Abstract
Frontotemporal degeneration (FTD) is an umbrella term encompassing a range of rare neurodegenerative disorders that cause progressive declines in cognition, behavior, and personality. Hearing directly from individuals living with FTD and their care partners is critical in optimizing care, identifying meaningful clinical trial endpoints, and improving research recruitment and retention. The current paper presents a subset of data from the FTD Insights Survey, chronicling the diagnostic journey, symptoms, and the impact of FTD on distress, quality of life, and independence, in the mild to moderate stages of the disease. Survey respondents included 219 individuals diagnosed with FTD and 437 current care partners, representing a range of FTD diagnoses. Around half of survey respondents reported seeing three or more doctors before an FTD diagnosis was given, and a range of prior diagnoses were noted. Most frequently endorsed symptoms tended to be consistent with clinical characteristics of the specific diagnosis, though there was significant variability in symptoms reported within diagnostic categories as well as considerable overlap in symptoms between diagnostic categories. Cognitive and language symptoms of FTD were generally most distressing to the person diagnosed, and a loss of independence was endorsed as affecting quality of life. The distinct perspectives of diagnosed persons and care partners regarding disease impact differed notably for bvFTD/Pick’s disease. Participating independently in a range of activities, within the home, outside the home, and with other people, were reported as challenging for people living with FTD, underscoring the degree to which the lives of these individuals are affected even at the mild and moderate stages of disease. Overall, by heeding the perspectives of those living with FTD, we can begin to design more meaningful research studies, provide better care, and develop therapies that improve quality of life.
Introduction
As experts on the lived experience of a disease, the perspectives of people diagnosed and their loved ones are key to consider in clinical research. Engaging persons diagnosed and their care partners in clinical research design has both practical and ethical benefits, including the identification of clinical trial endpoints that are meaningful for participants and their care partners, more positive participant research experiences, improved clinical trial recruitment and retention, and enhanced financial value for sponsors. 1 Within the United States, the past decade has seen changes to regulatory and legal requirements for such input. A leading framework is the 2014 FDA Roadmap to Patient-Focused Outcome Measurement in Clinical Trials, which includes (1) understanding the disease or condition, (2) conceptualizing treatment benefit, and (3) selecting/developing outcome measures. 2
Hearing directly from those impacted by a medical condition is even more crucial in rare diseases, where fundamental information about the diagnostic journey, most troublesome symptoms, disease impact, and efficacy of existing interventions may be less defined. It may also prove more difficult to document change in the domains relevant to patients, as needed for orphan drug regulatory approval. Heterogeneity in symptom manifestation, inadequate information about the natural course of the disease, potential inability for patients to report on their own condition, and a lack of familiarity on the part of healthcare providers can all lead to misconceptions about what matters most to people with lived experience. 3 Based on the FDA’s Roadmap to Patient-Focused Outcome Measurement in Clinical Trials, the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) Emerging Good Practices for Outcomes Research Task Force Report further outlined challenges specific to developing appropriate outcomes in rare disease research and highlighted solutions such as focusing on symptoms that are most common or most impactful based on patient perspective, better understanding of disease progression despite limited data, and working with patient advocacy organizations and registries to solicit patient and care partner input. 4
Frontotemporal lobar degeneration (FTLD) refers to a group of neuropathological disease processes that generally target the frontal and temporal cortices of the brain. FTLD is associated with a range of different clinical disorders or phenotypes, and here we use the term “frontotemporal degeneration” (FTD) to refer to this group of related, rare neurodegenerative disorders. FTD disorders typically involve some degree of cognitive and behavioral dysfunction, sometimes with concomitant motor disturbances. FTD phenotypes include: • Behavioral variant FTD (bvFTD), more commonly known as behavioral variant frontotemporal dementia, characterized by personality changes, apathy, and a progressive decline in appropriate social behavior, judgment, self-control, and empathy. • Primary progressive aphasia (PPA), a primary impairment of language, in which one may lose the ability to speak, read, write, and understand others’ speech. PPA is further broken down into nonfluent/agrammatic variant (nfvPPA), semantic variant (svPPA), and logopenic variant (lvPPA), based on the specific language dysfunction. • FTD with amyotrophic lateral sclerosis (FTD-ALS), in which FTD and ALS co-occur with both motor neuron and cognitive symptoms. • Corticobasal degeneration (CBD), in which brain regions associated with initiating, controlling, and coordinating movement are also impacted. Corticobasal syndrome (CBS) is a clinical phenotype associated with CBD. • Progressive supranuclear palsy (PSP), which primarily affects movement, such as balance, walking, and coordination.
While initially individuals may present with symptoms primarily consistent with one of these FTD phenotypes, over the course of the illness additional functional domains are impacted, ultimately leading to death. FTD disorders have a median onset in midlife and vary in clinical presentation, pathology, and etiology; however, diagnoses are typically made based on clinical manifestation. The heterogeneity of symptoms, which overlap with other neurodegenerative and psychiatric conditions; the absence of established biomarkers; and a lack of awareness of this young-onset dementia can lead to years of misdiagnosis.5-7 Epidemiologists estimate the prevalence of the FTD disorders in the United States at approximately 60,000, however, this figure is widely believed to be an underestimation given challenges with accessing timely, accurate diagnoses and lack of awareness of the diagnostic criteria or tests.8,9
While the experience of living with and caring for people with dementia, particularly Alzheimer’s disease, has been documented, there is little literature on the lived experience of FTD. Research in this area tends to focus specifically on caregiver burden or the experience of a single FTD disorder (e.g.10-16
To better understand the breadth of experience of FTD and inform clinical trial design from a patient- and care partner-centered perspective, The Association for Frontotemporal Degeneration and the FTD Disorders Registry collaborated on the FTD Insights Survey, the largest data source to-date on community experiences with FTD. This article represents the first scientific publication from this dataset. It will report a subset of the data collected from the FTD Insights Survey, documenting the lived experiences of those in the mild to moderate clinical stages of the disease as it relates to diagnostic journey, symptoms experienced, impact of the disease, and, briefly, use of treatments.
Method
Participants
Demographics of the individuals with FTD represented in the survey.

