB populations have a higher prevalence of several risk factors for dementia, including hypertension, 10 obesity, 11 and physical inactivity, 12 and some other research showed differences in risk factors by race/ethnicity for dementia among older adults.13,14 However, less is known about evidence regarding racial disparities in the profile of dementia risk factors in midlife.
To better quantify profile of risk factors for dementia in midlife, the Cardiovascular Risk Factors, Aging, and Incidence of Dementia (CAIDE) risk score, which includes age, sex, educational level, hypertension, hypercholesterolemia, obesity, and physical inactivity have been a validated measure for predicting 20-year risk of dementia among midlife adults, with a higher score indicating a greater risk of dementia.15,16 A higher CAIDE score has been associated with poorer neurocognitive outcomes in observational studies.17–20 The CAIDE score, the only midlife risk score for dementia available, 21 covers a wide range of risk factors, with a focus on modifiable risk factors. Also, the CAIDE has an advantage on its simplicity and accessibility to calculate the risk of dementia over a long period of time. Given these strengths, the CAIDE score in a representative sample in the U.S. may be used to compare between different racial groups to identify potential differences in the profile of risk factors for dementia. Furthermore, it is essential to examine whether potential racial differences are due to modifiable (hypertension, obesity, hypercholesterolemia, physical inactivity) or non-modifiable factors (age, sex, education), so that timely treatment and intervention may be implemented.
Using data from the Health and Retirement Study (HRS), we aimed to calculate CAIDE scores among NHW and NHB over a period of 10 years among adults aged 50 to 64 years in the U.S. We also aimed to estimate potential differences in CAIDE scores and prevalence of high dementia risk between NHW and NHB.
Methods
Study Population
Anonymized data and materials have been made publicly available at the website for the Health and Retirement Study and can be accessed at https://hrs.isr.umich.edu/data-products.
The HRS is a nationally representative, longitudinal study of adults aged over 50 years, and their spouses. 22 Since 1992, participants have biennially completed questionnaires on health-related issues. Starting from Wave 8 (2006), about 50% of the participants were randomly selected to have their blood pressure and blood-based biomarkers measured. The remaining half underwent the same process in Wave 9 (2008). Therefore, blood pressure and blood-based biomarkers were measured for each half of the sample every 4 years. The HRS received approval from the Institutional Review Boards at the University of Michigan and the National Institute on Aging, and all participants provided written consent.
For this study, we included NHW and NHB participants aged 50 to 64 years from six waves from 2006 to 2016 (Wave 8 (2006), Wave 9 (2008), Wave 10 (2010), Wave 11 (2012), Wave 12 (2014), Wave 13 (2016)). Participants may appear in more than one wave. At each wave, we included those who had normal cognition and had their blood pressure and blood-based biomarkers measured. We excluded participants with any missing information on age, sex, race, education, blood pressure, total cholesterol, body mass index, or physical activity.
Measurements of Variables for Analysis
Information on race (White, Black, other) and ethnicity (Hispanic or not), age (years), sex (men, women), education (years of education) of participants was obtained using questionnaires through the HRS. Body mass index (BMI) was calculated from self-reported weight and height at each wave, and obesity was defined as BMI >30 kg/m2. Information on physical activity was also obtained by self-report at each wave, with questions asking about leisure-time and work-related vigorous and moderate physical activity. Participants were asked to report frequency of physical activity as every day, more than once per week, once per week, one to three times per month, or never. Participants at each wave reporting never for both vigorous and moderate physical activity were categorized as physically inactive. Blood pressure was measured by trained interviewers using automated sphygmomanometer, 23 and hypertension was defined as systolic blood pressure ≥140 mmHg. Total cholesterol was assayed with blood droplets on filter paper, 24 and hypercholesterolemia was defined as total cholesterol ≥6.5 mmol/L.
Construction for CAIDE Score.
