Abstract
Background:
Delayed secretory activation can negatively impact maternal and child health through increasing the risk of early cessation of breastfeeding. Early cessation of breastfeeding is known to be associated with increased health risks.
Aim:
To determine the occurrence of, and risk factors associated with, delayed secretory activation in nulliparous women in Western Australia.
Methods:
This analysis used a subset of participants (n = 183) enrolled in a multicentre trial. Nulliparous low-risk pregnant women were enrolled between 28–32 weeks’ gestation and followed until 1 week after birth. The association between self-reported delayed secretory activation status (onset 4 days or more postpartum) and potential risk factors across the following five dimensions was examined: maternal demographics and health behaviours during pregnancy; maternal anthropometry; obstetric and birth outcomes; newborn characteristics; and infant feeding. Any variables that demonstrated significance (p ≤ 0.10) were entered into binary logistic regression models to quantify their association with delayed secretory activation and adjusted for known confounders.
Results:
The rate of delayed secretory activation was 43.2% (n = 79) and was significantly associated with body mass index (BMI) category 25 kg to <30 kg/m2: OR 2.83 (95% CI 1.39, 5.77), BMI ≥30 kg/m2: OR 2.51 (95% CI 1.13, 5.55), and gestational diabetes mellitus (GDM): OR (5.06; 95% CI 1.28, 20.08) in bivariate analysis and in the adjusted multivariable model. Among mothers with GDM, 78.6% experienced delayed secretory activation, compared to 21.4% of mothers who did not have GDM.
Conclusion:
Nulliparous women, particularly those with GDM, should be supported to initiate early, frequent, and optimal breast stimulation to exclusively breastfeed.
Keywords
Background
Breastfeeding provides both short- and long-term health benefits for infants and their mothers (Binns et al., 2016). Both the World Health Organization (WHO) and the National Health and Medical Research Council (NHMRC) in Australia recommend initiating breastfeeding within the first hour of birth and maintaining exclusive breastfeeding for the first 6 months of life to support optimal growth and development of the infant (National Health and Medical Research Council, 2012; World Health Organization, 2017). The breastfed infant has a reduced risk of gastrointestinal and respiratory infections (Horta et al., 2013); becoming overweight or obese; and developing type II diabetes in later life (Horta et al., 2023). The breastfeeding mother has a lower risk of developing type II diabetes and ovarian and breast cancers (Victora et al., 2016).
Lactation begins at conception, with alveolar development in early pregnancy leading to the formation of the milk secretion unit (Pang & Hartmann, 2007). During pregnancy, secretory differentiation (formerly lactogenesis I) occurs as mammary epithelial cells differentiate into lactocytes capable of synthesising breast milk components (Boss et al., 2020). Secretory activation (formerly lactogenesis II) marks the onset of copious milk secretion and occurs typically around 60 hours postpartum but may range from 24 to 72 hours, provided adequate prolactin, insulin, and cortisol are present (Kent, 2007). Secretory activation is commonly described as milk “coming in” and is associated with breast fullness, swelling, and occasional milk leakage (Lawrence & Lawrence, 2016; Preusting et al., 2017). Secretory activation occurring more than 72 hours after birth is considered delayed secretory activation (Lawrence & Lawrence, 2016).
Delayed secretory activation puts infants at a greater risk of excessive weight loss and subsequent infant formula supplementation even when their mothers intend to exclusively breastfeed (Nommsen-Rivers, 2016). Preglandular causes of impaired lactation can include diabetes, both pregestational and gestational diabetes, maternal prepregnancy overweightness or obesity, thyroid dysfunction, polycystic ovary syndrome, and retained placental fragments. Breast surgery and a lack of glandular tissue development are considered glandular causes of insufficient lactation, and post-glandular causes are infant dependent (e.g., inadequate latch or breast emptying), which can be brought about by prematurity (Farah et al., 2021). If delayed secretory activation occurs, healthcare professionals may advise mothers to supplement their infants with infant formula until copious breast milk is established. Understanding the risk factors for delayed secretory activation in nulliparous women should be regarded as a public health priority, as in-hospital formula supplementation may hamper breastfeeding efforts. In-hospital formula supplementation has been associated with shorter breastfeeding duration (McCoy & Heggie, 2020) and can reduce a mother’s breastfeeding confidence (Liu et al., 2023).
Primiparity has been well-established as a risk factor for delayed secretory activation (Peng et al., 2024). Multiparous mothers may experience secretory activation sooner than primiparous mothers because multiparous women are more likely to breastfeed their infants sooner in the first 24 hours postpartum (Neves et al., 2020). Multiparous mothers are also more likely to breastfeed more frequently in the first 24 hours postpartum (Hackman et al., 2015). On the other hand, primiparous mothers are more likely to have insecurities and anxiety around breastfeeding (Lindblad et al., 2022; Nakamura et al., 2020) and experience breastfeeding difficulties such as problems with feeding in hospital (Hackman et al., 2015).