Survey Design
The FTD Insights Survey included a total of 191 questions, but not all participants answered all questions. Rather, the survey used branching and skip logic based on responses to previous questions. The main points of branching were based on whether the respondent was (1) an individual diagnosed with FTD, (2) a biological relative of someone diagnosed who is not a care partner, (3) deemed to be at biological risk of developing FTD, (4) a current care partner of someone diagnosed with FTD, or (5) a past care partner for someone with FTD who is now deceased. There were 36 questions that required answers in order to progress through the survey, 13 of which were slightly reworded replicates depending on the respondent (i.e., diagnosed person vs care partner).
The development of the survey was guided by The Association for Frontotemporal Degeneration and the FTD Disorders Registry, with input from experts in the field of FTD including clinicians and researchers. Core survey elements were derived from existing surveys designed and used by the FTD Disorders Registry. Pilot testing among the intended respondents was used to refine and finalize the FTD Insights Survey. Survey questions captured demographic information, family history of FTD, the diagnostic journey, symptoms experienced, distress regarding symptoms, independence and quality of life, past experiences with treatments, hopes for future treatments, and perspectives on research participation and clinical trials. For the current paper, we focus only on survey questions relating to the following themes/topics:
Diagnostic Journey
Survey questions included “Prior to being diagnosed with FTD, was a different diagnosis given?”, and the response of “yes” would trigger a list of potential other diagnoses for the participant to select all that applied, including ALS, Alzheimer’s disease, anxiety, bipolar disorder, depression, Lewy Body disease, menopause, mid-life crisis, mild cognitive impairment, Parkinson’s disease, schizophrenia, stroke, vascular disease, other psychiatric diagnosis, other diagnosis, and “I’m not sure”. The survey also asked “How many different doctors were consulted before an FTD diagnosis was made?”, with response options including 1 or 2, 3 or 4, 4 or 5, more than 5, or “I’m not sure”.
Symptom Domains
Survey questions included “How would you describe the first indication that something was wrong?” and “Since the first symptom(s), what other symptoms have developed?”. Both questions allowed the respondent to select as many of the following as applied: language (e.g., speaking, finding words, understanding, knowing the meaning of objects); memory (e.g., remembering recent events, learning new information); thinking (e.g., solving problems, making judgments, organizing); spatial (e.g. judging distances, perceiving objects); personality (e.g., acting differently or inappropriately in a social situation); relationships (e.g., getting along with others); mood (e.g., anxious, not interested, depressed, irritable, emotional outbursts); eating/drinking (e.g., cravings, alcohol intake, weight gain); behavior (repetitive or compulsive behavior, rigid routines); sleep (e.g., not sleeping through the night, bad dreams, sleeping too much), delusions/hallucinations, a specific difficulty in everyday life, other, and “I’m not sure”.
Disease and Treatment Impact
The survey included the following questions relating to disease and treatment impact: “What symptoms, if any, distress [the person with FTD] the most?”, “What effect(s) of having FTD has had the most significant impact on the overall quality of life [of the person diagnosed?] (select up to three)”, “Do symptoms make it difficult for [the person diagnosed with FTD] to do any of the following activities independently? (select all that apply)”, “Do symptoms make it difficult for [the person diagnosed with FTD] to do any of the following activities outside of the home independently? (select all that apply)”, “Do symptoms make it difficult for [the person diagnosed with FTD] to do any of the following activities involving others? (select all that apply)”. All response options can be found in Tables 4 through 9. Regarding treatment impact, the survey included the question “Is there a treatment (medication or other) that has had the most positive impact on [the person with FTD’s] life?”, with response options including yes, no, and “I’m not sure”.
Procedure
The FTD Insights Survey was available online from October 9, 2020, through March 31, 2021. Survey responses were anonymous; no identifying information was collected. The survey link was promoted in newsletter notices, social media posts, and email invitations to The Association for Frontotemporal Degeneration and the FTD Disorders Registry constituent lists. Advertising materials were also disseminated to allied patient advocacy organizations. The survey was deemed to have “exempt” status by an Institutional Review Board (IRB) for data collection in the United States and Canada. Data collection in the United Kingdom was permitted under an existing research study protocol of the Genetic Frontotemporal dementia Initiative (GENFI) and was executed by special invitation. Geographic restrictions were largely based on a combination of survey design (i.e., language, pre-testing, geographic relevance of response options) and IRB factors. Further adaptions for language or geographic scope were outside the parameters of the current study.
Statistical Analyses
All survey data can be requested from the FTD Disorders Registry at https://ftdregistry.org/for-researchers. All frequencies were calculated using custom scripts in Python version 3.6. For ease of presentation, and based on broad phenotypic similarity across individual diagnoses, survey response frequencies were calculated for three overarching diagnostic categories: (1) bvFTD and Pick’s diseases; (2) PPA (all subtypes); and (3) PSP and CBS. Within these diagnostic categories, responses from diagnosed individuals and care partners were pooled, unless otherwise stated. Individuals with FTD-ALS were not included in the diagnostic category breakdowns, due to low numbers precluding meaningful interpretation of the data (N = 13). Percentages based on frequencies were calculated with the number of respondents who answered the specific survey question as the percentage denominator. Unanswered survey questions were coded as missing data. Note that survey responses cannot be interpreted as entirely independent as individuals with FTD may be represented by themselves and their care partners in separate survey responses. There was no way to account for this in analysis due to the anonymous nature of the survey.
