Abstract
Objective
This mixed-method pilot study aims to investigate the feasibility and preliminary effects of sport stacking on cognitive function in individuals with mild Alzheimer's disease (AD) and those with mild cognitive impairment (MCI).
Methods
Twenty-four community-dwelling subjects with confirmed mild AD or MCI were evenly randomly assigned to either the 12-week sport stacking intervention group (n = 12) or clinic routine management control group (n = 12). Outcome evaluation included the Auditory Verbal Learning Test (AVLT), Alzheimer's Disease Cooperative Study–Activities of Daily Living scale (ADCS-ADL), and plasma brain-derived neurotrophic factor (BDNF). A qualitative descriptive study was conducted to explore the overall perception and experience of the sport stacking from the subjects’ perspective.
Results
Twenty-two subjects completed the trial. At 12 weeks post-intervention, compared to the control group, the sport stacking group had significantly greater improvements in AVLT immediate recall (
Conclusion
To the best of our knowledge, this is the first study indicating that sport stacking is feasible among individuals with MCI and mild AD. The preliminary effect on episodic memory is encouraging, possibly via upregulation of BDNF.
Introduction
Dementia is a highly prevalent chronic condition characterized by cognitive decline and neuropsychiatric symptoms. 1 Among the various types of dementia, Alzheimer’s disease (AD) is most common among older adults. 2 According to Alzheimer Disease International (ADI), more than 50 million older people with AD worldwide in 2019, and this number is expected to reach 152 million by 2050, 3 with the global cost of care projected as $2 trillion in 2030. 4
One of the core treatment principles for AD is to delay the disease progression, especially when the disease is in milder and even preclinical form (i.e., mild cognitive impairment, MCI). For MCI, it is reported that 10%-15% of MCI cases will be converted to AD within the space of 12 months.5,6 At present, it is particularly challenging to identify those persons with preclinical AD using existing clinical diagnostic techniques because behavioral symptoms are typically absent at this stage. In this situation, the best way to deal with the rising cost of health care would be to introduce low-cost, non-invasive, and objective pre-clinically applied therapeutic options. The findings of several systematic reviews converged to support the benefits of cognitive interventions in delaying cognitive decline among individuals with MCI or mild AD. 7 However, in individuals with early AD, utilizing cognitive training alone frequently does not result in the required generalizability in the global cognitive state or functional ability. 8 Further, there is a current shift away from single-domain interventions, like cognitive training, in favor of integrated, multi-domain strategies that could benefit brain health generally. 9 Researchers found that people who participated in combination cognitive training programs improved the most when psychomotor training was included, to the extent that decreases in dementia-related symptoms were mostly noticed with only this additional training. 10 Also, previous preliminary studies have shown that people with early AD and MCI can benefit from cognitive-motor training to increase their global cognitive scores. 11 According to these findings, since motor and cognitive functions are interconnected and developed in parallel and interdependently in the brain, it would be beneficial to train them simultaneously.
In recent years, the concept of gamification has been applied to integrate motor and cognitive training components to support the cognitive function of persons with preclinical and clinical dementia. Some games (including “MINWii”, “Kitchen and Cooking” and “X-Torp”; game show; SHQ, Sea Hero Quest, etc.) are also applied to reduce the risk of AD and delay its progression. The researchers showed that these games could significantly improve the global cognitive function and episodic memory of patients with cognitive impairment.12-14 Moreover, studies have demonstrated that performing tasks, such as video game training (visuospatial/visuomotor tasks), resulted in improvements beyond simply visuospatial skills but also executive functions (eg, task switching, working memory, visual short-term memory, and reasoning). 15 Researchers have also proposed that there is an “interaction of large-scale brain networks during learning with diverse training procedures”. 16 All these games are highly accessible online games except for those who have barriers to the use of technology, such as people with a lower concentration, lower executive function, lower socio-economic condition, or higher technology anxiety. 17
Sport stacking (cup stacking) is a new activity with game characteristics. It began in the early 1980s and has become a globally popular physical activity led by the United States and Korea. 18 Participants use 3, 6, 9, and 12 specialized cups to make a pyramid with both hands (“stack up”). Then restore the cup to the original stack (“stack down”), which must be done in a predetermined order. 19 Many studies held in schools showed that sport stacking was beneficial for students in many aspects, such as hand-eye coordination, 20 reaction time, 21 bilateral coordination, 22 and dual hemispheric brain activity. 23
In recent years, some researchers tried to apply sport stacking to stroke patients and found that it significantly improved the reaction time of stroke patients. 24 In Korea, some researchers applied the sport stacking to healthy older people and found it feasible and acceptable for them to join this activity. According to the surveys and interviews, most participants reported benefits from the program (improved physical, psychological, and social health) and expressed their enjoyment of the sport stacking. They also reported that they would recommend it to others in the future and were willing to participate in it for a long time.18,25
Nevertheless, there is very limited application of sport stacking to the dementia context. The purpose of this pilot feasibility study was to explore the preliminary effects of sport stacking in persons with MCI or mild dementia (limited to AD for clinical cases). We hypothesized that sport stacking is feasible among individuals with MCI and mild AD, and it would improve their cognitive (global cognitive function, memory, executive function) and health outcomes (depression, activities of daily living).
Material and Methods
Study Design
This pilot randomized controlled trial (RCT) compared the effects of sport stacking with usual care on global cognitive function, episodic memory, executive function, depression, quality of sleep, and activities of daily living among persons with MCI and mild AD. A post-RCT interview was conducted to assess the feasibility and acceptance of the sport stacking. The study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University. It was conducted in compliance with the Declaration of Helsinki’s ethical standards. 26 It was registered on the Chinese Clinical Trial Registry: www.chictr.org.cn (Registration No.: ChiCTR2100045980). All patients provided written informed consent.