There is evidence that diabetes in pregnancy is associated with delayed secretory activation, although other potential confounders occur more frequently in women with diabetes in pregnancy, such as higher maternal prepregnancy body mass index (BMI), caesarean birth, and management of hypoglycaemia in the infant using infant formula during the birth hospitalisation (De Bortoli & Amir, 2015). Researchers found in a pilot study of pregnant women with type 2 diabetes that higher insulin doses in pregnancy were associated with increased time to secretory activation (Britten et al., 2022). In mothers without preexisting diabetes (type 1 or type 2), research has shown that the time until the first breastfeeding is associated with delayed secretory activation. Women whose first breastfeeding was ≥2 h postpartum were more likely to experience delayed secretory activation than women whose first breastfeeding occurred ≤1 h postpartum (OR 2.43, 95% [CI 1.09–5.45]) (Mullen et al., 2022). Moreover, the researchers found the time to first breastfeed was an independent predictor of delayed secretory activation after adjustment for age, race, prepregnancy BMI, primiparity, and mode of birth.
To date, the risk factors for delayed secretory activation have not been investigated in nulliparous women in Western Australia. Earlier research in Western Australia found primiparity and caesarean birth to be risk factors for delayed secretory activation; however, this research was conducted over a decade ago and included both primiparous and multiparous mothers without stratification or adjustment (Scott et al., 2007).
We therefore aimed to examine risk factors for delayed secretory activation across five dimensions: maternal demographics and health behaviours during pregnancy; maternal anthropometry; obstetric and birth outcomes; newborn characteristics; and infant feeding and their association with delayed secretory activation in a cohort of nulliparous women in Western Australia.
The hypothesis for this study was that maternal prepregnancy BMI >25 kg/m2 and gestational diabetes (GDM), diet-controlled and insulin-dependent diabetes, would be risk factors for delayed secretory activation in nulliparous women in Western Australia. We also hypothesised that delayed secretory activation would be associated with formula supplementation during the hospital stay.
Key Messages
• Delayed secretory activation can negatively impact maternal and child health through increasing the risk of early cessation of breastfeeding, hence this study aimed to determine the occurrence of, and risk factors associated with, delayed secretory activation in a cohort of nulliparous women in Western Australia (WA).
• Prepregnancy body mass index (BMI) 25 kg–29.9 kg/m2, BMI ≥30 kg/m2, and gestational diabetes mellitus were significantly associated with delayed secretory activation in a cohort of nulliparous women in WA who were intending to breastfeed.
• The findings of this study highlight the importance of educating women of childbearing age on the potential risks of having a BMI ≥25 kg/m2 prior to becoming pregnant and the associated increased risk of gestational diabetes mellitus.
Methods
Research Design
This was a secondary analysis of a randomised controlled trial. Data used in this analysis were obtained from a subset of participants in the control arm of the Antenatal Colostrum (ACE) Study in Perth, Western Australia.
The ACE Study is being conducted according to the guidelines laid down in the Declaration of Helsinki, and all procedures involving human subjects/patients were approved by the Human Research Ethics Committee at Edith Cowan University. The ACE Study has been approved by Ramsay Health Care Western Australia and South Australia, HREC (reference number: 1901), and Women and Newborn Health Service Ethics Committee (RGS0000001504). The ACE Study has been registered as a clinical trial with the Australian New Zealand Clinical Trials Registry (Universal Trial Number: U1111-1232-1397, https://anzctr.org.au/Trial/Registration/TrialReview.aspx?id=377448). The ACE Study is being conducted according to the strict protocols of the NHMRC National Statement (National Health and Medical Research Council et al., 2007).
Setting and Relevant Context
Data was obtained from participants who reside in Perth, the capital city of Western Australia, which accounts for 33% of the Australian continent and 10% of the country’s population. The participants gave birth at one of several hospitals: Joondalup Health Campus (JHC), the largest health care facility in Perth’s northern suburbs (a combined public and private hospital); Glengarry Private Hospital, a private hospital 15 km north of Perth’s central business district; and Armadale Hospital, a general public hospital in Perth’s southeastern suburbs. The hospital sites in this trial are not accredited as part of the Baby-Friendly Hospital Initiative (BFHI).