Results
Diagnostic Journey
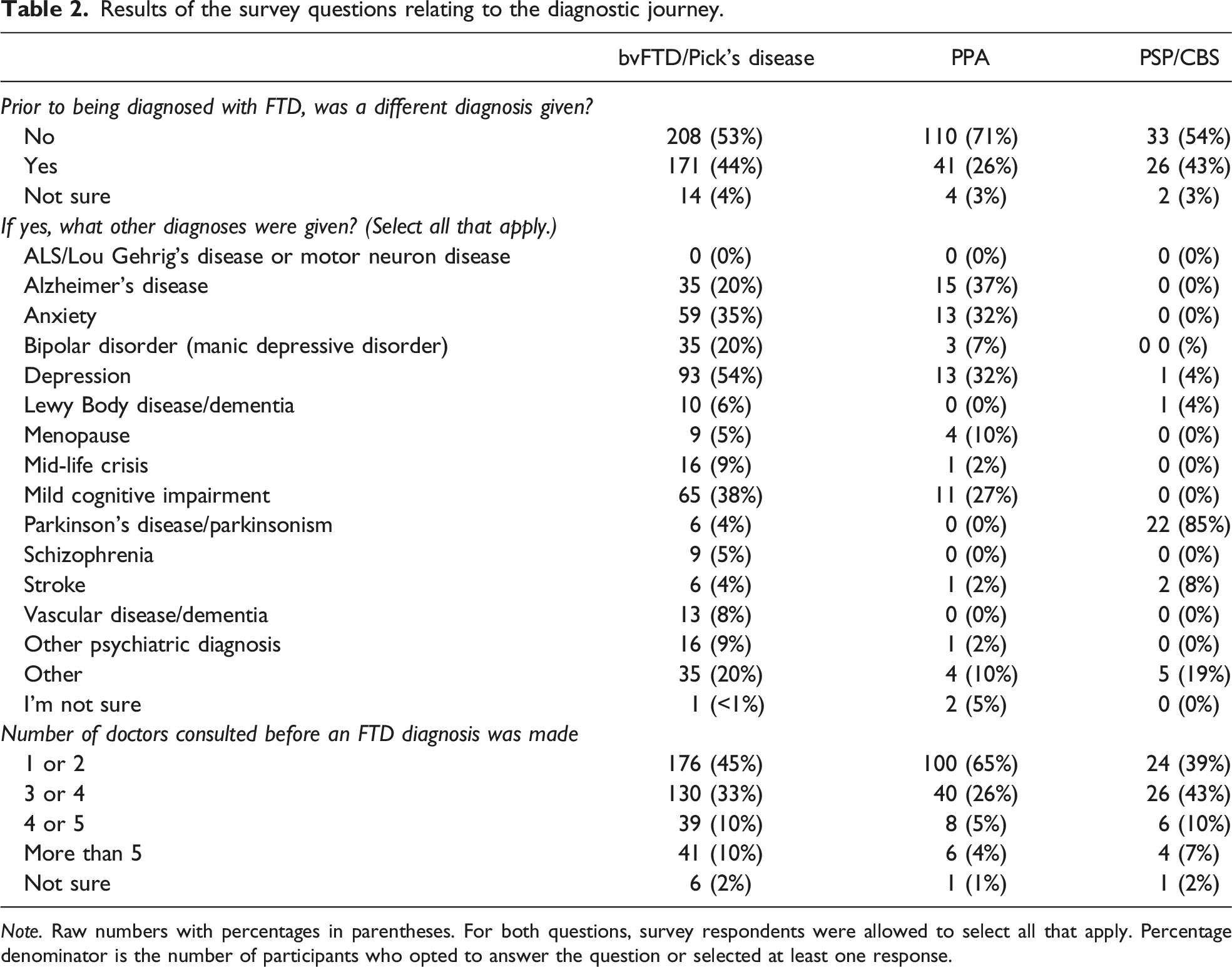
Results of the survey questions relating to the diagnostic journey.
Symptom Domains
Overview of symptoms experienced and the first indication of something wrong.
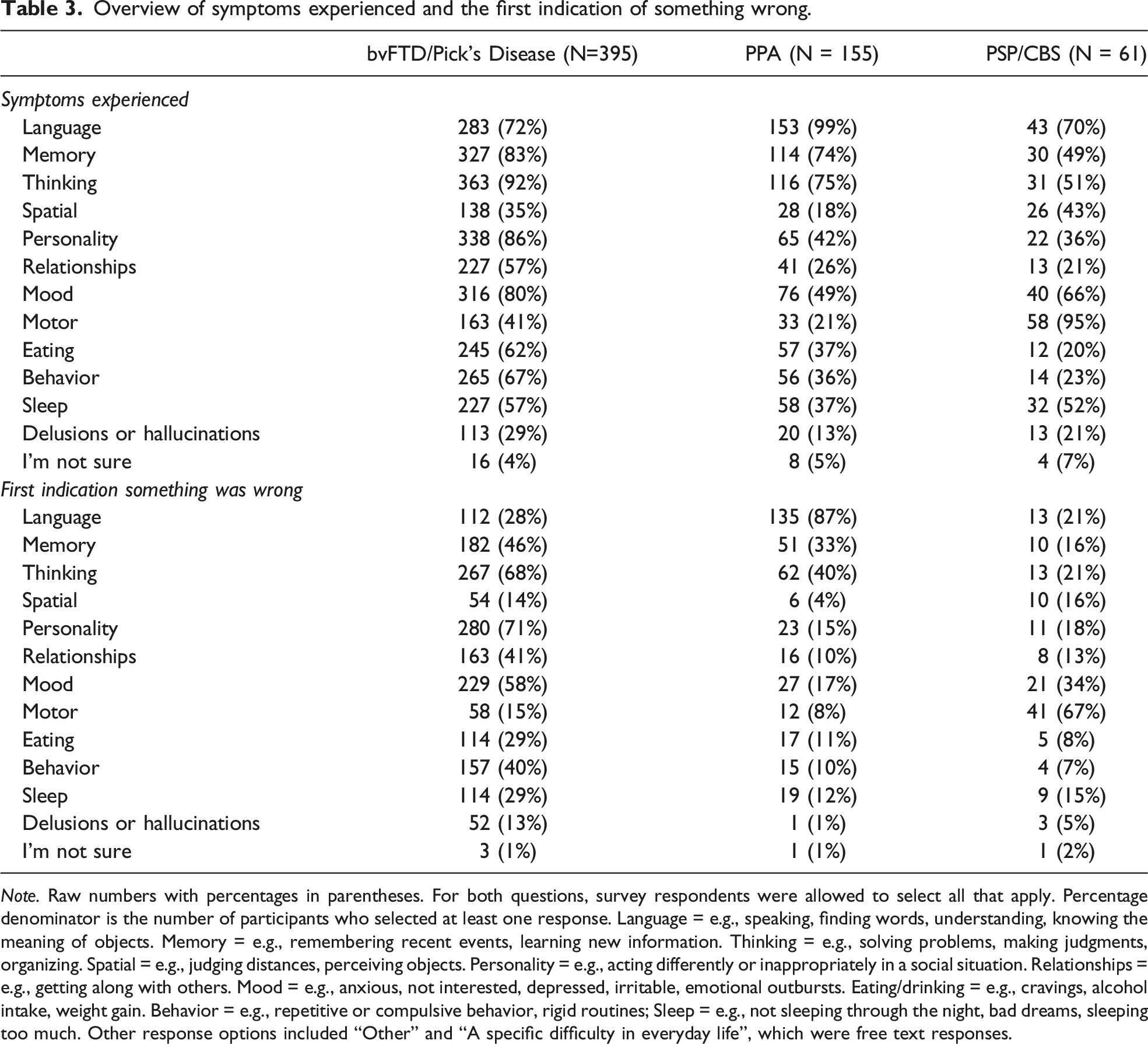
The first indication that something was wrong, as reported by persons diagnosed and care partners, also differed across diagnostic phenotypes. For those with bvFTD/Pick’s disease, the most frequently reported first symptom domain was a change in personality (71%), followed by problems with thinking skills (68%), and mood disturbances (58%). Within the PPA group, language difficulties were the most commonly reported first indication of something wrong (87%), though more than a third noted problems with thinking (40%) or memory (33%) as the first symptom. Around two thirds of individuals with PSP/CBS endorsed motor changes as the first symptom domain affected (67%), but a third reported initial mood disturbances (34%).