Participants
Participants were recruited through the geriatric memory clinic at the First Affiliated Hospital of Chongqing Medical University between February 2021 to May 2021. Inclusion criteria included: (1) aged 60 to 90, (2) Diagnosed by physicians as MCI or mild AD according to the modified Petersen criteria for the diagnosis of MCI, 27 and National Institute for Neurological and Communicative Disorders and Stroke–Alzheimer’s Disease and Related Disorders Association criteria (NINCDS-ADRDA) 28 respectively. The screening covered physical assessment, cognitive test, medical record review, and clinical laboratory screening; (3) at least 3 months of stable doses if receiving antidementia medication or mood-stabilizing medication; (4) had at least 1 family caregiver to live with or spend at least 4 hours a day for caregiving; (5) communicable with others and with adequate vision and hearing for social interaction; and (6) able to give informed consent. Participants were excluded from the study if they had: (1) severe psychiatric illness and the use of antidepressants; (2) participation in physical activity more than twice weekly on a regular basis; and (3) any medical condition precluding participation in the physical activity program (eg, severe neurological or medical illness and presence of several cardiovascular risk factors).
Eighty potential participants were eventually screened, and among them, twenty-four were eligible, all of them agreed to participate with a response rate of 100%. Eligible individuals were randomized in a 1:1 ratio to receive either the sport stacking or usual care based on a random sequence generated using a random number table, and the sequentially opaque numbered envelope was used to disclose the assignment.
Intervention
Sport Stacking Group
Sport Stacking Program for Participants.
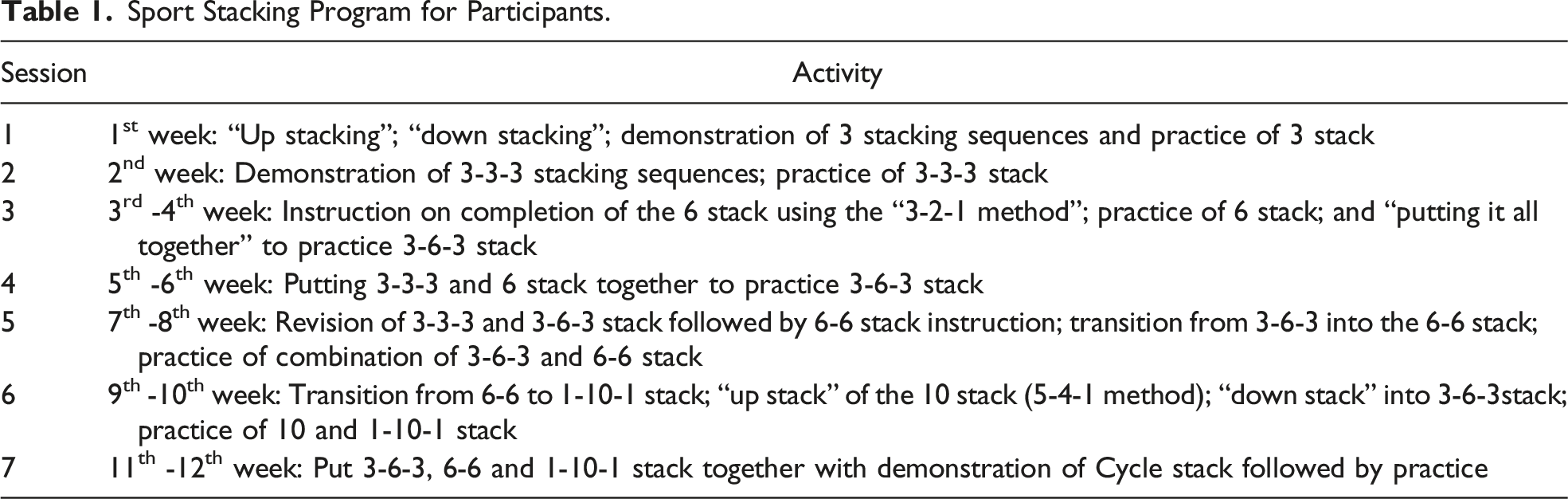

Schematic of the cup set and cup stack. (A1) Cups in an initial position and (A2) 3-6-3 pyramids in practice and retention.
To facilitate the self-practice at home, a set of online videos showing the skills trained in all 7 sessions was provided as a reminder to guide the participants’ self-practice. A self-recording logbook was given to record participants' compliance rates. The participants were asked to record each time they finished practicing sport stacking and the duration of their daily self-practice at home. The caregivers were encouraged to assist the participants in the practice and compliance recording process. The caregivers were asked to videotape the self-practice on the first and last day of each week to the research assistant for collecting feedback. These videos could be used to check the participants' mastery of the sport stacking mode and correct their inaccuracies promptly.
Control Group
Participants in the control group received routine management from the Memory Clinic, mainly including (1) Regular medication; (2) Basic health education (medication, diet, exercise, etc.). The intervention group also received the usual care.
Outcome Evaluation
The outcomes were evaluated at 3 points, including baseline before randomization, immediately at 4 weeks, and at 12 weeks thereafter. The plasma
Cognitive Function Assessment
The Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-cog) 30 was used to evaluate global cognition, behavior, and ability to manage daily life. The ADAS-cog, which scored 0 to 70, comprises 11 subscales. A higher score indicates poorer cognitive function. Cronbach's alpha was .91, and the scale showed satisfactory internal consistency reliability when used in the Chinese elder population. 31
The Auditory Verbal Learning Task (AVLT) 32 was used to assess their memory. The sum scores of the immediate recall trial and the delayed recall trial range from 0 to 45 and 0 to 15, respectively. It has good reliability and validity in the evaluation of episodic memory in patients with cognitive impairment. 33
The clock drawing test (CDT), 34 which scored 0 to 15, was used to assess executive functions. It has been widely used as a cognitive function screening tool because of its convenient operation and less influenced by the subject’s education, cultural background, and race. The CDT has good test-retest reliability, inter-rater reliability, and high levels of sensitivity and specificity for identifying cognitive decline. 35
Geriatric Depression Scale
The 30-item Geriatric Depression Scale (GDS-30) 36 was used to assess the severity of depressive symptoms. The brief statements and dichotomous response render GDS-30 an acceptable and feasible measure for depressive symptoms among the population with cognitive impairment. 37 Smarr and Keefer 38 used GDS to screen for depression with a sensitivity of .94 and a specificity of .80. The GDS-30, which scored 0 to 30, uses dichotomous questions, with high scores representing more symptoms of depression.
