Abstract
Background:
A disrupted gut microbiome during an infant’s first 1000 days of life can lead to long-lasting negative effects on child health. Cesarean delivery and formula feeding are two factors that can detrimentally impact infant microbiome development as well as maternal mental health. Donor human milk may be a superior supplementation alternative to formula.
Research Aim:
To examine donor human milk supplementation compared to formula supplementation in full-term infants born via Cesarean section and the impact on the infant gut microbiome, infant health outcomes, breastfeeding outcomes, and maternal mental health.
Methods and Planned Analyses:
We are conducting a pilot clinical randomized controlled trial, comparing donor human milk to formula supplementation for 187 full-term infants born via Cesarean section who are breastfeeding and require supplementation in the first postnatal week of life. Infant stool samples, breastfeeding outcomes, maternal mental health, and child health outcomes will be measured at 1-week, 3-, 6-, and 12-months postpartum. Additionally, child health and maternal mental health are being assessed at 18- and 36-months postpartum.
Discussion:
This study will generate essential data on the association between supplementation types and the full-term infant microbiome, breastfeeding exclusivity and duration, and infant health. It will also provide preliminary data to inform a multi-site, longitudinal mixed-methods randomized controlled trial that will assess longer term child health outcomes. This evidence may be used to inform guidelines and policies that will increase accessibility to and raise awareness of donor human milk as a supplementation option in this population.
Keywords
Background
The crucial period spanning from conception to 2 years of age, known as the first 1000 days, plays a pivotal role in shaping an infant’s immune, neurological, and hormonal systems, as well as their sleep patterns (Matenchuk et al., 2020), growth, and overall development (Robertson et al., 2019). During this period, the gut microbiome, comprised of bacteria, fungi, protozoa, and viruses, along with their metabolites, is highly influential to infant development (Tamburini et al., 2016). Commensal microbes in the gut help with the digestion of nutrients and produce important neurotransmitters such as Gamma-Aminobutyric Acid (GABA), serotonin, and dopamine (Guamán et al., 2024). Further, bioactive metabolites produced by microbes in the gut prevent the colonization and invasion of pathogenic microbes by producing mucin and upregulating the expression of tight junction proteins (Guamán et al., 2024).
A disrupted, or dysbiotic, gut microbiome during these early stages can lead to long-lasting negative effects on a child’s health. Ideally, a healthy infant gut microbiome is established through vertical transmission of microbes during vaginal birth, followed by exclusive breastfeeding (EBF) and no antibiotic treatment (Robertson et al., 2019). The typical development of a healthy infant gut microbiome involves a progression from low diversity dominated by Bifidobacteria in the early months to increased diversity as the infant encounters different foods and environments (Arrieta et al., 2014; Korpela & de Vos, 2022). However, many infants deviate from this norm due to factors such as antibiotic exposure, Cesarean delivery (c-section), and formula feeding, which can detrimentally affect their microbiome development (Persaud et al., 2015).
Globally, the c-section delivery rate is more than 20%, exceeding the World Health Organization (WHO) recommendation of 10%–15%; with the highest rates being in Europe (25.7%), North America (31.6%), and Latin America (42.8%; WHO, 2021). While c-sections are critically important interventions that save lives, they have many long-term sequelae that can adversely impact infant, child, and maternal health outcomes (Słabuszewska-Jóźwiak et al., 2020). Infants who are born via c-section are at an increased risk of developing non-communicable diseases (NCDs) such as allergy, asthma, and obesity (Keag et al., 2018; Słabuszewska-Jóźwiak et al., 2020), and altered neurodevelopment (Zhang et al., 2019).
In addition, infants born via c-section are significantly more likely to be supplemented with formula and experience earlier cessation of breastfeeding compared to infants who are born vaginally (Hobbs et al., 2016). This disruption of breastfeeding can negatively affect both mothers and infants. For example, disruption of breastfeeding in the first 6 months of life can increase the risk of developing NCDs in childhood and adulthood (Victora et al., 2016), while exclusive breastfeeding during this time may have a protective effect against developing NCDs after c-section delivery (Y. Liu et al., 2023). Additionally, many mothers who are required to supplement their infant with formula often experience negative feelings, including failure, guilt, and fear of being stigmatized (Fallon et al., 2017; Jackson et al., 2021). These negative experiences with breastfeeding and supplementation are associated with poor maternal mental health, particularly postpartum depression (Butler et al., 2021).
Key Messages
Infants born via Cesarean section are at increased risk of developing non-communicable diseases, likely due to early life disruptions to their developing microbiome.
Donor human milk from accredited milk banks is increasingly available for full-term infants; however, there is a lack of evidence about how donor human milk compared to formula can impact infant health, breastfeeding outcomes, and maternal mental health.