Disease and Treatment Impact
Results of the survey question that asks which symptoms of FTD distress the person with FTD the most.
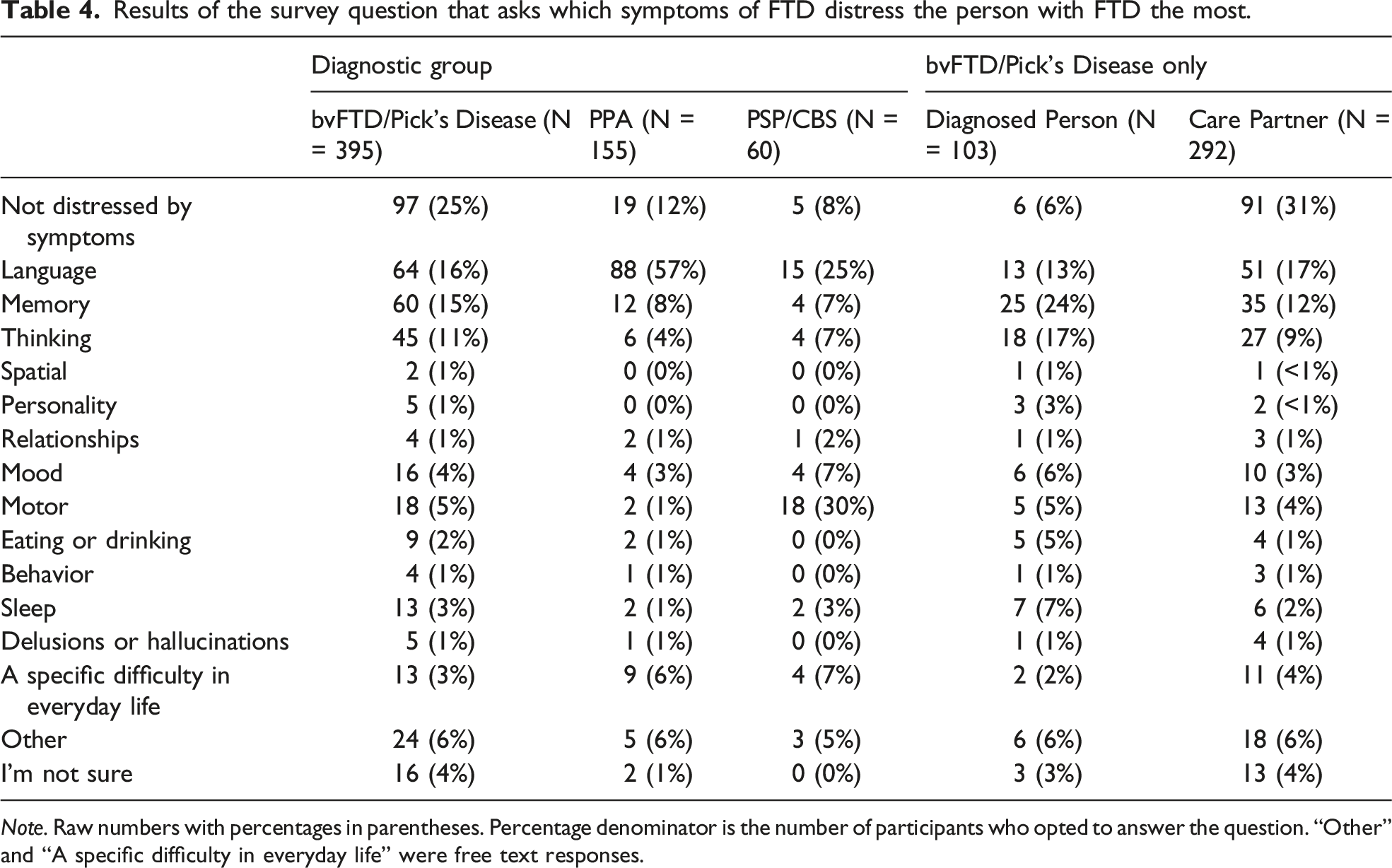
In the bvFTD/Pick’s disease and PPA groups, cognitive symptoms (problems with language, memory, or thinking) were reported to be most distressing to individuals diagnosed. Of the symptom domains, language problems were by far the most distressing symptom in the PPA group (57%), followed by memory deficits (8%). For those with bvFTD/Pick’s disease, thinking, memory and language problems were reported as the most distressing symptom at 11-16% of the time. In the PSP/CBS group, motor disturbances were reported to be most distressing (30%), followed by language problems (25%). Interestingly, 31% of bvFTD care partners reported that the diagnosed individual was not distressed by their symptoms, while only 6% of diagnosed individuals reported that they were not distressed. Lack of distress was reported for 12% of people diagnosed with PPA and 8% of people diagnosed with PSP/CBS. Results are shown in Table 4.