Activities of Daily Living Scale
The Alzheimer's Disease Cooperative Study–Activities of Daily Living scale (ADCS-ADL) 39 was used to assess the activities of daily living function of patients with AD and MCI. Galasko et al 39 found that the items comprising the ADCS-ADL had good individual sensitivity to functional decline in AD and MCI. The ADCS-ADL scores out of 78 points, with higher scores indicating better activities of daily living function. 40
ELISA Analysis of BDNF
A 5-mL blood sample was obtained by a qualified nurse from the antecubital vein to determine plasma levels of brain-derived neurotrophic factor (
Post-Intervention Interviews With Participants
Interview Questions.

Statistical Analysis
All statistical analyses were performed by IBM SPSS ver. 26.0 (IBM Corp., Armonk, USA). The Shapiro–Wilk test and the Levene test were used to determine the normality and homoscedasticity of the data. All data were expressed as mean ± standard deviation (SD). Group differences in demographics and baseline assessments were compared using the appropriate statistical tests (independent samples
Results
The CONSORT statement was used to facilitate complete and transparent reporting of this trial.
42
We started the recruitment in February 2021 and ended it in May 2021. The final follow-up visit was in August 2021. Twenty-four patients with mild AD or MCI satisfied the eligibility criteria and completed the baseline examination. The sample was composed of 11 MCI (4 males/7 females) and 13 patients with mild AD (2 males/11 females). They were randomized to receive either the intervention (n = 12) or control (n = 12). Twenty-three patients completed the 4-week follow-up, and twenty-two completed the 12-week follow-up. The flow diagram of patients through the trial and the details of dropouts are reported in Figure 2. Study flow chart.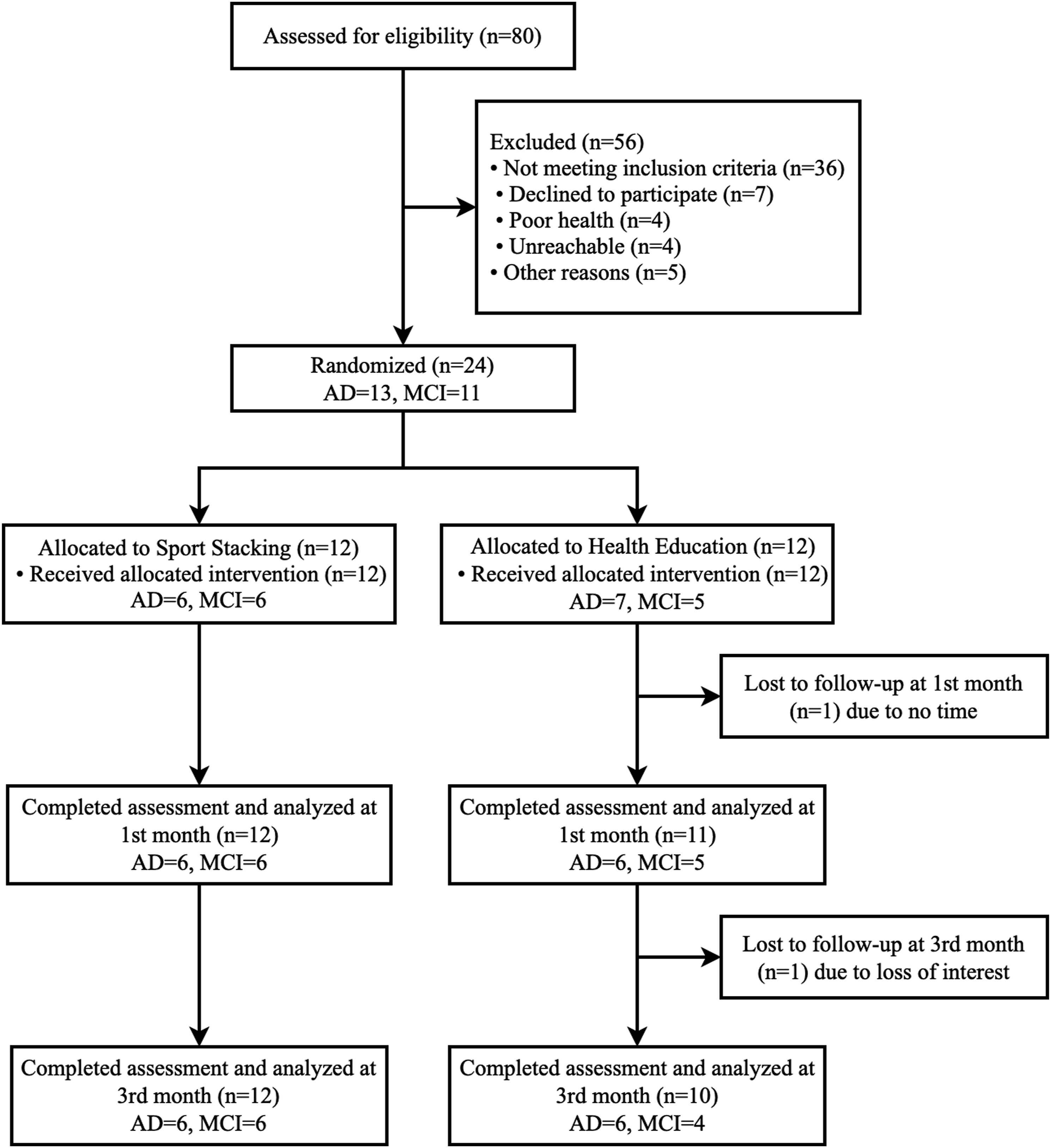
Patient Characteristics at Baseline
Demographics and Baseline Assessments of the Participants (n = 22).