Using a clinical, randomized controlled trial, this study examines donor human milk compared to formula supplementation and exclusive breastfeeding in infants who are born via Cesarean section.
We examine impacts on the developing infant microbiome, longer-term breastfeeding outcomes, maternal mental health, and child health outcomes.
Infant Microbiome
Infants who are born via c-section have markedly different microbiome signatures compared to infants who are born vaginally. Infants who are born vaginally have gut microbiomes that tend to be colonized with Bacteroides, Bifidobacteria, and Lactobacillus in the first days of life, whereas infants born via c-section tend to be colonized with Staphylococcus, Streptococcus, Klebsiella and Clostridium (Y. Liu et al., 2023), which are bacterial species reflective of the skin microbiome and those found in the hospital environment (Coelho et al., 2021). Further, infants who are born via c-section are at increased risk for developing adverse health outcomes such as respiratory tract infections, asthma, and obesity (Słabuszewska-Jóźwiak et al., 2020). It is well supported that NCDs are linked to disruptions in the developing infant microbiome (Ronan et al., 2021). While these differences gradually diminish over the 1st year of life, they are still present during important stages of early infant development (Tamburini et al., 2016).
Gut microbiome signatures are mediated by infant feeding type (Zimmermann & Curtis, 2020), and exclusive breastfeeding improves gut microbiota composition in infants who experience adverse early-life exposures, such as c-sections (Fok et al., 2019; Korpela & de Vos, 2022; Y. Liu et al., 2023; Princisval et al., 2021). The bioactive components in human milk contribute to homeostasis of the infant gut microbiome through pre-, pro-, and post-biotic properties, and may help to mitigate microbial dysbiosis caused by adverse early life exposures (Thai & Gregory, 2020). However, not all infants are able to receive their mother’s own milk (MOM) exclusively, and are instead supplemented with infant formula. Infants who are born via c-section are significantly more likely to be supplemented with formula compared to vaginally born infants, with some studies indicating supplementation rates as high as 50% (Hobbs et al., 2016; L. Y. Liu et al., 2024). These higher supplementation rates in c-section infants are likely due to maternal exhaustion, lower perceived milk supply, and other breastfeeding difficulties (Singh et al., 2023). Even one bottle of formula supplementation in the early postpartum period is associated with lower relative abundance of Bifidobacteriaceae in the gut microbiome at 3 months postpartum, despite exclusive breastfeeding occurring after hospital discharge (Forbes et al., 2018). Compared to infants who are exclusively fed MOM, infants who are supplemented with or exclusively fed infant formula demonstrate increased gut microbial diversity and decreased relative abundance in Bacteroides and Eubacterium; qualities which are reflective of microbial signatures found in adults, and which may adversely impact infant development (Ho et al., 2018).
Donor Human Milk
Donor human milk (DHM) from an accredited milk bank is an alternative to formula when supplementation is required. In North America, DHM is provided to hospitalized infants through milk banks regulated by the Human Milk Banking Association of North America (HMBANA). Certain milk banks within the HMBANA’s organization can provide DHM to families of non-hospitalized infants. This includes families in the postpartum and community settings who have given birth to full-term infants. While DHM is prioritized for ill and preterm infants in the Neonatal Intensive Care Unit (NICU), there is increasing supply of DHM which can be provided to full-term infants who may be at increased risk of poor health outcomes due to adverse early life exposures, such as c-section birth, antibiotic exposure, or other vulnerabilities.
DHM and the infant microbiome
From a microbiome perspective, DHM may be a superior alternative to formula when supplementation is required, allowing infants who are supplemented with DHM instead of formula to continue to be exclusively fed with human milk (Ames et al., 2023). Through its various biotic properties, DHM minimizes perturbations to the infant gut microbiome and may even help to repair the gut microbiome in infants who experience these adverse early-life exposures. As such, there are likely profound differences in the impact of formula supplementation compared to DHM supplementation on the developing full-term infant gut microbiome (Peila et al., 2016; Yadav et al., 2022).
DHM and maternal outcomes
Mothers who deliver via c-section are significantly more likely to have disrupted breastfeeding experiences, delayed lactogenesis, and be required to supplement their infants with formula (Hobbs et al., 2016). In addition to disrupting the infant’s microbiome, many mothers experience negative maternal mental health (guilt, shame, postpartum depression) when they are required to supplement their infant with formula, especially when they intend to exclusively breastfeed (Jackson et al., 2021, ). Perinatal mood disorders are associated with unmet breastfeeding goals (Butler et al., 2021) and formula supplementation (Pope & Mazmanian, 2016). While it is unclear if the association between maternal mental health and formula supplementation is bidirectional, evidence indicates that reassuring mothers that the human milk substitute their infant is receiving is the safest and best possible option ameliorates their negative emotions (Smyth & Hyde, 2020).