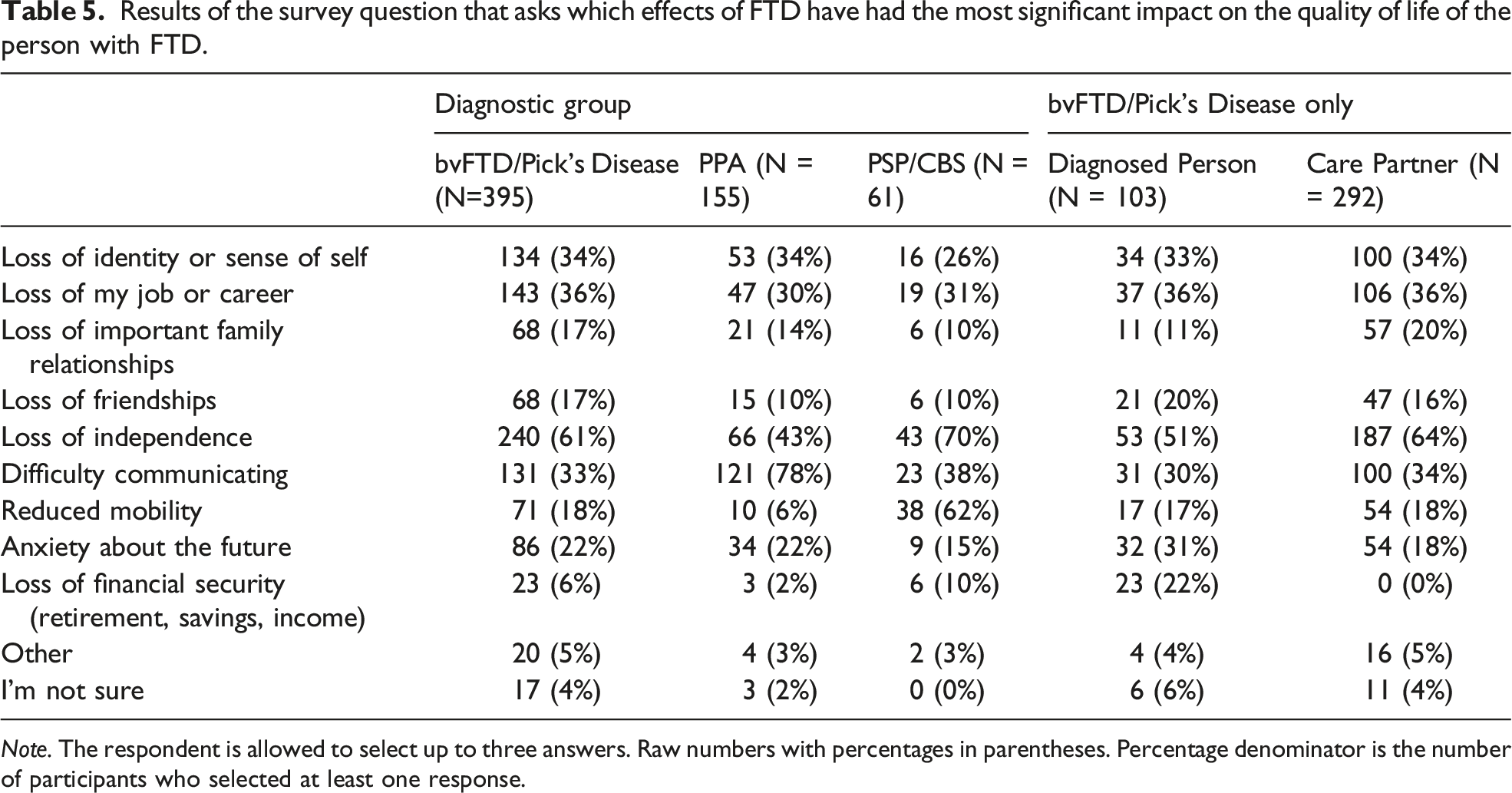
Results of the survey question that asks which effects of FTD have had the most significant impact on the quality of life of the person with FTD.
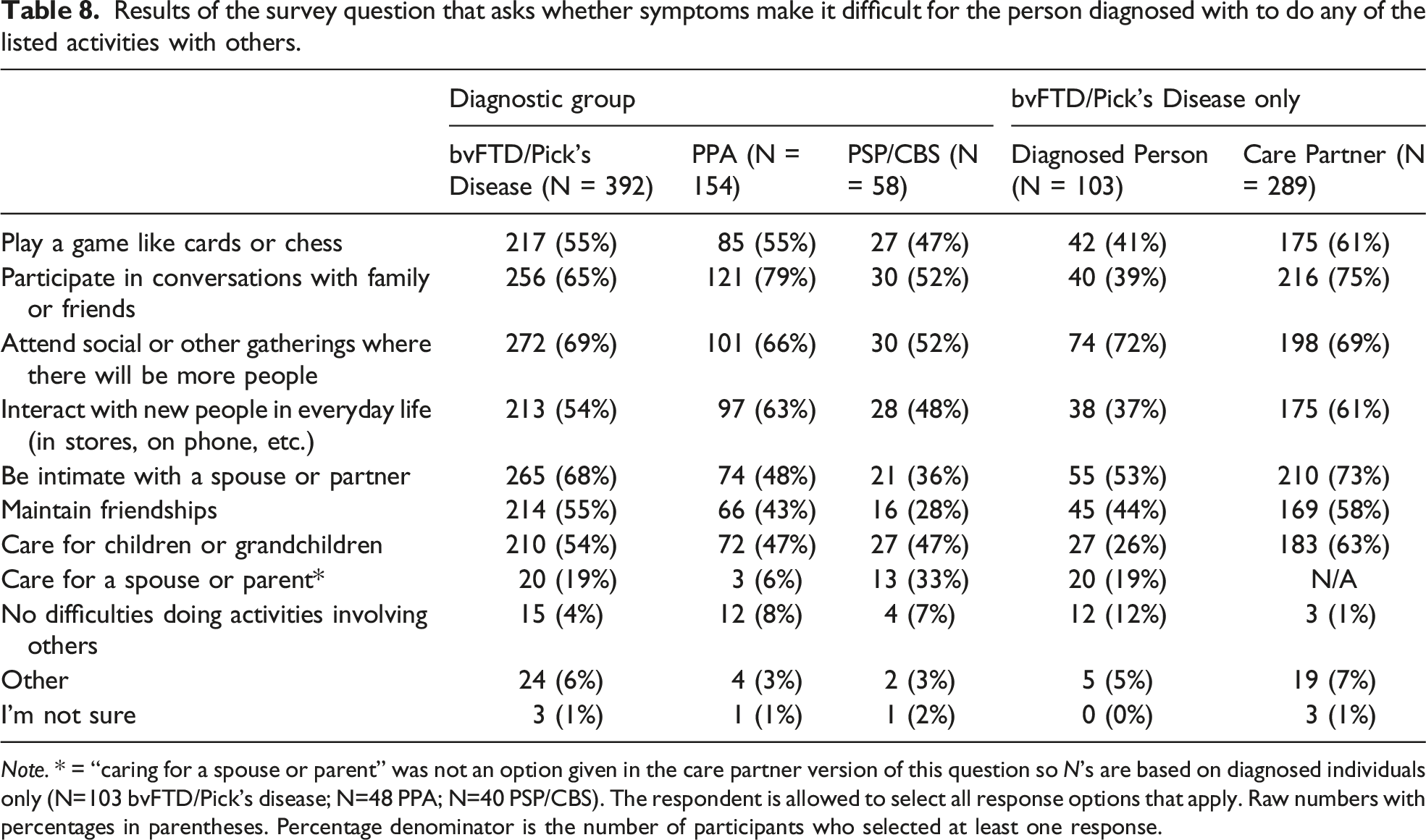
Results of the survey question that asks whether symptoms make it difficult