Abstract
Objective:
To identify determinants within 3 different domains (ie, somatic comorbidities, cognitive functioning, and neuropsychiatric symptoms [NPS]) of health-related quality of life (HRQoL) over time in memory clinic patients without dementia.
Methods:
This longitudinal multicenter cohort study with a 3-year observation period recruited 315 individuals (age: 69.8 ± 8.6, 64.4% males, Mini-Mental State Examination score 26.9 ± 2.6). A multivariable explanatory model was built using linear mixed effects models (forward selection per domain) to select determinants for self-perceived HRQoL over time, as measured by the EuroQoL-5D visual analogue scale (EQ VAS).
Results:
Mean HRQoL at study entry was 69.4 ± 15.6. The presence of agitation, appetite and eating abnormalities, and eyes/ears/nose (ie, sensory impairment) comorbidities were associated with a change in HRQoL over time. Agitation was most strongly associated with HRQoL over time.
Conclusions:
The association of somatic comorbidities and NPS in memory clinic patients with course of HRQoL shows that these should receive more awareness, detection, and monitoring by clinicians.
Keywords
Introduction
Cognitive decline is thought to have a profound negative impact on health-related quality of life (HRQoL), both in affected patients and their relatives. 1 As no disease-modifying treatment of (prodromal) dementia exists to date, enhancing and maintaining HRQoL is considered the most pivotal goal of management for all prodromal and clinical stages. 2 In the last decades, the broader concept of health and HRQoL has changed to become more dynamic and now includes the ability to adapt and self-manage in daily life despite certain impairments. 3
It has been argued that HRQoL follows the dementia process, where more severe diagnostic phases are associated with poorer HRQoL. 1,4 -8 Other, however, have reported limited changes in quality of life (QoL) over time in people with dementia (PwD), even in the presence of significant clinical deterioration, 5 and comparable HRQoL scores between mild cognitive impairment (MCI) and cognitively normal controls. 5,9 This suggests that other variables than disease stage determine HRQoL.
Several disease-related determinants have been reported to negatively influence HRQoL in PwD, such as the presence of neuropsychiatric symptoms (NPS), impaired activities of daily living, and severity of cognitive impairment (e.g., Study by Winzelberg et al 10 ). Data with regard to HRQoL in the prodromal stages of dementia are limited. In individuals with MCI, reduced HRQoL has been related to the presence of NPS, depressive symptoms in particular, 7,8 and decreased memory performance, 7 but not with general cognitive status 6 and executive functioning, language, and attention. 7
Most studies have been cross-sectional in nature, but research in this field is now moving forward to the examination of longitudinal changes in HRQoL. Depressive symptoms in cognitively impaired individuals have been associated with lower HRQoL at follow-up in some studies, 11,12 but not in others, 13 or only when both HRQoL and NPS were caregiver-rated. 14 Some studies showed that an increase in NPS over time was related to a decrease in HRQoL. 15,16 One study reported that the number of somatic comorbidites was related with a decrease in HRQoL at follow-up, 13 whereas another did not find any relation between general health and HRQoL. 17 Baseline cognition was unrelated to change of HRQoL in previous studies. 11 -14,18 However, none of these studies examined the course of HRQoL using multiple assessments over time, and all were limited to a sample of individuals with dementia.
An integrated view of determinants of the natural history of subjective HRQoL over time in prodromal stages of dementia is currently lacking. The relevance of identifying such determinants in memory clinic patients lies in their potential to target and personalize interventions with preventative and supportive strategies, thereby minimizing their impact on HRQoL. Hence, the aim of the present study was to identify the optimal combination of determinants of HRQoL over time in memory clinic patients without dementia at study entry.
Materials and Methods
The current study is part of the Dutch Clinical Course of Cognition and Comorbidity in Mild Cognitive Impairment (4C-MCI) study. 19 The 4C-MCI study is a longitudinal, multicenter study and focuses on the course of cognitive decline in nondemented memory clinic patients. The study included 315 participants at baseline, who were recruited at the memory clinics of Maastricht University Medical Centre, Radboud University Medical Center, and VU Medical Centre between January 2010 and May 2011, with a roughly equal distribution across centers (118, 98, 99 participants, respectively). Follow-up data were collected annually up to 3 years after baseline assessments. The medical ethical committee of each center approved the study. All participants gave written informed consent.
