Abstract
Importance:
Alzheimer’s disease (AD) is a complex neurodegenerative disorder and the most prevalent cause of dementia. In spite of the urgent need for more effective AD drug therapy strategies, evidence of the efficacy of combination therapy with existing drugs remains unclear.
Objective:
To assess the efficacy of combined drug therapy on cognition and progress in patients with AD in comparison to single agent drug therapy.
Methods:
The electronic databases MEDLINE and EMBASE were systematically searched to identify relevant publications. Only randomized controlled clinical trials were included, but no limits were applied to language or time published. Data were extracted from May 27th until December 29th, 2020.
Results:
Three trials found that a combination of ChEI with additional memantine provides a slight benefit for patients with moderate to severe AD over ChEI monotherapy and placebo. However, a further 4 trials could not replicate this effect. One trial reported benefits of add-on Gingko biloba in donepezil-treated patients with moderate AD (using a formula containing Gingko and other antioxidants) compared to donepezil with placebo. A further trial found no significant effect of combining EGb 761® and donepezil in patients with probable AD over donepezil with placebo. Approaches with idalopirdine, atorvastatin or vitamin supplementation in combination with ChEI have not proven effective and have not been retried since. Fluoxetine and ST101 have shown partial benefits in combination with ChEI over ChEI monotherapy and placebo. However, these effects must be replicated by further research.
Conclusion:
Additional memantine in combination with ChEI might be of slight benefit in patients with moderate to severe AD, but evidence is ambiguous. Longer trials are needed. No major cognitive benefit is missed, if solely appropriate ChEI monotherapy is initiated.
Introduction
Epidemiology and Relevance
Alzheimer’s disease (AD) is a complex, irreversible and progressive neurodegenerative disorder, representing the most prevalent cause of dementia worldwide. 1 Aging is undoubtedly the most significant risk factor regarding AD development. 2 Due to demographic change, AD has risen to be one of the biggest global health challenges of the century. In 2020, an estimated 5.8 million people were living with AD in the United States. 3 However, this number is projected to increase to 13.8 million by 2050. 3 Although AD patients live with the disease for an average of 8-10 years, its total time span, including presymptomatic and prodromal stages such as the mild cognitive impairment (MCI), is believed to extend over a period of 20 years. 4 In an elderly population of 65+ years of age, overall prevalence of AD amounts to 10-30% and incidence to 1%-3%. 4
Given its considerably high prevalence and long disease duration, AD represents an ever increasing burden to health care systems. In 2020, the total cost of care for AD patients in the U.S. amounted to $305 billion, a number projected to grow rapidly as the number of patients diagnosed each year is growing. 3
Despite this alarming trend, there is still an urgent need for new treatment approaches. Causal drug treatments are being discovered and in various stages toward approval. However, until those causative drug agents are firmly established, clinical practice must rely on already existent, predominantly symptomatic drug treatment options. By evaluating new combinations of those existing drugs, an effort can be made to maximize their limited potential.
Pathogenesis and Pharmacology
The pathogenesis of AD is complex and various factors have been investigated, the most common of which are amyloid/amyloid precursor protein and tau. However, less known factors, such as calcium, presenilins, the unfolded protein response and the Wnt/catenin system also play a role in the pathology of the AD brain. 5 These pathological changes in AD are accompanied by a decreased amount of acetylcholine in the basal forebrain, which leads to a reduction in cognitive function, as well as glutamate excitotoxicity, resulting in neuronal apoptosis. 6
Referring to this interconnected network of pathogenetic mechanisms, Fessel suggests that a combined treatment with several drugs may be needed to target these various factors more appropriately than monotherapy. 5
The U.S. Food and Drug Administration (FDA) has approved 5 individual drugs and one drug combination for the treatment of AD. Among the individual drugs are the acetylcholinesterase inhibitors donepezil, galantamine and rivastigmine for treatment of mild to moderate AD, the NMDA receptor antagonist memantine for moderate to severe AD and, most recently, aducanumab (Aduhelm), a monoclonal antibody targeting Aβ, that has been approved via the accelerated approval pathway. 7 The only drugs approved to be given in combination are donepezil and memantine. 3
Acetylcholinesterase inhibitors (ChEI) mitigate acetylcholine deficiency inside the synaptic cleft by inhibiting the enzyme acetylcholinesterase, thus preventing breakdown of acetylcholine. 8 Several meta-analyses found substantial evidence for donepezil, 9 galantamine 10 and rivastigmine 11 to be effective in improving cognition outcomes of patients with mild to moderate AD. While donepezil and galantamine are taken orally, rivastigmine can also be applied as a transdermal patch. Initially, ChEI were thought to provide only symptomatic treatment in AD patients. However, significant advances in MRI technology over the last 2 decades have revealed a different aspect of ChEI pharmacology, which extends beyond its initial scope. Especially in the case of donepezil, it is now well researched that this ChEI can actually slow down atrophy of brain tissue, and therefore provide anti-neurodegenerative effects. 12 -16 This is especially interesting, when it comes to atrophy in the hippocampal area, as a reduction in hippocampal volume is considered one of the key pathophysiologic markers of MCI and AD. 17 -19 Dubois et al even reported a 45%-decrease in the rate of hippocampal atrophy over a 1-year period in donepezil-receiving patients with prodromal AD, compared to patients receiving placebo treatment. 12 Findings like these contradict the popular belief, that ChEI can provide no more than symptomatic treatment.