Providing DHM instead of formula when supplementation is required may alleviate negative maternal feelings around supplementation and could enhance maternal mental health. Many mothers report improved well-being when being offered DHM instead of formula as a supplementation option (Brown et al., 2024). However, concerns remain among healthcare workers that families may equate DHM to MOM, creating a risk that DHM may displace mothers’ own milk and breastfeeding (Israel-Ballard et al., 2019). Examining breastfeeding outcomes (exclusivity and duration) in conjunction with maternal mental health outcomes is vital to ensure that families continue to breastfeed and do not displace mother’s own milk or breastfeeding with DHM. As such, it is important to empirically examine maternal mental health and breastfeeding outcomes when using DHM as a supplementation option.
This paper describes the protocol for a pilot clinical randomized controlled trial (RCT) to examine the impact of DHM compared to formula supplementation on the developing infant gut microbiome, infant health outcomes, breastfeeding outcomes, and maternal mental health of full-term mother–infant dyads who are born via c-section.
We aim to address the following research questions:
Primary research question: What are the impacts of replacing formula supplementation with DHM supplementation on the infant’s gut microbiome development over the 1st year of life for full-term infants who are born via c-section?
Secondary research questions include: What are the impacts of replacing formula supplementation with DHM supplementation on:
Infant health outcomes up to 36-months of age (growth, sleep, allergy, infection, neurodevelopment)?
Breastfeeding self-efficacy, exclusive breastfeeding up to 6-months postpartum, and breastfeeding duration?
Maternal mental health (depression, anxiety, and anger) across the first year postpartum?
Methods
Research Design
This study is a pilot, single-site, parallel group, clinical RCT to examine DHM compared to formula supplementation in full-term infants who are born via c-section and whose mothers are planning on breastfeeding and require supplementation in the first week of life (see the Appendix: Figure 1 in the online supplemental material). The RCT design will address the significant knowledge gap of how exclusive human milk feeding (via DHM supplementation) compared to formula supplementation can impact the developing infant microbiome using experimental methods. Randomization in this study will reduce potential confounding and will develop evidence that significantly improves our understanding of potential causal links between human milk feeding and the infant gut microbiome. This study was approved by the Conjoint Health Research Ethics Board at the University of Calgary on February 12, 2025 (Ethics ID: REB23-1356). The RCT has been registered at ClinicalTrials.gov: NCT05815433. The SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) checklist was adhered to.
Setting and Relevant Context
We are recruiting mothers from the postpartum and labor and delivery units at a tertiary care center in a large urban area in Western Canada. The hospital delivers approximately 16,000 infants per year, with a current c-section rate of greater than 30% and it is not Baby-Friendly Hospital Initiative (BFHI) designated. Breastfeeding initiation rates exceed 90% in the province of study, with 70% of infants continuing to breastfeed at 6 months (40.6% exclusively; Public Health Agency of Canada, 2022).
Nurses who work on the postpartum unit at the research hospital receive a 2-hour breastfeeding class and an 8-hour buddy shift with an International Board-Certified Lactation Consultant (IBCLC) as part of their orientation. Additionally, newly hired nurses are required to complete two modules on Breastfeeding Foundations and Breastfeeding Complications and Supplementation. In addition, there is an IBCLC available during the day shift for any patient in the postpartum unit who is experiencing lactation issues. All parents are provided with a hospital-grade pump if additional pumping is indicated while they are in the hospital. After discharge, families are followed by a public health nurse and have access to lactation and registered nurse (RN) support via an early-start hotline and public health clinics for 6 weeks postpartum.
Sample
The population of interest is primiparous individuals of full-term infants (≥ 37 weeks gestation) who deliver via c-section (both scheduled and emergency) and who are planning on breastfeeding. Inclusion criteria include: Scheduled for or has delivered via c-section, over 18 years of age, planning on breastfeeding, baby is full-term (born ≥ 37 weeks gestational age), singleton birth, can read and write in English, and consents to having their infant receive DHM if allocated. Exclusion criteria include: Group B Streptococcus (GBS) antibiotic prophylaxis, infant born with a congenital anomaly that may hinder breastfeeding, planning on exclusively formula feeding, multiparity, multiples (twins, triplets), NICU admission, and formula supplementation prior to enrollment.