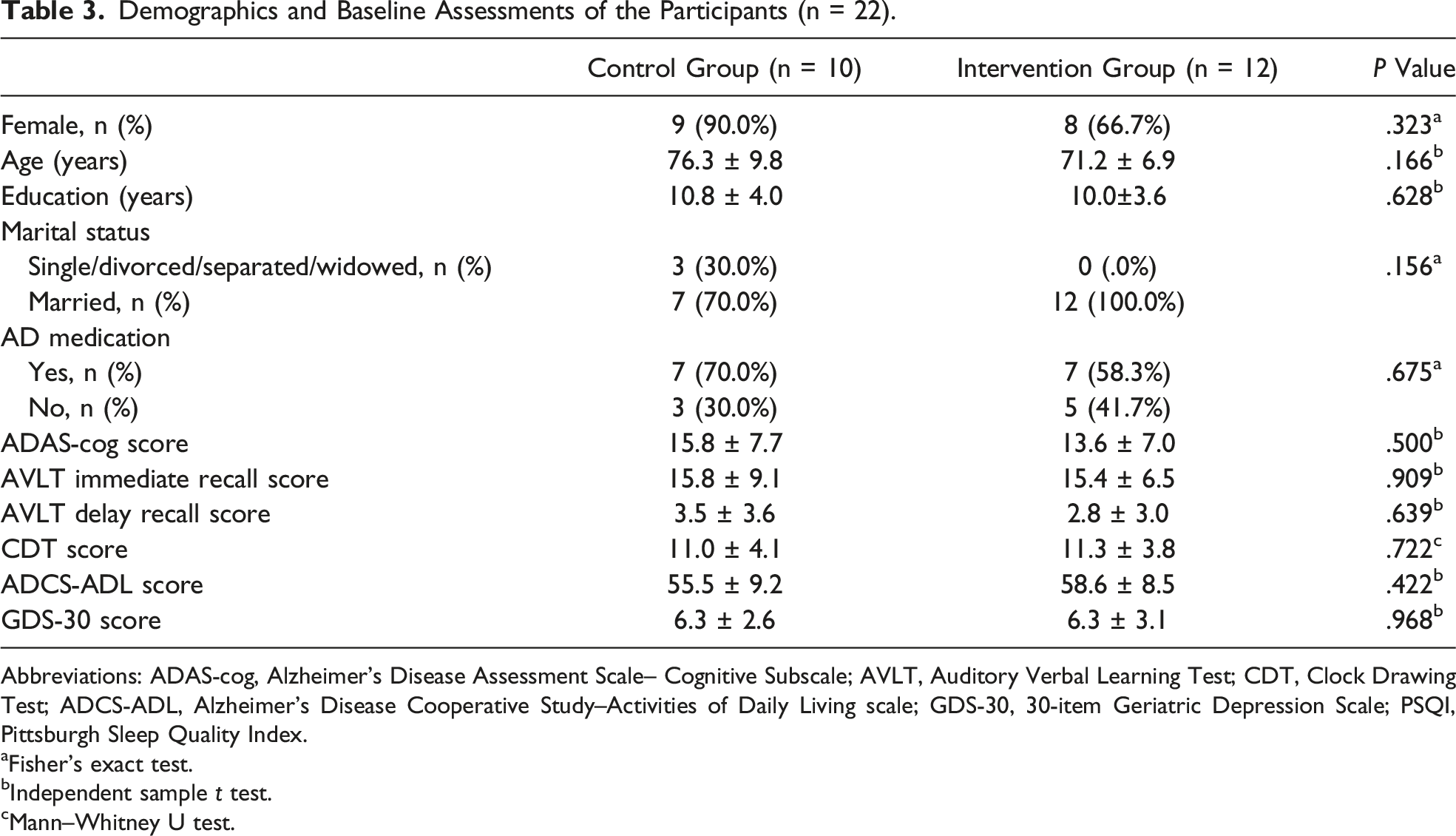
Abbreviations: ADAS-cog, Alzheimer's Disease Assessment Scale– Cognitive Subscale; AVLT, Auditory Verbal Learning Test; CDT, Clock Drawing Test; ADCS-ADL, Alzheimer's Disease Cooperative Study–Activities of Daily Living scale; GDS-30, 30-item Geriatric Depression Scale; PSQI, Pittsburgh Sleep Quality Index.
aFisher’s exact test.
bIndependent sample
cMann–Whitney U test.
Overall Sample Analysis
Comparison in Global Cognition, Memory, Executive Function, Depression Mood, and ADL Between the Groups of Overall Patients.
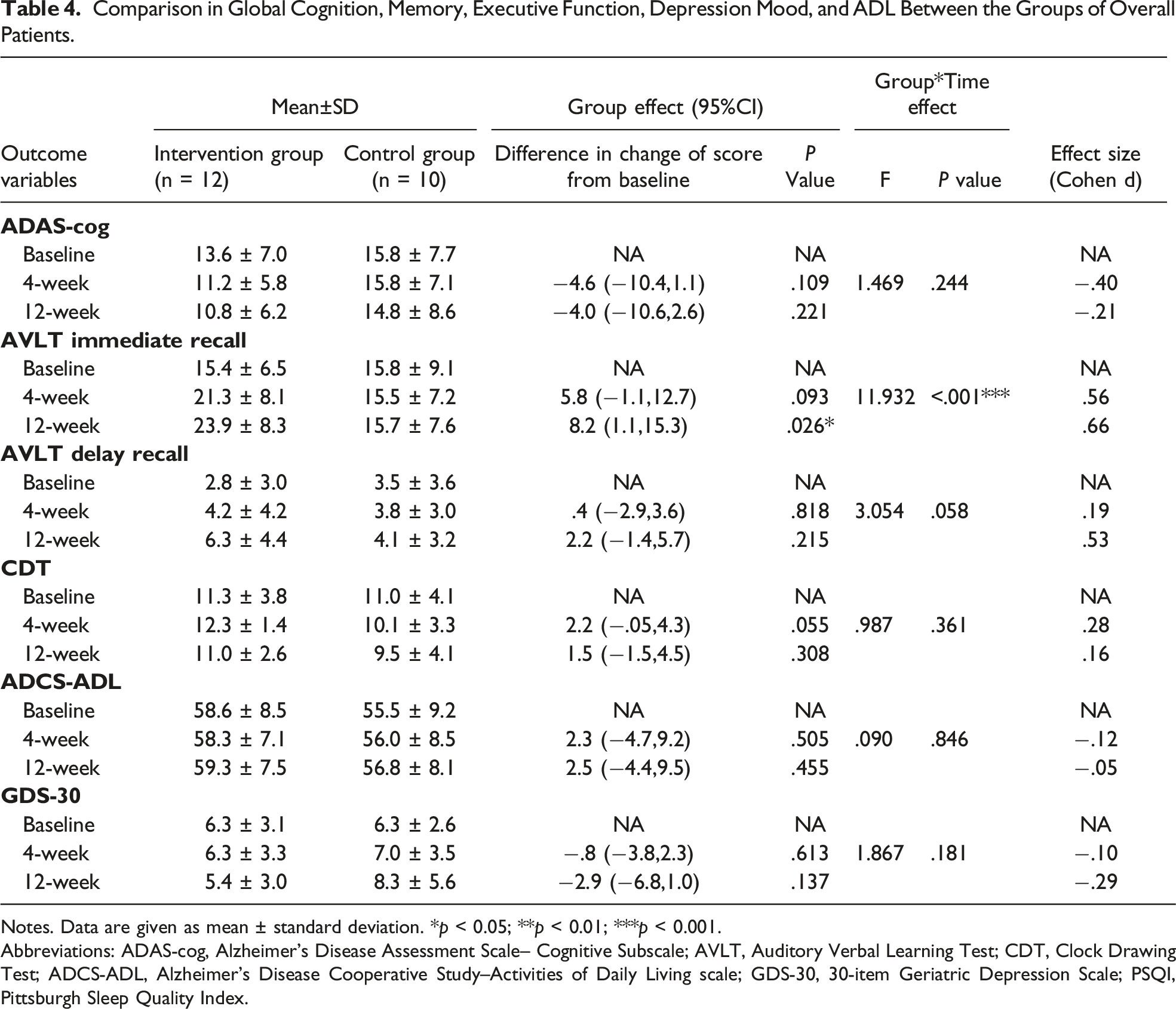
Notes. Data are given as mean ± standard deviation. *
Abbreviations: ADAS-cog, Alzheimer’s Disease Assessment Scale– Cognitive Subscale; AVLT, Auditory Verbal Learning Test; CDT, Clock Drawing Test; ADCS-ADL, Alzheimer’s Disease Cooperative Study–Activities of Daily Living scale; GDS-30, 30-item Geriatric Depression Scale; PSQI, Pittsburgh Sleep Quality Index.