It has also been discussed, whether ChEI like donepezil can also help to alleviate neuropsychiatric symptoms (NPS), which are often associated with AD. At least in mild-to-moderate AD patients, a Bayesian Network meta-analysis from 2016 could not confirm the benefit of ChEI on NPS over placebo. 20 However, other meta-analyses, which also included patients with severe AD, have indeed shown effects of ChEI treatment on NPS over placebo. 21,22 This may point to the conclusion, that ChEI show at least modest effectiveness in alleviating NPS in patients with AD of more severe stages.
In terms of cognition efficacy, another FDA approved AD drug, the NMDA receptor antagonist memantine, shows a significant but small cognitive effect over placebo in patients with moderate to severe AD. 23 As a neuroprotective agent reducing AD-related glutamate excitotoxicity, its mechanism of treatment might be considered rather disease-modifying than purely symptomatic. Therefore it will likely only show effectiveness in longer trials. Memantine also has the potential to slow hippocampal atrophy, as shown by Weiner et al in 2011. 24
Given the fact that ChEI are effective in mild to moderate AD and memantine provides a small benefit over placebo in moderate to severe AD, investigating a combination approach of these 2 drugs in patients with AD of moderate stages seems promising. As pointed out by Parsons et al, 25 ChEI and memantine target 2 different aspects of AD pathophysiology: While ChEI target deficits in the cholinergic system, memantine aims to regulate the dysfunctional glutamatergic system.
Individually, donepezil and memantine both have acceptable adverse event (AE) profiles. It must, however, be considered, whether they are safe to use in direct combination. As Periclou et al 26 reported, no pharmacodynamic or pharmacokinetic interactions were observed between donepezil and memantine, suggesting combined intake is safe. The National Institute for Health and Care Excellence (NICE) recommends the following in their 2011 guidelines (last updated in 2018) for patients who are already diagnosed with AD and treated with ChEI: To consider memantine in addition to ChEI for patients in moderate AD stage and offer memantine in addition to ChEI for patients in severe AD stage. 27 While the combination of donepezil and memantine is now an FDA approved treatment and is being increasingly investigated, recent evidence has raised the question, whether a different combination—galantamine and memantine—might even be superior to donepezil-memantine approaches. 28 This must be critically evaluated, especially with regard to patients with MCI, as galantamine as a monotherapy has been associated with an unexplained rise in death rates and is therefore suggested to be treated with caution in this patient group. 10
In addition to the memantine approach, a number of other substances have been tested in combination with ChEI. Among them were Gingko biloba, a neuroprotective agent (e.g. EGb 761 ®), the 5-HT6 receptor antagonist idalopirdine, several vitamins, the selective serotonin reuptake inhibitor fluoxetine as well as ST101, an azaindolizinone derivate, which has shown to reduce Aβ in mice. 29
As the number of possible drug combinations continues to increase, this review aims at providing a systematic overview of evidence (or lack thereof) regarding the most effective combinations in terms of improving cognition in patients. Safety issues are also taken into consideration.
The objective of this review is to assess whether a combined therapy with 2 or more individual drugs outperforms a monotherapy in patients with AD in terms of cognition efficacy. A systematic literature search was performed to evaluate the current evidence regarding this issue.
Methods
Inclusion Criteria
The requirements for inclusion of publications were a clinical trial in a human patient group diagnosed with either AD (all stages of severity) or MCI as well as the reporting of cognition efficacy as an outcome. Furthermore, a combination therapy of at least 2 separate drugs administered for a combined effect had to be investigated. Studies in which patients had significant comorbidities were excluded. Only randomized controlled clinical trials were included. However, no limits were applied to language or time published. Studies not evaluating combined drug therapy were excluded, as were studies featuring already discontinued drug therapy approaches or not reporting cognition as an outcome. Where cognition was measured, but not as a primary outcome, study inclusion was assessed on a case-by-case basis, as were studies not incorporating standard ChEI treatment as part of the drug combination.