Inclusion criteria were (1) age ≥ 55 years, (2) having cognitive complaints and/or cognitive impairments, in the absence of dementia, and (3) Clinical Dementia Rating score ≤ 0.5. 20 Exclusion criteria were (1) absence of a primary informant, (2) prognosis based on clinical judgment that the participant would not be able to have at least 1 follow-up contact, and (3) the presence of specific neurological disorders possibly causing cognitive impairment, such as Parkinson’s or Huntington’s disease, normal pressure hydrocephalus, Korsakoff’s syndrome, a medical history of brain tumor or encephalitis. Participants having any other comorbidities, including cerebrovascular and psychiatric disorders, were not excluded in this study.
Baseline and Follow-Up Assessment
At baseline, all participants underwent a standardized clinical assessment, which included a detailed history of the patient, a psychiatric, neurological and physical examination, assessments of daily functioning, an extensive neuropsychological assessment, and a cerebral magnetic resonance imaging scan. These assessments were part of the regular patient diagnostic procedures of the memory clinics. Participants were invited to take part in a follow-up assessment at 1, 2, and 3 years after baseline. For the current study, we extracted data on age, gender, education, HRQoL, comorbid disease burden, and cognitive and emotional functioning.
Diagnostic Procedures
Syndrome diagnoses were based on clinical assessment by the physician and the multidisciplinary team. The diagnosis of MCI was based on the Petersen criteria.
21
Individuals with an objective cognitive impairment, that is, a
Quality of Life
The Visual Analogue Scale (VAS) of the EuroQoL-5D (EQ-5D) 23 was used to measure self-rated HRQoL. The EQ VAS is a standard vertical scale to record individuals’ ratings for their current HRQoL state, ranging from 0 to 100, with a higher EQ VAS indicating better HRQoL. The rationale for using the VAS-score and not the 5 dimensions of the EQ-5D is that the latter focuses on functioning and consequent impairments on mobility, self-care, usual activities, pain or discomfort, and anxiety or depression, while our aim was to evaluate the subjective rating of current well-being and HRQoL, in line with previous conducted studies. 24,25 In addition, by using the VAS-score, the overlap with the domains of NPS and somatic comorbidities in the predictive model is minimized.
Somatic Comorbidities
The Cumulative Illness Rating Scale for Geriatrics (CIRS-G) 26 was used to rate all available data on medical comorbidities, medication use, smoking and drinking habits, and the physical examination. Scores between 0 (no problems) and 4 (extremely severe problems) were given to 14 categories of organ systems (ie, cardiac, vascular, hematopoietic, respiratory, eyes/ears/nose/throat/larynx, upper gastrointestinal tract, lower gastrointestinal tract, liver, renal, genitourinary, musculoskeletal, neurological, endocrine/metabolic and breast, and psychiatric). For the current analyses, we excluded the psychiatric category of the CIRS-G. By excluding this category, the CIRS-G was used as a measure of medical comorbid disease burden only, thus minimizing overlap with the cognitive and emotional functioning domain. Scores of the subcategories were dichotomized as comorbidity present (score of 2 or higher, ie, moderate, severe, and extremely severe disease severity) or absent (score of 0 or 1).
Cognitive Functioning
The neuropsychological assessment consisted of a standardized battery of cognitive tests. Global cognitive functioning was assessed by means of the Mini-Mental State Examination (MMSE).
27
Episodic memory was assessed by use of the Verbal Learning Task (VLT).
28
Information processing speed and executive functions were measured using the Stroop Color Word Test (SCWT),
29
the Letter Digit Substitution Test (LDST),
30
and the Trail Making Test (TMT).
31
Verbal fluency was assessed by use of the Category Fluency (1-minute animal naming).
32
In accordance with the available Dutch normative data, raw test scores were converted to
Neuropsychiatric Symptoms
The Neuropsychiatric Inventory (NPI) 33 was used to assess the frequency and severity of 12 NPS (ie, delusions, hallucinations, agitation, depression, anxiety, apathy, irritability, euphoria, disinhibition, aberrant motor behavior, nighttime behavior disturbances, and appetite/eating disturbances) through a structured interview with an informant. For each symptom, severity and frequency scores are multiplied to acquire a domain score, with higher scores indicating more severe problems. Symptom scores were dichotomized as present (domain score of 1 or higher) or absent (domain score of 0).