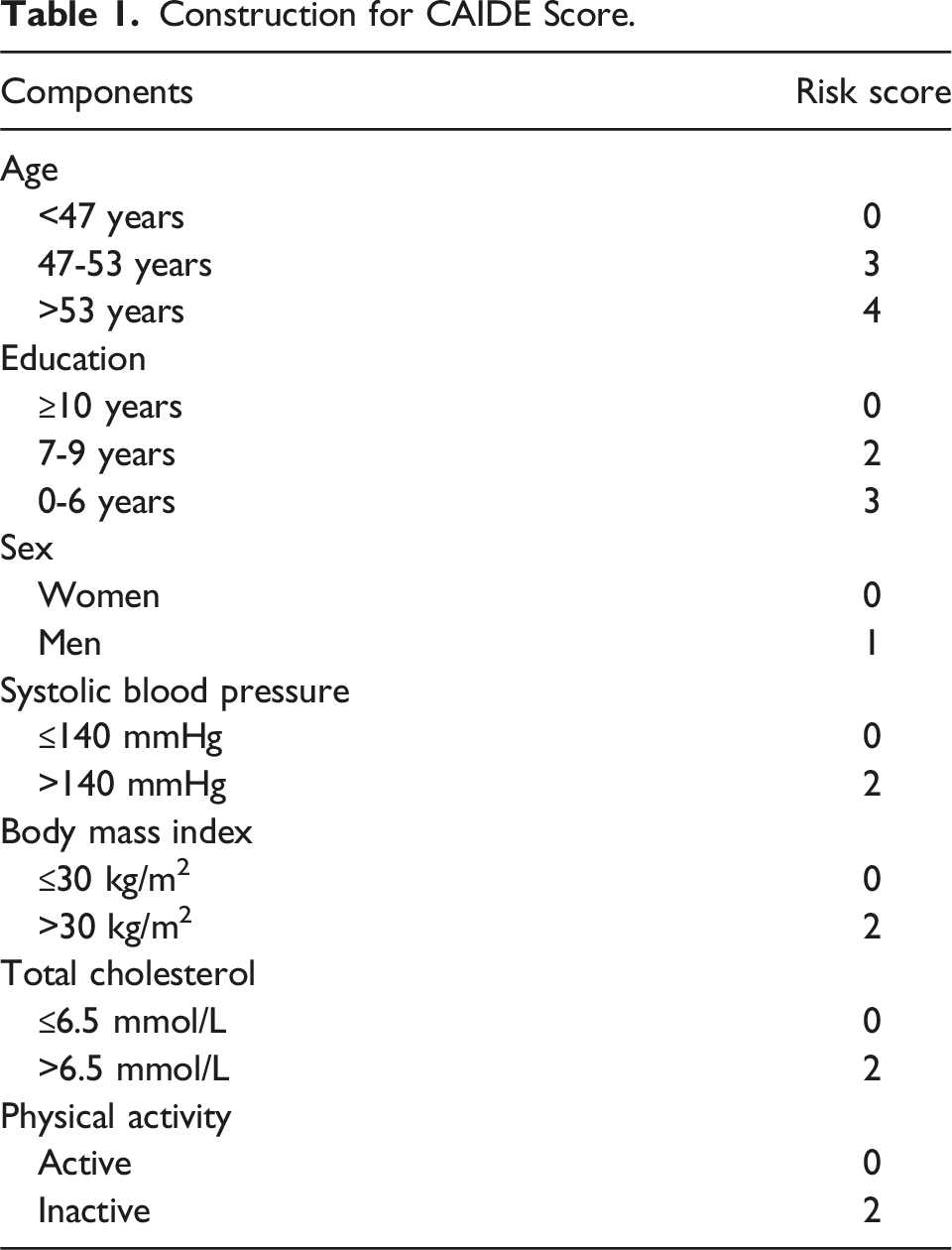
Other Variables
In addition to components of the CAIDE score, some additional variables were included for analysis. Specifically, cognitive status was determined by the Langa 26 -Weir Classification of Cognitive Function. 26 The Langa 26 -Weir Classifications was based on the summary scores of cognitive tests (including immediate and delayed recall test for memory (0 to 20 points), serial 7 s subtraction test for working memory (0 to 5 points), and counting backwards from 20 for speed of mental processing (0 to 2 points)), which were conducted with telephone interview. 26 The 27-point scale created by the Lang-Weir Classifications included three categories: normal (12-27), cognitively impaired but not demented (7-11), and demented (0-6). Only those who were categorized as normal were included in the analysis. Information about total household income (continuous amount) and health insurance (having any vs not) was obtained through self-report. The Langa-Weir Classification has been validated through a neuropsychiatric interview in the Aging, Demographics, and Memory Study, which shows an accuracy rate of 87% (95% confidence interval between 83% and 91%) in dementia categorization. 27
Statistical Analysis
As half of the sample provided blood pressure measures and blood-based biomarkers at each wave, the samples for Wave 8 (2006) and Wave 9 (2008), Wave 10 (2010) and Wave 11 (2012), Wave 12 (2014) and Wave 13 (2016) were combined. Three cohorts were analyzed separately for 2006-2008, 2010-2012, and 2014-2016.