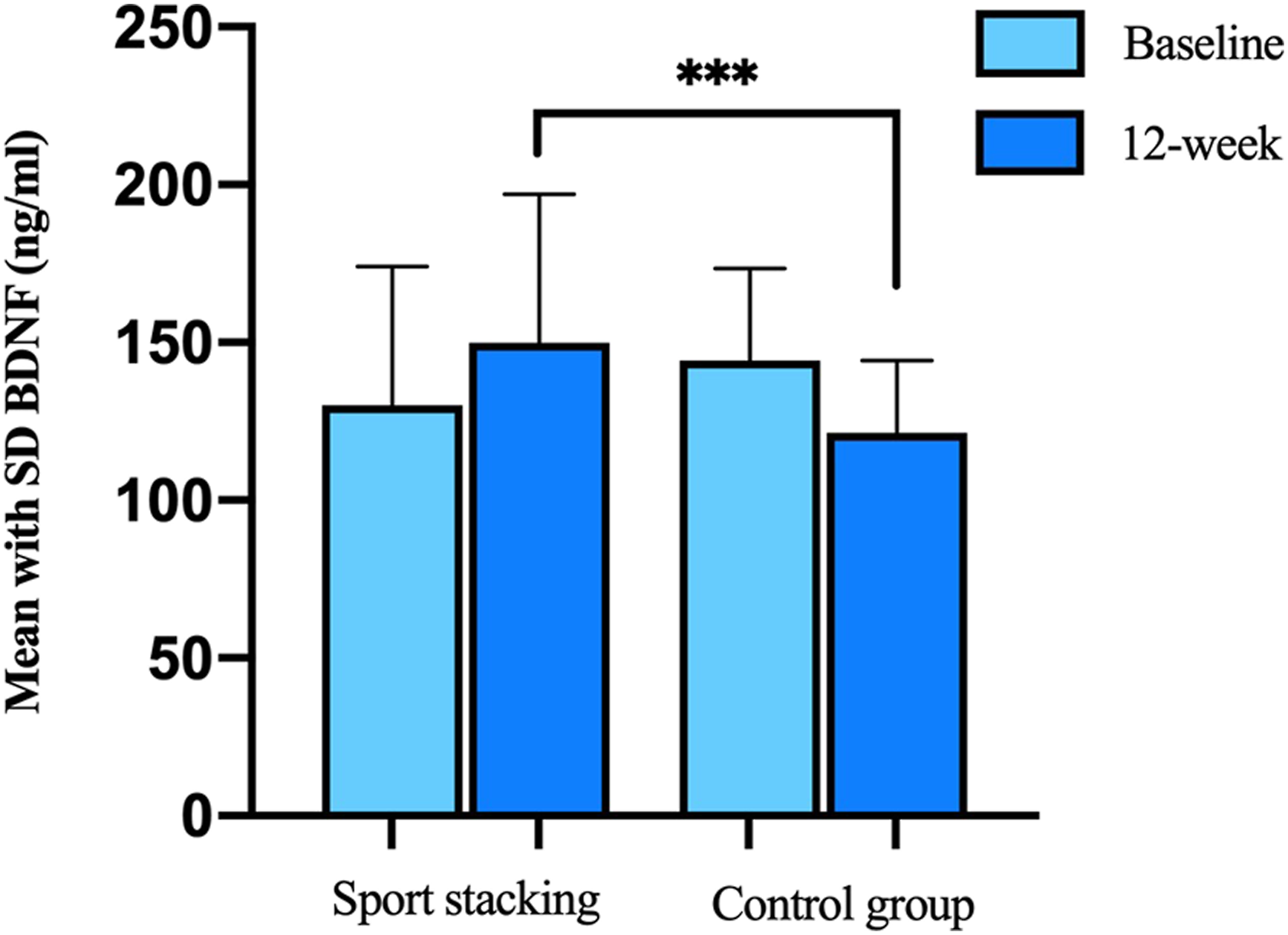
Comparison in concentration of plasma BDNF between 2 groups of overall patients. ***
Subgroup Analysis on Participants With MCI
Comparison in Global Cognition, Memory, Executive Function, Depression Mood, and ADL Between the Groups of Patients With MCI.