The Short Form of the Geriatric Depression Scale (GDS-15) 34 was used to determine the presence and severity of depression by self-rating. The questionnaire does not include somatic symptoms which might be present due to comorbid somatic disorders. Following prior studies, scores were dichotomized with a score of 6 or higher being indicative of depression. 35
Statistical Analyses
Analyses were performed using SPSS version 25 (Chicago, Illinois) for Mac OS X. Baseline differences between groups were analyzed using χ2 tests for categorical variables and
To test for multicollinearity, Spearman’s rank-order correlations were computed between all variables within a predictive domain (ie, somatic comorbidities, cognitive functioning, and NPS). Using a cutoff of 0.7, the VLT-delayed recall score was removed from the analyses, since it correlated highly with the VLT-immediate recall score (
The optimal combination of determinants of HRQoL over time was examined by modeling linear mixed effects (growth curve) models. This analysis models individual growth curves that take within-subject correlation between repeated measures into account, thus accounting for the hierarchical structure of the data (ie, time nested in individuals). Missing data can be considered at random when we include the covariates that are associated with missingness in the analyses. 36 The missing data points are estimated by maximum likelihood. Thus, these models allow the use of all available longitudinal data, including data from dropouts.
First, the association of each determinant with HRQoL over time was analyzed separately, corrected for age at baseline, sex, education (low, middle, and high), and study center. An unconditional means model was fitted with random intercepts (ie, patient factor as random), to account for the correlation between repeated measures within individuals. Next, cognitive measures, somatic comorbidities, NPS, and time and interaction terms between each variable and time were added as fixed effects. Time (ie, point of follow-up) was measured in years and used as a categorical variable to allow discontinuous change between the follow-ups. The variance component structure was specified according to best fit model based on likelihood ratio testing. Time as random slope was allowed if the model was significantly better compared to a model with only random intercepts.
Afterward, a multivariable model was built using forward selection. Per domain (ie, cognitive domain, somatic comorbidities domain, and NPS domain), the variable with the lowest
Results
Baseline participant characteristics are presented in Table 1. Participants (64.4% male) were on average 69.8 (SD = 8.6) years old. At study entry, 104 (33.0%) individuals had SCD, 27 (26.0%) of whom converted to MCI, and 16 (15.4%) to dementia over the course of the study period (up to 3 years); 211 (67.0%) individuals had MCI, 45 of whom (21.3%) converted to dementia. Of these 315 patients, 247 (78.4%) completed the 1-year follow-up assessment, 225 (71.4%) patients completed the 2-year follow-up assessment, and 198 (62.9%) patients completed the 3-year follow-up assessment. Dropouts were on average older, lower educated, performed worse on several cognitive tests (VLT, fluency, LDST, and MMSE) and had more often somatic comorbidities (hematopoietic, upper digestive tract, and kidney conditions) compared to those with at least 1 follow-up visit. Prevalence of NPS and HRQoL ratings were similar in both groups. The mean EQ VAS score of the entire group at baseline was 69.4 (SD = 15.6), which on average remained stable over time (