Because of the complex study design of the HRS with multi-stage national probability sampling, as well as the sample weights, clustering, and stratification variables developed in the study, the sample at each wave was representative of adults over 50 years in the U.S. Because the sampling of the HRS accounted for geographic stratification and clustering, so that the issue of nonindependence was addressed if a participant attended more than one of the three cohorts.28,29 Weighted average CAIDE total scores, scores for modifiable factors, scores for non-modifiable factors, and weighted prevalence of high dementia risk were calculated for cohorts for 2006-2008, 2010-2012, and 2014-2016. P-values for linear trends for CAIDE score and prevalence of high dementia risk were calculated with unadjusted linear regression models and logistic regression models, respectively.
Cross-sectional analysis, including multivariable linear and logistic regression models, were conducted. Multivariable linear regression models were conducted to examine the associations of race with CAIDE scores (including total score, score for modifiable factors, and scores for non-modifiable factors) for each cohort, adjusted for total household income and health insurance. Multivariable logistic regression models were conducted to examine the associations of race with high dementia risk with three models, including basic model (adjusted for total household income and health insurance), basic model with adjustment for non-modifiable factors (adjusted for total household income, health insurance, age, sex, education), and basic model with adjustment for modifiable factors (adjusted for total household income, health insurance, hypertension, obesity, hypercholesterolemia, physical inactivity). Sampling weights were applied to all the analyses above. All analyses were conducted using SAS 9.4 (Cary, NC).
Results
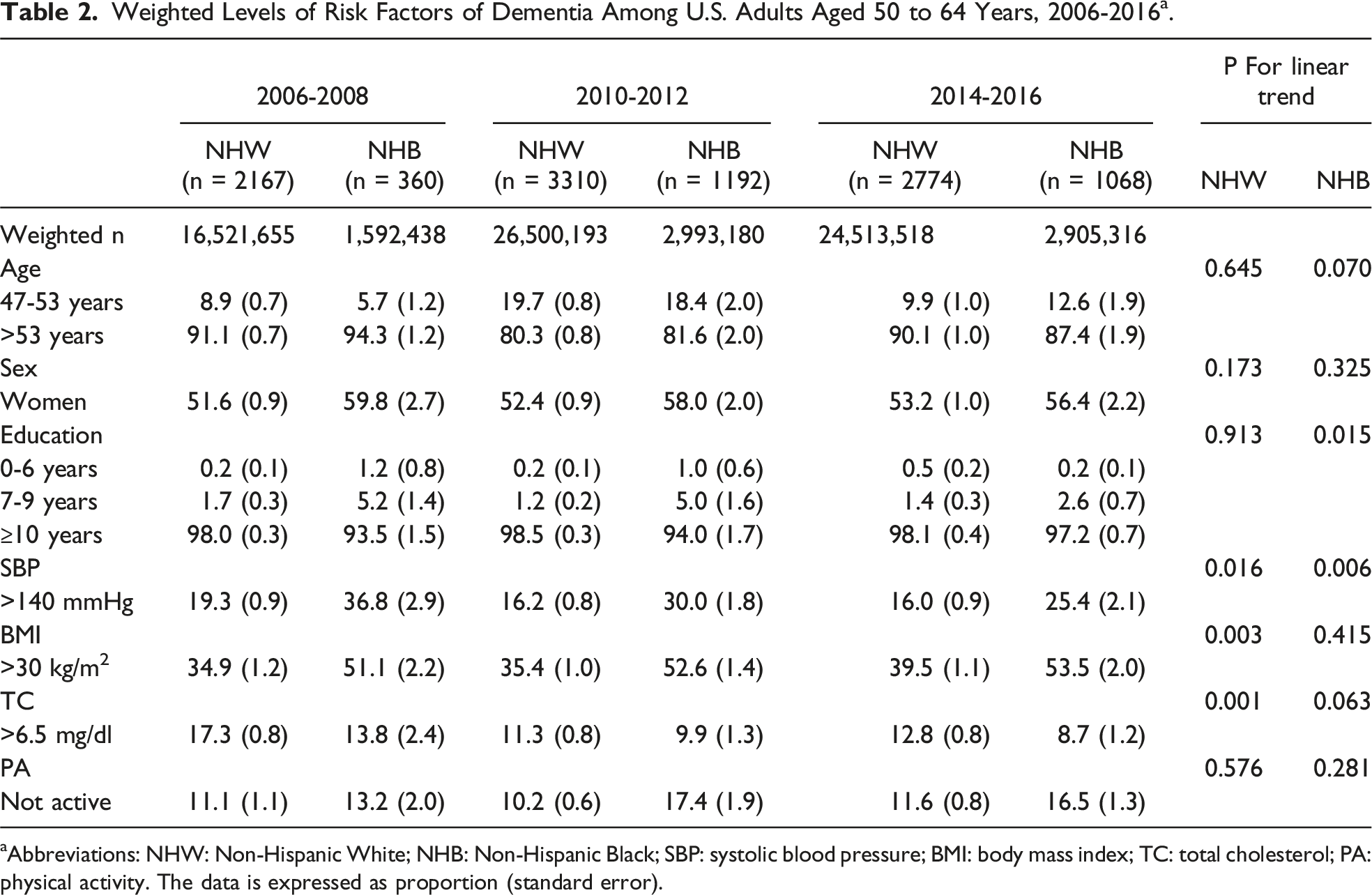
After applying eligibility criteria, 2,527, 4,502, and 3842 non-unique participants were included in cohorts for 2006-2008, 2010-2012, and 2014-2016, respectively (Figure 1). The characteristics of participants included in the study were shown in Table 2. Among all non-unique participants (N = 10,871), the weighted average age was 57.9 years, with 53.0% being women. NHB accounted for 10.0% of the study population. Most participants were older than 53 years, and the proportion of women was higher than men. More than 90% of participants had 10 or more years of education. The prevalence of having 10 or more years of education was improving among NHB (P = 0.015). The proportion of participants with 10 or more years of education was