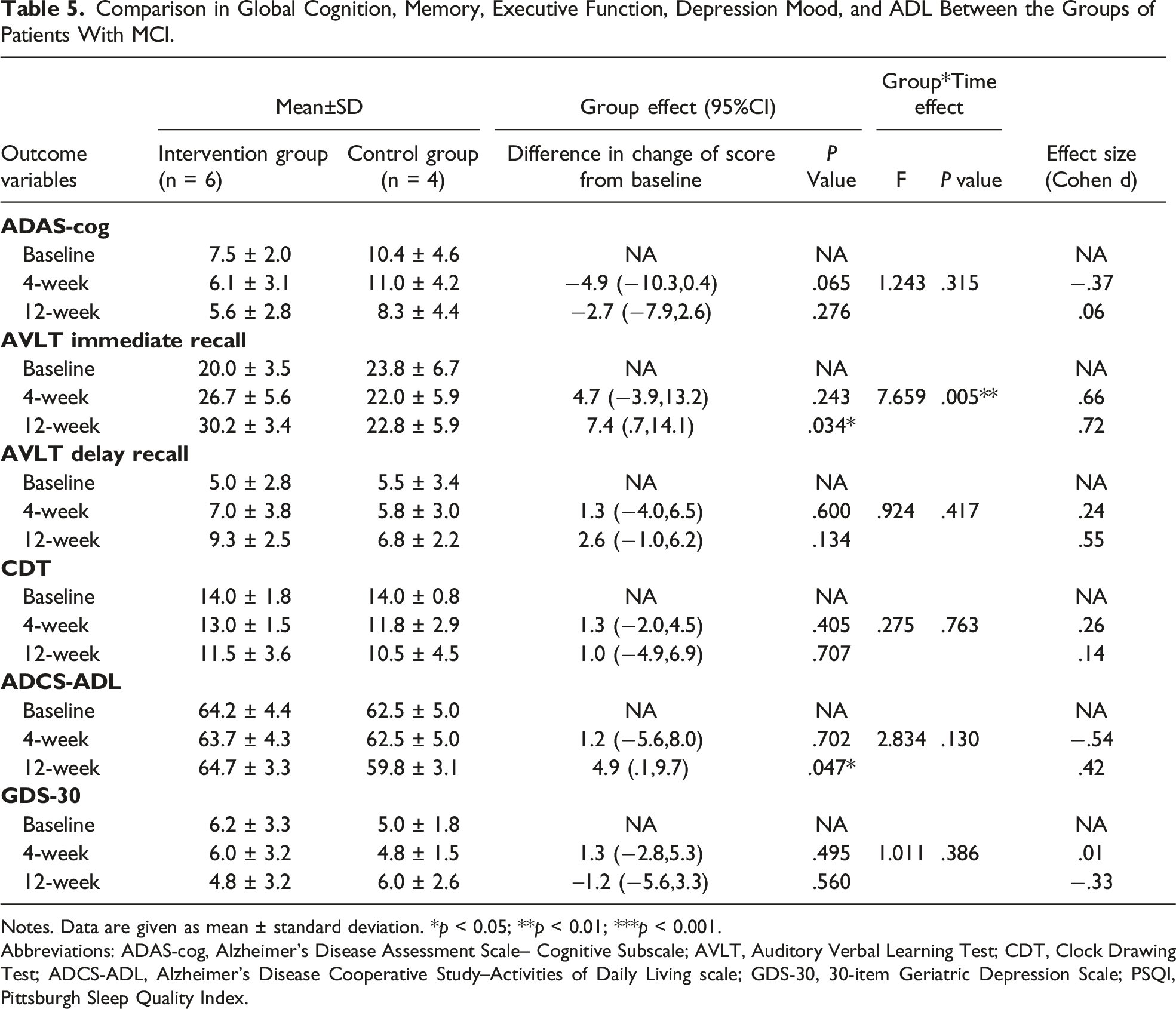
Notes. Data are given as mean ± standard deviation. *
Abbreviations: ADAS-cog, Alzheimer's Disease Assessment Scale– Cognitive Subscale; AVLT, Auditory Verbal Learning Test; CDT, Clock Drawing Test; ADCS-ADL, Alzheimer’s Disease Cooperative Study–Activities of Daily Living scale; GDS-30, 30-item Geriatric Depression Scale; PSQI, Pittsburgh Sleep Quality Index.
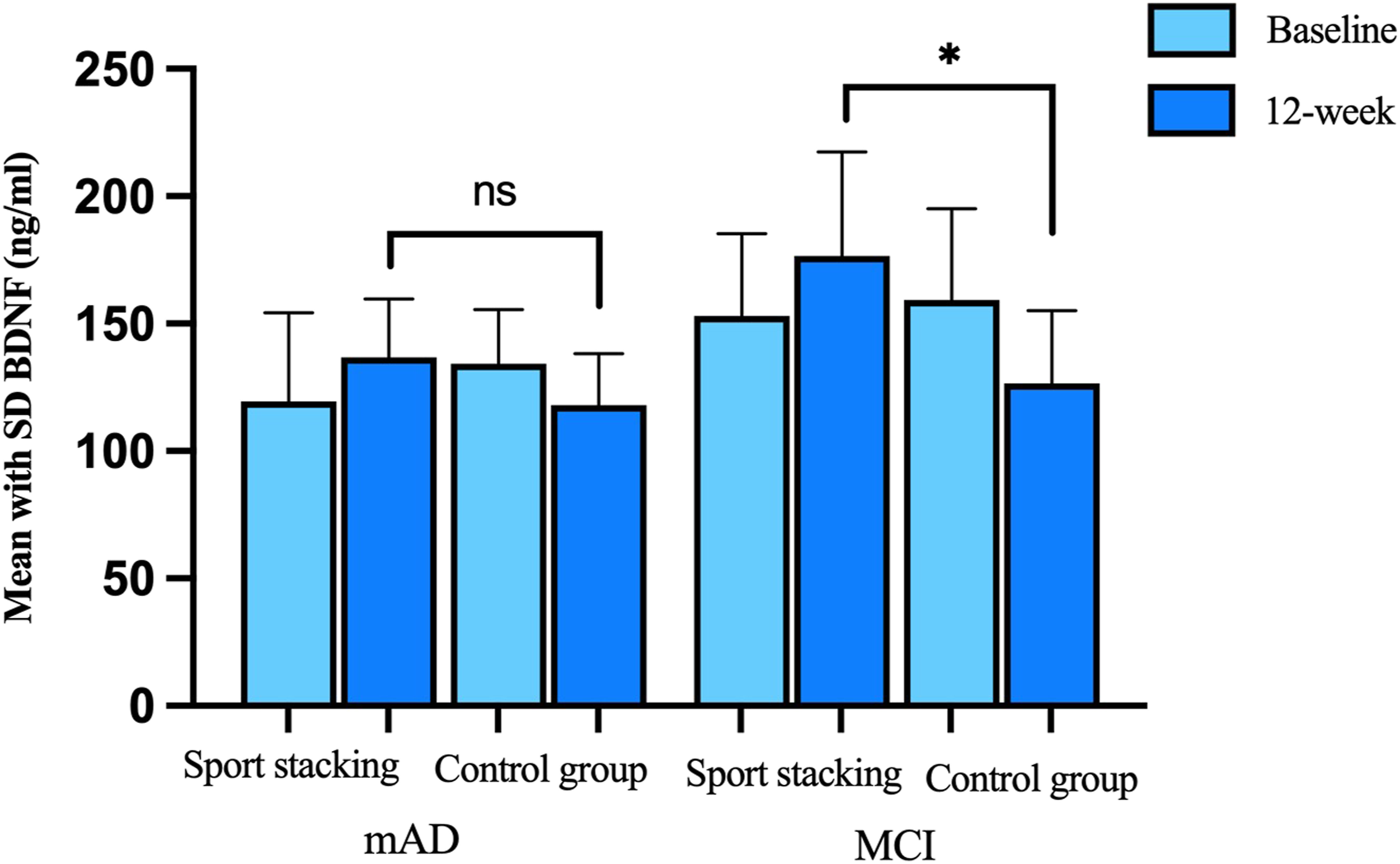
Comparison in concentration of plasma BDNF between the groups of patients with MCI and mild AD. No significant differences in T-mild AD subjects between intervention and control group were found. *