National Health Survey data in Australia show that the prevalence of exclusive breastfeeding at 4 months of age is 63.9% and drops to 37.5% by 6 months of age (Australian Bureau of Statistics, 2022). National Health Survey data in Australia is obtained to collect a wide range of information about the health of Australians, with data collected from approximately 13,100 households via personal interview. Households were randomly selected to participate.
Sample
The ACE Study is a stepped-wedge cluster (nested) randomised controlled trial in nulliparous pregnant women recruited from multiple hospital sites (Cuffe et al., 2024). Antenatal colostrum expressing has increased in popularity in Australia since the publication of research by Forster et al. (2017) that determined antenatal colostrum expressing was safe for women with low-risk diabetes in pregnancy from 36 weeks’ gestation. However, the safety and efficacy of antenatal colostrum expressing for improving breastfeeding outcomes in pregnant women without diabetes in Australia has not been established. The ACE Study aims to determine the safety of antenatal colostrum expressing; whether antenatal colostrum expressing improves short- and long-term breastfeeding rates; and whether learning to practice antenatal hand expressing via video instruction is noninferior to one-on-one instruction from a midwife (Cuffe et al., 2024).
The data used in this secondary data analysis were obtained from participants who received standard maternity care (no intervention provided), whereas participants in the intervention group of the ACE Study received antenatal colostrum-expressing instruction during the antenatal period via one-on-one instruction from a midwife or from an instructional video. Data used in this analysis were from participants who were followed until 1 week postpartum. Data were extracted from medical records and antenatal questionnaires that participants completed via an electronic survey link that was sent via email prior to birth. Participants were provided a unique access code for the online questionnaire. Postnatal questionnaires were completed at 1 week postpartum via telephone interview with a researcher.
Participants were recruited in their last trimester of pregnancy before 32 weeks’ gestation. Pregnant individuals birthing at hospital trial sites who met the inclusion criteria were approached to participate by either the midwife providing their antenatal care or a study researcher. The midwife conducted an eligibility screening to determine if the patient met the inclusion criteria before providing the patient with the Participant Information Form (PIF) and obtaining contact details (telephone/mobile number and email address). Screening was based on the medical and demographic information obtained before 32 weeks’ gestation. From medical records and this contact, the midwife determined if the patient met the inclusion criteria for the trial. The contact details of pregnant individuals who met the eligibility criteria and demonstrated interest in the trial were provided to the researcher. Individuals were contacted via telephone to discuss the trial and given an opportunity to ask questions before consenting. If verbal consent was given, the electronic consent form was sent via email. Alternatively, pregnant individuals were recruited in the antenatal care waiting room by a study researcher or at antenatal education group classes conducted at the hospital sites. The researcher conducted the eligibility screening, collected contact details from the patient, and provided the PIF to the participant prior to enrolment. Participants were not provided with compensation to participate in the ACE Study.
The inclusion criteria for the trial were 1) singleton pregnancy, 2) planning to breastfeed, and 3) able to read and speak in English. Pregnant women were ineligible to participate if they had the following maternal or infant medical conditions: Edinburgh Postnatal Depression Scale (EPDS) score ≥13, Perinatal Anxiety Screening Scale (PASS) score ≥26, antepartum haemorrhage, history of threatened/actual premature labour, cervical insufficiency, pre-eclampsia, fetal anomaly, and fetal compromise. Women remained eligible if they gave birth to a healthy infant born ≥37 weeks’ gestation. The additional inclusion criteria for this analysis were complete data for: 1) maternal prepregnancy height and weight measurements, 2) the time of secretory activation (number of days postpartum), and 3) data from the 1-week postpartum questionnaire and medical records. Women were excluded from this analysis if they did not breastfeed during the birth hospitalisation or never breastfed.
Measurement
The time of secretory activation was self-reported by mothers at 1 week postpartum during the postnatal questionnaire. Using a version of the two-question Assessment of Onset of Lactogenesis II adapted by Demirci et al. (2023), mothers were asked, “How long did it take your milk to ‘come in’ after your baby was born (i.e., when did you notice a big increase in the amount of milk and a feeling of breast fullness)?” Answers were recorded to the nearest day—that is, “1 day or less,” “2 days,” “3 days,” “4 days,” “more than 4 days,” “my milk never came in,” “I don’t remember when my milk came in” (Cuffe et al., 2024). The primary outcome was time of secretory activation and was categorised as not delayed (3 days or less postpartum) or delayed (4 days or more postpartum). Maternal self-reporting was used to determine secretory activation, as this was the most viable measure. Maternal perception is closely aligned with biochemical markers of secretory activation (Poeliniz et al., 2020).
Potential risk factors for delayed secretory activation were collected across five dimensions as follows.