higher among NHW in 2006-2008 and 2010-2012, but not statistically different from that of NHB in 2014-2016. The prevalence of hypertension decreased over time among NHW (P = 0.016) and NHB (P = 0.006), and NHB consistently had a higher prevalence than NHW. NHW had an increasing trend of prevalence of obesity (P = 0.003), and NHB consistently had a higher prevalence than NHW. The prevalence of hypercholesterolemia was decreasing among NHW (P = 0.001), and NHW had a higher prevalence than NHB in 2014-2016. No statistically significant change was found for prevalence of physical inactivity from 2006 to 2016, and NHB had a higher prevalence than NHW in 2010-2016. Flowchart of participant selection for (A) 2006-2008, (B) 2010-2012, (C) 2014-2016 into the analysis. Weighted Levels of Risk Factors of Dementia Among U.S. Adults Aged 50 to 64 Years, 2006-2016
a
. aAbbreviations: NHW: Non-Hispanic White; NHB: Non-Hispanic Black; SBP: systolic blood pressure; BMI: body mass index; TC: total cholesterol; PA: physical activity. The data is expressed as proportion (standard error).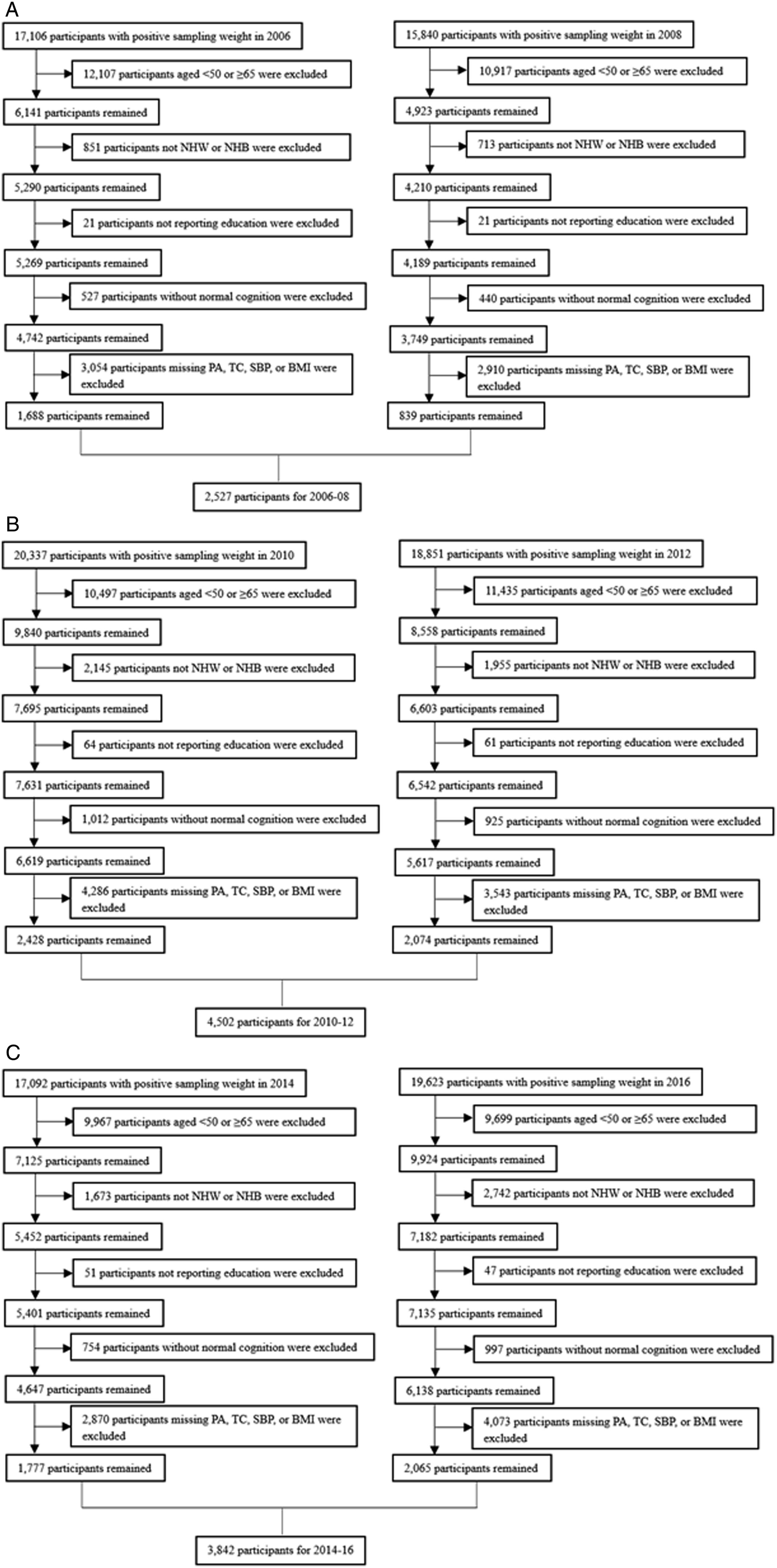
The Associations of Race With CAIDE Score Among Adults Aged 50 to 64 years in the U.S., 2006-2016 a .
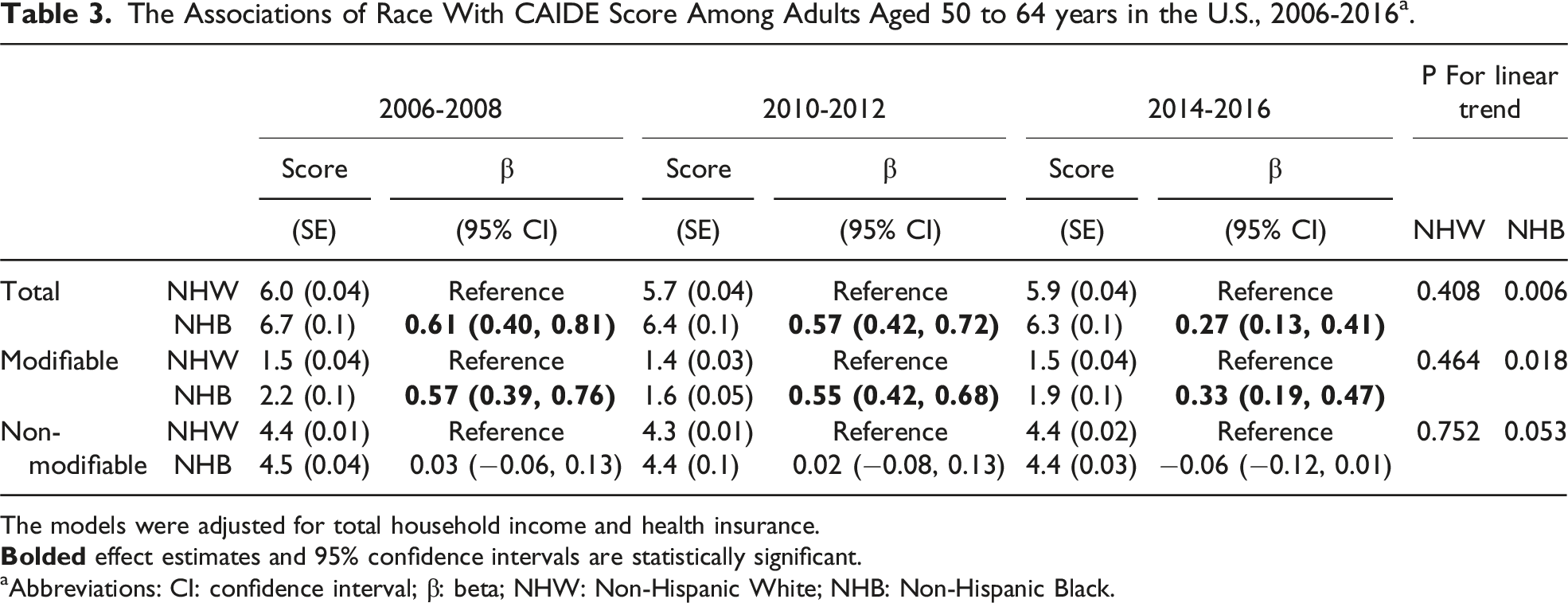
The models were adjusted for total household income and health insurance.