for the person diagnosed with to do any of the listed home activities independently.
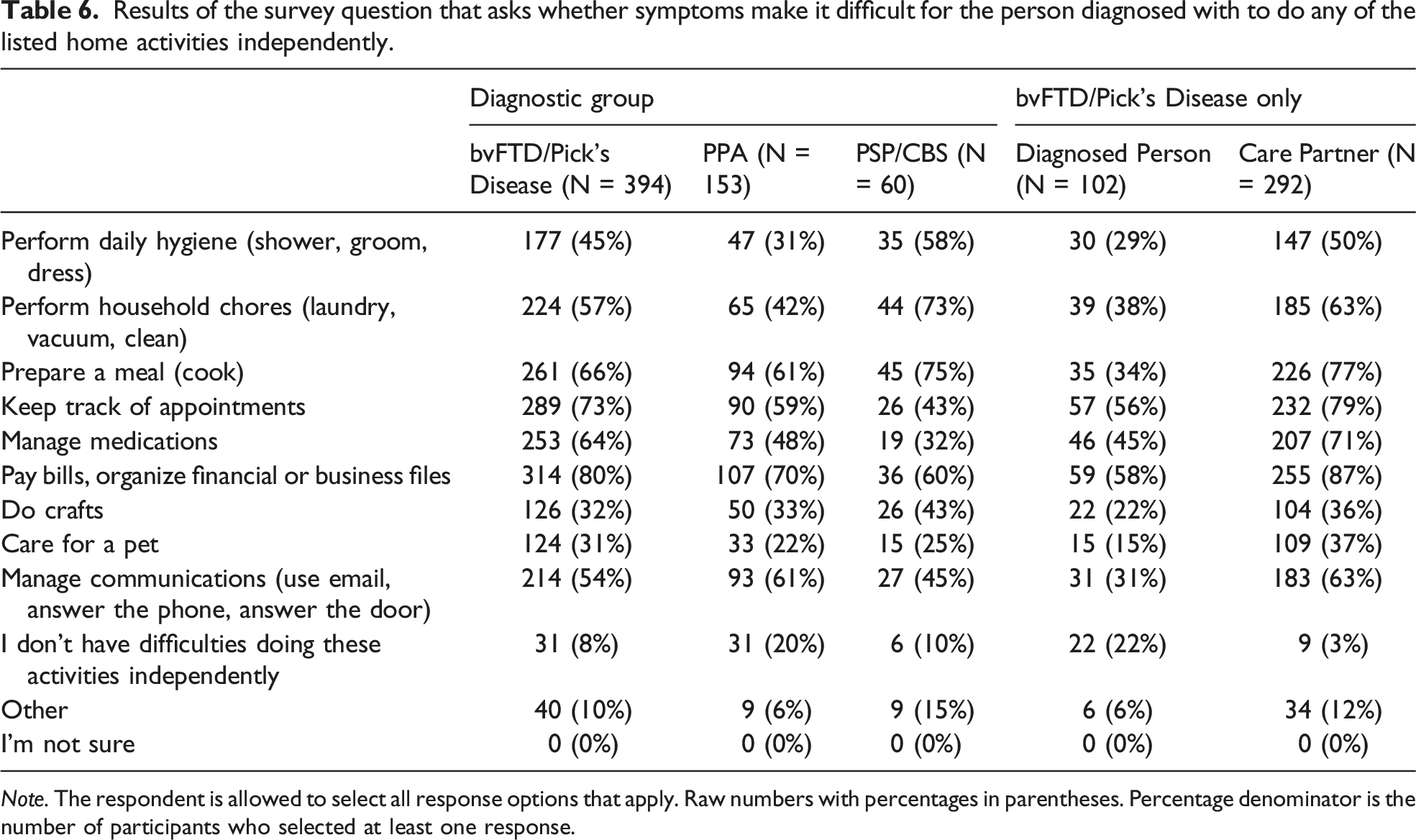
Results of the survey question that asks whether symptoms make it difficult for the person diagnosed with to do any of the listed activities outside the home independently.