Data Collection
The data used in this secondary data analysis was obtained from 183 participants who were a subset of participants recruited into the ACE Study between September 2019 and December 2023. The ACE Study extracted data from medical records and antenatal questionnaires that participants completed via an electronic survey link that was sent via email prior to birth. Participants were provided a unique access code to access the online questionnaire. Mothers’ breastfeeding intentions prior to birth were collected in the antenatal questionnaire. During the antenatal period, participants were informed they would be asked about infant feeding after birth. Data collection related to lactation and breastfeeding after birth was conducted via telephone-administered postnatal questionnaires by an ACE Study researcher at 1-week postpartum. Participants who could not complete the postnatal questionnaires via telephone (n = 7) were sent an email link to complete the questionnaire online. With written consent, the ACE Study accessed participants’ medical records to obtain maternal and infant medical details and birth-related factors. Participants in the ACE Study received a unique study identification number, and their medical notes were attached to this number to ensure confidentiality. The data used for this secondary analysis were provided by an ACE Study researcher.
Data Analysis
Statistical analysis was conducted using IBM software, Statistical Package for Social Sciences (SPSS) Version 29. Continuous variables were assessed visually for normality. Descriptive statistics were used for the continuous variables and summarised as mean ± standard deviation (SD) for variables normally distributed and using median (IQR) for variables that were not normally distributed. Categorical variables were summarised using proportions. Differences in independent variables (maternal demographics and health behaviours during pregnancy, maternal anthropometry, obstetric and birth outcomes, newborn characteristics, and infant feeding) were examined and summarised by delayed secretory activation status (delayed secretory activation versus not delayed). Dichotomous independent variables were assessed unadjusted using a comparison of proportions. Continuous variables that were normally distributed were assessed using Student’s t-test, and Mann-Whitney U test was used when independent variables were continuous and not normally distributed. Any variables that demonstrated significance (p ≤ 0.10) were entered into the binary logistic regression model to quantify their association with delayed secretory activation; for other comparisons, a value of p <0.05 was considered significant. Both an unadjusted and an adjusted (for two potential confounders: maternal age and caesarean birth, identified within the literature) model were examined.
Results
Characteristics
A total of 183 participants were eligible for inclusion in this analysis. Eligible participants were recruited from JHC (n = 154), Glengarry Private Hospital (n = 5), and Armadale Hospital (n = 24). Participants were predominantly born in Australia (53%), followed by the United Kingdom (24%), South Africa (4%), and New Zealand (4%). Among this nulliparous sample, 95% of participants were pregnant for the first time, and 5% had experienced a previous pregnancy loss. The average maternal age was 29.7 years (SD = 4.6), and 44.8% were university educated. The median prepregnancy BMI was 25.3 (IQR 22.1, 29.4). The rate of caesarean birth in this cohort of nulliparous women was 48%. The median gestational age was 39.7 weeks (39.0, 41.7). There were 86 (47%) male infants and 96 (52%) female infants born.
Results showed that 134 (73%) infants were breastfed at their first feed, 20 (11%) were fed infant formula, and 24 (13%) had expressed breast milk as their first feed after birth. The median time to first feed after birth was 66 minutes (49.0, 99.8). The overall incidence of perceived delayed secretory activation (4 or more days postpartum) was 43.2%.
Associations of Factors Across Five Dimensions With Secretory Activation
The characteristics of the study population are presented by maternal self-reported secretory activation status in Table 1. A total of 43.2% of participants perceived delayed secretory activation. Maternal anthropometric and obstetric measures that were significant in bivariate analysis (p ≤ 0.10) were prepregnancy BMI 25 kg to <30 kg/m2, BMI ≥30 kg/m2, and gestational diabetes mellitus (GDM). There were no statistically significant bivariate associations between any of the other variables across the five dimensions and delayed secretory activation.
Delayed Secretory Activation Status (n = 183) Across Five Dimensions of Characteristics.