aAbbreviations: CI: confidence interval; β: beta; NHW: Non-Hispanic White; NHB: Non-Hispanic Black.
The Associations of Race With High Dementia Risk Among Participants Aged 50 to 64 years in the U.S., 2006-2016a.
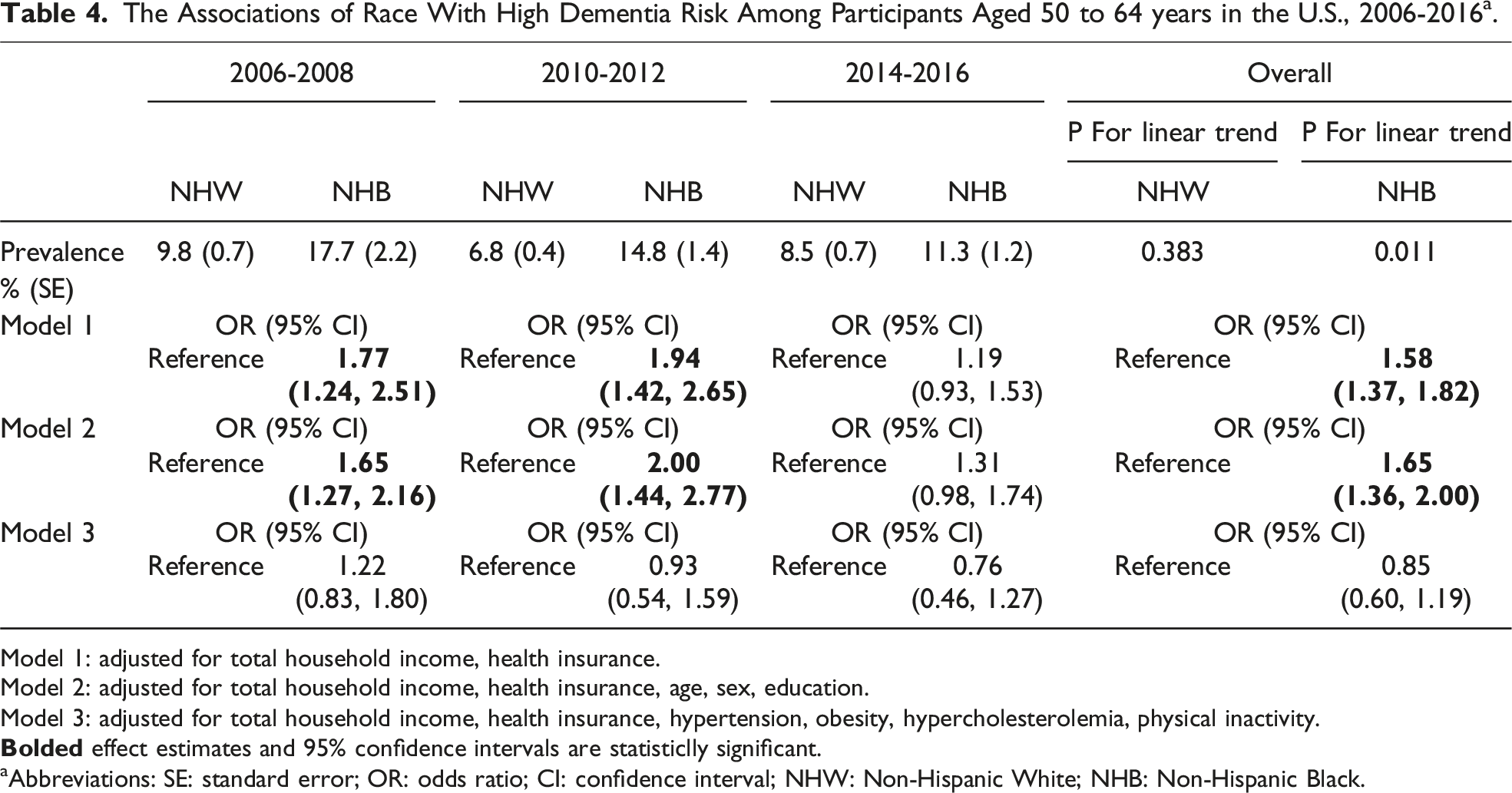
Model 1: adjusted for total household income, health insurance.