Subgroup Analysis on Participants With Mild AD
Comparison in Global Cognition, Memory, Executive Function, Depression Mood, and ADL Between the Groups of Patients With Mild AD
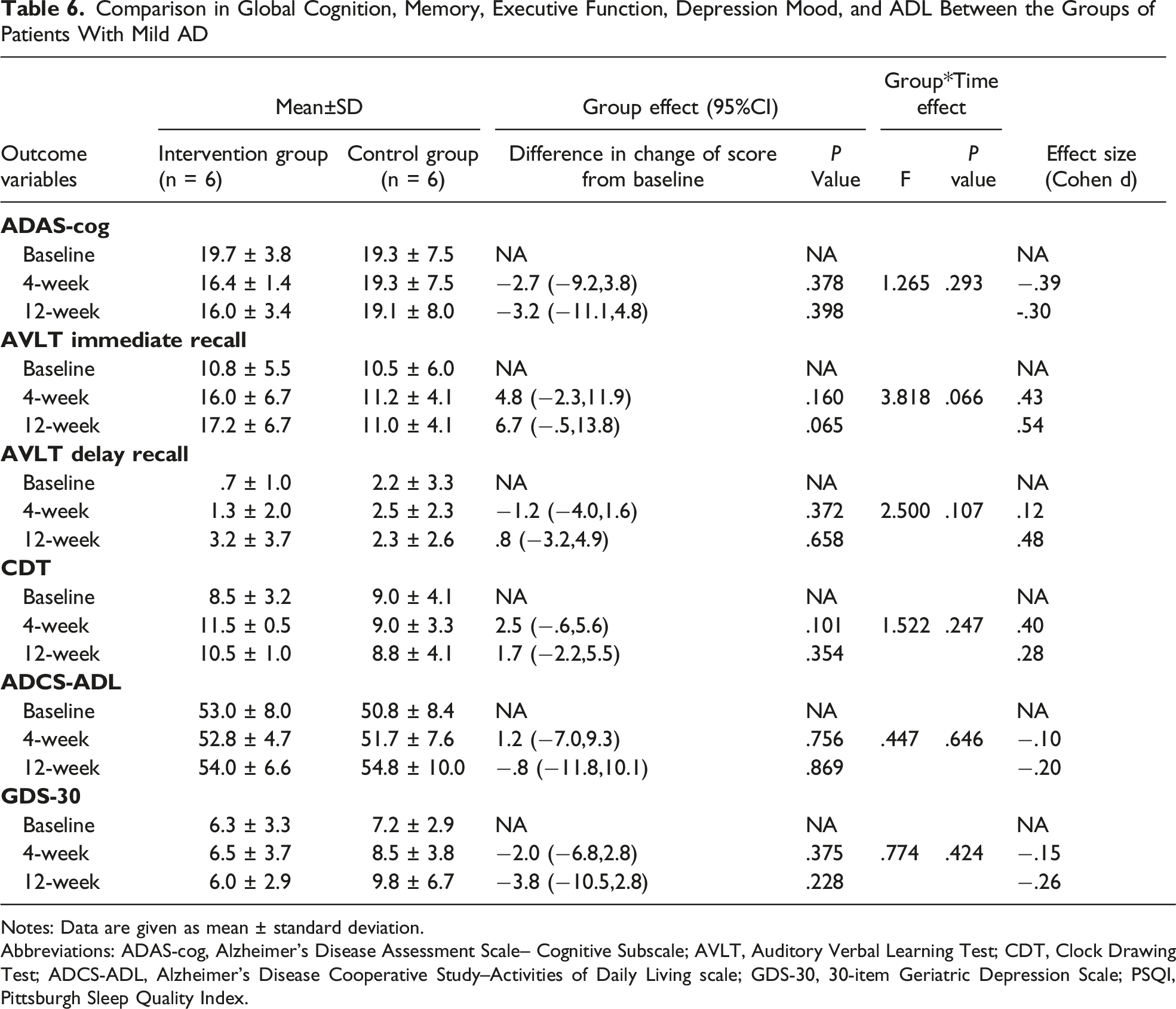
Notes: Data are given as mean ± standard deviation.
Abbreviations: ADAS-cog, Alzheimer's Disease Assessment Scale– Cognitive Subscale; AVLT, Auditory Verbal Learning Test; CDT, Clock Drawing Test; ADCS-ADL, Alzheimer's Disease Cooperative Study–Activities of Daily Living scale; GDS-30, 30-item Geriatric Depression Scale; PSQI, Pittsburgh Sleep Quality Index.