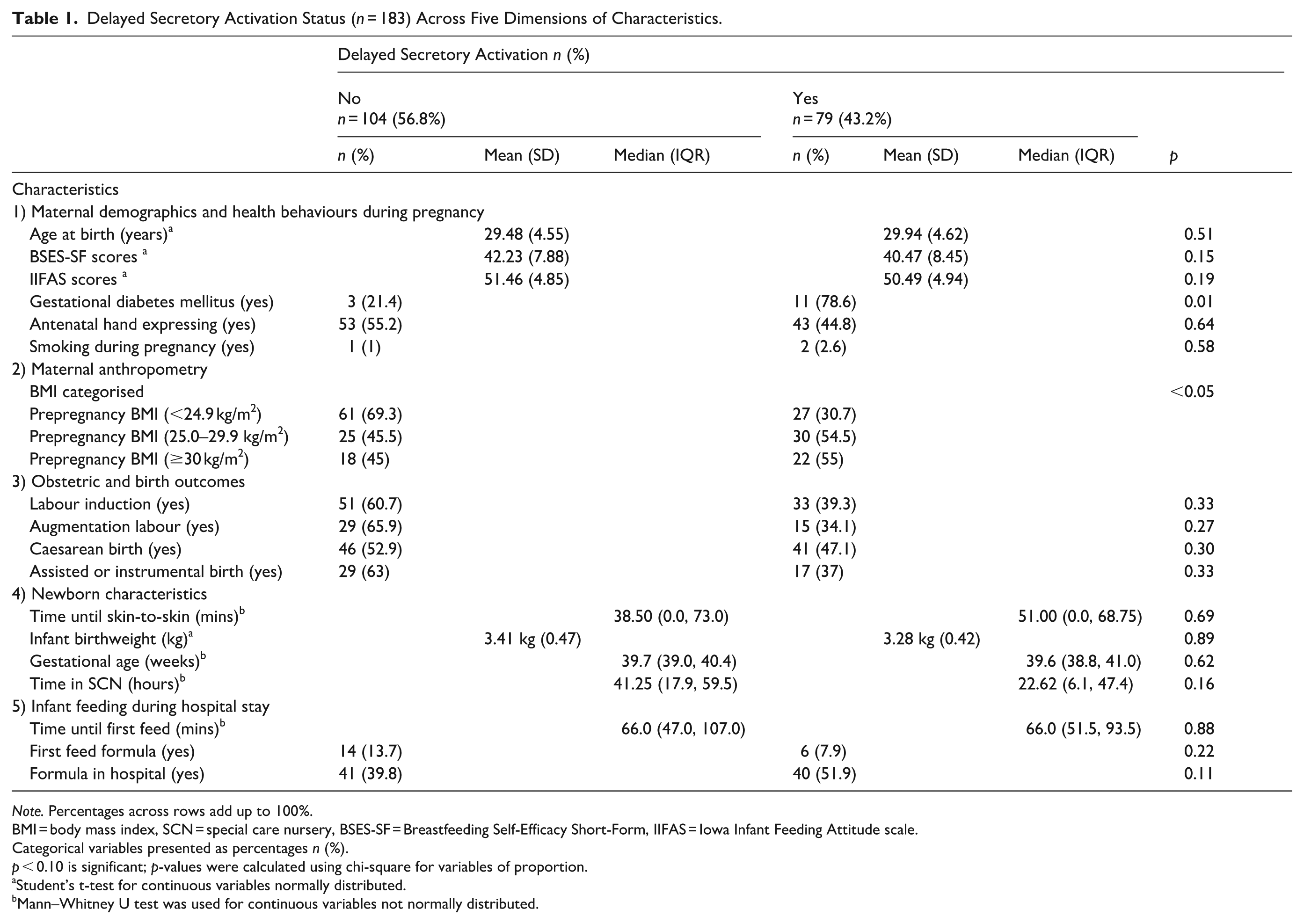
Note. Percentages across rows add up to 100%.
BMI = body mass index, SCN = special care nursery, BSES-SF = Breastfeeding Self-Efficacy Short-Form, IIFAS = Iowa Infant Feeding Attitude scale.
Categorical variables presented as percentages n (%).
p < 0.10 is significant; p-values were calculated using chi-square for variables of proportion.
Student’s t-test for continuous variables normally distributed.
Mann–Whitney U test was used for continuous variables not normally distributed.
Independent Risk Factors for Delayed Secretory Activation
Factors that were significant in bivariate analysis were eligible for inclusion in the multivariable logistic regression model. In the first model, all variables significant in bivariate analysis continued to be statistically significant (Table 2).
Predictor Coefficients for the Model Predicting Delayed Secretory Activation.