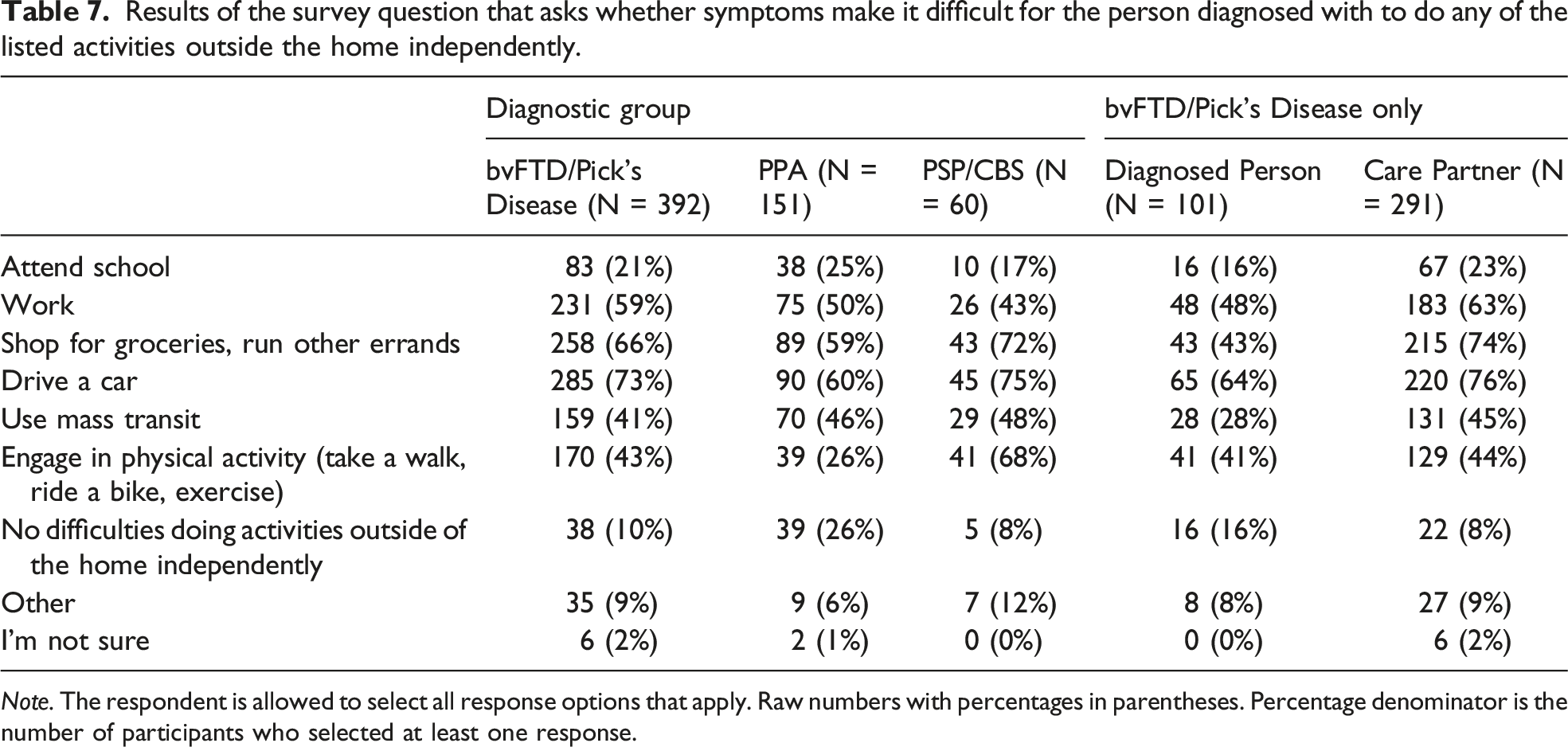
Results of the survey question that asks whether symptoms make it difficult for the person diagnosed with to do any of the listed activities with others.
Results showing whether any past treatment has had a positive impact on the life of the person diagnosed with FTD.

Discussion
The aim of this paper was to document aspects of the lived experience of FTD at the mild and moderate stages of disease, as reported by persons diagnosed and care partners in the FTD Insights Survey. Specifically, we sought to chronicle the experience of individuals with FTD regarding their diagnostic journey, symptoms, and past treatments, as well as the impact of the disease on quality of life and independence. This survey is the largest of its kind to date with additional questions, not reported here, related to care partner burden, specific symptom experiences, treatments, risk tolerance, genetic testing, and research readiness. Data are also available on the lived experience of FTD reported by caregivers of persons at severe stages of disease or who have passed away.
The diagnostic journey is often complicated for those with FTD and their families. Indeed, around half of our survey respondents reported seeing three or more doctors before an FTD diagnosis was made, and this was highest for the PSP/CBS group. Similarly, a 2017 web-based survey of almost 700 FTD caregivers reported that 65% saw 3 or more doctors before an FTD diagnosis was made. 19 Misdiagnoses are common and a source of burden for care partners. 10 Initial psychiatric diagnoses have been found to delay a correct diagnosis of FTD for up to 3-6 years,5,7 which has implications for medication and symptom management, as well as financial and family ramifications. Although in the current dataset we could not verify the prior diagnoses as “misdiagnoses” per se, given the lack of clinical, biomarker or diagnostic information available to verify the presence of FTD, it is nevertheless informative to understand the breadth of other diagnoses that were given to individuals who subsequently received an FTD diagnosis. Psychiatric disorders were common, particularly in bvFTD and PPA, which is unsurprising as psychiatric symptoms are often part of the clinical presentation in these disorders.20-25 By contrast, the most frequent diagnosis given to those who ultimately received a PSP or CBS clinical diagnosis was Parkinson’s disease, which is unsurprising given that both PSP and CBS are considered atypical parkinsonian disorders. Errors in an FTD diagnosis may lead to errors in treatment, with doctors prescribing medications that are ineffective or detrimental. 26 However, there remain multiple challenges to accurate clinical diagnosis of FTD, including a lack of established diagnostic biomarkers, and physicians’ poor familiarity with FTD(25).