Baseline Participant Characteristics.a
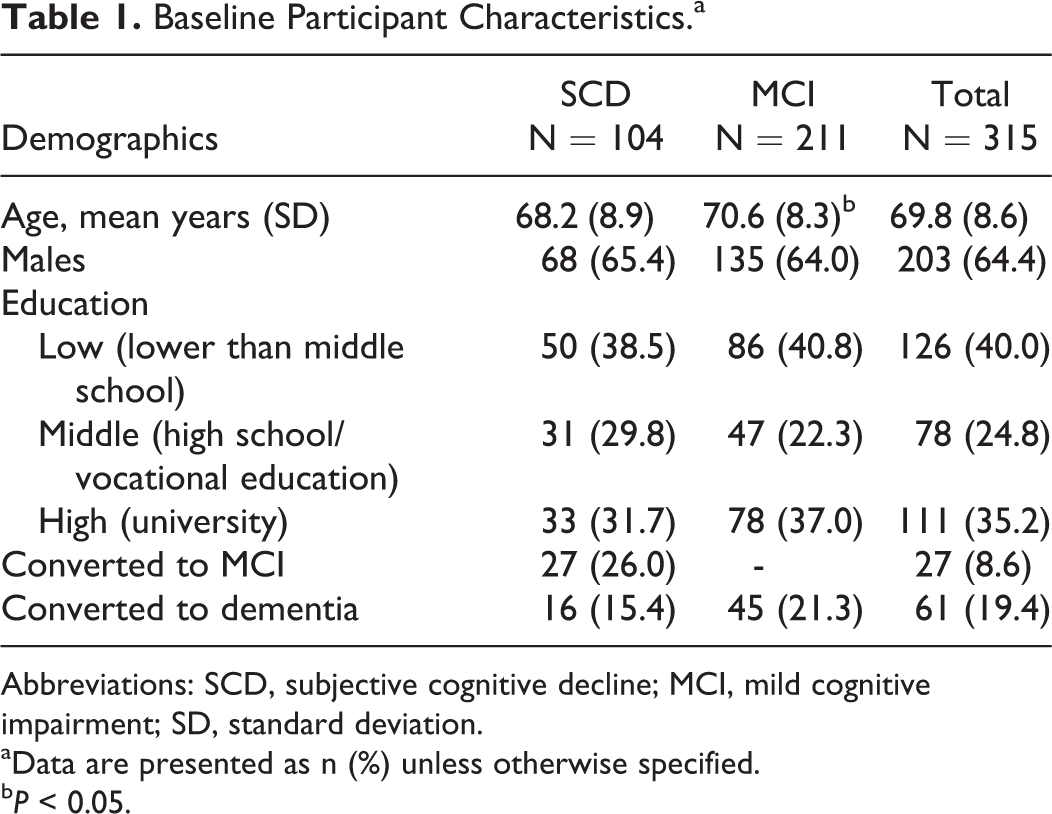
Abbreviations: SCD, subjective cognitive decline; MCI, mild cognitive impairment; SD, standard deviation.
aData are presented as n (%) unless otherwise specified.
b
At baseline, the presence of appetite and eating abnormalities, nighttime behavior disturbances, lower digestive tract comorbidities, and self-reported depression was significantly associated with lower HRQoL ratings (Table 2, Figure 1, and Online Appendix Table 2). Linear mixed models were used to evaluate the associations between the individual determinants and HRQoL over time. Eyes/ears/nose comorbidities, presence of agitation, lower digestive tract, urogenital comorbidities, presence of appetite and eating abnormalities, nighttime behavior disturbances, and self-reported depression were associated with HRQoL over time and included in the multivariable analyses.
Mean Differences in Baseline QoL Scores and in Rate of Change (Slopes) From Baseline to Follow-Up Between Participants With and Without Baseline Determinants.a
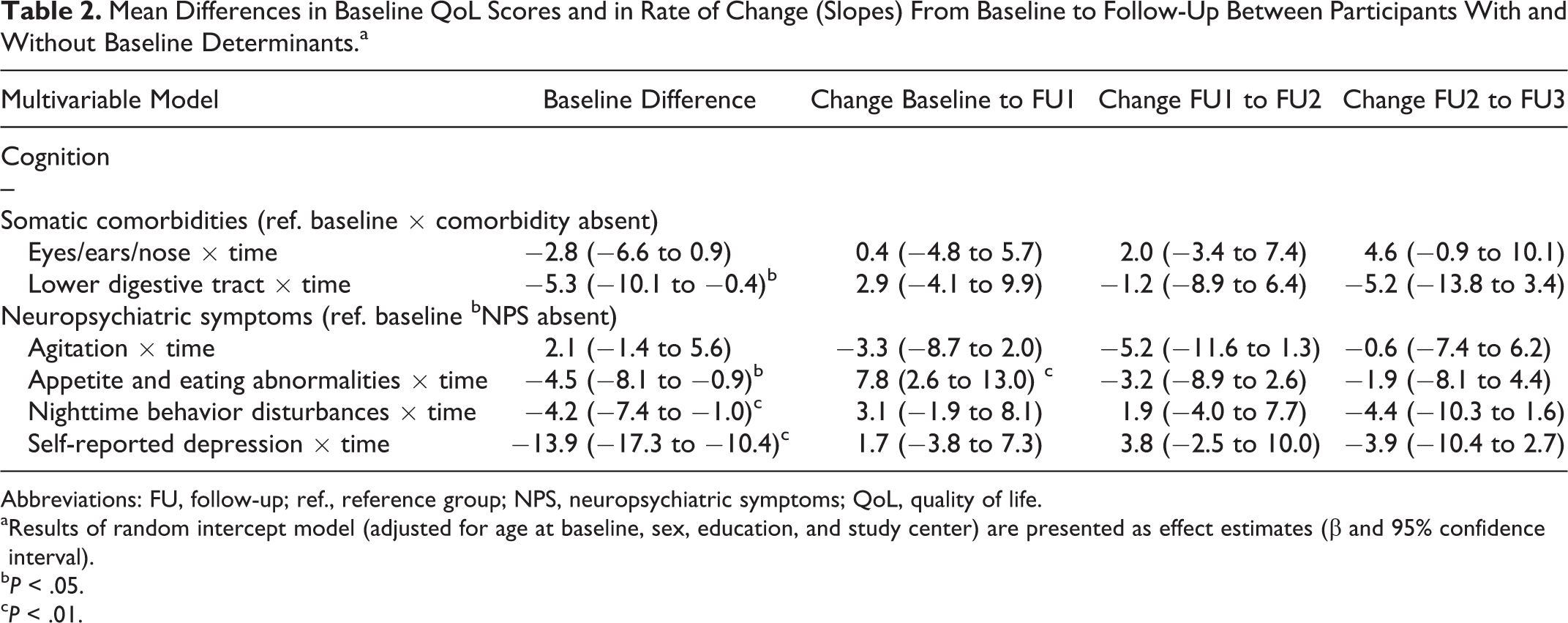
Abbreviations: FU, follow-up; ref., reference group; NPS, neuropsychiatric symptoms; QoL, quality of life.
aResults of random intercept model (adjusted for age at baseline, sex, education, and study center) are presented as effect estimates (β and 95% confidence interval).
b
c
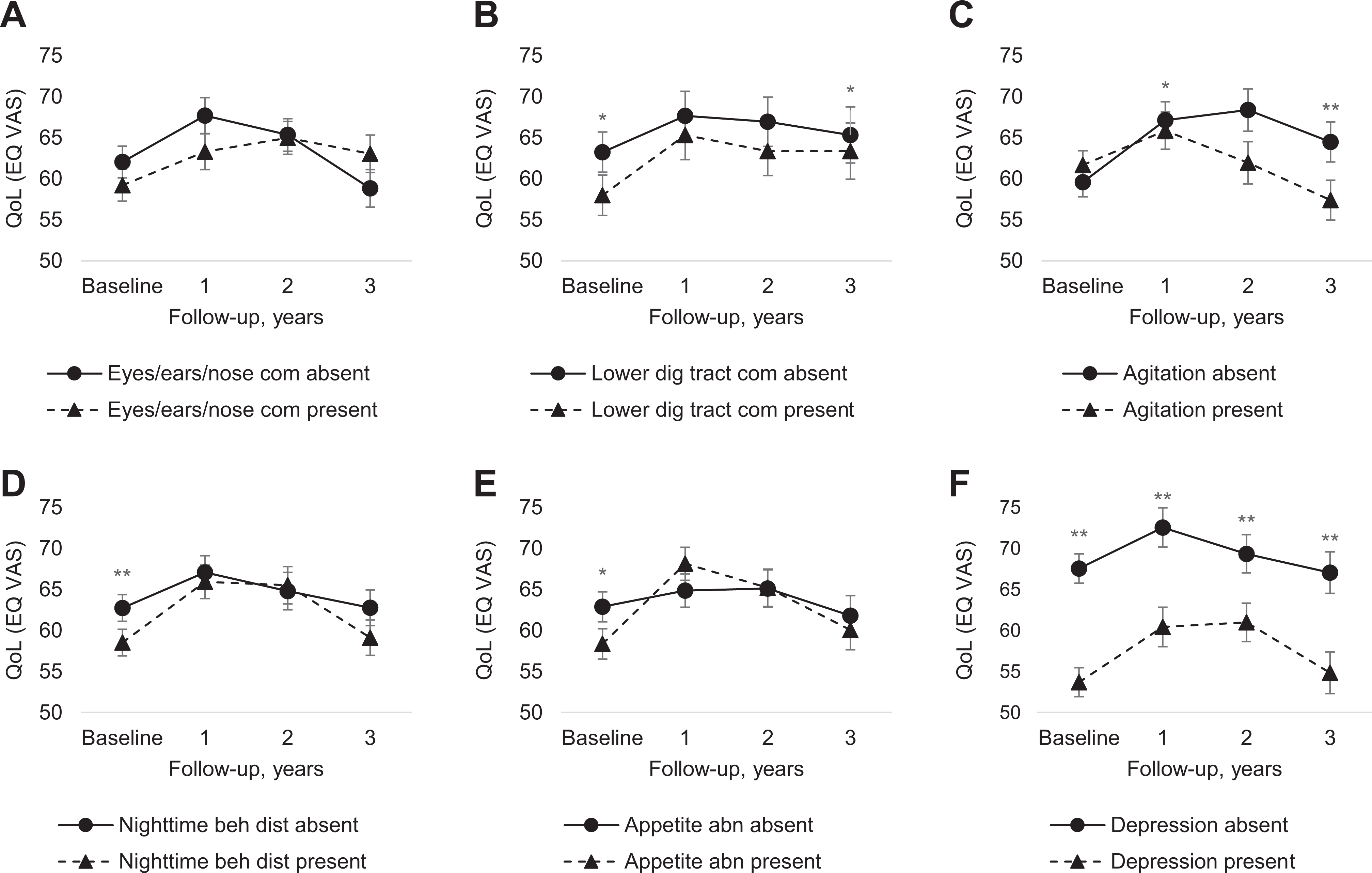