Individuals ≥ 37 weeks gestation admitted to the labor and delivery or postpartum unit who have a scheduled c-section or who have delivered via emergency c-section are screened by the clinical staff for eligibility according to the inclusion and exclusion criteria. Sample size considerations are based on the primary outcome of microbiome difference (β-diversity) between groups. We aim to determine realistic distance matrices between groups so that appropriate sample size calculations can be conducted for a full-scale study. Based on similar studies comparing β-diversity between vaginal and c-section born infants (Madan et al., 2016), we will recruit 187 mother–infant dyads to retain 175 participants. This will result in approximately 52 dyads in the DHM group, 52 dyads in the control group, and 75 dyads in the EBF (observation) group (primary outcome; Ferdous et al., 2022; Madan et al., 2016), providing 80% power to detect overall gut microbiome differences between groups (β-diversity), assuming a 25% dropout rate (preliminary data from a similar study) and a 60% supplementation rate.
Data Collection and Monitoring
Eligible individuals will be asked by clinical staff for permission to be approached by research staff. Formal consent of a participant, using the REB-approved consent form, must be provided, and the mother–infant dyad must be enrolled before supplementation of direct breastfeeding begins.
Following parental consent, researchers have access to both the mother and the infant’s medical information via medical charting systems. This ensures that the research team will have accurate medical information upon enrollment, throughout the study, and during follow-up.
Data collection and recruitment for this study started in October 2024 and are currently underway. We expect recruitment to be completed by January 2026, with data collection ending in July 2026. Table 1 describes the data collection timelines over the course of the study.
Proposed Study Schedule as per Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT).
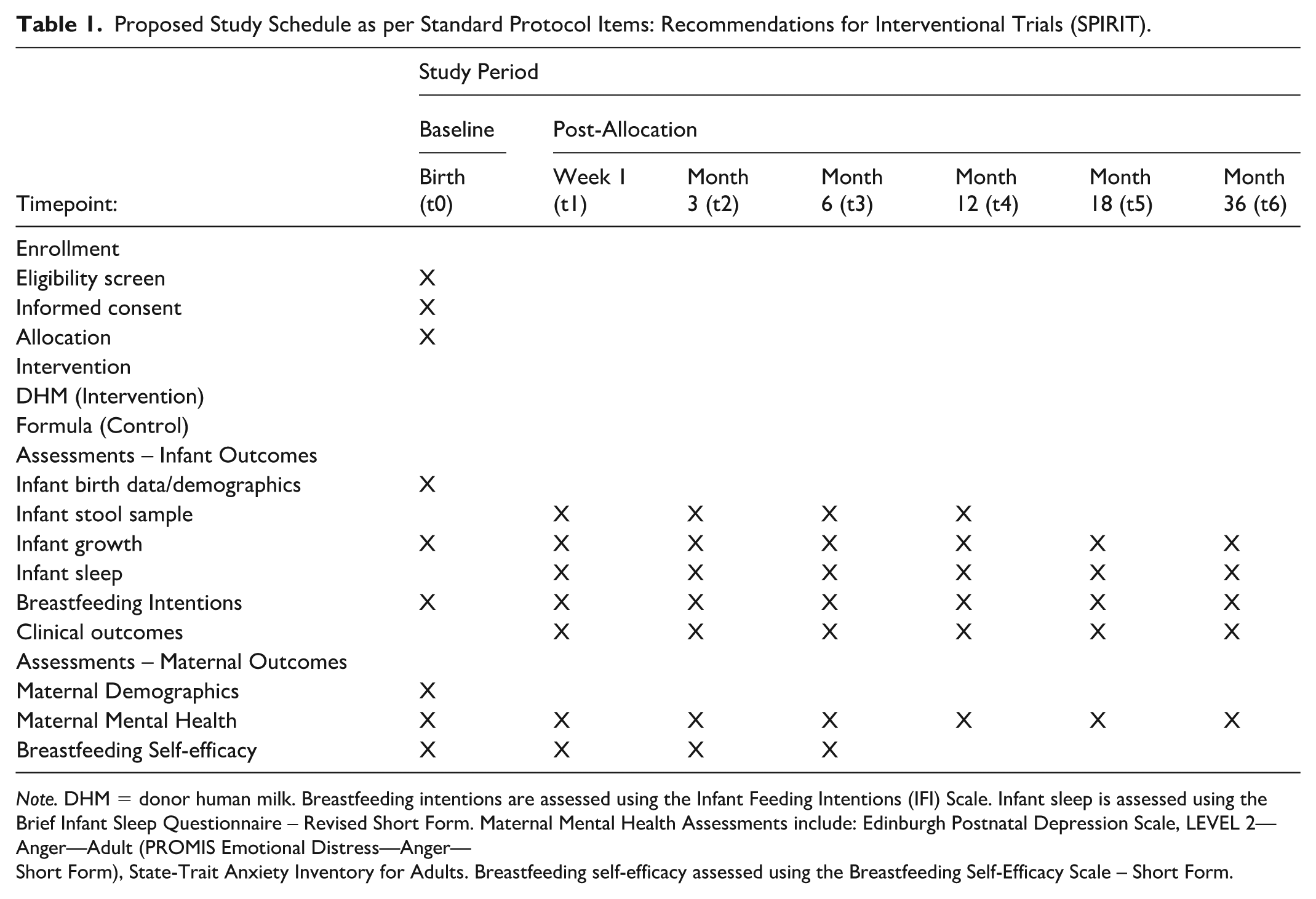
Note. DHM = donor human milk. Breastfeeding intentions are assessed using the Infant Feeding Intentions (IFI) Scale. Infant sleep is assessed using the Brief Infant Sleep Questionnaire – Revised Short Form. Maternal Mental Health Assessments include: Edinburgh Postnatal Depression Scale, LEVEL 2—Anger—Adult (PROMIS Emotional Distress—Anger—
Short Form), State-Trait Anxiety Inventory for Adults. Breastfeeding self-efficacy assessed using the Breastfeeding Self-Efficacy Scale – Short Form.