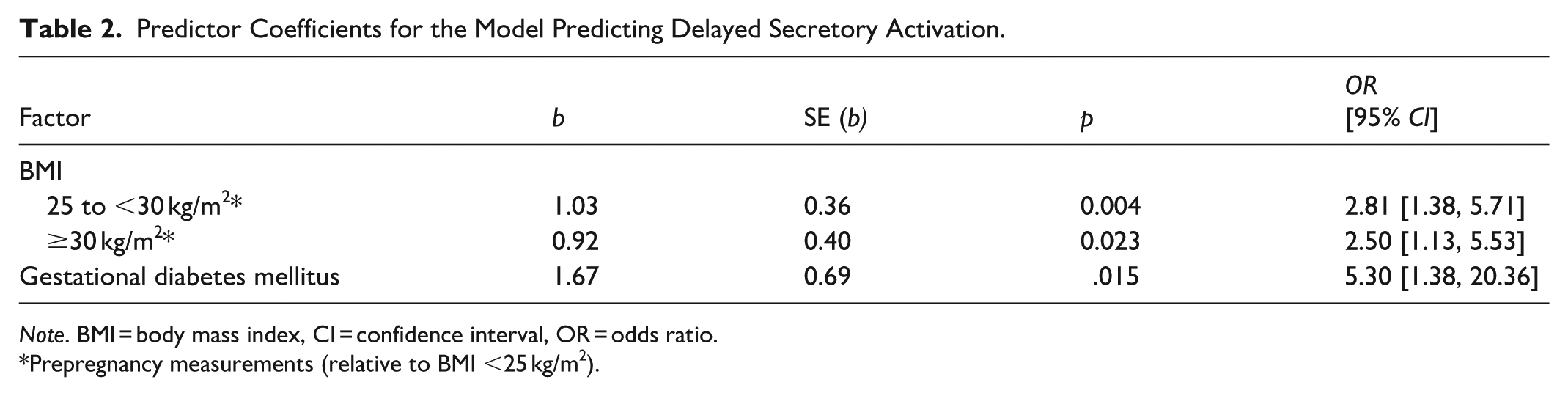
Note. BMI = body mass index, CI = confidence interval, OR = odds ratio.
Prepregnancy measurements (relative to BMI <25 kg/m2).
The second logistic regression model included adjustment for maternal age and caesarean birth (Table 3). After controlling for maternal age and caesarean birth associations GDM remained significant with an odds ratio of 5.06, 95% CI [1.28–20.07], BMI 25 to <30 kg/m2 with an odds ratio of 2.83, 95% CI [1.39–5.77], and BMI ≥30 kg/m2 with an odds ratio of 2.51, 95% CI [1.13–5.55]. The second and final logistic regression model was statistically significant, indicating that the predictors collectively improved the prediction of delayed lactogenesis compared with the intercept-only model, χ² (df = 5, N = 183) = 18.6, p = 0.002. The model explained between 9.7% (Cox & Snell R²) and 13.0% (Nagelkerke R²) of the variance in delayed lactogenesis. The Hosmer–Lemeshow goodness-of-fit test was nonsignificant, χ²(8) = 12.49, p = .131, indicating acceptable model fit. Coefficients for the final model are presented in Table 3.
Adjusted Logistic Regression of Factors Predicting Delayed Secretory Activation.
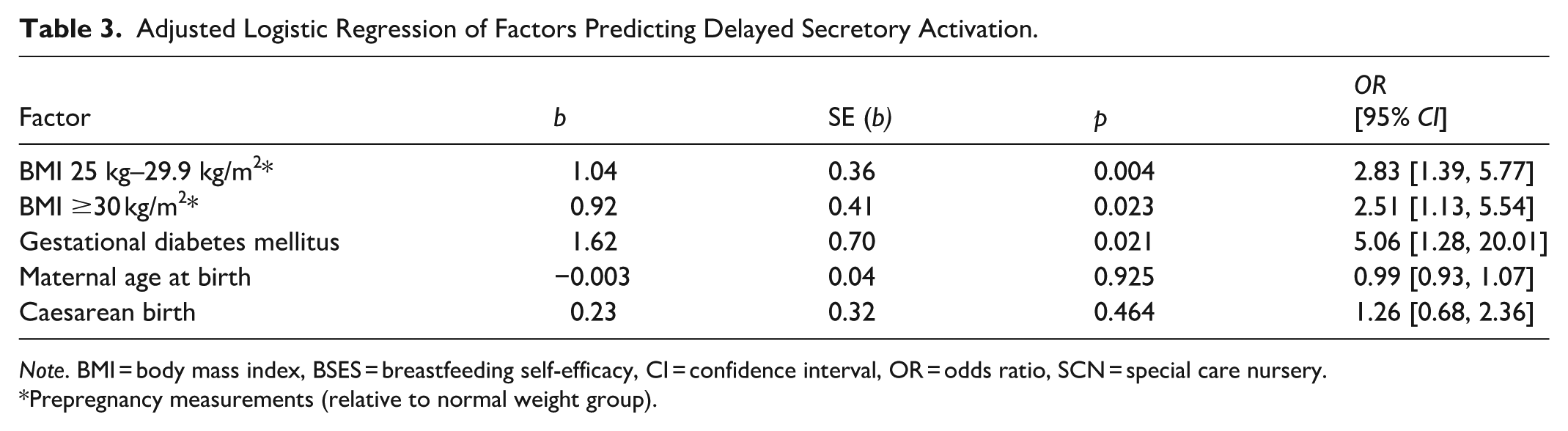
Note. BMI = body mass index, BSES = breastfeeding self-efficacy, CI = confidence interval, OR = odds ratio, SCN = special care nursery.