Search Strategy
The electronic biomedical database MEDLINE (1950-) was systematically searched via PubMed from May 27th, 2020 through June 10th, 2020 (and last updated on December 29th, 2020). To support search accuracy, “MeSH” (Medical Subject Headings) terms of the National Library of Medicine were used. For example, the “MeSH” heading term “Alzheimer disease” [MeSH] intrinsically includes 34 synonyms and closely related descriptions such as “Alzheimer sclerosis” or “Alzheimer Type Senile Dementia.” Furthermore, by using subheadings, also called qualifiers, the MeSH headings could be restricted to certain aspects, which were subsequently prioritized. This was proven useful in limiting the search to pharmacotherapy. To explicitly include studies targeting patients with Mild Cognitive Impairment (MCI), its proper MeSH term “Cognitive dysfunction” [MeSH] was added. The same was done to include combined drug therapy approaches. Using Boolean operators, the final search term printed out as follows: (“Alzheimer Disease/drug therapy” [Mesh] OR “Cognitive Dysfunction/drug therapy” [Mesh]) AND “Drug Therapy, Combination” [Mesh] (Alzheimer disease/dt [Drug Therapy] OR Alzheimer Disease.tw. OR mild cognitive impairment/dt [Drug Therapy] OR mild cognitive impairment.tw.) AND (combination drug therapy/ OR combination chemotherapy/)
Additional papers were manually identified. Initial search results were cross-checked for duplicates, which were removed. Results were then screened for inclusion by review of title and abstract. Studies clearly not meeting the inclusion criteria were excluded. Full text articles of all remaining studies were reviewed and assessed for inclusion.
Data Extraction
Data were extracted on study characteristics (number of patients, duration, groups, cognition outcomes) as well as reported findings with regard to cognition efficacy and adverse events (AEs).