The symptom domains reported are largely in line with what would be expected based on the clinical characteristics at early and moderate stages of each disorder. For example, overall, language problems were by far the most commonly endorsed symptom in those with PPA, motor symptoms were reported by the vast majority of individuals with PSP/CBS, and personality and cognitive changes were the most common symptoms in people with bvFTD/Pick’s disease. However, symptom domains varied considerably within diagnostic categories, particularly when it came to identifying initial symptoms. For example, over 25% of individuals with bvFTD/Pick’s disease endorsed changes in language, memory, thinking, personality, relationships, mood, behavior, eating, and sleep as among their initial symptoms experienced. Symptoms also heavily overlapped across diagnostic categories. For example, while language problems define PPA, over 70% of individuals with bvFTD/Pick’s disease or PSP/CBS endorsed difficulties with language. Likewise, personality, behavioral and relationship changes are pathognomonic of bvFTD/Pick’s disease, but a significant portion of those with PPA and PSP/CBS also reported changes within these domains (21-42%). While it is clear from the literature that FTD phenotypes ultimately evolve to encompass symptoms across many domains, these survey data shed light on the heterogeneous manifestations of FTD at its earliest clinical onset. Such information is often challenging to capture and to measure objectively in real time, as individuals with FTD typically are not being followed prior to disease onset unless they are part of a family with a known genetic mutation for FTD.
The symptoms found to be most distressing to the diagnosed person varied considerably both within and between diagnostic categories. Language changes were among the top two most distressing symptoms for individuals with PPA and PSP/CBS. However, the most frequently endorsed symptoms in each group were not necessarily reported to be the most distressing to the diagnosed person. For example, individuals with bvFTD/Pick’s disease reported cognitive symptoms (memory, thinking, language changes) to be most distressing, whereas personality changes, which were just as frequently experienced, tended not to be most distressing to the person diagnosed. Care partners in the bvFTD/Pick’s disease group frequently endorsed that that the diagnosed individual was not distressed by symptoms, consistent with known features of bvFTD including anosognosia, which is a lack of awareness of the disease, or “frontal anosodiaphoria”, which is a lack of concern about having the disorder.17,27
However, patients with bvFTD do in fact report some symptoms to be distressing, with cognitive difficulties including language (13%), memory (24%) and thinking (17%) reported more frequently than changes in personality (3%), relationships (1%) and behavior (1%). A similar pattern of endorsement was observed within the care partners of patients with bvFTD (Table 4) suggesting that metacognitive capacities may vary for different types of symptoms. Indeed, impairments in elements of social cognition which are believed to underlie changes in personality, relationships, and behavior28,29 may be more intimately linked with self-awareness30,31 and therefore inherently less distressing than impairments in primary aspects of cognition such as memory and language that can occur in bvFTD, for example. Work examining the neuroanatomic substrates of social cognitive changes in bvFTD point to a degradation in orbitofrontal 32 and ventromedial 33 regions of the prefrontal cortex (PFC), as well as the salience network,34,35 all of which have been linked to reductions in self-awareness. 36
When survey participants were asked to endorse the top three effects of FTD that impacted quality of life, patterns of responses were largely consistent with the specific challenges for different diagnostic groups. For example, the majority of individuals with PPA noted that difficulty communicating affected quality of life, while those with PSP/CBS endorsed reduced mobility as impacting quality of life. Otherwise, a loss of independence was the most highly endorsed determinant of quality of life in the full sample; this suggests that strategies to maintain independence should be of utmost importance to researchers and stakeholders in the field.
In the bvFTD/Pick’s disease group, responses varied across care partners and diagnosed individuals. For example, anxiety about the future and a loss of financial security were both reported by diagnosed individuals to affect their quality of life, but were rarely noted by care partners. This indicates that the source of the information is critical to consider when devising strategies to improve the quality of life for people with FTD. Whether these differences reflect reliable differences across care partners and persons diagnosed would need to be validated in studies with dyad pairs, as it could be due here to a responder bias where the persons diagnosed who were able and willing to complete the survey represent a highly functioning subset of those affected.
As aforementioned, losing independence was reported to significantly affect the quality of life of people living with FTD. When asked which activities
Likewise, maintaining independence in multiple different activities
Clinical trialists that seek to monitor the effect of a treatment on patient-centered outcomes will need to address the heterogeneity of both symptoms and priorities within and across FTD disorders with shared underlying etiology. They will also need to address the issues of anosognosia and frontal anosodiaphoria, where the person diagnosed may not be aware of or distressed by their own symptoms, despite significant decline in function, independence, and family well-being, even at early stages of disease.
While these survey results are informative, several limitations need to be taken into account. Survey respondents are not necessarily representative of the broader community of those affected by FTD. The vast majority of participants identified as white/Caucasian, with extremely limited representation from other racial and ethnic groups. This is an ongoing problem in all biomedical research with specific challenges for FTD research
37
that are beginning to be addressed. Survey respondents were also more likely to have achieved higher-than-average levels of education. Respondents were restricted to the United States, Canada, and the United Kingdom. We can anticipate that many perspectives were missing from the survey, particularly from communities that may have less access to resources and healthcare specialists. Survey responses of persons diagnosed may reflect a relatively high-functioning demographic of individuals with FTD who are both motivated and able to respond to a research study. Second, the diagnoses cannot be clinically confirmed and it is both possible and likely that some self-reported diagnosis are incorrect or may actually reflect undiagnosed comorbidities, such as underlying Alzheimer’s pathophysiology. Third, comparisons between the responses of care partner and persons diagnosed may need validation with dyad-based research studies, as: (
Overall, the FTD Insights Survey is an invaluable tool for researchers and other stakeholders to learn from the perspectives of people diagnosed with FTD, their family members, and care partners. By understanding the lived experience of FTD, we can design more successful research studies, develop new therapies, and provide better care to improve quality of life.
Footnotes
Acknowledgments
We would like to thank all survey respondents for their time and participation. We also thank experts in the field of FTD for their input on survey development and review of survey materials: Jonathan Rohrer, Caroline Greaves, Edward Huey, Adam Boxer, and David Knopman.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S. Cosentino: S.C. received funds from Association for Frontotemporal Degeneration to support her time for survey development, analysis, and manuscript planning.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: At the time of writing M.S.B. was supported by an FTD Insights Postdoctoral Fellowship from The Association for Frontotemporal Degeneration.