Model 2: adjusted for total household income, health insurance, age, sex, education.
Model 3: adjusted for total household income, health insurance, hypertension, obesity, hypercholesterolemia, physical inactivity.
aAbbreviations: SE: standard error; OR: odds ratio; CI: confidence interval; NHW: Non-Hispanic White; NHB: Non-Hispanic Black.
Discussion
In the present study, using a nationally representative sample, we found that NHB consistently had a higher CAIDE score than NHW among adults aged 50 to 64 years in the U.S. after controlling for total household income and health insurance, although the difference was getting smaller over 10 years. This difference was attributable to the CAIDE score for modifiable factors.
It is noticeable that the CAIDE scores, particularly among NHB were showing a declining trend. This trend may be attributed to several factors. Improved public health initiatives for better dietary habits, increased physical activity, and greater awareness of cardiovascular health may have contributed to lower blood pressure levels among the general population. Additionally, advances in medical treatments and more active screening and management of hypertension have likely played crucial roles. The shift towards personalized medicine, with tailored treatment plans for managing hypertension and other cardiovascular risk factors, could also be enhancing the effectiveness of interventions. These combined efforts not only improve individual health outcomes, but also reduce the burden of risk factors that contribute to the CAIDE score, which may predict a lower risk of dementia in the future. In addition, the improvement of social factors may contribute to the reduction of dementia risk factors, particularly the modifiable risk factors.
Previous studies have found that the CAIDE score is associated with cognitive outcomes, including poor cognitive function, 30 cognitive decline, 25 higher levels of biomarkers of neurodegeneration, 31 cerebral small vessel disease, 32 cognitive impairment, 33 and incident dementia. 20 As the CAIDE score is composed of a series of important risk factors for dementia, it is not only a useful tool for prediction of incident dementia, but also a profile indicating the overall levels of risk factors for dementia, which helps with the surveillance of long-term dementia risk. We found that NHB had a higher prevalence of elevated long-term risk of dementia compared to NHW from 2006 to 2016, which is consistent with the previous findings that NHB had persistently higher prevalence of dementia. 3 For both NHW and NHB, timely treatment and intervention of modifiable risk factors among individuals with elevated levels of dementia risk factors, and those at high risk of incident dementia may be beneficial. Given our findings showing that NHB had a higher prevalence of high dementia risk, and high CAIDE score is predictive of incident dementia, reducing the prevalence of high dementia risk may reduce the racial disparity of dementia between NHW and NHB.
In addition to treating individuals with high dementia risk, it is also essential to reduce the levels of risk factors in the whole population as our study showed that NHB had higher CAIDE scores than NHW. Similar to previous research, a higher CAIDE score was associated with poor cognitive outcomes, regardless of reaching the cutoff of elevated risk factors of dementia. 20 Therefore, the goal for interventions is not just among high-risk individuals to reduce the prevalence of high risk of dementia, but also reduce the levels of risk factors among the whole population. Since the overall CAIDE score among NHB is higher than NHW, if the same level of CAIDE score can be achieved between NHW and NHB, the racial disparity of dementia may be reduced.