As Esiri and Chance 2 accurately described, “most assessments of cognitive deficits are in fact measures of remaining function (that is, reduced measures of positive ability).” Among the most common cognition measurement scales are the Alzheimer’s Disease Assessment Scale Cognitive Subscale (ADAS-Cog), the Mini Mental State Examination (MMSE) and the Severe Impairment Battery (SIB). All scales are internationally approved and validated.
Quality Assessment
Methodological quality of the included studies was assessed using the Jadad Scale by Jadad et al. 30 The statutes of Charité University Medical School for assurance of good scientific practice were followed and respected throughout the review process.
Results
Overview of Results
After removal of duplicates, 119 search results were assessed for inclusion, 43 of which were excluded as they clearly failed the inclusion criteria. Full text articles of 76 studies were reviewed, from which 60 studies had to be excluded due to various reasons as listed in Figure 1.
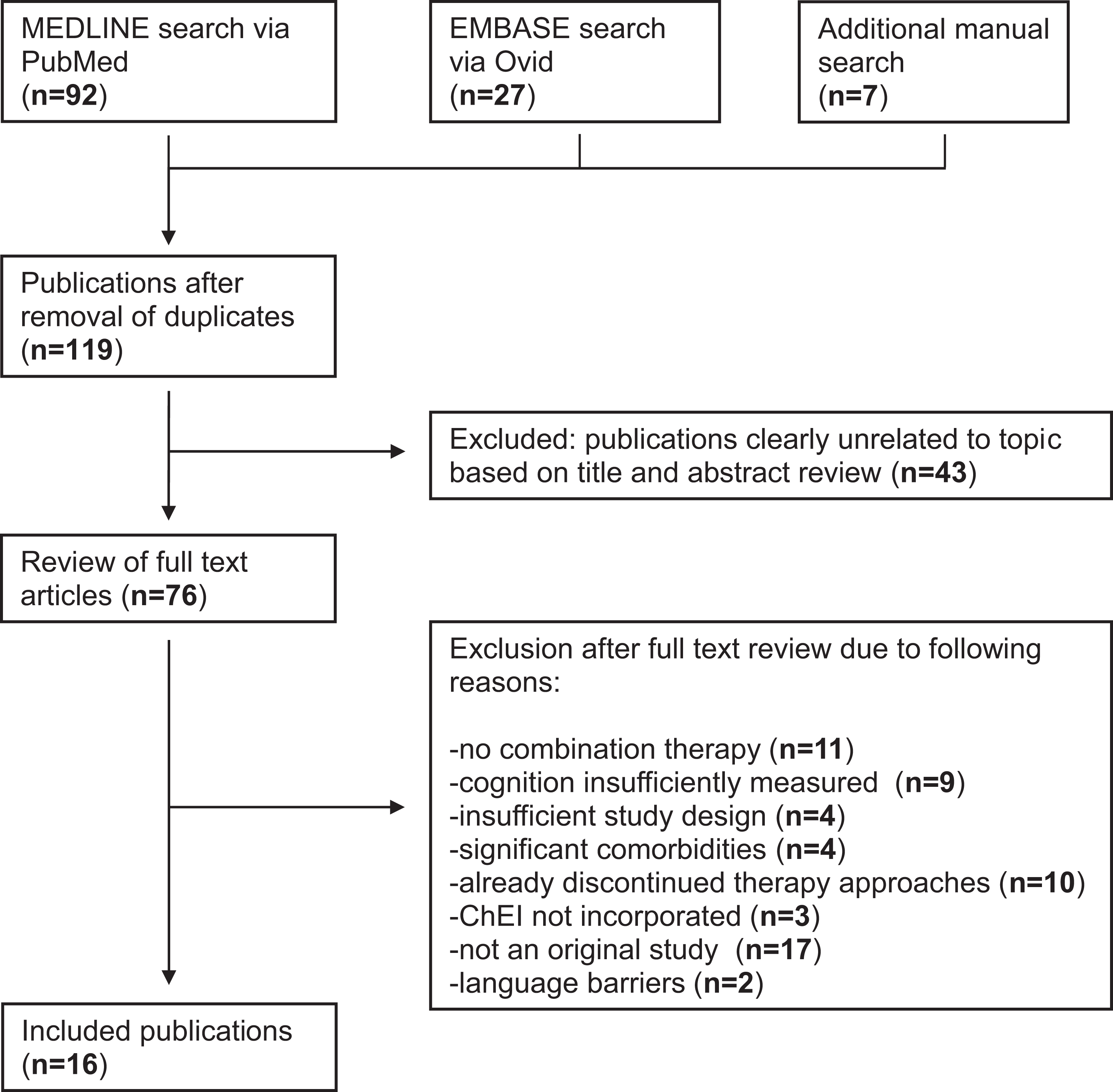
Procedural flow chart adapted from PRISMA. 31
A total of 16 studies were included in the systematic analysis, with 15 originating from the systematic search and one from the manual search. The following Table 1 lists all the included studies with characteristics and findings on cognition and AEs.
Overview of Combined Drug Therapies.
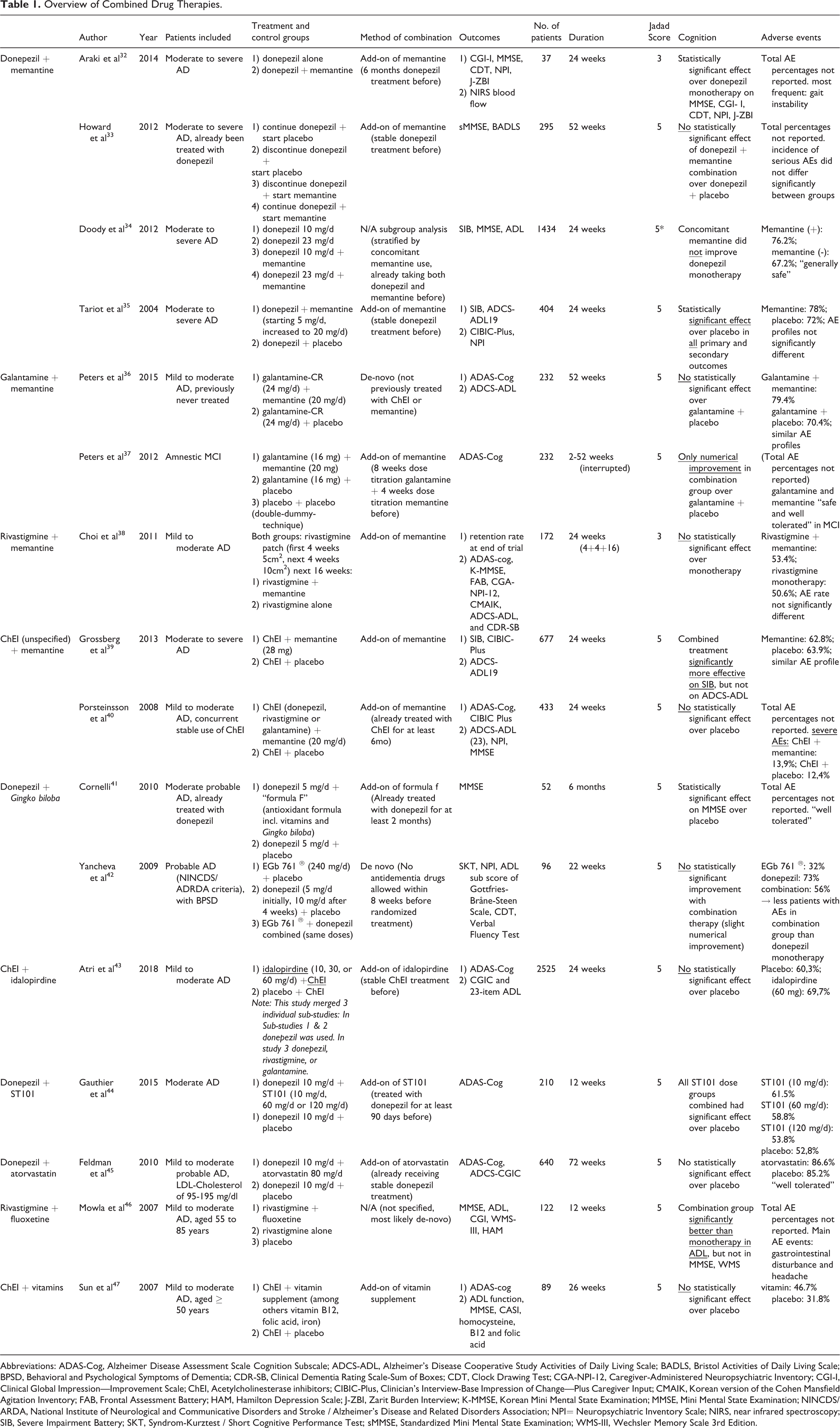
Abbreviations: ADAS-Cog, Alzheimer Disease Assessment Scale Cognition Subscale; ADCS-ADL, Alzheimer’s Disease Cooperative Study Activities of Daily Living Scale; BADLS, Bristol Activities of Daily Living Scale; BPSD, Behavioral and Psychological Symptoms of Dementia; CDR-SB, Clinical Dementia Rating Scale-Sum of Boxes; CDT, Clock Drawing Test; CGA-NPI-12, Caregiver-Administered Neuropsychiatric Inventory; CGI-I, Clinical Global Impression—Improvement Scale; ChEI, Acetylcholinesterase inhibitors; CIBIC-Plus, Clinician’s Interview-Base Impression of Change—Plus Caregiver Input; CMAIK, Korean version of the Cohen Mansfield Agitation Inventory; FAB, Frontal Assessment Battery; HAM, Hamilton Depression Scale; J-ZBI, Zarit