Baseline assessment
Upon enrollment to the study, research assistants collect infant birth metrics (weight, length, head circumference), APGAR scores, and gestational age via health records. An Infant Feeding Intentions Scale (Nommsen-Rivers & Dewey, 2009), demographics, ethnicity, and substance use are also collected via an electronic baseline survey. Maternal depressive symptoms (Edinburgh Postnatal Depression Screen (Cox et al., 1987)), anxiety (State-Trait Anxiety Inventory; Spielberger et al., 1970)), anger (PROMIS Emotional Distress-Anger – Short Form; Tarescavage et al., 2021) and breastfeeding self-efficacy (Breastfeeding Self-Efficacy Scale – Short Form; Dennis, 2003) are also measured as baseline to control for potential confounding of breastfeeding outcomes (Brockway et al., 2017; Henshaw, 2023). These are completed by maternal participants using online surveys via Research Electronic Data Capture (REDCap).
Study visits
Recent evidence suggests that exclusive breastfeeding is particularly effective at reversing many of the Caesarean-associated microbiota patterns in infants whose mothers are secretors (who produce the human milk oligosaccharide 2’-fucosyllactose in their bodily fluids, including saliva and human milk; Korpela et al., 2018). Infants born via c-section who consume human milk from secretor mothers have a gut microbiome profile more similar to vaginally-delivered infants in comparison to human milk-fed c-section infants born to non-secretors (Korpela et al., 2018). To control for this, upon enrollment, a research assistant collects 3-5 ml of saliva from each maternal participant using the passive drool technique. To determine the secretor status of participant mothers, the Wiener agglutination test is used (Rai et al., 2015).
In-person assessment
Research nurses visit families in their home between Days 6 and 8 postpartum, to weigh and measure the infant, and collect the hard copy 7-day infant feeding journal, which records the intensity and mode (direct or expressed) of breastfeeding, as well as other infant nutrition sources (see the online Supplementary File). The stool sample, which parents have been instructed to store at room temperature, will also be collected by the research nurse. Families are invited to attend a centrally located milk bank for the remaining follow-up visits (at 3-, 6-, 12-, 18-, and 36-months) where a research nurse weighs and measures their infant and collects the most recent stool sample (at 3-, 6-, and 12-months). If families are unable to attend the milk bank, provisions will be made for a research nurse to visit them in their home to reduce barriers to access to participate in the research.
Online surveys
Online surveys are provided to parents via REDCap at the Week 1, 3-, 12-, 18-, and 36-month timepoints. The specific surveys administered are discussed in the Outcomes/Measurement section. Families are reminded three times over a period of 7 days following the survey invitation to complete the survey. The ASQ3 survey is closed 30 days after the infant turns 6 months, as it is only valid until the infant is 6 months 30 days (Squires et al., 2009). All families will receive a $25 gift card for completing the 3-, 12-, 18-, and 36-month visits and surveys.
Data management
To ensure confidentiality, a unique participant ID is assigned to each participant and is used rather than identifiable information throughout the study. Participant information is anonymized and encrypted, and research data is stored and retained on secure servers according to institutional policies. All data and de-identified biospecimens will be retained for 5 years after study completion. Participants may consent to the use of this data for future research, which is required to undergo review by a Research Ethics Board. The final trial data set will only be available to the principal investigator, co-investigators, and research staff listed on the ethics application.
Data monitoring
There is no data monitoring committee/board.
Interventions
After enrollment in the study by a trained research assistant, participants and their infants are block randomized (four/block) with an equal number assigned to the control (formula) or intervention (DHM) group (1:1) using an independently developed randomization sequence through electronic assignment using REDCap. This will ensure a balanced allocation of participants across the groups.