Prepregnancy measurements (relative to normal weight group).
Differences in Infant Feeding at Discharge
There was no association found between delayed secretory activation and exclusive breastfeeding at discharge (including exclusively feeding at the breast and/or expressed breast milk). There were 80 mothers (59.6%) without delayed secretory activation who were exclusively breastfeeding at discharge, and 58 mothers (40.4%) with delayed secretory activation who were exclusively breastfeeding at discharge.
Discussion
This secondary data analysis examined the association of factors across five dimensions with delayed secretory activation. In the final multivariable adjusted model, women with prepregnancy BMI 25 to <30 kg/m2, BMI ≥30 kg/m2, and/or GDM had an increased likelihood of delayed secretory activation after adjusting for all other factors in the model. Increased BMI was a risk factor for delayed secretory activation, regardless of GDM status. Our findings align with recent research from other populations, including a systematic review and meta-analysis of 11 studies that concluded GDM negatively affects the time to secretory activation (Wu et al., 2021).
The prevalence of delayed secretory activation in nulliparous women in WA has not been previously determined, and the rate of delayed secretory activation in this analysis was 43.2%. In other studies, rates of delayed secretory activation have significantly varied between populations, with incidence rates between 9% and 58%. In previous studies rates were typically lower among mothers in China (8.7% [Tao et al., 2017], 9.8% [Zhu et al., 2013], 17.9% [Huang et al., 2020], 18.4% [Huang et al., 2019], 36% [Yu et al., 2019]); Peru (17% [Matias et al., 2010]); and Brazil (18.8% [Rocha et al., 2020]), and higher among mothers in the United States of America (22%, 23% [Brownell et al., 2012], 23.7% [Haile et al., 2017], 35% [Chapman & Pérez-Escamilla, 1999], 44% [Nommsen-Rivers et al., 2010], and 46.4% and 57.9% [Preusting et al., 2017]). However, study design and methods varied between studies. Given the prevalence of delayed secretory activation across other populations, the rates of delayed secretory activation in this analysis are comparatively high.
To our knowledge, there has been no research in the last decade in Australia that has identified the individual risk factors associated with delayed secretory activation in nulliparous women. While there has been previous research that shows GDM is associated with delayed secretory activation, women with GDM often have higher BMI and are potentially at risk for pregnancy complications that can impact secretory activation. From previous research, we know that women with prepregnancy BMI ≥25 kg/m2 have an increased risk of GDM (Zhang et al., 2021), and the results of this analysis show that prepregnancy BMI ≥25 kg/m2 and GDM independently increase a mother’s risk of delayed secretory activation. Our findings support a targeted care approach in which women with a prepregnancy BMI ≥25 kg/m² and/or gestational diabetes mellitus are offered additional, tailored breastfeeding support, monitoring, and lactation care, especially the first week post birth.
Mothers with prepregnancy BMI ≥25 kg/m2 and/or GDM may require additional breastfeeding support to exclusively breastfeed, particularly if delayed secretory activation occurs. Research shows women with diabetes in pregnancy are at risk of delayed secretory activation (Moorhead et al., 2024). Prior to birth, antenatal colostrum expression (ACE) may be a useful intervention for at-risk groups due to the availability of colostrum for infant feeding in the setting of delayed secretory activation. Women at risk of delayed secretory activation can learn how to hand express in pregnancy; colostrum can be collected and stored in the freezer during pregnancy and taken to the hospital at the time of birth and fed to infants that require additional supplementation. Colostrum expressed in pregnancy can be used in place of infant formula to ensure infants receive their mother’s own milk during any potential period of delayed secretory activation. Expressing colostrum antenatally has proven to be safe, including specifically for low-risk mothers with diabetes (Forster et al., 2017). There is anecdotal evidence that suggests using a mother’s colostrum collected in pregnancy to supplement infants instead of using infant formula may increase a mother’s confidence in her ability to successfully breastfeed by strengthening her commitment to breastfeeding (Demirci et al., 2019). Prior to giving birth, lifestyle management with a dietitian and exercise physiologist could help mitigate some of the effects of higher BMI and GDM.