QoL over the course of the study period by status of baseline determinants.(A) Course of QoL by eyes/ears/nose comorbidities group. (B) Course of QoL by lower digestive tract comorbidities group. (C) Course of QoL by agitation group. (D) Course of QoL by nighttime behavior disturbances group. (E) Course of QoL by appetite and eating abnormalities group. (F) Course of QoL by self-reported depression group. Based on random intercept analyses adjusted for age at baseline, sex, highest level of education, and study center. Shown is the predicted mean score (estimated marginal means of time by group) and standard error. Appetite abn indicates appetite and eating abnormalities; EQ VAS = EQ-5D Visual Analogue Scale; Eyes/ears/nose com = eyes/ears/nose comorbidities; Lower dig tract com = lower digestive tract comorbidities; Nighttime beh dist = nighttime behavior disturbances; QoL = quality of life.*
Using forward selection, the combined model included the following variables. The presence of nighttime behavior disturbances (
Discussion
In this longitudinal, multicenter study, the best fitting final model to explain HRQoL over time consisted of several somatic comorbidities (ie, eyes/ears/nose and lower digestive tract conditions) and NPS (ie, agitation, appetite, and eating abnormalities; nighttime behavior disturbances; and self-reported depression). In contrast, cognitive functioning did not predict HRQoL over time. The most consistent determinant of HRQoL over time was the presence of agitation.
Overall, an initial increase of HRQoL was observed at 1-year follow-up. This might (partially) be the result of a diagnosis disclosure effect as participants were included at first attendance at the memory clinic. 37 However, this effect appeared to diminish over time, as reflected by the decrease of HRQoL at 2 and 3-year follow-up. Hence, even when memory clinic patients show an initial increase in HRQoL after the diagnostic process, it is important to follow these individuals over time and continue to provide support.