Burden Interview; K-MMSE, Korean Mini Mental State Examination; MMSE, Mini Mental State Examination; NINCDS/ARDA, National Institute of Neurological and Communicative Disorders and Stroke / Alzheimer’s Disease and Related Disorders Association; NPI= Neuropsychiatric Inventory Scale; NIRS, near infrared spectroscopy; SIB, Severe Impairment Battery; SKT, Syndrom-Kurztest / Short Cognitive Performance Test; sMMSE, Standardized Mini Mental State Examination; WMS-III, Wechsler Memory Scale 3rd Edition.
Cognition
Nine studies examined a combination of ChEI and memantine. Four of these 32 -35 tested donepezil, 2 galantamine, 36,37 one rivastigmine 38 and the remaining 2 left choice of ChEI unspecified and dependent on the individual patients. 39,40
Tariot et al 35 and Araki et al 32 both found donepezil and memantine to have a statistically significant benefit over donepezil monotherapy on cognition outcomes. In the trials by Doody et al 34 and Howard et al, 33 however, the effect did not reach significance. All 4 trials examined patients with moderate to severe AD.
While Tariot et al 35 and Howard et al 33 displayed very good methodology, the trial by Araki et al 32 included a relatively low number of participants (n = 37), as it was aimed more at measuring NIRS blood flow. Furthermore, this trial was not double-blinded, resulting in a Jadad Score of 3.
The study by Doody et al 34 was a subgroup analysis of a previous clinical trial by Farlow et al. 48 While the original trial intended only to determine the ideal dosage of donepezil monotherapy, Doody et al 34 performed a subgroup analysis of concomitant memantine. Although the original study by Farlow et al 48 showed good methodology, it was not aimed at investigating combination therapy. As a consequence, no placebo-memantine was used in the non-memantine subgroup and patients and investigators were only blinded to donepezil dosages. The follow-up analysis of Doody et al 34 investigating concomitant memantine should therefore be treated with caution.