Interestingly, we found no significant association between formula feeding in hospital and delayed secretory activation. Although there were more mothers with delayed secretory activation who used infant formula in hospital (51.9%) compared to mothers without delayed secretory activation who used formula in hospital (39.8%), the difference was not significant. There are multiple reasons why first-time mothers who intend to exclusively breastfeed use formula in the hospital after birth. Factors associated with formula supplementation during the birth hospitalisation in a large cross-sectional study of 14,351 postpartum women in Brazil included maternal age ≥35, prenatal care in a private/public and private service, caesarean birth, multiple pregnancy, non-breastfeeding in the birthing room, birth in a private hospital, prematurity, and extremes of birth weight (Silva et al., 2022). In Australia, a large retrospective cohort of 24,713 mother infant dyads (Bentley et al., 2017) who intended to exclusively breastfeed found the strongest predictors of formula use in hospital included breastfeeding difficulties, including uncoordinated sucking or non/poor attachment; nipple problems (such as nipple damage, soreness, or discomfort); or undersupply, Asian-born mother, and neonatal conditions. First-time mothers should be supported without delay after birth to initiate early, frequent, and optimal breast stimulation if they intend to exclusively breastfeed. The median time to breastfeeding in this analysis was 66 minutes, whereas current WHO recommendations advise that infants should be breastfed within the first 60 minutes after birth, if mothers intend to exclusively breastfeed.
Notably, we did not find any significant association between delayed secretory activation and exclusive breastfeeding at discharge from the hospital. Although the percentage of mothers without delayed secretory activation who were exclusively breastfeeding at discharge from hospital was higher (59.6%) than mothers with delayed secretory activation (40.4%), the difference was not statistically significant. Rates of exclusive breastfeeding at discharge from hospital in this analysis are comparable to those reported in a large tertiary study in Australia of 5,836 infants (Vasilevski et al., 2021). Vasilevski et al. found that 69% of infants were exclusively breastfed at discharge from hospital, and factors that significantly reduced the odds of exclusive breastfeeding at discharge were maternal country of birth, birth type (caesarean or assisted vaginal birth), first-feed type (formula feeding or mixed feeding), and admission to SCN.
Limitations
There were several limitations in this analysis. Data used in this analysis were obtained from a subset of participants enrolled in the ACE Study, and although a sample size calculation was used for the ACE Study, we did not do a specific sample size calculation for this secondary analysis and may have been unable to detect small associations. The ACE Study is a study that focuses on mothers who can read and speak English and does not include non-English-speaking mothers, limiting generalisability.
Maternal prepregnancy weight and height measurements used to calculate prepregnancy BMI were obtained retrospectively in the ACE Study. Weight and height measurements were either self-reported by women in pregnancy between 28–32 weeks’ gestation or obtained from medical records from the first antenatal appointment. There may have been some recall bias in these women reporting prepregnancy weight; however, it was anticipated that most women would be able to reliably report their body weight prior to pregnancy. Previous research has shown that the majority of self-reported prepregnancy weight results in the same BMI classification when compared to measured weight at the first prenatal appointment (Holland et al., 2013). There may have been some measurement bias when using prepregnancy weight and height measurements from medical records to calculate BMI; therefore, maternal self-reported measurements were prioritised in this analysis.
The ACE Study does not collect data on race and ethnicity, so it could not be adjusted for in this analysis. Race and ethnicity have been reported as potential confounders of delayed secretory activation in other research.
Conclusion
The rate of delayed secretory activation was 43.2%. Women who were classified as overweight or obese prior to pregnancy and women with GDM were significantly more likely to experience delayed secretory activation after adjusting for maternal age and caesarean birth. Although other research suggests that mothers who experience delayed secretory activation are at risk of nonexclusive breastfeeding in hospital, we found no association with delayed secretory activation and formula feeding in hospital or exclusive breastfeeding at discharge.
Footnotes
Acknowledgements
The authors would like to thank all women who participated in the ACE Study for their commitment to helping future mothers with breastfeeding by sharing their own infant feeding experiences. The authors would also like to thank ACE Study research assistant Narelle Little and our wonderful research midwives across the different hospitals for their support with recruitment.
Consent for Publication
Not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The ACE Study has received funding from the Department of Health Western Australia and The Stan Perron Charitable Foundation. The first author, CC, is the recipient of a Research Excellence Fellowship Scholarship at Edith Cowan University (ECU) where she is a PhD candidate. The salary of JRL is supported by a National Heart Foundation Future Leader Fellowship (ID: 102817). The funders of the ACE Study had no role in the study design, data collection, analysis, interpretation of the findings, or writing the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analysed during the current analysis are not publicly available, although other investigators may request access to the dataset if a formal request describing their plans is approved by the principal investigators and the relevant ethics approval is in place.