Intervention group: Donor human milk
Families of infants randomized to the intervention group are provided with DHM for their baby for the first 7 days of life. Upon discharge from the hospital, families in the intervention group are sent home with three 120 ml bottles of DHM and are provided with additional DHM if needed for the 1st week of the baby’s life, at no cost to the family. If more DHM is required during the intervention timeframe, additional DHM is couriered to families. The exposure time of 7 days was selected due to the high cost of DHM and this being the period when breastfeeding is being established and most formula supplementation occurs (Kent et al., 2012). Currently, evidence is lacking as to the critical period when the microbiome is established. As evidence builds, and this study is scaled to a fully powered trial, different exposure periods will be assessed to determine the ideal period of human milk exposure to facilitate microbiome development. Additionally, there is no existing literature or guidelines for appropriate amounts of DHM supplementation for full-term infants in the 1st week of life. As such, existing guidelines for supplementing with formula provided by the local health region were used to guide how much DHM to send home with parents. Findings from the current study will allow researchers to better understand appropriate supplementation amounts for DHM in full-term infants. DHM for this study will be obtained from a HMBANA accredited community milk bank in Western Canada. All DHM for the research study is stored in a dedicated research freezer in the hospital nursery, which has continuous electronic monitoring to ensure adequate temperature and safe storage conditions.
Control group: Formula (standard care)
Infants randomized to the control group receive formula when supplementation is required. Formula consumed by infants on the postpartum unit is provided by the hospital at no cost to families. Formula required for supplementation after discharge from the postpartum unit is purchased by families, as this is the standard care practice. Researchers do not provide formula to participants as this presents a conflict of interest and violates the World Health Organization (WHO) International Code of Marketing of Breast-Milk Substitutes (Breastfeeding Committee for Canada, 2017). If parents indicate that they are supplementing with formula, the brand and type of formula will be recorded at each infant feeding assessment.
Infants who are recruited and randomized, but do not require supplementation, will be retained in the study as an observational group of EBF infants to serve as a gold-standard comparison. Blinding of participants is not feasible since we are assessing the impact of DHM supplementation compared to formula supplementation on maternal mental health and breastfeeding self-efficacy.
The 7-day infant feeding journals will be cross-checked to confirm that infants are receiving their allocated supplementation during the intervention timeframe to ensure intervention fidelity. If intervention contamination occurs (i.e., infants randomized to the DHM group receive formula, or vice versa), infants will be retained in the study but recorded as a protocol deviation. These participants will be included in a secondary, as-treated analysis to assess the impacts of supplementation on the microbiome. Supplementation after the intervention timeframe will be recorded via parent recall at the 12-week and 6-, 12-, and 18-month visits.
Staff on the postpartum unit have been provided with basic training about the research study by the study investigator, who is a registered nurse (RN) and IBCLC, including guidelines around provision of DHM as outlined by the local health authority, and safe thawing, preparation, and storage as outlined by HMBANA. Additionally, parents in the intervention group are sent home with instructions following HMBANA’s guidelines about the safe handling, storage, and preparation of DHM. Families can contact the study investigator via email or telephone if they have any questions or require additional DHM during the 7-day intervention time frame.
All protocol modifications are reported to the Conjoint Health Research Ethics Board (CHREB), co-investigators, trial participants, and trial registries. All participants will understand that taking part in the research is voluntary. Only infants who require supplementation (e.g., small for gestational age, hypoglycemia, poor latch, parental choice) will receive DHM or formula; this will never displace mother’s own milk. Randomization to DHM or formula is an ethical practice in this study as DHM is not readily available as an option for full-term infants and is not provided to families in the hospital setting. For adverse events, follow-up occurs within 7–15 calendar days of the PI becoming aware of them. All other events, such as protocol deviations or violations, are addressed within 7 calendar days to mitigate future deviations.
Outcomes/Measurement
Primary outcome measure
Infant gut microbiome up to 12 months of age
Infant stool samples are collected by families at 1-week and 3-, 6-, and 12-months postpartum, using shelf-stable DNA Genotek OMNIgene×GUT OM-200 stool collection kits for gut microbiome profiling. Samples are then transported to the International Microbiome Centre at the University of Calgary for analyzing. A 500 µl sample of each fecal sample is aliquoted into Qiagen PowerBead tubes and stored at −80 ºC until subsequent DNA extraction. Microbial DNA will be extracted using Qiagen DNeasy 96 PowerSoil Pro QIAcube HT Kit following the manufacturer’s protocol. A total of 75 µl of ZymoBIOMICS microbial community standard (Zymo Research) and lysis buffer are included as extraction positive and negative controls, respectively. DNA concentrations are measured using the Quant-iT™ PicoGreen™ dsDNA Assay Kit (Life Technologies).