Howard et al 33 had a duration of 52 weeks and was the only of these 4 studies extending over a period of time longer than 24 weeks. Interestingly, in this study, the slight benefit of memantine over placebo, measured as 1.5 points on the sMMSE at week 30, abated by week 52, measuring just 0.7 points. This naturally raises concerns over longevity of treatment effects and may illustrate a major issue with many AD drug trials. A duration of 24 weeks is a considerably short time frame for evaluating effects on a disease with a mean symptomatic duration of 8-10 years. However, as Horikoshi et al 49 discovered in 2020, a significant effect of antidementia drugs after 6 months is a predictor for a better outcome in patients even after up to 24 months. Nevertheless, RCTs of longer duration would ease concerns over longevity of treatment efficacy.
Two studies examined a combination of galantamine with memantine in patients with MCI 37 and with mild to moderate AD, who were previously never treated. 36 While memantine dosage was 20 mg/d in both trials, galantamine was dosed at 16 mg/d in the MCI population trial and 24 mg/d in the mild to moderate AD population trial. In mild to moderate AD, adding memantine had no significant effect either on ADAS-Cog or on ADCS-ADL over placebo after 52 weeks. 36 In MCI patients, only a numerical improvement over galantamine with placebo could be observed. 37 Important to note is that this trial was interrupted due to concerns over safety issues of galantamine observed in previous trials before. 50 Peters et al, 37 however, found galantamine and memantine to be safe and well tolerated in MCI in their trial.
A further study by Choi et al 38 investigated a combination of rivastigmine transdermal patches with memantine and found no statistically significant difference in treatment efficacy, when compared to rivastigmine monotherapy.
Two further studies, Porsteinsson et al 40 and Grossberg et al 39 also administered ChEI with memantine, however, allowed the choice of ChEI (donepezil, rivastigmine or galantamine) to be dependent on the individual patients. While Porsteinsson et al 40 found no significant effect of additional memantine in ChEI receiving patients with mild-to-moderate AD on ADAS Cog, CIBIC-Plus, ADCS-ADL (23-item) or MMSE over ChEI with placebo, Grossberg et al 39 reported a significant effect of add-on memantine on SIB, but not on ADCS-ADL in moderate-to-severe AD. Both trials had a duration of 24 weeks and good methodological quality with a Jadad Score of 5. It should be noted that Porsteinsson et al investigated a memantine dosage of 20 mg/d, while Grossberg et al used 28 mg/d. 39,40
Two studies examined the effects of ChEI combined with Gingko biloba. Yancheva et al 42 tested EGb 761®, an extract from Gingko leaves, in combination with donepezil in patients with probable AD and found only a numerical improvement over EGb 761® with placebo and over donepezil with placebo, but no statistically significant effect on the short cognitive test (SKT), ADL sub score of the Gottfried Bråne Steen Scale, Clock Drawing Test or Verbal Fluency Test after 22 weeks. An important aspect of this trial is that patients who had behavioral and psychological symptoms of dementia (BPSD) were explicitly included. However, no significant effect on scores on the Hamilton Depression Scale (HAMD) could be observed, indicating no benefit of EGb 761® augmentation for patients with BPSD. 42
Cornelli 41 on the other hand, investigated a formula (named “formula f”) in combination with donepezil 5 mg/d. The formula consisted of Gingko biloba as well as other antioxidants such as Vitamin E, B6, B9 and B12. In this trial, a significant effect of “formula f” in combination with donepezil over donepezil combined with placebo was observed on the MMSE after 6 months in patients with moderate AD. 41 Both studies investigating the combination of ChEI with Gingko biloba showed good methodological quality.
One further study by Atri et al 43 featured 3 sub-studies, all of which investigated the 5-HT6 receptor antagonist idalopirdine in combination with ChEI in patients with mild to moderate AD. Reaching a number of 2525 participants in total, the study investigated dosages of 10, 30 and 60 mg/d of idalopirdine. No significant effect of any of the administered doses in combination with ChEI could be observed when compared with ChEI and placebo after 24 weeks. 43
Feldman et al 45 examined a combination of donepezil 10 mg/d and atorvastatin 80 mg/d in patients with mild to moderate AD and LDL cholesterol of 95-195 mg/dl. No significant effect over donepezil with placebo could be reported after 72 weeks.
Gauthier et al 44 investigated the azaindolizinone derivate ST101 in combination with donepezil 10 mg/d in patients with moderate AD. ST101 dosages of 10,60 or 120 mg/d were administered. A significant effect over donepezil with placebo on the ADAS-Cog was observed in all ST101 dosage groups pooled together. In a subgroup analysis performed post-hoc, patients with MMSE 10-17 were proven to have the greatest benefit from high dosages of ST101 (120 mg/d). 44
While the study displayed good methodology and a sufficient patient number (n = 210), a limitation is its short duration of 12 weeks.