From the list of putative NPS determinants, agitation was the most consistently associated with HRQoL over time. Previous studies 7,8,9,38 which investigated the relationship between NPS and HRQoL in SCD or MCI only focused on depressive symptoms or total NPI scores, while in dementia, the presence of agitation has been associated with lower HRQoL cross-sectionally 39,40 and with a decrease of HRQoL over time. 41 Agitation can be interpreted as an expression of emotional distress, manifested in excessive motor activity, or verbal or physical aggression. 42 Clinicians should be aware of the influence of caregiver management strategies on patient behavior, as it has been shown that caregiver nonacceptance resulted in more patient hyperactivity behaviors. 43
Next to agitation, self-reported depressive symptoms were associated with lower HRQoL at baseline. The association between depressive symptoms and reduced HRQoL has been reported for both individuals with MCI 6 -8 and SCD. 2 More severe depressive symptoms have been associated with better insight into cognitive impairments, 44 which suggests that depressive symptoms might be a psychological reaction to the disease in individuals whose illness insight is intact (as can be expected in our sample with an average relatively mild cognitive deficits), although several other hypotheses have been posed to explain the presence of depression in individuals across the disease spectrum. For example, in the prodromal hypothesis, depression is considered a noncognitive manifestation of underlying neurodegenerative pathology. On the other hand, the risk factor hypothesis states that the presence of depression itself lowers the brains reserve to cope with Alzheimer's disease pathology, for example, via hypothalamic–pituitary–adrenal -axis dysregulation, and thus fastens progression of the disease. Here, we found a higher prevalence of informant-rated depressive symptoms compared to self-rated depressive symptoms (40% vs 20%), HRQoL only related to the latter one. In line with the psychological-reaction hypothesis, this suggests that subjective ratings of lower HRQoL by patients with cognitive deterioration reflect a psychological reaction to the decline and that this is not recognized by the informants, as corroborated by the finding that only 70% of the self-reported cases with high levels of depressive symptoms was recognized by the informant as such.
In addition, appetite and eating abnormalities were related to change in HRQoL over time. Previous studies have shown that appetite and eating abnormalities were unrelated to HRQoL in patients with mild to moderately severe dementia. 39,40 This discrepancy might be due to the lack of disease insight which occurs more often in the later stages of the disease, that is, the dementia phase versus the SCD and MCI stages, as in the current study. Furthermore, nighttime behavior disturbances were associated with baseline HRQoL. In a recent state-of-the-art review on persons with dementia, sleep disturbances, conceptualized as poor sleep efficiency, and increased night awakening, were associated with all four HRQoL domains as defined by Lawton 45 : physical function, social/behavioral function, emotional well-being, and cognitive function. 46
Although our findings reveal high informant-reported symptoms of irritability and apathy (respectively 51.7% and 43.7%), these were not related to HRQoL. This is in contrast to prior research where higher levels of irritability and apathy were associated with lower HRQoL in mild dementia. 39,47 However, Yeager and Hyer 47 assessed apathy by self-report, which is prone to information bias as reduced disease insight can influence the patient-reported HRQoL.
The effect of specific comorbidities on HRQoL in SCD and MCI has not been investigated before, although comorbidity burden has been related to the progression of disease. 48 In the current study, eyes/ears/nose and lower digestive tract comorbidities were found to be determinants of lower HRQoL over time. The association between hearing and vision problems and lower HRQoL has also been reported in older adults in general and in the nursing home population. 49,50 Sensory impairment in older adults resulted in the restriction of activities of daily living (ADL), 51 which might cause a decrease in self-esteem. Also, hearing difficulties may result in social isolation, 52 while positive social relationships have been shown to be associated with higher HRQoL. 39 Lower gastrointestinal tract disorders, such as functional gastrointestinal disorders (disorders of the gut–brain interaction), are common in patients with mild psychiatric disorders such as anxiety and depression, which are also often seen in individuals with cognitive dysfunction. 53 An increase in bowel symptoms could, therefore, link to deterioration of the central nervous system. 54 On the other hand, the gastrointestinal symptoms can have a significant impact on HRQoL in affected individuals due to the nature of the symptoms themselves, for example, via impaired physical or social functioning. 55
No association between cognition and HRQoL over time was found in the current study, which is in line with previous cross-sectional studies in SCD or MCI 7,38 and longitudinal association in SCD 25 and dementia. 11 -14,18,56 These prior studies only used global cognitive screening instruments 11 -14,38,56 or composite cognitive domain scores 7,18 to assess cognitive functioning, which are less sensitive to detect associations. Therefore, we looked at specific cognitive tests, but also in this manner, no association was found. Together these findings seem to imply that cognitive deterioration in itself is not associated with HRQoL in memory clinic patients, whereas NPS and somatic comorbidities seem to be more directly affecting the subjective burden of patients.