Another important finding from our study is that the disparity of dementia risk factors between NHW and NHB was mainly attributed to modifiable risk factors, including behavioral factors (physical inactivity) and clinical conditions (hypertension, obesity, hypercholesterolemia). This has important implications. Efforts have been made to improve the “macro environment” to reduce disparity of dementia risk factors between NHW and NHB. This can be reflected in the levels of education received. While NHW had a higher prevalence of having 10 or more years of education than NHB from 2006 to 2012, no difference existed in 2014-2016. Because of this change, the non-modifiable part of the CAIDE score was even significantly lower among NHB than NHW. Therefore, the focus for reducing racial disparities of dementia should be on modifiable risk factors, as NHB had higher CAIDE scores for modifiable factors, even after adjustment for clinical conditions. This leaves promise for minimizing racial disparities of dementia, as all these modifiable risk factors can be improved through intervention. Since there are multiple modifiable risk factors of dementia, it is desirable that multidomain interventions addressing these risk factors be conducted among individuals with elevated CAIDE scores. As inspired by the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) trial, targeting multiple risk factors simultaneously may increase the effect of preventing dementia. 34 In addition to this, another important factor that may potentially affect the difference of modifiable risk factors between NHW and NHB is the access to health care. For conditions such as hypertension and obesity, it is essential that they are treated in time. However, the current literature indicates that compared to NHW populations, NHB populations are less likely to have health insurance coverage and therefore received less health care,35,36 which may worsen their conditions and eventually lead to dementia. Future studies are recommended to examine the role of access to health care in the racial disparity of dementia.
Therefore, our study has important public health implications. There are two combinations of strategies to reduce the risk of dementia. First, the “high-risk” strategy should be combined with “population strategy”. While treatment should be conducted among individuals with immediate high risk of dementia risk, levels of risk factors need to be reduced in the whole population, which may be achieved through policy that promote activities to improve risk factors of dementia. Second, medical treatment and lifestyle should be promoted simultaneously. In addition to equally high-quality treatment on risk factors including hypertension, obesity, and hypercholesterolemia in both NHW and NHB, improvement of physical activity, which can be achieved among the whole population with little cost, should be considered another approach to reduce dementia risk. 37 It is worth noting that while education is considered non-modifiable in our analysis for adults aged 50 to 64 years, it has also been an established modifiable factor for dementia in early life. 38 Similarly, modifiable risk factors of dementia, such as blood pressure, BMI, and cholesterol, research has demonstrated that these risk factors are predictive of dementia in midlife, but not in late life.39–41 This emphasizes the importance of timing in the relationship between risk factors and dementia, as well as the early risk reduction of dementia.
This study has limitations. First, some measurements may not be accurate. For example, the measurement of physical activities was based on self-reported questionnaires without the amount of time reported, and body mass index was calculated based on self-reported weight and height, so there could be issues of misclassification. Second, the categories of cognitive status were based on results of cognitive tests but not actual diagnosis of dementia or mild cognitive impairment, and only participants with normal cognitive function were included, so we may have underestimated the prevalence of dementia risk factors. Also, our study only included participants who were 50 years or older, so we were not able to assess the level of dementia risk for younger individuals. In addition, survival bias can be another issue, since individuals with severe risk factors of dementia may be more likely to die or not respond, so they were less likely to be included in the cohort. Finally, the refreshment of the cohorts with younger participants may potentially affect the results of analysis, although the study design with sampling weights and stratification minimized the impact. Despite these limitations, with a nationally representative sample, and consecutive measurement of dementia risk factors, including accurate measurement of blood pressure and total cholesterol, we were able to examine dementia risk from 2006 to 2016 among NHW and NHB in the U.S.
In conclusion, among adults aged 50 to 64 years in the U.S., NHB had a higher level of dementia risk than NHW among adults from 2006 to 2016, and the difference is attributable to risk factors that may be modified. Multiple strategies may be taken to improve risk factors of dementia, which may promote cognitive health and reduce incident dementia among older adults.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the pilot project Carolina Center on Alzheimer’s Disease and Minority Research (to J.W.), funded by National Institute on Aging (5P30AG059294-05). Health and Retirement Study, (RAND HRS Longitudinal File 2020 (V1)) public use dataset. Produced and distributed by the University of Michigan with funding from the National Institute on Aging (grant number NIA U01AG009740). Ann Arbor, MI. RAND HRS Longitudinal File 2020 (V1). Produced by the RAND Center for the Study of Aging, with funding from the National Institute on Aging and the Social Security Administration. Santa Monica, CA. The HRS (Health and Retirement Study) is sponsored by the National Institute on Aging (grant number NIA U01AG009740) and is conducted by the University of Michigan.