Feasibility and Acceptance of the Sport Stacking
Participants’ Perceptions of Sport Stacking Intervention.

Discussion
To our best knowledge, this pilot study is the first 1 to apply sport stacking to improve the cognitive function of older people with mild cognitive impairment and mild Alzheimer’s disease. The results indicated that this intervention is highly feasible for them, even in the presence of cognitive affliction. The preliminary findings also suggested the potential of this intervention in improving the AVLT immediate recall and episodic memory, particularly among persons with MCI. The increase in plasma
The beneficial effects of sport stacking on immediate recall among persons with MCI converge with studies in which vision-based speed-of-processing training examined improving working memory. 43 Sport stacking is an activity that requires both hands and eyes to work together. As the participants learn a new stacking mode, they need to memorize the stacking method and then stack the cups in the prescribed order and restore them according to their memory. While stacking, hand-eye coordination is required, with the eyes determining the position and the left and right hands alternating between placing the cups appropriately or restoring them in sequence. In the process of stacking, participants are required to gradually increase their speed to improve their responsiveness to the whole process. The results of the study showed that cognitive and hand coordination activities, interactive activities for the upper and lower extremities, and trunk and functional activities could improve hand-eye coordination and have beneficial effects on the attention dimension of cognitive function, possibly delaying the decline in global cognitive function. Sport stacking can also be seen as a kind of visual-motor integration activity. Full visual-motor integration requires different extended brain regions to perceive visual stimuli and plan and execute movements. The occipital, parietal, and temporal lobes help distinguish and perceive visual stimuli. The frontal lobe facilitates the integration of sensory information and the planning of appropriate actions. The basal ganglia and cerebellum regulate muscle movement. It means that visual-motor integration training can stimulate specific brain regions to increase neuroplasticity and improve performance in everyday life. Therefore, visual-motor integration training is 1 of the most effective ways to improve cognitive function in older adults. In fact, there is more evidence to suggest the added benefit of simultaneous cognitive and exercise training,44,45 and such work also suggested that the visual-motor integration contributed to such difference. 46
In addition to immediate recall, engagement in sport stacking was also found to improve the ADCS-ADL among the MCI subject cohort. The improved hand movement coordination and hand-eye coordination resulting from sport stacking may explain the improved function on ADL. Previous study, in fact, indicated that improved hand movement, particularly the fingers, could stimulate the motor cortex 47 in the central nervous system to improve ADL. Hand-eye coordination is almost a pre-requisite to ensure performance quality for a wide range of basic and instrumental ADL such as bathing, dressing, exercising, and writing. 48 Impaired hand-eye coordination is even found to be associated with loss of independent living in older adults. 49 As such, it is likely that the improved ADL function associated with sport stacking is related to hand-movement coordination and hand-eye coordination.
Nevertheless, sport stacking does not demonstrate similar cognitive benefits in persons with a confirmed diagnosis of mild Alzheimer’s disease. The short training duration may explain this, as this clinical cohort takes far longer to learn a new pattern of behaviors and mental challenges than those with MCI. 50 Previous studies evaluating the effects of exergame, which exposed the participants to cognitive challenges integrated with demand on physical movement, also indicated the importance of the dose of training and engagement in shaping the treatment benefits, and the positive effects were detected at 6 months after training. 51 The evaluative timeframe of 3 months may be too short to allow for identifying the positive effects. By the way, optimizing the treatment effect of such more challenging activities for the clinical group may require additional strategies to enhance the skill mastery. 52
Another unique finding of this study was about the positive effect of 12-week sport stacking in increasing the plasma
Although the quantitative findings did not show any significant changes in the global cognition and executive function in patients with MCI and mild AD, the participants reported improved memory and better attention in the post-intervention interview after engaging in the 12-week sport stacking. Such findings reinforced the possibility that a full-scale evaluation of the cognitive benefit of sport stacking in persons with Alzheimer’s disease is needed. Indeed, the interview also identified the great feasibility and acceptance of sport stacking among this clinical cohort. More specifically, the participants enjoyed the engagement and regarded it as a game to allow individual and collaborative practice. The easy-to-follow instruction, simple equipment, free access, and no venue-related restriction rendered sport-stacking a highly acceptable day-to-day practice for persons with cognitive impairment. The accomplishment of progressively increasingly challenging steps also motivated their longer-term practice. The reported emotional benefit in alleviating loneliness and the poor mood was also crucial to support the adaptation of persons with MCI and clinical dementia to the disease process.
Even though this study is the first to examine the feasibility and preliminary cognitive benefits of sport stacking, it still has several limitations that deserve attention. Firstly, we recruited a heterogeneous sample of mild AD and MCI, for which the severity of cognitive affliction may shape the treatment effect. Secondly, the sample size is relatively small, and the inadequate sample is not appropriate to study the mediation effect of the biomarker on the relationship between sport stacking and cognitive improvement. Thirdly, the self-report method was used to assess compliance with self-practice. Finally, adequate objective quantitively measures were not used to capture the process variables (eg, frequency of self-practice, level of caregivers’ support, variation of sport stacking completion time) for evaluating feasibility.
Although our study had these limitations, it still showed the feasibility of sport stacking in older people with MCI or mild dementia. And we also found its positive effects on them. Our study provides a new method for cognitive training for patients with early stages of cognitive impairment.
Conclusion
To the best of our knowledge, this is the first study to report the effects of sport stacking on cognition in patients with MCI and mild AD. These preliminary results suggested the feasibility of sport stacking and its benefits on patients' episodic memory, possibly via upregulation of
Footnotes
Acknowledgments
This work was supported by the geriatric memory clinic at the First Affiliated Hospital of Chongqing Medical University, the Department of Histology and Embryology and Laboratory of Stem Cell and Tissue Engineering in Chongqing Medical University, Chongqing, China. The Authors wish to acknowledge the contributions of the Department of Histology and Embryology and Laboratory of Stem Cell and Tissue Engineering in Chongqing Medical University Prof. Yong Tang and the PhD candidate Lin Zhu.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Intelligent Medicine Project of Chongqing Medical University (ZHYX2019003).