This study has several strengths, of which most notably the longitudinal representative sample of memory clinic patients (by keeping the exclusion criteria to a minimum), repeated measures of HRQoL, its sample size, and the broad range of possible predictors of HRQoL. Validated measures were used to assess HRQoL, cognitive functioning, NPS, and somatic comorbidities. Certain limitations should also be acknowledged. Bias could have been introduced by the fact that the NPI was rated by informants. Salient symptoms might be more often reported because internal psychological reactions might be difficult to recognize for informants and difficult to communicate for patients with cognitive impairments. Indeed, the NPI items that refer more to concrete behavior (eg, eating and nighttime behavior disturbances) and are less likely to be influenced by the informants’ perception were related to HRQoL. Furthermore, a nondisease-specific HRQoL questionnaire was used. However, the EQ VAS has been shown to be a valid and reliable measure in individuals with cognitive impairments 57 and has been used in studies with similar research questions. 24 Moreover, the EQ VAS was specifically chosen for the present study to assess self-perceived HRQoL, in line with the conceptualization of HRQoL as the ability to adapt to the perceived consequences of dementia. 45,58 Still, a more specific HRQoL instrument for individuals with cognitive impairment could have been implemented. In addition, (selection-) bias could have been introduced, evidenced by the finding that individuals with follow-up data available were healthier at baseline. It might be argued that analyzing the SCD versus the MCI groups separately would have resulted in different outcomes. However, stratifying the sample according to diagnosis resulted in roughly similar average effect estimates (results not shown). Most importantly, within each subgroup, cognition was not associated with QoL. In this line, inclusion of the VLT delayed recall score rather than the immediate recall score could have changed the results as these represent two different constructs. Sensitivity analysis showed that neither the immediate nor the delayed VLT recall scores were significantly associated with HRQoL, while fitting the univariate linear mixed effects models. The choice of construct thus did not have an influence on the final model. Finally, although an extensive amount of predictors for HRQoL was used, other possible predictors could not be included, such as activities in daily life, socioeconomic status, and factors associated with autonomy and relationships. 40,59
Conclusions
In people without dementia visiting a memory clinic, specific somatic comorbidities and NPS predicted the level of HRQoL over time. Overall, there was an initial increase of HRQoL during the first year which was followed by a decrease of HRQoL in subsequent years. Therefore, it is important to follow individuals for a longer period of time and to continue providing support. These findings may give direction for tailoring interventions toward personalized needs and may improve HRQoL of memory clinic patients in the future. Future research should focus on the effect of treatment of somatic comorbidities and NPS on HRQoL in individuals with memory complaints.
Supplemental Material
Supplemental Material, Appendix_revised - Determinants of Cross-Sectional and Longitudinal Health-Related Quality of Life in Memory Clinic Patients Without Dementia
Supplemental Material, Appendix_revised for Determinants of Cross-Sectional and Longitudinal Health-Related Quality of Life in Memory Clinic Patients Without Dementia by Leonie C. P. Banning, Eveline P. C. J. Janssen, Renske E. G. Hamel, Marjolein de Vugt, Sebastian Köhler, Claire A. G. Wolfs, Saskia M. Oosterveld, Rene J. F. Melis, Marcel G. M. Olde Rikkert, Roy P. C. Kessels, Yolande A. L. Pijnenburg, Ted Koene, Wiesje M. van der Flier, Philip Scheltens, Pieter Jelle Visser, Frans R. J. Verhey, Pauline Aalten and Inez H. G. B. Ramakers in Journal of Geriatric Psychiatry and Neurology
Footnotes
Acknowledgments
The authors thank Nico Rozendaal for his help with the design of the database and data management of the 4C-study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was performed within the framework of the Clinical Course of Cognition and Comorbidity in MCI (4C-MCI) Study and was funded by Alzheimer Nederland (grant number: 20083494).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