Secondary outcome measures
Infant health and development up to 36 months of age
Infant health and development is assessed at 1-week and 3-, 6-, 12-, 18-, and 36-months. Research nurses weigh and measure (length and head circumference) infants and report growth data using standardized body mass index (BMI) z-scores (Villar et al., 2014). Maternal participants are asked to complete an online survey via REDCap to measure infant sleep duration using the Brief Infant Sleep Questionnaire (Sadeh, 2004; see the online Supplementary Materials). Infant health outcomes, including diagnoses of atopy, allergy, and asthma, are determined based on parental report of physician diagnosis and verified via electronic chart extraction (Netcare). Child neurodevelopment is assessed using the Ages & Stages Questionnaire®, Third Edition (Squires et al., 2009; Supplementary Materials), which is administered to maternal participants via REDCap at 6-, 18- and 36-months.
Breastfeeding outcomes
All infant feeds during the 7-day intervention window (1st week of life) are recorded by maternal participants in an infant feeding journal to capture the intensity and mode (direct or expressed) of breastfeeding as well as the type and amount of other infant nutrition sources (Supplementary Materials). A 7-day recall is used by maternal participants to assess infant feeding at all subsequent assessments up to 36 months of age, using the WHO infant feeding categorization (WHO, 2008). Maternal breastfeeding self-efficacy is also assessed at 1-week and 3-months postpartum using the Breastfeeding Self-Efficacy Scale – Short Form administered (Dennis, 2003; Supplementary Materials) via REDCap.
Maternal mental health
Maternal depressive symptoms (Edinburgh Postnatal Depression Scale; Cox et al., 1987; Supplementary Materials), anxiety (State-Trait Anxiety Inventory; Spielberger et al., 1970; Supplementary Materials), anger (PROMIS Emotional Distress–Anger – Short Form; Supplementary Materials; Tarescavage et al., 2021) are assessed at each follow-up visit via an online REDCap survey. Postpartum resources will be provided to all participants who score above 10 on the Edinburgh Postnatal Depression Scale.
Table 2 outlines the psychosocial outcome measures as described above.
Psychosocial Outcome Measures.

Planned Data Analysis
Analysis of primary outcome
Microbiome analysis
Data will be analyzed at each time point to explore effect changes over time. We will first compare differences in microbiome signatures at each time point (De Filippis et al., 2021) between EBF infants to those who were supplemented with DHM or formula (formula vs. DHM vs. EBF) during the 1-week intervention period. Secondarily, we will analyze microbiome signatures by infant feeding type at each time point (EBF, breastfeeding and formula, or exclusively formula fed as it is unlikely any babies will be receiving DHM at 3 or 6 months). We will compare differences in microbiome signatures (De Filippis et al., 2021), such as α- and β-diversity, between the DHM and formula groups. For assessing changes in β-diversity statistically, permutational multivariate analysis of variance (PERMANOVA) will be performed on Bray-Curtis dissimilarity indexes and beta-diversity will be visualized using principal coordinate analysis. Alpha-diversity metrics (Species Richness, Shannon index) will be analyzed using linear mixed-effects models and differentially-abundant taxa and functional pathways (determined from bacterial genomes) will be identified using repeated measures Compound Poisson Linear Models in MaAsLin2 (Mallick et al., 2021), while correcting for multiple comparisons using a false discovery rate (p < .05). Infant sex will be tested as an effect modifier in all models to determine whether microbial responses to DHM/formula supplementation are sex dependent.
Shotgun library preparation
Shotgun libraries will be prepared with 50 ng of DNA for each sample as input and barcoded with combinatorial dual indexes using Twist 96-Plex Library Preparation kit (Twist Bioscience). 50 ng of ZymoBIOMICS microbial community DNA standard (Zymo Research) and nuclease free water will be included as library positive and negative controls respectively. Agilent bioanalyzer system will be used to determine the average fragment size and concentration to estimate successful library preparation. Sequencing of libraries will be performed on Illumina Novaseq X platform, ~ 10 Million reads/sample, paired-end 150 base pairs.
Bioinformatics analysis of shotgun metagenomics reads
Fastqc will be used to check the quality of shotgun reads. Adaptor removal and quality filtering will be performed using cutAdapt, and BMTagger will be used for host contamination removal. Quality controlled reads will then be passed on to the MetaPhlAn3 pipeline for taxonomy assignment. For functional profiling, HUMAnN3 will be used to determine gene family and metabolic pathway abundances (i.e., MetaCyc, KEGG, eggnong). Strain-level profiling of metagenomes may also be performed using StrainPhlAn3 (Bell et al., 2013; De Filippis et al., 2021; Mallick et al., 2021; Martin, 2011).