Mowla et al 46 tested fluoxetine in combination with rivastigmine and found a statistically significant effect in ADL over rivastigmine monotherapy in patients with mild-to-moderate AD. No significant effects were observed on other outcomes, such as MMSE or WMS (Wechsler Memory Scale). This study also had a short duration of 12 weeks.
Lastly, Sun et al 47 investigated ChEI in combination with vitamin supplements containing Vitamin B6, B12 and folic acid in patients with mild to moderate AD. No significant effect was reported over ChEI with placebo after 26 weeks.
Adverse Events
Overall Adverse Event (AE) profiles of the drug combinations described above were comparable to control groups. Although not all studies reported absolute AE numbers, memantine, Gingko biloba extracts, idalopirdine, ST101, atorvastatin, fluoxetine and vitamin supplements were reported to be well-tolerated in the trial populations and generally safe to use in combination with ChEI. The study of Peters et al 37 was interrupted due to safety concerns raised by other studies, 50 regarding galantamine monotherapy in MCI patients. However, despite this, the combination therapy with galantamine and memantine proved to be safe and well-tolerated in the patient group of the study by Peters et al. 37
Regarding Gingko biloba, Yancheva et al 42 interestingly reported even less AEs in the combination group of memantine and donepezil than in the control group (donepezil with placebo). This suggests a high tolerability and safety of EGb 761®. 42
Discussion
Summary of Findings
Of 8 studies investigating ChEI in combination with memantine in various stages of AD, 3 have proven significant effects of additional memantine over ChEI monotherapy with placebo. In amnestic MCI, additional memantine in combination with galantamine led only to numerical effects over galantamine with placebo. While the results of one study investigating Gingko biloba in the form of EGb 761® in combination with donepezil did not reach significance, another study showed the effectiveness of a formula of Gingko biloba and other antioxidants in combination with donepezil over donepezil with placebo. Approaches using ChEI in combination with idalopirdine, atorvastatin or vitamin supplementation in mild to moderate AD did not show significant effects over ChEI monotherapy with placebo (at least in their singular trials). Combining fluoxetine with rivastigmine showed partial efficacy on ADL over rivastigmine monotherapy, however, this result must be replicated in trials of longer duration to justify clinical use. ST101 augmentation in donepezil-receiving patients with moderate AD has proven to be effective over donepezil and placebo in doses of 120 mg/d, especially in patients with MMSE of 10-17. The results of this single study should also be replicated in order to have clinical implications. All of the above-mentioned studies had too short a duration to be able to accurately predict cognition outcomes of the respective treatment approach after multiple years. However, exactly this is crucial in gaining a better understanding of the efficacy of AD drugs and the effects of various combinations, not just in the initial stages of therapy, but more importantly over the whole duration of the disease. Therefore, longer RCTs are essential, investigating all of the above-mentioned approaches.
Limitations
Although the MEDLINE and EMBASE database searches were carefully conducted using MeSH and Emtree terms, a possible evidence selection bias must be accounted for, especially considering the small initial result count of 114 articles (after duplicate removal) originating from the systematic search. The fact that 13.2% of these articles found by the systematic search qualified for inclusion, however, may indicate a strong initial search term. As the only databases searched were MEDLINE and EMBASE, relevant articles only available through other databases (e.g. Web of Science, PsycInfo) might have been missed. Seven additional sources were identified manually, of which only one (Peters et al 36 ) was included in the final analysis. Valuable evidence was gained from this study, however, the contribution of a manual search to an evidence selection bias cannot be ruled out.
Another important aspect to consider about this manuscript is that the newest study included had been published in 2018. A search via clinicaltrials.gov in June 2021 did not reveal any new studies completed after 2018, which fit all the inclusion criteria. One study published in 2019 by Atri et al is an extensive analysis of the study by Atri et al 43 in 2018 and therefore was not included.
Two possibly relevant studies in Chinese and Romanian had to be excluded due to the language barrier. Furthermore, and most importantly, no statistical analysis was performed in this systematic review, limiting its validity regarding clinical decision-making.