Analysis of secondary outcomes
To determine if group differences exist, secondary outcomes (Table 2) will be compared between intervention and control groups using linear mixed effects models. Group allocation will be treated as the between-subject factor, and each outcome will be treated as a within-subject factor. Baseline variables will be treated as covariates. Additional variables will be included as fixed effects in the linear mixed model. The frequency of dropouts will be compared between the intervention and control groups. Attrition bias will be assessed by testing whether those who dropout or who are lost to follow-up have traits that distinguish them from those who remain in the study.
Discussion
This study will examine the role of exclusive human milk feeding (via breastfeeding and DHM supplementation) as a mitigating factor on infant and child health when infants are born via c-section. While the evidence demonstrating the impact of human milk on child health outcomes is compelling, it is often critiqued for issues related to confounding, its observational nature, the high risk of selection bias, and poor assessment of breastfeeding and human milk intake (Smyth & Hyde, 2020). Due to the experimental design and randomization of participants, this study will be able to draw direct causal linkages between breastfeeding and child health outcomes.
Potential Limitations of the Work
Potential limitations for this study include the inability to blind, intervention compliance, and risk of attrition. In this study, blinding of participants is not feasible since we are assessing the impact of DHM supplementation compared to formula supplementation on maternal mental health and breastfeeding self-efficacy. Intervention compliance also presents limitations due to various factors. Caregiver supplementation feeding preferences may change or they may not be agreeable to the allocation group to which they were randomized, which may be attributed to personal preference, culture or faith, or support systems. Intervention compliance may also be impacted due to healthcare providers’ decision-making or recommendations. Risk of attrition is an additional limiting factor.
Another limitation of this study is the lack of baseline microbiome assessment at birth. While one of the biggest determinants of initial colonization of the gut in newborns is mode of delivery and exposure to intrapartum antibiotics (Turunen et al., 2024), baseline assessment of meconium microbiome would help to account for individual differences identified that may have already existed. Due to feasibility, it was decided to only assess the microbiome after the intervention period. However, future, full-scale studies will include a baseline microbiome assessment of each infant’s first stool.
While evidence from this study is limited to infants born via c-section, this population comprises over 20% of births globally, and these infants experience the highest supplementation rates within full-term populations. Studying DHM in this population will provide a large return on investment as it can address multiple aspects of adverse sequalae related to c-section delivery.
Conclusion
This pilot RCT will act as a first step in exploring knowledge gaps related to DHM supplementation compared to formula supplementation in full-term infants born via c-section, and the impact on maternal and child health outcomes, including the developing infant microbiome of these infants. By examining a population with high breastfeeding supplementation rates and subsequent adverse early life exposures to their developing microbiome, the results of this study will allow us to identify casual linkages between breastfeeding and child health outcomes, inform future recommendations for DHM supplementation in full-term infants, and provide initial data to inform a multi-site, longitudinal mixed-methods RCT. This study will also improve knowledge around supplementation for the full-term population, which will help healthcare providers to better educate families around DHM supplementation and advocate for increased DHM accessibility.
Supplemental Material
sj-docx-1-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-docx-1-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-docx-2-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-docx-2-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-3-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-3-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-4-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-4-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-5-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-5-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-6-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-6-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-7-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-7-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-8-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-8-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Supplemental Material
sj-pdf-9-jhl-10.1177_08903344251369442 – Supplemental material for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-9-jhl-10.1177_08903344251369442 for Comparing Impacts of Donor Human Milk to Formula Supplementation on the Gut Microbiome of Full-Term Infants Born Via Cesarean Section: Protocol for a Pilot Randomized Controlled Trial by Meredith (Merilee) Brockway, Maria Khalid, Yasmeen Khalil, Heather C Rusi, Michelle R Asbury, Marie-Claire Arrieta, Elizabeth Keys, Christine Ou and Jannette Festival in Journal of Human Lactation
Footnotes
Acknowledgements
We would like to acknowledge Olabisi Coker and the International Microbiome Centre staff at the University of Calgary for their input and guidance on the microbiome sample and analysis strategy. We would like to also acknowledge Vineetha Warriyar Kodalore Vijayan, biostatistician with the Alberta Children’s Hospital Research Institute for providing guidance on the sample size calculations and analysis of secondary outcomes.
ORCID iDs
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the Canadian Institutes of Health Research (CIHR), the Alberta Registered Nurses Educational Trust and the University of Calgary.
Disclosures and Conflicts of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Meredith Brockway is on the board of directors for the NorthernStar Mothers Milk Bank and on the Scientific Advisory Committee for the Human Milk Banking Association of North America. All other authors declare that they have no competing interests.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental Material
Supplementary Material may be found in the “Supplemental material” tab in the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