Outlook on Future Research Aims
Research on AD therapeutics is constantly evolving. According to a 2021 report, 126 AD drugs are currently being investigated in 152 trials, 28 of which are in phase 3 trials. 51 Of those 28 AD drugs in phase 3 trials in 2021, 60% are disease modifying therapies, most of them either targeting Aβ, or providing antioxidative or anti-inflammatory effects. 51 Aducanumab, the first drug to be approved by the FDA since almost 2 decades, is a monoclonal antibody targeting Aβ, and represents 1 of 4 biological drug approaches in phase 3 trial stage. 51 The others (gantenerumab, lecanemab and solanezumab) are also monoclonal antibodies with Aβ as a primary target. One has to note, however, that a phase 3 trial of gantenerumab in prodromal AD patients was stopped after futility analysis 52 and a phase 3 trial of solanezumab failed to prove effectiveness in mild AD, 53 diminishing hopes and expectations regarding these 2 drugs. In molecular comparison with those 2 antibodies, aducanumab, does shows significantly better Aβ aggregation kinetics, as Linse et al have shown in 2020. 54
In a clinical setting, on the other hand, aducanumab has produced ambiguous results in the EMERGE and ENGAGE trial and its preliminary FDA approval continues to be highly controversial.
Apart from the biological options, 24 approaches with small molecules in phase 3 trial stage, 13 of which can be considered disease-modifying and the 11 others rather symptomatic. 51
In this review, every included combination therapy uses standard ChEI treatment as part of the combined approach. ChEI, despite their limited efficacy on cognition outcomes, play a very important role in AD treatment strategies and will most likely continue to do so in coming years. However, the use of next-generation ChEI might offer expanded possibilities. As an example, agents like methanesulfonyl fluoride (MSF), an irreversible CNS-selective ChEI, that outperforms short acting inhibitors, 55 could have the potential to improve the well-established ChEI pillar of the AD treatment approach. Those new-line ChEI should be further evaluated against the established ones.
Speaking of well-established drugs, a continued effort is underway, exploring off-label use of drugs approved for other conditions and repurposing them to support treatment of AD patients. In this review, for example, off-label use was implemented in the form of add-on atorvastatin in combination with donepezil. Although this particular approach had failed, experiments like these could continue using drugs like the selective serotonin reuptake inhibitor paroxetine (FDA approved depression therapy) or lithium carbonate (FDA approved bipolar disorder treatment), which has been shown to inhibit GSK3β, a kinase involved in the development of tauopathy. 6
Dietary approaches might be of value, too. Caffeine, for example, has been associated with a significantly reduced risk of developing AD 56 and is currently investigated in a phase 3 trial in mild to moderate AD. 51
In terms of efficacy, there is still a long way to go. Also, as stated earlier, a critical issue is study duration. Of the AD phase 3 drug trials in 2021, the mean duration of exposure was 87 weeks for prodromal AD trials and 31 weeks for mild-to-moderate AD trials. 51 While this may represent an improvement over trials conducted in the past, it is still not entirely sufficient. Longer trials are needed.
While some treatments discussed in this review indeed have the potential to provide a small benefit for patients with AD, those benefits are only marginal and do not come without adverse events. It is clear, that we must set a considerably higher benchmark, when assessing the efficacy of these drug treatments in the future. As outlined by this review, combinations of already approved standard drug therapies (ChEI and memantine) are not likely to reach this benchmark. The evidence for a therapeutic benefit of additional memantine is ambiguous and even in studies favoring combination therapy this benefit is small, as was the case with Gingko biloba. As shown by one study in this review, combination approaches with ST101 could be of value, but need to be investigated further before use.
All in all, the extensive research efforts which are being made all over the world, especially those testing disease-modifying therapies, spark cautious optimism, that in the future, at least one of those drugs will meet the “efficacy benchmark” and provide the urgently needed relief for AD patients and their caregivers.
Conclusion
It can be concluded from this review that evidence for combination therapy in patients with AD remains highly contentious. While some well-designed trials suggest a slight cognitive benefit from use of memantine in combination with ChEI over ChEI monotherapy, other trials of at least comparable quality have failed to replicate this effect. Approaches with ChEI in combination with idalopirdine and atorvastatin have not shown success thus far, but each has been tested by only a single trial. Evidence on combination of ChEI with Gingko biloba is weak, with just one trial indicating slight benefits over ChEI monotherapy, as is the evidence for combinations of ST101 with donepezil as well as fluoxetine with rivastigmine in mild to moderate AD. The lack of clear evidence on these approaches warrants further research and effects should be replicated before clinically relevant decisions can be made.
The question of longevity of therapeutic effects remains to be an issue and should be investigated with further research into combined treatments and longer trials. Until then, cost-effectiveness relations must be individually considered by clinicians, although evidence suggests no major cognitive benefit will be missed, if no combination therapy is initiated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
